Coffee Berry Borer (Hypothenemus hampei), a Global Pest of Coffee: Perspectives from Historical and Recent Invasions, and Future Priorities
Abstract
Simple Summary
Abstract
1. Introduction
2. Taxonomy and Identification
3. Origin
4. Biology and Life History
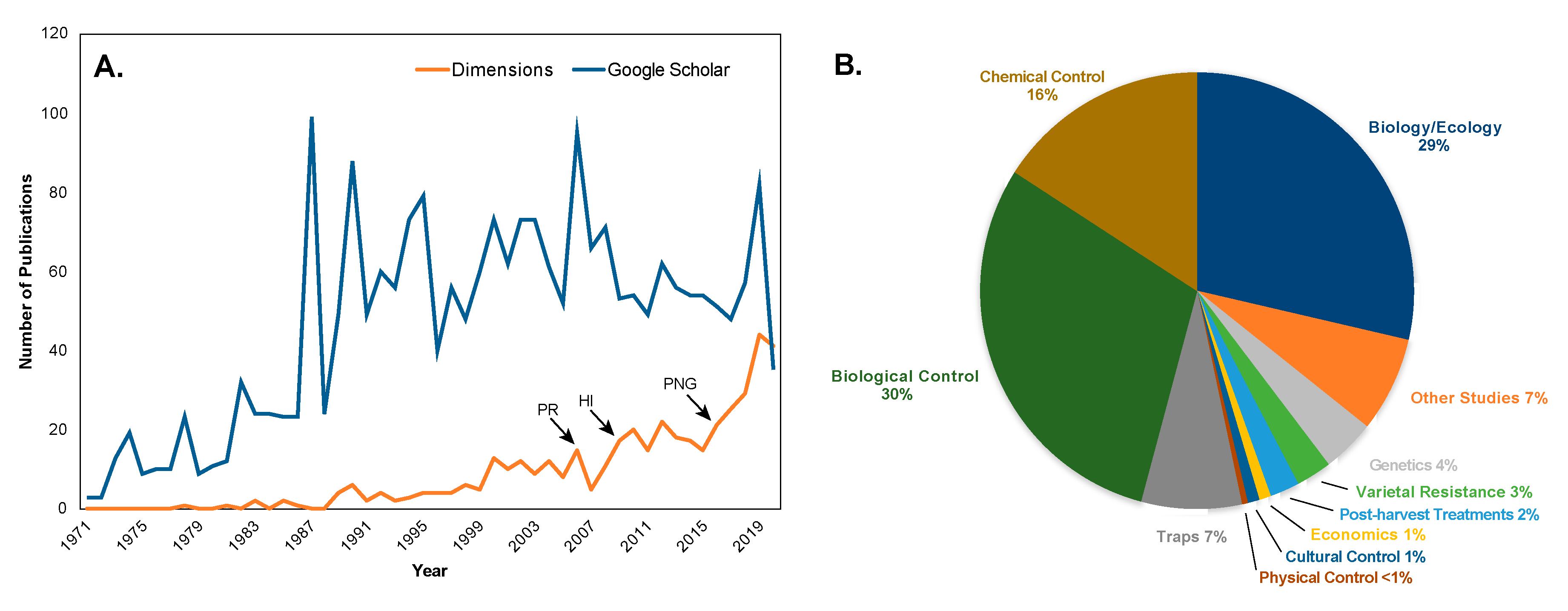
5. Worldwide Pest Status
6. Insights from Historical Invasions: Indonesia, Brazil, Ethiopia
6.1. Social and Economic Constraints Are Preventing Implementation of IPM: Indonesia as a Case Study
6.2. Chemical Controls Are Not Sustainable: Brazil as a Case Study
6.3. Climate Change Is Driving CBB’s Invasion Success: Ethiopia as a Case Study
6.4. Summary: What Have We Learned from Historical Invasions?
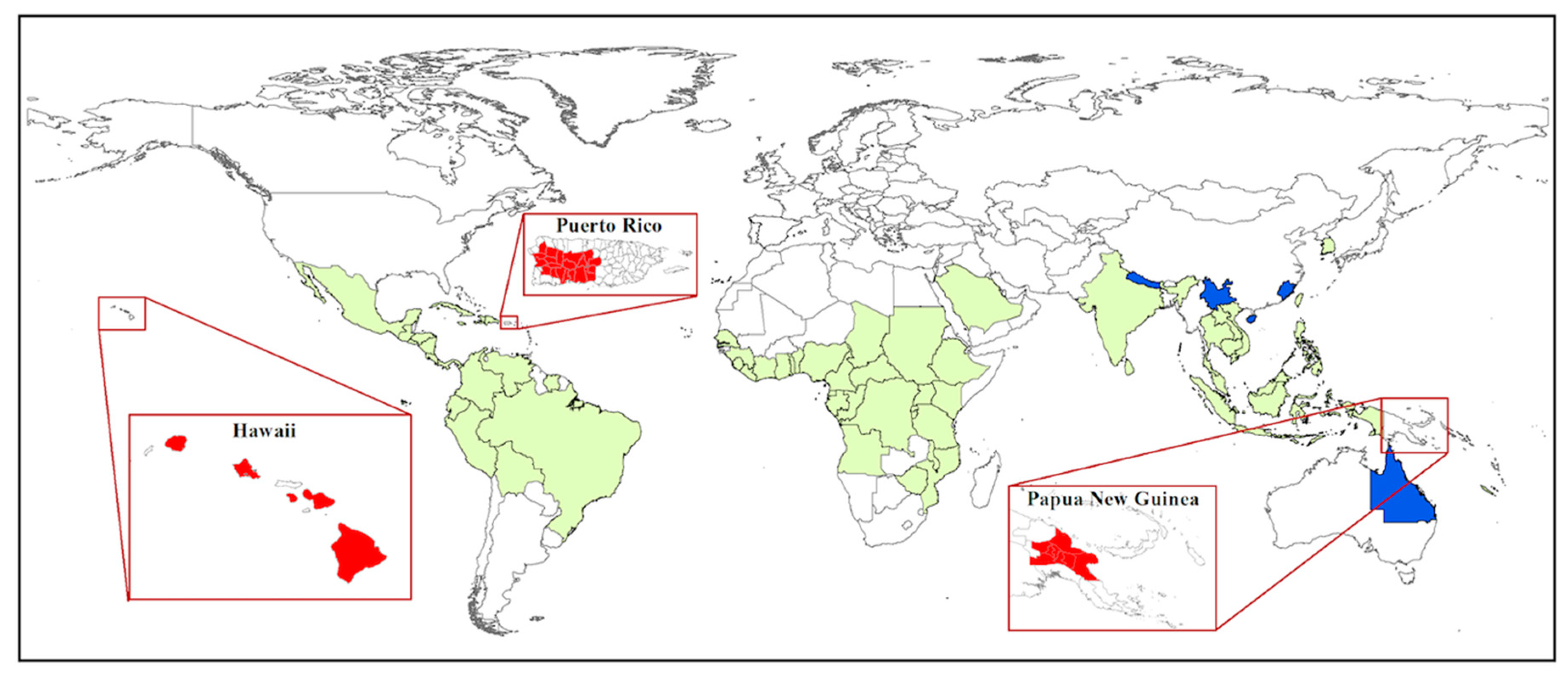
7. New Perspectives from Recent Invasions: Puerto Rico, Hawaii, Papua New Guinea
7.1. Puerto Rico
7.2. Hawaii
7.3. Papua New Guinea
7.4. Summary: Insights from Recent Invasions
8. Current State of CBB Management: Hawaii as a Case Study
8.1. Monitoring: Traps and Tree Sampling
8.2. Cultural Control: Pre- and Post-Harvest Sanitation
8.3. Biological Control: Natural Enemies
8.4. Chemical Control: Insecticides and Repellants
8.5. Physical Control: Exclusion Netting and Border Crops
9. Biosecurity Issues
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Le Pelley, R.H. Pests of Coffee; Longmans Green and Co.: London, UK, 1968; p. 590. [Google Scholar]
- Baker, P.S. The Coffee Berry Borer in Colombia; Final Report of the DFID-Cenicafé-CABI Bioscience IPM for Coffee Project; The Commodities Press: Chinchiná, Colombia, 1999; p. 154. [Google Scholar]
- Vega, F.E.; Benavides, P.; Stuart, J.A.; O’Neill, S.L. Wolbachia infection in the coffee berry borer (Coleoptera: Scolytidae). Ann. Entomol. Soc. Am. 2002, 95, 374–378. [Google Scholar] [CrossRef]
- Damon, A. A review of the biology and control of the coffee berry borer, Hypothenemus hampei (Coleoptera: Scolytidae). Bull. Entomol. Res. 2000, 90, 453–465. [Google Scholar] [CrossRef] [PubMed]
- Vega, F.E.; Infante, F.; Johnson, A.J. The genus Hypothenemus, with emphasis on H. hampei, the coffee berry borer. In Bark Beetles: Biology and Ecology of Native and Invasive Species; Vega, F.E., Hofstetter, R.W., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 427–494. [Google Scholar]
- Bottrill, M.; Ludowici, V.; Turner, R. Biosecurity Plan for the Australian Coffee Industry, V. 1.0-2018; Publication No. 18/023; AgriFutures: Wagga Wagga, NSW, Australia, 2018. [Google Scholar]
- Vega, F.E.; Infante, F.; Castillo, A.; Jaramillo, J. The coffee berry borer, Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae): A short review, with recent findings and future research directions. Terr. Arthropod Rev. 2009, 2, 129–147. [Google Scholar]
- Jaramillo, J.; Muchugu, E.; Vega, F.E.; Davis, A.; Borgemeister, C.; Chabi-Olaye, A. Some like it hot: The influence and implications of climate change on coffee berry borer (Hypothenemus hampei) and coffee production in East Africa. PLoS ONE 2011, 6, e24528. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.L.; Bright, D.E., Jr. A catalog of Scolytidae and Platypodidae (Coleoptera), Part 2: Taxonomic Index, Volume B. Great Basin Nat. Mem. 1992, 13, 835–1553. [Google Scholar]
- Bright, D.E.; Skidmore, R.E. A Catalog of Scolytidae and Platypodidae (Coleoptera); Supplement 1 (1990–1994); NRC Research Press: Ottawa, ON, Canada, 1997. [Google Scholar]
- Bright, D.E.; Skidmore, R.E. A Catalog of Scolytidae and Platypodidae (Coleoptera); Supplement 2 (1995–1999); NRC Research Press: Ottawa, ON, Canada, 2002. [Google Scholar]
- Bright, D.E. A catalog of Scolytidae and Platypodidae (Coleoptera), Supplement 3 (2000–2010). Insecta Mundi 2014, 0356, 1–336. [Google Scholar]
- Wood, S.L. A reclassification of the genera of Scolytidae (Coleoptera). Great Basin Nat. Mem. 1986, 10, 1–126. [Google Scholar]
- Ferrari, J.A.G. Die Forst- und Baumzuchtschädlichen Borkenkäfer; Druck und Verlag von Carl Gerold’s Sohn: Wien, Austria, 1867; pp. 12–13. [Google Scholar]
- Waterhouse, D.F.; Norris, K.R. Biological Control: Pacific Prospects; Supplement 1; ACIAR Monograph No. 12; Australian Centre for International Agricultural Research: Canberra, Australia, 1989; p. 125.
