Parthenogenetic Females of the Stick Insect Clitarchus hookeri Maintain Sexual Traits
Abstract
1. Introduction
2. Materials and Methods
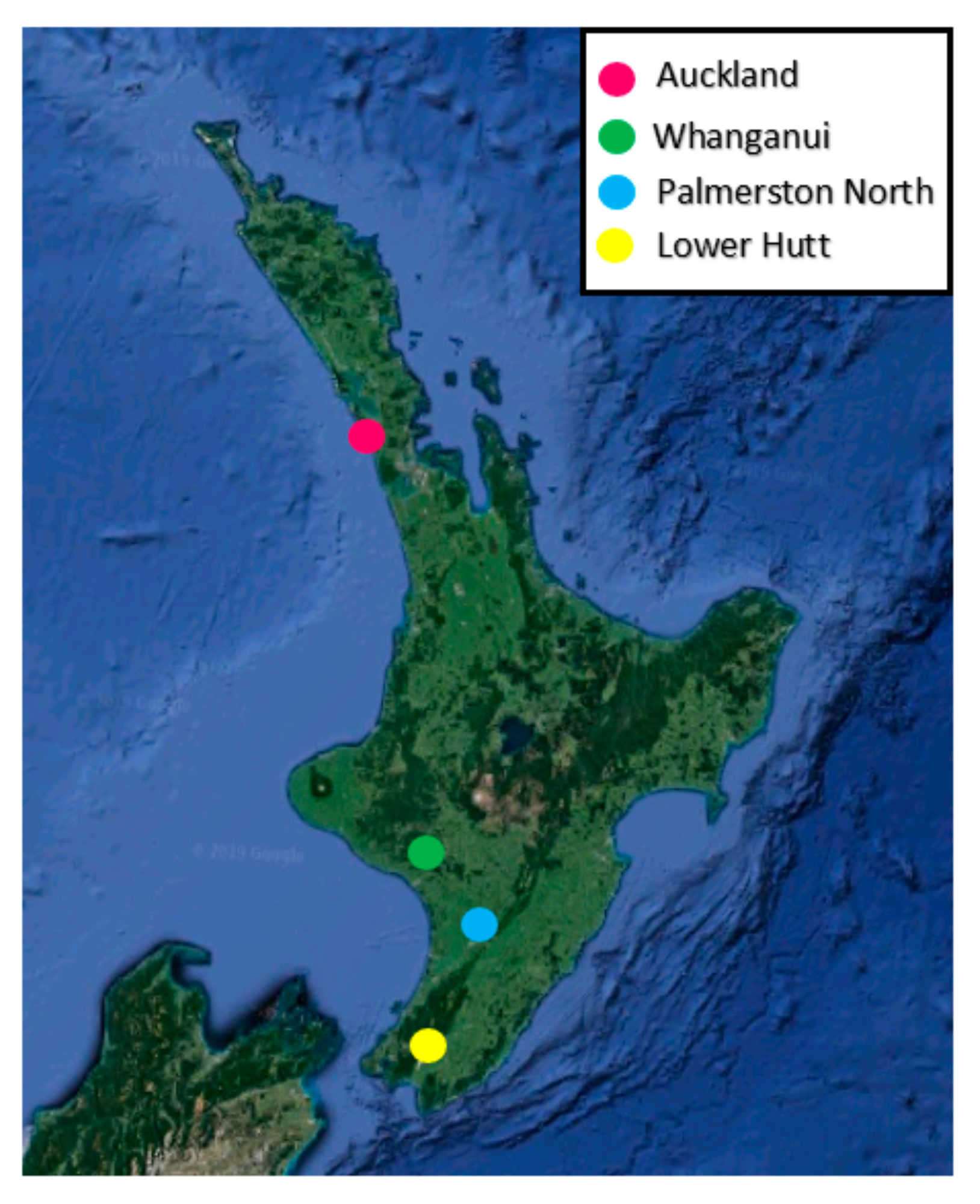
2.1. Sampling Location and Insect Husbandry
2.2. Morphological Analysis
2.3. Chemical Analysis
2.4. Male Choice
2.5. Data Analysis
3. Results
3.1. Males vs. Females
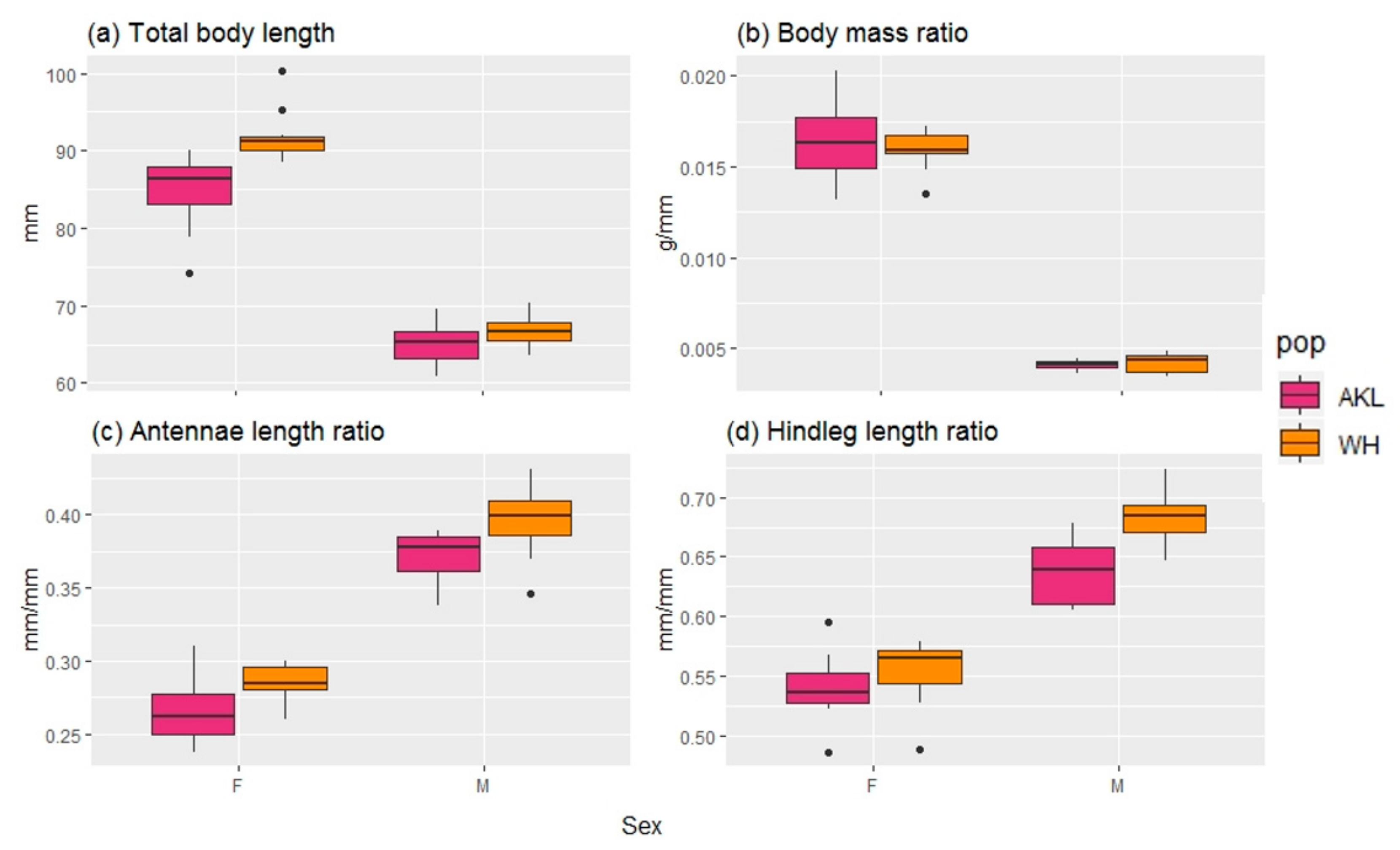
3.1.1. Morphological Analysis
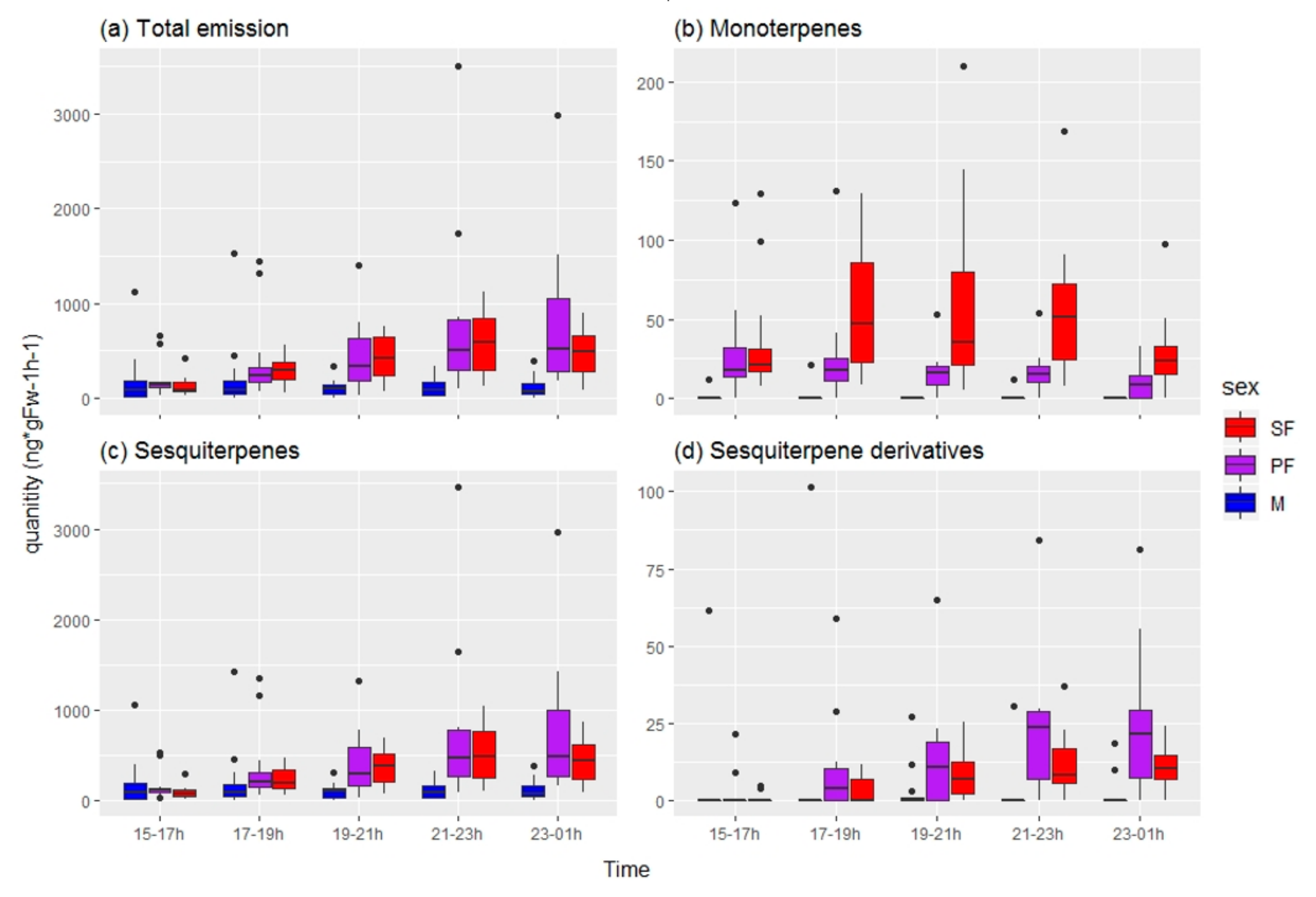
3.1.2. Chemical Analysis
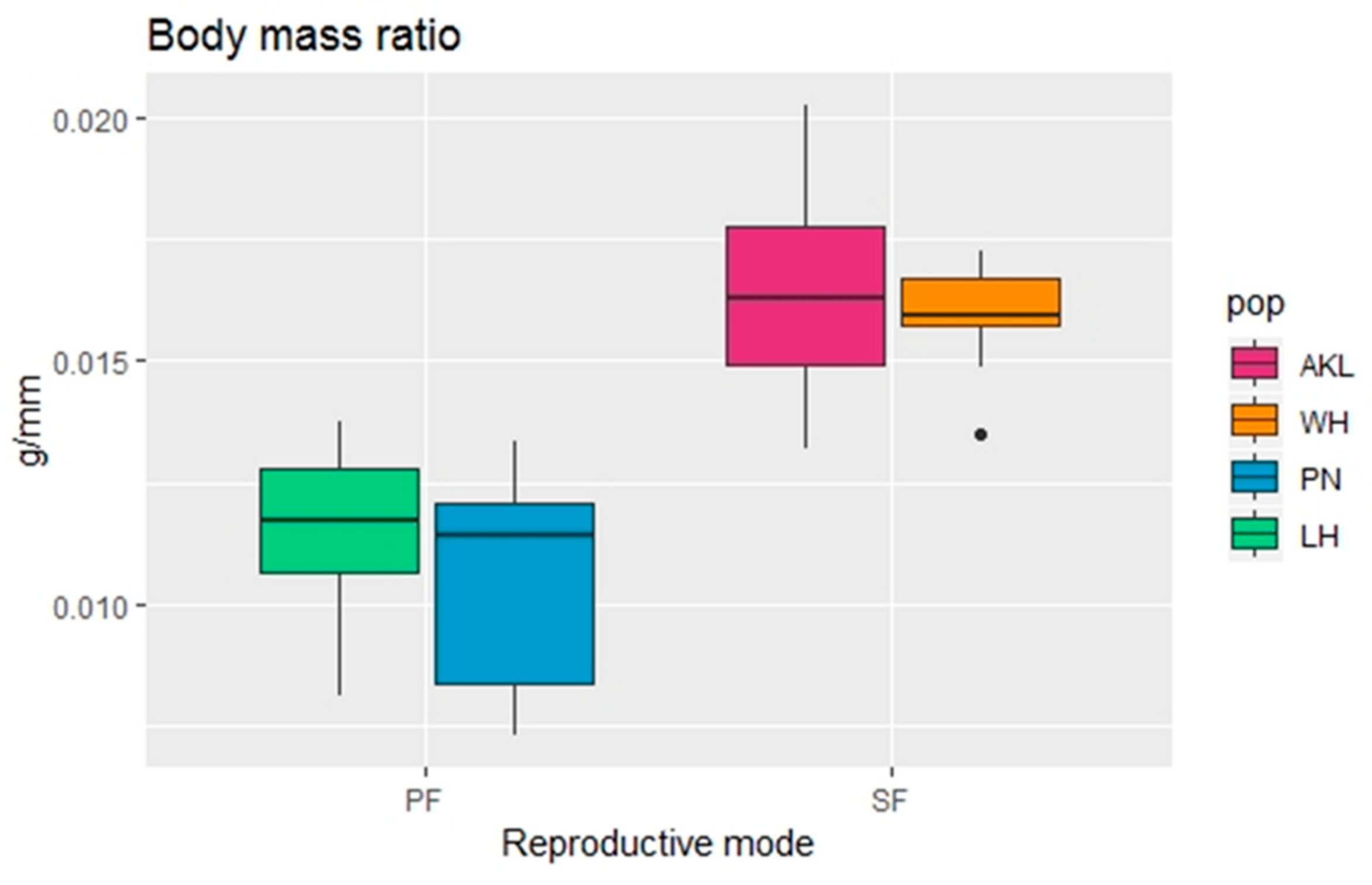
3.2. Sexual vs. Parthenogenetic Females
3.2.1. Morphological Analysis
3.2.2. Chemical Analysis
3.3. Male Choice
3.3.1. Copulatory Behaviors
3.3.2. Male Choice
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| RT | Compound | Chemical Class |
|---|---|---|
| 7.2 | α-Phellandrene | Monoterpenes |
| 7.5 | α-Pinene | Monoterpenes |
| 8.7 | Sabinene | Monoterpenes |
