Abstract
Climate change is a major environmental concern and is directly related to the increasing concentrations of greenhouse gases. The increase in concentrations of atmospheric carbon dioxide (CO2), not only affects plant growth and development, but also affects the emission of plant organic volatile compounds (VOCs). Changes in the plant odor profile may affect the plant-insect interactions, especially the behavior of herbivorous insects. In this study, we compared the foraging behavior of corn leaf aphid (Rhopalosiphum maidis) on barley (Hordeum vulgare L.) seedlings grown under contrasted CO2 concentrations. During the dual choice bioassays, the winged and wingless aphids were more attracted by the VOCs of barley seedlings cultivated under ambient CO2 concentrations (aCO2; 450 ppm) than barley seedlings cultivated under elevated CO2 concentrations (eCO2; 800 ppm), nymphs were not attracted by the VOCs of eCO2 barley seedlings. Then, volatile compositions from 14-d-old aCO2 and eCO2 barley seedlings were investigated by GC-MS. While 16 VOCs were identified from aCO2 barley seedlings, only 9 VOCs were found from eCO2 barley seedlings. At last, we discussed the potential role of these chemicals observed during choice bioassays. Our findings lay foundation for functional response of corn leaf aphid under climate change through host plant modifications.
1. Introduction
Since the industrial revolution, the atmospheric concentration of carbon dioxide (CO2) has been steadily rising from approximately 280 ppm to 401 ppm (Mauna Loa Observatory: NOAA-ESRL) worldwide. Forecasts suggest that the concentrations could double by the year 2100 [1]. As CO2 is a substrate for plant photosynthesis, an increase in its concentration in the atmosphere directly impacts plant growth and composition [2,3,4,5]. Hence, the carbon:nitrogen (C:N) ratios of plants increase with the concentration of CO2 [2,5,6,7,8,9], which enhanced the photosynthetic rate of C3 plants, such as wheat (Triticum aestivum L.) and barley (Hordeum vulgare L.) [10,11,12,13]. In addition, the alteration of secondary plant chemistry by CO2 rise was documented [14,15,16,17]. Indeed, plant grown under eCO2 condition usually elicits the production of phenolic compounds, tannins, and flavonoids and suppresses the production of terpenoids [17,18]. However, no consistent trend has been found in the response of insects to these allelochemicals adaptation [19].
Most of herbivorous insect species rely on olfactory signals from their environment to find a mate and to locate a host plant [20]. The VOCs from plants range from fatty acid derivatives, terpenoids, and sulfur compounds to phenylpropanoids [21]. The composition and amounts of plant VOCs can vary depending on several parameters: Plant taxon [22], stage of development [23], physiological status [24], and environmental stresses [25]. However, the effects of increasing the concentration of CO2 on the emissions of plant VOCs are not well-defined [26].
Aphids (Hemiptera: Aphididae) are the most important pest insects under temperate climate [27]. They are responsible for the transmission of more than 50% of insect-transmitted plant viruses [28]. Many aphid species produced two types of morphs: A winged (alate) morph, that is mainly responsible for the dispersal and the colonization of new plants, and an wingless (apterous) morph, that mostly stays on the plant on which it was born [29]. Aphids use olfaction to recognize plant hosts from non-plant hosts, which allows them to determine the suitability of different plants [30,31]. Moreover, the variation in behavioral responses to volatiles can also be found in winged and wingless morphs [32]. Volatile blends, based on headspace collections from wheat and oat (Avena sativa) plants, elicited similar behavioral responses from both morphs of R. padi in olfactometer studies. When compounds were tested individually, the two morphs responded differently [33]. Winged aphids were only attracted/arrested by four of the compounds, whereas wingless morphs responded to 11. However, studies dealing with the behavior of aphids, exposed to VOCs of host plants, grown under ambient and elevated atmospheric CO2, are not widespread [34,35,36,37].
As a worldwide pest insect, corn leaf aphid, Rhopalosiphum maidis (Fitch) (Hemiptera: Aphididae) caused significant damage on cereal crops, such as barley, corn, wheat, and broad bean [38]. Corn leaf aphid is also a vector of plant viruses including sugarcane mosaic virus (SCMV) and maize dwarf mosaic virus (MDMV), which result in serious damage [39,40,41]. This work aims to investigate the effects of elevated CO2 concentrations on foraging behavior of corn leaf aphids. The foraging behavior of aphids to chemical cues of host plants was assessed by using Y glass tube olfactometer. Then, volatile organic compounds from isolated barley seedlings Hordeum vulgare L. reared under ambient CO2 (aCO2), and elevated CO2 (eCO2) conditions were analyzed by GC-MS.
