Pollen Protein: Lipid Macronutrient Ratios May Guide Broad Patterns of Bee Species Floral Preferences
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
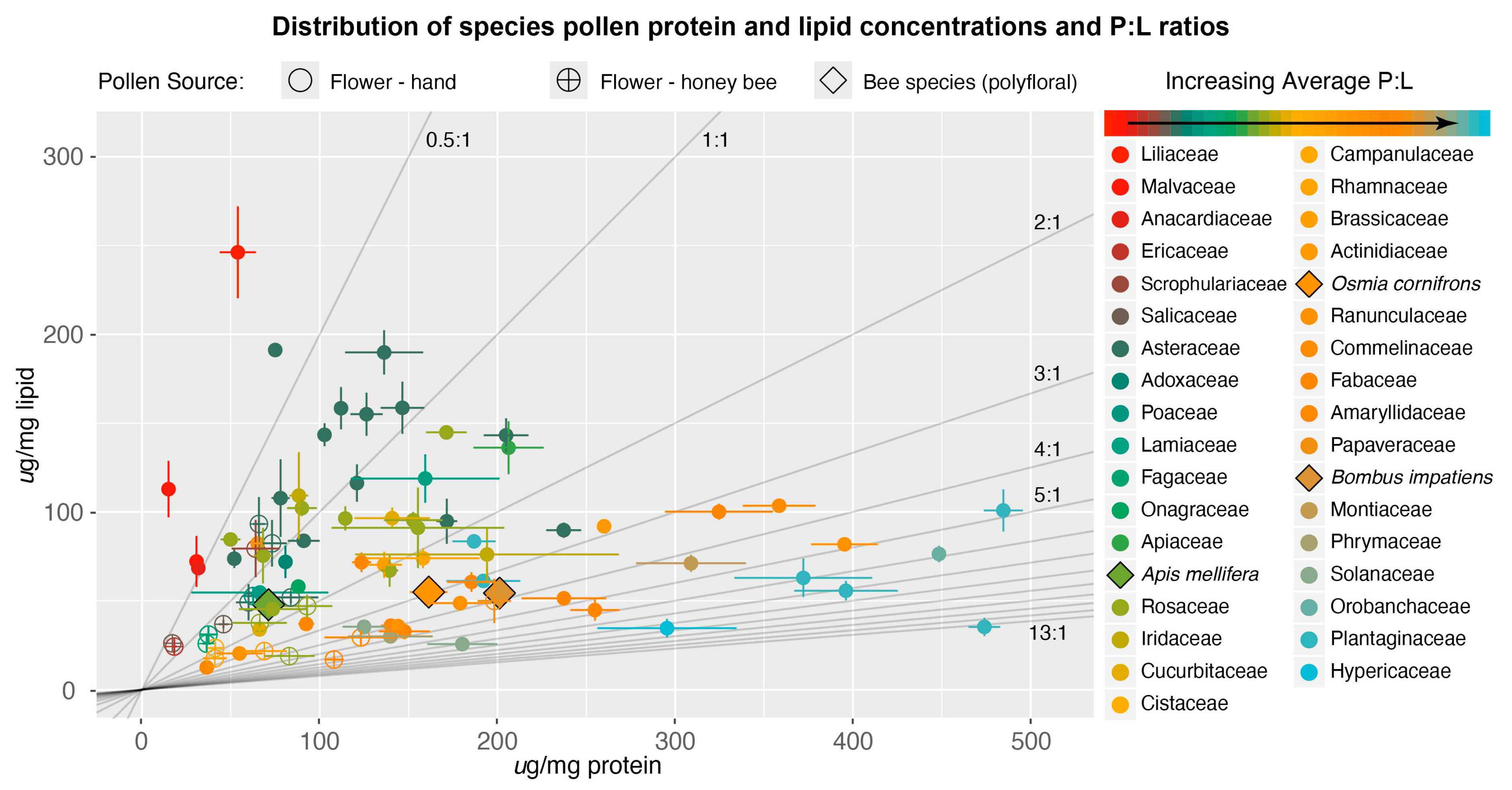
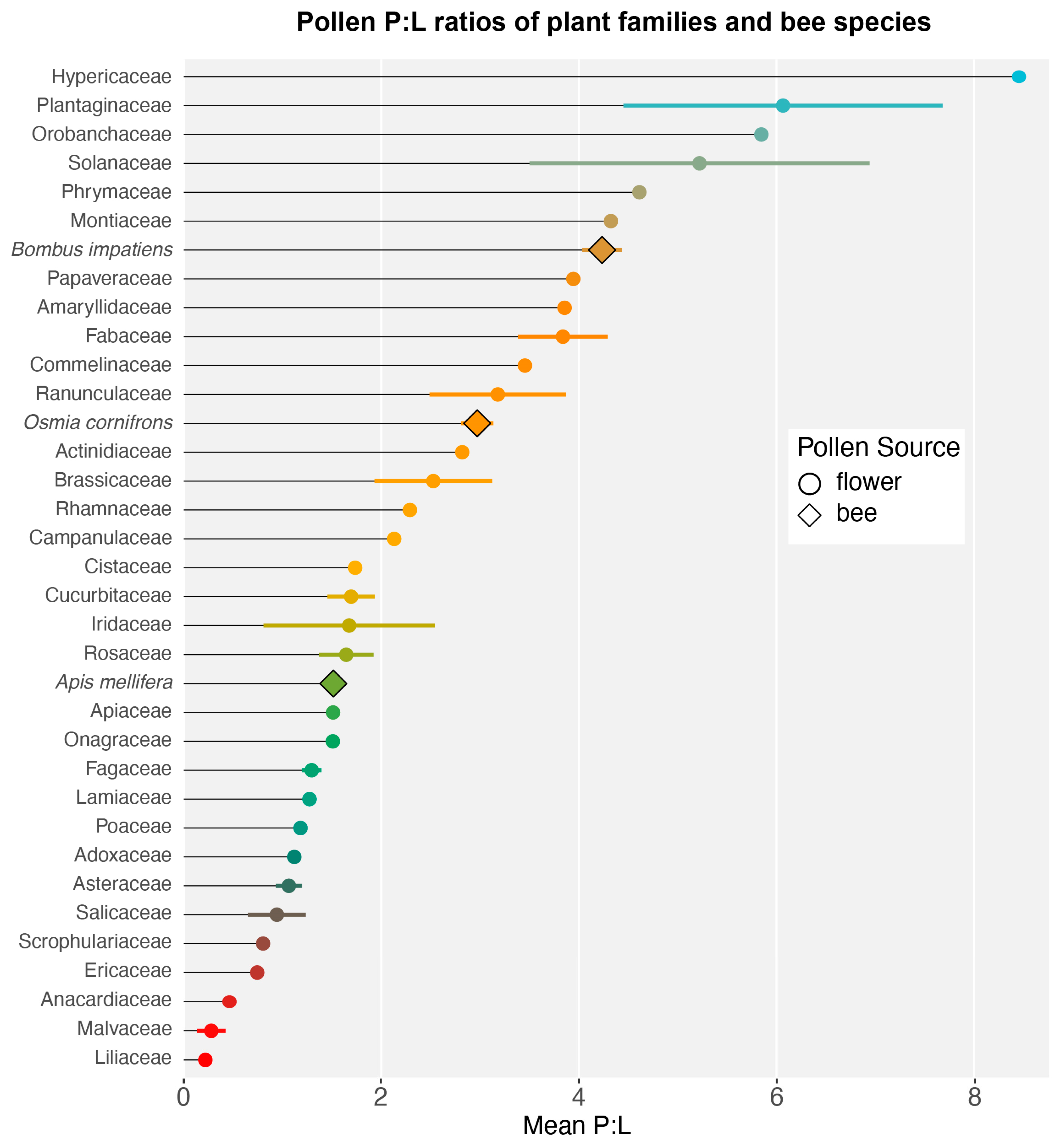
3.1. Nutritional Content of Pollen
3.2. P:L Trends in Bee–Flower Interactions
3.3. Applying Pollen P:L in the Future
3.4. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Biesmeijer, J.C.; Roberts, S.P.M.; Reemer, M.; Ohlemüller, R.; Edwards, M.; Peeters, T.; Schaffers, A.P.; Potts, S.G.; Kleukers, R.; Thomas, C.D.; et al. Parallel declines in pollinators and insect-pollinated plants in Britain and the Netherlands. Science 2006, 313, 351–354. [Google Scholar] [CrossRef]
- Carvell, C.; Roy, D.B.; Smart, S.M.; Pywell, R.F.; Preston, C.D.; Goulson, D. Declines in forage availability for bumblebees at a national scale. Biol. Conserv. 2006, 132, 481–489. [Google Scholar] [CrossRef]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global pollinator declines: Trends, impacts and drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef]
- Ollerton, J.; Erenler, H.; Edwards, M.; Crockett, R. Pollinator declines. Extinctions of aculeate pollinators in Britain and the role of large-scale agricultural changes. Science 2014, 346, 1360–1362. [Google Scholar] [CrossRef]
- Goulson, D.; Nicholls, E.; Botías, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef]
- Behmer, S.T. Insect herbivore nutrient regulation. Annu. Rev. Entomol. 2009, 54, 165–187. [Google Scholar] [CrossRef]
- Simpson, S.J.; Raubenheimer, D. The Nature of Nutrition: A Unifying Framework from Animal Adaptation to Human Obesity; Princeton University Press: Princeton, NJ, USA, 2012. [Google Scholar]
- Vaudo, A.D.; Stabler, D.; Patch, H.M.; Tooker, J.F.; Grozinger, C.M.; Wright, G.A. Bumble bees regulate their intake of essential protein and lipid pollen macronutrients. J. Exp. Biol. 2016, 219, 3962–3970. [Google Scholar] [CrossRef]
- Vaudo, A.D.; Farrell, L.M.; Patch, H.M.; Grozinger, C.M.; Tooker, J.F. Consistent pollen nutritional intake drives bumble bee (Bombus impatiens) colony growth and reproduction across different habitats. Ecol. Evol. 2018, 8, 5765–5776. [Google Scholar] [CrossRef]
- Kraus, S.; Gómez-Moracho, T.; Pasquaretta, C.; Latil, G.; Dussutour, A.; Lihoreau, M. Bumblebees adjust protein and lipid collection rules to the presence of brood. Curr. Zool. 2019, 65, 437–446. [Google Scholar] [CrossRef]
- Nicolson, S.W. Bee food: The chemistry and nutritional value of nectar, pollen and mixtures of the two. Afr. Zool. 2011, 46, 197–204. [Google Scholar] [CrossRef]
