Open Data for Open Questions in Comparative Nutrition
Abstract
1. The Multidimensional Nature of Nutrition
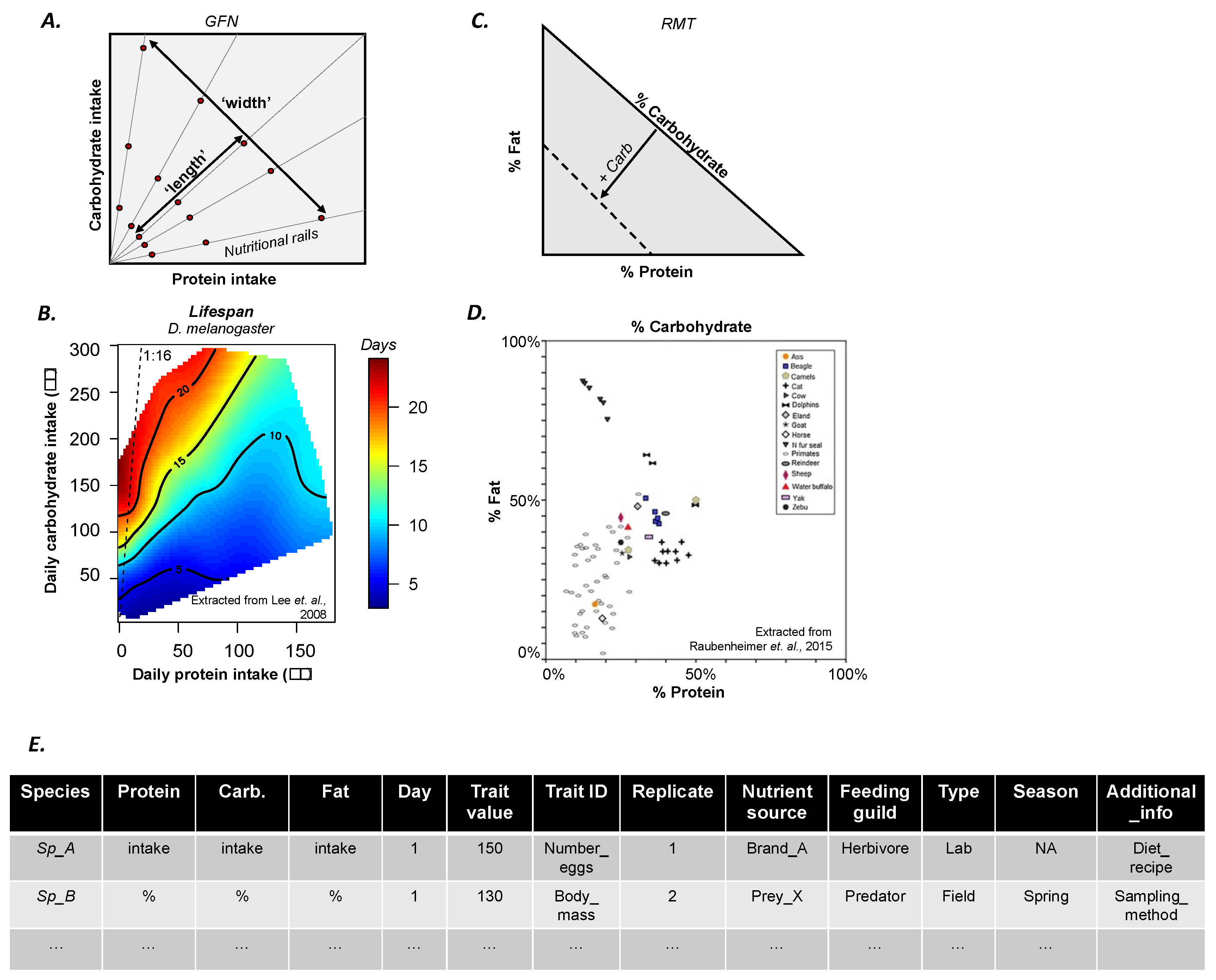
2. The Geometric Framework for Nutrition in a Nutshell
3. Raw Data Availability: Why Does It Matter?
4. More Data for A Bigger Picture
5. How to Share GFN Data?
- Provide raw data used to construct landscapes in a standardized and accessible format (see example in Figure 1E; see template file (.csv) in Appendix A).
