Seasonal Migrations of Pantala flavescens (Odonata: Libellulidae) in Middle Asia and Understanding of the Migration Model in the Afro-Asian Region Using Stable Isotopes of Hydrogen
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Stable Isotope Analysis
2.3. Data Analysis
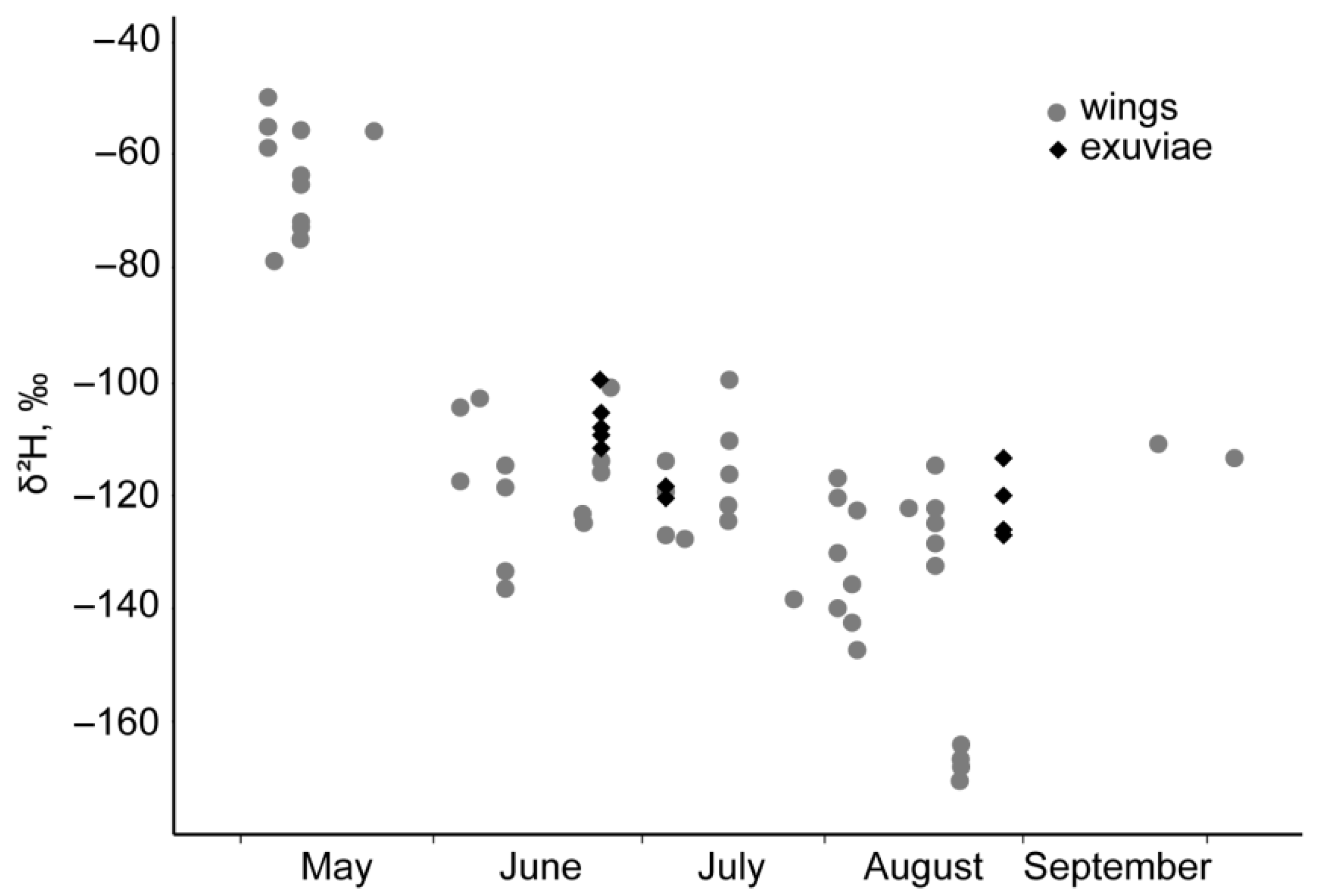
3. Results
4. Discussion
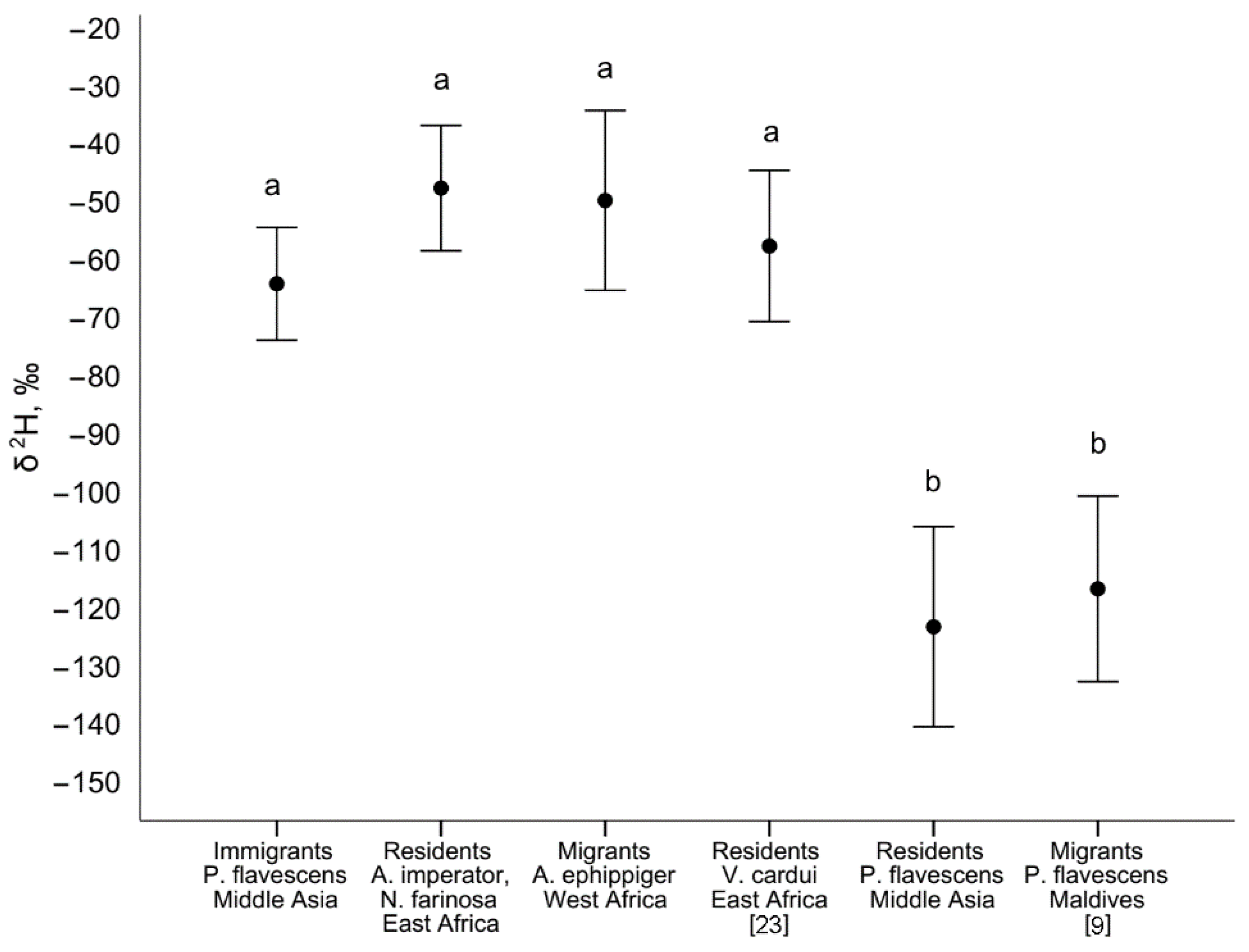
4.1. P. flavescens in Middle Asia and Isotope Evidence of Spring Immigration
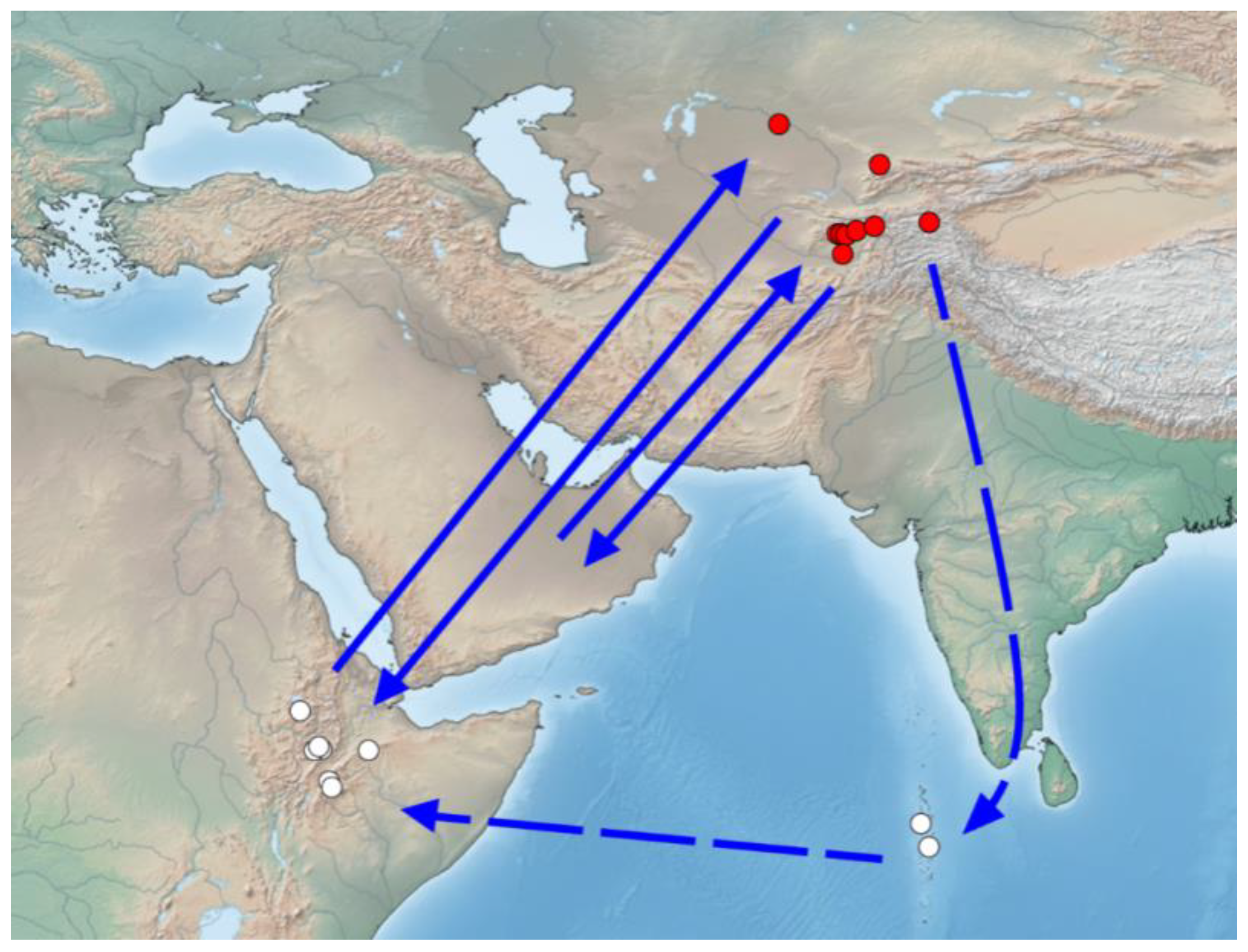
4.2. The Probable Region of Origin of Immigrants Is East Africa and the Arabian Peninsula
4.3. Probable Migration Pattern in the Afro-Asian Region
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kalkman, V.J.; Monnerat, C. Pantala flavescens (Fabricius, 1798). In Atlas of the European Dragonflies and Damselflies; Boudot, J.-P., Kalkman, V.J., Eds.; KNNNV Publishing: Amersfoort, The Netherlands, 2015; pp. 289–291. [Google Scholar]
- Devaud, M.; Lebouvier, M. First record of Pantala flavescens (Anisoptera: Libellulidae) from the remote Amsterdam Island, southern Indian Ocean. Polar Biol. 2019, 42, 1041–1046. [Google Scholar] [CrossRef]
- Moore, N.W. Behaviour of imaginal Pantala flavescens (Fabr.) on Eastern Island (Anisoptera: Libellulidae). Odonatologica 1993, 22, 71–76. [Google Scholar]
- Samways, M.; Osborn, R. Divergence in a transoceanic circumtropical dragonfly on a remote island. J. Biogeogr. 1998, 25, 935–946. [Google Scholar] [CrossRef]
- Corbet, P.S. Dragonflies: Behavior and Ecology of Odonata; Harley Books: Colchester, UK, 1999; p. 829. [Google Scholar]
- Feng, H.-Q.; Wu, K.-M.; Ni, Y.-X.; Cheng, D.-F.; Guo, Y.-Y. Nocturnal migration of dragonflies over the Bohai Sea in northern China. Ecol. Entomol. 2006, 31, 511–520. [Google Scholar] [CrossRef]
- Anderson, R.C. Do dragonflies migrate across the western Indian Ocean? J. Trop. Ecol. 2009, 25, 347–358. [Google Scholar] [CrossRef]
- May, M.L. A critical overview of progress in studies of migration of dragonflies (Odonata: Anisoptera), with emphasis on North America. J. Insect Conserv. 2013, 17, 1–15. [Google Scholar] [CrossRef]
