Unraveling the Genetic Structure of the Coconut Scale Insect Pest (Aspidiotus rigidus Reyne) Outbreak Populations in the Philippines
Abstract
1. Introduction
2. Materials and Methods
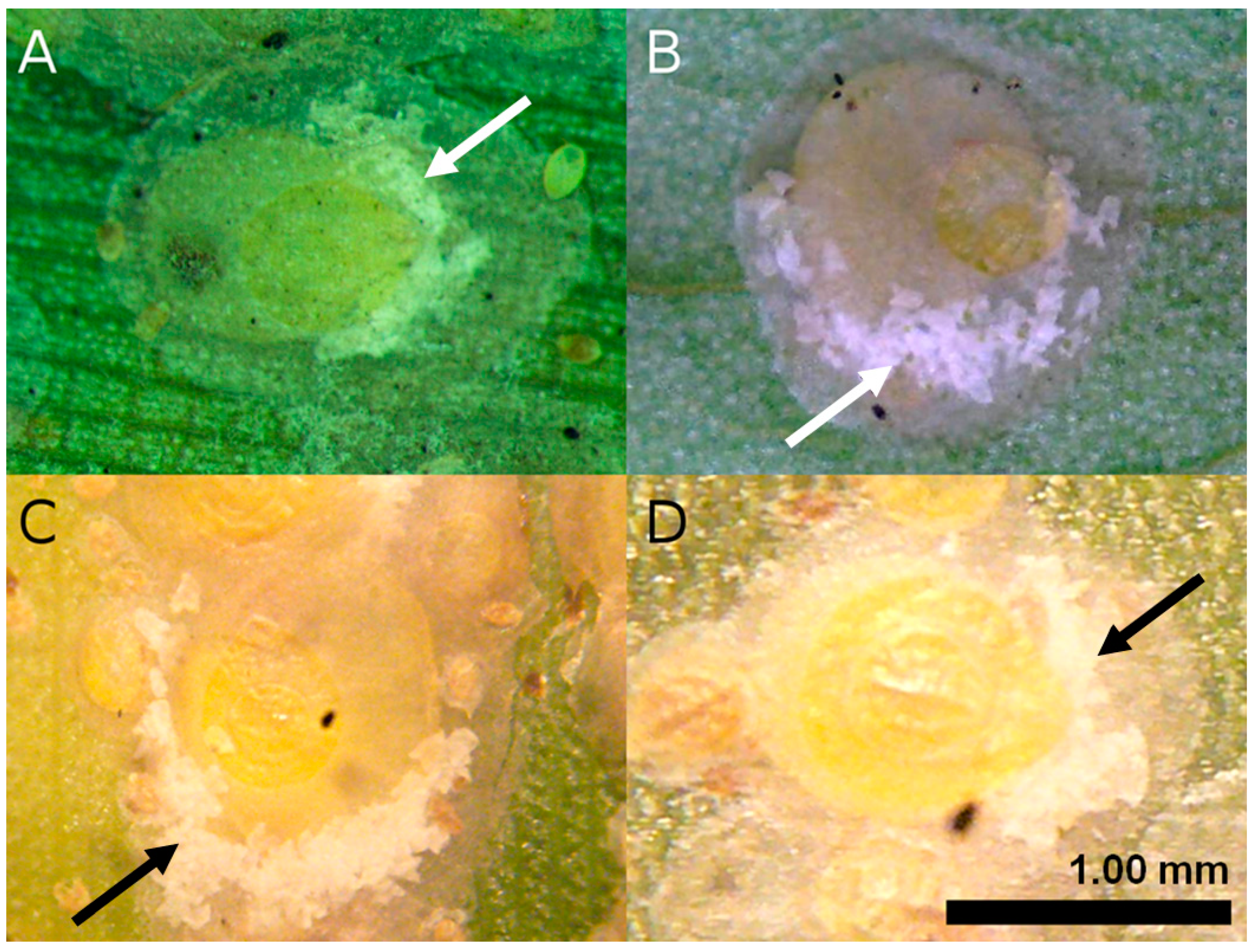
2.1. Sample Collection
2.2. DNA Extraction, PCR Amplification, and Sequencing
2.3. Genetic Diversity and Population Structure
2.4. Demographic Inference
2.5. Gene Flow Analysis and Median-joining Networks of Haplotypes
2.6. Phylogenetic Analysis
3. Results
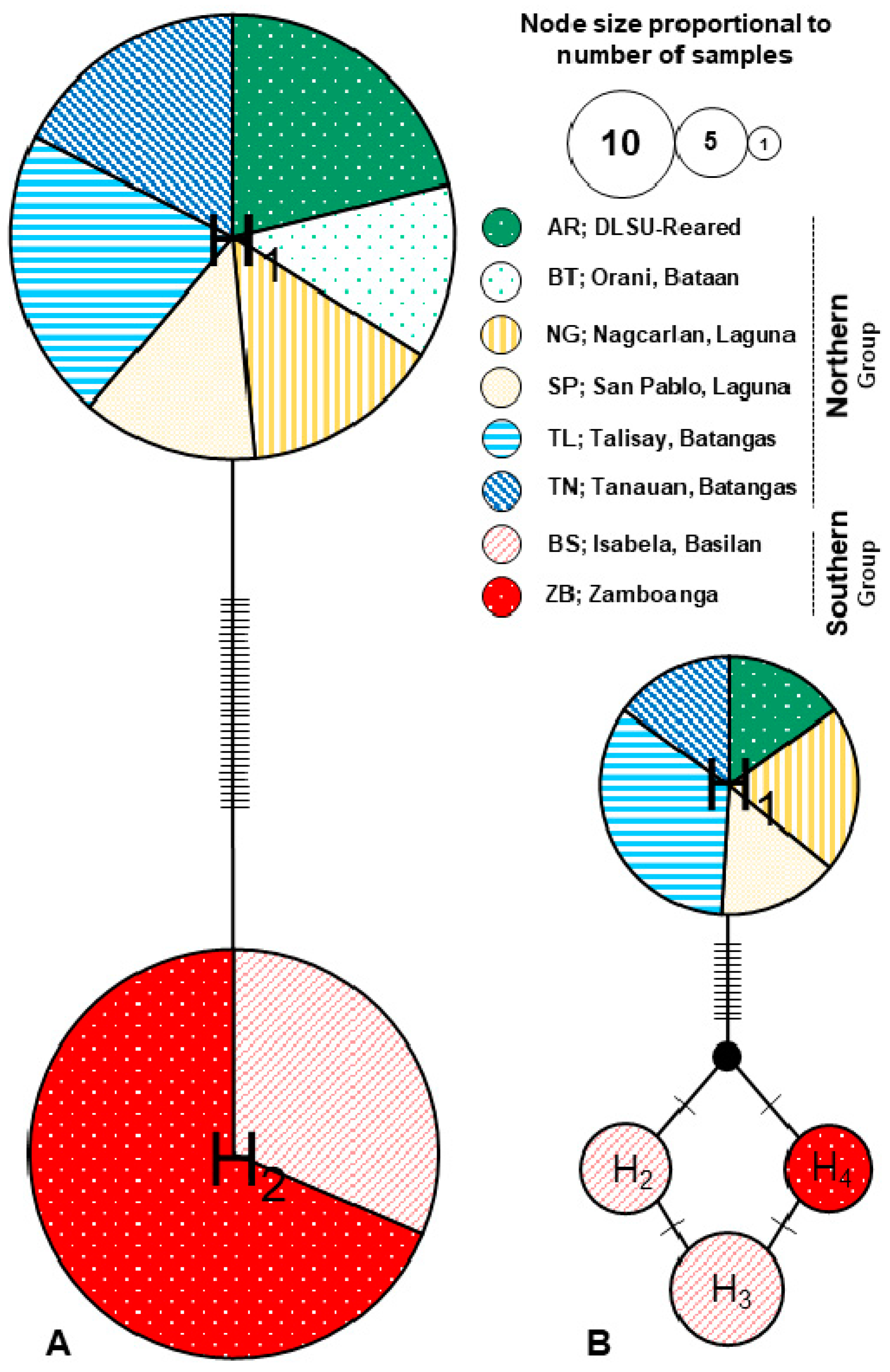
3.1. Genetic Diversity and Population Structure
3.2. Demographic History and Gene Flow
4. Discussion
4.1. Genetic Structure of A. rigidus: Evidence of Coconut Scale Insect “Superclones” in the Philippines
4.2. Demographic History: Resurgence of Resident Population or Recent Introduction?
4.3. Future Prospects: Applications for Biological Control
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Berryman, A.A. The theory and classification of outbreaks. In Insect Outbreaks; Academic Press, Inc.: San Diego, CA, USA, 1987; pp. 3–30. [Google Scholar]
- Risch, S.J. Agricultural ecology and insect outbreaks. In Insect Outbreaks; Academic Press, Inc.: San Diego, CA, USA, 1987; pp. 217–238. [Google Scholar]
