Changing Microarthropod Communities in Front of a Receding Glacier in the High Arctic
Abstract
1. Introduction
2. Materials and Methods
2.1. Chronosequence
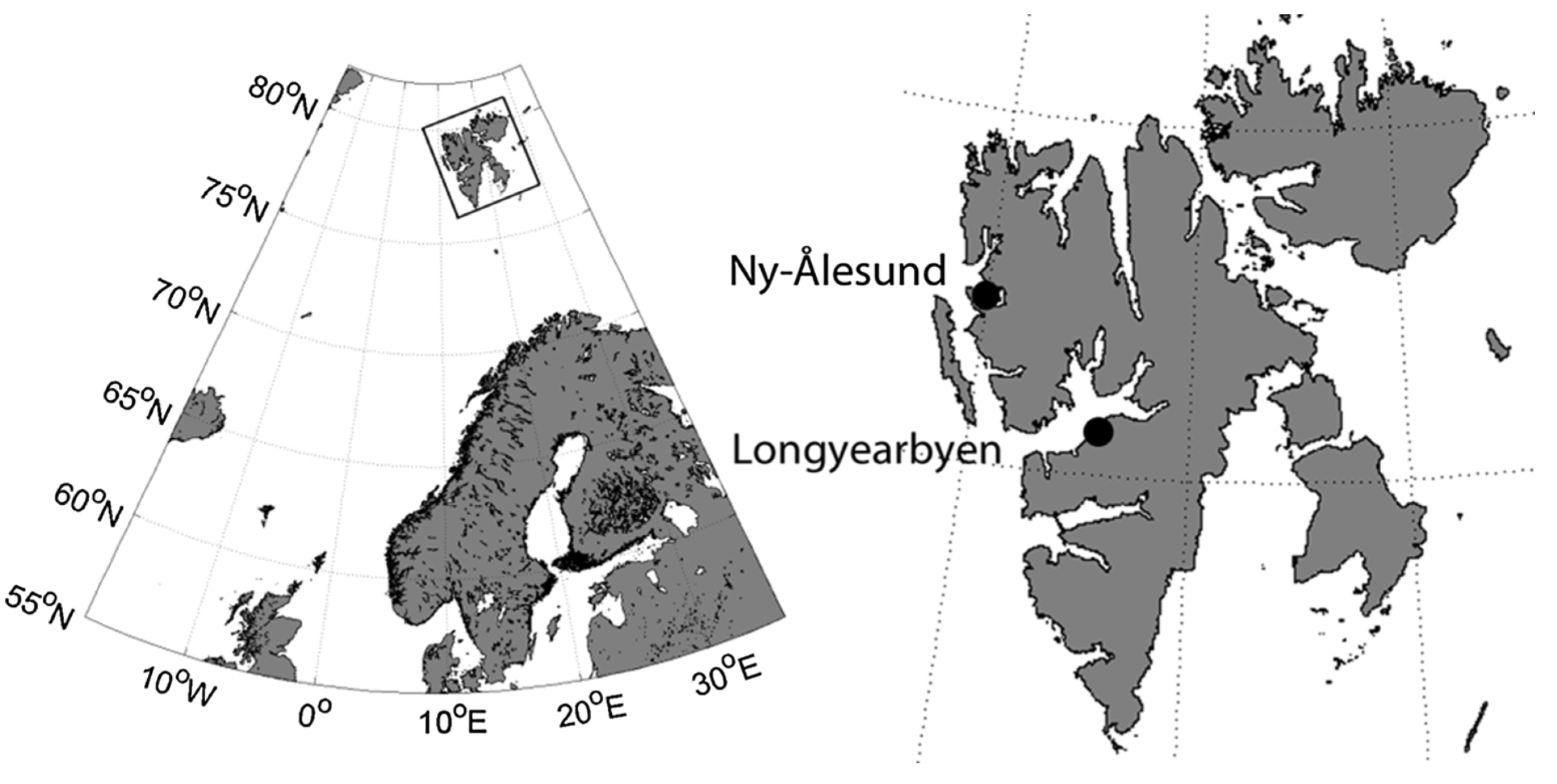
2.2. Study Area
2.3. Laboratory Procedures and Statistics
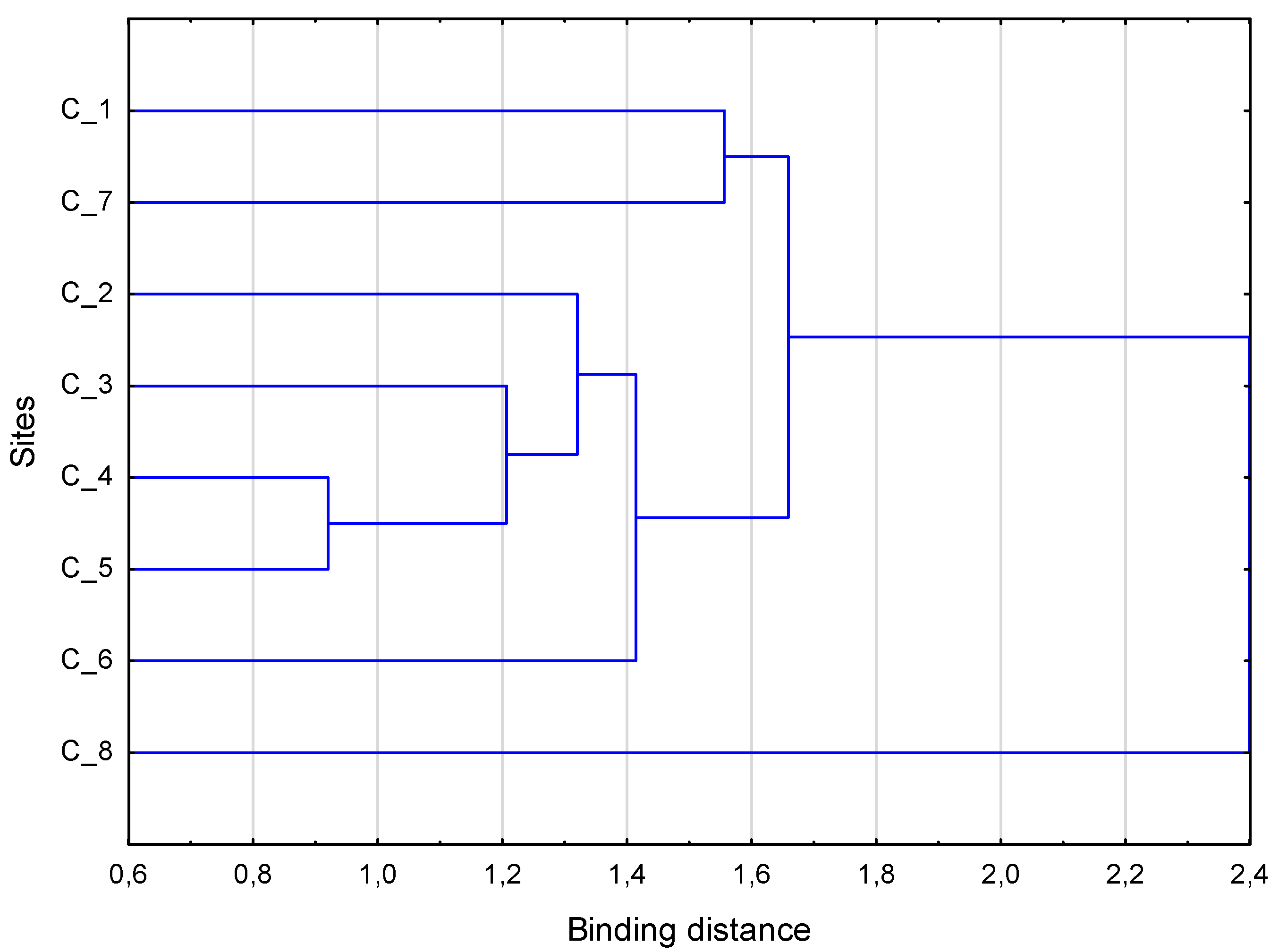
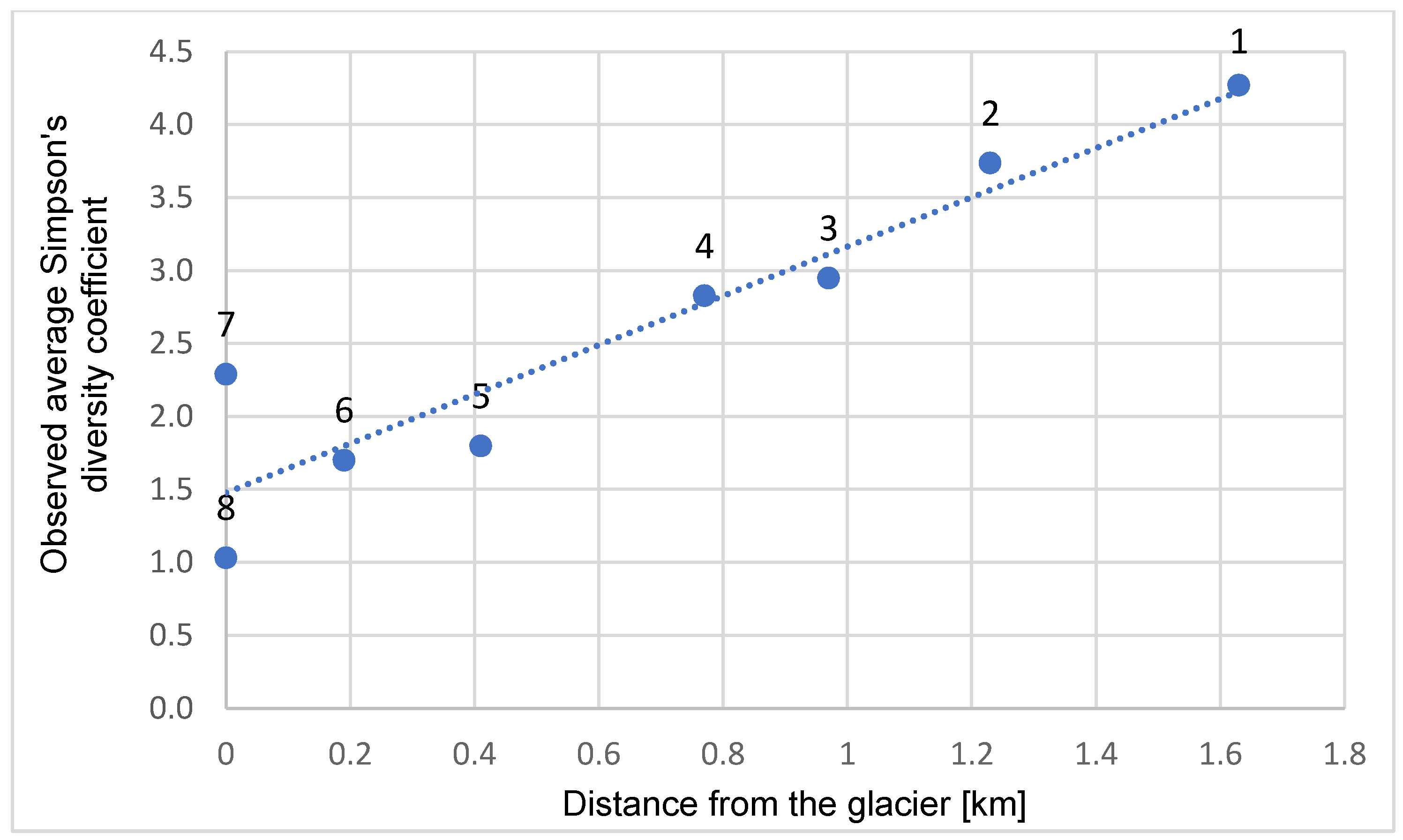
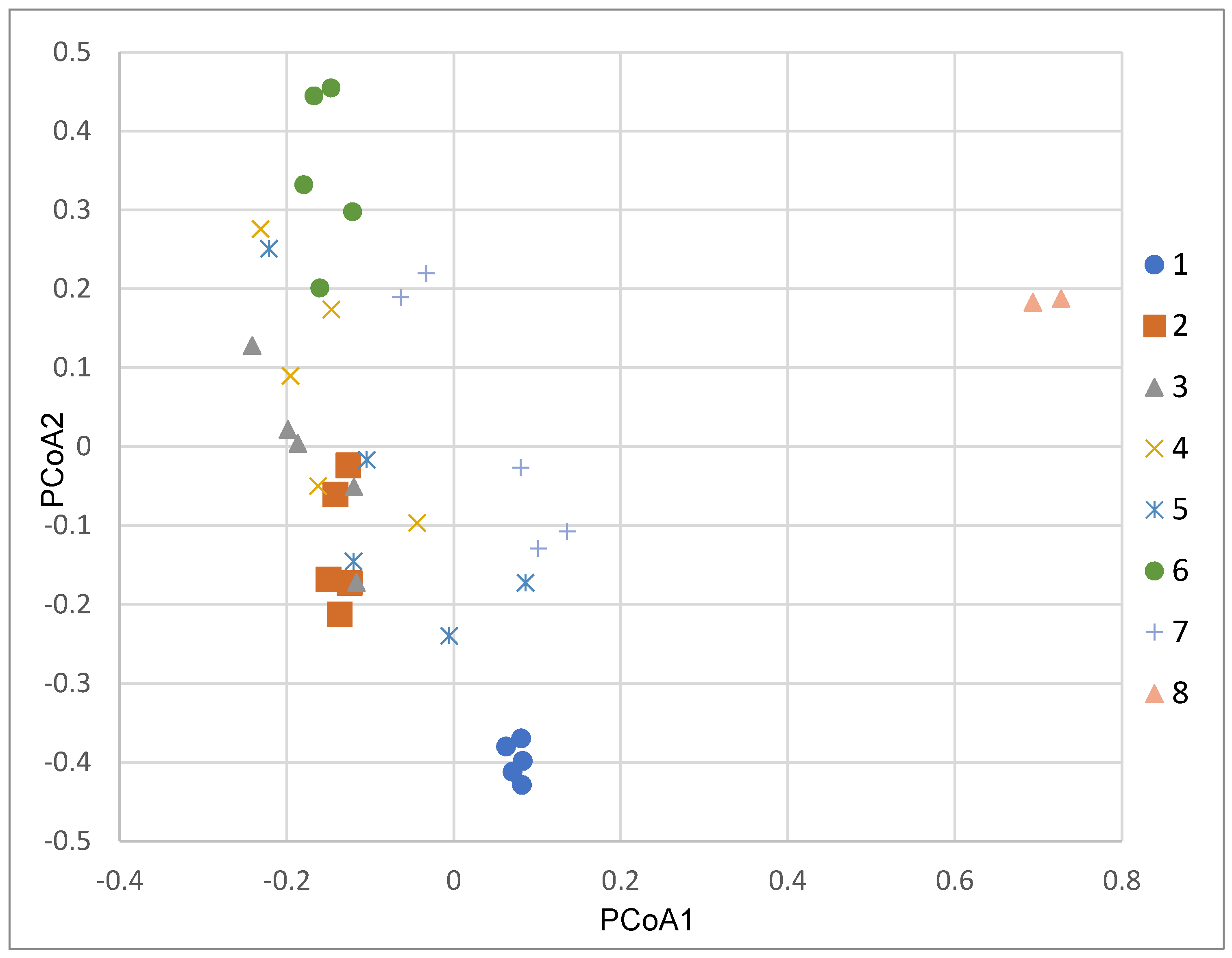
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Species | F (%) | A (ind.) | D (%) | F (%) | A (ind.) | D (%) | F (%) | A (ind.) | D (%) | F (%) | A (ind.) | D (%) | F (%) | A (ind.) | D (%) | F (%) | A (ind.) | D (%) | F (%) | A (ind.) | D (%) | F (%) | A (ind.) | D (%) |
| Oribatida | ||||||||||||||||||||||||
| Camisia dictyna Colloff 1993 | 80,00 | 70 | 4,60 | 100,0 | 189 | 2,54 | 100,0 | 1813 | 35,90 | 40,00 | 7 | 0,66 | - | - | - | - | - | - | - | - | - | - | - | - |
| Camisia foveolata Hammer 1955 | 20,00 | 1 | 0,07 | 60,00 | 13 | 0,17 | 100,0 | 73 | 1,45 | 60,00 | 9 | 0,84 | 20,00 | 3 | 0,33 | 80,00 | 138 | 11,23 | 40,00 | 6 | 5,94 | - | - | - |
| Conchogneta dalecarlica (Forsslund 1947) | - | - | - | - | - | - | 20,00 | 1 | 0,02 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Diapterobates notatus (Thorell 1871) | 100,0 | 91 | 5,98 | 100,0 | 79 | 1,06 | 100,0 | 203 | 4,02 | 100,0 | 133 | 12,46 | 100,0 | 563 | 62,21 | - | - | - | - | - | - | - | - | - |
| Hermannia reticulata Thorell 1871 | 100,0 | 174 | 11,44 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Liochthonius lapponicus (Trägårdh 1910) | 80,00 | 9 | 0,59 | 100,0 | 40 | 0,54 | 100,0 | 286 | 5,66 | 60,00 | 164 | 15,37 | 40,00 | 15 | 1,66 | 80,00 | 12 | 0,98 | - | - | - | - | - | - |
| Liochthonius tuxeni (Forsslund 1957) | 20,00 | 1 | 0,07 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Mycobates bicornis (Strenzke 1954) | - | - | - | 20,00 | 1 | 0,01 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Mycobates parmeliae (Michael 1884) | 60,00 | 7 | 0,46 | 20,00 | 4 | 0,05 | - | 1 | 0,02 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Oppiella (Moritzoppia) neerlandica (Oudemans 1900) | 100,0 | 134 | 8,81 | - | - | - | 20,00 | 1 | 0,02 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Steganacarus (Atropacarus) striculus (Koch 1835) | - | - | - | - | - | - | 40,00 | 2 | 0,04 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Tectocepheus velatus (Michael 1880) | - | - | - | 100,0 | 485 | 6,53 | 100,0 | 75 | 1,49 | 80,00 | 52 | 4,87 | 60,00 | 79 | 8,73 | 100,0 | 273 | 22,21 | 20,00 | 4 | 3,96 | - | - | - |
| Mesostigmata | ||||||||||||||||||||||||
| Arctoseius haarlovi Lindquist 1963 | 100,0 | 29 | 1,91 | 40,00 | 5 | 0,07 | - | - | - | 80,00 | 32 | 3,00 | 60,00 | 7 | 0,77 | - | - | - | - | - | - | - | - | - |
| Arctoseius multidentatus Evans 1955 | 80,00 | 8 | 0,53 | 100,0 | 19 | 0,26 | 100,0 | 27 | 0,53 | 20,00 | 2 | 0,19 | 20,00 | 2 | 0,22 | - | - | - | - | - | - | - | - | - |
| Neoseiulus magnanalis (Thor, 1930) | 40,00 | 19 | 1,25 | 40,00 | 2 | 0,03 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Proctolaelaps parvanalis (Thor 1930) | 40,00 | 3 | 0,20 | - | - | - | - | - | - | - | - | - | 20,00 | 2 | 0,22 | 20,00 | 1 | 0,08 | - | - | - | - | - | - |
| Zercon forsslundi Sellnick 1958 | 20,00 | 1 | 0,07 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Collembola | ||||||||||||||||||||||||
| Agrenia bidenticulata (Tullberg 1876) | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 80,00 | 29 | 96,67 |
| Anurida polaris (Hammer 1954) | - | - | - | 20,00 | 30 | 0,40 | - | - | - | 20,00 | 1 | 0,09 | - | - | - | - | - | - | - | - | - | - | - | - |
| Bonetogastrura nivalis (Martynova 1973) | 20,00 | 3 | 0,20 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Folsomia coeruleogrisea (Hammer, 1938) | 80,00 | 19 | 1,25 | - | - | - | - | - | - | - | - | - | 20,00 | 1 | 0,11 | - | - | - | 20,00 | 5 | 4,95 | - | - | - |
| Folsomia quadrioculata (Tullberg 1871) | 100,0 | 582 | 38,26 | 100,0 | 6353 | 85,48 | 80,00 | 1178 | 23,33 | 100,0 | 129 | 12,09 | 60,00 | 26 | 2,87 | 60,00 | 5 | 0,41 | 60,00 | 18 | 17,82 | - | - | - |
| Friesea quinquespinosa Wahlgren 1900 | 20,00 | 2 | 0,13 | 60,00 | 19 | 0,26 | 20,00 | 1 | 0,02 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Hypogastrura concolor (Carpenter 1900) | 100,0 | 315 | 20,71 | 80,00 | 65 | 0,87 | 100,0 | 1288 | 25,50 | 100,0 | 514 | 48,17 | 100,0 | 201 | 22,21 | 100,0 | 792 | 64,44 | 60,00 | 19 | 18,81 | 20,00 | 1 | 3,33 |
