Survival and Development of Striacosta albicosta (Smith) (Lepidoptera: Noctuidae) Immature Stages on Dry Beans, non-Bt, Cry1F, and Vip3A Maize
Abstract
:1. Introduction
2. Material and Methods
2.1. Insects, Plants and Laboratory Conditions
2.2. Larval Stage
2.3. Active Larval Stage
2.4. Inactive Larval Period (Prepupal Period)
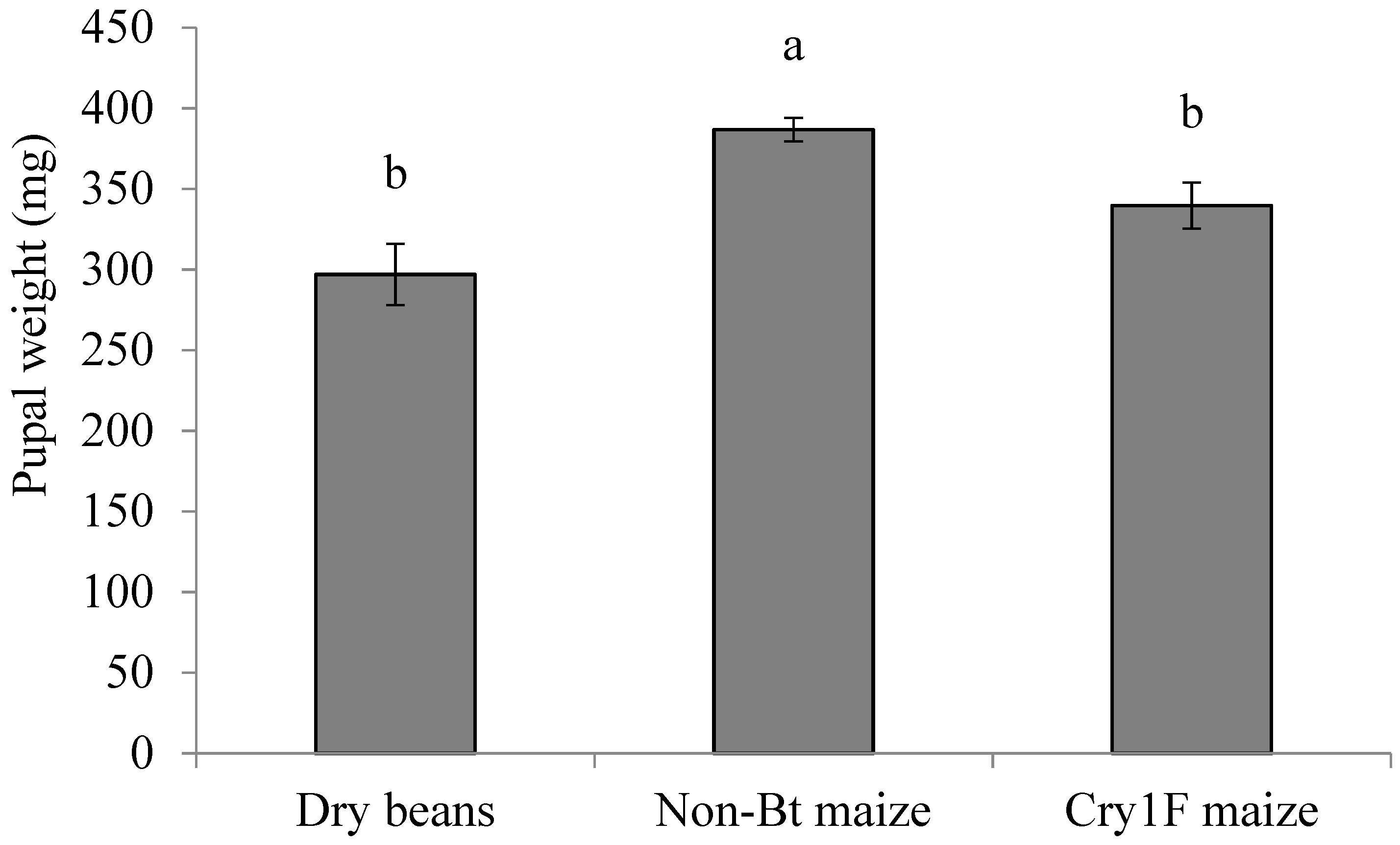
