Acoustic, Pitfall Trap, and Visual Surveys of Stored Product Insect Pests in Kenyan Warehouses
Abstract
:1. Introduction
2. Materials and Methods
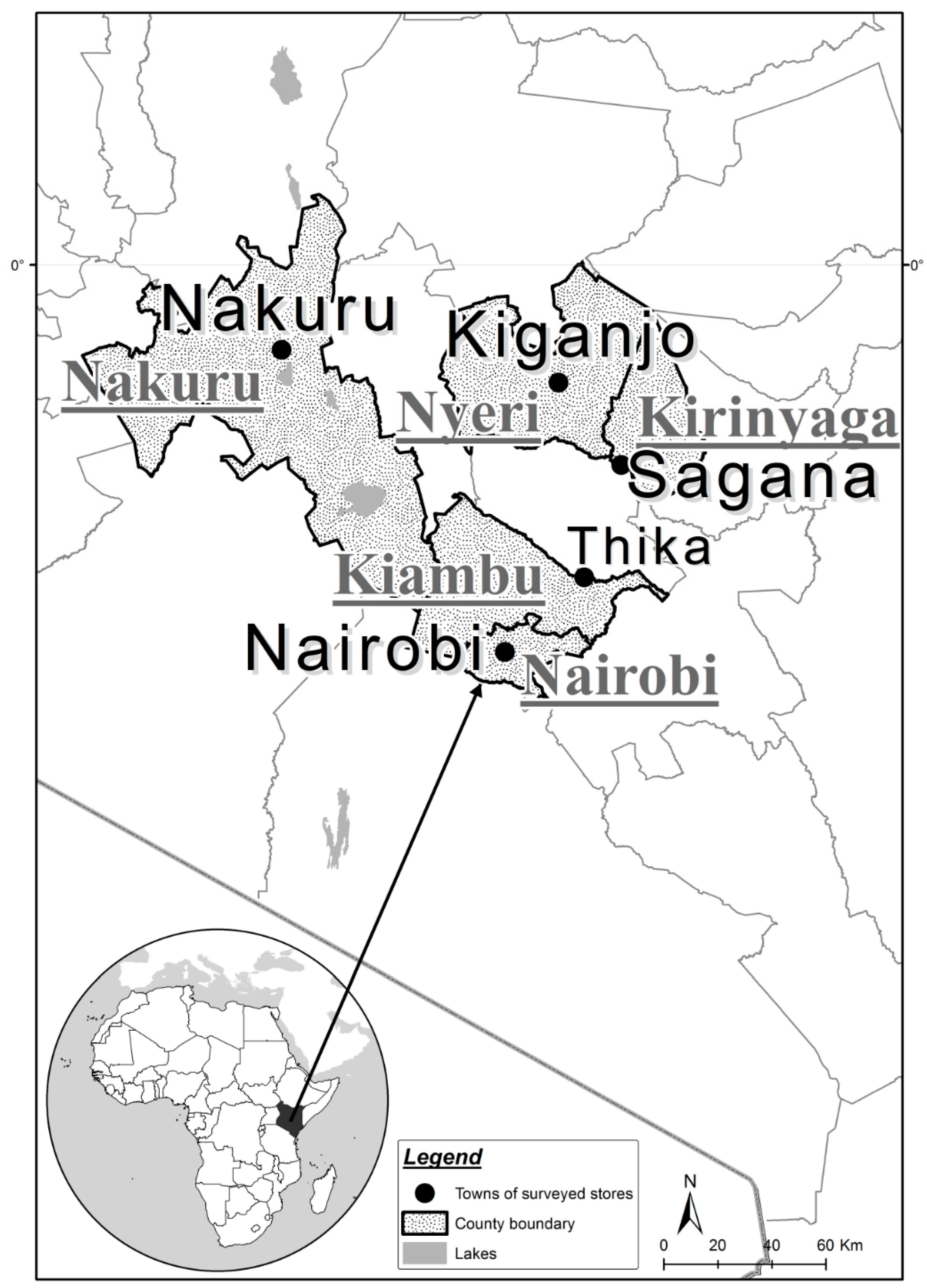
2.1. Recording Sites
2.2. Sampling Methods
2.3. Insect Trapping
2.4. Recording Equipment and Set-Up
2.5. Automated Classification of Insect Sounds and Background Noise Signals
3. Results
3.1. Pitfall Trap Counts and Visual Surveys
3.2. Acoustic Assessment of Infestation
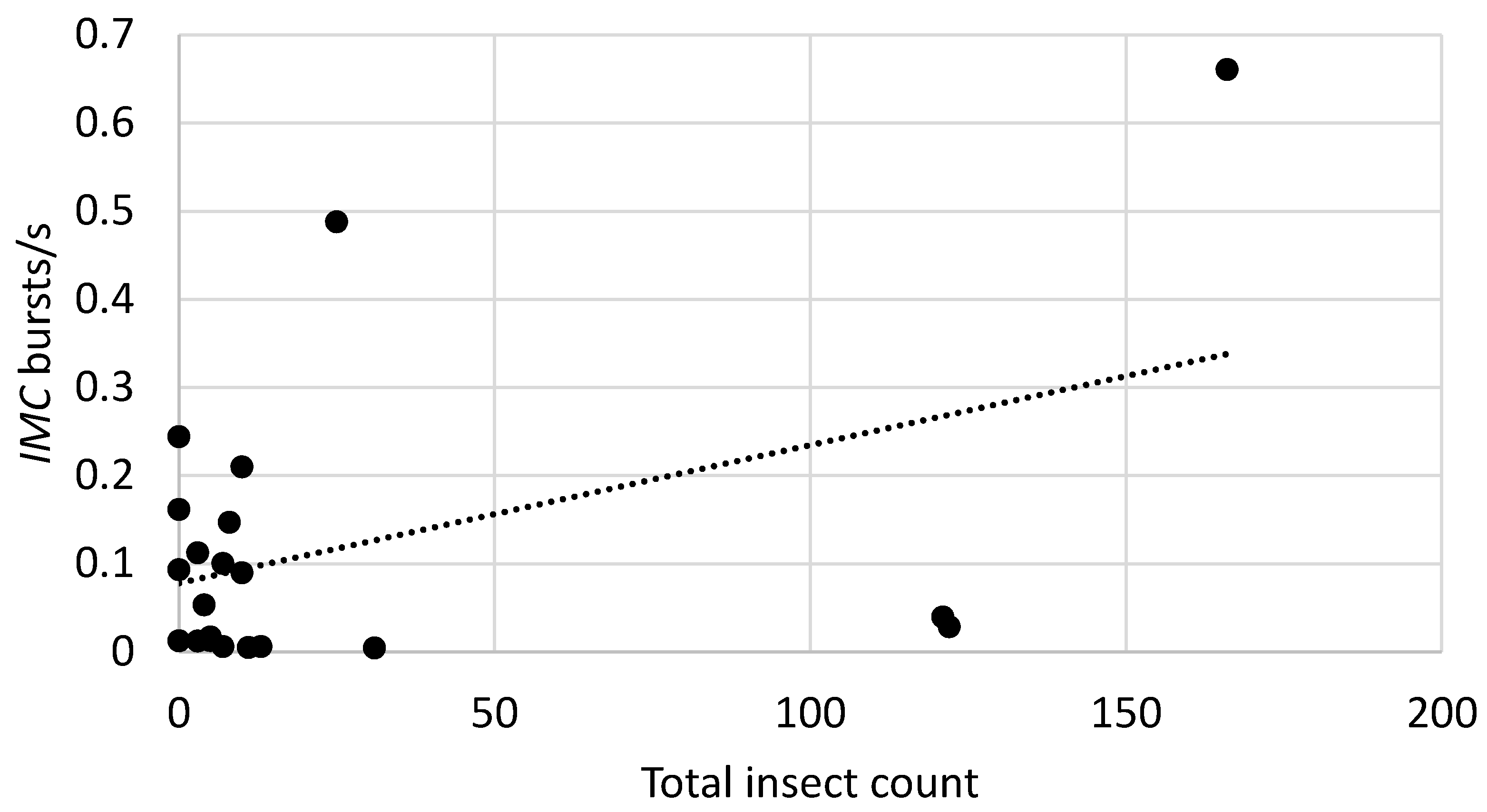
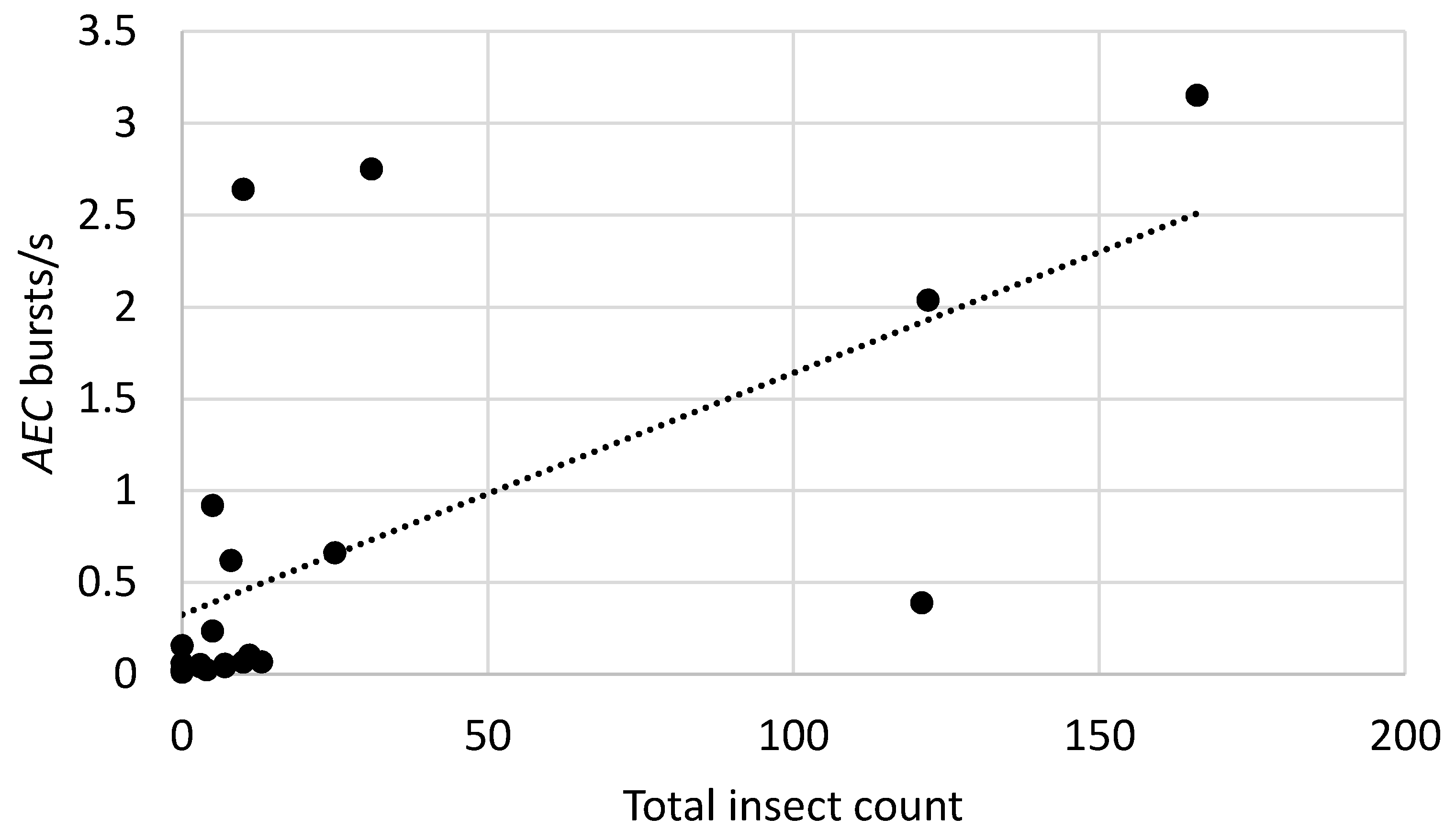
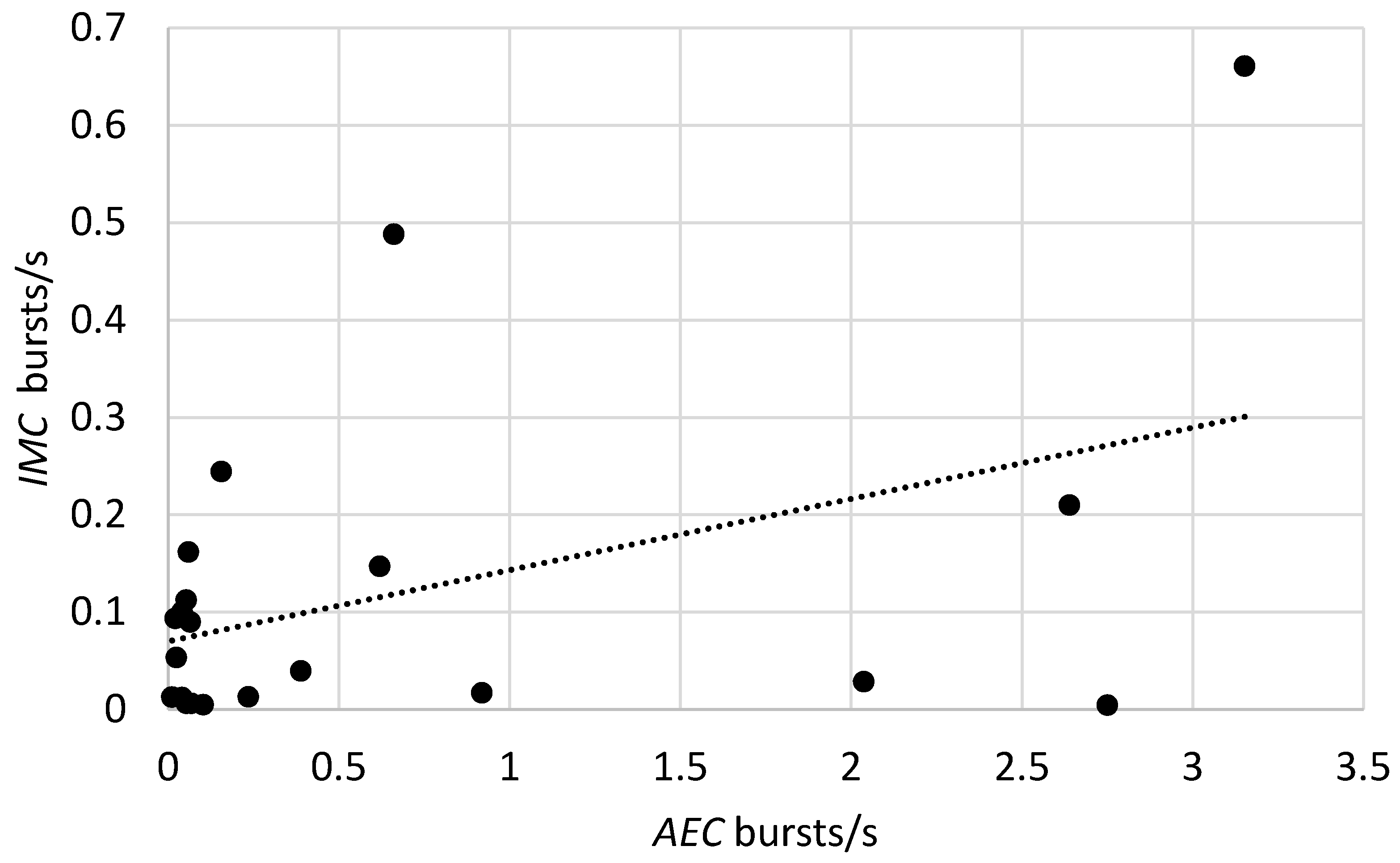
3.3. Relationship between Burst Rates and Counts of Insects Recovered from Bags
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zorya, S.; Morgan, N.; Diaz Rios, L.; Hodges, R.; Bennett, B.; Stathers, T.; Mwebaze, P.; Lamb, J. Missing Food: The Case of Postharvest Grain Losses in Sub-Saharan Africa; Report No. 60371-AFR; The World Bank: Washington, DC, USA; 2011; 96p. [Google Scholar]
- Murphy, S. Strategic Grain Reserves in an Era of Volatility; Institute for Agriculture and Trade Policy: Minneapolis, MN, USA, 2009; 15p. [Google Scholar]
- Early, R.; Moreno, P.G.; Murphy, S.T.; Day, R. Forecasting the global extent of invasion of the cereal pest Spodoptera frugiperda, the fall armyworm. NeoBiota 2018, 40, 25–50. [Google Scholar] [CrossRef]
- George, M.L. Effective Grain Storage for Better Livelihoods of African Farmers’ Project: Completion Report June 2008 to February 2011; International Maize and Wheat Improvement Centre (CIMMYT): Mexico City, Mexico, 2011; 65p. [Google Scholar]