| Isotoma anglicana Lubbock 1862 | 100,0 | 24 | 1,58 | 80,00 | 93 | 1,25 | 60,00 | 26 | 0,51 | 60,00 | 19 | 1,78 | 40,00 | 6 | 0,66 | 40,00 | 8 | 0,65 | 80,00 | 47 | 46,53 | - | - | - |
| Megaphorura arctica (Tullberg 1876) | 20,00 | 3 | 0,20 | 40,00 | 35 | 0,47 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Oligaphorura ursi (Fjellberg, 1984) | 60,00 | 15 | 0,99 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Parisotoma notabilis (Schaeffer 1896) | - | - | - | - | - | - | 40,00 | 68 | 1,35 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Tetracanthella arctica Cassagnau 1959 | - | - | - | - | - | - | 20,00 | 1 | 0,02 | 20,00 | 1 | 0,09 | - | - | - | - | - | - | - | - | - | - | - | - |
| Lepidocyrtus lignorum (Fabricius 1793) | - | - | - | - | - | - | 20,00 | 5 | 0,10 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Sminthurinus concolor (Meinert, 1896) | 60,00 | 11 | 0,72 | - | - | - | 20,00 | 1 | 0,02 | 20,00 | 4 | 0,37 | - | - | - | - | - | - | 20,00 | 2 | 1,98 | - | - | - |
| Total | 1521 | 100 | 7432 | 100 | 5050 | 100 | 1067 | 100 | 905 | 100 | 1229 | 100 | 101 | 100 | 30 | 100 | ||||||||
References
- Walker, L.R.; del Moral, R. Primary Succession and Ecosystem Rehabilitation; Cambridge University Press: Cambridge, UK, 2003; p. 443. [Google Scholar]
- Walker, L.R.; del Moral, R. Primary Succession; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2011. [Google Scholar] [CrossRef]
- Jones, C.D.; del Moral, R. Patterns of primary succession on the foreland of Coleman Glacier, Washington, USA. Plant Ecol. 2005, 180, 105–116. [Google Scholar] [CrossRef]
- Peet, R.K. Community structure and ecosystem function. In Plant Succession: Theory and Prediction; Glenn-Lewin, D.C., Peet, R.K., Veblen, T.T., Eds.; Chapman & Hall: London, UK, 1992; pp. 103–151. [Google Scholar]
- Odum, E.P. The strategy of ecosystem development. Science 1969, 164, 262–270. [Google Scholar] [CrossRef]
- Matthews, J.A. The Ecology of Recently Deglaciated Terrain; Cambridge University Press: Cambridge, UK, 1992; p. 386. [Google Scholar]
