Biological Control Potential and Drawbacks of Three Zoophytophagous Mirid Predators against Bemisia tabaci in the United States
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Plants and Colonies
2.2. Evaluation of Established and Native Mirids
2.3. Effect of Adding Sesame on Mirid Population Growth and Damage
2.4. Statistical Analyses
3. Results
3.1. Evaluation of Established and Native Mirids
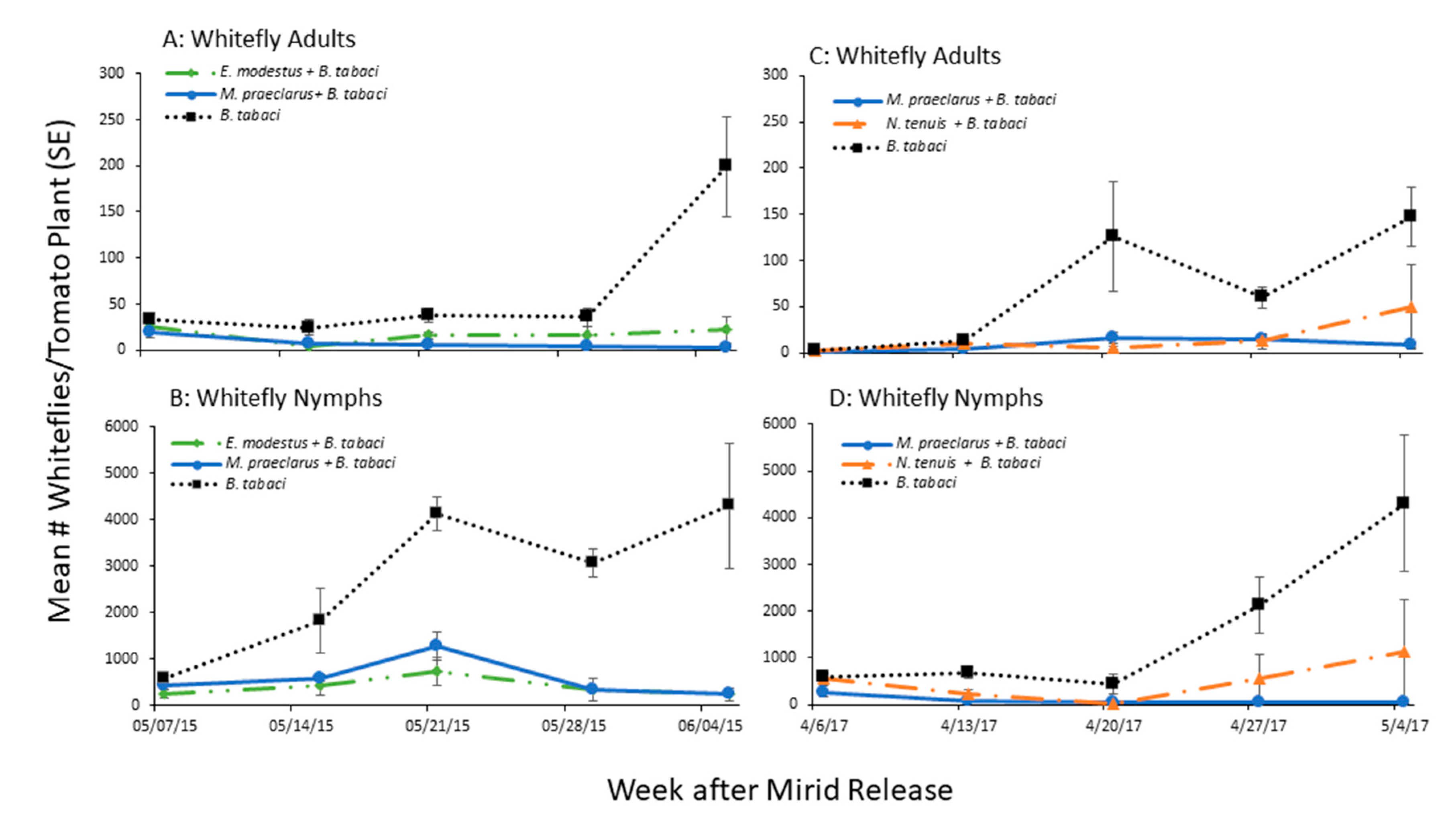
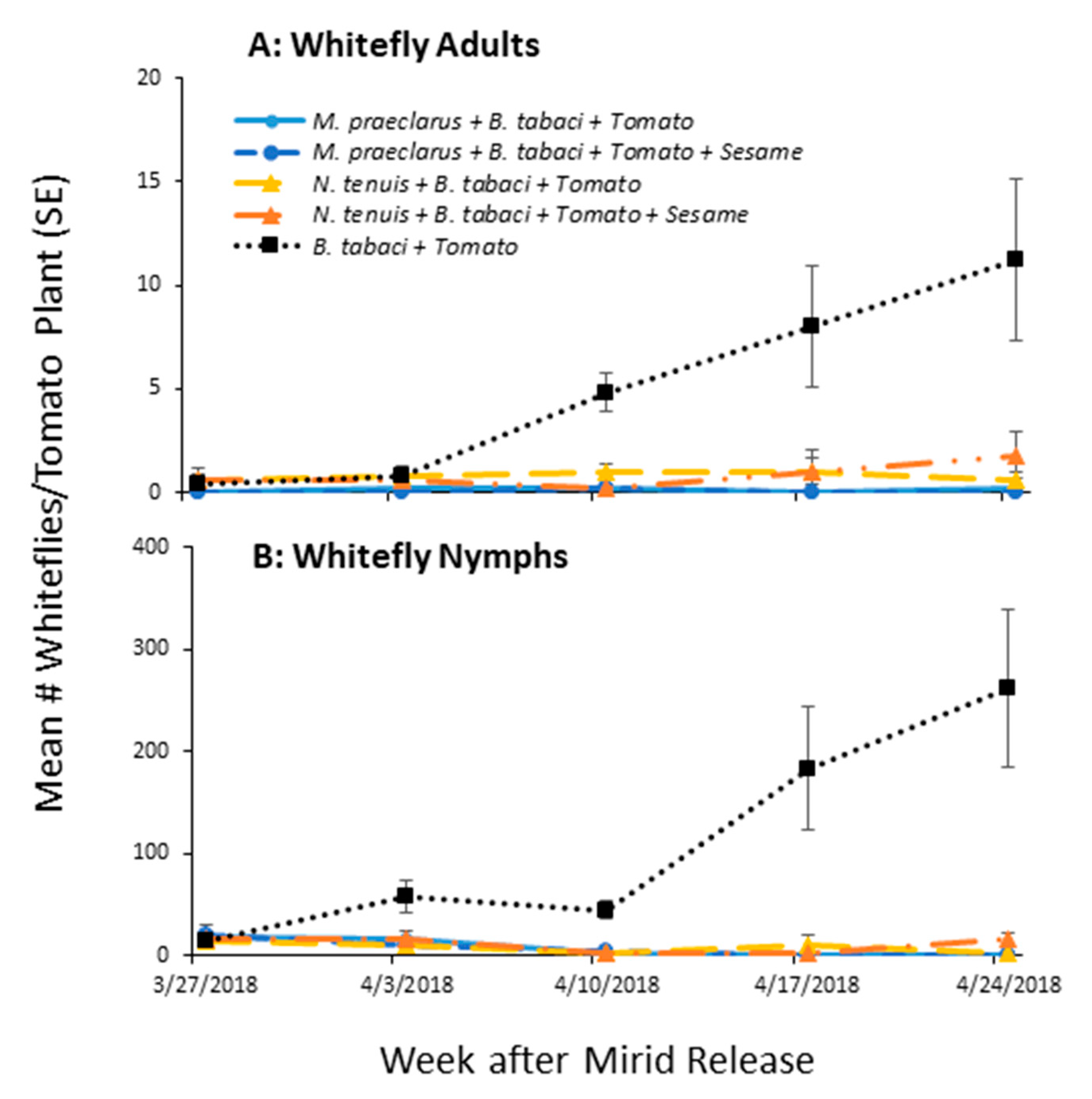
3.1.1. Whitefly Control by Established Mirids
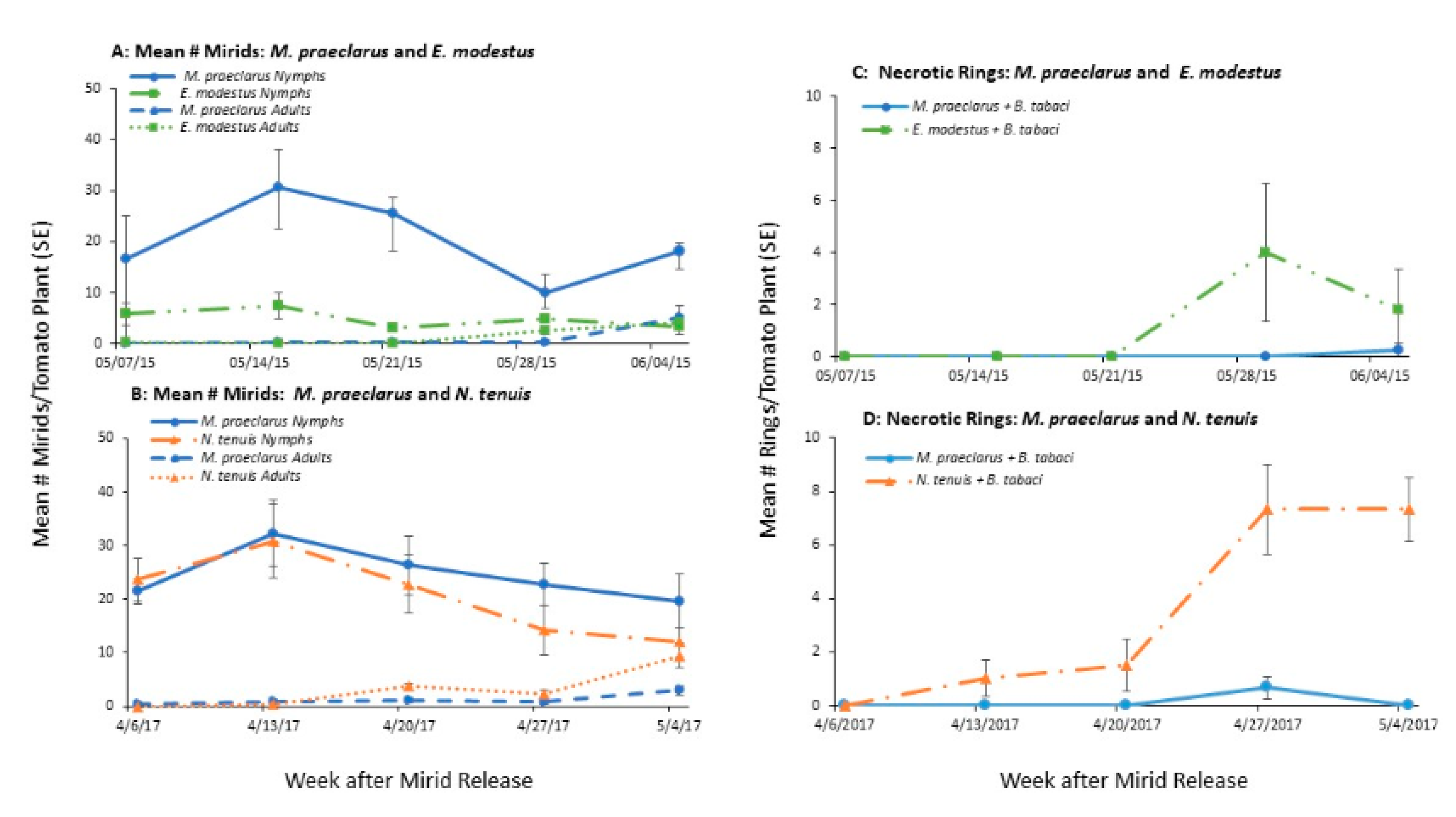
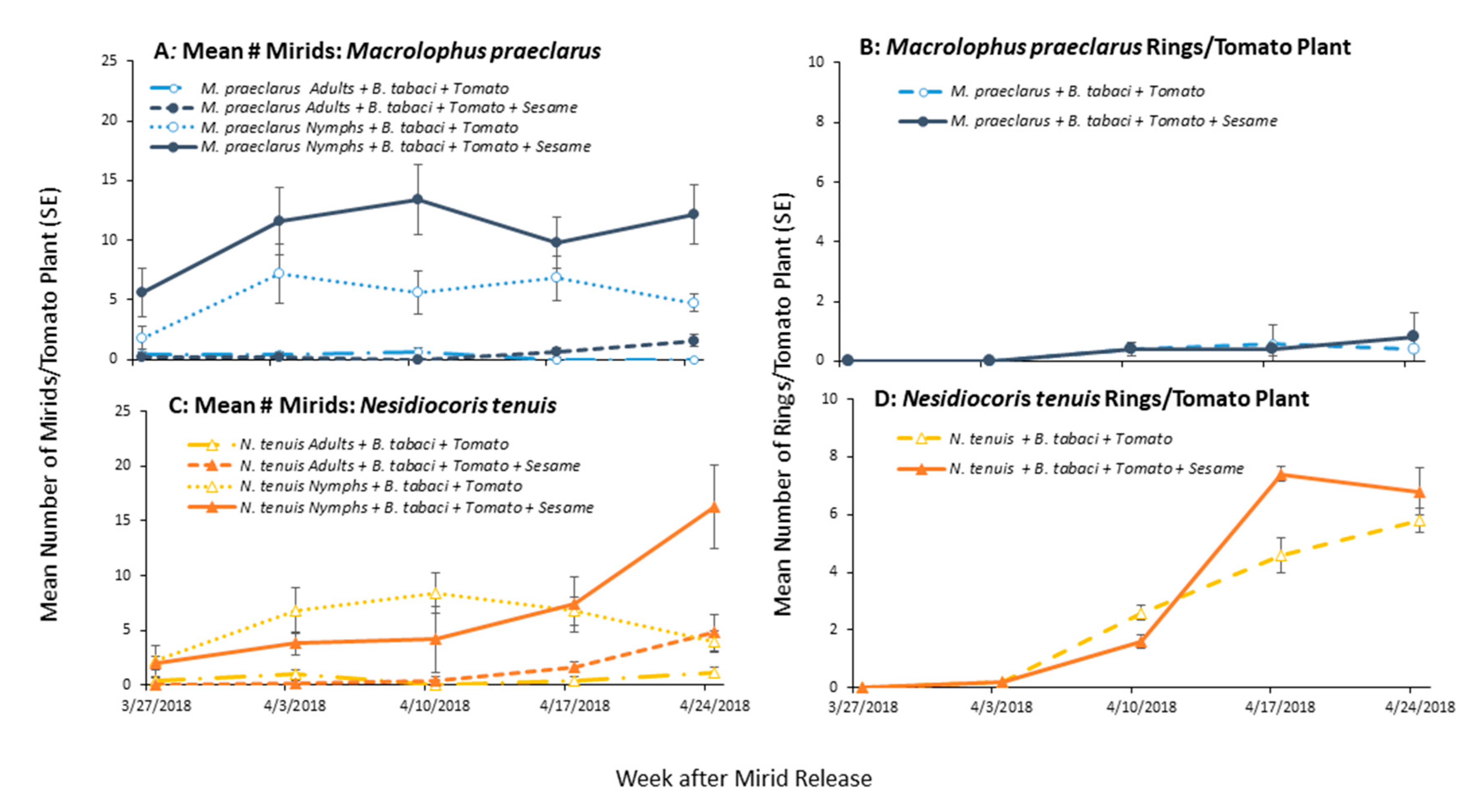
3.1.2. Mirid Population Growth and Plant Damage
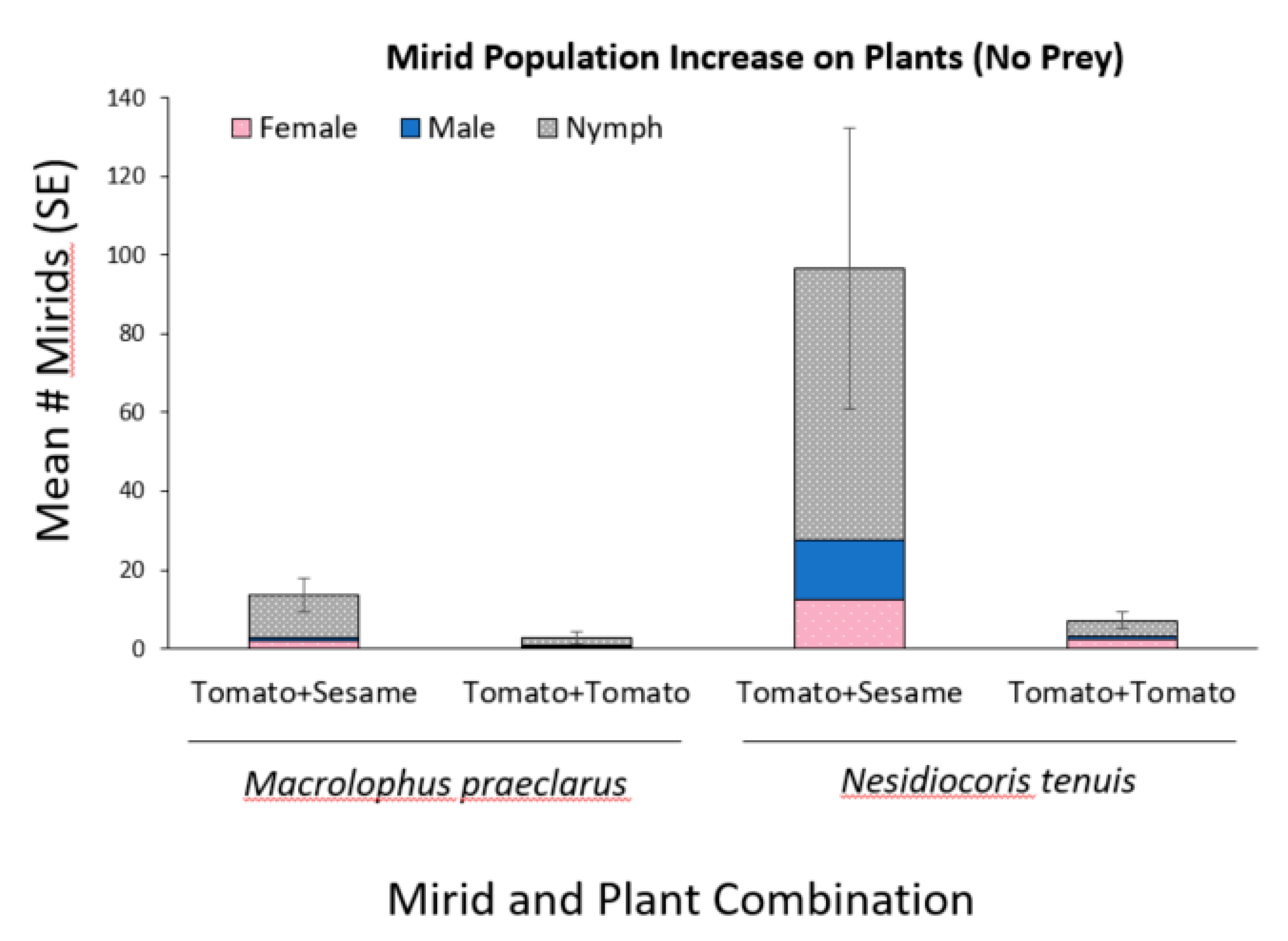
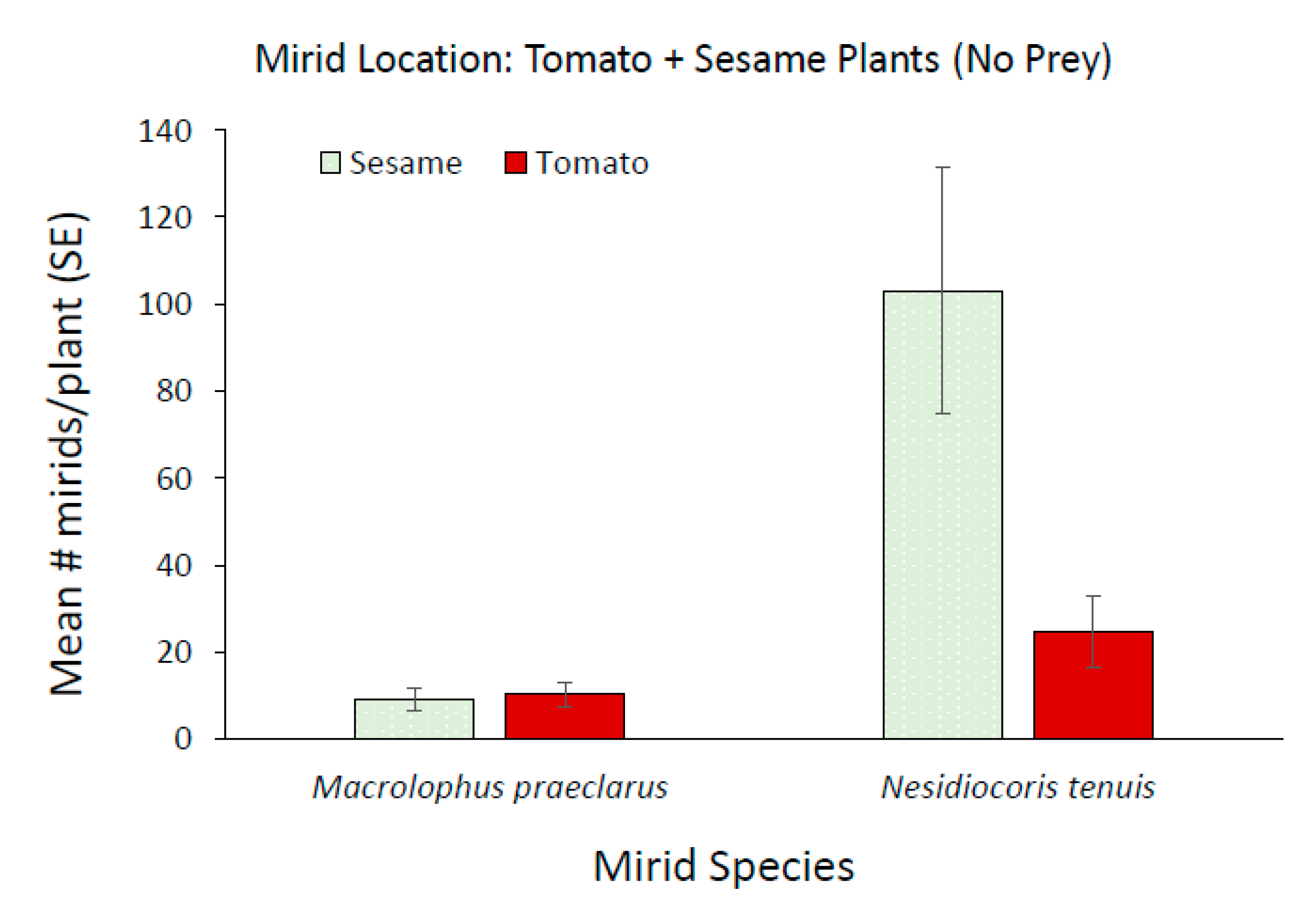
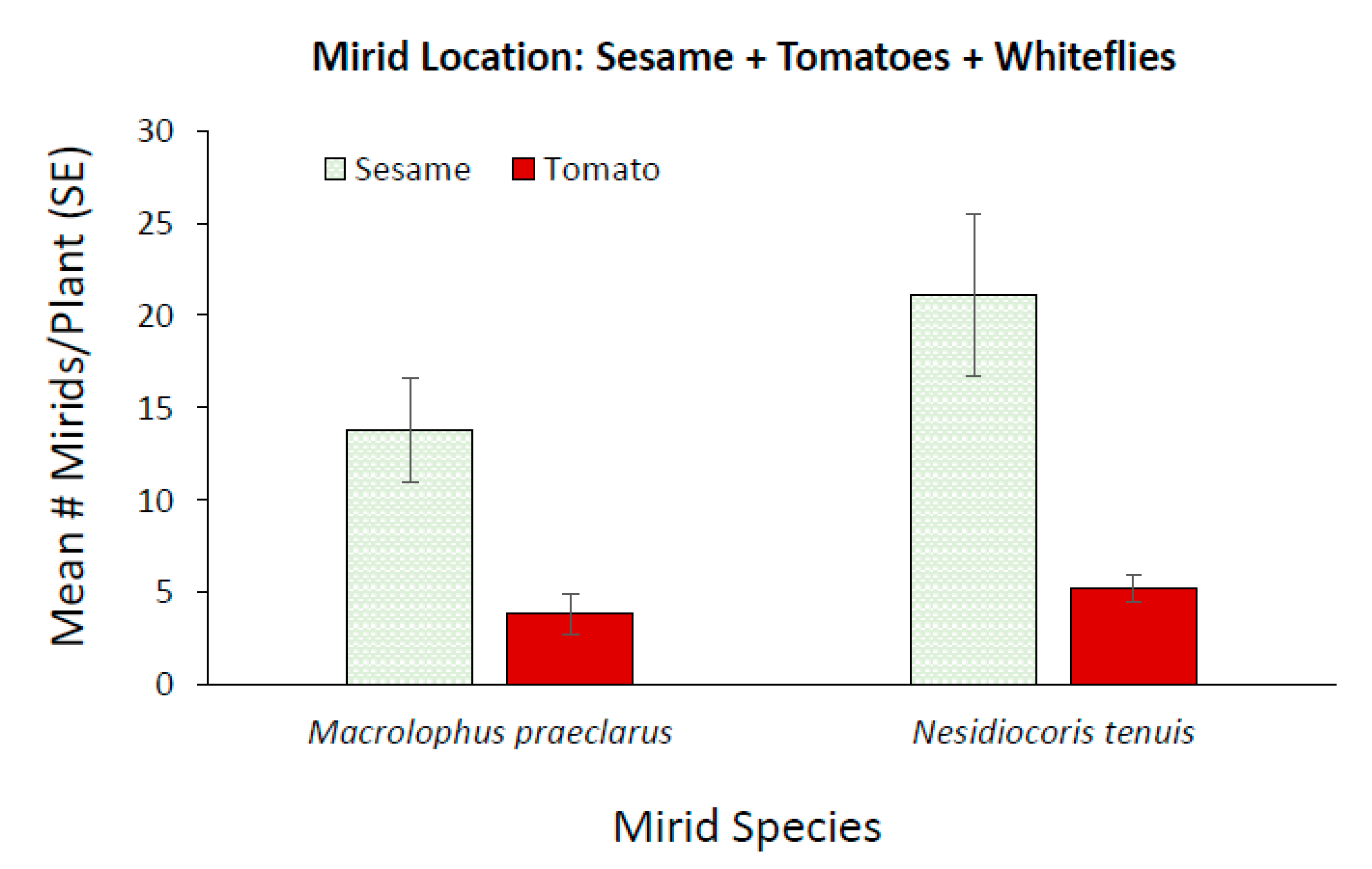
3.2. Effect of Adding Sesame on Mirid Population Growth and Plant Damage
3.3. Effect of Adding Sesame on Mirid Control of Whiteflies on Tomato
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van Lenteren, J.C.; Alomar, O.; Ravensberg, W.J.; Urbaneja, A. Integrated Pest and Disease Management in Greenhouse Crops. In Integrated Pest and Disease Management in Greenhouse Crops, Plant Pathology in the 21st Century 9; Gullino, M.L., Albajes, R., Nicot, P.C., Eds.; Springer: Cham, Switzerland, 2020; pp. 409–439. ISBN 978-3-030-22303-8. [Google Scholar]
- Van Lenteren, J.C.; Bolckmans, K.; Köhl, J.; Ravensberg, W.J.; Urbaneja, A. Biological control using invertebrates and microorganisms: Plenty of new opportunities. BioControl 2018, 63, 39–59. [Google Scholar] [CrossRef]
- Pérez-Hedo, M.; Riahi, C.; Urbaneja, A. Use of zoophytophagous mirid bugs in horticultural crops: Current challenges and future perspectives. Pest Manag. Sci. 2020. [Google Scholar] [CrossRef]
- Castañé, C.; Arnó, J.; Gabarra, R.; Alomar, O. Plant damage to vegetable crops by zoophytophagous mirid predators. Biol. Control 2011, 59, 22–29. [Google Scholar] [CrossRef]
- Urbaneja-Bernat, P.; Bru, P.; González-Cabrera, J.; Urbaneja, A.; Tena, A. Reduced phytophagy in sugar-provisioned mirids. J. Pest Sci. 2019, 92, 1139–1148. [Google Scholar] [CrossRef]
- Pérez-Hedo, M.; Suay, R.; Alonso, M.; Ruocco, M.; Giorgini, M.; Poncet, C.; Urbaneja, A. Resilience and robustness of IPM in protected horticulture in the face of potential invasive pests. Crop Prot. 2017, 97, 119–127. [Google Scholar] [CrossRef]