- Vowotor, K.A.; Meikle, W.G.; Ayertey, J.N.; Markham, R.H. Distribution of association between the larger grain borer Prostephanus truncatus (Horn) (Coleoptera: Bostrichidae) and the maize weevil Sitophilus zeamais Motschulsky (Coleoptera: Curculionidae) in maize stores. J. Stored Prod. Res. 2005, 41, 498–512. [Google Scholar] [CrossRef]
- Rembold, F.; Hodges, R.; Bernard, M.; Knipschild, H.; Léo, O. The African Postharvest Losses Information System (APHLIS). An Innovative Framework to Analyse and Compute Quantitative Postharvest Losses for Cereals Under Different Farming and Environmental Conditions in East and Southern Africa; JRC 62618 EUR 24712 EN; Publications Office of the European Union: Luxembourg, 2011; ISBN 978-02-79-19143-5. ISSN 1018-5593. [Google Scholar]
- Opit, G.P.; Phillips, T.W.; Aikins, M.J.; Hasan, M.M. Phosphine resistance in Tribolium castaneum and Rhyzopertha dominica from stored wheat in Oklahoma. J. Econ. Entomol. 2012, 105, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Toews, M.D.; Nansen, C. Trapping and interpreting captures of stored grain insects. In Stored Product Protection; Hagstrum, D.W., Phillips, T.W., Cuperus, G., Eds.; Kansas State University: Manhattan, KS, USA, 2012; pp. 243–261. [Google Scholar]
- Mankin, R.W.; Hagstrum, D.W.; Smith, M.T.; Roda, A.L.; Kairo, M.T.K. Perspective and promise: A century of insect acoustic detection and monitoring. Am. Entomol. 2011, 57, 30–44. [Google Scholar] [CrossRef]