- Eichhoff, W.J. Neue exotische Tomiciden-Arten. Berl. Entomol. Z. 1871, 15, 131–137. [Google Scholar] [CrossRef]
- Swaine, J.M. Catalogue of the described Scolytidae of America, north of Mexico. 24th Report of the State Entomologist on Injurious and other Insects of the State of New York 1908, Appendix B., Education Department Bulletin no. 455, New York State Museum. Mus. Bull. 1909, 134, 76–159. [Google Scholar]
- Browne, F.G. Taxonomic notes on Scolytidae (Coleoptera). Entomol. Ber. 1963, 23, 53–59. [Google Scholar]
- Hopkins, A.D. Classification of the Cryphalinæ, with Descriptions of New Genera and Species; Contributions from the Bureau of Entomology, Report No. 99; United States Department of Agriculture: Washington, DC, USA, 1915; p. 75.
- Hulcr, J.; Atkinson, T.H.; Cognato, A.I.; Jordal, B.H.; McKenna, D.D. Morphology, taxonomy, and phylogenetics of bark beetles. In Bark Beetles: Biology and Ecology of Native and Invasive Species; Vega, F.E., Hofstetter, Eds.; Academic Press: San Diego, CA, USA, 2015; Chapter 2; pp. 41–84. [Google Scholar]
- Murphy, S.T.; Moore, D. Biological control of the coffee berry borer, Hypothenemus hampei (Ferrari) (Coleoptera, Scolytidae): Previous programmes and possibilities for the future. Biocon. News Inf. 1990, 11, 107–117. [Google Scholar]
- Davis, A.P.; Govaerts, R.; Bridson, D.M.; Stoffelen, P. An annotated taxonomic conspectus of the genus Coffea (Rubiaceae). Bot. J. Linn. Soc. 2006, 152, 465–512. [Google Scholar] [CrossRef]
- Davidson, A. The occurrence of coffee berry borer Hypothenemus (Stephanoderis) hampei in Ethiopia. Café 1967, 8, 1–3. [Google Scholar]
- Abebe, M. Further Evidence on the Occurrence of Coffee Berry Borer in Ethiopia. In Proceedings of the 2nd Intercontinental Conference on Coffee Berry Borer, Tapachula, Mexico, 1998; pp. 75–76. [Google Scholar]
- Jaramillo, J.; Chabi-Olaye, A.; Kamonjo, C.; Jaramillo, A.; Vega, F.E.; Poehling, H.; Borgemeister, C. Thermal tolerance of the coffee berry borer Hypothenemus hampei: Predictions of climate change impact on a tropical insect pest. PLoS ONE 2009, 4, e6487. [Google Scholar] [CrossRef] [PubMed]
- Baker, P.S. Some aspects of the behavior of the coffee berry borer in relation to its control in southern Mexico (Coleoptera, Scolytidae). Folia Entomol. Mex. 1984, 61, 9–24. [Google Scholar]
- Garavito, A.; Montagnon, C.; Guyot, R.; Bertrand, B. Identification by the DArTseq method of the genetic origin of the Coffea canephora cultivated in Vietnam and Mexico. BMC Plant Biol. 2016, 16, 242. [Google Scholar] [CrossRef] [PubMed]
- Ceja-Navarro, J.A.; Vega, F.E.; Karaoz, U.; Hao, Z.; Jenkins, S.; Lim, H.C.; Kosina, P.; Infante, F.; Northen, T.R.; Brodie, E.L. Gut microbiota mediate caffeine detoxification in the primary insect pest of coffee. Nat. Commun. 2015, 6, 7618. [Google Scholar] [CrossRef]
- Barrera, J.F. Dynamique des Populations du Scolyte des Fruits du Caféier, Hypothenemus Hampei (Coleoptera: Scolytidae), et Lutte Biologique Avec le Parasitoide Cephalonomia Stephanoderis (Hymenoptera: Bethylidae), au Chiapas, Mexique. Ph.D. Thesis, Université Paul Sabatier Toulouse, Toulouse, France, 1994; p. 301. [Google Scholar]
- Bustillo, A.E.; Cárdenas, R.; Villalba, D.A.; Benavides, P.; Orozco, J.; Posada, F.J. Manejo Integrado de la Broca del Café Hypothenemus Hampei (Ferrari) en Colombia; Centro Nacional de Investigaciones de Café (Cenicafé): Chinchiná, Colombia, 1998; p. 134. [Google Scholar]
- Varón, E.H.; Hanson, P.; Borbón, O.; Carballo, M.; Hilje, L. Potencial de hormigas como depredadoras de la broca del café (Hypothenemus hampei) in Costa Rica. Manejo Integr. Plagas Agroecol. 2004, 73, 42–50. [Google Scholar]
- Costa, J.N.M.; da Silva, R.B.; Ribeiro, P.A.D.; Teixeira, C.A.D. Flutuação Populacional da Broca-Do-Café (Hypothenemus Hampei, Ferrari) no Estado de Rondônia. In Proceedings of the 2nd Simpósio de Pesquisa dos Cafés do Brasil, Vitória, Brazil, 24–27 September 2001; pp. 1944–1950. [Google Scholar]
- Cárdenas, M.R.; Posada Flórez, F.J. Los Insectos y Otros Habitantes de Platanales y Cafetales; Centro Nacional de Investigaciones de Café: Chinchiná, Colombia, 2001; p. 250. [Google Scholar]
- Wrigley, G. Coffee; Longman Scientific & Technical: Essex, UK, 1988; p. 369. [Google Scholar]
- Mendesil, E.; Jembere, B.; Seyoum, E. Population dynamics and distribution of the coffee berry borer, Hypothenemus hampei (Ferrari) (Coleoptera: Scolytidae) on Coffea arabica L. in southwestern Ethiopia. Sinet 2004, 27, 127–134. [Google Scholar]
- Vega, F.E.; Kramer, M.; Jaramillo, J. Increasing coffee berry borer (Coleoptera: Curculionidae: Scolytinae) female density in artificial diet decreases fecundity. J. Econ. Entomol. 2011, 104, 87–93. [Google Scholar] [CrossRef]
- Penagos-Dardón, H.; Flores, J.C. Hábito y tiempo de penetración de la broca del café, Hypothenemus hampei (Ferrari). Rev. Cafe. 1974, 137, 5–15. [Google Scholar]
- Sponagel, K.W. La Broca del Café Hypothenemus Hampei en Plantaciones de Café Robusta en la Amazonía Ecuatoriana: Presencia, Posibilidades de Control y Consideraciones Socio-Económicas del Cultivo en Relación a Sistemas Alternativos de Producción Agropecuaria en la Region; Wissenschaftlicher Fachverlag: Berlin, Germany, 1994; p. 185. [Google Scholar]
- Bergamin, J. Contribuição para o conhecimento da biologia da broca do café “Hypothenemus hampei (Ferrari, 1867)” (Col. Ipidae). Arq. Inst. Biológico São Paulo 1943, 14, 31–72. [Google Scholar]
- Friederichs, K. De Bestrijding van de Koffiebessenboeboek op de Onderneming Karang Redjo. Meded. Koffiebessenboeboek Fonds 1922, 1, 7–21. [Google Scholar]
- Baker, P.S.; Barrera, J.F.; Rivas, A. Life-history studies of the coffee berry borer (Hypothenemus hampei, Scolytidae) on coffee trees in southern Mexico. J. App. Ecol. 1992, 29, 656–662. [Google Scholar] [CrossRef]
- Jaramillo, J.; Chabi-Olaye, A.; Borgemeister, C. Temperature dependent development and emergence pattern of Hypothenemus hampei (Coleoptera: Curculionidae: Scolytinae) from coffee berries. J. Econ. Entomol. 2010, 103, 1159–1165. [Google Scholar] [CrossRef]
- Giraldo-Jaramillo, M.; Garcia, A.G.; Parra, J.R. Biology, thermal requirements, and estimation of the number of generations of Hypothenemus hampei (Ferrari, 1867) (Coleoptera: Curculionidae) in the state of São Paulo. Brazil J. Econ. Entomol. 2018, 111, 2192–2200. [Google Scholar] [CrossRef]
- Hamilton, L.J.; Hollingsworth, R.G.; Sabado-Halpern, M.; Manoukis, N.C.; Follett, P.A.; Johnson, M.A. Coffee berry borer (Hypothenemus hampei) (Coleoptera: Curculionidae) development across an elevational gradient on Hawai’i Island: Applying laboratory degree-day predictions to natural field populations. PLoS ONE 2019, 14, e0218321. [Google Scholar] [CrossRef]
- Brun, L.O.; Stuart, J.; Gaudichon, V.; Aronstein, K.; French-Constant, R.H. Functional haplodiploidy: A mechanism for the spread of insecticide resistance in an important international insect pest. Proc. Natl. Acad. Sci. USA 1995, 92, 9861–9865. [Google Scholar] [CrossRef]
- Ticheler, J.H.G. Étude Analytique de l’Épidémiologie du Scolyte des Graines de Café, Stephanoderes Hampei Ferr., en Côte d’Ivoire; Diss. Veenman: Utrecht, The Netherlands, 1961. [Google Scholar]
- Ticheler, J. Estudio analítico de la epidemiología del escolítido de los granos de café, Stephanoderis hampei Ferr. en Costa de Marfil. Rev. Cenicafé 1963, 14, 223–294. [Google Scholar]
- Breilid, H.; Brun, L.O.; Andreev, D.; French-Constant, R.H.; Kirkendall, L.R. Phylogeographic patterns of introduced populations of the coffee berry borer Hypothenemus hampei (Ferrari) (Coleoptera: Scolytidae) inferred from mitochondrial DNA sequences. In Proceedings of the 17th International Scientific Colloquium on Coffee; Association Scientifique Internationale du Café (ASIC): Nairobi, Kenya, 1997; pp. 653–655. [Google Scholar]