- Gillespie, D.R.; Mcgregor, R.R. The functions of plant feeding in the omnivorous predator Dicyphus hesperus: Water places limits on predation. Ecol. Entomol. 2000, 25, 380–386. [Google Scholar] [CrossRef]
- Dumont, F.; Aubry, O.; Lucas, E. From evolutionary aspects of zoophytophagy to biological control. Front. Ecol. Evol. 2018, 6, 222. [Google Scholar] [CrossRef]
- Chinchilla-Ramírez, M.; Pérez-Hedo, M.; Pannebakker, B.A.; Urbaneja, A. Genetic Variation in the Feeding Behavior of Isofemale Lines of Nesidiocoris tenuis. Insects 2020, 11, 513. [Google Scholar] [CrossRef]
- Perdikis, D.; Fantinou, A.; Lykouressis, D. Enhancing pest control in annual crops by conservation of predatory Heteroptera. Biol. Control 2011, 59, 13–21. [Google Scholar] [CrossRef]
- Ingegno, B.L.; Ferracini, C.; Gallinotti, D.; Alma, A.; Tavella, L. Evaluation of the effectiveness of Dicyphus errans (Wolff) as predator of Tuta absoluta (Meyrick). Biol. Control 2013, 67, 246–252. [Google Scholar] [CrossRef]
- Madeira, F.; Edo, E.; Sossai, S.; Callizo, J.; Albajes, R. Pre-planting inoculation for early establishment of Dicyphus bolivari and D. errans on tomatoes. BioControl 2019, 64, 33–41. [Google Scholar] [CrossRef]
- Shipp, J.L.; Wang, K. Evaluation of Dicyphus hesperus (Heteroptera: Miridae) for biological control of Frankliniella occidentalis (Thysanoptera: Thripidae) on greenhouse tomato. J. Econ. Entomol. 2006, 99, 414–420. [Google Scholar] [CrossRef]
- Gillespie, D.R.; McGregor, R.R.; Sánchez, J.A. Dicyphus hesperus (Hemiptera: Miridae) as a success story in development of endemic natural enemies as biological control agents. In Case Studies in Biological Control: A Global Perspective; Vincent, C.M., Goettel, M.S., Lazarovits, G., Eds.; CABI Publishing: Wallingford, UK, 2007; pp. 128–135. [Google Scholar]
- Calvo, F.J.; Torres-Ruiz, A.; Velázquez-González, J.C.; Rodríguez-Leyva, E.; Lomeli-Flores, J.R. Evaluation of Dicyphus hesperus for biological control of sweet potato whitefly and potato psyllid on greenhouse tomato. BioControl 2016, 61, 415–424. [Google Scholar] [CrossRef]
- Sanchez, J.A. Zoophytophagy in the plantbug Nesidiocoris tenuis. Agric. For. Entomol. 2008, 10, 75–80. [Google Scholar] [CrossRef]
- Albajes, R.; Castañé, C.; Gabarra, R.; Alomar, Ò. Risks of plant damage caused by natural enemies introduced for arthropod biological control. In Environmental Impact of Invertebrates for Biological Control of Arthropods: Methods and Risk Assessment; CABI: Wallingford, UK, 2006; pp. 132–144. ISBN 0851990584. [Google Scholar]