- Leblanc, M.P.; Gaunt, D.; Fleurat-Lessard, F. Experimental study of acoustic equipment for real-time insect detection in grain bins—Assessment of their potential for infestation risk prediction during long term storage periods. IOBC/WPRS Bull. 2011, 69, 79–88. [Google Scholar]
- Eliopoulos, P.A.; Potamitis, I.; Kontodimas, D.C.; Givropoulou, E.G. Detection of adult beetles inside the stored wheat mass based on their acoustic emissions. J. Econ. Entomol. 2015, 108, 2808–2814. [Google Scholar] [CrossRef]
- Eliopoulos, P.A.; Potamitis, I.; Kontodimas, D.C. Estimation of population density of stored grain pests via bioacoustic detection. Crop Prot. 2016, 85, 71–78. [Google Scholar] [CrossRef]
- Hagstrum, D.W.; Flinn, P.W.; Shuman, D. Automated monitoring using acoustical sensors for insects in farm-stored wheat. J. Econ. Entomol. 1996, 89, 211–217. [Google Scholar] [CrossRef]
- Mankin, R.W. Eavesdropping on coconut rhinoceros beetles, red palm weevils, Asian longhorned beetles and other invasive travellers. In Proceedings of the Meetings on Acoustics 162ASA, San Diego, CA, USA, 31 October–4 November 2011; Volume 14. [Google Scholar]
- Njoroge, A.W.; Mankin, R.W.; Smith, B.W.; Baributsa, D. Effects of hermetic storage on adult Sitophilus oryzae L. (Coleoptera: Curculionidae) acoustic activity patterns and mortality. J. Econ. Entomol. 2017, 110, 2707–2715. [Google Scholar] [CrossRef]
- Njoroge, A.W.; Mankin, R.W.; Smith, B.W.; Baributsa, D. Oxygen consumption and acoustic activity of adult Callosobruchus maculatus (F.) (Coleoptera: Chrysomelidae) during hermetic storage. Insects 2018, 9, 45. [Google Scholar] [CrossRef]
