Chronic High Glyphosate Exposure Delays Individual Worker Bee (Apis mellifera L.) Development under Field Conditions
Simple Summary
Abstract
1. Introduction
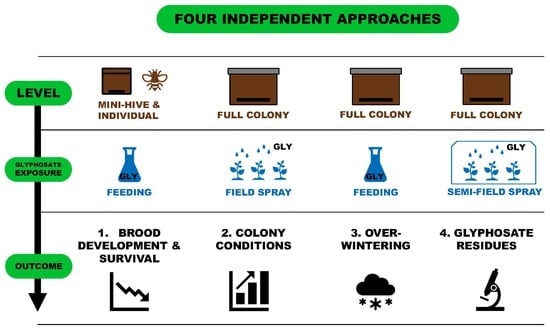
2. Experiment 1: Brood Development and Survival
2.1. Methods
2.1.1. Experimental Colonies and Field Site
2.1.2. Chemical Treatment and Sampling
2.1.3. Colony Conditions
2.1.4. Brood Development and Photographic Assessment
2.1.5. Survival of Individual Bees and Hatching Weight
2.1.6. Statistical Analyses
2.2. Results
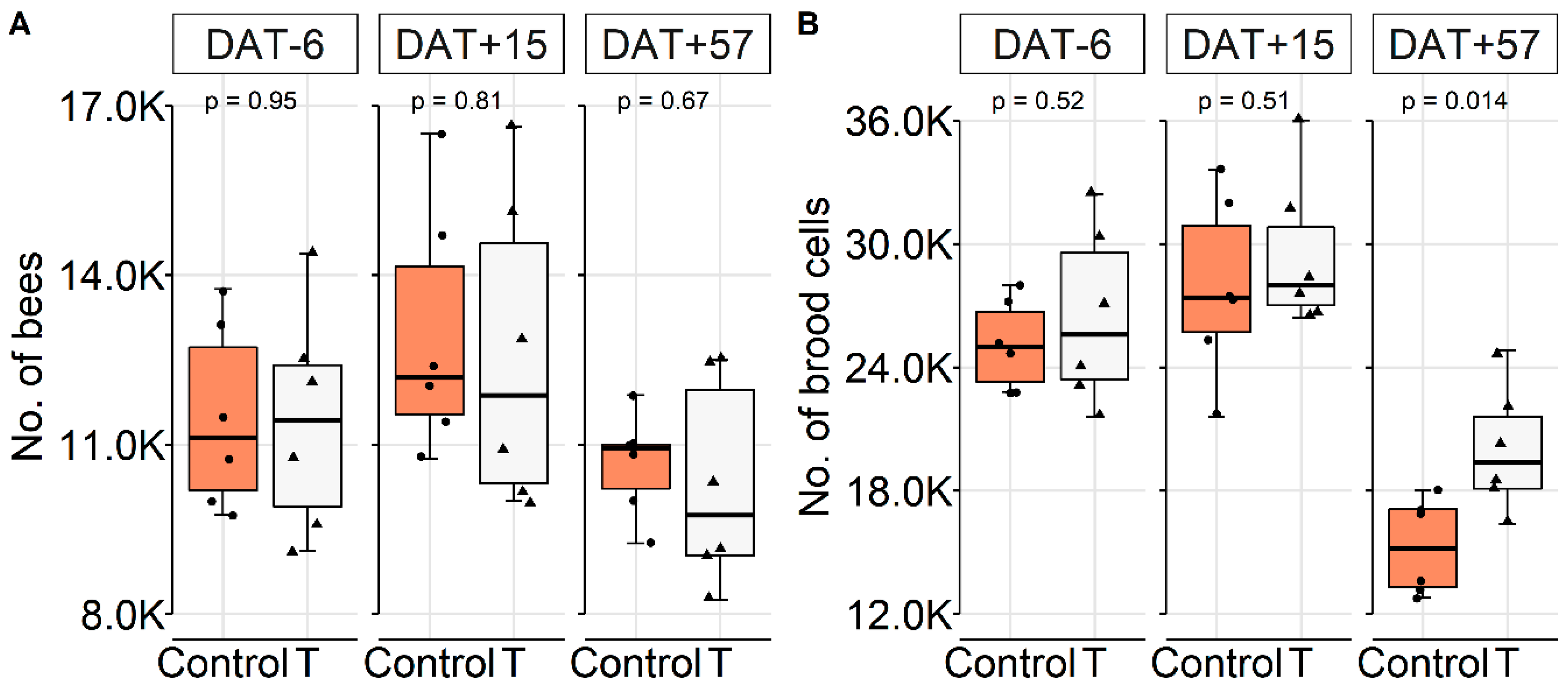
2.2.1. Colony Conditions
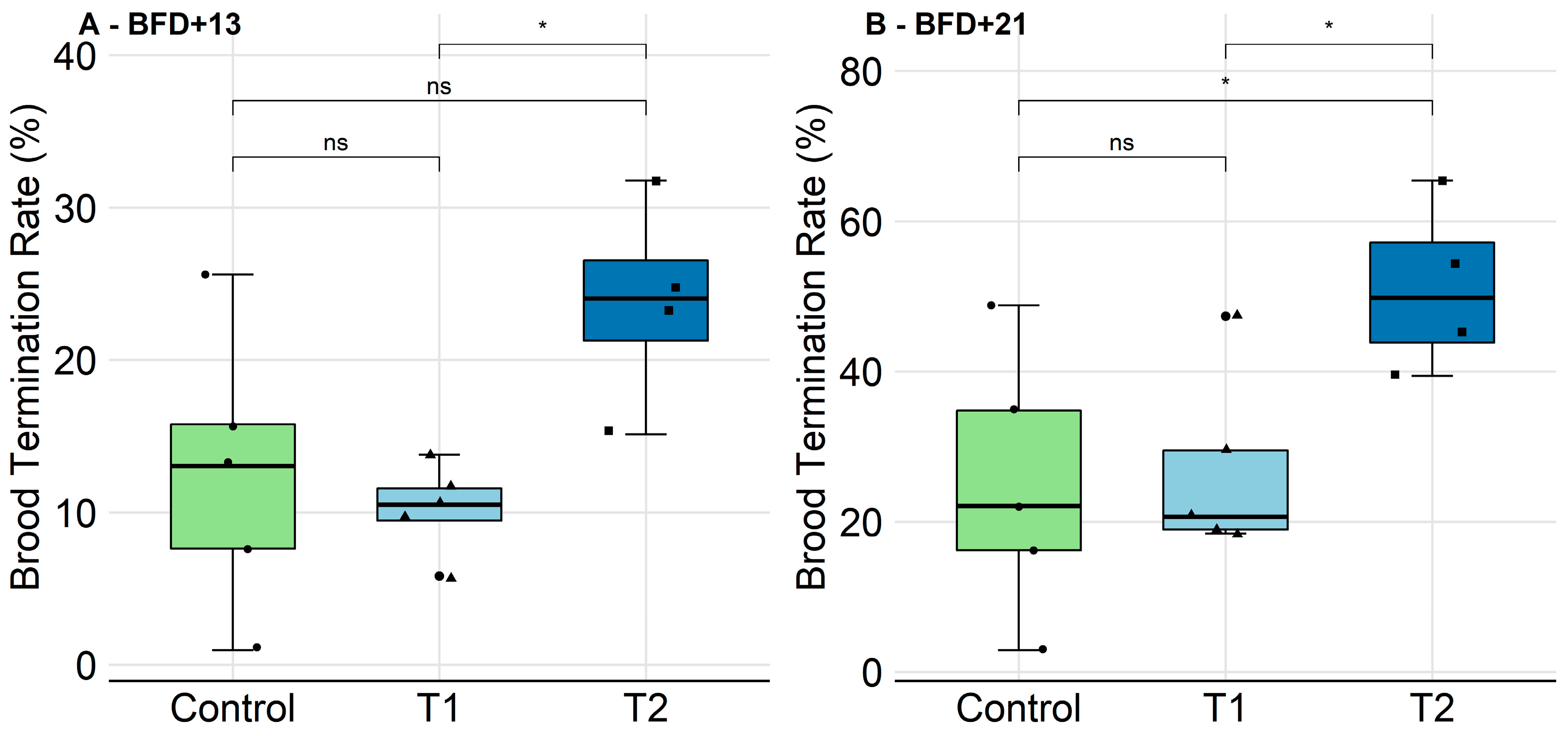
2.2.2. Brood Development and Photographic Assessment
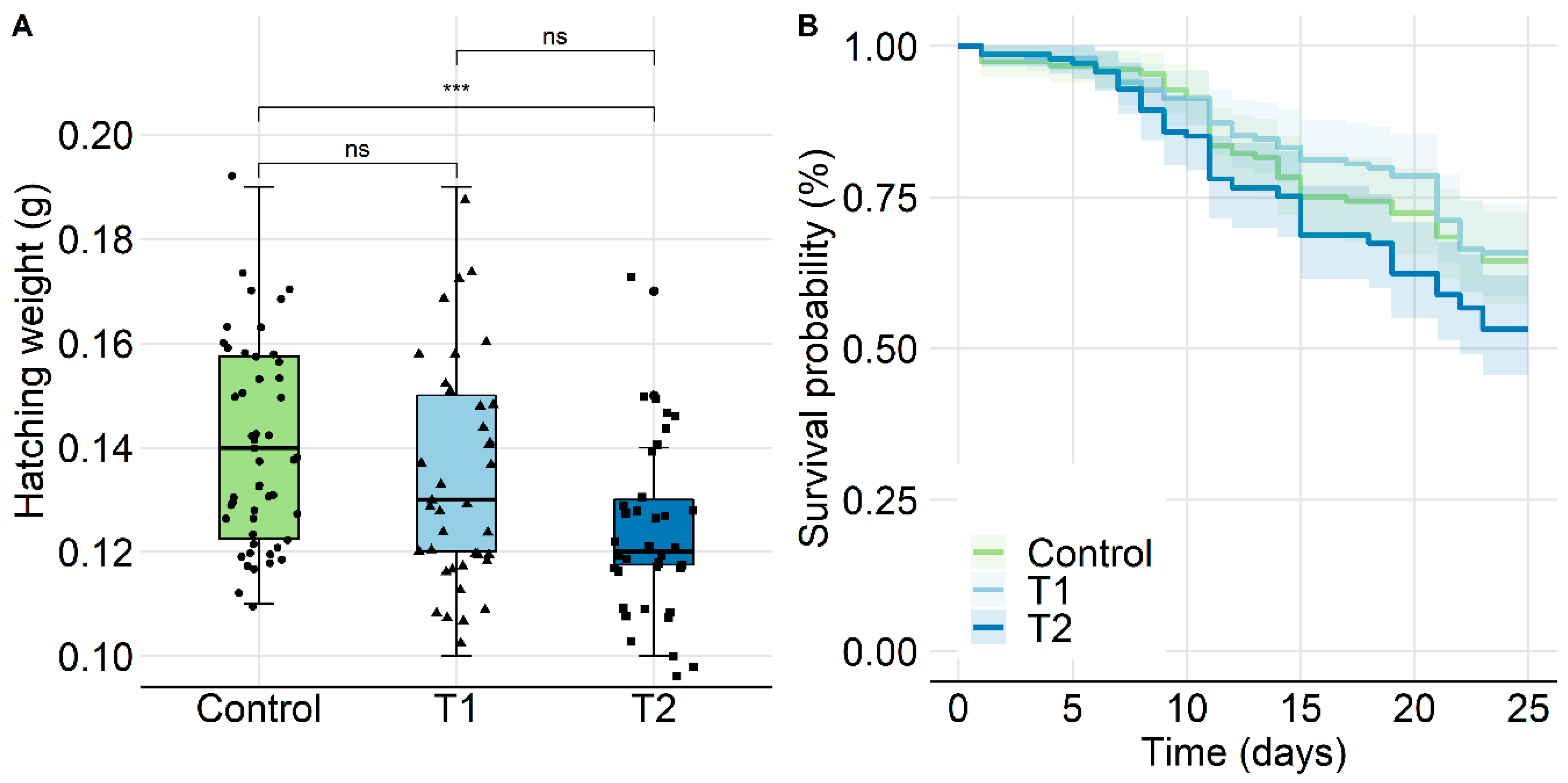
2.2.3. Survival of Individual Bees and Hatching Weight
3. Experiment 2: Field Exposure
3.1. Methods
3.1.1. Experimental Colonies and Field Sites
3.1.2. Chemical Treatment and Sampling
3.1.3. Colony Conditions
3.1.4. Statistical Analyses
3.2. Results
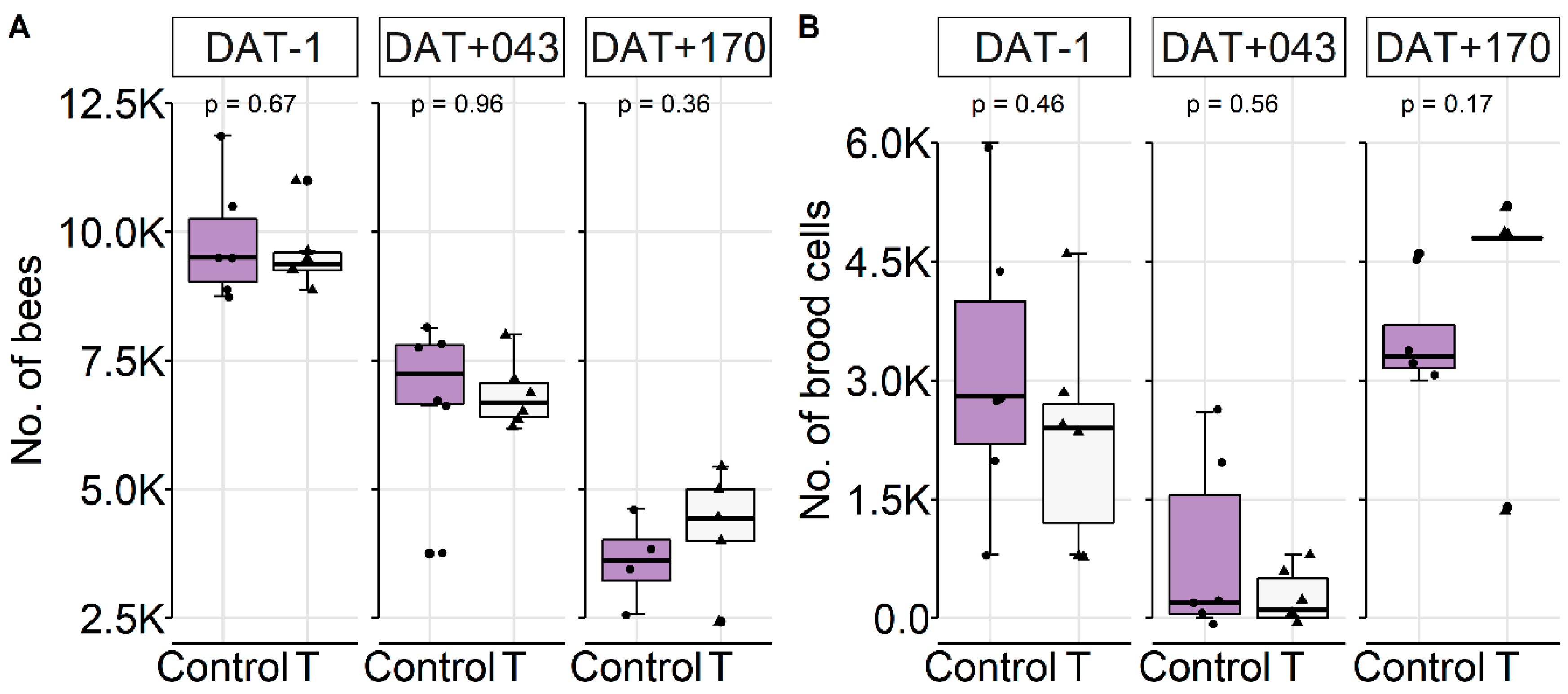
Colony Conditions
4. Experiment 3: Overwintering
4.1. Methods
4.1.1. Experimental Colonies and Field Sites
4.1.2. Chemical Treatment and Sampling
4.1.3. Colony Conditions
4.1.4. Statistical Analyses
4.2. Results
Colony Conditions
5. Experiment 4: Determination of GBH Residues
5.1. Methods
5.1.1. Experimental Colonies and Field Sites
5.1.2. Chemical Treatment and Sampling
5.1.3. Residue Analyses
5.2. Results
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AMPA | Aminomethylphosphonic acid |
| ANOVA | Analysis of variance |
| BBCH | Biologische Bundesanstalt für Land—und Forstwirtschaft, Bundessortenamt und CHemische Industrie |
| BFD | Brood area fixing day |
| BTR | Brood termination rate |
| DAT | Day after treatment |
| EC | European commission |