- van Lenteren, J.C.; Bueno, V.H.P.; Burgio, G.; Lanzoni, A.; Montes, F.C.; Silva, D.B.; de Jong, P.W.; Hemerik, L. Pest kill rate as aggregate evaluation criterion to rank biological control agents: A case study with Neotropical predators of Tuta absoluta on tomato. Bull. Entomol. Res. 2019, 109, 812–820. [Google Scholar] [CrossRef]
- Stansly, P.A.; Naranjo, S.E. Bemisia: Bionomics and Management of a Global Pest; Springer: Dordrecht, The Netherlands, 2010; ISBN 978-90-481-2460-2. [Google Scholar]
- Horowitz, A.R.; Ghanim, M.; Roditakis, E.; Nauen, R.; Ishaaya, I. Insecticide resistance and its management in Bemisia tabaci species. J. Pest Sci. 2020, 93, 893–910. [Google Scholar] [CrossRef]
- Stansly, P.A.; Sánchez, P.A.; Rodríguez, J.M.; Cañizares, F.; Nieto, A.; Leyva, M.J.L.; Fajardo, M.; Suárez, V.; Urbaneja, A. Prospects for biological control of Bemisia tabaci (Homoptera, Aleyrodidae) in greenhouse tomatoes of southern Spain. Crop Prot. 2004, 23, 701–712. [Google Scholar] [CrossRef]
- Pérez-Hedo, M.; Urbaneja, A. The zoophytophagous predator Nesidiocoris tenuis: A successful but controversial biocontrol agent in tomato crops. In Advances in Insect Control and Resistance Management; Horowitz, A.R., Ishaaya, I., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 121–138. ISBN 978-3-319-31798-4. [Google Scholar]
- Moerkens, R.; Pekas, A.; Bellinkx, S.; Hanssen, I.; Huysmans, M.; Bosmans, L.; Wäckers, F. Nesidiocoris tenuis as a pest in Northwest Europe: Intervention threshold and influence of Pepino mosaic virus. J. Appl. Entomol. 2020, 144, 566–577. [Google Scholar] [CrossRef]
- Calvo, J.; Bolckmans, K.; Stansly, P.A.; Urbaneja, A. Predation by Nesidiocoris tenuis on Bemisia tabaci and injury to tomato. BioControl 2009, 54, 237–246. [Google Scholar] [CrossRef]
- Arnó, J.; Castañé, C.; Riudavets, J.; Gabarra, R. Risk of damage to tomato crops by the generalist zoophytophagous predator Nesidiocoris tenuis (Reuter) (Hemiptera: Miridae). Bull. Entomol. Res. 2010, 100, 105–115. [Google Scholar] [CrossRef]
- Sanchez, J.A.; Lacasa, A. Impact of the Zoophytophagous Plant Bug Nesidiocoris tenuis (Heteroptera: Miridae) on Tomato Yield. J. Econ. Entomol. 2008, 101, 1864–1870. [Google Scholar] [CrossRef] [PubMed]
