The Neurophysiological Bases of the Impact of Neonicotinoid Pesticides on the Behaviour of Honeybees
Abstract
:1. Introduction
2. Neonicotinoids as Agonists of nAchRs
2.1. Structure of nAchRs and Binding Properties
2.2. Location of nAchRs in the Brain
2.3. Neuronal Depolarization after Neonicotinoid Binding to nAchR
3. Neuronal Plasticity Following Neonicotinoid Exposure
3.1. Changes in Gene Expression Levels
3.2. Impact on Brain Function
3.3. Impact on Brain Structure
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Simon-Delso, N.; Amaral-Rogers, V.; Belzunces, L.P.; Bonmatin, J.M.; Chagnon, M.; Downs, C.; Furlan, L.; Gibbons, D.W.; Giorio, C.; Girolami, V.; et al. Systemic insecticides (neonicotinoids and fipronil): Trends, uses, mode of action and metabolites. Environ. Sci. Pollut. Res. Int. 2015, 22, 5–34. [Google Scholar] [CrossRef] [PubMed]
- Tomizawa, M.; Casida, J.E. Neonicotinoid Insecticide Toxicology: Mechanisms of Selective Action. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 247–268. [Google Scholar] [CrossRef]
- Blacquière, T.; Smagghe, G.; van Gestel, C.A.M.; Mommaerts, V. Neonicotinoids in bees: A review on concentrations, side-effects and risk assessment. Ecotoxicology 2012, 21, 973–992. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, E.A.D.; Mulhauser, B.; Mulot, M.; Mutabazi, A.; Glauser, G.; Aebi, A. A worldwide survey of neonicotinoids in honey. Science 2017, 358, 109–111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Commission. Commission implementing regulation (EU) 2018/783 of 29 May 2018 amending Implementing Regulation (EU) No 540/2011 as regards the conditions of approval of the active substance imidacloprid. Off. J. Eur. Union 2018, 61, 31–34. [Google Scholar]
- European Commission. Commission implementing regulation (EU) 2018/784 of 29 May 2018 amending Implementing Regulation (EU) No 540/2011 as regards the conditions of approval of the active substance clothianidin. Off. J. Eur. Union 2018, 61, 35–39. [Google Scholar]
- European Commission. Commission implementing regulation (EU) 2018/785 of 29 May 2018 amending Implementing Regulation (EU) No 540/2011 as regards the conditions of approval of the active substance thiamethoxam (Text with EEA relevance.). Off. J. Eur. Union 2018, 61, 40–44. [Google Scholar]
- Klein, S.; Cabirol, A.; Devaud, J.-M.; Barron, A.B.; Lihoreau, M. Why bees are so vulnerable to environmental stressors. Trends Ecol. Evol. 2017, xx, 1–11. [Google Scholar] [CrossRef]
- Godfray, H.C.J.; Blacquière, T.; Field, L.M.; Hails, R.S.; Petrokofsky, G.; Potts, S.G.; Raine, N.E.; Vanbergen, A.J.; Mclean, A.R. A restatement of the natural science evidence base concerning neonicotinoid insecticides and insect pollinators. Proc. R. Soc. B 2014, 281, 20140558. [Google Scholar] [CrossRef] [Green Version]