- Whittaker, R.J.; Bush, M.B.; Richards, K. Plant recolonization and vegetation succession on the Krakatau Islands, Indonesia. Ecol. Monogr. 1989, 59, 59–123. [Google Scholar] [CrossRef]
- Reiners, W.A.; Worley, I.A.; Lawrence, D.B. Plant diversity in a chronosequence at Glacier Bay, Alaska. Ecology 1971, 52, 55–69. [Google Scholar] [CrossRef]
- Birks, H.J.B. The present flora and vegetation of the moraines of the Klutlan Glacier, Yukon Territory, Canada. Quat. Res. 1980, 14, 60–86. [Google Scholar] [CrossRef]
- Connell, J.H. Diversity in tropical rain forests and coral reefs. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef]
- Hågvar, S.; Solhøy, T.; Mong, C.E. Primary Succession of Soil Mites (Acari) in a Norwegian Glacier Foreland, with Emphasis on Oribatid Species. Arct. Antarct. Alp. Res. 2009, 41, 219–227. [Google Scholar] [CrossRef]
- Matthews, J.A.; Whittaker, R.J. Vegetation succession on the Storbreen glacier foreland, Jotunheimen, Norway: A review. Arct. Alp. Res. 1987, 19, 385–395. [Google Scholar] [CrossRef]
- Moreau, M.; Laffly, D.; Joly, D.; Brossard, T. Analysis of plant colonization on an arctic moraine since the end of the Little Ice Age using remotely sensed data and a Bayesian approach. Remote Sens. Environ. 2005, 99, 244–253. [Google Scholar] [CrossRef]
- Chapin, F.S.; Walker, L.R.; Fastie, C.L.; Sharman, L.C. Mechanisms of Primary Succession Following Deglaciation at Glacier Bay, Alaska. Ecol. Monogr. 1994, 64, 149–175. [Google Scholar] [CrossRef]
- Kaufmann, R. Invertebrate succession on an alpine glacier foreland. Ecology 2001, 82, 2261–2278. [Google Scholar] [CrossRef]
- Hodkinson, I.D.; Webb, N.R.; Coulson, S.J. Primary community assembly on land- the missing stages: Why are the heterotrophic organisms always there first? J. Ecol. 2002, 90, 569–577. [Google Scholar] [CrossRef]
- Kaufmann, R.; Fuchs, M.; Gosterxerier, N. The soil fauna of an alpine glacier foreland: Colonization and succession. Arct. Antarct. Alp. Res. 2002, 34, 242–250. [Google Scholar] [CrossRef]
- Hodkinson, I.D.; Coulson, S.J.; Webb, N.R. Invertebrate community assembly along pro-glacial chronosequences in the high Arctic. J. Anim. Ecol. 2004, 73, 556–568. [Google Scholar] [CrossRef]