- Chinchilla-Ramírez, M.; Garzo, E.; Fereres, A.; Gavara-Vidal, J.; ten Broeke, C.J..; van Loon, J.J.A.; Urbaneja, A.; Pérez-Hedo, M. Plant feeding by Nesidiocoris tenuis: Quantifying its behavioral and mechanical components. Biol. Control 2021, 152, 104402. [Google Scholar] [CrossRef]
- Biondi, A.; Zappalà, L.; Di Mauro, A.; Tropea Garzia, G.; Russo, A.; Desneux, N.; Siscaro, G. Can alternative host plant and prey affect phytophagy and biological control by the zoophytophagous mirid Nesidiocoris tenuis? BioControl 2016, 61, 79–90. [Google Scholar] [CrossRef]
- Tanada, Y.; Holdaway, F.G. Feeding habits of the tomato bug, Cyrtopeltis (Engytatus) modestus (distant), with special reference to the feeding lesion on tomato. Tech. Bull. Hawaii 1954, 24, 1–41. [Google Scholar]
- Ferguson, G.; Shipp, L. New pests in Ontario greenhouse vegetables. IOBC Wprs Bull. 2002, 25, 69–72. [Google Scholar]
- Parrella, M.; Bethke, J. Biological studies with Cyrtopeltis modestus (Hemiptera, Miridae), a facultative predator of Liriomyza spp. (Dipt, Agromyzidae). In Proceedings of the 3rd Annual International Conference on Leafminer, San Diego, CA, USA, 8–10, 1982; Poe, S., Ed.; Society of American Florists: Alexandria, VA, USA, 1983; pp. 180–185. [Google Scholar]
- González-Jaime, E.J.; Torres-Ruíz, A.; González-Velázquez, J.; Rodríguez-Enríquez, C. Biología de Engytatus modestus (Distant) (Hemiptera: Miridae) depredador generalista, ¿una alternativa en el control biológico. In Memorias XXV Curso Nacional de Control Biológico; González-Hernández, H., Rodríguez-Leyva, E., Lomelí-Flores, J., Eds.; Sociedad Mexicana de Control Biológico: Mérida, México, 2014; pp. 190–195. [Google Scholar]
- Brown, L.; Ellsworth, P.; Bundy, S.; Kerns, D.; Mostafa, A.; Hughes, G.; Porter, P.; Naranjo, S.; Fournier, A. Natural Enemies of the Southwest: A field Guide to the Arthropod Natural Enemies of Southwestern Field Crops; University of Arizona: Tucson, AZ, USA, 2013; ISBN 1304664074. [Google Scholar]
- Martínez, M.A.; Duarte, L.; Baños, H.L.; Rivas, A.; Sánchez, A. Predatory mirids (Hemiptera: Heteroptera: Miridae) in tomato and tobacco in Cuba. Rev. Protección Veg. 2014, 29, 204–207. [Google Scholar]
- Serra, C.; van Lenteren, J.C. Biological Control in the Dominican Republic. In Biological Control in Latin America and the Caribbean: Its Rich History and Bright Future; Lenteren, J.C., van Bueno, V.H.P., Luna, M.G., Colmeneraz, Y.C., Eds.; CABI: Wallingford, UK, 2020; pp. 199–219. [Google Scholar]