| EFSA | European Food Safety Authority |
| EPPO | European and Mediterranean Plant Protection Organization |
| GBH | Glyphosate-based herbicides |
| HPLC | High-performance liquid chromatography |
| ICP-PR | International Commission for Plant Pollinator Relationships |
| IPA | Isopropylamine |
| OECD | Organization for Economic Co-operation and Development |
| UV | Ultraviolet |
References
- De María, N.; Becerril, J.M.; García-Plazaola, J.I.; Hernández, A.; De Felipe, M.R.; Fernández-Pascual, M. New insights on glyphosate mode of action in nodular metabolism: Role of shikimate accumulation. J. Agric. Food Chem. 2006, 54, 2621–2628. [Google Scholar] [CrossRef] [PubMed]
- Benbrook, C.M. Trends in glyphosate herbicide use in the United States and globally. Environ. Sci. Eur. 2016, 28, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Richmond, M.E. Glyphosate: A review of its global use, environmental impact, and potential health effects on humans and other species. J. Environ. Stud. Sci. 2018, 8, 416–434. [Google Scholar] [CrossRef]
- Steinmann, H.H.; Dickeduisberg, M.; Theuvsen, L. Uses and benefits of glyphosate in German arable farming. Crop Prot. 2012, 42, 164–169. [Google Scholar] [CrossRef]
- Bauernzeitung. Glyphosat im Honig. Available online: https://www.bauernzeitung.de/news/glyphosat-im-honig/ (accessed on 7 August 2020).
- European Commission (EC). Earlier Assessment of Glyphosate. Available online: https://ec.europa.eu/food/plant/pesticides/glyphosate/earlier-assessment_en (accessed on 15 February 2020).
- Thompson, T.S.; van den Heever, J.P.; Limanowka, R.E. Determination of glyphosate, AMPA, and glufosinate in honey by online solid-phase extraction-liquid chromatography-tandem mass spectrometry. Food Addit. Contam. Part A 2019, 36, 434–446. [Google Scholar] [CrossRef]