- Kiobia, D.O.; Tumbo, S.D.; Cantillo, J.; Rohde, B.B.; Mallikarjunan, P.K.; Mankin, R.W. Characterization of sounds in maize produced by internally feeding insects: Investigations to develop inexpensive devices for detection of Prostephanus truncatus (Coleoptera: Bostrichidae) and Sitophilus zeamais (Coleoptera: Curculionidae) in small-scale storage facilities in sub-Saharan Africa. Fla. Entomol. 2015, 98, 405–409. [Google Scholar]
- Njoroge, A.W.; Affognon, H.; Mutungi, C.; Rohde, B.; Richter, U.; Hensel, O.; Mankin, R.W. Frequency and time pattern differences in acoustic signals produced by Prostephanus truncatus (Horn) (Coleoptera: Bostrichidae) and Sitophilus zeamais (Motschulsky) (Coleoptera: Curculionidae) in stored maize. J. Stored Prod. Res. 2016, 69, 31–40. [Google Scholar] [CrossRef]
- Njoroge, A.W.; Affognon, H.; Mutungi, C.; Richter, U.; Hensel, O.; Rohde, B.; Mankin, R.W. Bioacoustics of Acanthoscelides obtectus (Say) (Coleoptera: Chrysomelidae: Bruchinae) on Phaseolus vulgaris L. (Fabaceae). Fla. Entomol. 2017, 110, 2707–2715. [Google Scholar]
- ISO. Storage of Cereals and Pulses—Part 1 G: General Recommendations for the Keeping of Cereals; ISO 6322-1:1996; International Organization for Standardization: Geneva, Switzerland, 1996. [Google Scholar]
- Greig, D.J.; Reeves, M. Prevention of Post-Harvest Food Losses: A Training Manual; Food and Agriculture Organization: Rome, Italy, 1985. [Google Scholar]
- Mankin, R.W. Recent developments in the use of acoustic sensors and signal processing tools to target early infestations of red palm weevil in agricultural environments. Fla. Entomol. 2011, 94, 761–765. [Google Scholar] [CrossRef]