- Hobson, K.A.; Anderson, R.C.; Soto, D.X.; Wassenaar, L.I. Isotopic evidence that dragonflies (Pantala flavescens) migrating through the Maldives come from the northern Indian Subcontinent. PLoS ONE 2012, 7, e52594. [Google Scholar] [CrossRef]
- Holland, R.A.; Wikelski, M.; Wilcove, D.S. How and why do insects migrate? Science 2006, 313, 794–796. [Google Scholar] [CrossRef]
- Borisov, S.N. Migrant dragonflies in Middle Asia. 3. Pantala flavescens (Fabricius, 1798) (Odonata, Libellulidae). Evraziatskii Entomol. Zhurnal 2012, 11, 37–41. (In Russian) [Google Scholar]
- Hobson, K.A. Application of Isotopic Methods to Tracking Animal Movements. In Tracking Animal Migration with Stable Isotopes; Academic Press: London, UK, 2019; pp. 85–115. [Google Scholar]
- Brattström, O.; Bensch, S.; Wassenaar, L.I.; Hobson, K.A.; Akesson, S. Understanding the migration ecology of European red admirals Vanessa atalanta using stable hydrogen isotopes. Ecography 2010, 33, 720–729. [Google Scholar] [CrossRef]
- Hobson, K.A.; Soto, D.X.; Paulson, D.R.; Wassenaar, L.I.; Matthews, J.H. A dragonfly (2H: 1H) isoscape for North America: A new tool for determining natal origins of migratory aquatic emergent insects. Method Ecol. Evol. 2012, 3, 766–772. [Google Scholar] [CrossRef]
- van Hardenbroek, M.; Gröcke, D.R.; Sauer, P.E.; Elias, S.A. North American transect of stable hydrogen and oxygen isotopes in water beetles from a museum collection. J. Paleolimnol. 2012, 48, 461–470. [Google Scholar] [CrossRef]
- Hobson, K.A. Tracking migratory insects using stable isotopes. Bull. Entomol. Soc. Can. 2012, 44, 127–132. [Google Scholar]
- Hallworth, M.T.; Marra, P.P.; McFarland, K.P.; Zahendra, S.; Studds, C.E. Tracking dragons: Stable isotopes reveal the annual cycle of a long-distance migratory insect. Biol. Lett. 2018, 14, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.-Z.; Fu, X.-W.; Hu, C.-H.; Wu, K.-M. Seasonal Migration of Pantala flavescens Across the Bohai Strait in Northern China. Environ. Entomol. 2018, 47, 264–270. [Google Scholar] [CrossRef]