2. Material and Methods
2.1. CO2 Condition Chambers
Six chambers (60 cm in length, 50 cm in width, and 50 cm in height, PLEXIGLAS® GS, clear 0F00 GT, 8 mm thick; Evonik Industries, Essen, Germany) were used for rearing plants and insects under two different concentrations of CO2. In each chamber, a constant airflow (30 L·min−1) was pushed through an air pump (Koi flow 30; Superfish, Netherlands). Two levels of CO2 concentrations were applied: Ambient level (aCO2, 450 ± 50 ppm) and elevated level (eCO2, aCO2 + 350 ppm) by using a CO2 gas tank (>99% purity; Airliquide®, Paris, France). Three chambers were used for each CO2 treatment. These chambers were maintained at 23 ± 1 °C and 65 ± 10% relative humidity (RH), with a 16:8 h (L:D) photoperiod. Carbon dioxide concentrations, temperature, and RH were continuously monitored in each chamber with MCH-383 SD data loggers (Lutron, Taipei, Taiwan).
2.2. Plant Material
Barley, Hordeum vulgare L., Etincel cultivar was sown in single plastic plots (7.5 cm diameter, 9.0 cm high), with 25 to 30 seedlings per pot. After sowing, these pots were introduced in aCO2 and eCO2 chambers separately. Two weeks old barley seedlings (decimal code 12 [42]) were used for host finding behavior tests and volatile analysis.
2.3. Aphid Rearing
A colony of Corn leaf aphid, Rhopalosiphum maidis, was originally collected from a corn field in the experiment station of Chinese Academy of Agricultural Sciences (39°30′42″N, 116°36′7″E) in Hebei Province, China, was maintained under ambient CO2 concentration at a constant temperature of 23 ± 1 °C, 65 ± 10% RH and a photoperiod of 16:8 h (l:d). The colony was reared on barley seedlings in a cage (36 cm in length, 27 cm in width, and 28 cm in height).
To ensure the experiments remained uniform, numerous apterous reproductive adults were transferred to new pots. After 24 h, the adults were removed from the plants, and their offspring were reared on the barley seedlings. Three days old nymphs and eight days old winged and wingless adults were used for the host finding behavior tests.
2.4. Foraging Behavior Bioassay
A two-arms glass olfactometer (Y tube olfactometer) was used to investigate the behavioral response of nymphs, winged, and wingless adults to different olfactory stimuli from barley grown under different concentration of carbon dioxide (CO2) (eCO2: 800 ± 50 ppm; aCO2: 450 ± 50 ppm). The aphids were offered one of the following three odor source combinations: Control (clean air) versus aCO2 barley seedling, control versus eCO2 barley seedling, or eCO2 barley seedling versus aCO2 barley seedling.
All trials were conducted at 23 ± 1 °C in an observation chamber (60 cm in length, 50 cm in width and 40 cm in height) lightened with three 16-W cool white fluorescent lights, which provided uniform lightening. The main arm of Y-olfactometer (15 cm long and 1.5 cm I.D.) and the two arms (20 cm long and 1.5 cm I.D.) were made of glass. Three black lines (two centimeters from the bottom of the stem or two arms) were drawn on the stem and two arms of Y tube olfactometer separately in order to observe the position of aphids. Plants grown under elevated or ambient concentrations of CO2 in glass pot were placed into sealed glass chamber (4 L, 20 cm I.D.) (Analytical Research Systems, Gainesville, FL, USA) and randomly connected to each arm of the Y-olfactometer with Teflon® pipes. A push pump system (PVAS11; Volatile Assay Systems®, Rensselaer, NY, USA) was connected to each chamber to carry volatile organic compounds (VOCs) released by barley to the Y-olfactometer. The air was first purified through a charcoal filter to avoid any outdoor contamination. The pushed air flow was kept at 0.7 L·min−1.
Aphids were individually placed at the entrance of the stem part, alternating with nymphs, wingless and winged adult aphids. Each insect was allowed to spend five minutes in the Y tube olfactometer. In total 180 aphids were tested for each life stage. The host finding behaviors of aphids were visually observed and simultaneously encoded using The Observer 5.0 software (Noldus®, Wageningen, The Netherlands). The following behaviors were recorded during the experiment:
No response: When aphids stayed at the entrance, they did not cross the black line marked on the stem part.
Only searching: When aphids crossed the black line marked on the stem, but did not cross the black line marked on the chosen arm.
Selection: When aphids made a choice and crossed the black line marked on the arm of the Y tube olfactometer.
Between each experiment, new plants were introduced to the chambers. The Y-olfactometer were cleaned with pure n-hexane (>99.7%; VWR®, Radnor, PA, USA) and dried at room temperature for about five minutes after testing 15 aphids. Moreover, the chambers and all of the Teflon pipes were washed with n-hexane (>99.7%; VWR®, Radnor, PA, USA).