- Charif, R.A.; Waack, A.M.; Strickman, L.M. Raven Pro 1.3 User’s Manual; Cornell Laboratory of Ornithology: Ithaca, NY, USA, 2008. [Google Scholar]
- Mankin, R.W.; Mizrach, A.; Hetzroni, A.; Levsky, S.; Nakache, Y.; Soroker, V. Temporal and spectral features of sounds of wood-boring beetle larvae: Identifiable patterns of activity enable improved discrimination from background noise. Fla. Entomol. 2008, 91, 241–248. [Google Scholar] [CrossRef]
- Mankin, R.W.; Smith, M.T.; Tropp, J.M.; Atkinson, E.B.; Jong, D.Y. Detection of Anoplophora glabripennis (Coleoptera: Cerambycidae) larvae in different host trees and tissues by automated analyses of sound-impulse frequency and temporal patterns. J. Econ. Entomol. 2008, 101, 838–849. [Google Scholar] [CrossRef]
- Potamitis, I.; Ganchev, T.; Kontodimas, D. On automatic bioacoustic detection of pests: The cases of Rhynchophorus ferrugineus and Sitophilus oryzae. J. Econ Entomol. 2009, 102, 1681–1690. [Google Scholar] [CrossRef]
- Mankin, R.; Hagstrum, D. Chapter 22. Acoustic monitoring of insects. In Stored Product Protection; Hagstrum, D.W., Phillips, T.W., Cuperus, G., Eds.; Kansas State Univ. Press: Manhattan, KS, USA, 2011; Publication S156-22. [Google Scholar]
- Flynn, T.; Salloum, H.; Hull-Sanders, H.; Sedunov, A.; Sedunov, N.; Sinelnikov, Y.; Sutin, A.; Masters, D. Acoustic methods of invasive species detection in agriculture shipments. In Proceedings of the IEEE Symposium on Technologies for Homeland Security (HST), Waltham, MA, USA, 10–11 May 2016; pp. 1–5. [Google Scholar]