2.5. Pupal Stage
2.6. Data Analysis
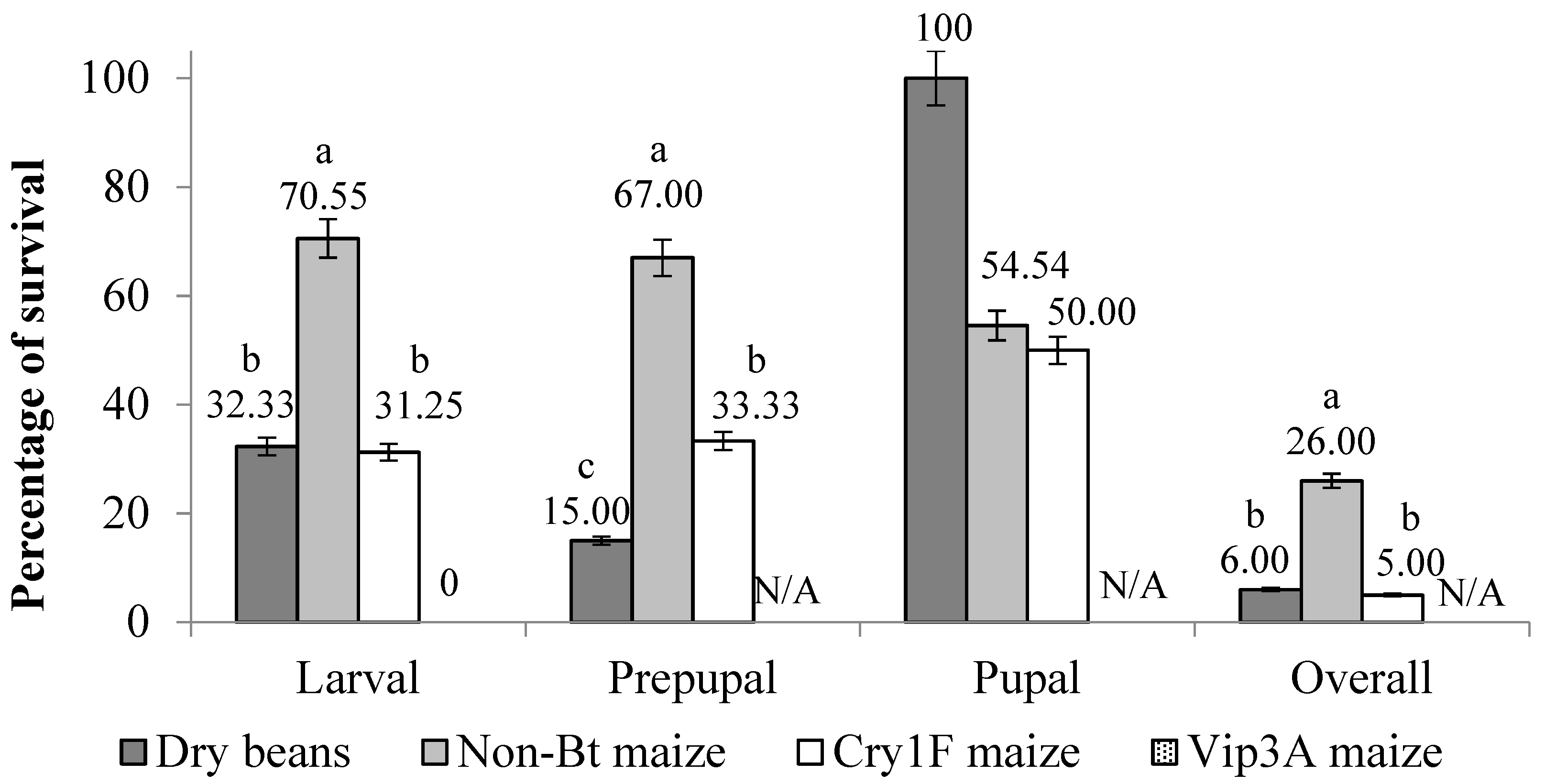
3. Results
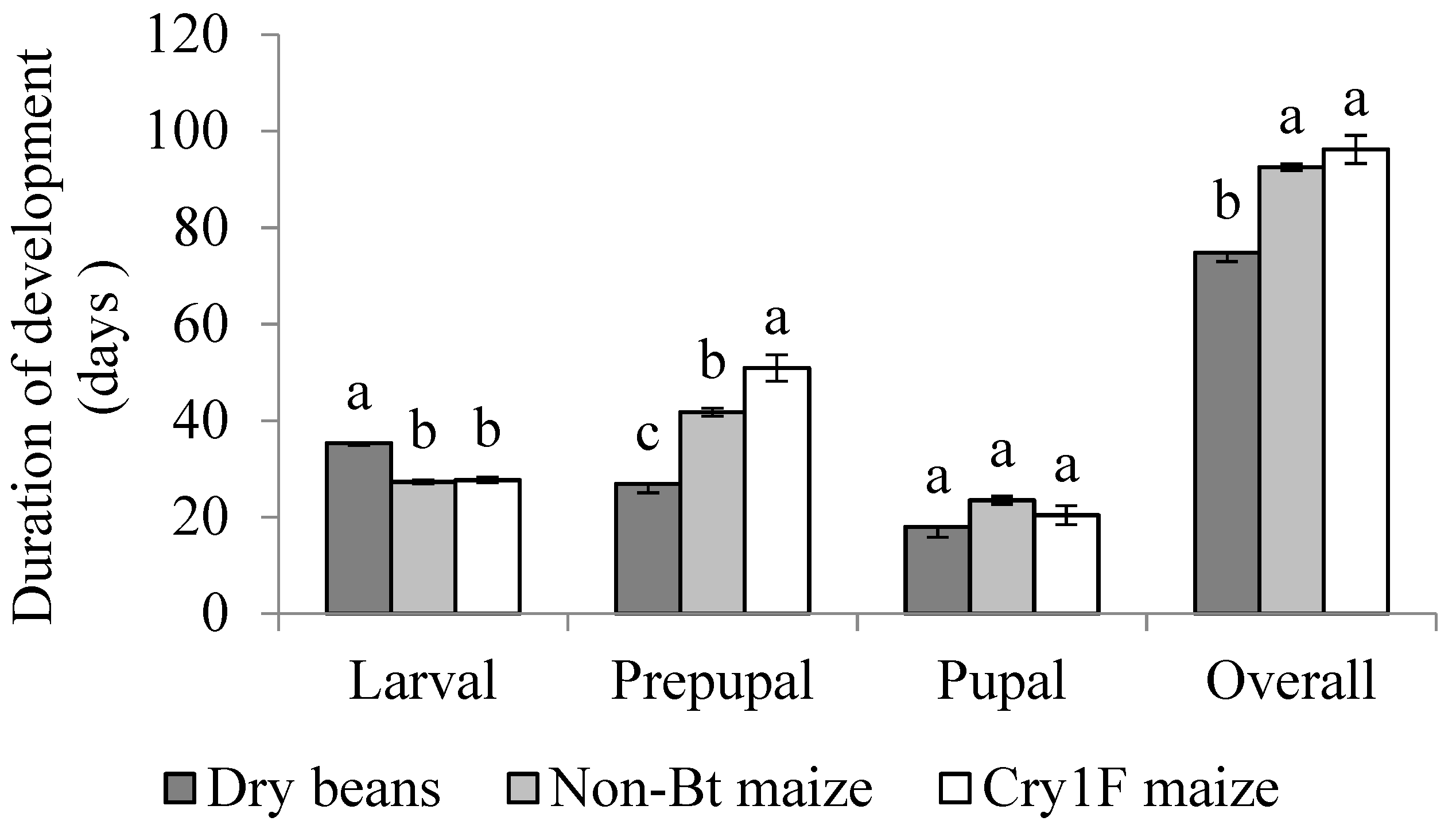
3.1. Overall Development
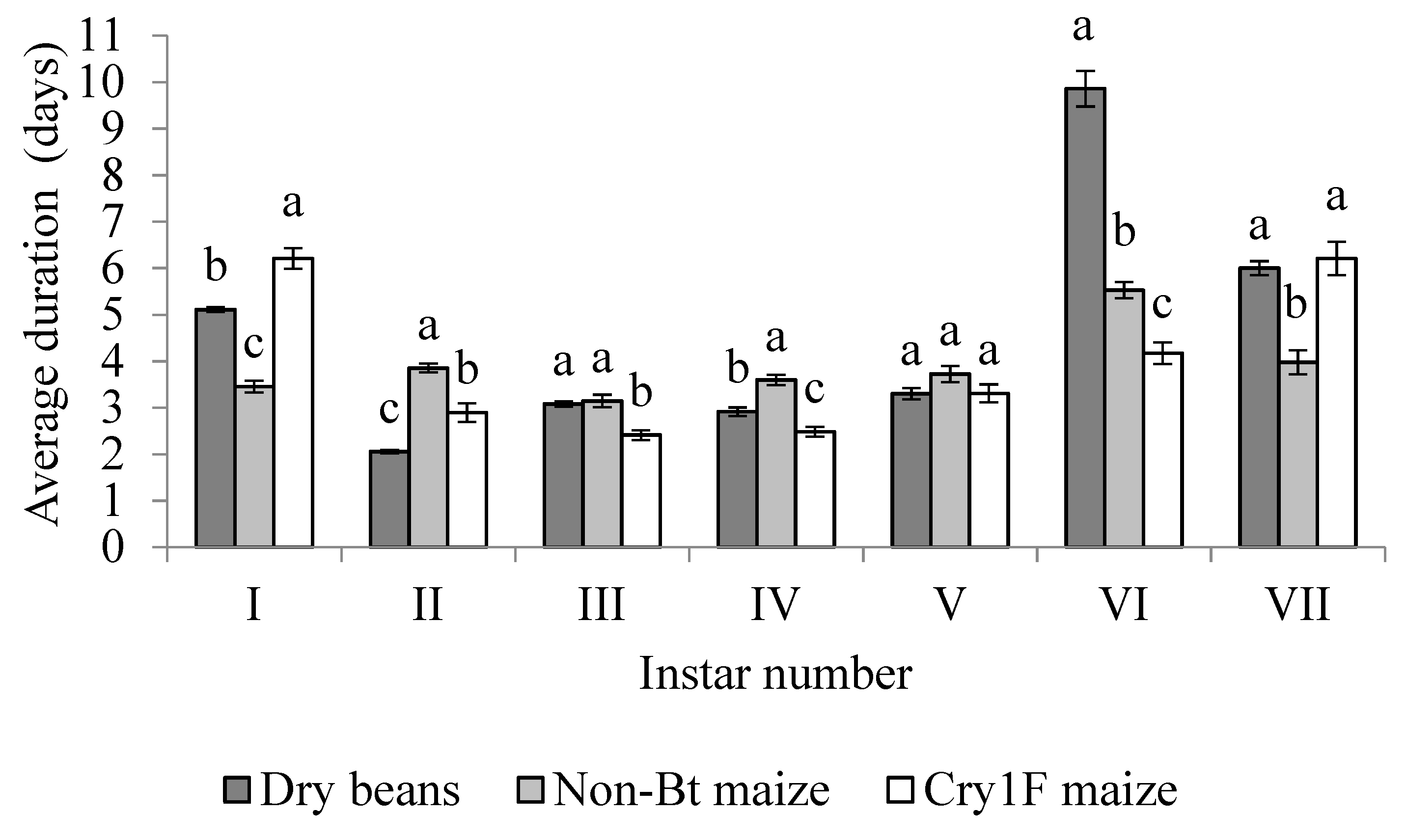
3.2. Active Larval Period
3.3. Inactive Larval Period (Prepupal Period)
3.4. Pupal Stage
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hoerner, J.L. The cutworm Loxagrotis albicosta on beans. J. Econ. Entomol. 1948, 41, 631–635. [Google Scholar] [CrossRef]
- McCampbell, S.C. Cutworm control. In Annual Report; Colorado State University Cooperative Extension: Colorado, CO, USA, 1941. [Google Scholar]
- Hagen, A.F. The biology and control of the western bean cutworm in dent corn in Nebraska. J. Econ. Entomol. 1962, 55, 628–631. [Google Scholar] [CrossRef]
- Douglass, J.R.; Ingram, J.W.; Gibson, K.E.; Peay, W.E. The western bean cutworm as a pest of corn in Idaho. J. Econ. Entomol. 1957, 50, 543–545. [Google Scholar] [CrossRef]
- Western Bean Cutworm and Its Control. Available online: http://agris.fao.org/agris-search/search.do?recordID=US201300613361 (accessed on 12 October 2019).