- Ayala, J.L.; Grillo, H.; Vera, E.R. Enemigos naturales de Heliothis virescens (Fabricius) (Lepidoptera: Noctuidae) en las provincias centrales de Cuba. Cent. Agrícola 1982, 9, 3–14. [Google Scholar]
- Soto, S.S.; Nakano, O. The occurrence of Macrolophus praeclarus (Distant) Hemiptera: Miridae) in the state of São Paulo, Brazil. Rev. Agric. 2009, 84, 149–151. [Google Scholar]
- Perez-Hedo, M.; Gallego, C.; Roda, A.; Kostyk, B.; Triana, M.; Alférez, F.; Stansly, P.A.; Qureshi, J.; Urbaneja, A. Biological traits of the predatory mirid Macrolophus praeclarus, a candidate biocontrol agent for the Nearctic region. Bull. Entomol. Res. 2020. under review. [Google Scholar]
- Urbaneja-Bernat, P.; Mollá, O.; Alonso, M.; Bolkcmans, K.; Urbaneja, A.; Tena, A. Sugars as complementary alternative food for the establishment of Nesidiocoris tenuis in greenhouse tomato. J. Appl. Entomol. 2015, 139, 161–167. [Google Scholar] [CrossRef]
- SAS Institute. JMP®, Version 13; SAS Institute: Cary, NC, USA, 2013. [Google Scholar]
- Siegel, S.; Castellan, N.J. Nonparametric Statistics for the Behavioural Sciences, 2nd ed.; McGraw-Hill Book Company: New York, NY, USA, 1988. [Google Scholar]
- Wheeler, A.G., Jr. Biology of the Plant Bugs (Hemiptera: Miridae): Pests, Predators, Opportunists; Comstock Publishing Associates: Ithaca, NY, USA, 2001; ISBN 0801438276. [Google Scholar]
- Coll, M.; Guershon, M. Omnivory in Terrestrial Arthropods: Mixing Plant and Prey Diets. Annu. Rev. Entomol. 2002, 47, 267–297. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, A.G.; Henry, T.J. A Synthesis of the Holarctic Miridae (Heteroptera): Distribution, Biology, and Origin, with Emphasis on North America; Entomological Society of America: Lanham, MD, USA, 1992. [Google Scholar]
- Pérez-Hedo, M.; Urbaneja-Bernat, P.; Jaques, J.A.; Flors, V.; Urbaneja, A. Defensive plant responses induced by Nesidiocoris tenuis (Hemiptera: Miridae) on tomato plants. J. Pest Sci. 2015, 88, 543–554. [Google Scholar] [CrossRef]
- Pérez-Hedo, M.; Bouagga, S.; Jaques, J.A.; Flors, V.; Urbaneja, A. Tomato plant responses to feeding behavior of three zoophytophagous predators (Hemiptera: Miridae). Biol. Control 2015, 86, 46–51. [Google Scholar] [CrossRef]
- Raman, K.; Sanjayan, K.P. Histology and Histopathology of the Feeding Lesions by Cyrtopeltis tenuis Reut (Hemiptera, Miridae) on Lycopersicon esculentum Mill (Solanaceae). Proc. Indian Acad. Sci. 1984, 93, 543–547. [Google Scholar] [CrossRef]