- Nicolson, S.W.; Nepi, M.; Pacini, E.; Nicolson, S.W.; Nepi, M.; Pacini, E. (Eds.) Nectaries and Nectar; Springer Science & Business Media: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Vaudo, A.D.; Tooker, J.F.; Grozinger, C.M.; Patch, H.M. Bee nutrition and floral resource restoration. Curr. Opin. Insect Sci. 2015, 10, 133–141. [Google Scholar] [CrossRef]
- Cane, J.H. Adult pollen diet essential for egg maturation by a solitary Osmia bee. J. Insect Physiol. 2016, 95, 105–109. [Google Scholar] [CrossRef]
- Cane, J.H.; Dobson, H.E.M.; Boyer, B. Timing and size of daily pollen meals eaten by adult females of a solitary bee (Nomia melanderi) (Apiformes: Halictidae). Apidologie 2016, 48, 17–30. [Google Scholar] [CrossRef]
- Di Pasquale, G.; Salignon, M.; Le Conte, Y.; Belzunces, L.P.; Decourtye, A.; Kretzschmar, A.; Suchail, S.; Brunet, J.-L.; Alaux, C. Influence of pollen nutrition on honey bee health: Do pollen quality and diversity matter? PLoS ONE 2013, 8, e72016. [Google Scholar] [CrossRef]
- Roulston, T.H.; Cane, J.H.; Buchmann, S.L. What governs protein content of pollen: Pollinator preferences, pollen-pistil interactions, or phylogeny? Ecol. Monogr. 2000, 70, 617–643. [Google Scholar]
- Vaudo, A.D.; Patch, H.M.; Mortensen, D.A.; Tooker, J.F.; Grozinger, C.M. Macronutrient ratios in pollen shape bumble bee (Bombus impatiens) foraging strategies and floral preferences. Proc. Natl. Acad. Sci. USA 2016, 113, E4035–E4042. [Google Scholar] [CrossRef]
- Russo, L.; Vaudo, A.D.; Fisher, C.J.; Grozinger, C.M.; Shea, K. Bee community preference for an invasive thistle associated with higher pollen protein content. Oecologia 2019, 190, 901–912. [Google Scholar] [CrossRef]
- Treanore, E.D.; Vaudo, A.D.; Grozinger, C.M.; Fleischer, S.J. Examining the nutritional value and effects of different floral resources in pumpkin agroecosystems on Bombus impatiens worker physiology. Apidologie 2019, 50, 542–552. [Google Scholar] [CrossRef]
- Weiner, C.N.; Hilpert, A.; Werner, M.; Linsenmair, K.E.; Blüthgen, N. Pollen amino acids and flower specialisation in solitary bees. Apidologie 2010, 41, 476–487. [Google Scholar] [CrossRef]
- Roulston, T.H.; Cane, J.H. Pollen nutritional content and digestibility for animals. Plant Syst. Evol. 2000, 222, 187–209. [Google Scholar] [CrossRef]
- Arien, Y.; Dag, A.; Zarchin, S.; Masci, T.; Shafir, S. Omega-3 deficiency impairs honey bee learning. Proc. Natl. Acad. Sci. USA 2015, 112, 15761–15766. [Google Scholar] [CrossRef]