- Godfray, H.C.J.; Blacquière, T.; Field, L.M.; Hails, R.S.; Potts, S.G.; Raine, N.E.; Vanbergen, A.J.; McLean, A.R. A restatement of recent advances in the natural science evidence base concerning neonicotinoid insecticides and insect pollinators. Proc. R. Soc. B Biol. Sci. 2015, 282, 20151821. [Google Scholar] [CrossRef] [Green Version]
- Brown, L.A.; Ihara, M.; Buckingham, S.D.; Matsuda, K.; Sattelle, D.B. Neonicotinoid insecticides display partial and super agonist actions on native insect nicotinic acetylcholine receptors. J. Neurochem. 2006, 99, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Pitman, M. Transmitter substances in insects: A review. Comp. gen. Pharmac. 1971, 2, 347–371. [Google Scholar] [CrossRef]
- Matsuda, K.; Buckingham, S.D.; Kleier, D.; Rauh, J.J.; Grauso, M.; Sattelle, D.B. Neonicotinoids: insecticides acting on insect nicotinic acetylcholine receptors. Trends Pharmacol. Sci. 2001, 22, 573–580. [Google Scholar] [CrossRef]
- Siviter, H.; Koricheva, J.; Brown, M.J.F.; Leadbeater, E. Quantifying the impact of pesticides on learning and memory in bees. J. Appl. Ecol. 2018, 55, 2812–2821. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gauthier, M. State of the Art on Insect Nicotinic Acetylcholine Receptor Function in Learning and Memory. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2010; Volume 683, pp. 97–115. ISBN 9781441964441. [Google Scholar]
- Hebb, D.O. The Organization of Behavior: A Neuropsychological Theory; Wiley Book in Clinical Psychology: New York, NY, USA, 1949. [Google Scholar]
- Turrigiano, G.G.; Nelson, S.B. Hebb and homeostasis in neuronal plasticity. Curr. Opin. Neurobiol. 2000, 10, 358–364. [Google Scholar] [CrossRef]
- Thompson, H.M. Behavioural effects of pesticides in bees--their potential for use in risk assessment. Ecotoxicology 2003, 12, 317–330. [Google Scholar] [CrossRef]
- Taillebois, E.; Cartereau, A.; Jones, A.K.; Thany, S.H. Neonicotinoid insecticides mode of action on insect nicotinic acetylcholine receptors using binding studies. Pestic. Biochem. Physiol. 2018, 151, 59–66. [Google Scholar] [CrossRef] [Green Version]
- Jones, A.K.; Raymond-Delpech, V.; Thany, S.H.; Gauthier, M.; Sattelle, D.B. The nicotinic acetylcholine receptor gene family of the honey bee, Apis mellifera. Genome Res. 2006, 16, 1422–1430. [Google Scholar] [CrossRef]
- Thany, S.H.; Lenaers, G.; Raymond-Delpech, V.; Sattelle, D.B.; Lapied, B. Exploring the pharmacological properties of insect nicotinic acetylcholine receptors. Trends Pharmacol. Sci. 2007, 28, 14–22. [Google Scholar] [CrossRef]
- Schroeder, M.E.; Flattum, R.F. The mode of action and neurotoxic properties of the nitromethylene heterocycle insecticides. Pestic. Biochem. Physiol. 1984, 22, 148–160. [Google Scholar] [CrossRef]
- Bai, D.; Lummis, S.C.R.; Leicht, W.; Breer, H.; Sattelle, D.B. Actions of imidacloprid and a related nitromethylene on cholinergic receptors of an identified insect motor neurone. Pestic. Sci. 1991, 33, 197–204. [Google Scholar] [CrossRef]