- Blickenstaff, C.C.; Jolley, P.M. Host plants of western bean cutworm. Environ. Entomol. 1982, 11, 421–425. [Google Scholar] [CrossRef]
- Hutchison, W.D.; Hunt, T.E.; Hein, G.L.; Steffey, K.L.; Pilcher, C.D.; Rice, M.E. Genetically engineered Bt corn and range expansion of the western bean cutworm (Lepidoptera: Noctuidae) in the United States: A response to Greenpeace Germany. J. Integr. Pest Manag. 2011, 3, B1–B8. [Google Scholar] [CrossRef]
- Smith, J.L.; Baute, T.S.; Sebright, M.M.; Schaafsma, A.W.; DiFonzo, C.D. Establishment of Striacosta albicosta (Lepidoptera: Noctuidae) as a primary pest of corn in the Great Lakes region. J. Econ. Entomol. 2018, 4, 1732–1744. [Google Scholar] [CrossRef]
- Baute, T. Current distribution of western bean cutworm in the Great Lakes region. CropPest Ont. Newsl. 2009, 14, 7–8. [Google Scholar]
- Tooker, J.F.; Fleischer, S.J. First report of western bean cutworm (Striacosta albicosta) in Pennsylvania. Crop Manag. 2010, 9, 1. [Google Scholar] [CrossRef]
- Ingerson-Mahar, J. Western bean cutworm found in New Jersey. Plant Pest Advis. 2012, 18, 1. [Google Scholar]
- Sánchez-Peña, S.R.; Torres-Acosta, R.I.; Camacho-Ponce, D. The second report of the western bean cutworm, Striacosta albicosta (Smith) (Lepidoptera: Noctuidae) as a dominant corn pest in Mexico. Proc. Entomol. Soc. Wash. 2016, 118, 389–392. [Google Scholar] [CrossRef]
- Chludzinski, M.M. Biology and Management of Western Bean Cutworm (Striacosta albicosta Smith) in Michigan Dry Beans (Phaseolus vulgaris L.). Master’s Thesis, Michigan State University, East Lansing, MI, USA, 2013. [Google Scholar]
- Paula-Moraes, S.V.; Hunt, T.E.; Wright, R.J.; Hein, G.I.; Blankenship, E.E. On-plant movement and feeding of western bean cutworm (Lepidoptera: Noctuidae) early instars on corn. Environ. Entomol. 2012, 41, 1494–1500. [Google Scholar] [CrossRef] [PubMed]
- Western Bean Cutworm in Corn and Dry Beans. Available online: http://extensionpublications.unl.edu/assets/pdf/g2013.pdf (accessed on 12 October 2019).
- Paula-Moraes, S.V.; Hunt, T.E.; Wright, R.J.; Hein, G.I.; Blankenship, E.E. Western bean cutworm survival and the development of economic injury levels and economic thresholds in field corn. J. Econ. Entomol. 2013, 106, 1274–1285. [Google Scholar] [CrossRef] [PubMed]
- Archibald, W.R.; Bradshaw, J.D.; Golick, D.A.; Wright, R.J.; Peterson, J.A. Nebraska growers’ and crop consultants’ knowledge and implementation of Integrated Pest Management of western bean cutworm. J. Integr. Pest Manag. 2017, 9, 1–7. [Google Scholar] [CrossRef]