- Ziska, L.H.; Blumenthal, D.M.; Runion, G.B.; Hunt, E.R.; Diaz-Soltero, H. Invasive species and climate change: An agronomic perspective. Clim. Chang. 2011, 105, 13–42. [Google Scholar] [CrossRef]
- Handley, L.J.L.; Estoup, A.; Evans, D.M.; Thomas, C.E.; Lombaert, E.; Facon, B.; Aebi, A.; Roy, H.E. Ecological genetics of invasive alien species. BioControl 2011, 56, 409–428. [Google Scholar] [CrossRef]
- Strayer, D.L.; D’Antonio, C.M.; Essl, F.; Fowler, M.S.; Geist, J.; Hilt, S.; Jarić, I.; Jöhnk, K.; Jones, C.G.; Latzka, A.W.; et al. Boom-bust dynamics in biological invasions: Towards an improved application of the concept. Ecol. Lett. 2017, 20, 1337–1350. [Google Scholar] [CrossRef] [PubMed]
- Prentis, P.J.; Wilson, J.R.; Dormontt, E.E.; Richardson, D.M.; Lowe, A.J. Adaptive evolution in invasive species. Trends. Plant. Sci. 2008, 13, 288–294. [Google Scholar] [CrossRef]
- Renault, D.; Laparie, M.; McCauley, S.J.; Bonte, D. Environmental adaptations, ecological filtering, and dispersal central to insect invasions. Annu. Rev. Ent. 2018, 63, 345–368. [Google Scholar] [CrossRef]