- Liu, M.Y.; Casida, J.E. High Affinity Binding of [3H]Imidacloprid in the Insect Acetylcholine Receptor. Pestic. Biochem. Physiol. 1993, 46, 40–46. [Google Scholar] [CrossRef]
- Lansdell, S.J.; Millar, N.S. The influence of nicotinic receptor subunit composition upon agonist, α-bungarotoxin and insecticide (imidacloprid) binding affinity. Neuropharmacology 2000, 39, 671–679. [Google Scholar] [CrossRef]
- Zhang, N.; Tomizawa, M.; Casida, J.E. Drosophila nicotinic receptors: Evidence for imidacloprid insecticide and α-bungarotoxin binding to distinct sites. Neurosci. Lett. 2004, 371, 56–59. [Google Scholar] [CrossRef] [PubMed]
- Taillebois, E.; Beloula, A.; Quinchard, S.; Jaubert-Possamai, S.; Daguin, A.; Servent, D.; Tagu, D.; Thany, S.H.; Tricoire-Leignel, H. Neonicotinoid Binding, Toxicity and Expression of Nicotinic Acetylcholine Receptor Subunits in the Aphid Acyrthosiphon pisum. PLoS ONE 2014, 9, e96669. [Google Scholar] [CrossRef] [PubMed]
- Buckingham, S.; Lapied, B.; Corronc, H.L.; Sattelle, F. Imidacloprid actions on insect neuronal acetylcholine receptors. J. Exp. Biol. 1997, 200, 2685–2692. [Google Scholar]
- Sun, H.; Liu, Y.; Li, J.; Cang, X.; Bao, H.; Liu, Z. The potential subunits involved in two subtypes of α-Bgt-resistant nAChRs in cockroach dorsal unpaired median (DUM) neurons. Insect Biochem. Mol. Biol. 2017, 81, 32–40. [Google Scholar] [CrossRef]
- Benzidane, Y.; Goven, D.; Abd-Ella, A.A.; Deshayes, C.; Lapied, B.; Raymond, V. Subchronic exposure to sublethal dose of imidacloprid changes electrophysiological properties and expression pattern of nicotinic acetylcholine receptor subtypes in insect neurosecretory cells. Neurotoxicology 2017, 62, 239–247. [Google Scholar] [CrossRef]
- Kreissl, S.; Bicker, G. Histochemistry of acetylcholinesterase and immunocytochemistry of an acetylcholine receptor-like antigen in the brain of the honeybee. J. Comp. Neurol. 1989, 286, 71–84. [Google Scholar] [CrossRef] [Green Version]
- Goldberg, F.; Grünewald, B.; Rosenboom, H.; Menzel, R. Nicotinic acetylcholine currents of cultured Kenyon cells from the mushroom bodies of the honey bee Apis mellifera. J. Physiol. 1999, 514, 759–768. [Google Scholar] [CrossRef]
- Dupuis, J.P.; Gauthier, M.; Raymond-Delpech, V. Expression patterns of nicotinic subunits α2, α7, α8, and β1 affect the kinetics and pharmacology of ACh-induced currents in adult bee olfactory neuropiles. J. Neurophysiol. 2011, 106, 1604–1613. [Google Scholar] [CrossRef] [PubMed]
- Thany, S.H.; Crozatier, M.; Raymond-Delpech, V.; Gauthier, M.; Lenaers, G. Apisα2, Apisα7-1 and Apisα7-2: Three new neuronal nicotinic acetylcholine receptor α-subunits in the honeybee brain. Gene 2005, 344, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Wang, M.; Kang, M.; Liu, L.; Guo, X.; Xu, B. Molecular cloning and characterization of two nicotinic acetylcholine receptor β subunit genes from Apis cerana cerana. Arch. Insect Biochem. Physiol. 2011, 77, 163–178. [Google Scholar] [CrossRef] [PubMed]
