Antiviral Effectors and Gene Drive Strategies for Mosquito Population Suppression or Replacement to Mitigate Arbovirus Transmission by Aedes aegypti
Abstract
1. Introduction
2. Aedes aegypti Population Suppression Strategies
3. Antiviral Effector Strategies for Aedes aegypti Population Replacement
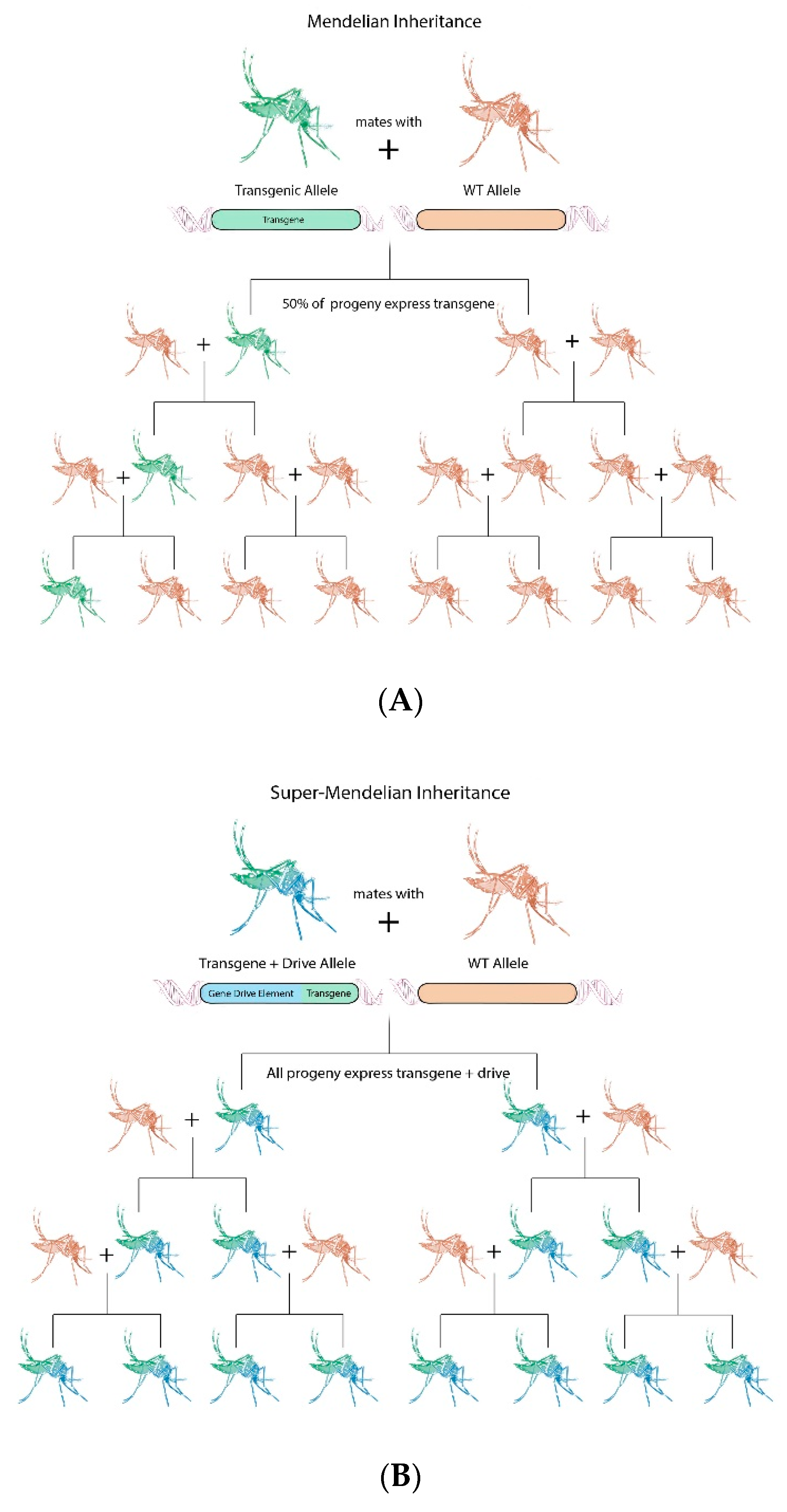
4. Gene Drive Principles
- (1)
- Compensate for any loss of fitness associated with the effector gene;
- (2)
- Link tightly to complex effector genes that are associated with a fluorescent marker;
- (3)
- Drive the effector gene relatively quickly to fixation within the target population;
- (4)
- Adapt to genetically diverse strains of mosquitoes;
- (5)
- Remain confined to the targeted species irrespective of population structure and mating dynamics between species;
- (6)
- Resist mutations that diminish or block drive to be sustained in nature;
- (7)
- Be socially accepted by those communities who might benefit.