- Vanderplanck, M.; Moerman, R.; Rasmont, P.; Lognay, G.; Wathelet, B.; Wattiez, R.; Michez, D. How does pollen chemistry impact development and feeding behaviour of polylectic bees? PLoS ONE 2014, 9, e86209. [Google Scholar] [CrossRef]
- Moerman, R.; Vanderplanck, M.; Fournier, D.; Jacquemart, A.-L.; Michez, D. Pollen nutrients better explain bumblebee colony development than pollen diversity. Insect Conserv. Diver. 2017, 10, 171–179. [Google Scholar] [CrossRef]
- Stevenson, P.C. For antagonists and mutualists: The paradox of insect toxic secondary metabolites in nectar and pollen. Phytochem. Rev. 2019. [Google Scholar] [CrossRef]
- Roulston, T.H.; Cane, J.H. The effect of pollen protein concentration on body size in the sweat bee Lasioglossum zephyrum (Hymenoptera: Apiformes). Evol. Ecol. 2002, 16, 49–65. [Google Scholar] [CrossRef]
- Human, H.; Nicolson, S.W.; Strauss, K.; Pirk, C.W.W.; Dietemann, V. Influence of pollen quality on ovarian development in honeybee workers (Apis mellifera scutellata). J. Insect Physiol. 2007, 53, 649–655. [Google Scholar] [CrossRef]
- Tasei, J.-N.; Aupinel, P. Nutritive value of 15 single pollens and pollen mixes tested on larvae produced by bumblebee workers (Bombus terrestris, Hymenoptera: Apidae). Apidologie 2008, 39, 397–409. [Google Scholar] [CrossRef]
- Li, C.; Xu, B.; Wang, Y.; Feng, Q.; Yang, W. Effects of dietary crude protein levels on development, antioxidant status, and total midgut protease activity of honey bee (Apis mellifera ligustica). Apidologie 2012, 43, 576–586. [Google Scholar] [CrossRef]
- van der Moezel, P.G.; Delfs, J.C.; Pate, J.S.; Loneragan, W.A.; Bell, D.T. Pollen selection by honeybees in shrublands of the Northern Sand plains of Western Australia. J. Apicult. Res. 1987, 26, 224–232. [Google Scholar] [CrossRef]
- Pernal, S.; Currie, R. Pollen quality of fresh and 1-year-old single pollen diets for worker honey bees (Apis mellifera L.). Apidologie 2000, 31, 387–409. [Google Scholar] [CrossRef]
- Hanley, M.E.; Franco, M.; Pichon, S.; Darvill, B.; Goulson, D. Breeding system, pollinator choice and variation in pollen quality in British herbaceous plants. Funct. Ecol. 2008, 22, 592–598. [Google Scholar] [CrossRef]
- Cardoza, Y.J.; Harris, G.K.; Grozinger, C.M. Effects of soil quality enhancement on pollinator-plant interactions. Psyche J. Entomol. 2012, 2012, 581458. [Google Scholar] [CrossRef]
- Konzmann, S.; Lunau, K. Divergent rules for pollen and nectar foraging bumblebees – A laboratory study with artificial flowers offering diluted nectar substitute and pollen surrogate. PLoS ONE 2014, 9, e91900. [Google Scholar] [CrossRef] [PubMed]