- Oleskevich, S. Cholinergic Synaptic Transmission in Insect Mushroom Bodies In Vitro. J. Neurophysiol. 1999, 82, 1091–1096. [Google Scholar] [CrossRef] [PubMed]
- Barbara, G.S.; Grünewald, B.; Paute, S.; Gauthier, M.; Raymond-Delpech, V. Study of nicotinic acetylcholine receptors on cultured antennal lobe neurons from adult honeybee brains. Invertebr. Neurosci. 2008, 8, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Nauen, R.; Ebbinghaus-Kintscher, U.; Schmuck, R. Toxicity and nicotinic acetylcholine receptor interaction of imidacloprid and its metabolites inApis mellifera (Hymenoptera: Apidae). Pest Manag. Sci. 2001, 57, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Jepson, J.E.C.; Brown, L.A.; Sattelle, D.B. The actions of the neonicotinoid imidacloprid on cholinergic neurons of Drosophila melanogaster. Invertebr. Neurosci. 2006, 6, 33–40. [Google Scholar] [CrossRef]
- Déglise, P.; Grünewald, B.; Gauthier, M. The insecticide imidacloprid is a partial agonist of the nicotinic receptor of honeybee Kenyon cells. Neurosci. Lett. 2002, 321, 13–16. [Google Scholar] [CrossRef]
- Wüstenberg, D.G.; Grünewald, B. Pharmacology of the neuronal nicotinic acetylcholine receptor of cultured Kenyon cells of the honeybee, Apis mellifera. J. Comp. Physiol. A 2004, 190, 807–821. [Google Scholar]
- Moffat, C.; Buckland, S.T.; Samson, A.J.; McArthur, R.; Chamosa Pino, V.; Bollan, K.A.; Huang, J.T.J.; Connolly, C.N. Neonicotinoids target distinct nicotinic acetylcholine receptors and neurons, leading to differential risks to bumblebees. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef]
- Palmer, M.J.; Moffat, C.; Saranzewa, N.; Harvey, J.; Wright, G.A.; Connolly, C.N. Cholinergic pesticides cause mushroom body neuronal inactivation in honeybees. Nat. Commun. 2013, 4, 1634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, J.; Galligan, J.J.; Hollingworth, R.M. Agonist actions of neonicotinoids on nicotinic acetylcholine receptors expressed by cockroach neurons. Neurotoxicology 2007, 28, 829–842. [Google Scholar] [CrossRef] [PubMed]
- Decourtye, A.; Armengaud, C.; Renou, M.; Devillers, J.; Cluzeau, S.; Gauthier, M.; Pham-Delègue, M.-H. Imidacloprid impairs memory and brain metabolism in the honeybee (Apis mellifera L.). Pestic. Biochem. Physiol. 2004, 78, 83–92. [Google Scholar] [CrossRef]
- Christen, V.; Mittner, F.; Fent, K. Molecular Effects of Neonicotinoids in Honey Bees (Apis mellifera). Environ. Sci. Technol. 2016, 50, 4071–4081. [Google Scholar] [CrossRef]
- Shi, T.F.; Wang, Y.F.; Liu, F.; Qi, L.; Yu, L.S. Sublethal Effects of the Neonicotinoid Insecticide Thiamethoxam on the Transcriptome of the Honey Bees (Hymenoptera: Apidae). J. Econ. Entomol. 2017, 110, 2283–2289. [Google Scholar] [CrossRef]
- Markussen, M.D.K.; Kristensen, M. Cytochrome P450 monooxygenase-mediated neonicotinoid resistance in the house fly Musca domestica L. Pestic. Biochem. Physiol. 2010, 98, 50–58. [Google Scholar] [CrossRef]