- Berg, C.J.; King, H.P.; Delenstarr, G.; Kumar, R.; Rubio, F.; Glaze, T. Glyphosate residue concentrations in honey attributed through geospatial analysis to proximity of large-scale agriculture and transfer off-site by bees. PLoS ONE 2018, 13, e0198876. [Google Scholar] [CrossRef]
- Karise, R.; Raimets, R.; Bartkevics, V.; Pugajeva, I.; Pihlik, P.; Keres, I.; Williams, I.H.; Viinalass, H.; Mänd, M. Are pesticide residues in honey related to oilseed rape treatments? Chemosphere 2017, 188, 389–396. [Google Scholar] [CrossRef]
- Rubio, F.; Guo, E.; Kamp, L. Survey of Glyphosate Residues in Honey, Corn and Soy Products. J. Environ. Anal. Toxicol. 2014, 05, 1–8. [Google Scholar] [CrossRef]
- Dickeduisberg, M.; Steinmann, H.H.; Theuvsen, L. Erhebungen zum Einsatz von Glyphosat im deutschen Ackerbau. In Proceedings of the 25 th Germany Conference Weed Biology Weed Control, Braunschweig, Germany, 13–15 March 2012; pp. 2010–2013. [Google Scholar] [CrossRef]
- Di Pasquale, G.; Salignon, M.; Le Conte, Y.; Belzunces, L.P.; Decourtye, A.; Kretzschmar, A.; Suchail, S.; Brunet, J.L.; Alaux, C. Influence of Pollen Nutrition on Honey Bee Health: Do Pollen Quality and Diversity Matter? PLoS ONE 2013, 8, e72016. [Google Scholar] [CrossRef]
- Schonbrunn, E.; Eschenburg, S.; Shuttleworth, W.A.; Schloss, J.V.; Amrhein, N.; Evans, J.N.S.; Kabsch, W. Interaction of the herbicide glyphosate with its target enzyme 5-enolpyruvylshikimate 3-phosphate synthase in atomic detail. Proc. Natl. Acad. Sci. USA 2001, 98, 1376–1380. [Google Scholar] [CrossRef]
- EFSA (European Food Safety Authority). Conclusion on the peer review of the pesticide risk assessment of the active substance glyphosate. EFSA J. 2015, 13. [Google Scholar] [CrossRef]