- Kobayashi, T.; Sakurai, T.; Sakakibara, M.; Watanabe, T. Multiple origins of outbreak populations of a native insect pest in an agro-ecosystem. Bull. Entomol. Res. 2011, 101, 313–324. [Google Scholar] [CrossRef]
- Reyne, A. Studies on a serious outbreak of Aspidiotus destructor rigidus in the coconut-palms of Sangi (North Celebes). Tijdschr. Entomol. 1948, 89, 83–123. [Google Scholar]
- Watson, G.W.; Adalla, C.B.; Shepard, B.M.; Carner, G.R. Aspidiotus rigidus Reyne (Hemiptera: Diaspididae): A devastating pest of coconut in the Philippines. Agric. For. Entomol. 2015, 17, 1–8. [Google Scholar] [CrossRef]
- Reyne, A. Notes on the biology of Comperiella unifasciata Ishii and its host Aspidiotus destructor rigidus nov. subspec. Tijdschr. Entomol. 1947, 88, 294–302. [Google Scholar]
- Manohar, E.C. Infestation Status, IPM Initiatives and Recovery Risk Assessment Protocol. In Proceedings of the NSTW Agri-Aqua Forum: Coping up with Cocolisap, SMX Convention Center, Mall of Asia, Pasay City, Philippines, 26 July 2015. [Google Scholar]
- Javier, E. Science to the Rescue of Coconut Industry. Philippine Daily Inquirer. 2014. Available online: http://opinion.inquirer.net/76268/science-to-the-rescue-of-coconut-industry (accessed on 8 February 2017).