- Borisov, S.N.; Iakovlev, I.K.; Borisov, A.S.; Zuev, A.G.; Tiunov, A.V. Isotope evidence for latitudinal migrations of the dragonfly Sympetrum fonscolombii (Odonata: Libellulidae) in Middle Asia. Ecol. Entomol. 2020, 45, 1445–1456. [Google Scholar] [CrossRef]
- Dumont, H.J.; Desmet, K. Transsahara and transmediterranean migratory activity of Hemianax ephippiger (Burm.) in 1988 and 1989 (Anisoptera: Aeshnidae). Odonatologica 1990, 19, 181–185. [Google Scholar]
- Dumont, H.J. Odonata from the Tibesti Mountains and the Ounianga Lakes in Chad, with notes on Hemianax ephippiger accumulating in the desert. Odonatologica 2014, 43, 13–24. [Google Scholar]
- Lambret, P.; Boudot, J.-P. Hemianax ephippiger (Burmeister, 1839) (Odonata, Anisoptera: Aeshnidae): Présentation générale. In Martinia, Hors-série, Hemianax Ephippiger—Migration 2011; Société Française d’Odonatologie: Bois-d’Arcy, France, 2013; pp. 13–27. [Google Scholar]
- Stefanescu, C.; Soto, D.X.; Talavera, G.; Vila, R.; Hobson, K.A. Long-distance autumn migration across the Sahara by painted lady butterflies: Exploiting resource pulses in the tropical savannah. Biol. Lett. 2016, 12, 20160561. [Google Scholar] [CrossRef]
- Kalnay, E.; Kanamitsu, M.; Kistler, R.; Collins, W.; Deaven, D.; Gandin, L.; Zhu, Y. The NCEP/NCAR 40-year reanalysis project. Bull. Am. Meteorol. Soc. 1996, 77, 437–471. [Google Scholar] [CrossRef]
- Wassenaar, L.I.; Hobson, K.A. Comparative equilibration and online technique for determination of non-exchangeable hydrogen of keratins for use in animal migration studies. Isot. Environ. Health Stud. 2003, 39, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Shorthouse, D.P. SimpleMappr, an Online Tool to Produce Publication-Quality Point Maps. 2010. Available online: http://www.simplemappr.net (accessed on 13 February 2020).
- Ichikawa, Y.; Yokoi, T.; Watanabe, M. Thermal factors affecting egg development in the wandering glider dragonfly, Pantala flavescens (Odonata: Libellulidae). Appl. Entomol. Zool. 2017, 52, 89–95. [Google Scholar] [CrossRef]