- Chmiel, J.A.; Daisley, B.A.; Pitek, A.P.; Thompson, G.J.; Reid, G. Understanding the Effects of Sublethal Pesticide Exposure on Honey Bees: A Role for Probiotics as Mediators of Environmental Stress. Front. Ecol. Evol. 2020, 8, 1–19. [Google Scholar] [CrossRef]
- Farina, W.M.; Balbuena, M.S.; Herbert, L.T.; Mengoni Goñalons, C.; Vázquez, D.E. Effects of the Herbicide Glyphosate on Honey Bee Sensory and Cognitive Abilities: Individual Impairments with Implications for the Hive. Insects 2019, 10, 354. [Google Scholar] [CrossRef] [PubMed]
- Balbuena, M.S.; Tison, L.; Hahn, M.L.M.-L.M.L.; Greggers, U.; Menzel, R.; Farina, W.M. Effects of sublethal doses of glyphosate on honeybee navigation. J. Exp. Biol. 2015, 218, 2799–2805. [Google Scholar] [CrossRef] [PubMed]
- Motta, E.V.S.; Raymann, K.; Moran, N.A. Glyphosate perturbs the gut microbiota of honey bees. Proc. Natl. Acad. Sci. USA 2018, 115, 10305–10310. [Google Scholar] [CrossRef]
- Dai, P.; Yan, Z.; Ma, S.; Yang, Y.; Wang, Q.; Hou, C.; Wu, Y.; Liu, Y.; Diao, Q. The Herbicide Glyphosate Negatively Affects Midgut Bacterial Communities and Survival of Honey Bee during Larvae Reared in Vitro. J. Agric. Food Chem. 2018, 66, 7786–7793. [Google Scholar] [CrossRef]
- Blot, N.; Veillat, L.; Rouzé, R.; Delatte, H. Glyphosate, but not its metabolite AMPA, alters the honeybee gut microbiota. PLoS ONE 2019, 14, e0215466. [Google Scholar] [CrossRef]
- Faita, M.R.; Oliveira, E.M.; Alves, V.V.; Orth, A.I.; Nodari, R.O. Changes in hypopharyngeal glands of nurse bees (Apis mellifera) induced by pollen-containing sublethal doses of the herbicide Roundup®. Chemosphere 2018, 211, 566–572. [Google Scholar] [CrossRef]
- Vázquez, D.E.; Ilina, N.; Pagano, E.A.; Zavala, J.A.; Farina, W.M. Glyphosate affects the larval development of honey bees depending on the susceptibility of colonies. PLoS ONE 2018, 13, e205074. [Google Scholar] [CrossRef]