- Sampson, C.; Jacobson, R.J. Macrolophus caliginosus Wagner (Heteroptera: Miridae): A predator causing damage in UK tomatoes. IOBC WPRS Bull. 1999, 22, 213–216. [Google Scholar]
- Sanchez, J.A.; López-Gallego, E.; Pérez-Marcos, M.; Perera-Fernández, L.G.; Ramírez-Soria, M.J. How Safe Is It to Rely on Macrolophus pygmaeus (Hemiptera: Miridae) as a Biocontrol Agent in Tomato Crops? Front. Ecol. Evol. 2018, 6, 1–10. [Google Scholar] [CrossRef]
- Moerkens, R.; Berckmoes, E.; Van Damme, V.; Ortega-Parra, N.; Hanssen, I.; Wuytack, M.; Wittemans, L.; Casteels, H.; Tirry, L.; De Clercq, P.; et al. High population densities of Macrolophus pygmaeus on tomato plants can cause economic fruit damage: Interaction with Pepino mosaic virus? Pest Manag. Sci. 2016, 72, 1350–1358. [Google Scholar] [CrossRef]
- Rim, H.; Hattori, S.; Arimura, G. Mint companion plants enhance the attraction of the generalist predator Nesidiocoris tenuis according to its experiences of conspecific mint volatiles. Sci. Rep. 2020, 10, 2078. [Google Scholar] [CrossRef]
- Abbas, S.; Pérez-Hedo, M.; Colazza, S.; Urbaneja, A. The predatory mirid Dicyphus maroccanus as a new potential biological control agent in tomato crops. BioControl 2014, 59, 565–574. [Google Scholar] [CrossRef]
- Lucas, É.; Alomar, O. Impact of the presence of Dicyphus tamaninii Wagner (Heteroptera: Miridae) on whitefly (Homoptera: Aleyrodidae) predation by Macrolophus caliginosus (Wagner) (Heteroptera: Miridae). Biol. Control 2002, 25, 123–128. [Google Scholar] [CrossRef]
- Urbaneja, A.; Tapia, G.; Stansly, P. Influence of host plant and prey availability on developmental time and surviorship of Nesidiocoris tenius (Het.: Miridae). Biocontrol Sci. Technol. 2005, 15, 513–518. [Google Scholar] [CrossRef]
- Lins, J.C.; van Loon, J.J.A.; Bueno, V.H.P.; Lucas-Barbosa, D.; Dicke, M.; van Lenteren, J.C. Response of the zoophytophagous predators Macrolophus pygmaeus and Nesidiocoris tenuis to volatiles of uninfested plants and to plants infested by prey or conspecifics. BioControl 2014, 59, 707–718. [Google Scholar] [CrossRef]
- Naselli, M.; Zappalà, L.; Gugliuzzo, A.; Tropea Garzia, G.; Biondi, A.; Rapisarda, C.; Cincotta, F.; Condurso, C.; Verzera, A.; Siscaro, G. Olfactory response of the zoophytophagous mirid Nesidiocoris tenuis to tomato and alternative host plants. Arthropod. Plant Interact. 2017, 11, 121–131. [Google Scholar] [CrossRef]