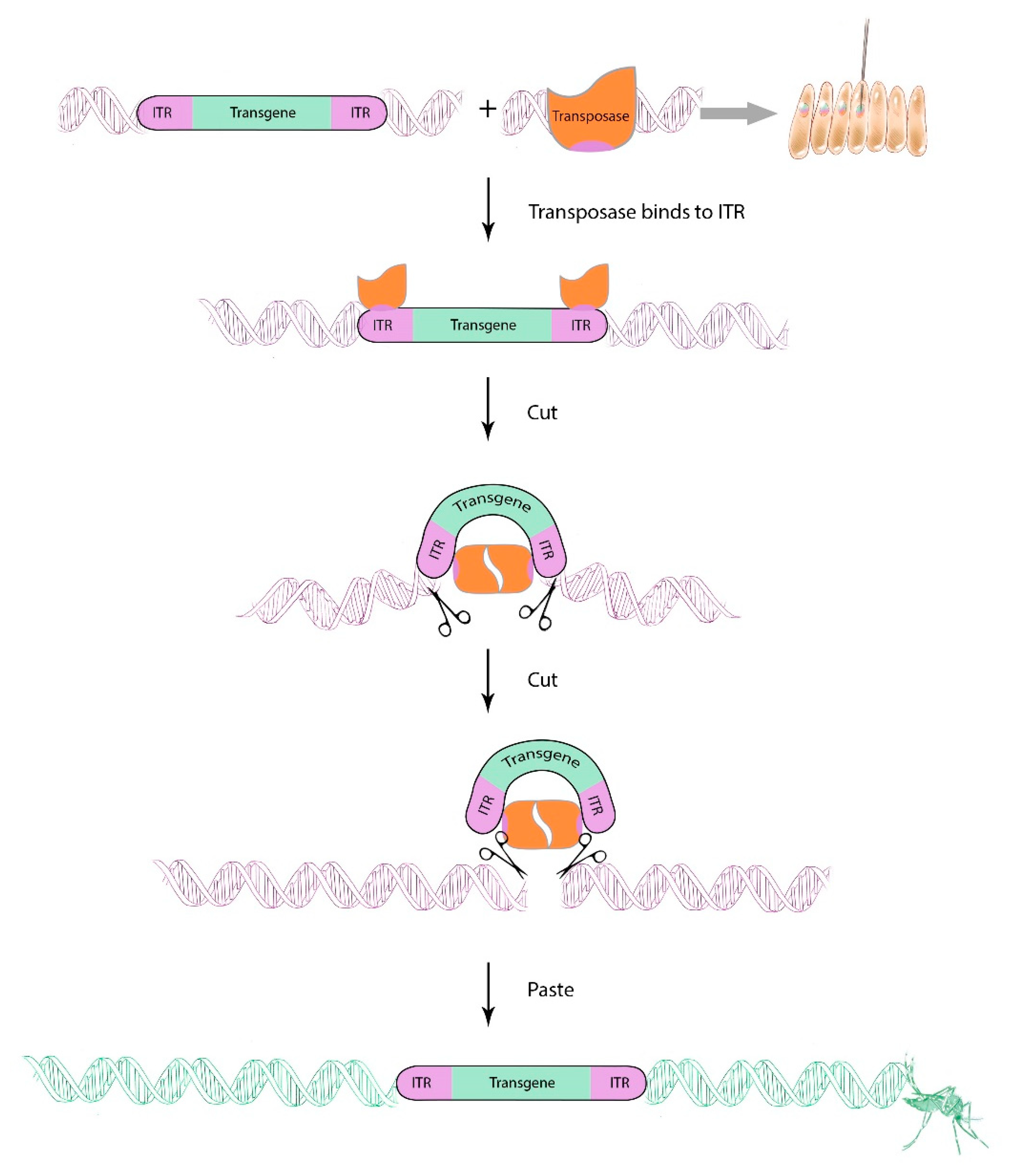
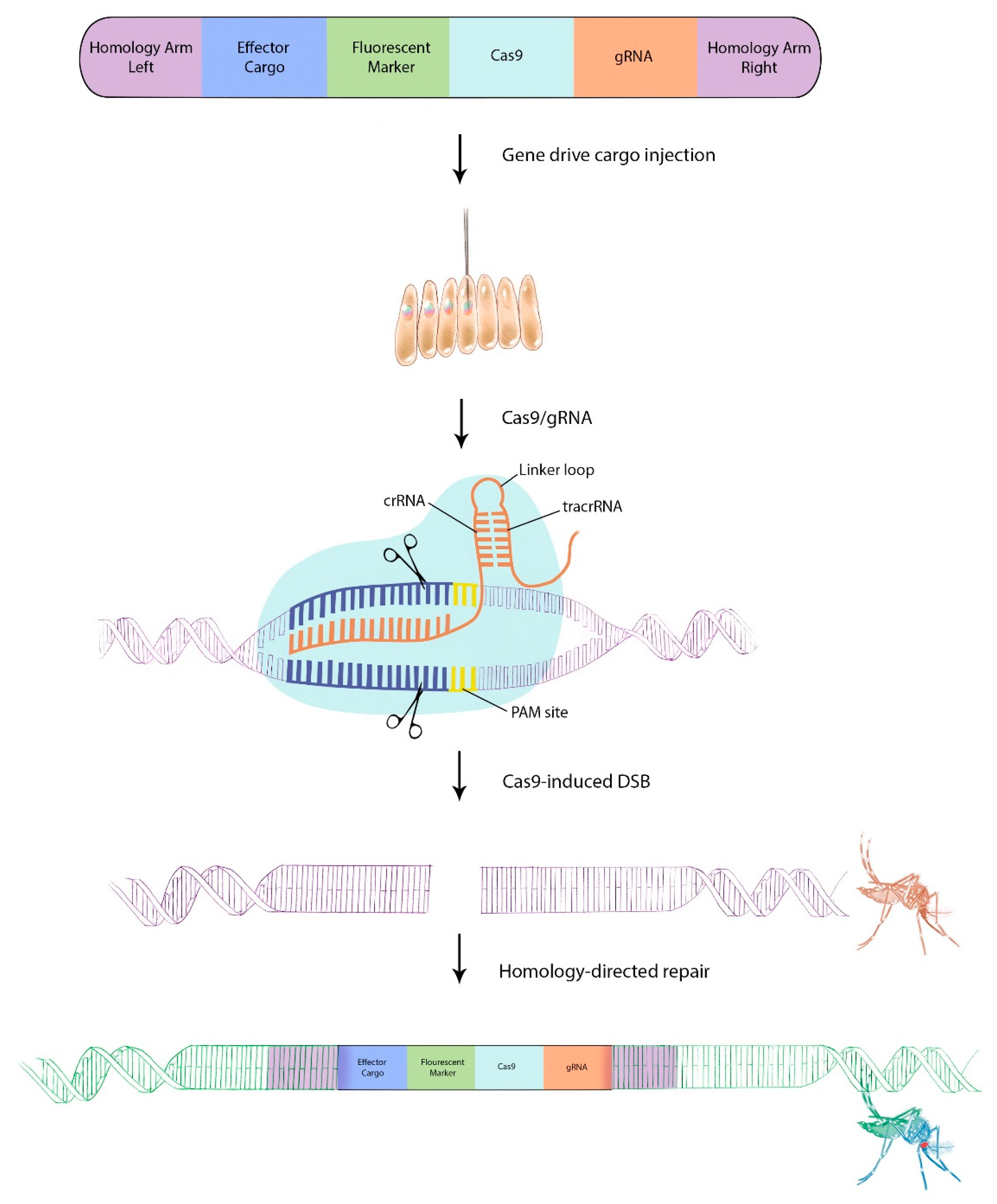
5. Gene Drive Technologies for Aedes aegypti
6. Aedes aegypti Population Replacement Models
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wilder-Smith, A.; Gubler, D.J.; Weaver, S.C.; Monath, T.P.; Heymann, D.L.; Scott, T.W. Epidemic arboviral diseases: Priorities for research and public health. Lancet Infect. Dis. 2017, 17, 101–106. [Google Scholar] [CrossRef]
- Powell, J.R.; Tabachnick, W.J. History of domestication and spread of Aedes aegypti—A review. Mem. Inst. Oswaldo Cruz 2013, 108 (Suppl. 1), 11–17. [Google Scholar] [CrossRef]
- Luckhart, S.; Lindsay, S.W.; James, A.A.; Scott, T.W. Reframing critical needs in vector biology and management of vector-borne disease. PLoS Negl. Trop. Dis. 2010, 4, e566. [Google Scholar] [CrossRef]
- Moyes, C.L.; Vontas, J.; Martins, A.J.; Ng, L.C.; Koou, S.Y.; Dusfour, I.; Raghavendra, K.; Pinto, J.; Corbel, V.; David, J.P.; et al. Contemporary status of insecticide resistance in the major Aedes vectors of arboviruses infecting humans. PLoS Negl. Trop. Dis. 2017, 11, e0005625. [Google Scholar] [CrossRef] [PubMed]
- Gould, F. Broadening the application of evolutionarily based genetic pest management. Evolution 2008, 62, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Bargielowski, I.; Nimmo, D.; Alphey, L.; Koella, J.C. Comparison of life history characteristics of the genetically modified OX513A line and a wild type strain of Aedes aegypti. PLoS ONE 2011, 6, e20699. [Google Scholar] [CrossRef] [PubMed]
- Harris, A.F.; Nimmo, D.; McKemey, A.R.; Kelly, N.; Scaife, S.; Donnelly, C.A.; Beech, C.; Petrie, W.D.; Alphey, L. Field performance of engineered male mosquitoes. Nat. Biotechnol. 2011, 29, 1034–1037. [Google Scholar] [CrossRef]
- Paes de Andrade, P.; Aragao, F.J.L.; Colli, W.; Dellagostin, O.A.; Finardi-Filho, F.; Hirata, M.H.; Lira-Neto, A.C.; Almeida de Melo, M.; Nepomuceno, A.L.; Gorgônio da Nóbrega, F.; et al. Use of transgenic Aedes aegypti in Brazil: Risk perception and assessment. Bull. World Health Organ. 2016, 10, 766–771. [Google Scholar] [CrossRef]
- Facchinelli, L.; Valerio, L.; Ramsey, J.M.; Gould, F.; Walsh, R.K.; Bond, G.; Robert, M.A.; Lloyd, A.L.; James, A.A.; Alphey, L.; et al. Field cage studies and progressive evaluation of genetically-engineered mosquitoes. PLoS Negl. Trop. Dis. 2013, 7, e2001. [Google Scholar] [CrossRef]
- Bargielowski, I.; Kaufmann, C.; Alphey, L.; Reiter, P.; Koella, J. Flight performance and teneral energy reserves of two genetically-modified and one wild-type strain of the yellow fever mosquito Aedes aegypti. Vector Borne Zoonotic Dis. 2012, 12, 1053–1058. [Google Scholar] [CrossRef]
- McMeniman, C.J.; Lane, R.V.; Cass, B.N.; Fong, A.W.C.; Sidhu, M.; Wang, Y.-F.; Neill, S.L. Stable introduction of a life-shortening Wolbachia infection into the mosquito Aedes aegypti. Science 2009, 323, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Moreira, L.A.; Iturbe-Ormaetxe, I.; Jeffery, J.A.; Lu, G.; Pyke, A.T.; Hedges, L.M.; Rocha, B.C.; Hall-Mendelin, S.; Day, A.; Riegler, M.; et al. A Wolbachia symbiont in Aedes aegypti limits infection with dengue, Chikungunya, and Plasmodium. Cell 2009, 139, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.; Johnson, P.H.; Moreira, L.A.; Iturbe-Ormaetxe, I.; Frentiu, F.D.; McMeniman, C.J.; Leong, Y.S.; Dong, Y.; Axford, J.; Kriesner, P.; et al. The wMel Wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature 2011, 476, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Sicard, M.; Bonneau, M.; Weill, M. Wolbachia prevalence, diversity, and ability to induce cytoplasmic incompatibility in mosquitoes. Curr. Opin. Insect Sci. 2019, 34, 12–20. [Google Scholar] [CrossRef]