- Sponsler, D.B.; Johnson, R.M. Mechanistic modeling of pesticide exposure: The missing keystone of honey bee toxicology. Environ. Toxicol. Chem. 2017, 36, 871–881. [Google Scholar] [CrossRef]
- Straub, L.; Williams, G.R.; Pettis, J.; Fries, I.; Neumann, P. Superorganism resilience: Eusociality and susceptibility of ecosystem service providing insects to stressors. Curr. Opin. Insect Sci. 2015, 12, 109–112. [Google Scholar] [CrossRef]
- Scharf, I.; Modlmeier, A.P.; Beros, S.; Foitzik, S. Ant societies buffer individual-level effects of parasite infections. Am. Nat. 2012, 180, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.M.; Levine, S.L.; Doering, J.; Norman, S.; Manson, P.; Sutton, P.; von Mérey, G. Evaluating exposure and potential effects on honeybee brood (Apis mellifera) development using glyphosate as an example. Integr. Environ. Assess. Manag. 2014, 10, 463–470. [Google Scholar] [CrossRef]
- Ciarlo, T.J.; Mullin, C.A.; Frazier, J.L.; Schmehl, D.R. Learning Impairment in Honey Bees Caused by Agricultural Spray Adjuvants. PLoS ONE 2012, 7, e40848. [Google Scholar] [CrossRef] [PubMed]
- Mullin, C.A.; Fine, J.D.; Reynolds, R.D.; Frazier, M.T. Toxicological Risks of Agrochemical Spray Adjuvants: Organosilicone Surfactants May Not Be Safe. Front. Public Heal. 2016, 4, 1–8. [Google Scholar] [CrossRef]
- Oomen, P.A.; De Ruijter, A.; Van Der Steen, J.J.M. Method for honeybee brood feeding tests with insect growth-regulating insecticides. EPPO Bull. 1992, 22, 613–616. [Google Scholar] [CrossRef]
- OECD (Organisation for Economic Co-operation and Development). Guidance Document on the Honey Bee (Apis mellifera L.) Brood Test under Semi-Field Conditions No. 75; OECD: Paris, France, 2007.