- Eichenseer, H.; Strohbehn, R.; Burks, J. Frequency and severity of western bean cutworm (Lepidoptera: Noctuidae) ear damage in transgenic corn hybrids expressing different Bacillus thuringiensis Cry toxins. J. Econ. Entomol. 2008, 101, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Ostrem, J.S.; Pan, Z.; Flexner, J.L.; Owens, E.; Binning, R.; Higgins, L.S. Monitoring susceptibility of western bean cutworm (Lepidoptera: Noctuidae) field populations to Bacillus thuringiensis Cry1F protein. J. Econ. Entomol. 2016, 109, 847–853. [Google Scholar] [CrossRef]
- Smith, J.L.; Lepping, M.D.; Rule, D.M.; Farhan, Y.; Schaafsma, A.W. Evidence for field-evolved resistance of Striacosta albicosta (Lepidoptera: Noctuidae) to Cry1F Bacillus thuringiensis protein and transgenic corn hybrids in Ontario, Canada. J. Econ. Entomol. 2017, 110, 2217–2228. [Google Scholar] [CrossRef]
- Bowers, E.; Hellmich, R.; Munkvold, G. Comparison of fumonisin contamination using HPLC and ELISA methods in Bt and near-isogenic maize hybrids infested with European corn borer or western bean cutworm. J. Agric. Food Chem. 2014, 62, 6463–6472. [Google Scholar] [CrossRef]
- Storer, N.P. The Corn Earworm, Bt Transgenic Corn and Bt-Resistance Evolution in a Mixed Cropping System. Ph.D. Thesis, North Carolina State University, Raleigh, NC, USA, 1999. [Google Scholar]
- Horner, T.A.; Dively, G.P.; Herbert, D.A. Development, survival and fitness performance of Helicoverpa zea (Lepidoptera: Noctuidae) in MON810 Bt field corn. J. Econ. Entomol. 2003, 96, 914–924. [Google Scholar] [CrossRef]
- Bilbo, T.R.; Reay-Jones, F.P.; Reisig, D.D.; Musser, F.R.; Greene, J.K. Effects of Bt corn on the development and fecundity of corn earworm (Lepidoptera: Noctuidae). J. Econ. Entomol. 2018, 111, 2233–2241. [Google Scholar] [CrossRef]
- Liu, Y.B.; Tabashnik, B.E.; Dennehy, T.J.; Patin, A.L.; Bartlett, A.C. Development time and resistance to Bt crops. Nature 1999, 400, 519. [Google Scholar] [CrossRef]
- Gould, F. Sustainability of transgenic insecticidal cultivars: Integrating pest genetics and ecology. Annu. Rev. Entomol. 1998, 43, 701–726. [Google Scholar] [CrossRef] [PubMed]
- Bates, S.L.; Zhao, J.Z.; Roush, R.T.; Shelton, A.M. Insect resistance management in GM crops: Past, present and future. Nat. Biotechnol. 2005, 23, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Vélez, A.M.; Alves, A.P.; Blankenship, E.E.; Siegfried, B.D. Effect of Cry1F maize on the behavior of susceptible and resistant Spodoptera frugiperda and Ostrinia nubilalis. Entomol. Exp. Appl. 2016, 159, 37–45. [Google Scholar] [CrossRef]
- Montezano, D.G.; Hunt, T.E.; Specht, A.; Luz, P.C.; Peterson, A.J. Life-history parameters of Striacosta albicosta (Lepidoptera: Noctuidae) under laboratory conditions. J. Insect Sci. 2019, 19, 1–8. [Google Scholar] [CrossRef]