- Andreev, D.; Breilid, H.; Kirkendall, L.; Brun, L.O.; ffrench-Constant, R.H. Lack of nucleotide variability in a beetle pest with extreme inbreeding. Insect Mol. Biol. 1998, 7, 197–200. [Google Scholar] [CrossRef]
- Benavides, P.; Vega, F.E.; Romero-Severson, J.; Bustillo, A.E.; Stuart, J.J. Biodiversity and biogeography of an important inbreed pest of coffee, coffee berry borer (Coleoptera: Curculionidae: Scolytinae). Ann. Entomol. Soc. Am. 2005, 98, 359–366. [Google Scholar] [CrossRef]
- Benavides, P.; Stuart, J.J.; Vega, F.E.; Romero-Severson, J.; Bustillo, A.E.; Navarro, L.; Constantino, L.M.; Acevedo, F.E. Genetic variability of Hypothenemus hampei (Ferrari) in Colombia and development of molecular markers. In Proceedings, 21st International Scientific Colloquium on Coffee; Association Scientifique Internationale du Café (ASIC): Montpellier, France, 2006; pp. 1301–1315. [Google Scholar]
- Benavides Machado, P.; Stuart, J.J.; Vega, F.E.; Romero-Severson, J.; Bustillo, A.E. Biogeografía y aspectos genéticos de la broca del café Hypothenemus hampei. In Proceedings Manejo da Broca-do-Café; Workshop Internacional: Londrina, Brasil, 2007; pp. 11–36. [Google Scholar]
- Chapman, E.G.; Messing, R.H.; Harwood, J.D. Determining the origin of the coffee berry borer invasion of Hawaii. Ann. Entomol. Soc. Am. 2015, 108, 585–592. [Google Scholar] [CrossRef]
- Gauthier, N. Multiple cryptic genetic units in Hypothenemus hampei (Coleoptera: Scolytinae): Evidence from microsatellite and mitochondrial DNA sequence data. Biol. J. Linn. Soc. 2010, 101, 113–129. [Google Scholar] [CrossRef]
- Trujillo, E.; Ferreira, S.; Schmitt, D.; Mitchell, W. Serious economic pests of coffee that may accidentally be introduced to Hawaii. College of Tropical Agriculture and Human Resources University of Hawaii. Res. Ext. Ser. 1995, 156, 21. [Google Scholar]
- International Coffee Organization (ICO). Trade Statistics. 2018. Available online: http://www.ico.org/trade_statistics.asp?section=Statistics (accessed on 13 August 2019).
- Mawardi, S.; Wiryadiputra, S. Recent Status of Coffee Berry Borer in Indonesia. In Proceedings of the International Coffee Organization Seminar on Coffee Berry Borer, London, UK, 17 March 2009. [Google Scholar]
- Byrareddy, V.; Kouadio, L.; Mushtaq, S.; Stone, R. Sustainable production of robusta coffee under a changing climate: A 10-year monitoring of fertilizer management in coffee farms in Vietnam and Indonesia. Agronomy 2019, 9, 499. [Google Scholar] [CrossRef]
- Saragih, J.R. Socioeconomic and ecological dimension of certified and conventional arabica coffee production in North Sumatra, Indonesia. Asian J. Agric. Rural Dev. 2013, 3, 93–107. [Google Scholar]
- Hagedorn, M. Wieder ein neuer Kaffeeschädling. Entomol. Blätter 1910, 6, 1–4. [Google Scholar]
- Corbett, G.H. Some preliminary observations on the coffee berry beetle borer Stephanoderes (Cryphalus) hampei Ferr. Malay. Agric. J. 1933, 21, 8–22. [Google Scholar]
- Corporaal, J.B. De Koffiebesboorder op Sumatra’s Oostkust en Atjeh. Mededeelingen van het Algemeen Proefstation der A.V.R.O.S.; Algèmeene Serie No. 12; 1921; p. 20. [Google Scholar]
- Wiryadiputra, S.; Cilas, C.; Morin, J.-P. Effectiveness of the Brocap Trap in Controlling the Coffee Berry Borer (Hypothenemus Hampei Ferr.) in Indonesia: [PA589]; ASIC: Montpellier, France, 2008.
- Dufour, B.; Frérot, B. Optimization of coffee berry borer, Hypothenemus hampei Ferrari (Col., Scolytidae), mass trapping with an attractant mixture. J. Appl. Entomol. 2008, 132, 591–600. [Google Scholar] [CrossRef]
- Dufour, B.P.; Kerana, I.W.; Ribeyre, F. Effect of coffee tree pruning on berry production and coffee berry borer infestation in the Toba highlands (North Sumatra). Crop Prot. 2019, 122, 151–158. [Google Scholar] [CrossRef]
- Bustillo Pardey, A.E. Una revisión sobre la broca del café, Hypothenemus hampei (Coleoptera: Curculionidae: Scolytinae), en Colombia. Rev. Colomb. Entomol. 2006, 32, 101–116. [Google Scholar]
- Aristizábal, L.F.; Johnson, M.A.; Shriner, S.; Hollingsworth, R.; Manoukis, N.C.; Myers, R.; Bayman, P.; Arthurs, S.P. Integrated pest management of coffee berry borer in Hawaii and Puerto Rico: Current status and prospects. Insects 2017, 8, 123. [Google Scholar]
- Kawabata, A.M.; Nakamoto, S.T.; Curtiss, R.T. Recommendations for Coffee Berry Borer Integrated Pest Management in Hawai’i 2015; Insect Pests-33; College of Tropical Agriculture and Human Resources, University of Hawaii at Manoa: Honolulu, HI, USA, 2016. [Google Scholar]
- Fausto, B. A Concise History of Brazil; Cambridge University Press: Cambridge, UK, 1999; ISBN 978-0-521-56526-4. [Google Scholar]
- Crocitti, J.J.; Vallance, M.M. Brazil Today: An Encyclopedia of Life in the Republic; ABC-CLIO: Santa Barbara, CA, USA, 2011; p. 238. [Google Scholar]
- Bethell, L. Brazil: Empire and Republic, 1822–1930; Cambridge University Press: Cambridge, UK, 1985; p. 85. [Google Scholar]
- Volsi, B.; Telles, T.S.; Caldarelli, C.E.; Camara, M.R.G. The dynamics of coffee production in Brazil. PLoS ONE 2019, 14, e0219742. [Google Scholar] [CrossRef] [PubMed]
- Watson, K.; Achinelli, M.L. Context and contingency: The coffee crisis for conventional small-scale coffee farmers in Brazil. Geogr. J. 2008, 174, 223–234. [Google Scholar] [CrossRef]
- Helfand, S.M.; de Rezende, G.C. The impact of sector-specific and economy-wide policy reforms on the agricultural sector in Brazil: 1980–1998. Contemp. Econ. Policy 2004, 22, 194–212. [Google Scholar] [CrossRef]
- Graham, D.H.; Gauthier, H.; De Barros, J.R.M. Thirty years of agricultural growth in Brazil: Crop performance, regional profile, and recent policy review. Econ. Dev. Cult. Chang. 1987, 36, 1–34. [Google Scholar] [CrossRef]
- Atkins, P.; Bowler, I. Food in Society: Economy, Culture, Geography; Arnold: London, UK, 2001. [Google Scholar]
- Gwynne, R.; Kay, C. Latin America transformed: Globalization and neoliberalism. In Latin America Transformed: Globalization and Modernity, 2nd ed.; Gwynne, R., Kay, C., Eds.; Oxford University Press: New York, NY, USA, 2004; pp. 211–225. [Google Scholar]
- Vakis, R.; Kruger, D.; Mason, A. Shocks and Coffee: Lessons from Nicaragua; The World Bank: Washington, DC, USA, 2004. [Google Scholar]
- Franco, F.S.; Couto, L.; Carvalho, A.F.; Jucksch, I.; Filho, E.I.F.; Silva, E.; Neto, J.A.A.M. Quantificação de erosão em sistemas agroflorais e convencionais na Zona da Mata de Minas Gerais. Rev. Árvore 2002, 26, 751–760. [Google Scholar] [CrossRef]
- Rice, R. A place unbecoming: The coffee farm of northern Latin America. Geogr. Rev. 1999, 89, 554–579. [Google Scholar] [CrossRef]
- Perfecto, I.; Rice, R.A.; Greenburg, R.; VanderVoort, M.E. Shade coffee: A disappearing refuge for biodiversity. Bioscience 1996, 46, 598–608. [Google Scholar] [CrossRef]
- Andrade, R. A Expansão da Cafeicultural em Minas Gerais: Da Intervenção do Estado à Liberalização do Mercado Centro de Desenvolvimento e Planejamento Regional; da Facultade Federal de Minas Gerais Universidade Federal de Minas Gerais: Belo Horizonte, Brazil, 1994. [Google Scholar]
- Berthet, J.J.A. Caruncho do café: Informação prestada pelo Snr; Boletim de Agricultura; Director do Instituto Arthaud Agronômico a Respeito de Amostras de Café Vindas do Congo Belga: São Paulo, Brazil, 1913; Volume 14, pp. 312–313. [Google Scholar]
- Neiva, A. Os Trabalhos da Commissão de Estudo e Debellação da Praga Cafeeira Desde seu Inicio; Publicação No. 21; Commissão de Estudo e Debellação da Praga Cafeeira, 1928; p. 27.