- McMeniman, C.J.; Lane, A.M.; Fong, A.W.; Voronin, D.A.; Iturbe-Ormaetxe, I.; Yamada, R.; McGraw, E.A.; O’Neill, S.L. Host adaptation of a Wolbachia strain after long-term serial passage in mosquito cell lines. Appl. Environ. Microbiol. 2008, 74, 6963–6969. [Google Scholar] [CrossRef]
- Joubert, D.A.; Walker, T.; Carrington, L.B.; De Bruyne, J.T.; Kien, D.H.; Hoang, N.; Chau, N.V.; Iturbe-Ormaetxe, I.; Simmons, C.P.; O’Neill, S.L. Establishment of a Wolbachia superinfection in Aedes aegypti mosquitoes as a potential approach for future resistance management. PLoS Pathog. 2016, 12, e1005434. [Google Scholar] [CrossRef]
- Turley, A.P.; Zalucki, M.P.; O’Neill, S.L.; McGraw, E.A. Transinfected Wolbachia have minimal effects on male reproductive success in Aedes aegypti. Parasites Vectors 2013, 6, 36. [Google Scholar] [CrossRef]
- O’Neill, S.L.; Ryan, P.A.; Turley, A.P.; Wilson, G.; Retzki, K.; Iturbe-Ormaetxe, I.; Dong, Y.; Kenny, N.; Paton, C.J.; Ritchie, S.A.; et al. Scaled deployment of Wolbachia to protect the community from dengue and other Aedes transmitted arboviruses. Gates Open Res. 2019, 2, 36. [Google Scholar] [CrossRef]
- Ford, S.A.; Allen, S.L.; Ohm, J.R.; Sigle, L.T.; Sebastian, A.; Albert, I.; Chenoweth, S.F.; McGraw, E.A. Selection on Aedes aegypti alters Wolbachia-mediated dengue virus blocking and fitness. Nat. Microbiol. 2019, 4, 1832–1839. [Google Scholar] [CrossRef]
- Miller, B.R.; Mitchell, C.J. Genetic selection of a flavivirus-refractory strain of the yellow fever mosquito Aedes aegypti. Am. J. Trop. Med. Hyg. 1991, 45, 399–407. [Google Scholar] [CrossRef]
- Bennett, K.E.; Olson, K.E.; Muñoz Mde, L.; Fernandez-Salas, I.; Farfan-Ale, J.A.; Higgs, S.; Black, W.C., 4th; Beaty, B.J. Variation in vector competence for dengue 2 virus among 24 collections of Aedes aegypti from Mexico and the United States. Am. J. Trop. Med. Hyg. 2002, 67, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Bosio, C.F.; Fulton, R.E.; Salasek, M.L.; Beaty, B.J.; Black, W.C., 4th. Quantitative trait loci that control vector competence for dengue-2 virus in the mosquito Aedes aegypti. Genetics 2000, 156, 687–698. [Google Scholar] [PubMed]
- Lambrechts, L.; Quillery, E.; Noël, V.; Richardson, J.H.; Jarman, R.G.; Scott, T.W.; Chevillon, C. Specificity of resistance to dengue virus isolates is associated with genotypes of the mosquito antiviral gene Dicer-2. Proc. Biol. Sci. 2013, 280, 20122437. [Google Scholar] [CrossRef] [PubMed]
- Blair, C.D.; Olson, K.E. The role of RNA interference (RNAi) in arbovirus-vector interactions. Viruses 2015, 7, 820–843. [Google Scholar] [CrossRef]
- Franz, A.W.E.; Sanchez-Vargas, I.; Adelman, Z.N.; Blair, C.D.; Beaty, B.J.; James, A.A.; Olson, K.E. Engineering RNA interference-based resistance to dengue virus type 2 in genetically modified Aedes aegypti. Proc. Natl. Acad. Sci. USA 2006, 103, 4198–4203. [Google Scholar] [CrossRef]
- Mathur, G.; Sanchez-Vargas, I.; Alvarez, D.; Olson, K.E.; Marinotti, O.; James, A.A. Transgene-mediated suppression of dengue viruses in the salivary glands of the yellow fever mosquito, Aedes aegypti. Insect Mol. Biol. 2010, 19, 753–763. [Google Scholar] [CrossRef]