- Kitaoka, T.K.; Nieh, J.C. Bumble bee pollen foraging regulation: Role of pollen quality, storage levels, and odor. Behav. Ecol. Siciobiol. 2008, 63, 501–510. [Google Scholar] [CrossRef]
- Leonhardt, S.D.; Blüthgen, N. The same, but different: Pollen foraging in honeybee and bumblebee colonies. Apidologie 2011, 43, 449–464. [Google Scholar] [CrossRef]
- Génissel, A.; Aupinel, P.; Bressac, C.; Tasei, J.N.; Chevrier, C. Influence of pollen origin on performance of Bombus terrestris micro-colonies. Entomol. Exp. Appl. 2002, 104, 329–336. [Google Scholar] [CrossRef]
- Moerman, R.; Vanderplanck, M.; Roger, N.; Declèves, S.; Wathelet, B.; Rasmont, P.; Fournier, D.; Michez, D. Growth rate of bumblebee larvae is related to pollen amino acids. J. Econ. Entomol. 2016, 109, 25–30. [Google Scholar] [CrossRef]
- Alaux, C.; Ducloz, F.; Crauser, D.; Le Conte, Y. Diet effects on honeybee immunocompetence. Biol. Lett. 2010, 6, 562–565. [Google Scholar] [CrossRef]
- Ruedenauer, F.A.; Leonhardt, S.D.; Lunau, K.; Spaethe, J. Bumblebees are able to perceive amino acids via chemotactile antennal stimulation. J. Comp. Physiol. A 2019, 205, 321–331. [Google Scholar] [CrossRef]
- Dobson, H. Survey of pollen and pollen kitt lipids – Chemical cues to flower visitors? Am. J. Bot. 1988, 75, 170–182. [Google Scholar] [CrossRef]
- Ruedenauer, F.A.; Raubenheimer, D.; Kessner Beierlein, D.; Grund Mueller, N.; Noack, L.; Spaethe, J.; Leonhardt, S.D. Best be(e) on low fat: Linking nutrient perception, regulation and fitness. Ecol. Lett. 2020, 23, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Arien, Y.; Dag, A.; Shafir, S. Omega-6:3 ratio more than absolute lipid level in diet affects associative learning in honey bees. Front. Psychol. 2018, 9, 1001. [Google Scholar] [CrossRef] [PubMed]
- Muth, F.; Breslow, P.R.; Masek, P.; Leonard, A.S. A pollen fatty acid enhances learning and survival in bumblebees. Behav. Ecol. 2018, 29, 1371–1379. [Google Scholar] [CrossRef]
- Giri, S.; Rule, D.C.; Dillon, M.E. Fatty acid composition in native bees – Associations with thermal and feeding ecology. Comp. Biochem. Phys. 2018, 218, 70–79. [Google Scholar] [CrossRef]
- Vanderplanck, M.; Zerck, P.L.; Lognay, G.; Michez, D. Generalized host-plant feeding can hide sterol-specialized foraging behaviors in bee–plant interactions. Ecol. Evol. 2019, 42, 91–113. [Google Scholar] [CrossRef]
- Raubenheimer, D.; Simpson, S.J. Integrating nutrition: A geometrical approach. Entomol. Exp. Appl. 1999, 91, 67–82. [Google Scholar] [CrossRef]