| 8.8 | β-Pinene | Monoterpenes |
| 9.34 | β-Myrcene | Monoterpenes |
| 11.19 | α-Ocimene | Monoterpenes |
| 11.5 | γ-Terpinene | Monoterpenes |
| 17.15 | α-Cubebene | Sesquiterpenes |
| 17.4 | Ylangene | Sesquiterpenes |
| 17.54 | Copaene | Sesquiterpenes |
| 17.63 | β-Elemene | Sesquiterpenes |
| 17.73 | γ-Elemene | Sesquiterpenes |
| 18.02 | α-Gurjunene | Sesquiterpenes |
| 18.16 | β-Caryophyllene | Sesquiterpenes |
| 18.4 | Aromandrene | Sesquiterpenes |
| 18.5 | Isoledene | Sesquiterpenes |
| 18.6 | α-Caryophyllene | Sesquiterpenes |
| 18.8 | Cadina-1(10),4diene | Sesquiterpenes |
| 18.93 | GermacreneD | Sesquiterpenes |
| 19.05 | Eudesma-4(14),11-diene | Sesquiterpenes |
| 19.12 | α-Seline | Sesquiterpenes |
| 19.24 | Butylated Hydroxytoluene | Sesquiterpenes |
| 19.43 | Calamenene | Sesquiterpene derivatives |
| 19.53 | Cadine-1,4-diene | Sesquiterpenes |