- Schur, A.; Tornier, I.; Brasse, D.; Mühlen, W.; Von Werner Ohe, D.E.R.; Wallner, K.; Wehling, M. Honey bee brood ring-test in 2002: Method for the assessment of side effects of plant protection products on the honey bee brood under semi-field conditions. Bull. Insectology 2003, 56, 91–96. [Google Scholar]
- Odemer, R.; Nilles, L.; Linder, N.; Rosenkranz, P. Sublethal effects of clothianidin and Nosema spp. on the longevity and foraging activity of free flying honey bees. Ecotoxicology 2018, 27, 527–538. [Google Scholar] [CrossRef]
- Wetter BW Wetterdienst Baden-Württemberg. Agrarmeteorologie. Available online: https://www.wetter-bw.de (accessed on 25 December 2019).
- Imdorf, A.; Buehlmann, G.; Gerig, L.; Kilchenmann, V. Überprüfung der Schätzmethode zur Ermittlung der Brutfläche und der Anzahl Arbeiterinnen in freifliegenden Bienenvölkern. Apidologie 1987, 18, 137–146. [Google Scholar] [CrossRef]
- Therneau, T. A Package for Survival Analysis in S 2015. Available online: https://CRAN.R-project.org/package=survival (accessed on 25 September 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Kassambara, A.; Kosinski, M.; Biecek, P. survminer: Drawing Survival Curves Using “ggplot2” 2019. Available online: https://CRAN.R-project.org/package=survminer (accessed on 25 September 2020).
- Kassambara, A. ggpubr: “ggplot2” Based Publication Ready Plots 2019. Available online: https://CRAN.R-project.org/package=ggpubr (accessed on 25 September 2020).
- Wang, Y.; Ma, L.-T.; Xu, B.-H. Diversity in life history of queen and worker honey bees, Apis mellifera L. J. Asia Pac. Entomol. 2015, 18, 145–149. [Google Scholar] [CrossRef]
- Jay, S.C. The development of honeybees in their cells. J. Apic. Res. 1963, 2, 117–134. [Google Scholar] [CrossRef]
- Chamkasem, N.; Morris, C.; Harmon, T. Direct Determination of Glyphosate, Glufosinate, and AMPA in milk by Liquid chromatography/tandem mass spectrometry. J. Regul. Sci. 2015, 2, 20–26. [Google Scholar]
- Chamkasem, N.; Harmon, T. Direct determination of glyphosate, glufosinate, and AMPA in soybean and corn by liquid chromatography/tandem mass spectrometry. Anal. Bioanal. Chem. 2016, 18. [Google Scholar] [CrossRef] [PubMed]
- Chamkasem, N.; Vargo, J.D. Development and Independent Laboratory Validation of an Analytical Method for the Direct Determination of Glyphosate, Glufosinate, and Aminomethylphosphonic Acid in Honey by Liquid Chromatography/Tandem Mass. Spectrometry. J. Regul. Sci. 2017, 5, 1–9. [Google Scholar]
- Chamkasem, N.; Morris, C.; Harmon, T. Direct Determination of Glyphosate, Glufosinate, and AMPA in honey by liquid chromatography/tandem mass spectrometer. In Proceedings of the PittCon, Atlanta, GA, USA, 6–10 March 2016. [Google Scholar]
- SANTE/12682/2019. Method Validation and Quality Control Procedures for Pesticide Residues Analysis in Food and Feed; European Union Reference Laboratory Residues Pesticide: Geel, Belgium, 2020. [Google Scholar]
- Odemer, R. Data: Chronic high glyphosate exposure delays individual worker bee (Apis mellifera L.) development under field conditions. Open Sci. Framew. 2020. [Google Scholar] [CrossRef]
- Imdorf, A.; Ruoff, K.; Fluri, P. Volksentwicklung bei der Honigbiene. ALP Forum 2008, 68, 1–88. [Google Scholar]
- Kunert, K.; Crailsheim, K. Seasonal Changes in Carbohydrate, Lipid and Protein Content in Emerging Worker Honeybees and their Mortality. J. Apic. Res. 1988, 27, 13–21. [Google Scholar] [CrossRef]
- Zółtowska, K.; Fraczek, R.; Lipiński, Z. Hydrolases of developing worker brood and newly emerged worker of Apis mellifera carnica. J. Apic. Sci. 2011, 55, 27–36. [Google Scholar]
- Mesnage, R.; Bernay, B.; Séralini, G.-E. Ethoxylated adjuvants of glyphosate-based herbicides are active principles of human cell toxicity. Toxicology 2013, 313, 122–128. [Google Scholar] [CrossRef]