- Lever, R.J.A.W. Pests of Coconut Palm, 2nd ed.; United Nations Food & Agriculture Organization: Rome, Italy, 1979. [Google Scholar]
- Velasquez, F.J. Some Philippine armored scale insects of the tribe Aspidiotini (Diaspididae, Homoptera). Philipp. Entomol. 1971, 2, 89–154. [Google Scholar]
- Wilby, A.; Thomas, M.B. Natural enemy diversity and pest control: Patterns of pest emergence with agricultural intensification. Ecol. Lett. 2002, 5, 353–360. [Google Scholar] [CrossRef]
- Rugman-Jones, P.F.; Hoddle, M.S.; Phillips, P.A.; Jeong, G.; Stouthamer, R. Strong genetic structure among populations of the invasive avocado pest Pseudacysta perseae (Heidemann) (Hemiptera: Tingidae) reveals the source of introduced populations. Biol. Invasions 2002, 14, 1079–1100. [Google Scholar] [CrossRef]
- Kébé, K.; Alvarez, N.; Espíndola, A.; Justy, F.; Olivieri, I.; Sembène, M. Insights into the genetic structure of the cowpea pest Callosobruchus maculatus in Africa. J. Pest. Sci. 2016, 89, 449–458. [Google Scholar] [CrossRef]
- Yang, W.Y.; Tang, X.T.; Ju, R.T.; Zhang, Y.; Du, Y.Z. The population genetic structure of Corythucha ciliata (Say) (Hemiptera: Tingidae) provides insights into its distribution and invasiveness. Sci. Rep. 2017, 7, 635. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Cai, W.; Luo, J.; Zhang, S.; Li, W.; Wang, C.; Lv, L.; Cui, J. Population genetic structure and expansion patterns of the cotton pest Adelphocoris fasciaticollis. J. Pest. Sci. 2018, 91, 539–550. [Google Scholar] [CrossRef]
- Provencher, L.M.; Morse, G.E.; Weeks, A.R.; Normark, B.B. Parthenogenesis in the Aspidiotus nerii complex (Hemiptera: Diaspididae): A single origin of a worldwide, polyphagous lineage associated with Cardinium bacteria. Ann. Entomol. Soc. Am. 2005, 98, 629–635. [Google Scholar] [CrossRef]
- Andersen, J.C. A Phylogenetic Analysis of Armored Scale Insects, based upon Nuclear, Mitochondrial, and Endosymbiont Gene Sequences. Master’s Thesis, University of Massachusetts Amherst, Amherst, MA, USA, 2014. [Google Scholar]
- Andersen, J.C.; Gruwell, M.E.; Morse, G.E.; Normark, B.B. Cryptic diversity in the Aspidiotus nerii complex in Australia. Ann. Entomol. Soc. Am. 2010, 103, 844–854. [Google Scholar] [CrossRef]
- Schneider, S.A.; Okusu, A.; Normark, B.B. Molecular phylogenetics of Aspidiotini armored scale insects (Hemiptera: Diaspididae) reveals rampant paraphyly, curious species radiations, and multiple origins of association with Melissotarsus ants (Hymenoptera: Formicidae). Mol. Phylogenet. Evol. 2018, 129, 291–303. [Google Scholar] [CrossRef]