- Franz, A.W.E.; Sanchez-Vargas, I.; Raban, R.R.; Black, W.C., 4th; James, A.A.; Olson, K.E. Fitness impact and stability of a transgene conferring resistance to dengue-2 virus following introgression into a genetically diverse Aedes aegypti strain. PLoS Negl. Trop. Dis. 2014, 8, e2833. [Google Scholar] [CrossRef]
- Magalhaes, T.; Bergren, N.A.; Bennett, S.L.; Borland, E.M.; Hartman, D.A.; Lymperopoulos, K.; Sayre, R.; Borlee, B.R.; Campbell, C.L.; Foy, B.; et al. Induction of RNA interference to block Zika virus replication and transmission in the mosquito Aedes aegypti. Insect Biochem. Mol. Biol. 2019, 111, 103169. [Google Scholar] [CrossRef]
- Lilley, D.M. Structure, folding, and mechanisms of ribozymes. Curr. Opin. Struct. Biol. 2005, 15, 313–323. [Google Scholar] [CrossRef]
- Nawtaisong, P.; Keith, J.; Fraser, T.; Balaraman, V.; Kolokoltsov, A.; Davey, R.; Higgs, S.; Mohammad, A.; Rongsriyam, Y.; Komalamisra, N.; et al. Effective suppression of dengue fever virus in mosquito cell cultures using retroviral transduction of hammerhead ribozymes targeting the viral genome. Virol. J. 2009, 6, 73. [Google Scholar] [CrossRef]
- Carter, J.R.; Taylor, S.; Fraser, T.S.; Kucharski, C.A.; Dawson, J.L.; Fraser, M.J. Suppression of the arboviruses dengue and Chikungunya using a dual-acting group-I intron coupled with conditional expression of the Bax C-terminal domain. PLoS ONE 2015, 10, e0139899. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.; Furey, C.; Balaraman, V.; Fraser, M.J. Antiviral hammerhead ribozymes are effective for developing transgenic suppression of Chikungunya virus in Aedes aegypti mosquitoes. Viruses 2016, 8, 163. [Google Scholar] [CrossRef] [PubMed]
- Jupatanakul, N.; Sim, S.; Angleró-Rodríguez, Y.I.; Souza-Neto, J.; Das, S.; Poti, K.E.; Rossi, S.L.; Bergren, N.; Vasilakis, N.; Dimopoulos, G. Engineered Aedes aegypti JAK/STAT pathway-mediated immunity to dengue virus. PLoS Negl. Trop. Dis. 2017, 11, e0005187. [Google Scholar] [CrossRef] [PubMed]
- Souza-Neto, J.A.; Sim, A.; Dimopoulos, G. An evolutionary conserved function of the JAK-STAT pathway in anti-dengue defense. Proc. Natl. Acad. Sci. USA 2009, 106, 17841–17846. [Google Scholar] [CrossRef]
- Yen, P.S.; James, A.A.; Li, J.C.; Chen, C.H.; Failloux, A.B. Synthetic miRNAs induce dual arboviral-resistance phenotypes in the vector mosquito Aedes aegypti. Commun. Biol. 2018, 1, 11. [Google Scholar] [CrossRef]
- Buchman, A.; Gamez, S.; Li, M.; Antoshechkin, I.; Li, H.-H.; Wang, H.-W.; Chen, C.-H.; Klein, M.J.; Duchemin, J.-B.; Paradkar, P.N.; et al. Engineered resistance to Zika virus in transgenic Aedes aegypti expressing a polycistronic cluster of synthetic small RNAs. Proc. Natl. Acad. Sci. USA 2019, 116, 3656–3661. [Google Scholar] [CrossRef]
- Buchman, A.; Gamez, S.; Li, M.; Antoshechkin, I.; Lee, S.-H.; Wang, S.-W.; Chen, C.-H.; Klein, M.J.; Duchemin, J.-B.; Crowe, J.E., Jr.; et al. Broad dengue neutralization in mosquitoes expressing an engineered antibody. Cell 2019. [Google Scholar] [CrossRef]
- Smith, S.A.; de Alwis, A.R.; Kose, N.; Harris, E.; Ibarra, K.D.; Kahle, K.M.; Pfaff, J.M.; Xiang, X.; Doranz, B.J.; de Silva, A.M.; et al. The potent and broadly neutralizing human dengue virus-specific monoclonal antibody 1C19 reveals a unique cross-reactive epitope on the bc loop of domain II of the envelope protein. mBio 2013, 4, e00873. [Google Scholar] [CrossRef]