- Vaudo, A.D.; Patch, H.M.; Mortensen, D.A.; Grozinger, C.M.; Tooker, J.F. Bumble bees exhibit daily behavioral patterns in pollen foraging. Arthropod-Plant Inte. 2014, 8, 273–283. [Google Scholar] [CrossRef]
- Russo, L.; Keller, J.; Vaudo, A.D.; Grozinger, C.M.; Shea, K. Warming increases pollen lipid concentration in an invasive thistle, with minor effects on the associated floral-visitor community. Insects 2020, 11, 20. [Google Scholar] [CrossRef]
- Vanderplanck, M.; Leroy, B.; Wathelet, B.; Wattiez, R.; Michez, D. Standardized protocol to evaluate pollen polypeptides as bee food source. Apidologie 2013, 45, 192–204. [Google Scholar] [CrossRef]
- Vanderplanck, M.; Michez, D.; Vancraenenbroeck, S.; Lognay, G. Micro-quantitative method for analysis of sterol levels in honeybees and their pollen loads. Anal. Lett. 2011, 44, 1807–1820. [Google Scholar] [CrossRef]
- Stabler, D.; Power, E.F.; Borland, A.M.; Barnes, J.D.; Wright, G.A. A method for analysing small samples of floral pollen for free and protein-bound amino acids. Methods Ecol. Evol. 2018, 9, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Ruedenauer, F.A.; Spaethe, J.; van der Kooi, C.J.; Leonhardt, S.D. Pollinator or pedigree: Which factors determine the evolution of pollen nutrients? Oecologia 2019, 191, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Human, H.; Nicolson, S.W. Nutritional content of fresh, bee-collected and stored pollen of Aloe greatheadii var. davyana (Asphodelaceae). Phytochemistry 2006, 67, 1486–1492. [Google Scholar] [CrossRef] [PubMed]
- Behmer, S.T.; Joern, A. Coexisting generalist herbivores occupy unique nutritional feeding niches. Proc. Natl. Acad. Sci. USA 2008, 105, 1977–1982. [Google Scholar] [CrossRef] [PubMed]
- Kämper, W.; Werner, P.K.; Hilpert, A.; Westphal, C.; Blüthgen, N.; Eltz, T.; Leonhardt, S.D. How landscape, pollen intake and pollen quality affect colony growth in Bombus terrestris. Landsc. Ecol. 2016, 31, 2245–2258. [Google Scholar]
- Rasmont, P.; Mersch, P. Première estimation de la dérive faunique chez les bourdons de la Belgique (Hymenoptera: Apidae). Ann. Soc. R. Zool. Bel. 1988, 118, 141–147. [Google Scholar]
- Nicolson, S.W.; Human, H. Chemical composition of the “low quality” pollen of sunflower (Helianthus annuus, Asteraceae). Apidologie 2013, 44, 144–152. [Google Scholar] [CrossRef]
- Richardson, R.T.; Lin, C.-H.; Sponsler, D.B.; Quijia, J.O.; Goodell, K.; Johnson, R.M. Application of ITS2 metabarcoding to determine the provenance of pollen collected by honey bees in an agroecosystem. Appl. Plant Sci. 2015, 3, 1400066. [Google Scholar] [CrossRef]