- Da Silva, A.F.C. A Campanha Contra a Broca-do-Café em São Paulo (1924–1927); História, Ciências, Saúde—Manguinhos, 2006; Volume 13, pp. 957–993. [Google Scholar]
- Pamplona, A. Divulgação, Pelo Cinema, dos Methodos de Combate á Broca do Café no Estado de São Paulo; Publicação No. 19; Comissão Para o Estudo e Debellação da Praga Caféeira, 1927; pp. 101–104.
- Hambleton, E.J. Economic entomology in South America. Sci. Mon. 1944, 59, 283–292. [Google Scholar]
- Hempel, A. O combate á broca do café por meio da vespa de Uganda. Bol. Agric. Zootec. Vet. Minas Gerais 1933, 6, 551–555. [Google Scholar]
- Infante, F. Biological Control of Hypothenemus Hampei (Coleoptera. Scolytidae) in Mexico, Using the Parasitoid Prorops Nasuta (Hymenoptera: Bethylidae); No. TE/633.73976 I5; Diss., University of London, Department of Biology: London, UK, 1998; p. 173. [Google Scholar]
- Benavides, P.; Bustillo, A.E.; Portilla, M.; Orozco, J. Classical Biological Control of Coffee Berry Borer, Hypothenemus Hampei (Coleoptera: Scolytidae) in Colombia with African Parasitoids. In Proceedings of the 1st International Symposium on Biological Control of Arthropods, Honolulu, HI, USA, 14–18 January 2002; pp. 430–434. [Google Scholar]
- Sauer, H.F.G.; Duval, G.; Falanghe, O. Combate á broca do café e a possibilidae do emprêgo de inseticidas. Bol. Supt. Serviços Café 1947, 23, 184–192. [Google Scholar]
- Mansingh, A. Limitations of insecticides in the management of the coffee berry borer. J. Coffee Res. 1991, 21, 67–98. [Google Scholar]
- Brun, L.O.; Marcillaud, C.; Gaudichon, V.; Suckling, D.M. Endosulfan resistance in Hypothenemus hampei (Coleoptera: Scolytidae) in New Caledonia. J. Econ. Entomol. 1989, 82, 1311–1316. [Google Scholar] [CrossRef]
- Brun, L.O.; Marcillaud, C.; Gaudichon, V. Etudes sur le Ravageur Majeur du café en Nouvelle Calédonie: Hypothenemus hampei (Coleoptera: Scolytidae) 2ème Annèe; Conventions Sciences de la vie, Zoologie Appliquée No. 4; ORSTOM: Nouméa, New Caledonia, 1990; p. 58. [Google Scholar]
- Lubick, N. Endosulfan’s exit: US EPA pesticide review leads to a ban. Science 2010, 328, 1466. [Google Scholar] [CrossRef]
- Janssen, M.P.M. Endosulfan: A Closer Look at the Arguments against a Worldwide Phase Out; Letter Report 601356002; National Institute for Public Health and the Environment: Ministry of Health, Welfare and Sport: Bilthoven, The Netherlands, 2011; p. 90. [Google Scholar]
- Villalba, D.; Bustillo, A.E.; Chaves, B. Evaluación de insecticidas para el control de la broca del café en Colombia. Cenicafé 1995, 46, 152–163. [Google Scholar]
- Torres, F.Z.V.; de Oliveira Rigitano, R.L.; Torres, L.C. Occurrence of thiamethoxam in coffee (Coffea arabica L.) leaves and fruits following a soil drench application of the insecticide. Coffee Sci. 2010, 5, 148–153. [Google Scholar]
- Arcila Moreno, A.; Duarte Cano, A.F.; Villalba Guott, D.A.; Benavides Machado, P. Nuevo Producto en el Manejo Integrado de la Broca del Café en Colombia; Avances Técnicos Cenicafé No. 437; Cenicafé: Chinchiná, Colombia, 2013; p. 8. [Google Scholar]
- Plata-Rueda, A.; Martínez, L.C.; Costa, N.C.R.; Zanuncio, J.C.; de Sena Fernandes, M.E.; Serrão, J.E.; Guedes, R.N.C.; Fernandes, F.L. Chlorantraniliprole–mediated effects on survival, walking abilities, and respiration in the coffee berry borer, Hypothenemus hampei. Ecotoxicol. Environ. Saf. 2019, 172, 53–58. [Google Scholar] [CrossRef]
- Vijayalakshmi, C.K.; Tintumol, K.; Vinod Kumar, P.K. Effect of few commercial neem-based insecticides in the management of coffee berry borer, Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae). J. Zool. Stud. 2014, 1, 22–25. [Google Scholar]
- Kawabata, A.M.; Nakamoto, S.T.; Miyahara, M.; Curtiss, R.T. Recommendations for Coffee Berry Borer Integrated Pest Management in Hawai’i 2020; Insect Pests-47; College of Tropical Agriculture and Human Resources, University of Hawaii at Manoa: Honolulu, HI, USA, 2020. [Google Scholar]
- Nicholson, M.; Mano, A. Berry Borer Beetle Damages Brazil’s Coffee Crop. Available online: //www.reuters.com/article/us-brazil-coffee-pix/exclusive-berry-borer-beetle-damages-brazils-coffee-crop-idUSKBN1A92JU (accessed on 24 July 2017).