- Corbet, P.S. (Ed.) Current topics in dragonfly biology. 3. A discussion focussing on the seasonal ecology of Pantala flavescens in the Indian subcontinent. Rapid Commun. 1988, 8, 1–24. [Google Scholar]
- Borisov, S.N.; Malikova, E.I. Distribution and migration strategy of Pantala flavescens (Fabricius, 1798) (Odonata, Libellulidae) near the northern limit of the range in Transbaikalia and in the Far East of Russia Evraziatskii Entomol. Zhurnal 2019, 18, 155–162. [Google Scholar] [CrossRef]
- Borisov, A.S.; Borisov, S.N. Spring immigration of dragonflies (Odonata) in Tajikistan. Evraziatskii Entomol. Zhurnal 2019, 18, 305–311. [Google Scholar] [CrossRef]
- Kumar, A. On the life history of Pantala flavescens (Fabricius) (Libellulidae: Odonata). Ann. Entomol. 1984, 2, 43–50. [Google Scholar]
- Hawking, J.H.; Ingram, B.A. Rate of larval development of Pantala flavescens (Fabricius) at its southern limit of range in Australia (Anisoptera: Libellulidae). Odonatologica 1994, 23, 63–68. [Google Scholar]
- Suhling, F.; Schenk, K.; Padeffke, T.; Martens, A. A field study of larval development in a dragonfly assemblage in African desert ponds (Odonata). Hydrobiologia 2004, 528, 75–85. [Google Scholar] [CrossRef]
- Kumar, A. The phenology of dragonflies in the Dehra Dun Valley, India. Odonatologica 1972, 1, 199–207. [Google Scholar]
- Kulkarni, A.S.; Subramanian, K.A. Habitat and seasonal distribution of Odonata (Insecta) of Mula and Mutha river basins, Maharashtra, India. J. Threat. Taxa 2013, 5, 4084–4095. [Google Scholar] [CrossRef]
- Sharma, G. Studies on the reproductive behaviour of dragonfly, Pantala flavescens (Fabricius, 1798) (Odonata: Insecta: Arthropoda) in Aravalli range and desert ecosystem of Rajasthan, India. Biol. Bull. 2017, 3, 67–73. [Google Scholar]
- Schneider, T.; Ikemeyer, D.; Müller, O.; Dumont, H. Checklist of the dragonflies (Odonata) of Iran with new records and notes on distribution and taxonomy. Zootaxa 2018, 4394, 1–40. [Google Scholar] [CrossRef]