References
- Dimijian, G.G. Evolution of sexuality: Biology and behavior. Baylor Univ. Med. Cent. Proc. 2005, 18, 244–258. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schwander, T.; Vuilleumier, S.; Dubman, J.; Crespi, B.J. Positive feedback in the transition from sexual reproduction to parthenogenesis. Proc. R. Soc. B Biol. Sci. 2010, 277, 1435–1442. [Google Scholar] [CrossRef] [PubMed]
- Otto, S.P. The evolutionary enigma of sex. Am. Nat. 2009, 174, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Normark, B.B.; Kirkendall, L.R. Parthenogenesis in Insects and Mites. In Encyclopedia of Insects, 2nd ed.; Resh, V.H., Cardé, R.T., Eds.; Elsevier Inc.: Berkeley, CA, USA, 2009; pp. 753–757. [Google Scholar]
- Butlin, R. The costs and benefits of sex: New insights from old asexual lineages. Nat. Rev. Genet. 2002, 3, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Scali, V. Metasexual stick insects: Model pathways to losing sex and bringing it back. In Lost Sex: The Evolutionary Biology of Parthenogenesis; Schön, I., Martens, K., van Dijk, P., Eds.; Springer: Dordrecht, The Netherlands; New York, NY, USA, 2009; pp. 317–345. [Google Scholar]
- Scali, V.; Passamonti, M.; Marescalchi, O.; Mantovani, B. Linkage between sexual and asexual lineages: Genome evolution in Bacillus stick insects. Biol. J. Linn. Soc. 2003, 79, 137–150. [Google Scholar] [CrossRef]
- Morgan-Richards, M.; Trewick, S.A.; Stringer, I.A.N. Geographic parthenogenesis and the common tea-tree stick insect of New Zealand. Mol. Ecol. 2010, 19, 1227–1238. [Google Scholar] [CrossRef] [PubMed]
- Morgan-Richards, M.; Langton-Myers, S.S.; Trewick, S.A. Loss and gain of sexual reproduction in the same stick insect. Mol. Ecol. (under review).