- Oliveira, C.M.; Auad, A.M.; Mendes, S.M.; Frizzas, M.R. Economic impact of exotic insect pests in Brazilian agriculture. J. Appl. Entomol. 2013, 137, 1–15. [Google Scholar] [CrossRef]
- Souza, J.C.; Reis, P.R. Broca do Café: Histórico, Reconhecimento, Biologia, Prejuízos, Monitoramento e Controle; Epamig: Belo Horizonte, Brazil, 1997. [Google Scholar]
- Ferreira, A.J.; Bueno, V.H.P.; Moraes, J.C.; Carvalho, G.A.; de, S. Bueno Filho, J.S. Dinâmica populacional da broca-do-café Hypothenemus hampei (Ferr.) (Coleoptera: Scolytidae) em Lavras, MG. Anais Soc. Entomológica Braz. 2000, 29, 237–244. [Google Scholar] [CrossRef]
- Costa, J.N.M.; da Silva, R.B.; Ribeiro, P.A.D.; Teixeira, C.A.D. Flutuação Populacional da Broca-Do-Café (Hypothenemus Hampei, Ferrari) no Estado de Rondônia. In Proceedings of the 19th Congresso Brasileiro de Entomologia, Manaus, Brazil, 16–17 June 2002. [Google Scholar]
- Oliveira, C.M.; Santos, M.J.; Amabile, R.F.; Frizzas, M.R.; Bartholo, G.F. Coffee berry borer in conilon coffee in the Brazilian Cerrado: An ancient pest in a new environment. Bull. Entomol. Res. 2018, 108, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.A.; Machado, J.L.; Souza, J.C.; Alcântara, E.A.; Carvalho, T.A.F. Flotation population of coffee berry borer Hypothenemus hampei (Ferrari, 1867) (Coleoptera—Scolytidae) in southern state of Minas Gerais-Brasil. In Proceedings, 24th International Scientific Colloquium on Coffee; Association Scientifique Internationale du Café (ASIC): San José, CA, USA, 2013; pp. 969–972. [Google Scholar]
- Mathieu, F.; Brun, L.O.; Frérot, B.; Suckling, D.M.; Frampton, C. Progression in field infestation is linked with trapping of coffee berry borer, Hypothenemus hampei (Col., Scolytidae). J. Appl. Entomol. 1999, 123, 535–540. [Google Scholar] [CrossRef]
- Pereira, A.E.; Vilela, E.F.; Tinoco, R.S.; de Lima, J.O.; Fantine, A.K.; Morais, E.G.; Franca, C.F. Correlation between numbers captured and infestation levels of the coffee berry-borer, Hypothenemus hampei: A preliminary basis for an action threshold using baited traps. Int. J. Pest Manag. 2012, 58, 183–190. [Google Scholar] [CrossRef]
- Mariño, Y.A.; Pérez, M.-E.; Gallardo, F.; Trifilio, M.; Cruz, M.; Bayman, P. Sun vs. shade affects infestation, total population and sex ratio of the coffee berry borer (Hypothenemus hampei) in Puerto Rico. Agric. Ecosyst. Environ. 2016, 222, 258–266. [Google Scholar] [CrossRef]
- Infante, F. Pest management strategies against the coffee berry borer (Coleoptera: Curculionidae: Scolytinae). J. Agric. Food Chem. 2018, 66, 5275–5280. [Google Scholar] [CrossRef]
- Yilma, T.; Olana, G.; Zegeye, T. Some Socio Economic Issue Related to Fungicide Use against CD in Ethiopia. In Proceedings of the Workshop on the Command (CBI) in Ethiopia, Addis Ababa, Ethiopia, 13–14 August 1999; pp. 72–84. [Google Scholar]
- Woldemariam, T.; Denich, M.; Gatzweler, F.; Balcha, G.; Tekatay, D. In Situ Conservation of Genetic Resources of Wild Arabica Coffee in Montane Rain Forest Ethiopia. In Coffee Diversity & Knowledge; Adugna, G., Shimber, B.B.T., Taye, E., Kufa, T., Eds.; EIRO: Addis Ababa, Ethiopia, 2008; pp. 29–343. [Google Scholar]
- FDRE. Comprehensive Coffee Development and Marketing Plan. In Coffee Improvement Programme IV; Federal Democratic Republic of Ethiopia, Ministry of Agriculture and Rural Development: Addis Ababa, Ethiopia, 2003. [Google Scholar]
- Koebler, J. Buzzkill? How Climate Change Could Eventually End Coffee. Available online: http://www.usnews.com/news/articles/2013/03/27/buzzkill-how-climate-change-could-eventually-end-coffee (accessed on 27 March 2013).
- James, W.; Tim, S.; Leulsegged, K. Woreda Level Crop Production Ranking in Ethiopia; International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2015; p. 43. [Google Scholar]
- Tadesse, T.; Tesfaye, B.; Abera, G. Coffee production constraints and opportunities at major growing districts of southern Ethiopia. Cogent Food Agri. 2020, 6, 1741982. [Google Scholar] [CrossRef]
- Crowe, T.J.; Tadesse, G.M.; Tsedeke, A. An Annotated List of Insect Pests of Field Crops in Ethiopia; Institute of Agricultural Research: Addis Ababa, Ethiopia, 1977; p. 71. [Google Scholar]
- Crowe, T.J.; Gebremedhin, T. Coffee Pests in Ethiopia; Their Biology and Control; Institute of Agricultural Research: Addis Ababa, Ethiopia, 1984; p. 44. [Google Scholar]
- Abebe, M. Insect pests of coffee with special emphasis on antestia, Antestiopsis intricata, in Ethiopia. Int. J. Trop. Insect Sci. 1987, 8, 977–980. [Google Scholar] [CrossRef]
- Asfaw, E.; Mendesil, E.; Mohammed, A. Altitude and coffee production systems influence extent of infestation and bean damage by the coffee berry borer. Arch. Phytopathol. Plant Prot. 2019, 52, 170–183. [Google Scholar] [CrossRef]
- Kucel, P.; Kangire, A.; Egonyu, J.P. Status and Current Research Strategies for Management of the Coffee Berry Borer (Hypothenemus Hampei Ferr) in Africa; National Crop Resources Research Institute, 2009. [Google Scholar]
- Trenberth, K.E.; Jones, P.D.; Ambenje, P.; Bojariu, R.; Easterling, D.; Klein Tank, A.; Parker, D.; Rahimzadeh, F.; Renwick, J.A.; Rusticucci, M.; et al. Observations: Surface and atmospheric climate change. In Climate Change 2007: The Physical Science Basis—Contribution of WG 1 to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Solomon, S., Qin, D., Manning, M., Chen, Z., Marquis, M., Averyt, K.B., Tignor, M., Miller, H.L., Eds.; Cambridge University Press: Cambridge, UK, 2007; pp. 235–336. [Google Scholar]
- Kyamanywa, S.; Kucel, P.; Uringi, N.; Kovach, J.; Roberts, A.; Erbaugh, M. Arabica Coffee Pest Profiles in the Mt. Elgon Area of Uganda; Report on Arabica IPM-CRSP Project in Uganda; Virginia Tech: Blacksburg, VA, USA, 2009. [Google Scholar]
- Moat, J.; Williams, J.; Baena, S.; Wilkinson, T.; Gole, T.D.; Challa, Z.K.; Demissew, S.; Davis, A.P. Resilience potential of the Ethiopian coffee sector under climate change. Nat. Plants 2017, 3, 1–14. [Google Scholar] [CrossRef]
- Bosselman, A.S.; Dons, K.; Oberthur, T.; Smith, C.; Raebild, A.; Usma, H. The influence of shade trees on coffee quality in smallholder coffee agroforesty systems in Southern Colombia. Agric. Ecosyst. Environ. 2009, 129, 253–260. [Google Scholar] [CrossRef]
- Larsen, A.; Philpott, S.M. Twig-nesting ants: The hidden predators of the coffee berry borer in Chiapas, Mexico. Biotropica 2010, 42, 342–347. [Google Scholar] [CrossRef]
- Jaramillo, J. Coffee insects in a changing world. In Proceedings 24th International Scientific Colloquium on Coffee; Association Scientifique Internationale du Café (ASIC): San José, CA, USA, 2013; pp. 645–652. [Google Scholar]
- Jonsson, M.; Ijala, A.R.; Ekbom, B.; Kyamanywa, S.; Karungi, J. Contrasting effect of shade levels and altitude on two important coffee pests. J. Pest Sci. 2015, 88, 281–287. [Google Scholar] [CrossRef]
- Teodoro, A.; Klein, A.; Reist, P.; Tscharntke, T. Agroforestry management affects coffee pests contingent on season and developmental stage. Agric. For. Entomol. 2009, 11, 295–300. [Google Scholar] [CrossRef]
- Soto-Pinto, L.; Perfecto, I.; Caballero-Nieto, J. Shade over coffee: Its effects on berry borer, leaf rust and spontaneous herbs in Chiapas, Mexico. Agrofores. Syst. 2002, 55, 37–45. [Google Scholar] [CrossRef]
- Meylan, L.; Gary, C.; Allinne, C.; Ortiz, J.; Jackson, L.; Rapidel, B. Evaluating the effect of shade trees on provision of ecosystem services in intensively managed coffee plantations. Agric. Ecosyst. Environ. 2017, 245, 32–42. [Google Scholar] [CrossRef]
- Johnson, M.A.; Fortna, S.; Manoukis, N.C. Evaluation of Exclusion Netting for Coffee Berry Borer (Hypothenemus hampei) Management. Insects 2020, 11, 364. [Google Scholar] [CrossRef]
- Saldaña, Jorge. El Café en Puerto Rico; Real Hnos. Inc.: San Juan, PR, USA, 1935. [Google Scholar]
- Bergad, L.W. Agrarian history of Puerto Rico, 1870–1930. Lat. Am. Res. Rev. 1978, 13, 63–94. [Google Scholar]
- Dietz, J.L. Economic History of Puerto Rico: Institutional Change and Capitalist Development; Princeton University Press: Princeton, NJ, USA, 1986. [Google Scholar]
- Mariño, Y.A.; Verle Rodrigues, J.C.; Bayman, P. Wolbachia affects reproduction and population dynamics of the coffee berry borer (Hypothenemus hampei): Implications for biological control. Insects 2017, 8, 8. [Google Scholar] [CrossRef]
- Flores, C.A. Informe Empresa de Cafe 2011–2012; Universidad de Puerto Rico, Recinto Univesitario de Mayaguez, Colegio de Ciencias Agrícolas. Estación Experimental Agrícola Río Piedras: Rio Piedras, Puerto Rico, 2011; p. 29. [Google Scholar]
- Borkhataria, R.; Collazo, J.A.; Groom, M.J.; Jordan-Garcia, A. Shade-grown coffee in Puerto Rico: Opportunities to preserve biodiversity while reinvigorating a struggling agricultural commodity. Agric. Ecosyst. Environ. 2012, 164–170. [Google Scholar] [CrossRef]
- Vicente-Chandler, J. Intensive Coffee Culture in Puerto Rico; University of Puerto Rico, Mayaguez Campus, Agricultural Experiment Station: Rio Piedras, Puerto Rico, 1968; p. 84. [Google Scholar]
- Rodriguez, R.; Monroig, M. Coffee leaf rust in Puerto Rico. Phytopathology 1991, 81, 699. [Google Scholar]
- Gallardo, F.; Hernández, E.; Daza, M.; Pagán, J. Crianza Masiva Mirax Insularis Muesebeck, el Parasitoide Exótico del Minador del café Leucoptera Coffeella Guérin-Ménéville (Lepidoptera: Lyonetíidae) en Puerto Rico. In Proceedings of the Symposium: The Caribbean Food Crops Society, Miami, FL, USA, 13–17 July 2008; pp. 554–562. [Google Scholar]
- James, T.Y.; Marino, J.A.; Perfecto, I.; Vandermeer, J. Identification of putative coffee rust mycoparasites using single molecule DNA sequencing of infected pustules. Appl. Environ. Microbiol. 2015, 82, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Álamo, C.; Verle Rodrigues, J.C.; Rámos, J.E.; Baiges, S.; Bengoa, K.; Fernandez, D.; Lugo, V.; Sepulveda, R.; Cordero, M. Improving Quality of Coffee of Puerto Rico and the Impact of Irma and Maria Hurricanes on Coffee Production and Its Potential Effect on Education in the Control of CBB. In Proceedings of the Coffee Berry Borer Conference, Kona, HI, USA, 25 April 2018. [Google Scholar]
- Census of Agriculture: Puerto Rico Island and Municipio Data; Geographic Area Series, Part 52; USDA National Agricultural Statistics Service: Washington, DC, USA, 2007; Volume 1. Available online: https://www.nass.usda.gov/Publications/AgCensus/2007/Full_Report/Outlying_Areas/prv1.pdf (accessed on 23 October 2019).