- Mesnage, R.; Antoniou, M.N. Ignoring Adjuvant Toxicity Falsifies the Safety Profile of Commercial Pesticides. Front. Public Health 2018, 5, 1–8. [Google Scholar] [CrossRef]
- Frey, E.; Odemer, R.; Blum, T.; Rosenkranz, P. Activation and interruption of the reproduction of Varroa destructor is triggered by host signals (Apis mellifera). J. Invertebr. Pathol. 2013, 113, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Odemer, R. Reproductive capacity of Varroa destructor in four different honey bee subspecies. Saudi J. Biol. Sci. 2020, 27, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Odemer, R.; Rosenkranz, P. Chronic exposure to a neonicotinoid pesticide and a synthetic pyrethroid in full-sized honey bee colonies. J. Apic. Res. 2020, 59, 2–11. [Google Scholar] [CrossRef]
- Manjon, C.; Troczka, B.J.; Zaworra, M.; Beadle, K.; Randall, E.; Hertlein, G.; Singh, K.S.; Zimmer, C.T.; Homem, R.A.; Lueke, B.; et al. Unravelling the Molecular Determinants of Bee Sensitivity to Neonicotinoid Insecticides. Curr. Biol. 2018, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Berenbaum, M.R.; Johnson, R.M. Xenobiotic detoxification pathways in honey bees. Curr. Opin. Insect Sci. 2015, 10, 51–58. [Google Scholar] [CrossRef]
- Wang, M.; Braasch, T.; Dietrich, C. Reduction of variability for the assessment of side effects of toxicants on honeybees and understanding drivers for colony development. PLoS ONE 2020, 15, e0229295. [Google Scholar] [CrossRef]
- Lückmann, J.; Becker, R.; Schmitzer, S. Variability of brood termination rates in bee brood studies according to Oomen and OECD GD 75. In Proceedings of the SETAC Europe Conference, Barcelona, Spain, 3–7 May 2015. [Google Scholar]
- Pistorius, J.; Lückmann, J.; Schur, A.; Barth, M.; Jeker, L.; Schmitzer, S.; von der Ohe, W. Effectiveness of method improvements to reduce variability of brood termination rate in honey bee brood 694 studies under semi-field conditions. In Proceedings of the 11th International Symposium of the ICP-BR Bee Protection Group, Wageningen, The Netherlands, 2–4 November 2012; Volume 2011, pp. 45–54. [Google Scholar]
- Colin, T.; Bruce, J.; Meikle, W.G.; Barron, A.B. The development of honey bee colonies assessed using a new semi-automated brood counting method: CombCount. PLoS ONE 2018, 13, e0205816. [Google Scholar] [CrossRef]
- Bermig, S.; Odemer, R.; Gombert, A.; Frommberger, M.; Rosenquist, R.; Pistorius, J. Experimental validation of an electronic counting device to determine flight activity of honey bees (Apis mellifera L.). J. Cultiv. Plants 2020, 72, 132–140. [Google Scholar] [CrossRef]
- Szekacs, A.; Darvas, B. Forty Years with Glyphosate. In Herbicides-Properties, Synthesis and Control of Weeds; InTech: London, UK, 2012. [Google Scholar]
- Curran, W.S.; Lingenfelter, D.D. Adjuvants for Enhancing Herbicide Performance; Pennsylvania State University: State College, PA, USA, 2009. [Google Scholar]
- Sviridov, A.V.; Shushkova, T.V.; Ermakova, I.T.; Ivanova, E.V.; Epiktetov, D.O.; Leontievsky, A.A. Microbial degradation of glyphosate herbicides (review). Appl. Biochem. Microbiol. 2015, 51, 188–195. [Google Scholar] [CrossRef]
- Israili, Z.H. Antimicrobial properties of honey. Am. J. Ther. 2014, 21, 304–323. [Google Scholar] [CrossRef]
- Chirife, J.; Herszage, L.; Joseph, A.; Kohn, E.S. In vitro study of bacterial growth inhibition in concentrated sugar solutions: Microbiological basis for the use of sugar in treating infected wounds. Antimicrob. Agents Chemother. 1983, 23, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Assalin, M.R.; De Moraes, S.G.; Queiroz, S.C.N.; Ferracini, V.L.; Duran, N. Studies on degradation of glyphosate by several oxidative chemical processes: Ozonation, photolysis and heterogeneous photocatalysis. J. Environ. Sci. Health Part B 2009, 45, 89–94. [Google Scholar] [CrossRef] [PubMed]
- EPPO (European and Mediterranean Plant Protection Organization). Side-effects on honeybees (PP 1/170 (4). EPPO Bull. 2010, 40, 313–319. [Google Scholar] [CrossRef]