- Skubała, P.; Gulvik, M. Pioneer oribatid mite communities (Acari, Oribatida) in newly exposed natural (glacier foreland) and anthropogenic (post-industrial dump) habitats. Pol. J. Ecol. 2005, 53, 395–407. [Google Scholar]
- Ávila-Jiménez, M.L.; Gwiazdowicz, D.J.; Coulson, S.J. The mesostigmatid mite (Acari: Parasitiformes) fauna of Svalbard: A revised inventory of a high Arctic archipelago. Zootaxa 2011, 3091, 33–41. [Google Scholar] [CrossRef]
- Kolodochka, L.A.; Gwiazdowicz, D.J. A new species of predaceous mite of the genus Neoseiulus Hughes (Acari, Phytoseiidae), with redescriptions of N.magnanalis (Thor) and N. ellesmerei (Chant & Hansell), from Svalbard, High Arctic. Zootaxa 2014, 3793, 441–452. [Google Scholar] [CrossRef]
- Gwiazdowicz, D.J.; Teodorowicz, E. Description of Halolaelaps coulsoni n.sp. (Acari, Halolaelapidae) from the High Arctic. Acarologia 2017, 57, 393–406. [Google Scholar] [CrossRef]
- Gwiazdowicz, D.J.; Rakowski, R. Redescription of Proctolaelaps parvanalis (Thor, 1930) (Acari: Ascidae) from Spitsbergen. Entomol. Fenn. 2009, 20, 281–286. [Google Scholar] [CrossRef]
- Gwiazdowicz, D.J.; Teodorowicz, E.; Coulson, S.J. Redescription of Arctoseius haarlovi Lindquist, 1963 (Acari: Ascidae) from Spitsbergen. Entomol. Fenn. 2011, 22, 140–148. [Google Scholar] [CrossRef]
- Coulson, S.J.; Fjellberg, A.; Gwiazdowicz, D.J.; Lebedeva, N.V.; Melekhina, E.N.; Solhøy, T.; Erséus, C.; Maraldo, K.; Miko, L.; Schatz, H.; et al. Introduction of invertebrates into the High Arctic via imported soils: The case of Barentsburg in the Svalbard. Biol. Invasions 2013, 15, 1–5. [Google Scholar] [CrossRef]
- Coulson, S.J.; Fjellberg, A.; Melekhina, E.N.; Taskaeva, A.A.; Lebedeva, N.V.; Belkina, O.; Seniczak, S.; Seniczak, A.; Gwiazdowicz, D.J. Microarthropod communities of industrially disturbed or imported soils in the High Arctic; the abandoned coal mining town of Pyramiden, Svalbard. Biodivers. Conserv. 2015, 24, 1671–1690. [Google Scholar] [CrossRef]
- Gwiazdowicz, D.J.; Coulson, S.J. High-Arctic gamasid mites (Acari, Mesostigmata): Community composition in Spitsbergen, Svalbard. Polar Res. 2011, 30, 8311. [Google Scholar] [CrossRef]