2.5. Headspace Analysis of Volatiles from Plants by GC-MS
The upper seedling parts (about 12 cm in length) of aCO2 and eCO2 barley were carefully sealed in the bell-like glass collection chamber (2 L) separately. To avoid volatile contamination, the base root parts were wrapped with aluminum foil and placed in a cleaned glass bottle. Headspace volatiles from aCO2 and eCO2 barley seedlings were collected using a dynamic ‘push–pull’ pump system. The pushed airflow was set at 0.7 L·min−1 and the pulled air flow was set at 0.3 L·min−1. The air entering into the chamber was cleaned by an activated charcoal filter. A 60 mg Tenax TA® thermodesorption tube (Gerstel, Germany), which is made of a microporous polymer of 2,6-diphenylen oxide, was placed at the exit of the glass chamber to trap the volatile compounds carried by the air pulling from the chamber. The tubes were previously cleaned by a thermal conditioner (TC2, Gerstel, Mülheim an der Ruhr, Deutschland), for a period of 11 h at 300 °C. Volatile collection took place over a 24-h period. Straight after volatile collection, the entire aerial portion of the plants was removed to determine dry weight. It allowed the calculation of the amount of VOCs in nanogram per gram of above-ground dry plant. Six replicates were conducted for each condition of CO2 concentration of growing, along with the same number of controls (only soil and glass pots wrapped with aluminum).
The volatiles were analyzed by Gas Chromatography coupled with a Mass Spectrometer (GC-MS) (model 7890A; Agilent Technologies Inc., Santa Clara, CA, USA). In this system, the Tenax TA cartridge was thermally desorbed (Thermal Desorption Unit, Gerstel, Mülheim an der Ruhr, Deutschland) at 250 °C for 10 min prior to the injection. In each sample, one microliter of butylbenzene (2.15 ng/μL) was injected as an internal standard.
The entire sample was injected in a HP-5 capillary column (5% Phenyl Methyl, 30.0 m, internal diameter: 0.25 mm, thickness: 0.25 μm, Agilent Technologies®, Santa Clara, CA, USA). The carrier gas used was Helium (Initial flow: 1.5 mL/min, Post flow: 0.4 mL/min). The temperature program started at 40 °C for 2 min, and was increased at 4 °C min−1 to 95 °C, and then increased at 6 °C min−1 to 155 °C for 10 min, and was finally increased at 25 °C min−1 to 280 °C hold for 5 min. The detected peaks were identified based on their mass spectrum by using spectral libraries, Pal 600k and Wiley 275 (the MS spectra match factor was minimum 70%).
2.6. Statistical Analyses
Binomial proportion tests (equal distribution hypothesized) were used to compare the foraging behavior of nymphs, wingless and winged aphids in Y tube olfactometer. The residence time of each choice was subjected to an analysis by using a general linear model (GLM). The treatment means were compared using the Tukey’s multiple range tests to determine significant difference at a 95% confidence level. Plant VOCs between two CO2 levels were tested with independent samples t-test. All analyses were performed using SAS version 9.2 (SAS Institute, Cary, NC, USA).
3. Results
3.1. Foraging Behaviors of Aphids
We tested the foraging behavior of corn leaf aphid for three developmental stages (Figure 1), according to different dual choice, namely control versus aCO2 barley seedling, control versus eCO2 barley seedling and eCO2 versus aCO2 barley seedlings. The winged and wingless aphids were more attracted by odors of aCO2 barley seedlings when tested in combination with control air or in combination with eCO2 barley seedlings. However, nymphs were only attracted by aCO2 barley seedlings when it was tested in combination with control, otherwise no significant difference was observed when eCO2 was tested in combination with aCO2 or with control.
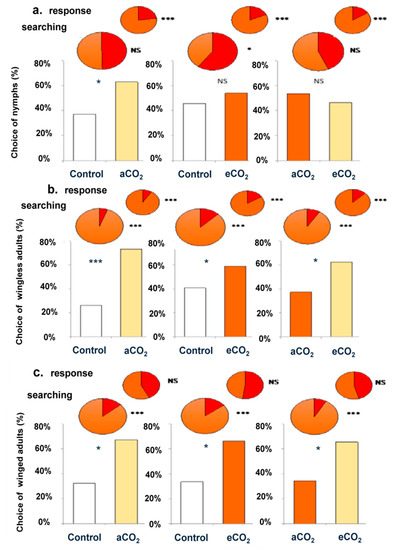
Figure 1.
Foraging behavior (in %) of nymph (a), wingless (b) and winged (c) corn leaf aphid during three dual choices including control, aCO2 and eCO2 barley seedling combinations. Response and searching status assessment corresponded to mobility in the first 2 cm, and before the split of the olfactometer 2 arms, respectively. Red color in pies was negative behaviors (For example, orange color in pies pointed to response, and red color pointed to no response). There were three replicates for each treatment, and a total of 180 aphids were tested. *, *** and NS for p ≤ 0.05, p ≤ 0.001 and not significant at α = 0.05, respectively.
3.2. Residence Duration for Searching and Selection Behaviors
The typical behavior of nymphs in Y tube olfactometer consisted mainly of searching activities with more than 38% of experimental time. Also, more than 33% of experimental duration of winged aphids corresponded to no response to odor sources, being stationary at the entrance of the olfactometer stem. When the aphids made a choice, the residence duration on each arm was largely affected by kind of life stages (Table 1 and Figure 2). The wingless and winged aphids spent significantly more time in the arms of aCO2 barley seedlings when tested against control or eCO2 barley seedlings. However, the residence duration of three life stages did not show any significant difference between control versus eCO2 barley seedlings.