- Wang, X.; Sun, H.; Zhang, Y.; Liu, C.; Liu, Z.; Jurenka, R.A. Transcriptional changes in nAChRs, interactive proteins and P450s in Locusta migratoria manilensis (Orthoptera: Acrididae) CNS in response to high and low oral doses of imidacloprid. J. Insect Sci. 2015, 15. [Google Scholar] [CrossRef]
- Cresswell, J.E. A meta-analysis of experiments testing the effects of a neonicotinoid insecticide (imidacloprid) on honey bees. Ecotoxicology 2011, 20, 149–157. [Google Scholar] [CrossRef]
- Peng, Y.-C.C.; Yang, E.-C.C. Sublethal dosage of imidacloprid reduces the microglomerular density of honey bee mushroom bodies. Sci. Rep. 2016, 6, 19298. [Google Scholar] [CrossRef]
- De Almeida Rossi, C.; Roat, T.C.; Tavares, D.A.; Cintra-Socolowski, P.; Malaspina, O. Brain Morphophysiology of Africanized Bee Apis mellifera Exposed to Sublethal Doses of Imidacloprid. Arch. Environ. Contam. Toxicol. 2013, 65, 234–243. [Google Scholar] [CrossRef] [Green Version]
- Catae, A.F.; Roat, T.C.; Pratavieira, M.; da Silva Menegasso, A.R.; Palma, M.S.; Malaspina, O. Exposure to a sublethal concentration of imidacloprid and the side effects on target and nontarget organs of Apis mellifera (Hymenoptera, Apidae). Ecotoxicology 2018, 27, 109–121. [Google Scholar] [CrossRef] [PubMed]
- Bantz, A.; Camon, J.; Froger, J.A.; Goven, D.; Raymond, V. Exposure to sublethal doses of insecticide and their effects on insects at cellular and physiological levels. Curr. Opin. Insect Sci. 2018, 30, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Alptekin, S.; Bass, C.; Nicholls, C.; Paine, M.J.I.; Clark, S.J.; Field, L.; Moores, G.D. Induced thiacloprid insensitivity in honeybees (Apis mellifera L.) is associated with up-regulation of detoxification genes. Insect Mol. Biol. 2016, 25, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.C.; Chang, Y.W.; Lu, K.H.; Yang, E.C. Gene expression changes in honey bees induced by sublethal imidacloprid exposure during the larval stage. Insect Biochem. Mol. Biol. 2017, 88, 12–20. [Google Scholar] [CrossRef]
- Moffat, C.; Pacheco, J.G.; Sharp, S.; Samson, A.J.; Bollan, K.A.; Huang, J.; Buckland, S.T.; Connolly, C.N. Chronic exposure to neonicotinoids increases neuronal vulnerability to mitochondrial dysfunction in the bumblebee (Bombus terrestris). FASEB J. 2015. [Google Scholar] [CrossRef]
- Paoli, M.; Andrione, M.; Haase, A. Imaging Techniques in Insects. In Lateralized Brain Functions: Methods in Human and Non-Human Species; Rogers, L.J., Vallortigara, G., Eds.; Springer: New York, NY, USA, 2017; pp. 471–519. ISBN 978-1-4939-6725-4. [Google Scholar]
- Haase, A.; Rigosi, E.; Trona, F.; Anfora, G.; Vallortigara, G.; Antolini, R.; Vinegoni, C. In-vivo two-photon imaging of the honey bee antennal lobe. Biomed. Opt. Express 2010, 2, 131–138. [Google Scholar] [CrossRef] [Green Version]
- Galizia, C.G.; Sachse, S.; Rappert, A.; Menzel, R. The glomerular code for odor representation is species specific in the honeybee Apis mellifera. Nat. Neurosci. 1999, 2, 473–478. [Google Scholar] [CrossRef] [Green Version]