- Boudot, J.-P.; Kalkman, V.J.; Amorín, M.F.; Bogdanović, T.; Rivera, A.C.; Degabriele, G.; Dommanget, J.L.; Ferreira, S.; Garrigós, B.; Jović, M.; et al. Atlas of the Odonata of the Mediterranean and North Africa. Libellula Suppl. 2009, 9, 1–256. [Google Scholar]
- Clausnitzer, V.; Dijkstra, K.-D.B.; Koch, R.; Boudot, J.-P.; Darwall, W.R.T.; Kipping, J.; Samraoui, B.; Samways, M.J.; Simaika, J.P.; Suhling, F. Focus on African Freshwaters: Hotspots of dragonfly diversity and conservation concern. Front. Ecol. Environ. 2012, 10, 129–134. [Google Scholar] [CrossRef]
- Corbet, P.S. Orientation and reproductive condition of migrating dragonflies (Anisoptera). Odonatologica 1984, 13, 81–88. [Google Scholar]
- Waterston, A.R.; Pittaway, A.R. The Odonata or dragonflies of Oman and neighboring territories. J. Oman Stud. 1991, 10, 131–168. [Google Scholar]
- Lambret, P.; Boudot, J.-P.; Chelmick, D.; De Knijf, G.; Durand, É.; Judas, J.; Stoquert, A. Odonata surveys 2010–2016 in the United Arab Emirates and the Sultanate of Oman, with emphasis on some regional heritage species. Odonatologica 2017, 46, 153–205. [Google Scholar]
- Schneider, T.; Ikemeyer, D. Dragonflies of Oman—A revised illustrated checklist (Odonata). Entomol. Zeit. 2016, 126, 137–147. [Google Scholar]
- Reels, G.T. Emergence patterns and adult flight season of Anisoptera at a managed wetland site in Hong Kong, southern China. Int. J. Odonatol. 2011, 14, 33–48. [Google Scholar] [CrossRef]
- Tajikistan. Nature and Natural Resources; Donish: Dushanbe, Tajikistan, 1982; p. 601. (In Russian)
- Campbell, O.J.; Reimer, R.W. An influx of Sympetrum fonscolombii Selys in the United Arab Emirates. Agrion 2011, 15, 20–21. [Google Scholar]
- Monnerat, C.; Al Dhafer, H.M. Odonata records from southwestern Saudi Arabia. Not. Odonatol. 2016, 8, 203–245. [Google Scholar]
- Borisov, S.N. Migrations of dragonflies (Odonata) in Central Asia: A review. Part 1. Latitudinal migrations. Evraziatskii Entomol. Zhurnal 2015, 14, 241–256. (In Russian) [Google Scholar]