Table 1.
Summary of a general linear model (GLM) analysis of the effect of life stages (nymphs, wingless and winged adults) and choices on residence duration of Rhopalosiphon maidis in Y tube olfactometer, during three dual choices including control, aCO2 and eCO2 barley seedling combinations.
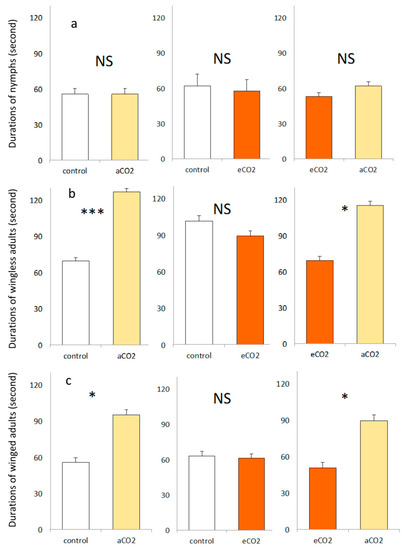
Figure 2.
(mean ± se) of nymph (a), wingless (b) and winged (c) corn leaf aphid during three dual choices including control, aCO2 and eCO2 barley seedling combinations. There were three replicates for each treatment, and a total of 180 aphids were tested. *, *** and NS for p ≤ 0.05, p ≤ 0.001 and not significant at α = 0.05, respectively.
3.3. Volatiles Analysis
According to the GC-MS analysis, 16 and 9 VOCs were identified in aCO2, and eCO2 barley seedlings, respectively (Figure 3). While, 1,3-butanediol was the main volatile compound emitted by aCO2 barley seedling, linalool was the most abundant volatile compound emitted by eCO2 barley seedling. Six volatiles were found in both aCO2 and eCO2 barley seedlings, including heptanal, 1,3-butanediol, 2,3-butanediol, 2-methyl-propanoic acid, heptadecane, and pentadecane. However, the relative abundances decreased in eCO2 barley seedlings. The volatile pattern from aCO2 barley seedlings was more diversified, including seven supplementary volatiles, namely 2-hexenal, cyclohexane decyl, 3-methyl-hexadecane, 3-methyl-pentadecane, 4-methyl-pentadecane, 1-methyl-2-propyl-benzene, propyl-benzene, 1,2,4-trimethyl- benzene, 1,3,5-trimethyl-benzene, and indane.
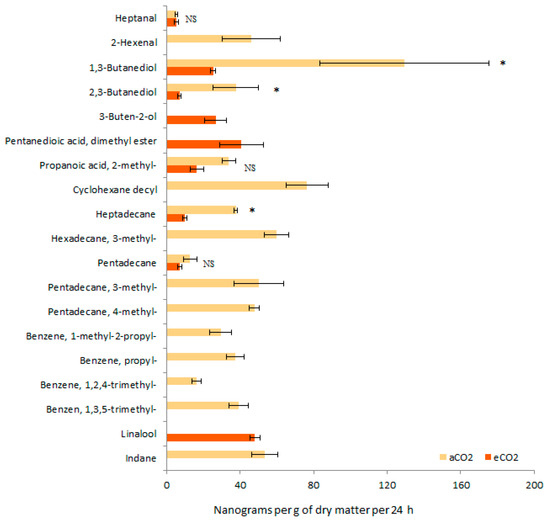
Figure 3.
Diversity and abundance of volatile emission (mean ± se in ng per g of dry matter per 24 h) from aCO2 and eCO2 barley seedling (n = 6 replicates). * and NS for p ≤ 0.05 and not significant at α = 0.05, respectively.
4. Discussion
In the study, aphid foraging behaviors were found to be influenced by host plants reared in different CO2 concentrations. The diversity and abundance of plant VOCs were also differently induced by elevated CO2 when compared with aCO2.
In our experiment, the wingless and winged aphids were more attracted to the odors of aCO2 barley seedlings when tested against eCO2 barley seedlings or control. They spent more time during the dual choice on aCO2 barley seedlings. However, nymphs were only attracted by aCO2 when it was tested in combination with the control. There was some evidence that aphids can detect a variety of individual plant odor components using the hairs on the tips of antennae [43] or that a sensilla at the tibia-tarsus junction may respond to non-volatile chemicals [44]. In aphids, the semio-chemicals are perceived by sensory structures called rhinaria, that are classified in two main groups: Primary and secondary rhinaria. For example, distal (DPR) and proximal (PPR) primary rhinaria allow all morphs and life stages of Aphis fabae Scopoli to detect 2-hexenal, a common volatile of their host-plants [45] that is not detected by secondary rhinaria. PPR are usually associated with the perception of host and non-host volatile chemicals, and DPR are probably involved in the perception of the alarm pheromone [30,46].