- Park, D.S.; Leem, Y.J.; Hahn, K.W.; Suh, S.J.; Hong, K.J.; Oh, H.W. Molecular identification of mealybugs (Hemiptera: Pseudococcidae) found on Korean pears. J. Econ. Entomol. 2010, 103, 25–33. [Google Scholar] [CrossRef]
- Morse, G.E.; Normark, B.B. A molecular phylogenetic study of armoured scale insects (Hemiptera: Diaspididae). Syst. Entomol. 2006, 31, 338–349. [Google Scholar] [CrossRef]
- Palumbi, S.R. Nucleic acids II: The polymerase chain reaction. In Molecular Systematics; Oxford University Press: Oxford, England, 1996; pp. 205–247. [Google Scholar]
- Park, D.S.; Suh, S.J.; Hebert, P.D.; Oh, H.W.; Hong, K.J. DNA barcodes for two scale insect families, mealybugs (Hemiptera: Pseudococcidae) and armored scales (Hemiptera: Diaspididae). Bull. Entomol. Res. 2011, 101, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Talavera, G.; Castresana, J. Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst. Biol. 2007, 56, 564–577. [Google Scholar] [CrossRef] [PubMed]
- Rozas, J.; Ferrer-Mata, A.; Sánchez-DelBarrio, J.C.; Guirao-Rico, S.; Librado, P.; Ramos-Onsins, S.E.; Sánchez-Gracia, A. DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol. Biol. Evol. 2017, 34, 3299–3302. [Google Scholar] [CrossRef] [PubMed]
- Excoffier, L.; Lischer, H.E. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.X.; Li, W.H. Statistical tests of neutrality of mutations. Genetics 1993, 133, 693–709. [Google Scholar]
- Beerli, P. Comparison of Bayesian and maximum-likelihood inference of population genetic parameters. Bioinformatics 2005, 22, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Beerli, P.; Palczewski, M. Unified framework to evaluate panmixia and migration direction among multiple sampling locations. Genetics 2010, 185, 313–326. [Google Scholar] [CrossRef]
- Bandelt, H.J.; Forster, P.; Röhl, A. Median-joining networks for inferring intraspecific phylogenies. Mol. Biol. Evol. 1999, 16, 37–48. [Google Scholar] [CrossRef]
- Leigh, J.W.; Bryant, D. POPART: Full-feature software for haplotype network construction. Methods Ecol. Evol. 2015, 6, 1110–1116. [Google Scholar] [CrossRef]
- Darriba, D.; Taboada, G.L.; Doallo, R.; Posada, D. jModelTest 2: More models, new heuristics and parallel computing. Nat. Methods 2012, 9, 772. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, A.; Darriba, D.; Flouri, T.; Morel, B.; Stamatakis, A. RAxML-NG: A fast, scalable, and user-friendly tool for maximum likelihood phylogenetic inference. Bioinformatics 2019. [Google Scholar] [CrossRef] [PubMed]
- Abbot, P. A closer look at the spatial architecture of aphid clones. Mol. Ecol. 2011, 20, 4587–4589. [Google Scholar] [CrossRef] [PubMed]