- Mankin, R.W.; Shuman, D.; Weaver, D.K. Thermal treatments to increase acoustic detectability of Sitophilus oryzae (Coleoptera: Curculionidae) in stored grain. J. Econ. Entomol. 1999, 92, 453–462. [Google Scholar] [CrossRef]
- Navarro, S. The use of modified and controlled atmospheres for the disinfestation of stored products. J. Pest Sci. 2012, 85, 301–322. [Google Scholar] [CrossRef]
- Murdock, L.L.; Baoua, I.B. On Purdue Improved Cowpea Storage (PICS) technology, background, mode of action, future prospects. J. Stored Prod. Res. 2014, 58, 3–11. [Google Scholar] [CrossRef]
- Kardous, C.A.; Shaw, P.B. Evaluation of smartphone sound measurement applications (apps) using external microphones—A follow-up study. J. Acoust. Soc. Am. Express Lett. 2016, 140, EL327–EL333. [Google Scholar] [CrossRef]
- Mukundarajan, H.; Hol, F.J.J.; Castillo, E.A.; Newby, C.; Prakash, M. Using mobile phones as acoustic sensors for high-throughput mosquito surveillance. eLife 2017, 6, e27854. [Google Scholar] [CrossRef]
- Zhu, C.; Wang, J.; Liu, H.; Mi, H. Insect identification and counting in stored grain: Image processing approach and application embedded in smart phones. Mob. Inform. Syst. 2018, 2018, 5491706. [Google Scholar] [CrossRef]
| Insect Category | Location | ||||
|---|---|---|---|---|---|
| Kiganjo | Nairobi | Nakuru | Sagana | Thika | |
| Prostephanus truncatus | 37.69 ± 13.80aA | 0.25 ± 0.25aB | 0.89 ± 0.54aB | 0.57 ± 0.42aB | 0.00 ± 0.00aB |
| Sitophilus zeamais | 6.23 ± 5.82bA | 20.42 ± 6.71bB | 24.56 ± 8.70bB | 0.00 ± 0.00aC | 0.25 ± 0.25aC |