- Tassetto, M.; Kunitomi, M.; Whitfield, Z.J.; Dolan, P.T.; Sanchez-Vargas, I.; Garcia-Knight, M.; Ribiero, I.; Chen, T.; Olson, K.E.; Andino, R. Control of RNA viruses in mosquito cells through the acquisition of vDNA and endogenous viral elements. eLife 2019, 8, e41244. [Google Scholar] [CrossRef]
- Blair, C.D.; Olson, K.E.; Bonizzoni, M. The widespread occurrence and potential biological roles of endogenous viral elements in insect genomes. Curr. Issues Mol. Biol. 2019, 34, 13–30. [Google Scholar] [CrossRef]
- Houe, V.; Bonizzoni, M.; Bailloux, A.B. Endogenous non-retroviral elements in genomes of Aedes mosquitoes and vector competence. Emerg. Microbes Infect. 2019, 8, 542–555. [Google Scholar] [CrossRef] [PubMed]
- Crow, J.F. Unmasking a cheating gene. Science 1999, 283, 1651–1652. [Google Scholar] [CrossRef] [PubMed]
- Hickey, W.A.; Craig, G.B. Genetic distortion of sex ratio in a mosquito, Aedes aegypti. Genetics 1966, 53, 1177–1196. [Google Scholar] [PubMed]
- Craig, G.B., Jr.; Hickey, W.A.; Vandehey, R.C. An inherited male-producing factor in Aedes aegypti. Science 1960, 132, 1887–1889. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.; Behura, S.K.; Severson, D.W. Genome-wide transcriptome profiling reveals genes associated with meiotic drive system of Aedes aegypti. Insects 2019, 10, 25. [Google Scholar] [CrossRef]
- Sinkins, S.P.; Gould, F. Gene drive systems for insect disease vectors. Nat. Rev. Genet. 2006, 7, 427–435. [Google Scholar] [CrossRef]
- Cha, S.-J.; Mori, A.; Chadee, D.D.; Severson, D.W. Cage trials using an endogenous meiotic drive gene in the mosquito Aedes aegypti to promote population replacement. Am. J. Trop. Med. Hyg. 2006, 74, 62–68. [Google Scholar] [CrossRef]
- Werren, J.H. Selfish genetic elements, genetic conflict, and evolutionary innovation. Proc. Natl. Acad. Sci. USA 2011, 108, 10863–10870. [Google Scholar] [CrossRef]
- Burt, A. Site-specific selfish genes as tools for the control and genetic engineering of natural populations. Proc. Biol. Sci. 2003, 270, 921–928. [Google Scholar] [CrossRef]
- Akbari, O.S.; Papathanos, P.A.; Sandler, J.E.; Kennedy, K.; Hay, B.A. Identification of germline transcriptional regulatory elements in Aedes aegypti. Sci. Rep. 2014, 4, 3954. [Google Scholar] [CrossRef]
- Deredec, A.; Burt, A.; Godfray, H.C. The population genetics of using homing endonuclease genes in vector and pest management. Genetics 2008, 179, 2013–2026. [Google Scholar] [CrossRef] [PubMed]
- Burt, A.; Trivers, R. Genes in Conflict: The Biology of Selfish Genetic Elements; Belknap: Cambridge, MA, USA, 2008. [Google Scholar]
- James, A.A. Gene drive systems in mosquitoes: Rules of the road. Trends Parasitol. 2005, 21, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.H.; Huang, H.; Ward, C.M.; Su, J.T.; Schaeffer, L.V.; Guo, M.; Hay, B.A. A synthetic maternal-effect selfish genetic element drives population replacement in Drosophila. Science 2007, 316, 597–600. [Google Scholar] [CrossRef]
- Ward, C.M.; Su, J.T.; Huang, Y.; Lloyd, A.L.; Gould, F.; Hay, B.A. Medea selfish genetic elements as tools for altering traits of wild populations: A theoretical analysis. Evolution 2011, 65, 1149–1162. [Google Scholar] [CrossRef] [PubMed]
- Morris, A.C.; Eggleston, P.; Crampton, J.M. Genetic transformation of the mosquito Aedes aegypti by micro-injection of DNA. Med. Vet. Entomol. 1989, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kokoza, V.; Ahmed, A.; Wimmer, E.A.; Raikhel, A.S. Efficient transformation of the yellow fever mosquito Aedes aegypti using the piggyBac transposable element vector pBac[3xP3-EGFP afm]. Insect Biochem. Mol. Biol. 2001, 31, 1137–1143. [Google Scholar] [CrossRef]