- Pernal, S.; Currie, R. The influence of pollen quality on foraging behavior in honeybees (Apis mellifera L.). Behav. Ecol. Sociobiol. 2001, 51, 53–68. [Google Scholar] [CrossRef]
- Degrandi-Hoffman, G.; Gage, S.L.; Corby-Harris, V.; Carroll, M.; Chambers, M.; Graham, H.; Watkins Dejong, E.; Hidalgo, G.; Calle, S.; Azzouz-Olden, F.; et al. Connecting the nutrient composition of seasonal pollens with changing nutritional needs of honey bee (Apis mellifera L.) colonies. J. Insect Physiol. 2018, 109, 114–124. [Google Scholar] [CrossRef]
- Hendriksma, H.P.; Shafir, S. Honey bee foragers balance colony nutritional deficiencies. Behav. Ecol. Sociobiol. 2016, 70, 509–517. [Google Scholar] [CrossRef]
- Zarchin, S.; Dag, A.; Salomon, M.; Hendriksma, H.P.; Shafir, S. Honey bees dance faster for pollen that complements colony essential fatty acid deficiency. Behav. Ecol. Sociobiol. 2017, 71, 1–11. [Google Scholar] [CrossRef]
- McKinney, M.I.; Park, Y.-L. Nesting activity and behavior of Osmia cornifrons (Hymenoptera: Megachilidae) elucidated using videography. Psyche J. Entomol. 2012, 2012, 814097. [Google Scholar]
- Haider, M.; Dorn, S.; Sedivy, C.; Müller, A. Phylogeny and floral hosts of a predominantly pollen generalist group of mason bees (Megachilidae: Osmiini). Biol. J. Linn. Soc. 2014, 111, 78–91. [Google Scholar] [CrossRef]
- Nagamitsu, T.; Suzuki, M.F.; Mine, S.; Taki, H.; Shuri, K.; Kikuchi, S.; Masaki, T. Effects of forest loss and fragmentation on pollen diets and provision mass of the mason bee, Osmia cornifrons, in central Japan. Ecol. Entomol. 2017, 43, 245–254. [Google Scholar] [CrossRef]
- Cane, J.H.; Sipes, S.; Waser, N.M.J. (Eds.) Characterizing floral specialization by bees: Analytical methods and a revised lexicon for oligolecty. In Plant–Pollinator Interactions: From Specialization to Generalization; University of Chicago Press: Chicago, IL, USA, 2006. [Google Scholar]
- Crumière, A.J.J.; Stephenson, C.J.; Nagel, M.; Shik, J.Z. Using nutritional geometry to explore how social insects navigate nutritional landscapes. Insects 2020, 11, 53. [Google Scholar] [CrossRef]
- Omar, E.; Abd-Ella, A.A.; Khodairy, M.M.; Moosbeckhofer, R.; Crailsheim, K.; Brodschneider, R. Influence of different pollen diets on the development of hypopharyngeal glands and size of acid gland sacs in caged honey bees (Apis mellifera). Apidologie 2016, 48, 425–436. [Google Scholar] [CrossRef]
- Renzi, M.T.; Rodríguez-Gasol, N.; Medrzycki, P.; Porrini, C.; Martini, A.; Burgio, G.; Maini, S.; Sgolastra, F. Combined effect of pollen quality and thiamethoxam on hypopharyngeal gland development and protein content in Apis mellifera. Apidologie 2016, 47, 779–788. [Google Scholar] [CrossRef]