| Tribolium castaneum | 39.08 ± 7.37aA | 39.42 ± 4.75cA | 27.11 ± 7.64bB | 5.86 ± 1.74bC | 12.50 ± 1.84bD |
| Other | 4.07 ± 0.78bA | 0.08 ± 0.08aB | 0.56 ± 0.44aB | 0.00 ± 0.00aB | 0.00 ± 0.00aB |
| Mixed larvae | 7.54 ± 1.75bA | 0.00 ± 0.00aB | 3.67 ± 1.09aC | 0.14 ± 0.14aB | 0.42 ± 0.42aB |
| Location | Mean ± SEM of Acoustic Parameter Measured by AEC or IMC System | |||||
|---|---|---|---|---|---|---|
| Rb | Nb | Rbimp | ||||
| AEC | IMC | AEC | IMC | AEC | IMC | |
| Kiganjo | 1.204a ± 0.432 | 0.209b ± 0.085 | 11.52 ± 3.96 | 41.63 ± 19.86 | 15.32 ± 7.13 | 6.87 ± 4.29 |
| Nairobi | 0.581 ± 0.381 | 0.0446 ± 0.0170 | 4.84 ± 0.42 | 20.27 ± 11.43 | 2.91 ± 1.96 | 0.777 ± 0.367 |
| Nakuru | 0.358 ± 0.121 | 0.125 ± 0.056 | 32.26 ± 13.78 | 9.79 ± 3.45 | 15.18 ± 7.67 | 1.85 ± 1.02 |
| Thika | 0.200 ± 0.067 | - | 43.60 ± 22.94 | - | 7.23 ± 3.77 | |
| Sagana | 0.093 ± 0.034 | 0.094 ± 0.038 | 35.44 ± 13.24 | 11.41 ± 8.45 | 5.38 ± 3.26 | 1.28 ± 0.64 |
| Acoustic Parameter | Df (Parameter, Error) | F | p |
|---|---|---|---|
| AEC Rb | 4, 27 | 2.22 | 0.094 |
| IMC Rb | 3, 22 | 1.50 | 0.242 |
| AEC Nb | 4, 27 | 1.90 | 0.139 |
| IMC Nb | 3, 22 | 1.17 | 0.343 |
| AEC Rbimp | 4, 27 | 1.18 | 0.340 |
| IMC Rbimp | 3, 22 | 1.22 | 0.325 |
| Acoustic Parameter | Df | t | p |
|---|---|---|---|
| Kiganjo Rb | 8 | 2.26 | 0.028 * |
| Nakuru Rb | 5 | 1.75 | 0.117 |
| Nairobi Rb | 7 | 1.40 | 0.210 |
| Sagana Rb | 6 | 0.008 | 0.992 |
| Kiganjo Nb | 8 | 1.48 | 0.178 |
| Nakuru Nb | 5 | 1.58 | 0.181 |
| Nairobi Nb | 7 | 1.35 | 0.226 |
| Sagana Nb | 6 | 1.75 | 0.132 |
| Kiganjo Rbimp | 8 | 1.02 | 0.327 |