- Buckley, T.R.; Marske, K.; Attanayake, D. Phylogeography and ecological niche modelling of the New Zealand stick insect Clitarchus hookeri (White) support survival in multiple coastal refugia. J. Biogeogr. 2010, 37, 682–695. [Google Scholar] [CrossRef]
- Law, J.H.; Crespi, B.J. The evolution of geographic parthenogenesis in Timema walking-sticks. Mol. Ecol. 2002, 11, 1471–1489. [Google Scholar] [CrossRef]
- Peck, J.R.; Yearsley, J.M.; Waxman, D. Explaining the geographic distributions of sexual and asexual populations. Nature 1998, 391, 889–892. [Google Scholar] [CrossRef]
- Trewick, S.A.; Goldberg, J.; Morgan-Richards, M. Fewer species of Argosarchus and Clitarchus stick insects (Phasmida, Phasmatinae): Evidence from nuclear and mitochondrial DNA sequence data. Zool. Scr. 2005, 34, 483–491. [Google Scholar] [CrossRef]
- Edward, D.A.; Chapman, T. The evolution and significance of male mate choice. Trends Ecol. Evol. 2011, 26, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Bonduriansky, R. The evolution of male mate choice in insects: a synthesis of ideas and evidence. Biol. Rev. Camb. Philos. Soc. 2001, 76, 305–339. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.D. Sexual selection, phenotypic variation, and allometry in genitalic and non-genitalic traits in the sexually size-dimorphic stick insect Micrarchus hystriculeus. Biol. J. Linn. Soc. 2014, 113, 471–484. [Google Scholar] [CrossRef]
- Kelly, C.D.; Bussière, L.F.; Gwynne, D.T. Sexual selection for male mobility in a giant insect with female-biased size dimorphism. Am. Nat. 2008, 172, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Holwell, G.I.; Barry, K.L.; Herberstein, M.E. Mate location, antennal morphology, and ecology in two praying mantids (Insecta: Mantodea). Biol. J. Linn. Soc. 2007, 91, 307–313. [Google Scholar] [CrossRef]
- Baena, M.L.; Macías-Ordóñez, R. Mobility and mating frequency in the scramble competition polygyny of a chrysomelid beetle. Behav. Ecol. 2014, 26, 416–424. [Google Scholar] [CrossRef]
- Myers, S.S.; Buckley, T.R.; Holwell, G.I. Mate detection and seasonal variation in stick insect mating behaviour (Phamatodea: Clitarchus hookeri). Behaviour 2015, 152, 1325–1348. [Google Scholar] [CrossRef]
- Kokko, H.; Wong, B.B.M. What determines sex roles in mate searching? Evolution 2007, 61, 1162–1175. [Google Scholar] [CrossRef]
- Herberstein, M.E.; Painting, C.J.; Holwell, G.I. Scramble competition polygyny in terrestrial arthropods. In Advances in the Study of Behavior; Elsevier Ltd.: Amstedam, The Netherlands, 2017; Volume 49, pp. 237–295. [Google Scholar]