- Cary, L.C.; Goebel, M.; Corsaro, B.G.; Wang, H.G.; Rosen, E.; Fraser, M.J. Transposon mutagenesis of baculoviruses: Analysis of Trichoplusia ni transposon IFP2 insertions within the FP-locus of nuclear polyhedrosis viruses. Virology 1989, 172, 156–169. [Google Scholar] [CrossRef]
- Jasinskiene, N.; Coates, C.J.; Benedict, M.Q.; Cornel, A.J.; Rafferty, C.S.; James, A.A.; Collins, F.H. Stable transformation of the yellow fever mosquito, Aedes aegypti, with the Hermes element from the housefly. Proc. Natl. Acad. Sci. USA 1998, 95, 3743–3747. [Google Scholar] [CrossRef]
- Warren, W.D.; Atkinson, P.W.; O’Brochta, D.A. The Hermes transposable element from the house fly, Musca domestica, is a short inverted repeat-type element of the hobo, Ac, and Tam3 (hAT) element family. Genet. Res. 1994, 64, 87–97. [Google Scholar] [CrossRef]
- Coates, C.J.; Jasinskiene, N.; Miyashiro, L.; James, A.A. Mariner transposition and transformation of the yellow fever mosquito, Aedes aegypti. Proc. Natl. Acad. Sci. USA 1998, 95, 3748–3751. [Google Scholar] [CrossRef]
- Medhora, M.; Maruyama, K.; Hartl, D.L. Molecular and functional analysis of the mariner mutator element Mos1 in Drosophila. Genetics 1991, 128, 311–318. [Google Scholar] [PubMed]
- Adelman, Z.N.; Jasinskiene, N.; James, A.A. Development and applications of transgenesis in the yellow fever mosquito, Aedes aegypti. Mol. Biochem. Parasitol. 2002, 121, 1–10. [Google Scholar] [CrossRef]
- Criscione, F.; O’Brochta, D.A.; Reid, W. Genetic technologies for disease vectors. Curr. Opin. Insect Sci. 2015, 10, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Sethuraman, N.; Fraser, M.J.; Eggleston, P.; O’Brochta, D.A. Post-integration stability of piggyBac in Aedes aegypti. Insect Biochem. Mol. Biol. 2008, 37, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Aryan, A.; Anderson, M.A.E.; Myles, K.M.; Adelman, Z.N. Germline excision of transgenes in Aedes aegypti by homing endonucleases. Sci. Rep. 2013, 3, 1603. [Google Scholar] [CrossRef]
- Traver, B.E.; Anderson, M.A.; Adelman, Z.N. Homing endonucleases catalyze double-stranded DNA breaks and somatic transgene excision in Aedes aegypti. Insect Mol. Biol. 2009, 18, 623–633. [Google Scholar] [CrossRef]
- Jacquier, A.; Dujon, B. An intron-encoded protein is active in a gene conversion process that spreads an intron into a mitochondrial gene. Cell 1985, 41, 383–394. [Google Scholar] [CrossRef]
- Windbichler, N.; Papathanos, P.A.; Catteruccia, F.; Ranson, H.; Burt, A.; Crisanti, A. Homing endonuclease mediated gene targeting in Anopheles gambiae cells and embryos. Nucleic Acids Res. 2007, 35, 5922–5933. [Google Scholar] [CrossRef]
- Windbichler, N.; Menichelli, M.; Papathanos, P.A.; Thyme, S.B.; Li, H.; Ulge, U.Y.; Hovde, B.T.; Baker, D.; Monnat, R.J., Jr.; Burt, A.; et al. A synthetic homing endonuclease-based gene drive system in the human malaria mosquito. Nature 2011, 473, 212–215. [Google Scholar] [CrossRef]
- Liu, Q.; Segal, D.J.; Ghiara, J.B.; Barbas, C.F. Design of polydactyl zinc-finger proteins for unique addressing within complex genomes. Proc. Natl. Acad. Sci. USA 1997, 94, 5525–5530. [Google Scholar] [CrossRef]