- Pattamayutanon, P.; Peng, C.-C.; Sinpoo, C.; Chantawannakul, P. Effects of pollen feeding on quality of royal jelly. J. Econ. Entomol. 2018, 111, 2974–2978. [Google Scholar] [CrossRef]
- Pereboom, J.J.M. The composition of larval food and the significance of exocrine secretions in the bumblebee Bombus terrestris. Insect Soc. 2000, 47, 11–20. [Google Scholar] [CrossRef]
- Jandt, J.M.; Huang, E.; Dornhaus, A. Weak specialization of workers inside a bumble bee (Bombus impatiens) nest. Behav. Ecol. Sociobiol. 2009, 63, 1829–1836. [Google Scholar] [CrossRef]
- Praz, C.J.; Müller, A.; Dorn, S. Specialized bees fail to develop on non-host pollen: Do plants chemically protect their pollen? Ecology 2008, 89, 795–804. [Google Scholar] [CrossRef]
- Lawson, S.P.; Ciaccio, K.N.; Rehan, S.M. Maternal manipulation of pollen provisions affects worker production in a small carpenter bee. Behav. Ecol. Sociobiol. 2016, 70, 1891–1900. [Google Scholar] [CrossRef]
- Sipes, S.D.; Tepedino, V.J. Pollen-host specificity and evolutionary patterns of host switching in a clade of specialist bees (Apoidea: Diadasia). Biol. J. Linn. Soc. 2005, 86, 487–505. [Google Scholar] [CrossRef]
- Eardley, C.D.; Kuhlmann, M.; Pauly, A. The Bee Genera and Subgenera of sub-Saharan Africa. Abc Taxa 2010, 7, 1–138. [Google Scholar]
- Müller, A.; Kuhlmann, M. Pollen hosts of western palaearctic bees of the genus Colletes (Hymenoptera: Colletidae): The Asteraceae paradox. Biol. J. Linn. Soc. 2008, 95, 719–733. [Google Scholar] [CrossRef]
- Cane, J.H. Specialist bees collect Asteraceae pollen by distinctive abdominal drumming (Osmia) or tapping (Melissodes, Svastra). Arthropod-Plant Inte. 2016, 11, 257–261. [Google Scholar] [CrossRef]
- Fowler, R.E. Specialist bees of the Mid-Atlantic: Host plants and habitat conservation. Md. Entomol. 2016, 6, 2–40. [Google Scholar]
- Egan, P.A.; Adler, L.S.; Irwin, R.E.; Farrell, I.W.; Palmer-Young, E.C.; Stevenson, P.C. Crop domestication alters floral reward chemistry with potential consequences for pollinator health. Front. Plant Sci. 2018, 9, 409–414. [Google Scholar] [CrossRef]
- Toshack, M.; Elle, E. Wild bumble bee foraging preferences and fat content in highbush blueberry agro-ecosystems. Apidologie 2019, 50, 425–435. [Google Scholar] [CrossRef]