- Coulson, S.J.; Hodkinson, I.D.; Webb, N.R.; Mikkola, K.; Harrison, J.A.; Pedgley, D.E. Aerial colonisation of high Arctic islands by invertebrates: The diamondback moth, Plutella xylostella (Lepidoptera: Yponomeutidae) as a potential indicator species. Divers. Distrib. 2002, 8, 327–334. [Google Scholar] [CrossRef]
- Coulson, S.J.; Hodkinson, I.D.; Webb, N.R. Aerial dispersal of invertebrates over a high-Arctic glacier foreland: Midtre Lovénbreen, Svalbard. Polar Biol. 2003, 26, 530–537. [Google Scholar] [CrossRef]
- Hodkinson, I.D.; Webb, N.R.; Bale, A.J.S.; Blocks, W.; Coulson, S.J.; Strathdeet, A.T. Global Change and Arctic Ecosystems: Conclusions and Predictions from Experiments with Terrestrial Invertebrates on Spitsbergen. Arct. Alp. Res. 1998, 30, 306–313. [Google Scholar] [CrossRef]
- Førland, E.J.; Benestad, R.; Hanssen-Bauer, I.; Haugen, J.E.; Skaugen, T.E. Temperature and precipitation development at Svalbard 1900–2100. Adv. Meteorol. 2011, 2011, 893790. [Google Scholar] [CrossRef]
- Ghilarov, M.S.; Krivolutsky, D.A. (Eds.) Identification of Soil Mites, Sarcoptiformes; Izd, Nauka: Moscow, Russia, 1975; p. 491. (In Russian) [Google Scholar]
- Colloff, M.J. A taxonomic revision of the oribatid mite genus Camisia (Acari: Oribatida). J. Nat. Hist. 1993, 27, 1325–1408. [Google Scholar] [CrossRef]
- Weigmann, G. Hornmilben (Oribatida). Die Tierwelt Deutschlands, 76. Teil; Goecke and Evers: Keltern, Germany, 2006; p. 520. [Google Scholar]
- De Caceres, M.; Legendre, P. Associations between Species and Groups of Sites: Indices and Statistical Inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R package Version 2.5-6. 2019. Available online: https://CRAN.R-project.org/package=vegan (accessed on 15 August 2011).
- Hervé, M. RVAideMemoire: Testing and Plotting Procedures for Biostatistics. R Package Version 0.9-74. 2020. Available online: https://CRAN.R-project.org/package=RVAideMemoire (accessed on 1 February 2020).
- Subias, L.S. Listado sistemáTico, sinoníMico y Biogeográfico de los Ácaros Oribátidos (Acariformes, Oribatida) del mundo (1758–2002). Graellsia 60. 2014. Available online: http://www.ucm.es/info/zoo/Artropodos/Catalogo.pdf (accessed on 1 February 2020).