GC-MS analysis showed that volatiles come from barley seedlings, including aldehydes, alcohol compounds, acid compounds, alkanes, phenyl compounds and others. We found 2-hexenal in the volatile blends of aCO2 barley seedlings. Previous study has proved that it is attractive to pea aphid, Acyrthosiphon pisum Harris [47]. In our experiment, an amount of linalool was emitted by eCO2 barley seedling, which had showed a repellent effect on green peach aphid, Myzus persicae (Sulzer) [48], corn leaf aphid, R. maidis, and bird cherry-oat aphid, R. padi [49]. Therefore, the presence of those compounds in the odor blends of barley, could explain the preference of aphids towards odors of aCO2 barley.
The volatiles emitted by barley aerial parts differ both qualitatively and quantitatively, probably because of the experimental treatments or plant stage. Bukovinszky et al. (2005) analyzed the headspace volatiles of 3–4 week old barley; they detected 15 compounds and pointed the volatile profile of barley had the greatest dissimilarity [50]. Wenda-Piesik et al. (2010) collected 11 different volatiles from third leaf stage uninfested barley [51]. Piesik et al. (2010) tested six-week barley, identified about 19 volatiles, and mentioned that mechanical injury and insect feeding caused barley to quantitatively release the highest total VOC concentrations after injury [52]. In our study reported here in, we used the intact barley seedling after 14 days of growth, and identified 16 VOCs in aCO2 barley seedlings, and 9 VOCs in eCO2 barley seedlings. The barley seedlings were so young and not infested by insects or any fungus, which is probably the reason why we collected less volatiles compared to other research.
The common feature of wingless and winged aphids in the foraging behaviors in the study was that, once they had a response to the volatiles of the plant, their search time was relatively short and they could make a choice quickly. The average time of response at the entrance of Y tube for winged aphid was longer than wingless. The sensitivity to the plant volatiles and the variation in behavioral responses were partly as a result of differences in morphs. Walking is the main way of wingless aphids expanding to nearby plants in the field. When induced by host-plant odors, the wingless aphids will actively walk towards the odor source in the absence of other host cues [53,54]. Winged morphs are capable of making targeted landings on plants under low wind conditions [55,56,57]. However, winged aphids will not attempt to fly without a certain speed of wind prevails [58,59], which indicate that wind probably is an important precondition for the movement of winged aphid. Behavior. The results showed that the nymphs spent more time searching around in the central tube of the olfactometer compared to adults, perhaps they spend more energy moving than adults because of their slower walking speed [60,61,62].
5. Conclusions
The aphid behavioral response to plant VOCs is complex. Presented as a blend, these volatile compounds may be integrated as host cues, leading to aphid attraction/arrestment toward the odor source [63]. Every compound of the constitutive blend is going to present individually in olfactometric bioassays to test which one have effect on aphid responses. Future work may focus on how aphids gain information on the identity and quality of a plant from the composition of its volatile blend, and how the interactions between volatiles can affect aphid behavior in changing climatic conditions.
Author Contributions
Y.C. performed the experiments and data statistics processing. C.M. helped with plant GC-MS test. J.C.F.M. helped with the aphid foraging behavior test. F.V. contributed to designing the experimental protocol. Y.C., C.M., Z.W., K.L.H., and F.F. contributed to the preparation of the manuscript. F.F. supervised the research.
Funding
This research was supported by National Key R and D Program of China (2017YFD0201802) and Erasmus Mundus program.
Acknowledgments
This study would not have been possible without the following members: Nicolas Poncelet, Nicolas Leroy, Antoine Boullis, Solene Blanchard, Laurent Serteyn, Emilie Bosquée.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Stocker, T.; Qin, D.; Plattner, G.; Tignor, M.; Allen, S.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, V.; Midgley, P. IPCC, 2013: Climate Change 2013: The physical science basis. contributionof working group i to the fifth assessment report of the intergovernmental panel on climate change. Comput. Geom. 2013, 18, 95–123. [Google Scholar]