- Normark, B.B.; Johnson, N.A. Niche explosion. Genetica 2011, 139, 551–564. [Google Scholar] [CrossRef] [PubMed]
- Loxdale, H.D.; Edwards, O.; Tagu, D.; Vorburger, C. Population genetic issues: New insights using conventional molecular markers and genomics tools. In Aphids as Crop Pests, 2nd ed.; van Emden, H.F., Harrington, R., Eds.; CABI: Oxford, UK, 2017; pp. 50–80. [Google Scholar]
- Figueroa, C.C.; Simon, J.C.; Le Gallic, J.F.; Prunier-Leterme, N.; Briones, L.M.; Dedryver, C.A.; Niemeyer, H.M. Genetic structure and clonal diversity of an introduced pest in Chile, the cereal aphid Sitobion avenae. Heredity 2005, 95, 24. [Google Scholar] [CrossRef]
- Peccoud, J.; Figueroa, C.C.; Silva, A.X.; Ramirez, C.C.; Mieuzet, L.; Bonhomme, J.; Stoeckel, S.; Plantegenest, M.; Simon, J.C. Host range expansion of an introduced insect pest through multiple colonizations of specialized clones. Mol. Ecol. 2008, 17, 4608–4618. [Google Scholar] [CrossRef]
- Cifuentes, D.; Chynoweth, R.; Bielza, P. Genetic study of Mediterranean and South American populations of tomato leafminer Tuta absoluta (Povolny, 1994) (Lepidoptera: Gelechiidae) using ribosomal and mitochondrial markers. Pest Manag. Sci. 2011, 67, 1155–1162. [Google Scholar] [CrossRef]
- Harrison, J.S.; Mondor, E.B. Evidence for an invasive aphid “superclone”: Extremely low genetic diversity in oleander aphid (Aphis nerii) populations in the southern United States. PLoS ONE 2011, 6, e17524. [Google Scholar] [CrossRef]
- Caron, V.; Ede, F.J.; Sunnucks, P. Unravelling the paradox of loss of genetic variation during invasion: Superclones may explain the success of a clonal invader. PLoS ONE 2014, 9, e97744. [Google Scholar] [CrossRef]
- Knowles, L.L.; Carstens, B.C.; Keat, M.L. Coupling genetic and ecological-niche models to examine how past population distributions contribute to divergence. Curr. Biol. 2007, 17, 940–946. [Google Scholar] [CrossRef]
- Hewitt, G.M. Genetic consequences of climatic oscillations in the Quaternary. Philos. Trans. R Soc. Lond. B Biol. Sci. 2004, 359, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, N.D.; Suarez, A.V.; Holway, D.A.; Case, T.J. Reduced genetic variation and the success of an invasive species. Proc. Nat. Acad. Sci. USA 2000, 97, 5948–5953. [Google Scholar] [CrossRef] [PubMed]
- Navia, D.; De Moraes, G.J.; Roderick, G.; Navajas, M. The invasive coconut mite Aceria guerreronis (Acari: Eriophyidae): Origin and invasion sources inferred from mitochondrial (16S) and nuclear (ITS) sequences. Bull. Entom. Res. 2005, 95, 505–516. [Google Scholar] [CrossRef]