- Christian, M.; Cermak, T.; Doyle, E.L.; Schmidt, C.; Zhang, F.; Hummel, A.; Bogdanove, A.J.; Voytas, D.F. Targeting DNA double-strand breaks with TAL effector nucleases. Genetics 2010, 186, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A programmable dual-RNA–guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Lin, J.; Held, N.L.; Clem, R.J.; Passarelli, A.L.; Franz, A.W.E. Heritable CRISPR/Cas9-mediated genome editing in the yellow fever mosquito, Aedes aegypti. PLoS ONE 2015, 10, e0122353. [Google Scholar] [CrossRef] [PubMed]
- Kistler, K.E.; Vosshall, L.B.; Matthews, B.J. Genome engineering with CRISPR-Cas9 in the mosquito Aedes aegypti. Cell Rep. 2015, 11, 51–60. [Google Scholar] [CrossRef]
- Basu, S.; Aryan, A.; Overcash, J.M.; Samuel, G.H.; Anderson, M.A.E.; Dahlem, T.J.; Myles, K.M.; Adelman, Z.N. Silencing of end-joining repair for efficient site-specific gene insertion after TALEN/CRISPR mutagenesis in Aedes aegypti. Proc. Natl. Acad. Sci. USA 2015, 112, 4038–4043. [Google Scholar] [CrossRef]
- Gantz, V.M.; Jasinskiene, N.; Tatarenkova, O.; Fazekas, A.; Macias, V.M.; Bier, E.; James, A.A. Highly efficient Cas9-mediated gene drive for population modification of the malaria vector mosquito Anopheles stephensi. Proc. Natl. Acad. Sci. USA 2015, 112, 6736–6743. [Google Scholar] [CrossRef]
- Konet, D.S.; Anderson, J.; Piper, J.; Akkina, R.; Suchman, E.; Carlson, J. Short-hairpin RNA expressed from polymerase III promoters mediates RNA interference in mosquito cells. Insect Mol. Biol. 2007, 16, 199–206. [Google Scholar] [CrossRef]
- Dang, Y.; Jia, G.; Choi, J.; Ma, H.; Anaya, E.; Ye, C.; Shankar, P.; Wu, H. Optimizing sgRNA structure to improve CRISPR-Cas9 knockout efficiency. Genome Biol. 2015, 16, 280. [Google Scholar] [CrossRef]
- Li, M.; Yang, T.; Kandul, N.P.; Bui, M.; Gamez, S.; Raban, R.; Bennett, J.; Sanchez, C.H.M.; Lanzaro, G.C.; Schmidt, H.; et al. Development of a Confinable Gene-Drive System in the Human Disease Vector, Aedes aegypti. bioRxiv 2019. [Google Scholar] [CrossRef]
- Drury, D.W.; Dapper, A.L.; Siniard, D.J.; Zentner, G.E.; Wade, M.J. CRISPR/Cas9 gene drives in genetically variable and nonrandomly mating wild populations. Sci. Adv. 2017, 3, e1601910. [Google Scholar] [CrossRef]
- Li, M.; Bui, M.; Yang, T.; Bowman, C.S.; White, B.J.; Akbari, O.S. Germline Cas9 expression yields highly efficient genome engineering in a major worldwide disease vector, Aedes aegypti. Proc. Natl. Acad. Sci. USA 2017, 114, 10540–10549. [Google Scholar] [CrossRef] [PubMed]
- Champer, J.; Liu, J.; Oh, S.Y.; Reeves, R.; Luthra, A.; Oakes, N.; Clark, A.G.; Messer, P.W. Reducing resistance allele formation in CRISPR gene drive. Proc. Natl. Acad. Sci. USA 2018, 115, 5522–5527. [Google Scholar] [CrossRef] [PubMed]
- Bier, E.; Harrison, M.M.; O’Connor-Giles, K.M.; Wildonger, J. Advances in engineering the fly genome with the CRISPR-Cas System. Genetics 2018, 208, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Magori, K.; Legros, M.; Puente, M.E.; Focks, D.A.; Scott, T.W.; Lloyd, A.L.; Gould, F. Skeeter buster: A stochastic, spatially explicit modeling tool for studying Aedes aegypti population replacement and population suppression strategies. PLoS Negl. Trop. Dis. 2009, 3, e508. [Google Scholar] [CrossRef]
- Haller, B.C.; Messer, P.W. SLiM 2: Flexible, interactive forward genetic simulations. Mol. Biol. Evol. 2017, 34, 230–240. [Google Scholar] [CrossRef]
- Haller, B.C.; Messer, P.W. SLiM3: Forward genetic simulations beyond the Wright-Fisher model. Mol. Biol. Evol. 2019, 36, 632–637. [Google Scholar] [CrossRef]
- Sanchez, C.H.M.; Wu, S.L.; Bennett, J.B.; Marshall, J.M. MGDrivE: A modular simulation framework for the spread of gene drives through spatially-explicit mosquito populations. Methods Ecol. Evol. 2019. [Google Scholar] [CrossRef]