- Andrione, M.; Vallortigara, G.; Antolini, R.; Haase, A. Neonicotinoid-induced impairment of odour coding in the honeybee. Sci. Rep. 2016, 6, 38110. [Google Scholar] [CrossRef] [Green Version]
- Parkinson, R.H.; Gray, J.R. Neural conduction, visual motion detection, and insect flight behaviour are disrupted by low doses of imidacloprid and its metabolites. Neurotoxicology 2019, 72, 107–113. [Google Scholar] [CrossRef]
- Rigosi, E.; O’Carroll, D.C. The cholinergic pesticide imidacloprid impairs contrast and direction sensitivity in motion detecting neurons of an insect pollinator. bioRxiv 2018, 295576, 1–28. [Google Scholar]
- Withers, G.S.; Fahrbach, S.E.; Robinson, G.E. Selective neuroanatomical plasticity and division of labour in the honeybee. Nature 1993, 364, 238–240. [Google Scholar] [CrossRef] [PubMed]
- Muenz, T.S.; Groh, C.; Maisonnasse, A.; Le Conte, Y.; Plettner, E.; Rössler, W. Neuronal plasticity in the mushroom body calyx during adult maturation in the honeybee and possible pheromonal influences. Dev. Neurobiol. 2015, 75, 1368–1384. [Google Scholar] [CrossRef] [PubMed]
- Tomé, H.V.V.; Martins, G.F.; Lima, M.A.P.; Campos, L.A.O.; Guedes, R.N.C. Imidacloprid-Induced Impairment of Mushroom Bodies and Behavior of the Native Stingless Bee Melipona quadrifasciata anthidioides. PLoS ONE 2012, 7, e38406. [Google Scholar] [CrossRef] [PubMed]
- Rössler, W.; Spaethe, J.; Groh, C.; Peng, Y.C.; Yang, E.C. Pitfalls of using confocal-microscopy based automated quantification of synaptic complexes in honeybee mushroom bodies (response to Peng and Yang 2016). Sci. Rep. 2017, 7, 9786. [Google Scholar] [CrossRef] [Green Version]
- Lozano, V.; Armengaud, C.; Gauthier, M. Memory impairment induced by cholinergic antagonists injected into the mushroom bodies of the honeybee. J. Comp. Physiol. A 2001, 187, 249–254. [Google Scholar] [CrossRef]
- Gauthier, M.; Dacher, M.; Thany, S.H.; Niggebrügge, C.; Déglise, P.; Kljucevic, P.; Armengaud, C.; Grünewald, B. Involvement of α-bungarotoxin-sensitive nicotinic receptors in long-term memory formation in the honeybee (Apis mellifera). Neurobiol. Learn. Mem. 2006, 86, 164–174. [Google Scholar] [CrossRef]
- Louis, T.; Musso, P.Y.; De Oliveira, S.B.; Garreau, L.; Giurfa, M.; Raymond, V.; Gauthier, M. Amelα8 subunit knockdown the mushroom body vertical lobes impairs olfactory retrieval the honeybee, Apis mellifera. Eur. J. Neurosci. 2012, 36, 3438–3450. [Google Scholar] [CrossRef]
- Gronenberg, W. Subdivisions of hymenopteran mushroom body calyces by their afferent supply. J. Comp. Neurol. 2001, 435, 474–489. [Google Scholar] [CrossRef]
- Henry, M.; Béguin, M.; Requier, F.; Rollin, O.; Odoux, J.-F.; Aupinel, P.; Aptel, J.; Tchamitchian, S.; Decourtye, A. A common pesticide decreases foraging success and survival in honey bees. Science 2012, 336, 348–350. [Google Scholar] [CrossRef]