- Blanckenhorn, W.U. Behavioral causes and consequences of sexual size dimorphism. Ethology 2005, 111, 977–1016. [Google Scholar] [CrossRef]
- Tabata, J.; Ichiki, R.T.; Moromizato, C.; Mori, K. Sex pheromone of a coccoid insect with sexual and asexual lineages: Fate of an ancestrally essential sexual signal in parthenogenetic females. J. R. Soc. Interf. 2017, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Carson, H.L.; Chang, L.S.; Lyttle, T.W. Decay of female sexual behavior under parthenogenesis. Science 1982, 218, 68–70. [Google Scholar] [CrossRef]
- Burke, N.W.; Crean, A.J.; Bonduriansky, R. The role of sexual conflict in the evolution of facultative parthenogenesis: A study on the spiny leaf stick insect. Anim. Behav. 2015, 101, 117–127. [Google Scholar] [CrossRef]
- Van der Kooi, C.J.; Schwander, T. On the fate of sexual traits under asexuality. Biol. Rev. Camb. Philos. Soc. 2014, 89, 805–819. [Google Scholar] [CrossRef] [PubMed]
- Schwander, T.; Crespi, B.J.; Gries, R.; Gries, G. Neutral and selection-driven decay of sexual traits in asexual stick insects. Proc. R. Soc. B Biol. Sci. 2013, 280, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, G.U.C.; Siozios, S.; Bourtzis, K.; Reinhold, K.; Lehmann, A.W. Thelytokous parthenogenesis and the heterogeneous decay of mating behaviours in a bushcricket (Orthopterida). J. Zool. Syst. Evol. Res. 2011, 49, 102–109. [Google Scholar] [CrossRef]
- Stringer, I.A.N. The nymphal and imaginal stages of the bisexual stick insect Clitarchus hookeri (Phasmidae: Phasminae). N. Z. Entomol. 1970, 4, 85–95. [Google Scholar] [CrossRef]
- Perry, N.B.; Brennan, N.J.; Klink, J.W.V.; Harris, W.; Douglas, M.H.; Mcgimpsey, J.A.; Smallfield, B.M.; Anderson, R.E. Essential oils from New Zealand manuka and kanuka: Chemotaxonomy of Leptospermum. Phytochemistry 1997, 44, 1485–1494. [Google Scholar] [CrossRef]
- Van Vuuren, S.F.; Docrat, Y.; Kamatou, G.P.P.; Viljoen, A.M. Essential oil composition and antimicrobial interactions of understudied tea tree species. South Afr. J. Bot. 2014, 92, 7–14. [Google Scholar] [CrossRef]
- Douglas, M.H.; van Klink, J.W.; Smallfield, B.M.; Perry, N.B.; Anderson, R.E.; Johnstone, P.; Weavers, R.T. Essential oils from New Zealand manuka: Triketone and other chemotypes of Leptospermum scoparium. Phytochemistry 2004, 65, 1255–1264. [Google Scholar] [CrossRef]