- Montezano, D.G.; Specht, A.; Bortolin, T.M.; Fronza, E.; Sosa-Gomez, D.R.; Roque-Specht, V.F.; Pezzi, P.P.; Luz, P.C.; Barros, N.M. Immature stages of Spodoptera albula (Walker) (Lepidoptera: Noctuidae): Developmental parameters & host plants. Anais Acad. Bras. Ciênc. 2013, 85, 271–284. [Google Scholar]
- Angulo, A.O.; Jana, S. La pupa de Spodoptera Guenée, 1852, en el norte de Chile (Lepidoptera: Noctudae). Agricultura Técnica 1982, 42, 347–349. [Google Scholar]
- Efron, B. Logistic regression, survival analysis, and the Kaplan-Meier curve. J. Am. Stat. Assoc. 1998, 83, 414–425. [Google Scholar] [CrossRef]
- War, A.R.; Paulraj, M.G.; Ahmad, T.; Buhroo, A.A.; Hussain, B.; Ignacimuthu, S.; Sharma, H.C. Mechanisms of plant defense against insect herbivores. Plant Signal. Behav. 2012, 7, 1306–1320. [Google Scholar] [CrossRef] [Green Version]
- Blickenstaff, C.C. History and Biology of the Western Bean Cutworm in Southern Idaho, 1942–1977; University of Idaho, College of Agriculture: Idaho, Moscow, 1979. [Google Scholar]
- Antonelli, A.L. Resistance of Phaseolus vulgaris Cultivars to Western Bean Cutworm, Loxagrotis albicosta (Smith), with Notes on the Bionomics and Culture of the Cutworm. Ph.D. Thesis, University of Idaho, Moscow, ID, USA, 1974. [Google Scholar]
- Doyle, M.S. Pheromone Trapping and Laboratory Rearing of Western Bean Cutworm (Lepidoptera: Noctuidae). Master’s Thesis, Colorado State University, Ft. Collins, CO, USA, 1994. [Google Scholar]
- Dyer, J.; Lewis, L.; Sappington, T.; Coates, B.; Abel, C.; Bidne, K.; Gunnarson, B.; Hellmich, R. Western Bean Cutworm Laboratory Rearing Manual; Iowa State University: Ames, IA, USA, 2013. [Google Scholar]
- DiFonzo, C.D. Managing Western Bean Cutworm in Dry Beans; Michigan State University: East Lansing, MI, USA, 2010. [Google Scholar]
- Esperk, T.; Tammaru, T.; Nylin, S. Intraspecific variability in number of larval instars in insects. J. Econ. Entomol. 2007, 100, 627–645. [Google Scholar] [CrossRef]
- Nijhout, H.F. A threshold size for metamorphosis in the tobacco hornworm, Manduca sexta. Biol. Bull. 1975, 149, 214–225. [Google Scholar] [CrossRef]
- Milks, M.L.; Burnstyn, I.; Myers, J.W. Influence of larval age on the lethal and sublethal effects of the nucleopolyhedrovirus of Trichoplusia ni in the cabbage looper. Biol. Control. 1998, 12, 119–126. [Google Scholar] [CrossRef]
- Reisig, D.D.; Reay-Jones, P.P. Inhibition of Helicoverpa zea (Lepidoptera: Noctuidae) growth by transgenic corn expressing Bt toxins and development of resistance to Cry1Ab. Environ. Entomol. 2015, 44, 1275–1285. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Buschman, L.L.; Higgins, R.A. Susceptibility of different instars of European corn borer (Lepidoptera: Crambidae) to diet containing Bacillus thuringiensis. Biol. Microb. Control. 1999, 92, 547–550. [Google Scholar] [CrossRef]