- Fischer, J.; Müller, T.; Spatz, A.-K.; Greggers, U.; Grünewald, B.; Menzel, R. Neonicotinoids Interfere with Specific Components of Navigation in Honeybees. PLoS ONE 2014, 9, e91364. [Google Scholar] [CrossRef]
- Jin, N.; Klein, S.; Leimig, F.; Bischoff, G.; Menzel, R. The neonicotinoid clothianidin interferes with navigation of the solitary bee Osmia cornuta in a laboratory test. J. Exp. Biol. 2015. [Google Scholar] [CrossRef] [PubMed]
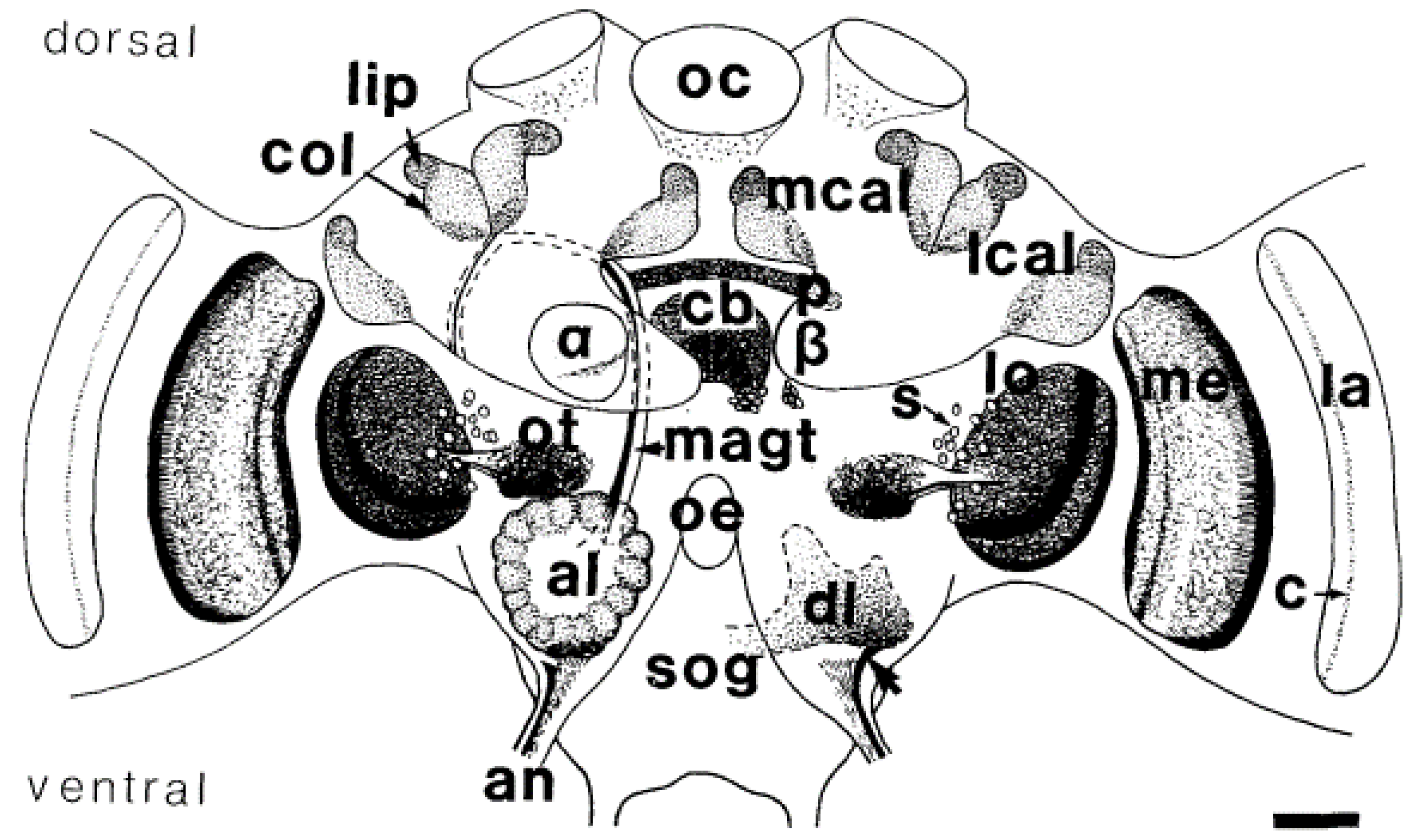
| Reference | Neonicotinoid | Treatment Duration | Minimal Dose | Effects | |
|---|---|---|---|---|---|
| Gene Expression | [46] | Acetamiprid | 24 h, 48 h, 72 h | 80,000 ppb | Increased expression of nAchRα1 after 72 h |
| Clothianidin | 0.3 ppb | Increased expression of nAchRα1 after 72 h | |||
| Imidacloprid | 3 ppb | Increased expression of nAchRα1 after 72 h | |||
| 3 ppb | Increased expression of nAchRα2 after 48 h, but not after 72 h | ||||
| Thiametoxam | 1 ppb | Increased expression of nAchRα1 after 48 h | |||
| 10 ppb | Increased expression of nAchRα1 after 72 h | ||||
| 1 ppb | Increased expression of nAchRα2 after 48 h, but not after 72 h | ||||
| [47] | Thiametoxam | 10 days | 10 ppb | 225 upregulated genes, 384 downregulated genes | |
| Increased expression of nAchRα9 and nAchRβ2 | |||||
| [35] * | Acetamiprid | 0.5 h, 1 h, 2 h, 3 h | 10,000 ppb | Increased expression of nAchRβ1 and nAchRβ2 at 0.5 h | |
| Imidacloprid | 1 h, 2 h, 3 h, 4 h | 10,000 ppb | Decreased expression of nAchRβ1 and nAchRβ2 at 1–2 h | ||
| Neuronal | [51] | Imidacloprid | Larval stage (effects | 10 ppb | Decreased synaptic bouton density in the MB lateral calyces |
| Structure | 20 days after eclosion) | 100 ppb | Decreased synaptic bouton density in the MB median calyces | ||
| [52] * | Imidacloprid | 1, 3, 5, 7 or 10 days | 800 ppb | Increased cell death after 1 day in the optic lobes | |
| 8100 ppb | Increased cell death after 1 day in the mushroom bodies | ||||
| [53] | Imidacloprid | 1, 4, 8 days | 14.6 ppb | Cellular alterations (mitochondria, chromatin, phagosomes) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabirol, A.; Haase, A. The Neurophysiological Bases of the Impact of Neonicotinoid Pesticides on the Behaviour of Honeybees. Insects 2019, 10, 344. https://doi.org/10.3390/insects10100344
Cabirol A, Haase A. The Neurophysiological Bases of the Impact of Neonicotinoid Pesticides on the Behaviour of Honeybees. Insects. 2019; 10(10):344. https://doi.org/10.3390/insects10100344
Chicago/Turabian StyleCabirol, Amélie, and Albrecht Haase. 2019. "The Neurophysiological Bases of the Impact of Neonicotinoid Pesticides on the Behaviour of Honeybees" Insects 10, no. 10: 344. https://doi.org/10.3390/insects10100344
APA StyleCabirol, A., & Haase, A. (2019). The Neurophysiological Bases of the Impact of Neonicotinoid Pesticides on the Behaviour of Honeybees. Insects, 10(10), 344. https://doi.org/10.3390/insects10100344