- Akhila, A. Metabolic engineering of biosynthetic pathways leading to isoprenoids: Mono- and sesquiterpenes in plastids and cytosol. J. Plant Interact. 2007, 2, 195–204. [Google Scholar] [CrossRef]
- Dudareva, N.; Andersson, S.; Orlova, I.; Gatto, N.; Reichelt, M.; Rhodes, D.; Boland, W.; Gershenzon, J. The nonmevalonate pathway supports both monoterpene and sesquiterpene formation in snapdragon flowers. Proc. Natl. Acad. Sci. USA 2005, 102, 933–938. [Google Scholar] [CrossRef] [PubMed]
- McKinney, R.M.; Vernier, C.; Ben-Shahar, Y. The neural basis for insect pheromonal communication. Curr. Opin. Insect Sci. 2015, 12, 86–92. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rouger, R.; Reichel, K.; Malrieu, F.; Masson, J.P.; Stoeckel, S. Effects of complex life cycles on genetic diversity: Cyclical parthenogenesis. Heredity 2016, 117, 336–347. [Google Scholar] [CrossRef]
- Allen, D.E.; Lynch, M. The effect of variable frequency of sexual reproduction on the genetic structure of natural populations of a cyclical parthenogen. Evolution 2012, 66, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Hochkirch, A.; Gröning, J. Sexual size dimorphism in Orthoptera—A review. J. Orthoptera Res. 2008, 17, 189–196. [Google Scholar] [CrossRef]
- Arbuthnott, D.; Crespi, B.J. Courtship and mate discrimination within and between species of Timema walking-sticks. Anim. Behav. 2009, 78, 53–59. [Google Scholar] [CrossRef]
- Lehmann, G.U.C.; Strauß, J.; Lakes-Harlan, R. Listening when there is no sexual signalling? Maintenance of hearing in the asexual bushcricket Poecilimon intermedius. J. Comp. Physiol. A Neuroethol. Sens. Neural. Behav. Physiol. 2007, 193, 537–545. [Google Scholar] [CrossRef]

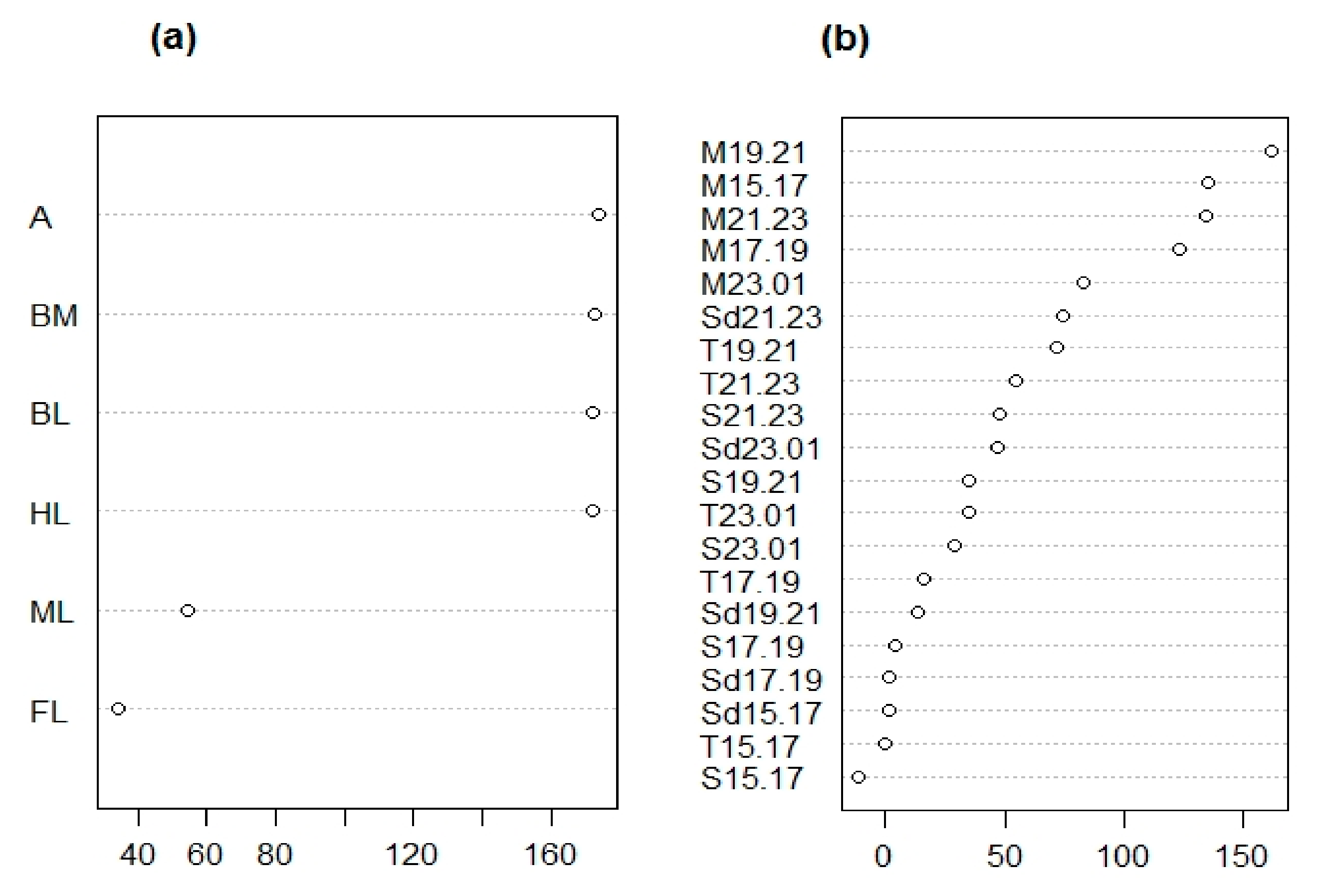
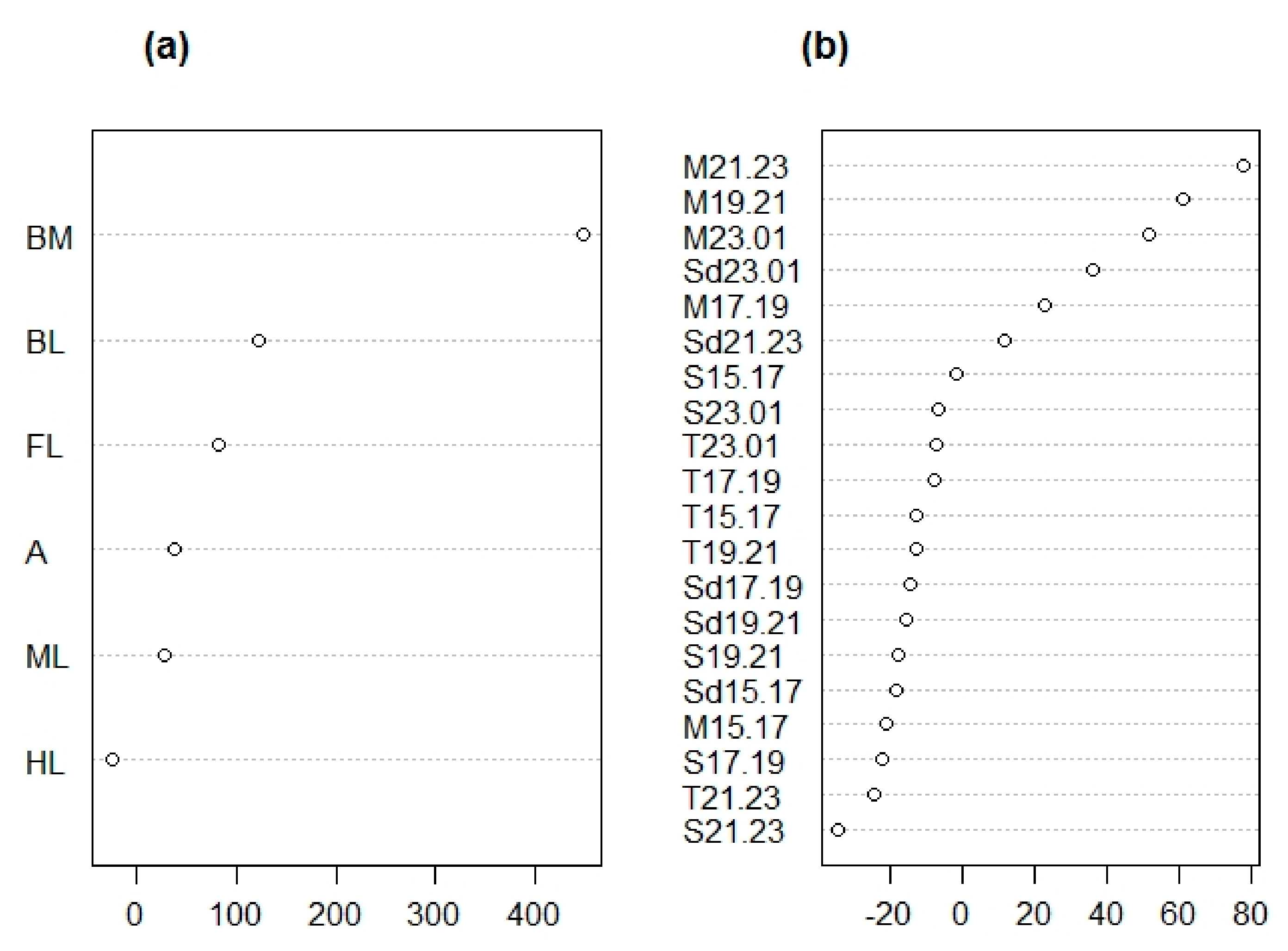
| Trait ID | Trait Description | Measure | Type of Trait |
|---|---|---|---|
| BL | Total Body length | mm | Morphological |
| BM | Body mass ratio | g/mm | Morphological |
| A | Antennae length ratio | mm/mm | Morphological |
| FL | Front leg length ratio | mm/mm | Morphological |
| ML | Mid leg ratio | mm/mm | Morphological |
| HL | Hind leg ratio | mm/mm | Morphological |
| M15.17 | Monoterpene emission 15–17h | ng*gFw−1h−1 | Chemical |
| M17.19 | Monoterpene emission 17–19h | ng*gFw−1h−1 | Chemical |
| M19.21 | Monoterpene emission 19–21h | ng*gFw−1h−1 | Chemical |
| M21.23 | Monoterpene emission 21–23h | ng*gFw−1h−1 | Chemical |
| M23.01 | Monoterpene emission 23–01h | ng*gFw−1h−1 | Chemical |
| S15.17 | Sesquiterpene emission 15–17h | ng*gFw−1h−1 | Chemical |
| S17.19 | Sesquiterpene emission 17–19h | ng*gFw−1h−1 | Chemical |