- Farhan, Y.; Smith, J.L.; Schaafsma, A.W. Baseline susceptibility of Striacosta albicosta (Lepidoptera: Noctuidae) in Ontario, Canada to Vip3A Bacillus thuringiensis protein. J. Econ. Entomol. 2017, 111, 65–71. [Google Scholar] [CrossRef]
- Pannuti, L.E.R.; Paula-Moraes, S.V.; Hunt, T.E.; Baldin, E.L.L.; Dana, L.; Malaquias, J.V. Plant-to-plant movement of Striacosta albicosta (Lepidoptera: Noctuidae) and Spodoptera frugiperda (Lepidoptera: Noctuidae) in maize (Zea mays). J. Econ. Entomol. 2016, 109, 1125–1131. [Google Scholar] [CrossRef]
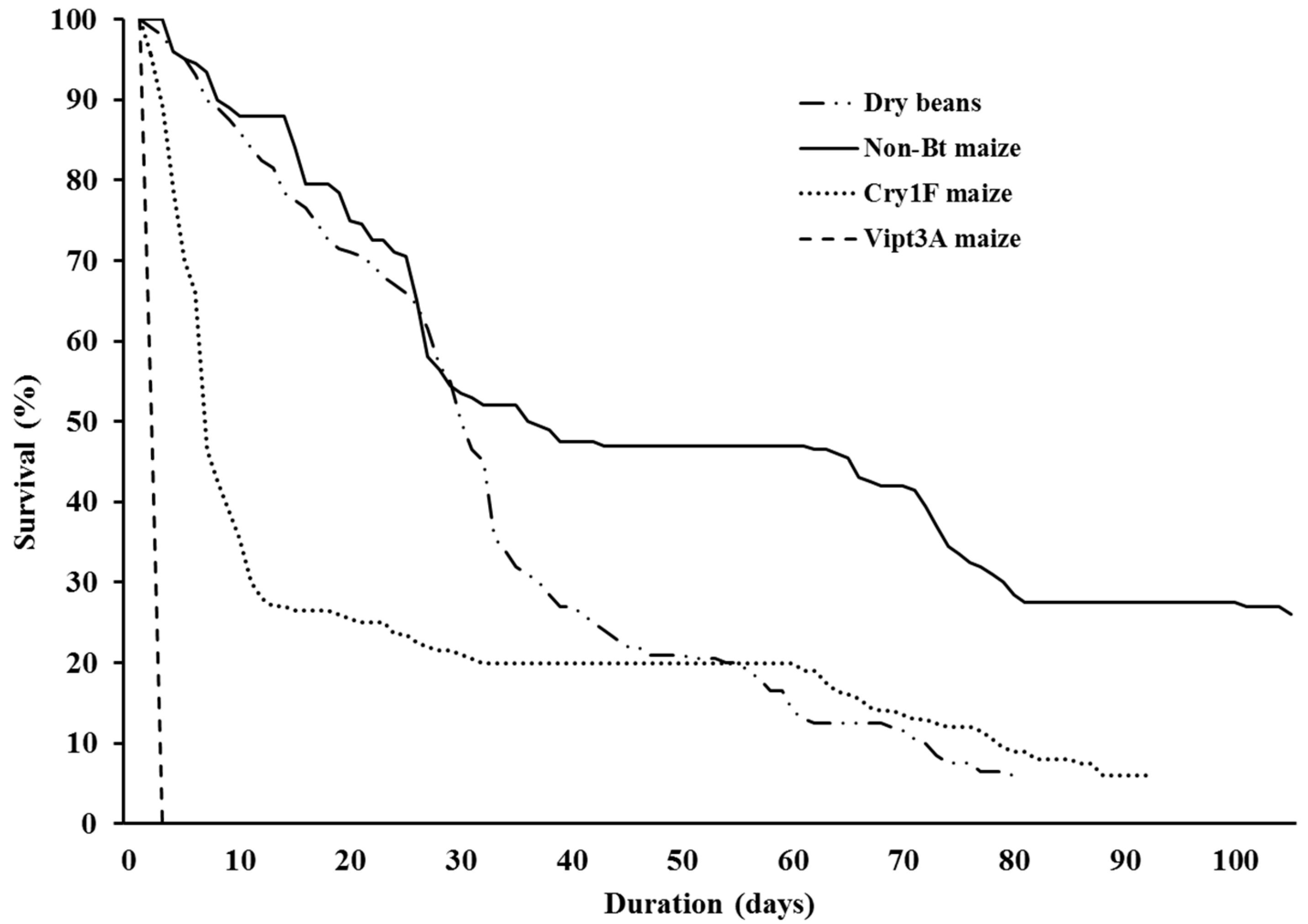
| Survival Curves Comparisons | df | χ2 | p |
|---|---|---|---|
| General comparison among all curves | 3 | 913.00 | <0.001 |
| Dry beans x Cry1F maize | 1 | 19.0 | <0.001 |
| Dry beans x Non-Bt maize | 1 | 32.2 | <0.001 |
| Dry bean x Vip3A maize | 1 | 422.0 | <0.001 |
| Cry1F maize x Non-Bt maize | 1 | 88.8 | <0.001 |
| Cry1F maize x Vip3A maize | 1 | 331.0 | <0.001 |
| Vip3A maize x Non-Bt maize | 1 | 345.0 | <0.001 |
| Dry Beans (n = 16) | Non-Bt (n = 34) | Cry1F (n = 30) | ||||
|---|---|---|---|---|---|---|
| Instar | Mean ± SE | Growth ratio | Mean ± SE | Growth ratio | Mean ± SE | Growth ratio |
| I | 0.39 ± 0.01 a | --- | 0.37 ± 0.01 a | --- | 0.39 ± 0.00 a | --- |
| II | 0.62 ± 0.01 a | 1.59 | 0.55 ± 0.01 b | 1.49 | 0.56 ± 0.01 b | 1.44 |
| III | 0.92 ± 0.02 a | 1.48 | 0.88 ± 0.01 b | 1.6 | 0.83 ± 0.02 b | 1.48 |
| IV | 1.46 ± 0.03 a | 1.59 | 1.32 ± 0.02 b | 1.5 | 1.32 ± 0.04 b | 1.60 |
| V | 2.12 ± 0.06 a | 1.45 | 1.96 ± 0.02 a | 1.48 | 2.00 ± 0.04 a | 1.51 |
| VI | 2.92 ± 0.04 a | 1.39 | 2.85 ± 0.01 a | 1.45 | 2.93 ± 0.04 a | 1.46 |
| VII | 3.33 ± 0.06 b | 1.13 | 3.64 ± 0.02 a | 1.28 | 3.66 ± 0.02 a | 1.25 |
| Mean | --- | 1.44 | --- | 1.47 | --- | 1.46 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
G. Montezano, D.; Hunt, T.E.; Specht, A.; C. Luz, P.M.; Peterson, J.A. Survival and Development of Striacosta albicosta (Smith) (Lepidoptera: Noctuidae) Immature Stages on Dry Beans, non-Bt, Cry1F, and Vip3A Maize. Insects 2019, 10, 343. https://doi.org/10.3390/insects10100343
G. Montezano D, Hunt TE, Specht A, C. Luz PM, Peterson JA. Survival and Development of Striacosta albicosta (Smith) (Lepidoptera: Noctuidae) Immature Stages on Dry Beans, non-Bt, Cry1F, and Vip3A Maize. Insects. 2019; 10(10):343. https://doi.org/10.3390/insects10100343
Chicago/Turabian StyleG. Montezano, Débora, Thomas E. Hunt, Alexandre Specht, Priscila M. C. Luz, and Julie A. Peterson. 2019. "Survival and Development of Striacosta albicosta (Smith) (Lepidoptera: Noctuidae) Immature Stages on Dry Beans, non-Bt, Cry1F, and Vip3A Maize" Insects 10, no. 10: 343. https://doi.org/10.3390/insects10100343