- Seniczak, S. The morphology of juvenile stages of moss mites of the family Camisiidae (Acari: Oribatida), V. Zool. Anz. 1991, 227, 173–184. [Google Scholar]
- Luxton, M. The oribatid mites of J. E. Hull. J. Nat. Hist. 1987, 21, 1273–1291. [Google Scholar] [CrossRef]
- Wissuwa, J.; Salamon, J.-A.; Frank, T. Oribatida (Acari) in grassy arable fallows are more affected by soil properties than habitat age and plant species. Eur. J. Soil Biol. 2013, 59, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Weigmann, G.; Kratz, W. Oribatid mites in urban zones of West Berlin. Biol. Fertily Soils 1987, 3, 81–84. [Google Scholar] [CrossRef]
- Skubała, P.; Rola, K.; Osyczka, P. Oribatid communities and heavy metal bioaccumulation in selected species associated with lichens in a strongly metal contaminated habitat. Environ. Sci. Pollut. Res. 2016, 23, 8861–8871. [Google Scholar] [CrossRef]
- Skubała, P.; Marzec, A. Importance of different types of beech dead wood for soil microarthropod fauna. Pol. J. Ecol. 2013, 61, 543–557. [Google Scholar]
- Cianciolo, J.M.; Norton, R.A. The ecological distribution of reproductive mode in oribatid mites, as related to biological complexity. Exp. Appl. Acarol. 2006, 40, 1–25. [Google Scholar] [CrossRef]
- Luxton, M. Studies on the oribatid mites of a Danish beech wood soil. I. Nutritional biology. Pedobiologia 1972, 12, 434–463. [Google Scholar]
- Skarżyński, D. The springtails (Collembola) of epilittoral of selected rivers and streams of Lower Silesia. Wiadomości Entomol. 1999, 17, 133–143. (In Polish) [Google Scholar]
- Skarżyński, D. Springtails (Collembola) of the Karkonosze Mountains (Poland). Fragm. Faun. 2001, 44, 203–212. [Google Scholar] [CrossRef]
- Traeger, G. Springtails of the Aggtelek National Park (Hexapoda: Collembola). In The Fauna of the Aggtelek National Park; Hungarian Natural History Museum: Budapest, Hungary, 1999; pp. 49–59. [Google Scholar]
- Fjellberg, A.; Bernard, E.C. Review of Agrenia Börner, 1906 with descriptions of four new species from North America (Collembola, Isotomidae). Zootaxa 2009, 2306, 17–28. [Google Scholar] [CrossRef]
- Fjellberg, A. Collembola from Jan Mayen, Bjornoya and Hopen with additions to the species list from Spitsbergen. Fauna Nor. Ser. B. 1984, 3, 69–76. [Google Scholar]
- Potapov, M. Synopses on Palearctic Collembola. Abh. Ber. Nat. Görlitz 2002, 73, 1–600. [Google Scholar]
- Fjellberg, A. The Collembola of Fennoscancandia and Denmark. Part II: Entomobryomorpha and Symphypleon. Fauna Entomol. Scand. 2007, 42, 1–266. [Google Scholar]
- Zmudczynska-Skarbek, K.; Zwolicki, A.; Convey, P.; Barcikowski, M.; Stempniewicz, L. Is ornithogenic fertilization important for collembolan communities in Arctic terrestrial ecosystems? Polar Res. 2015, 34. [Google Scholar] [CrossRef]
- Ponge, J.F. Biocenoses of Collembolga in Atlantic temperate grass-woodland ecosystems. Pedobiologia 1993, 37, 223–244. [Google Scholar]
- Ponge, J.F. Vertical distribution of Collembola (Hexapoda) and their food resources in organic horizons of beech forests. Biol. Fertil. Soils 2000, 32, 508–522. [Google Scholar] [CrossRef]
- Hågvar, S. Primary succession of springtails (Collembola) in a Norwegian glacier foreland. Arct. Antarct. Alp. Res. 2010, 42, 422–429. [Google Scholar] [CrossRef]
- Sengupta, S.; Ergon, T.; Leinaas, H.P. Genotypic differences in embryonic life history traits of Folsomia quadrioculata (Collembola: Isotomidae) across a wide geographical range. Ecol. Entomol. 2016, 41, 72–84. [Google Scholar] [CrossRef]
- Coulson, S.J.; Hodkinson, I.D.; Webb, N.R.; Harrison, J.A. Survival of terrestrial soil-dwelling arthropods on and in seawater: Implications for trans-oceanic dispersal. Funct. Ecol. 2002, 16, 353–356. [Google Scholar] [CrossRef]
- Huhta, V.; Siira-Pietikäinen, A.; Penttinen, R.; Räty, M. Soil fauna of Finland: Acarina, Collembola and Enchytraeidae. Memoranda Societatis pro Fauna et Flora Fennica 2010, 86, 59–82. [Google Scholar]
- Fjellberg, A. The Collembola of the Norwegian Arctic Islands. Medd. Nor. Polarinst. 1994, 133, 1–57. [Google Scholar]
- Coulson, S.J.; Schatz, H.; Gwiazdowicz, D.J.; Solhøy, T. On the oribatid and mesostigmatid mites (Acari) of the High Arctic island of Hopen. Pol. Polar Res. 2014, 35, 133–139. [Google Scholar] [CrossRef]
- Binns, E.S. Arctoseius cetratus (Sellnick) (Acarina: Ascidae) phoretic on mushroom sciarid flies. Acarologia 1972, 14, 350–356. [Google Scholar]
- Seyd, E.L. Studies on the moss mites of Snowdonia (Acari: Oribatei). 2. The Cnicht. Biol. J. Linn. Soc. 1981, 15, 287–298. [Google Scholar] [CrossRef]