- Hartley, S.E.; Jones, C.G.; Couper, G.C.; Jones, T.H. Biosynthesis of plant phenolic compounds in elevated atmospheric CO2. Glob. Chang. Biol. 2000, 6, 497–506. [Google Scholar] [CrossRef]
- Bae, H.; Sicher, R. Changes of soluble protein expression and leaf metabolite levels in Arabidopsis thaliana grown in elevated atmospheric carbon dioxide. Field Crops Res. 2004, 90, 61–73. [Google Scholar] [CrossRef]
- Seneweera, S.P.; Conroy, J.P. Enhanced leaf elongation rates of wheat at elevated CO2: Is it related to carbon and nitrogen dynamics within the growing leaf blade? Environ. Exp. Bot. 2005, 54, 174–181. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Rogers, A. The response of photosynthesis and stomatal conductance to rising CO2: Mechanisms and environmental interactions. Plant Cell Environ. 2007, 30, 258–270. [Google Scholar] [CrossRef] [PubMed]
- Johns, C.V.; Hughes, L. Interactive effects of elevated CO2 and temperature on the leaf-miner Dialectica scalariella Zeller (Lepidoptera: Gracillariidae) in Paterson’s Curse, Echium plantagineum (Boraginaceae). Glob. Chang. Biol. 2002, 8, 142–152. [Google Scholar] [CrossRef]
- Chen, F.; Ge, F.; Parajulee, M.N. Impact of elevated CO2 on tri-trophic interaction of Gossypium hirsutum, Aphis gossypii, and Leis axyridis. Environ. Entomol. 2005, 34, 37–46. [Google Scholar] [CrossRef]
- Sun, Y.; Guo, H.; Yuan, E.; Ge, F. Elevated CO2 increases R gene-dependent resistance of Medicago truncatula against the pea aphid by up-regulating a heat shock gene. New Phytol. 2018, 217, 1696–1711. [Google Scholar] [CrossRef]
- Zhang, Y.; Dai, Y.; Wan, G.; Liu, B.; Xing, G.; Chen, F. Effects of elevated CO2 on plant chemistry, growth, yield of resistant soybean, and feeding of a target lepidoptera pest, Spodoptera litura (Lepidoptera: Noctuidae). Environ. Entomol. 2018, 47, 848–856. [Google Scholar]
- Drake, B.G.; Azcon-Bieto, J.; Berry, J.; Bunce, J.; Dijkstra, P.; Farrar, J.; Gifford, R.; Gonzalez-meler, M.A.; Koch, G.; Lambers, H. Does elevated atmospheric CO2 concentration inhibit mitochondrial respiration in green plants? Plant Cell Environ. 1999, 22, 649–657. [Google Scholar] [CrossRef]
- Ziska, L.H.; Namuco, O.; Moya, T.; Quilang, J. Growth and yield response of field-grown tropical rice to increasing carbon dioxide and air temperature. Agron. J. 1997, 89, 45–53. [Google Scholar] [CrossRef]
- Kim, K.N.; Cheong, Y.H.; Grant, J.J.; Pandey, G.K.; Luan, S. CIPK3, a calcium sensor-associated protein kinase that regulates abscisic acid and cold signal transduction in Arabidopsis. Plant Cell 2003, 15, 411–423. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Long, S.P. What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytol. 2005, 165, 351–372. [Google Scholar] [CrossRef] [PubMed]
- Karowe, D.N.; Seimens, D.H.; Mitchell-Olds, T. Species-specific response of glucosinolate content to elevated atmospheric CO2. J. Chem. Ecol. 1997, 23, 2569–2582. [Google Scholar] [CrossRef]
- Coviella, C.E.; Stipanovic, R.D.; Trumble, J.T. Plant allocation to defensive compounds: Interactions between elevated CO2 and nitrogen in transgenic cotton plants. J. Exp. Bot. 2002, 53, 323–331. [Google Scholar] [CrossRef]
- Karowe, D.N.; Grubb, C. Elevated CO2 increases constitutive phenolics and trichomes, but decreases inducibility of phenolics in Brassica rapa (Brassicaceae). J. Chem. Ecol. 2011, 37, 1332–1340. [Google Scholar] [CrossRef]
- Stiling, P.; Cornelissen, T. How does elevated carbon dioxide CO2 affect plant–herbivore interactions? A field experiment and meta-analysis of CO2-mediated changes on plant chemistry and herbivore performance. Glob. Chang. Biol. 2007, 13, 1823–1842. [Google Scholar] [CrossRef]
- Robinson, M.T.; Weeks, A.R.; Hoffmann, A.A. Geographic patterns of clonal diversity in the earth mite species Penthaleus major with particular emphasis on species margins. Evolution 2002, 56, 1160–1167. [Google Scholar] [CrossRef] [PubMed]
- Bidart-Bouzat, M.G.; Imeh-Nathaniel, A. Global change effects on plant chemical defenses against insect herbivores. J Integr. Plant Biol. 2008, 50, 1339–1354. [Google Scholar] [CrossRef] [PubMed]
- Hansson, B.; Wicher, D. Chemical ecology in insects. In Chemosensory Transduction: The Detection of Odors, Tastes, and other Chemostimuli; Zufall, F., Munger, S.D., Eds.; Academic press: London, UK, 2006; pp. 29–45. [Google Scholar]
- Qualley, A.V.; Dudareva, N. Metabolomics of plant volatiles. Methods Mol. Biol. 2009, 553, 329–343. [Google Scholar]