- Nakaishi, K.; Fukui, Y.; Arakawa, R. Reproduction of Nesidiocoris tenuis (Reuter) on Sesame. Jpn. J. Appl. Entomol. Zool. 2011, 55, 199–205. [Google Scholar] [CrossRef]
- Ingegno, B.L.; Pansa, M.G.; Tavella, L. Plant preference in the zoophytophagous generalist predator Macrolophus pygmaeus (Heteroptera: Miridae). Biol. Control 2011, 58, 174–181. [Google Scholar] [CrossRef]
- Symondson, W.O.C.; Sunderland, K.D.; Greenstone, M.H. Can generalist predators be effective biocontrol agents? Ann. Rev. Entomol. 2002, 47, 561–594. [Google Scholar] [CrossRef]
- Zappalà, L.; Biondi, A.; Alma, A.; Al-Jboory, I.J.; Arnò, J.; Bayram, A.; Chailleux, A.; El-Arnaouty, A.; Gerling, D.; Guenaoui, Y.; et al. Natural enemies of the South American moth, Tuta absoluta, in Europe, North Africa and Middle East, and their potential use in pest control strategies. J. Pest Sci. 2013, 86, 635–647. [Google Scholar] [CrossRef]
- Bouvet, J.P.R.; Urbaneja, A.; Pérez-Hedo, M.; Monzó, C. Contribution of predation to the biological control of a key herbivorous pest in citrus agroecosystems. J. Anim. Ecol. 2019, 88, 915–926. [Google Scholar] [CrossRef]
- Labbé, R.M.; Gagnier, D.; Kostic, A.; Shipp, L. The function of supplemental foods for improved crop establishment of generalist predators Orius insidiosus and Dicyphus hesperus. Sci. Rep. 2018, 8, 17790. [Google Scholar] [CrossRef]
- Biondi, A.; Desneux, N. Special issue on Tuta absoluta: Recent advances in management methods against the background of an ongoing worldwide invasion. J. Pest Sci. 2019, 92, 1313–1315. [Google Scholar] [CrossRef]
- Tembrock, L.R.; Timm, A.E.; Zink, F.A.; Gilligan, T.M. Phylogeography of the Recent Expansion of Helicoverpa armigera (Lepidoptera: Noctuidae) in South America and the Caribbean Basin. Ann. Entomol. Soc. Am. 2019, 112, 388–401. [Google Scholar] [CrossRef]
- Urbaneja, A.; González-Cabrera, J.; Arnó, J.; Gabarra, R. Prospects for the biological control of Tuta absoluta in tomatoes of the Mediterranean basin. Pest Manag. Sci. 2012, 68, 1215–1222. [Google Scholar] [CrossRef]
| Experiment | Source | Response Variable | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Whiteflies | Mirids | Necrotic Rings | ||||||||
| d.f. | F | p | d.f. | F | p | d.f. | F | p | ||
| M.p. vs. E.m.: Tomato + B.t. | Treatment | 2, 11 | 15.71 | <0.001 | 1, 7 | 13.50 | 0.008 | 1, 7 | 2.82 | 0.14 |
| Week | 4, 44 | 5.46 | <0.001 | 4, 28 | 0.99 | 0.43 | 4, 28 | 2.13 | 0.10 | |