| S19.21 | Sesquiterpene emission 19–21h | ng*gFw−1h−1 | Chemical |
| S21.23 | Sesquiterpene emission 21–23h | ng*gFw−1h−1 | Chemical |
| S23.01 | Sesquiterpene emission 23–01h | ng*gFw−1h−1 | Chemical |
| Sd15.17 | Sesquiterpene derivative emission 15–17h | ng*gFw−1h−1 | Chemical |
| Sd17.19 | Sesquiterpene derivative emission 17–19h | ng*gFw−1h−1 | Chemical |
| Sd19.21 | Sesquiterpene derivative emission 19–21h | ng*gFw−1h−1 | Chemical |
| Sd21.23 | Sesquiterpene derivative emission 21–23h | ng*gFw−1h−1 | Chemical |
| Sd23.01 | Sesquiterpene derivative emission 23–01h | ng*gFw−1h−1 | Chemical |
| T15.17 | Total emission 15–17h | ng*gFw−1h−1 | Chemical |
| T17.19 | Total emission 17–19h | ng*gFw−1h−1 | Chemical |
| T19.21 | Total emission 19–21h | ng*gFw−1h−1 | Chemical |
| T21.23 | Total emission 21–23h | ng*gFw−1h−1 | Chemical |
| T23.01 | Total emission 23–01h | ng*gFw−1h−1 | Chemical |
| Trait ID | p | Males | Females | ||
|---|---|---|---|---|---|
| AKL | WH | AKL | WH | ||
| BL | <0.001 | a | a | b | c |
| BM | <0.001 | a | a | b | b |
| A | <0.001 | a | b | c | d |
| HL | <0.001 | a | b | c | c |
| M15.17 | <0.001 | a | a | b | b |
| M17.19 | <0.001 | a | a | b | b |
| M19.21 | <0.001 | a | a | b | b |
| M21.23 | <0.001 | a | a | b | b |
| Trait ID | p | Sexual | Parthenogenetic | ||
|---|---|---|---|---|---|
| AKL | WH | PN | LH | ||
| BM | <0.001 | a | a | b | b |
| M19.21 | 0.02 | a,b | a | a,b | b |
| M21.23 | 0.08 | ||||
| M23.01 | 0.25 | ||||
| Sd23.01 | 1 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakano, M.; Morgan-Richards, M.; Godfrey, A.J.R.; Clavijo McCormick, A. Parthenogenetic Females of the Stick Insect Clitarchus hookeri Maintain Sexual Traits. Insects 2019, 10, 202. https://doi.org/10.3390/insects10070202
Nakano M, Morgan-Richards M, Godfrey AJR, Clavijo McCormick A. Parthenogenetic Females of the Stick Insect Clitarchus hookeri Maintain Sexual Traits. Insects. 2019; 10(7):202. https://doi.org/10.3390/insects10070202
Chicago/Turabian StyleNakano, Mari, Mary Morgan-Richards, A. Jonathan R. Godfrey, and Andrea Clavijo McCormick. 2019. "Parthenogenetic Females of the Stick Insect Clitarchus hookeri Maintain Sexual Traits" Insects 10, no. 7: 202. https://doi.org/10.3390/insects10070202
APA StyleNakano, M., Morgan-Richards, M., Godfrey, A. J. R., & Clavijo McCormick, A. (2019). Parthenogenetic Females of the Stick Insect Clitarchus hookeri Maintain Sexual Traits. Insects, 10(7), 202. https://doi.org/10.3390/insects10070202