- If publishing in an open-access journal, authors are likely (although not always) to be required to deposit the raw data in a public data repository (e.g., Dryad). If publishing in a subscription journal, we encourage authors to make raw data available as Supplementary Material. In both cases, it is important that the data format is also standardised (Figure 1C) and any additional information needed for the understanding of the dataset is also provided (e.g., README file). These steps will benefit the visibility and citations of the original paper.
- Include ‘geometric framework for nutrition’ and ‘performance landscapes’ as keywords in publications to increase exposure on search engines.
- If the study is already published but the data are not available, we encourage authors to deposit the raw data in a public data repository.
6. Towards Quantitative and Comparative Nutrition Research
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Species | Protein | Carb. | Fat | Day | Trait Value | Trait_ID | Replicate | Nutrient Source | Feeding Guild | Type of Experiment | Season | Additional_Info |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sp_A | intake | intake | intake | 1 | 150 | Number_eggs | 1 | Brand_A | Herbivores | Lab | NA | Diet_recipe |
| Sp_B | % | % | % | 1 | 130 | Body_mass | 2 | Prey_X | Predator | Field | Spring | Sampling_method |
References
- Branca, F.; DeMaio, A.; Udomkesmalee, E.; Baker, P.; Aguayo, V.M.; Barquera, S.; Dain, K.; Keir, L.; Lartey, A.; Mugambi, G.; et al. A new nutrition manifesto for a new nutrition reality. Lancet 2019, 395, 8–10. [Google Scholar] [CrossRef]
- Simpson, S.J.; Raubenheimer, D. The Nature of Nutrition: A Unifying Framework from Animal Adaptation to Human Obesity, 1st ed.; Princeton University Press: Princeton, NJ, USA, 2012. [Google Scholar]
- Simpson, S.J.; Raubenheimer, D. A multi-level analysis of feeding behaviour: The geometry of nutritional decisions. Philos. Trans. R. Soc. B Boil. Sci. 1993, 342, 381–402. [Google Scholar]
- Simpson, S.J.; Sibly, R.M.; Lee, K.P.; Behmer, S.; Raubenheimer, D. Optimal foraging when regulating intake of multiple nutrients. Anim. Behav. 2004, 68, 1299–1311. [Google Scholar] [CrossRef]
- Raubenheimer, D.; Simpson, S. Eat Like the Animals: What Nature Teaches Us about the Science of Healthy Eating; Houghton Mifflin: London, UK, 2020. [Google Scholar]
- Raubenheimer, D.; Simpson, S.J. Nutritional Ecology and Human Health. Annu. Rev. Nutr. 2016, 36, 603–626. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.P.; Simpson, S.J.; Clissold, F.J.; Brooks, R.C.; Ballard, J.W.O.; Taylor, P.; Soran, N.; Raubenheimer, D. Lifespan and reproduction in Drosophila: New insights from nutritional geometry. Proc. Natl. Acad. Sci. USA 2008, 105, 2498–2503. [Google Scholar] [CrossRef] [PubMed]
- Mayntz, D.; Behrens, M.M.; Ali, S.S.; Dao, D.N.; Lucero, J.D.; Shekhtman, G.; Quick, K.L.; Dugan, L.L. Nutrient-Specific Foraging in Invertebrate Predators. Science 2005, 307, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Solon-Biet, S.M.; McMahon, A.C.; Ballard, J.W.O.; Ruohonen, K.; Wu, L.E.; Cogger, V.C.; Warren, A.; Huang, X.; Pichaud, N.; Melvin, R.G.; et al. The ratio of macronutrients, not caloric intake, dictates cardiometabolic health, aging, and longevity in ad libitum-fed mice. Cell Metab. 2014, 19, 418–430. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, J.; Senior, A.; Ruiz, K.; Wali, J.A.; Pulpitel, T.; Solon-Biet, S.M.; Cogger, V.C.; Raubenheimer, D.; Le Couteur, D.G.; Simpson, S.J.; et al. Sucrose and starch intake contribute to reduced alveolar bone height in a rodent model of naturally occurring periodontitis. PLoS ONE 2019, 14, e0212796. [Google Scholar] [CrossRef] [PubMed]