- Lu, Y.; Wu, K.; Jiang, Y.; Xia, B.; Li, P.; Feng, H.; Wyckhuys, K.A.; Guo, Y. Mirid bug outbreaks in multiple crops correlated with wide-scale adoption of Bt cotton in China. Science 2010, 328, 1151–1154. [Google Scholar] [CrossRef]
- Bommarco, R.; Miranda, F.; Bylund, H.; Björkman, C. Insecticides suppress natural enemies and increase pest damage in cabbage. J. Econ. Entomol. 2011, 104, 782–791. [Google Scholar] [CrossRef]
- Ward, S.F.; Aukema, B.H. Anomalous outbreaks of an invasive defoliator and native bark beetle facilitated by warm temperatures, changes in precipitation, and interspecific interactions. Ecography 2019. [Google Scholar] [CrossRef]
- Schwartzberg, E.G.; Jamieson, M.A.; Raffa, K.F.; Reich, P.B.; Montgomery, R.A.; Lindroth, R.L. Simulated climate warming alters phenological synchrony between an outbreak insect herbivore and host trees. Oecologia 2014, 175, 1041–1049. [Google Scholar] [CrossRef]
- Ahern, R.G.; Hawthorne, D.J.; Raupp, M.J. Founder effects and phenotypic variation in Adelges cooleyi, an insect pest introduced to the eastern United States. Biol. Invasions 2009, 11, 959. [Google Scholar] [CrossRef]
- Burdon, J.J.; Thrall, P.H. Genetic structure of natural plant and pathogen populations. In Genetics, Evolution, and Biological Control; Ehler, L.E., Sforza, R., Mateille, T., Eds.; CABI Publishing: Wallingford, UK, 2003; pp. 1–17. [Google Scholar]
- Almarinez, B.J.M.; Amalin, D.M.; Carandang, J.S.R.; Navasero, M.V.; Navasero, M.M. First Philippine record of the parasitoid, Comperiella sp. (Hymenoptera: Encyrtidae): A potential biocontrol agent against Aspidiotus rigidus (Hemiptera: Diaspididae). J. Appl. Entomol. 2015, 139, 237–240. [Google Scholar] [CrossRef]
- Navasero, M.M.; Corpuz-Raros, L.A.; Navasero, M.V.; Cayabyab, B.F. Survey of mites (Acari) associated with outbreak populations of Aspidiotus rigidus Reyne (Hemiptera: Diaspididae) on coconut in Calabarzon. Philipp. Entomol. 2015, 29, 104–113. [Google Scholar]
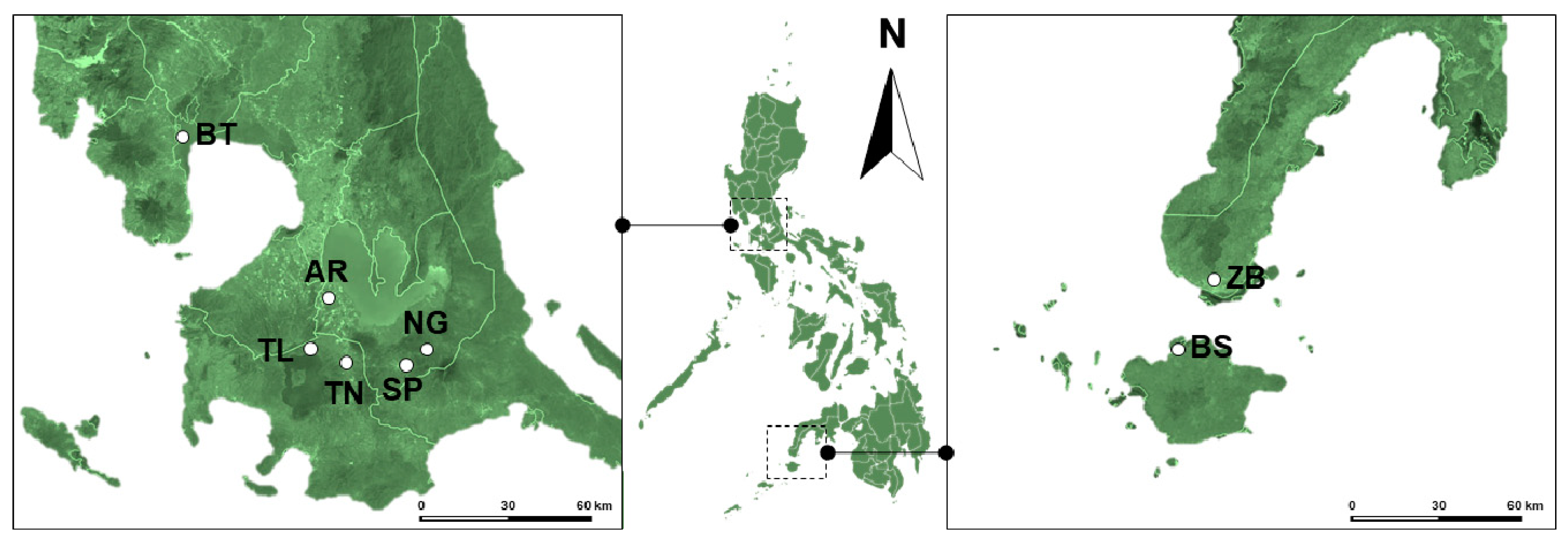
| Locality | Code | Collection Date | mtCOI | EF-1α | |||
|---|---|---|---|---|---|---|---|
| N | H | N | H | ||||
| Northern Region | DLSU-STC, Laguna a | AR | July 2017 | 35 | H1 | 8 | H1 |
| Orani, Bataan | BT | September 2015 | 21 | H1 | 0 | — | |
| Nagcarlan, Laguna | NG | January 2015 | 24 | H1 | 11 | H1 | |
| San Pablo, Laguna | SP | December 2014 | 21 | H1 | 8 | H1 | |
| Talisay, Batangas | TL | December 2014 | 35 | H1 | 18 | H1 | |
| Tanauan, Batangas | TN | December 2014 | 29 | H1 | 8 | H1 | |
| Southern Region | Isabela, Basilan | BS | November 2016 | 44 | H2 | 16 | H2; H3 |
| Zamboanga City, Zamboanga | ZB | April 2017 | 96 | H2 | 6 | H4 | |