- Knudsen, J.T.; Tollsten, L.; Bergström, L.G. Floral scents-a checklist of volatile compounds isolated by head-space techniques. Phytochemistry 1993, 33, 253–280. [Google Scholar] [CrossRef]
- Holopainen, J.K.; Heijari, J.; Oksanen, E.; Alessio, G.A. Leaf volatile emissions of Betula pendula during autumn coloration and leaf fall. J. Chem. Ecol. 2010, 36, 1068–1075. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Martínez, E.; Bosque-Pérez, N.; Berger, P.; Zemetra, R. Life history of the bird cherry-oat aphid, Rhopalosiphum padi (Homoptera: Aphididae), on transgenic and untransformed wheat challenged with Barley yellow dwarf virus. J. Econ. Entomol. 2004, 97, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Maffei, M.E. Sites of synthesis, biochemistry and functional role of plant volatiles. S. Afr. J. Bot. 2010, 76, 612–631. [Google Scholar] [CrossRef]
- Boullis, A.; Detrain, C.; Francis, F.; Verheggen, F.J. Will climate change affect insect pheromonal communication? Curr. Opin. Insect Sci. 2016, 17, 87–91. [Google Scholar] [CrossRef]
- Harrewijn, P.; Minks, A.K. Aphids: Their Biology, Natural Enemies, and Control, Vol. A.; Elsevier: Amsterdam, The Netherlands, 1987. [Google Scholar]
- Nault, L. Arthropod transmission of plant viruses: A new synthesis. Ann. Entomol. Soc. Am. 1997, 90, 521–541. [Google Scholar] [CrossRef]
- Braendle, C.; Davis, G.K.; Brisson, J.A.; Stern, D.L. Wing dimorphism in aphids. Heredity 2006, 97, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Pickett, J.; Wadhams, L.; Woodcock, C.; Hardie, J. The chemical ecology of aphids. Annu. Rev. Entomol. 1992, 37, 67–90. [Google Scholar] [CrossRef]
- Pettersson, J.; Ninkovic, V.; Glinwood, R.; Al Abassi, S.; Birkett, M.; Pickett, J.; Wadhams, L. Chemical stimuli supporting foraging behaviour of Coccinella septempunctata L. (Coleoptera: Coccinellidae): Volatiles and allelobiosis. Appl. Entomol. Zool. 2008, 43, 315–321. [Google Scholar] [CrossRef][Green Version]
- Webster, B. The role of olfaction in aphid host location. Physiol. Entomol. 2012, 37, 10–18. [Google Scholar] [CrossRef]
- Quiroz, A.; Niemeyer, H. Olfactometer-assessed responses of aphid Rhopalosiphum padi to wheat and oat volatiles. J. Chem. Ecol. 1998, 24, 113–124. [Google Scholar] [CrossRef]
- Boullis, A.; Francis, F.; Verheggen, F.J. Climate change and tritrophic interactions: Will modifications to greenhouse gas emissions increase the vulnerability of herbivorous insects to natural enemies? Environ. Entomol. 2015, 44, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Boullis, A.; Fassotte, B.; Sarles, L.; Lognay, G.; Heuskin, S.; Vanderplanck, M.; Bartram, S.; Haubruge, E.; Francis, F.; Verheggen, F.J. Elevated carbon dioxide concentration reduces alarm signaling in aphids. J. Chem. Ecol. 2017, 43, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Boullis, A.; Blanchard, S.; Francis, F.; Verheggen, F. Elevated CO2 concentrations impact the semiochemistry of aphid honeydew without having a cascade effect on an aphid predator. Insects 2018, 9, 47. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, S.; Lognay, G.; Verheggen, F.; Detrain, C. Today and tomorrow: Impact of climate change on aphid biology and potential consequences on their mutualism with ants. Physiol. Entomol. 2019, 44, 77–86. [Google Scholar] [CrossRef]
- El-Ibrashy, M.T.; El-ziadys, S.; Riad, A.A. Laboratory studies on the biology of the corn leaf aphid, Rhopalosiphum maidis (Homoptera: Aphididae). Entomol. Exp. Appl. 1972, 15, 166–174. [Google Scholar] [CrossRef]
- Everly, R.T. Loss in corn yield associated with the abundance of the corn leaf aphid, Rhopalosiphum maidis, in Indiana. J. Econ. Entomol. 1960, 53, 924–932. [Google Scholar] [CrossRef]
- Foott, W.H.; Timmins, P.R. Effects of infestations by the corn leaf aphid, Rhopalosiphum maidis (Homoptera: Aphididae), on field corn in southwestern Ontario. Can. Entomol. 1973, 105, 449–458. [Google Scholar] [CrossRef]
- Bing, J.W.; Guthrie, W.D.; Dicke, F.F.; Obrycki, J.J. Seedling stage feeding by corn leaf aphid (Homoptera: Aphididae): Influence on plant development in maize. J. Econ. Entomol. 1991, 84, 625–632. [Google Scholar] [CrossRef]
- Tottman, D.; Makepeace, R.; Broad, H. An explanation of the decimal code for the growth stages of cereals, with illustrations. Ann. Appl. Biol. 1979, 93, 221–234. [Google Scholar] [CrossRef]
- Powell, G.; Hardie, J.; Pickett, J. Behavioural evidence for detection of the repellent polygodial by aphid antennal tip sensilla. Physiol. Entomol. 1995, 20, 141–146. [Google Scholar] [CrossRef]