- Arganda, S.; Bouchebti, S.; Bazazi, S.; Le Hesran, S.; Puga, C.; Latil, G.; Simpson, S.J.; Dussutour, A. Parsing the life-shortening effects of dietary protein: Effects of individual amino acids. Proc. R. Soc. B Boil. Sci. 2017, 284, 20162052. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, D.; Machovsky-Capuska, G.E.; Chapman, C.A.; Rothman, J.M. Geometry of nutrition in field studies: An illustration using wild primates. Oecologia 2014, 177, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, D. Toward a quantitative nutritional ecology: The right-angled mixture triangle. Ecol. Monogr. 2011, 81, 407–427. [Google Scholar] [CrossRef]
- Nychka, D.; Reinhard, F.; Paige, J.; Sain, S. Fields: Tools for Spatial Data; R Packag. 9.9; University Corporation for Atmospheric Research: Boulder, CO, USA, 2017. [Google Scholar]
- Rosas, A.; Bastir, M. Thin-plate spline analysis of allometry and sexual dimorphism in the human craniofacial complex. Am. J. Phys. Anthropol. Off. Publ. Am. Assoc. Phys. Anthropol. 2002, 117, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Rapkin, J.; Jensen, K.; Archer, C.R.; House, C.M.; Sakaluk, S.K.; Del Castillo, E.; Hunt, J. The Geometry of Nutrient Space-Based Life-History Trade-Offs: Sex-Specific Effects of Macronutrient Intake on the Trade-Off between Encapsulation Ability and Reproductive Effort in Decorated Crickets. Am. Nat. 2018, 191, 452–474. [Google Scholar] [CrossRef]
- Morimoto, J.; Lihoreau, M. Quantifying Nutritional Trade-Offs across Multidimensional Performance Landscapes. Am. Nat. 2019, 193, E168–E181. [Google Scholar] [CrossRef]
- Culina, A.; Baglioni, M.; Crowther, T.W.; Visser, M.E.; Woutersen-Windhouwer, S.; Manghi, P. Navigating the unfolding open data landscape in ecology and evolution. Nat. Ecol. Evol. 2018, 2, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Reichman, O.J.; Jones, M.; Schildhauer, M. Challenges and Opportunities of Open Data in Ecology. Science 2011, 331, 703–705. [Google Scholar] [CrossRef] [PubMed]
- Fanson, B.G.; Weldon, C.W.; Simpson, S.J.; Taylor, P.; Perez-Staples, D. Nutrients, not caloric restriction, extend lifespan in Queensland fruit flies (Bactrocera tryoni). Aging Cell 2009, 8, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Machovsky-Capuska, G.E.; Raubenheimer, D. The Nutritional Ecology of Marine Apex Predators. Annu. Rev. Mar. Sci. 2020, 12, 361–387. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Wang, Z.; Zhang, S.; Wang, B.; Lu, J.; Raubenheimer, D. Living near the limits: Effects of interannual variation in food availability on diet and reproduction in a temperate primate, the Taihangshan macaque (Macaca mulatta tcheliensis). Am. J. Primatol. 2019, 82, e23080. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, D.; Rothman, J.M. Nutritional Ecology of Entomophagy in Humans and Other Primates. Annu. Rev. Entomol. 2013, 58, 141–160. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morimoto, J.; Lihoreau, M. Open Data for Open Questions in Comparative Nutrition. Insects 2020, 11, 236. https://doi.org/10.3390/insects11040236
Morimoto J, Lihoreau M. Open Data for Open Questions in Comparative Nutrition. Insects. 2020; 11(4):236. https://doi.org/10.3390/insects11040236
Chicago/Turabian StyleMorimoto, Juliano, and Mathieu Lihoreau. 2020. "Open Data for Open Questions in Comparative Nutrition" Insects 11, no. 4: 236. https://doi.org/10.3390/insects11040236
APA StyleMorimoto, J., & Lihoreau, M. (2020). Open Data for Open Questions in Comparative Nutrition. Insects, 11(4), 236. https://doi.org/10.3390/insects11040236