- Nolla, J.A.B. Annual Report for the Fiscal Year 1942–1943; University of Puerto Rico, Agricultural Experiment Station: San Juan, PR, USA, 1944. [Google Scholar]
- Roque, A. Annual Report for the Fiscal Year 1943–1944; University of Puerto Rico, Agricultural Experiment Station: San Juan, PR, USA, 1946. [Google Scholar]
- Wolcott, G.N. The Insects of Puerto Rico. J. Agric. Univ. Puerto Rico 1948, 32, 417–748. [Google Scholar] [CrossRef]
- Vega, F.E.; Franqui, R.A.; Benavides, P. The presence of the coffee berry borer, Hypothenemus hampei, in Puerto Rico: Fact or fiction? J. Insect Sci. 2002, 2, 1–3. [Google Scholar] [CrossRef]
- Diario Oficial. Norma Oficial Mexicana NOM-019-FITO- 1995, por la que se Establece la Cuarentena Exterior Para Prevenir la Introducción de Plagas del Café. Diario Oficial, Primera Sección, Martes 10 de Diciembre de. 1996. Available online: http://ns1.oirsa.org.sv/DI07/Di0703/Di070301/ Di07030102/NORMA19.htm (accessed on 20 October 2019).
- Detection of the Coffee Berry Borer, Hypothenemus Hampei, in Puerto Rico—United States. Official Pest Reports. North American Plant Protection Organization. 2007. Available online: http://www.pestalert.org/oprDetail. cfm?oprID=281 (accessed on 9 October 2019).
- Department of Consumer Affairs (DACO). Orden De Descuento De Peso Por Café Brocado 2009-07. 2009. Available online: https://www.daco.pr.gov/wp-content/uploads/2018/11/Orden-2009-07-Orden-Descuento-Peso-Por-Cafe-Brocado.pdf (accessed on 9 October 2019).
- Census of Agriculture: Specialty Crops Puerto Rico; Subject Series, Part 9; USDA National Agricultural Statistics Service: Washington, DC, USA, 2012; Volume 2. Available online: https://www.nass.usda.gov/Publications/AgCensus/2012/Full_Report/Outlying_Areas/prv1.pdf (accessed on 23 October 2019).
- Vega, F.E.; Davis, A.P.; Jaramillo, J. From forest to plantation? Obscure articles reveal alternative host plants for the coffee berry borer, Hypothenemus hampei (Coleoptera: Curculionidae). Biol. J. Linn. Soc. 2012, 107, 86–94. [Google Scholar] [CrossRef]
- Vega, V.J.; Mariño, Y.A.; Deynes, D.; Greco, E.B.; Bright, D.E.; Bayman, P. A Beetle in a Haystack: Are There Alternate Hosts of the Coffee Berry Borer (Hypothenemus hampei) in Puerto Rico? Agronomy 2020, 10, 228. [Google Scholar] [CrossRef]
- Gallardo, F.; González, O. Manejo Integrado de la Broca del Café en Puerto Rico; Guía Técnica; Departamento de Agricultura: San Juan, PR, USA, 2015; p. 12. [Google Scholar]
- Mariño, Y.A.; García-Peña, J.M.; Vega, V.J.; Verle-Rodrigues, J.C.; Bayman, P. After the fall: Did coffee plants in Puerto Rico survive the 2017 hurricanes? Agric. Ecosyst. Environ. 2018, 266, 10–16. [Google Scholar] [CrossRef]
- Seneviratne, S.; Nicholls, N.; Easterling, D.; Goodess, C.; Kanae, S.; Kossin, J.; Luo, Y.; Marengo, J.; McInnes, K.; Rahimi, M.; et al. Changes in climate extremes and their impacts on the natural physical environment. In Managing the Risks of Extreme Events and Disasters to Advance Climate Change Adaptation; A Special Report of Working Groups I and II of the Intergovernmental Panel on Climate Change (IPCC); Field, C.B., Barros, V., Stocker, T.F., Qin, D., Dokken, D.J., Ebi, K.L., Mastrandrea, M.D., Mach, K.J., Plattner, G.-K., Allen, S.K., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2012; pp. 109–230. [Google Scholar]
- Hatfield, J.; Lead, M.H.; Swanston, C.; Lead, N.F.S.H.; Janowiak, M.; Hub, N.F.S.; Steele, R.F.; Hub, M.; Cole, A.P.H.I.S.; Sharon Hestvik, R.M.A.; et al. USDA Midwest and Northern Forests Regional Climate Hub: Assessment of Climate Change Vulnerability and Adaptation and Mitigation Strategies; U.S. Department of Agriculture: Washington, DC, USA, 2015; p. 55.
- Gowda, P.; Steiner, J.L.; Olson, C.; Boggess, M.; Farrigan, T.; Grusak, M.A.; Reidmiller, D.R.; Avery, C.W.; Easterling, D.R.; Kunkel, K.E.; et al. Agriculture and Rural Com-munities. In Impacts, Risks, and Adaptation in the United States: Fourth National Climate Assessment; Reidmiller, D.R., Avery, C.W., Easterling, D.R., Kunkel, K.E., Lewis, K.L.M., Maycock, T.K., Stewart, B.C., Eds.; U.S. Global Change Research Program: Washington, DC, USA, 2018; Volume II, pp. 391–437. [Google Scholar] [CrossRef]
- Mariño, Y.A.; Ospina, O.E.; Verle Rodrigues, J.C.; Bayman, P. High diversity and variability in the bacterial microbiota of the coffee berry borer (Coleoptera: Curculionidae), with emphasis on Wolbachia. J. Appl. Microbiol. 2018, 125, 528–543. [Google Scholar] [CrossRef]
- USDA National Agricultural Statistics Service, in Cooperation with the Hawaii Department of Agriculture. Pacific Region—Hawaii Coffee Marketings Final Season Estimates. Available online: https://www.nass.usda.gov/Statistics_by_State/Hawaii/Publications/Fruits_and_Nuts/201807FinalCoffee.pdf (accessed on 26 July 2018).
- Kinro, G. A Cup of Aloha: The Kona Coffee Epic; University of Hawaii Press: Honolulu, HI, USA, 2003. [Google Scholar]
- Bittenbender, H.C.; Smith, V.E. Growing Coffee in Hawaii; College of Tropical Agriculture and Human Resources, University of Hawai’i: Honolulu, HI, USA, 2008. [Google Scholar]
- Elevitch, C.; Idol, T.; Friday, J.B.; Lepczyk, C.; Easton Smith, V.; Nelson, S.C. Shade-Grown Coffee for Hawaii: Results of a Twelve Farm Study in Kona. Permanent Agriculture Resources: Holualoa, HI, USA; Available online: http://agroforesty.net/caf (accessed on 5 December 2019).
- Burbano, E.; Wright, M.; Bright, D.E.; Vega, F.E. New record for the coffee berry borer, Hypothenemus hampei, in Hawaii. J. Insect Sci. 2011, 11, 117. [Google Scholar] [CrossRef]
- Johnson, M.A.; Manoukis, N.C. Abundance of coffee berry borer in feral, abandoned, and managed coffee on Hawai’i Island. J. Appl. Entomol. 2020, 00, 1–9. [Google Scholar]
- USDA. Proposed Rule for Movement of Unroasted Coffee into Hawaii and Puerto Rico. Marketing and Regulatory Programs, Animal and Plant Health Inspection Service; Environmental Assessment; USDA: Washington, DC, USA, 2005.