| Nakuru Rbimp | 5 | 1.72 | 0.157 |
| Nairobi Rbimp | 7 | 1.07 | 0.323 |
| Sagana Rbimp | 6 | 1.24 | 0.245 |
| Model | Intercept ± SEM | t for Intercept | p > t | Slope ± SEM | t for Slope | p > t |
|---|---|---|---|---|---|---|
| aecrb = Tc | 0.326 ± 0.210 | 1.55 | 0.137 | 0.0132 ± 0.0039 * | 3.34 | 0.004 |
| imcrb = Tc | 0.078 ± 0.039 | 2.0 | 0.063 | 0.0016 ± 0.0007 * | 2.13 | 0.047 |
| imcrb = aecrb | 0.070 ± 0.041 | 1.72 | 0.102 | 0.073 ± 0.033 * | 2.16 | 0.044 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Njoroge, A.; Affognon, H.; Richter, U.; Hensel, O.; Rohde, B.; Chen, D.; Mankin, R. Acoustic, Pitfall Trap, and Visual Surveys of Stored Product Insect Pests in Kenyan Warehouses. Insects 2019, 10, 105. https://doi.org/10.3390/insects10040105
Njoroge A, Affognon H, Richter U, Hensel O, Rohde B, Chen D, Mankin R. Acoustic, Pitfall Trap, and Visual Surveys of Stored Product Insect Pests in Kenyan Warehouses. Insects. 2019; 10(4):105. https://doi.org/10.3390/insects10040105
Chicago/Turabian StyleNjoroge, Anastasia, Hippolyte Affognon, Uwe Richter, Oliver Hensel, Barukh Rohde, Davie Chen, and Richard Mankin. 2019. "Acoustic, Pitfall Trap, and Visual Surveys of Stored Product Insect Pests in Kenyan Warehouses" Insects 10, no. 4: 105. https://doi.org/10.3390/insects10040105
APA StyleNjoroge, A., Affognon, H., Richter, U., Hensel, O., Rohde, B., Chen, D., & Mankin, R. (2019). Acoustic, Pitfall Trap, and Visual Surveys of Stored Product Insect Pests in Kenyan Warehouses. Insects, 10(4), 105. https://doi.org/10.3390/insects10040105