| Treatment × Week | 8, 44 | 6.34 | <0.001 | 4, 28 | 2.08 | 0.11 | 4, 28 | 1.75 | 0.17 | |
| M.p. vs. N.t.: Tomato + B.t. | Treatment | 2, 15 | 17.38 | <0.001 | 1, 10 | 1.15 | 0.29 | 1, 10 | 22.37 | <0.001 |
| Week | 4, 60 | 9.01 | <0.001 | 4, 40 | 1.75 | 0.16 | 4, 40 | 26.41 | <0.001 | |
| Treatment × Week | 8, 60 | 7.96 | <0.001 | 4, 40 | 0.86 | 0.49 | 4, 40 | 5.48 | <0.001 | |
| M.p. vs. N.t.: Tomato + Sesame + B.t. | Treatment | 4, 20 | 20.57 | <0.001 | 3, 16 | 2.72 | 0.08 | 3, 16 | 21.78 | <0.001 |
| Week | 4, 90 | 9.01 | <0.001 | 4, 64 | 8.22 | <0.001 | 4, 64 | 29.65 | <0.001 | |
| Treatment × Week | 16, 90 | 7.16 | <0.001 | 12, 64 | 2.07 | 0.03 | 12, 64 | 5.88 | <0.001 | |
| M.p. only: Tomato + Sesame + B.t. | Treatment | 1, 8 | 6.95 | 0.03 | 1, 8 | 0.20 | 0.88 | |||
| Week | 4, 32 | 4.37 | 0.006 | 4, 32 | 1.61 | 0.19 | ||||
| Treatment × Week | 4, 32 | 0.35 | 0.87 | 4, 32 | 0.04 | 0.997 | ||||
| N.t. only: Tomato+ Sesame + B.t. | Treatment | 1, 8 | 0.32 | 0.58 | 1, 8 | 0.17 | 0.69 | |||
| Week | 4, 32 | 6.66 | <0.001 | 4, 32 | 36.33 | <0.001 | ||||
| Treatment × Week | 4, 32 | 3.50 | 0.02 | 4, 32 | 1.38 | 0.26 | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roda, A.; Castillo, J.; Allen, C.; Urbaneja, A.; Pérez-Hedo, M.; Weihman, S.; Stansly, P.A. Biological Control Potential and Drawbacks of Three Zoophytophagous Mirid Predators against Bemisia tabaci in the United States. Insects 2020, 11, 670. https://doi.org/10.3390/insects11100670
Roda A, Castillo J, Allen C, Urbaneja A, Pérez-Hedo M, Weihman S, Stansly PA. Biological Control Potential and Drawbacks of Three Zoophytophagous Mirid Predators against Bemisia tabaci in the United States. Insects. 2020; 11(10):670. https://doi.org/10.3390/insects11100670
Chicago/Turabian StyleRoda, Amy, Jose Castillo, Carina Allen, Alberto Urbaneja, Meritxell Pérez-Hedo, Scott Weihman, and Philip A. Stansly. 2020. "Biological Control Potential and Drawbacks of Three Zoophytophagous Mirid Predators against Bemisia tabaci in the United States" Insects 11, no. 10: 670. https://doi.org/10.3390/insects11100670
APA StyleRoda, A., Castillo, J., Allen, C., Urbaneja, A., Pérez-Hedo, M., Weihman, S., & Stansly, P. A. (2020). Biological Control Potential and Drawbacks of Three Zoophytophagous Mirid Predators against Bemisia tabaci in the United States. Insects, 11(10), 670. https://doi.org/10.3390/insects11100670