- Leung, P.S.; Kawabata, A.M.; Nakamoto, S.T. Estimated economywide impact of CBB for the crop years 2011/12 and 2012/13; Brief Report at Request of Hawaii Congressional Delegation; 2014; p. 2. [Google Scholar]
- Hicks, S. CBB in Kona as Experienced by Greenwell Farms, Inc. In 2016 Coffee Berry Borer Summit and Conference; Kawabata, A.M., Nakamoto, S.T., Curtiss, R.T., Eds.; Greenwell Farms, Inc.: Kona, HI, USA, 2016. [Google Scholar]
- Greenwell Farms, Inc. The Impact of CBB Infestation in Kona, Hawaii: A Market Perception; Presentation Given by S. Hicks to the Hawaii Coffee Association Annual Meeting; Greenwell Farms, Inc.: Kona, HI, USA, 2013. [Google Scholar]
- Greenwell Farms, Inc. The Impact of CBB in the Flavor of Kona Coffee; Presentation Given by S. Hicks to the Hawaii Coffee Association Annual Meeting; Greenwell Farms, Inc., 2015. [Google Scholar]
- Holzapfel, B.; Bowmen, A. Improving coffee-based production systems for smallholder farmers in Papua New Guinea: A Scoping Study; ACIAR Report HORT/2015/023; Department of Foreign Affairs and Trade, 2015. [Google Scholar]
- CIC. PNG Coffee Industry under Threat of CBB; Coffee Industry Corporation Quarterly Newsletter; CIC: Goroka, Papua New Guinea, 2017; pp. 4–5. [Google Scholar]
- CIC. The Coffee Industry Corporation Strategic Plan to Support the Papua New Guinea Coffee Industry 2008–2018; Coffee Industry Corporation, Eastern Highlands Province, Papua New Guinea; CIC: Goroka, Papua New Guinea, 2008; p. 40. [Google Scholar]
- CIC. CBB Fight Persists; Coffee Industry Corporation Quarterly Newsletter; CIC: Goroka, Papua New Guinea, 2017; pp. 3–4. [Google Scholar]
- CIC. Biocontrol for CBB Underway; Coffee Industry Corporation Quarterly Newsletter; CIC: Goroka, Papua New Guinea, 2018; pp. 5–9. [Google Scholar]
- CIC. CBB Biocontrol under Field Trial; Coffee Industry Corporation Quarterly Newsletter; CIC: Goroka, Papua New Guinea, 2018; pp. 2–3. [Google Scholar]
- World Bank Group. The Fruit of Her Labor: Promoting Gender-Equitable Agribusiness in Papua New Guinea; World Bank: Washington, DC, USA, 2014. [Google Scholar]
- Aristizábal, L.F.; Jiménez, M.; Bustillo, A.E.; Trujillo, H.I.; Arthurs, S.P. Monitoring coffee berry borer, Hypothenemus hampei (Coleoptera: Curculionidae), populations with alcohol-baited funnel traps in coffee farms in Colombia. Fla. Entomol. 2015, 98, 381–383. [Google Scholar] [CrossRef]
- Mendesil, E.; Bruce, T.J.; Woodcock, C.M.; Caulfield, J.C.; Seyoum, E.; Pickett, J.A. Semiochemicals used in host location by the coffee berry borer, Hypothenemus hampei. J. Chem. Ecol. 2009, 35, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Messing, R.H. The coffee berry borer (Hypothenemus hampei) invades Hawaii: Preliminary investigations on trap response and alternate hosts. Insects 2012, 3, 640–652. [Google Scholar] [CrossRef] [PubMed]
- Aristizábal, L.F.; Shriner, S.; Hollingsworth, R.; Arthurs, S. Flight activity and field infestation relationships for coffee berry borer in commercial coffee plantations in Kona and Kau districts, Hawaii. J. Econ. Entomol. 2017, 110, 2421–2427. [Google Scholar] [CrossRef]
- Fernandes, F.L.; Picanco, M.C.; Campos, S.O.; Bastos, C.S.; Chediak, M.; Guedes, R.N.C.; da Silva, R.S. Economic injury level for the coffee berry borer (Coleoptera: Curculionidae: Scolytinae) using attractive traps in Brazilian coffee fields. J. Econ. Entomol. 2011, 104, 1909–1917. [Google Scholar] [CrossRef]
- Centro Nacional de Investigaciones de Café (CENICAFÉ). Como Determinar la Infestación de Broca en un Cafetal; Cenicafé: Bogotá, Colombia, 1993. [Google Scholar]
- Aristizábal, L.F.; Bustillo, A.E.; Arthurs, S.P. Integrated pest management of coffee berry borer: Strategies from Latin America that could be useful for coffee farmers in Hawaii. Insects 2016, 7, 6. [Google Scholar] [CrossRef]
- Johnson, M.A.; Hollingsworth, R.; Fortna, S.; Aristizábal, L.F.; Manoukis, N.C. The Hawaii protocol for scientific monitoring of coffee berry borer: A model for coffee agroecosystems worldwide. J. Vis. Exp. 2018, 133, e57204. [Google Scholar] [CrossRef]
- Kuno, E. A new method of sequential sampling to classify populations relative to a critical density. Res. Pop. Ecol. 1969, 11, 127–136. [Google Scholar] [CrossRef]
- Green, R.H. On fixed precision level sequential sampling. Res. Pop. Ecol. 1970, 12, 249–251. [Google Scholar] [CrossRef]
- Aristizábal, L.F.; Shriner, S.; Hollingsworth, R.; Mascarin, G.M.; Chaves, B.; Matsumoto, T.; Arthurs, S.P. Field sampling strategies for coffee berry borer (Coleoptera: Curculionidae: Scolytinae) infesting berries in coffee farms in Hawaii. Int. J. Trop. Insect Sci. 2018, 38, 418–426. [Google Scholar] [CrossRef]
- Pulakkatu-thodi, I.; Gutierrez-Coarite, R.; Wright, M.G. Dispersion and optimization of sequential sampling plans for coffee berry borer (Coleoptera: Curculionidae) infestations in Hawaii. Env. Entomol. 2018, 47, 1306–1313. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.A.; Fortna, S.; Hollingsworth, R.G.; Manoukis, N.C. Postharvest population reservoirs of coffee berry borer (Coleoptera: Curculionidae) on Hawaii Island. J. Econ. Entomol. 2019, 112, 2833–2841. [Google Scholar] [CrossRef] [PubMed]
- Aristizábal-Aristizábal, L.F. Controlling the Coffee Berry Borer through Integrated Pest Management: A Practical Manual for Coffee Growers & Field Workers in Hawaii; Kailua-Kona, HI, USA, 2018; p. 79. [Google Scholar]
- Bustillo Pardey, A.E. El Manejo de Cafetales y su Relación con el Control de la Broca del café en Colombia; Boletín Técnico Cenicafé No. 24; Cenicafé: Chinchiná, Colombia, 2002; p. 40. [Google Scholar]
- Hollingsworth, R.G.; Jang, E.B.; Follett, P.A. Freezing as a treatment to prevent the spread of Hypothenemus hampei (Coleoptera: Curculionidae), in coffee. J. Econ. Entomol. 2013, 106, 653–660. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Matsumoto, T.K.; Lopez, J. Coffee harvest management by manipulation of coffee flowering with plant growth regulators. In XXIX International Horticultural Congress on Horticulture: Sustaining Lives, Livelihoods and Landscapes (IHC2014); Acta Horticulturae: Brisbane, Australia, 2014; Volume 1130, pp. 219–224. [Google Scholar]
- Zimmermann, G. Review on safety of the entomopathogenic fungi Beauveria bassiana and Beauveria brongniartii. Biocontrol Sci. Technol. 2007, 17, 553–596. [Google Scholar] [CrossRef]
- Jaronski, S.T. Ecological factors in the inundative use of fungal entomopathogens. BioControl 2010, 55, 159–185. [Google Scholar] [CrossRef]
- Wraight, S.P.; Galaini-Wraight, S.; Howes, R.L.; Castrillo, L.A.; Carruthers, R.I.; Smith, R.H.; Matsumoto, T.K.; Keith, L.M. Prevalence of naturally-occurring strains of Beauveria bassiana in populations of coffee berry borer Hypothenemus hampei on Hawaii Island, with observations on coffee plant-H. hampei-B. bassiana interactions. J. Invertebr. Pathol. 2018, 156, 54–72. [Google Scholar] [CrossRef] [PubMed]
- Greco, E.B.; Wright, M.G.; Burgueño, J.; Jaronski, S.T. Efficacy of Beauveria bassiana applications on coffee berry borer across an elevation gradient in Hawaii. Bioncontrol Sci. Technol. 2018, 28, 995–1013. [Google Scholar] [CrossRef]
- Hollingsworth, R.G.; Aristizábal, L.F.; Shriner, S.; Mascarin, G.M.; Moral, R.D.A.; Arthurs, S.P. Incorporating Beauveria bassiana into an integrated pest management plan for Coffee Berry Borer in Hawaii. Front. Sustain. Food Syst. 2020, 4, 22. [Google Scholar] [CrossRef]
- Woodill, A.J.; Nakamoto, S.T.; Kawabata, A.M.; Leung, P. To spray or not to spray: A decision analysis of coffee berry borer in Hawaii. Insects 2017, 8, 116. [Google Scholar] [CrossRef]