- Hammond, A.M.; Kyrou, K.; Bruttini, M.; North, A.; Galizi, R.; Karlsson, X.; Kranjc, N.; Carpi, F.M.; D’Aurizio, R.; Crisanti, A.; et al. The creation and selection of mutations resistant to a gene drive over multiple generations in the malaria mosquito. PLoS Genet. 2017, 13, e1007039. [Google Scholar] [CrossRef]
- Adelman, Z.; Akbari, O.; Bauer, J.; Bier, E.; Bloss, C.; Carter, S.R.; Callender, C.; Denis, A.C.; Cowhey, P.; Dass, B.; et al. Rules of the road for insect gene drive research and testing. Nat. Biotechnol. 2017, 35, 716–718. [Google Scholar] [CrossRef]
- Benedict, M.Q.; Burt, A.; Capurro, M.L.; De Barro, P.; Handler, A.M.; Hayes, K.R.; Marshall, J.M.; Tabachnick, W.J.; Adelman, Z.N. Recommendations for laboratory containment and management of gene drive systems in arthropods. Vector Borne Zoonotic Dis. 2018, 18, 2–13. [Google Scholar] [CrossRef]
- Adelman, Z.N.; Pledger, D.; Myles, K.M. Developing standard operating procedures for gene drive research in disease vector mosquitoes. Pathog. Glob. Health 2018, 111, 436–447. [Google Scholar] [CrossRef] [PubMed]
| Study | Transgenic Strategy | Virus Targeted | Method | Promoter | Prevalence of Disseminated Infections in Transgenics |
|---|---|---|---|---|---|
| Franz et al. 2006 [25] | † IR triggering siRNA antiviral pathway | DENV2 | mariner Mos1 | ††AeCPA | 0% (14 dpi §) |
| Franz et al. 2014 [27] | |||||
| Mathur et al. 2010 [26] | IR triggering siRNA antiviral pathway | DENV2 | mariner Mos1 | ‡Ae30K b | 0% (saliva, 14 dpi) |
| Mishra et al. 2016 [32] | Antiviral hammerhead ribozymes | CHIKV 181/25 | piggyBac | ‡‡AetRNAval Pol III | 0% (7 dpi) |
| Jupatanakul et al. 2017 [33] | Overexpression of components in the JAK/STAT pathway | DENV2/4 | piggyBac | * AeVg1 | ≥43% (14 dpi) |
| Yen et al. 2018 [35] | Synthetic RNAs targeting virus genome, triggering miRNA antiviral immunity | DENV3/CHIKV | mariner Mos1 | ** AePUb/AeCPA | ~10% (DENV3, 21 dpi) |
| ~10–50% (CHIKV, 6 dpi) | |||||
| Buchman et al. 2019 [36] | Synthetic RNAs targeting virus genome, triggering miRNA antiviral immunity | ZIKV | piggyBac | AeCPA | 0% (14 dpi) |
| Buchman et al. 2019 [37] | Broadly neutralizing single chain antibody | DENV1-4 | piggyBac | AeCPA | 0% (14 dpi) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Williams, A.E.; Franz, A.W.E.; Reid, W.R.; Olson, K.E. Antiviral Effectors and Gene Drive Strategies for Mosquito Population Suppression or Replacement to Mitigate Arbovirus Transmission by Aedes aegypti. Insects 2020, 11, 52. https://doi.org/10.3390/insects11010052
Williams AE, Franz AWE, Reid WR, Olson KE. Antiviral Effectors and Gene Drive Strategies for Mosquito Population Suppression or Replacement to Mitigate Arbovirus Transmission by Aedes aegypti. Insects. 2020; 11(1):52. https://doi.org/10.3390/insects11010052
Chicago/Turabian StyleWilliams, Adeline E., Alexander W. E. Franz, William R. Reid, and Ken E. Olson. 2020. "Antiviral Effectors and Gene Drive Strategies for Mosquito Population Suppression or Replacement to Mitigate Arbovirus Transmission by Aedes aegypti" Insects 11, no. 1: 52. https://doi.org/10.3390/insects11010052
APA StyleWilliams, A. E., Franz, A. W. E., Reid, W. R., & Olson, K. E. (2020). Antiviral Effectors and Gene Drive Strategies for Mosquito Population Suppression or Replacement to Mitigate Arbovirus Transmission by Aedes aegypti. Insects, 11(1), 52. https://doi.org/10.3390/insects11010052