- Eckhardt, M.; Haider, M.; Dorn, S.; Müller, A. Pollen mixing in pollen generalist solitary bees: A possible strategy to complement or mitigate unfavourable pollen properties? J. Anim. Ecol. 2014, 83, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Vanderplanck, M.; Vereecken, N.J.; Grumiau, L.; Esposito, F.; Lognay, G.; Wattiez, R.; Michez, D. The importance of pollen chemistry in evolutionary host shifts of bees. Sci. Rep. 2017, 7, 43058. [Google Scholar] [CrossRef] [PubMed]
- Drossart, M.; Michez, D.; Vanderplanck, M. Invasive plants as potential food resource for native pollinators: A case study with two invasive species and a generalist bumble bee. Sci. Rep. 2017, 7, 16242. [Google Scholar] [CrossRef] [PubMed]
- Ziska, L.H.; Pettis, J.S.; Edwards, J.; Hancock, J.E.; Tomecek, M.B.; Clark, A.; Dukes, J.S.; Loladze, I.; Polley, H.W. Rising atmospheric CO2 is reducing the protein concentration of a floral pollen source essential for North American bees. P. R. Soc. B. 2016, 283, 1–7. [Google Scholar] [CrossRef]
- Roger, N.; Moerman, R.; Carvalheiro, L.G.; Aguirre-Guitiérrez, J.; Jacquemart, A.-L.; Kleijn, D.; Lognay, G.; Moquet, L.; Quinet, M.; Rasmont, P.; et al. Impact of pollen resources drift on common bumblebees in NW Europe. Glob. Chang. Biol. 2016, 23, 68–76. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vaudo, A.D.; Tooker, J.F.; Patch, H.M.; Biddinger, D.J.; Coccia, M.; Crone, M.K.; Fiely, M.; Francis, J.S.; Hines, H.M.; Hodges, M.; et al. Pollen Protein: Lipid Macronutrient Ratios May Guide Broad Patterns of Bee Species Floral Preferences. Insects 2020, 11, 132. https://doi.org/10.3390/insects11020132
Vaudo AD, Tooker JF, Patch HM, Biddinger DJ, Coccia M, Crone MK, Fiely M, Francis JS, Hines HM, Hodges M, et al. Pollen Protein: Lipid Macronutrient Ratios May Guide Broad Patterns of Bee Species Floral Preferences. Insects. 2020; 11(2):132. https://doi.org/10.3390/insects11020132
Chicago/Turabian StyleVaudo, Anthony D., John F. Tooker, Harland M. Patch, David J. Biddinger, Michael Coccia, Makaylee K. Crone, Mark Fiely, Jacob S. Francis, Heather M. Hines, Mackenzie Hodges, and et al. 2020. "Pollen Protein: Lipid Macronutrient Ratios May Guide Broad Patterns of Bee Species Floral Preferences" Insects 11, no. 2: 132. https://doi.org/10.3390/insects11020132
APA StyleVaudo, A. D., Tooker, J. F., Patch, H. M., Biddinger, D. J., Coccia, M., Crone, M. K., Fiely, M., Francis, J. S., Hines, H. M., Hodges, M., Jackson, S. W., Michez, D., Mu, J., Russo, L., Safari, M., Treanore, E. D., Vanderplanck, M., Yip, E., Leonard, A. S., & Grozinger, C. M. (2020). Pollen Protein: Lipid Macronutrient Ratios May Guide Broad Patterns of Bee Species Floral Preferences. Insects, 11(2), 132. https://doi.org/10.3390/insects11020132