- Sim, S.B.; Yoneishi, N.M.; Brill, E.; Geib, S.M.; Follett, P.A. Molecular markers detect cryptic predation on coffee berry borer (Coleoptera: Curculionidae) by silvanid and laemophloeid flat bark beetles (Coleoptera: Silvanidae, Laemophloeidae) in coffee beans. J. Econ. Entomol. 2015, 109, 100–105. [Google Scholar] [CrossRef]
- Follett, P.A.; Kawabata, A.; Nelson, R.; Asmus, G.; Burt, J.; Goschke, K.; Ewing, C.; Gaertner, J.; Brill, E.; Geib, S. Predation by flat bark beetles (Coleoptera: Silvanidae and Laemophloeidae) on coffee berry borer (Coleoptera: Curculionidae) in Hawaii coffee. Biol. Control 2016, 101, 152–158. [Google Scholar] [CrossRef]
- Kawabata, A.; Follett, P.; Wright, M.; Brill, E.; Curtiss, R. An introduction to the Square-Necked Grain Beetle as a Predator of Coffee Berry Borer in Hawaii; Insect Pests-40; College of Tropical Agriculture and Human Resources, University of Hawaii at Manoa: Honolulu, HI, USA, 2016. [Google Scholar]
- Lara, G.J.C.; Lopez, N.J.C.; Bustillo, P.A.E. Efecto de entomonématodos sobre poblaciónes de la broca del café, Hypothenemus hampei (Coleoptera: Scolytidae), en frutos en el suelo. Rev. Colomb. Entomol. 2004, 30, 179–185. [Google Scholar]
- Allard, G.B.; Moore, D. Heterorhabditis sp. nematodes as control agents for coffee berry borer, Hypothenemus hampei (Scolytidae). J. Invertebr. Pathol. 1989, 54, 45–48. [Google Scholar] [CrossRef]
- Castillo, A.; Marbán-Mendoza, N. Evaluación en laboratorio de nematodos Steinernematidos y Heterohabditidos para el control biológico de la broca del café, Hypothenemus hampei Ferr. Nematropica 1996, 26, 101–109. [Google Scholar]
- Molina, J.P.; Lopez, J.C. Desplazamiento y parasitismo de los entomonematodos Steinernema feltiae (Rhabditida: Steinernematidae) y Heterorhabditis bacteriophora (Rhabditida: Heterorhabditidae) hacia frutos infestados con la broca del café Hypothenemus hampei (Coleoptera: Scolytidae). Rev. Colomb. Entomol. 2002, 28, 145–151. [Google Scholar]
- Manton, J.L.; Hollingsworth, R.G.; Cabos, R.Y.M. Potential of Steinernema carpocapsae (Rhabditida: Steinernematidae) against Hypothenemus hampei (Coleoptera: Curculionidae) in Hawaii. Fla. Entomol. 2012, 95, 1194–1197. [Google Scholar] [CrossRef]
- Arthurs, S.; Heinz, K.M.; Prasifka, J.R. An analysis of using entomopathogenic nematodes against above-ground pests. Bull. Entomol. Res. 2004, 94, 297–306. [Google Scholar] [CrossRef]
- Pérez-Lachaud, G.; Hardy, I.C.W.; Lachaud, J.P. Insect gladiators: Competitive interactions between three species of bethylid wasps attacking the coffee berry borer, Hypothenemus hampei (Coleoptera: Scolytidae). Biol. Control 2002, 25, 231–238. [Google Scholar] [CrossRef]
- Infante, F.; Mumford, J.; Baker, P. Life history studies of Prorops nasuta, a parasitoid of the coffee berry borer. BioControl 2005, 50, 259–270. [Google Scholar] [CrossRef]
- Espinoza, J.C.; Infante, F.; Castillo, A.; Pérez, J.; Nieto, G.; Pinson, E.P.; Vega, F.E. The biology of Phymastichus coffea LaSalle (Hymenoptera: Eulophidae) under field conditions. Biol. Control 2009, 49, 227–233. [Google Scholar] [CrossRef]
- Jaramillo, J.; Borgemeister, C.; Baker, P.S. Coffee berry borer Hypothenemus hampei (Coleoptera: Curculionidae): Searching for sustainable control strategies. Bull. Entomol. Res. 2006, 96, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, A. Biosecurity after the Event: Risk Politics and Animal Disease. Environ. Plan A 2008, 40, 1552–1567. [Google Scholar] [CrossRef]
- Hewitt, C.L.; Campbell, M.L. Mechanisms for the prevention of marine bioinvasions for better biosecurity. Mar. Pollut. Bull. Mar. Bioinvasions A Collect. Rev. 2007, 55, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Waage, J.K.; Mumford, J.D. Agricultural biosecurity. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 863–876. [Google Scholar] [CrossRef] [PubMed]
- Lakoff, A.; Collier, S.J. Biosecurity Interventions: Global Health and Security in Question; Columbia University Press: New York City, NY, USA, 2008. [Google Scholar]
- Meyerson, L.A.; Reaser, J.K. Bioinvasions, bioterrorism, and biosecurity. Front. Ecol. Environ. 2003, 1, 307–314. [Google Scholar] [CrossRef]
- New Zealand Biosecurity Council. The Biosecurity Strategy for New Zealand; New Zealand Biosecurity Council: Wellington, New Zealand, 2003. Available online: https://www.maf.govt.nz/biosecurity-strategy (accessed on 15 August 2019).
- Simberloff, D.; Martin, J.-L.; Genovesi, P.; Maris, V.; Wardle, D.A.; Aronson, J.; Courchamp, F.; Galil, B.; García-Berthou, E.; Pascal, M.; et al. Impacts of biological invasions: what’s what and the way forward. Trends Ecol. Evol. 2013, 28, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, D.; McNair, S.; Janecka, J.; Wightman, J.; Simmonds, C.; O’Connell, C.; Wong, E.; Russel, L.; Zern, J.; Aquino, T.; et al. Economic and environmental threats of alien plant, animal, and microbe invasions. Agric. Ecosyst. Environ. 2001, 84, 1–20. [Google Scholar] [CrossRef]
- Davis, A.P.; Woldemariam Gole, T.; Baena, S.; Moat, J. The Impact of Climate Change on Indigenous Arabica Coffee (Coffea arabica): Predicting Future Trends and Identifying Priorities. PLoS ONE 2012, 7, e47981. [Google Scholar] [CrossRef] [PubMed]
- Paramaguru, K. Coffee Under Threat: How Wild Arabica Could Go Extinct. TIME. Available online: http://newsfeed.time.com/2012/11/16/coffee-under-threat-how wild-arabica-could-go-extinct/ (accessed on 16 November 2012).
- Serrato-Diaz, L.M.; Mariño, Y.A.; Bayman, P. Pathogens causing Anthracnose and fruit rots of coffee associated with the coffee berry borer and the entomopathogenic fungus Beauveria bassiana in Puerto Rico. Phytopathology 2020, 110, 1541–1552. [Google Scholar] [CrossRef]
- Follett, P.A. Irradiation for Quarantine Control of Coffee Berry Borer, Hypothenemus hampei (Coleoptera: Curculionidae: Scolytinae) in Coffee and a Proposed Generic Dose for Snout Beetles (Coleoptera: Curculionoidea). J. Econ. Entomol. 2018, 111, 1633–1637. [Google Scholar] [CrossRef]
- Kiran, R.; Shenoy, K.B.; Venkatesha, M.G. Effect of gamma radiation as a post-harvest disinfestation treatment against life stages of the coffee berry borer, Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae). Int. J. Radiat. Biol. 2019, 95, 1301–1308. [Google Scholar] [CrossRef] [PubMed]
- Langley, E.; Hannan, T. Biosecurity New Zealand, Ministry for Primary Industries; Colmar Brunton, a Kantar Millward Brown Company: Wellington, New Zealand, 2018; pp. 2–50. [Google Scholar]
- Walker, H.E.; Lehman, K.A.; Wall, M.; Siderhurst, M.S. Analysis of volatile profiles of green Hawaiian coffee beans damaged by the coffee berry borer (Hypothenemus hampei). J. Sci. Food Agric. 2019, 99, 1954–1960. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, M.A.; Ruiz-Diaz, C.P.; Manoukis, N.C.; Verle Rodrigues, J.C. Coffee Berry Borer (Hypothenemus hampei), a Global Pest of Coffee: Perspectives from Historical and Recent Invasions, and Future Priorities. Insects 2020, 11, 882. https://doi.org/10.3390/insects11120882
Johnson MA, Ruiz-Diaz CP, Manoukis NC, Verle Rodrigues JC. Coffee Berry Borer (Hypothenemus hampei), a Global Pest of Coffee: Perspectives from Historical and Recent Invasions, and Future Priorities. Insects. 2020; 11(12):882. https://doi.org/10.3390/insects11120882
Chicago/Turabian StyleJohnson, Melissa A., Claudia Patricia Ruiz-Diaz, Nicholas C. Manoukis, and Jose Carlos Verle Rodrigues. 2020. "Coffee Berry Borer (Hypothenemus hampei), a Global Pest of Coffee: Perspectives from Historical and Recent Invasions, and Future Priorities" Insects 11, no. 12: 882. https://doi.org/10.3390/insects11120882
APA StyleJohnson, M. A., Ruiz-Diaz, C. P., Manoukis, N. C., & Verle Rodrigues, J. C. (2020). Coffee Berry Borer (Hypothenemus hampei), a Global Pest of Coffee: Perspectives from Historical and Recent Invasions, and Future Priorities. Insects, 11(12), 882. https://doi.org/10.3390/insects11120882