- Chapman, J.W.; Bell, J.R.; Burgin, L.E.; Reynolds, D.R.; Pettersson, L.B.; Hill, J.K.; Bonsall, M.B.; Thomas, J.A. Seasonal migration to high latitudes results in major reproductive benefits in an insect. Proc. Natl. Acad. Sci. USA 2012, 109, 14924–14929. [Google Scholar] [CrossRef] [PubMed]
- Satterfield, D.A.; Sillett, T.S.; Chapman, J.W.; Altizer, S.; Marra, P.P. Seasonal insect migrations: Massive, influential, and overlooked. Front. Ecol. Environ. 2020, 18, 335–344. [Google Scholar] [CrossRef]

| Locality | n | Wings/ Exuviae | Date of Collection | Country | Nearest Settlement | Altitude, m a.s.l. | Latitude, N | Longitude, E |
|---|---|---|---|---|---|---|---|---|
| 1 | 1 | wings | 22.V.1976 | Kazakhstan | Kyzylorda | 170 | 44°50′ | 64°55′ |
| 1 | 5 | wings | 18.VIII.1976 | Kazakhstan | Kyzylorda | 170 | 44°50′ | 64°55′ |
| 2 | 1 | wings | 14.VIII.1973 | Kazakhstan | Chokpak | 1183 | 42°31′ | 70°36′ |
| 2 | 1 | wings | 04.X.2010 | Kazakhstan | Chokpak | 1183 | 42°31′ | 70°36′ |
| 3 | 2 | wings | 05.VI.1978 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 3 | 1 | wings | 08.VI.1978 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 3 | 4 | wings | 12.VI.1979 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 3 | 2 | wings | 24.VI.1978 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 3 | 1 | wings | 28.VI.1982 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 3 | 1 | wings | 10.VII.1984 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 3 | 1 | wings | 27.VII.1977 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 3 | 2 | wings | 05.VIII.1978 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 3 | 2 | wings | 06.VIII.1979 | Tajikistan | Tigrovaya Balka | 340 | 37°25′ | 68°30′ |
| 4 | 3 | wings | 06.V.2019 | Tajikistan | Regar | 732 | 38°32′ | 68°13′ |
| 4 | 1 | wings | 07.V.2019 | Tajikistan | Regar | 732 | 38°32′ | 68°13′ |
| 4 | 7 | wings | 11.V.2019 | Tajikistan | Regar | 732 | 38°32′ | 68°13′ |
| 5 | 5 | wings | 17.VII.1988 | Tajikistan | Shakhrinau | 850 | 38°34′ | 68°20′ |