- Schatz, H. Catalogus Fauna Austriae. Teil IX U.-Ordn.: Oribatei, Hornmilben. Ősterreichischen; Akademie der Wissenschaften: Vienna, Austria, 1983; p. 118. ISBN 13 978-3-7001-0613-5. [Google Scholar]
- Babenko, A.B. Collembolan assemblages of polar deserts and subarctic nival communities. Pedobiologia 2000, 44, 421–429. [Google Scholar] [CrossRef]
- Block, W. Low temperature Effects on Microarthropods. J. Therm. Biol. 1981, 6, 215–218. [Google Scholar] [CrossRef]
- Sømme, L. Adaptations of Terrestrial Arthropods to the Alpine Environment. Biol. Rev. 1989, 64, 367–407. [Google Scholar] [CrossRef]
- Babenko, A.B. The Structure of Springtail Fauna (Collembola) of the Arctic. Entomol. Rev. 2005, 85, 878–901. [Google Scholar]
- Coulson, S.J.; Birkemoe, T. Long-term cold tolerance in Arctic invertebrates: Recovery after 4 years at below −20 °C. Can. J. Zool. 2000, 78, 2055–2058. [Google Scholar] [CrossRef]
- Birkemoe, T.; Leinaas, H.P. Reproductive biology of the arctic collembolan Hypogastrura tullbergi. Ecography 1999, 22, 31–39. [Google Scholar] [CrossRef]
- Dunger, W.; Schulz, H.J.; Zimdars, B. Colonization behavior of Collembola under different conditions of dispersal. Pedobiologia 2002, 46, 316–327. [Google Scholar]
- Petersen, H. General aspects of collembolan ecology at the turn of the millennium. Pedobiologia 2002, 46, 246–260. [Google Scholar] [CrossRef]
- Thor, S. Beiträge zur Kenntnis der Invertebraten Fauna von Svalbard; Skrifer om Svalbard og Ishavet 27; Norway’s Svalbard and Arctic Ocean Research Survey: Oslo, Norway, 1930; p. 156. [Google Scholar]
- Petrova, A.D. Family Zerconidae Cannestrini, 1891. In Key to Mites Living in Soil [Mesostigmata]; Ghilarov, M.S., Bregetova, N.G., Eds.; Nauka: Leningrad, Russia, 1977; pp. 577–621. (In Russian) [Google Scholar]
| Parameters | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Total number of species | 22 | 16 | 18 | 13 | 11 | 7 | 7 | 2 |
| Mean number of species ± SD | 14 ± 1.41 | 10.6 ± 0.89 | 10.6 ± 1.82 | 6.6 ± 0.55 | 5.4 ± 1.14 | 4.8 ± 1.30 | 3 ± 1.00 | 1 ± 0,71 |
| Total abundance | 1521 | 7432 | 5050 | 1067 | 905 | 1229 | 101 | 30 |
| Mean abundance ± SD | 304.2 ± 162.48 | 1486.4 ± 2698.67 | 1010 ± 967.12 | 213.4 ± 139.46 | 181 ± 199.35 | 245.8 ± 145.08 | 20.2 ± 7.95 | 6 ± 6.67 |
| Simpson (D) ± SD | 4.61 ± 0.06 | 1.36 ± 0.29 | 3.94 ± 0.11 | 3.46 ± 0.17 | 2.25 ± 0.19 | 2.09 ± 0.19 | 3.43 ± 0.16 | 1.07 ± 0.05 |
| Evenness (E) | 0.6277 | 0.2452 | 0.5551 | 0.5950 | 0.4740 | 0.4980 | 0.7707 | 0.2108 |
| Dominance | 0.2170 | 0.7361 | 0.2538 | 0.2893 | 0.4452 | 0.4774 | 0.2916 | 0.9356 |
| Effects | Df | SS | MS | Pseudo F | p |
|---|---|---|---|---|---|
| Locations | 7 | 5.9071 | 0.84386 | 9.436 | 0.001 |
| Residuals | 31 | 2.7724 | 0.08943 | ||
| Total | 38 | 8.6795 |
| Groups | Species | Stat | p Value |
|---|---|---|---|
| 1 | Oppiella neerlandica | 0.843 | 0.001 |
| Hermannia reticulata | 0.789 | 0.001 | |
| Arctoseius haarlovi | 0.712 | 0.006 | |
| Oligaphorura ursi | 0.687 | 0.008 | |
| 2 | Friesea quinquespinosa | 0.488 | 0.044 |
| 3 | Camisia dictyna | 0.792 | 0.001 |
| 5 | Diapterobates notatus | 0.676 | 0.002 |
| 8 | Agrenia bidenticulata | 0.785 | 0.001 |
| 2 + 3 | Arctoseius multidentatus | 0.743 | 0.001 |
| 2 + 6 | Tectocepheus velatus | 0.551 | 0.046 |
| 3 + 4 | Liochthonius lapponicus | 0.581 | 0.026 |
| 3 + 6 | Camisia foveolata | 0.627 | 0.017 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gwiazdowicz, D.J.; Zawieja, B.; Olejniczak, I.; Skubała, P.; Gdula, A.K.; Coulson, S.J. Changing Microarthropod Communities in Front of a Receding Glacier in the High Arctic. Insects 2020, 11, 226. https://doi.org/10.3390/insects11040226
Gwiazdowicz DJ, Zawieja B, Olejniczak I, Skubała P, Gdula AK, Coulson SJ. Changing Microarthropod Communities in Front of a Receding Glacier in the High Arctic. Insects. 2020; 11(4):226. https://doi.org/10.3390/insects11040226
Chicago/Turabian StyleGwiazdowicz, Dariusz J., Bogna Zawieja, Izabella Olejniczak, Piotr Skubała, Anna K. Gdula, and Stephen J. Coulson. 2020. "Changing Microarthropod Communities in Front of a Receding Glacier in the High Arctic" Insects 11, no. 4: 226. https://doi.org/10.3390/insects11040226
APA StyleGwiazdowicz, D. J., Zawieja, B., Olejniczak, I., Skubała, P., Gdula, A. K., & Coulson, S. J. (2020). Changing Microarthropod Communities in Front of a Receding Glacier in the High Arctic. Insects, 11(4), 226. https://doi.org/10.3390/insects11040226