| Gene | Group | N | S | h | Haplotype a | k | Hd (SD) | Pi (SD) | Db | Fs | D *b | F * |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| mtCOI | Northern | 165 | — | — | H1 | — | — | — | — | — | — | — |
| Southern | 140 | — | — | H2 | — | — | — | — | — | — | — | |
| All | 305 | 31 | 2 | — | 15.45 | 0.4980 (0.005) | 0.0239 (0.00025) | 5.85 *** | 59.59 | 2.04 ** | 4.43 | |
| EF-1α | Northern | 53 | — | — | H1 | — | — | — | — | — | — | — |
| Southern | 22 | 2 | 3 | H2; H3; H4 | 0.87 | 0.6880 (0.039) | 0.0009 (0.00010) | 1.33 | 0.99 | 0.85 | 1.13 | |
| All | 75 | 14 | 4 | — | 5.71 | 0.4770 (0.064) | 0.0057 (0.00061) | 2.83 ** | 12.81 | 1.55 * | 2.38 |
| Gene | Source of Variation | df. | Sum of Squares | Variance Components | Percentage of Variation | Fixation Indices |
|---|---|---|---|---|---|---|
| mtCOI | Among groups | 1 | 2347.869 | 15.50000 Va | 100 | FCT = 1.00000 * |
| Among populations within groups | 6 | 0 | 0.00000 Vb | 0 | FSC = 0.00000 | |
| Within populations | 297 | 0 | 0.00000 Vc | 0 | FST = 1.00000 ** | |
| EF-1α | Among groups | 1 | 202.010 | 6.45405 Va | 97.63 | FCT = 0.97630 * |
| Among populations within groups | 5 | 5.199 | 0.09876 Vb | 1.49 | FSC = 0.63040 ** | |
| Within populations | 68 | 3.938 | 0.05790 Vc | 0.88 | FST = 0.99124 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrana, J.M.; Ishitani, N.; Carvajal, T.M.; Almarinez, B.J.M.; Barrion, A.T.; Amalin, D.M.; Watanabe, K. Unraveling the Genetic Structure of the Coconut Scale Insect Pest (Aspidiotus rigidus Reyne) Outbreak Populations in the Philippines. Insects 2019, 10, 374. https://doi.org/10.3390/insects10110374
Serrana JM, Ishitani N, Carvajal TM, Almarinez BJM, Barrion AT, Amalin DM, Watanabe K. Unraveling the Genetic Structure of the Coconut Scale Insect Pest (Aspidiotus rigidus Reyne) Outbreak Populations in the Philippines. Insects. 2019; 10(11):374. https://doi.org/10.3390/insects10110374
Chicago/Turabian StyleSerrana, Joeselle M., Naoto Ishitani, Thaddeus M. Carvajal, Billy Joel M. Almarinez, Alberto T. Barrion, Divina M. Amalin, and Kozo Watanabe. 2019. "Unraveling the Genetic Structure of the Coconut Scale Insect Pest (Aspidiotus rigidus Reyne) Outbreak Populations in the Philippines" Insects 10, no. 11: 374. https://doi.org/10.3390/insects10110374
APA StyleSerrana, J. M., Ishitani, N., Carvajal, T. M., Almarinez, B. J. M., Barrion, A. T., Amalin, D. M., & Watanabe, K. (2019). Unraveling the Genetic Structure of the Coconut Scale Insect Pest (Aspidiotus rigidus Reyne) Outbreak Populations in the Philippines. Insects, 10(11), 374. https://doi.org/10.3390/insects10110374