| 6 | 3 | wings | 07.VII.1988 | Tajikistan | Gissar | 712 | 38°28′ | 68°36′ |
| 6 | 2 | exuviae | 07.VII.1988 | Tajikistan | Gissar | 712 | 38°28′ | 68°36′ |
| 6 | 1 | wings | 22.IX.1991 | Tajikistan | Gissar | 712 | 38°28′ | 68°36′ |
| 7 | 2 | wings | 27.VI.1981 | Tajikistan | Ramit | 1195 | 38°44′ | 69°19′ |
| 7 | 5 | exuviae | 27.VI.1981 | Tajikistan | Ramit | 1195 | 38°44′ | 69°19′ |
| 8 | 4 | wings | 22.VIII.1987 | Tajikistan | Garm | 1300 | 39°00′ | 70°18′ |
| 8 | 4 | exuviae | 29.VIII.1987 | Tajikistan | Garm | 1300 | 39°00′ | 70°18′ |
| 9 | 4 | wings | 03.VIII.1980 | Tajikistan | Uy-bulak pass 1 | 4260 | 39°12′ | 73°26′ |
Publisher′s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borisov, S.N.; Iakovlev, I.K.; Borisov, A.S.; Ganin, M.Y.; Tiunov, A.V. Seasonal Migrations of Pantala flavescens (Odonata: Libellulidae) in Middle Asia and Understanding of the Migration Model in the Afro-Asian Region Using Stable Isotopes of Hydrogen. Insects 2020, 11, 890. https://doi.org/10.3390/insects11120890
Borisov SN, Iakovlev IK, Borisov AS, Ganin MY, Tiunov AV. Seasonal Migrations of Pantala flavescens (Odonata: Libellulidae) in Middle Asia and Understanding of the Migration Model in the Afro-Asian Region Using Stable Isotopes of Hydrogen. Insects. 2020; 11(12):890. https://doi.org/10.3390/insects11120890
Chicago/Turabian StyleBorisov, Sergey N., Ivan K. Iakovlev, Alexey S. Borisov, Mikhail Yu. Ganin, and Alexei V. Tiunov. 2020. "Seasonal Migrations of Pantala flavescens (Odonata: Libellulidae) in Middle Asia and Understanding of the Migration Model in the Afro-Asian Region Using Stable Isotopes of Hydrogen" Insects 11, no. 12: 890. https://doi.org/10.3390/insects11120890
APA StyleBorisov, S. N., Iakovlev, I. K., Borisov, A. S., Ganin, M. Y., & Tiunov, A. V. (2020). Seasonal Migrations of Pantala flavescens (Odonata: Libellulidae) in Middle Asia and Understanding of the Migration Model in the Afro-Asian Region Using Stable Isotopes of Hydrogen. Insects, 11(12), 890. https://doi.org/10.3390/insects11120890