- Pettersson, J.; Tjallingii, W.F.; Hardie, J. Host-Plant Selection and Feeding. In Aphids as Crop Pests; van Emden, H.F., Harrington, R., Eds.; CABI: Wallingford, UK, 2007; pp. 87–113. [Google Scholar]
- Park, K.C.; Hardie, J. Electrophysiological characterisation of olfactory sensilla in the black bean aphid, Aphis fabae. J. Insect Physiol. 2004, 50, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Wohlers, P.; Tjallingii, W. Electroantennogram responses of aphids to the alarm pheromone (E)-β-farnesene. Entomol. Exp. Appl. 1983, 33, 79–82. [Google Scholar] [CrossRef]
- Babikova, Z.; Gilbert, L.; Randall, K.C.; Bruce, T.J.; Pickett, J.A.; Johnson, D. Increasing phosphorus supply is not the mechanism by which arbuscular mycorrhiza increase attractiveness of bean (Vicia faba) to aphids. J. Exp. Bot. 2014, 65, 5231–5241. [Google Scholar] [CrossRef] [PubMed]
- Harmel, N.; Almohamad, R.; Fauconnier, M.L.; Du Jardin, P.; Verheggen, F.; Marlier, M.; Haubruge, E.; Francis, F. Role of terpenes from aphid-infested potato on searching and oviposition behavior of Episyrphus balteatus. Insect Sci. 2007, 14, 57–63. [Google Scholar] [CrossRef]
- Halbert, S.; Corsini, D.; Wiebe, M.; Vaughn, S. Plant-derived compounds and extracts with potential as aphid repellents. Ann. Appl. Biol. 2009, 154, 303–307. [Google Scholar] [CrossRef]
- Bukovinszky, T.; Gols, R.; Posthumus, M.; Vet, L.; Van Lenteren, J. Variation in plant volatiles and attraction of the parasitoid Diadegma semiclausum (Hellen). J. Chem. Ecol. 2005, 31, 461–480. [Google Scholar] [CrossRef] [PubMed]
- Wenda-Piesik, A.; Piesik, D.; Ligor, T.; Buszewski, B. Volatile organic compounds (VOCs) from cereal plants infested with crown rot: Their identity and their capacity for inducing production of VOCs in uninfested plants. Int. J. Pest Manag. 2010, 56, 377–383. [Google Scholar] [CrossRef]
- Piesik, D.; Łyszczarz, A.; Tabaka, P.; Lamparski, R.; Bocianowski, J.; Delaney, K. Volatile induction of three cereals: Influence of mechanical injury and insect herbivory on injured plants and neighbouring uninjured plants. Ann. Appl. Biol. 2010, 157, 425–434. [Google Scholar] [CrossRef]
- Visser, J.; Taanman, J. Odour-conditioned anemotaxis of apterous aphids (Cryptomyzus korschelti) in response to host plants. Physiol. Entomol. 1987, 12, 473–479. [Google Scholar] [CrossRef]
- Nottingham, S.F.; Hardie, J.; Dawson, G.W.; Hick, A.J.; Pickett, J.A.; Wadhams, L.J.; Woodcock, C.M. Behavioral and electrophysiological responses of aphids to host and nonhost plant volatiles. J. Chem. Ecol. 1991, 17, 1231–1242. [Google Scholar] [CrossRef] [PubMed]
- Storer, J.R.; Young, S.; Hardie, J. Three-dimensional analysis of aphid landing behaviour in the laboratory and field. Physiol. Entomol. 1999, 24, 271–277. [Google Scholar] [CrossRef]
- Vargas, R.R.; Troncoso, A.J.; Tapia, D.H.; Olivares-Donoso, R.; Niemeyer, H.M. Behavioural differences during host selection between alate virginoparae of generalist and tobacco-specialist Myzus persicae. Entomol. Exp. Appl. 2005, 116, 43–53. [Google Scholar] [CrossRef]
- Goldansaz, S.H.; McNeil, J.N. Effect of wind speed on the pheromone-mediated behavior of sexual morphs of the potato aphid, Macrosiphum euphorbiae (Thomas) under laboratory and field conditions. J. Chem. Ecol. 2006, 32, 1719. [Google Scholar] [CrossRef] [PubMed]
- Hardie, J.; Gao, N.; Timar, T.; Sebok, P.; Honda, K.I. Precocene derivatives and aphid morphogenesis. Arch. Insect Biochem. Physiol. 1996, 32, 493–501. [Google Scholar] [CrossRef]
- Kennedy, J. Behavioural post-inhibitory rebound in aphids taking flight after exposure to wind. Anim. Behav. 1990, 39, 1078–1088. [Google Scholar] [CrossRef]
- Sørensen, J.G.; Kristensen, T.N.; Loeschcke, V. The evolutionary and ecological role of heat shock proteins. Ecol. Lett. 2003, 6, 1025–1037. [Google Scholar] [CrossRef]
- Gish, M.; Inbar, M. Host location by apterous aphids after escape dropping from the plant. J. Insect Behav. 2006, 19, 143. [Google Scholar] [CrossRef]
- Zhang, W.; Chang, X.Q.; Hoffmann, A.A.; Zhang, S.; Ma, C.S. Impact of hot events at different developmental stages of a moth: The closer to adult stage, the less reproductive output. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef]
- Webster, B.; Bruce, T.; Pickett, J.; Hardie, J. Volatiles functioning as host cues in a blend become nonhost cues when presented alone to the black bean aphid. Anim. Behav. 2010, 79, 451–457. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).