- Havard, T.; Laurent, M.; Chauzat, M. Impact of Stressors on Honey Bees (Apis mellifera; Hymenoptera: Apidae): Some Guidance for Research Emerge from a Meta-Analysis. Diversity 2019, 12, 7. [Google Scholar] [CrossRef]
- Cullen, M.G.; Thompson, L.J.; Carolan, J.C.; Stout, J.C.; Stanley, D.A. Fungicides, herbicides and bees: A systematic review of existing research and methods. PLoS ONE 2019, 14, e0225743. [Google Scholar] [CrossRef]


| Stock Solution | Stored Food/Nectar | Honey Sac | Beebread | Corbicular Pollen | Plants | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Time | Samples (n) | GLY | AMPA | GLY | AMPA | GLY | AMPA | GLY | AMPA | GLY | AMPA | GLY | AMPA | |
| Experiment 1: Brood development & survival | DAT0 (T1) | 1 | 4.764 ± 0.424 + | 0.020 ± 0.003 + | - | - | - | - | - | - | - | - | - | - |
| DAT0 (T2) | 1 | 137.566 ± 4.318 + | 0.428 ± 0.001 + | - | - | - | - | - | - | - | - | - | - | |
| DAT+21 (T1) | 5 * | - | - | 5.103 | 0.013 | - | - | - | - | - | - | - | - | |
| DAT+21 (T2) | 4 * | - | - | 99.861 | 0.319 | - | - | - | - | - | - | - | - | |
| Experiment 2: Field exposure | DAT0 (+0.5 h) | 6 * | - | - | n.d. | n.d. | - | - | - | - | - | - | 96.429 ± 13.540 + | 0.360 ± 0.005 + |
| DAT+1 | 6 | - | - | 0.327 ± 0.246 | 0.087 ± 0.194 | - | - | - | - | - | - | 44.715 ± 1.429 + | 0.255 ± 0.007 + | |
| DAT+3 | 6 | - | - | 0.490 ± 0.455 | n.d. | - | - | - | - | - | - | 33.634 ± 0.016 + | 0.218 ± 0.017 + | |
| DAT+6 | 6 | - | - | 0.347 ± 0.362 | n.d. | - | - | 5.512 ± 6.922 | 0.007 ± 0.017 | - | - | - | - | |
| DAT+7 | 6 | - | - | - | - | - | - | - | - | - | - | 31.051 ± 1.930 + | 0.224 ± 0.007 + | |
| Experiment 3: Overwintering | DAT −1 | 12 | - | - | 0.004 ± 0.012 | n.d. | - | - | - | - | - | - | - | - |
| DAT+6 | 6 | - | - | 4.462 ± 1.651 | 0.006 ± 0.005 | - | - | - | - | - | - | - | - | |
| DAT+13 | 6 | - | - | 5.159 ± 1.011 | 0.012 ± 0.004 | - | - | - | - | - | - | - | - | |
| DAT+43 | 6 | - | - | 7.148 ± 3.234 | 0.033 ± 0.015 | - | - | - | - | - | - | - | - | |
| DAT+170 | 6 | - | - | 4.985 ± 0.613 | 0.019 ± 0.004 | - | - | - | - | - | - | - | - | |
| Experiment 4: Residues under semi-field conditions | DAT0 (+1 h) | 3 * | - | - | - | - | 24.918 | 0.092 | - | - | 614.841 | 2.122 | 82.407 | 0.217 |
| DAT+1 | 3 * | - | - | - | - | - | - | - | - | - | - | 103.892 | 0.362 | |
| DAT+2 | 3 * | - | - | - | - | - | - | - | - | - | - | 62.845 | 0.229 | |
| DAT+3 | 3 * | - | - | - | - | - | - | - | - | - | - | 92.886 | 0.395 | |
| DAT+5 | 4 | - | - | 0.182 ± 0.225 | n.d. | - | - | - | - | - | - | - | - | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Odemer, R.; Alkassab, A.T.; Bischoff, G.; Frommberger, M.; Wernecke, A.; Wirtz, I.P.; Pistorius, J.; Odemer, F. Chronic High Glyphosate Exposure Delays Individual Worker Bee (Apis mellifera L.) Development under Field Conditions. Insects 2020, 11, 664. https://doi.org/10.3390/insects11100664
Odemer R, Alkassab AT, Bischoff G, Frommberger M, Wernecke A, Wirtz IP, Pistorius J, Odemer F. Chronic High Glyphosate Exposure Delays Individual Worker Bee (Apis mellifera L.) Development under Field Conditions. Insects. 2020; 11(10):664. https://doi.org/10.3390/insects11100664
Chicago/Turabian StyleOdemer, Richard, Abdulrahim T. Alkassab, Gabriela Bischoff, Malte Frommberger, Anna Wernecke, Ina P. Wirtz, Jens Pistorius, and Franziska Odemer. 2020. "Chronic High Glyphosate Exposure Delays Individual Worker Bee (Apis mellifera L.) Development under Field Conditions" Insects 11, no. 10: 664. https://doi.org/10.3390/insects11100664
APA StyleOdemer, R., Alkassab, A. T., Bischoff, G., Frommberger, M., Wernecke, A., Wirtz, I. P., Pistorius, J., & Odemer, F. (2020). Chronic High Glyphosate Exposure Delays Individual Worker Bee (Apis mellifera L.) Development under Field Conditions. Insects, 11(10), 664. https://doi.org/10.3390/insects11100664