Effects of Reduced Graphene Oxide (rGO) at Different Concentrations on Tribological Properties of Liquid Base Lubricants
Abstract
1. Introduction
2. Method
2.1. Materials
2.2. Sample Preparation
2.3. Sample Characterization
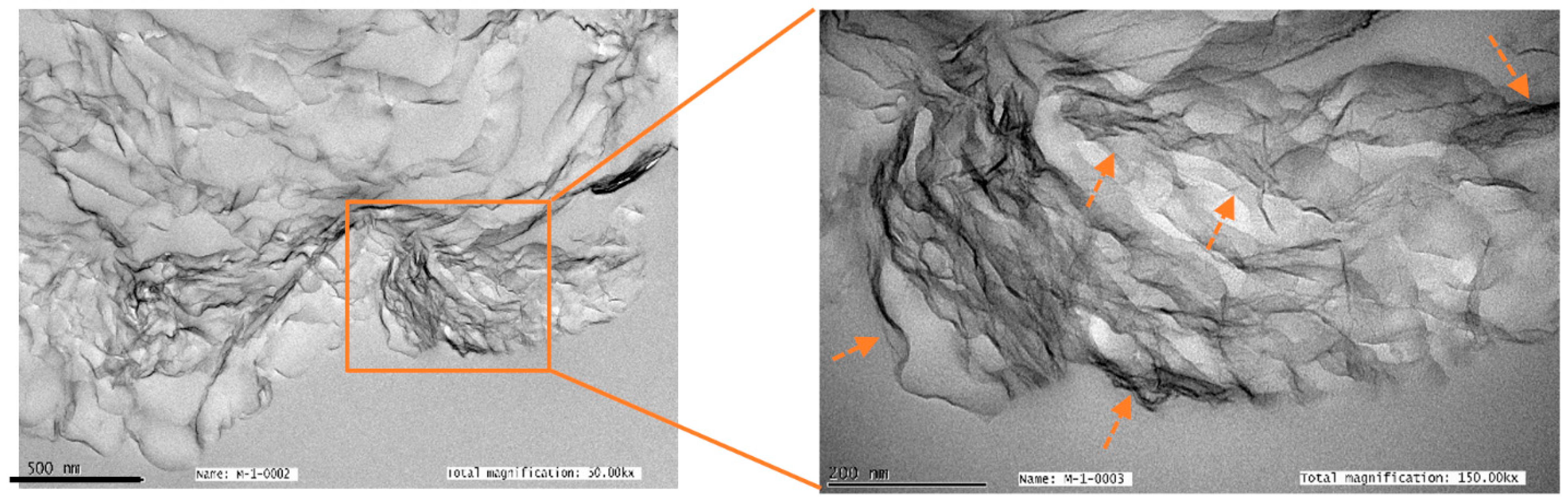
2.3.1. TEM and SEM Analysis
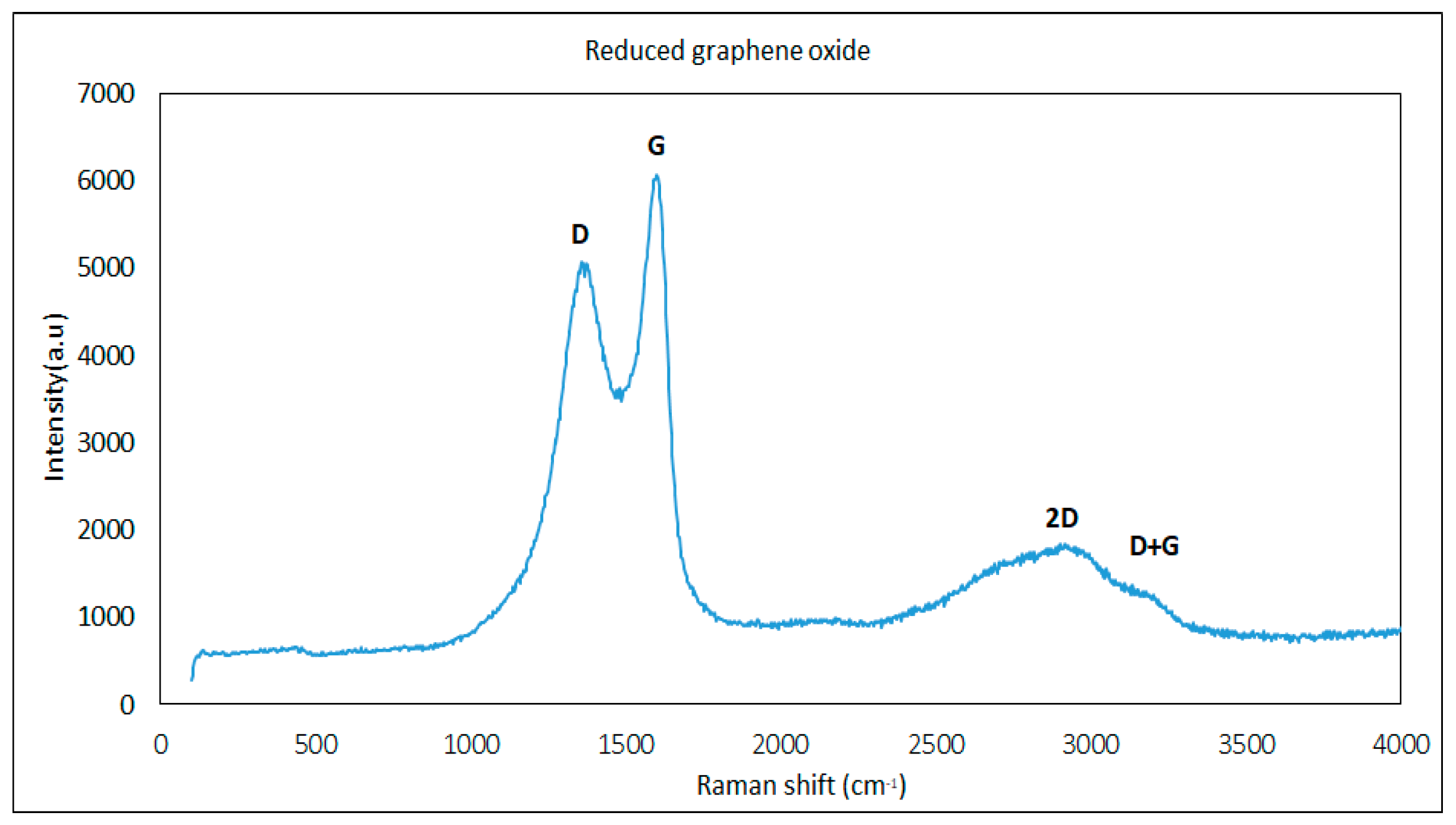
2.3.2. Raman Spectroscopy
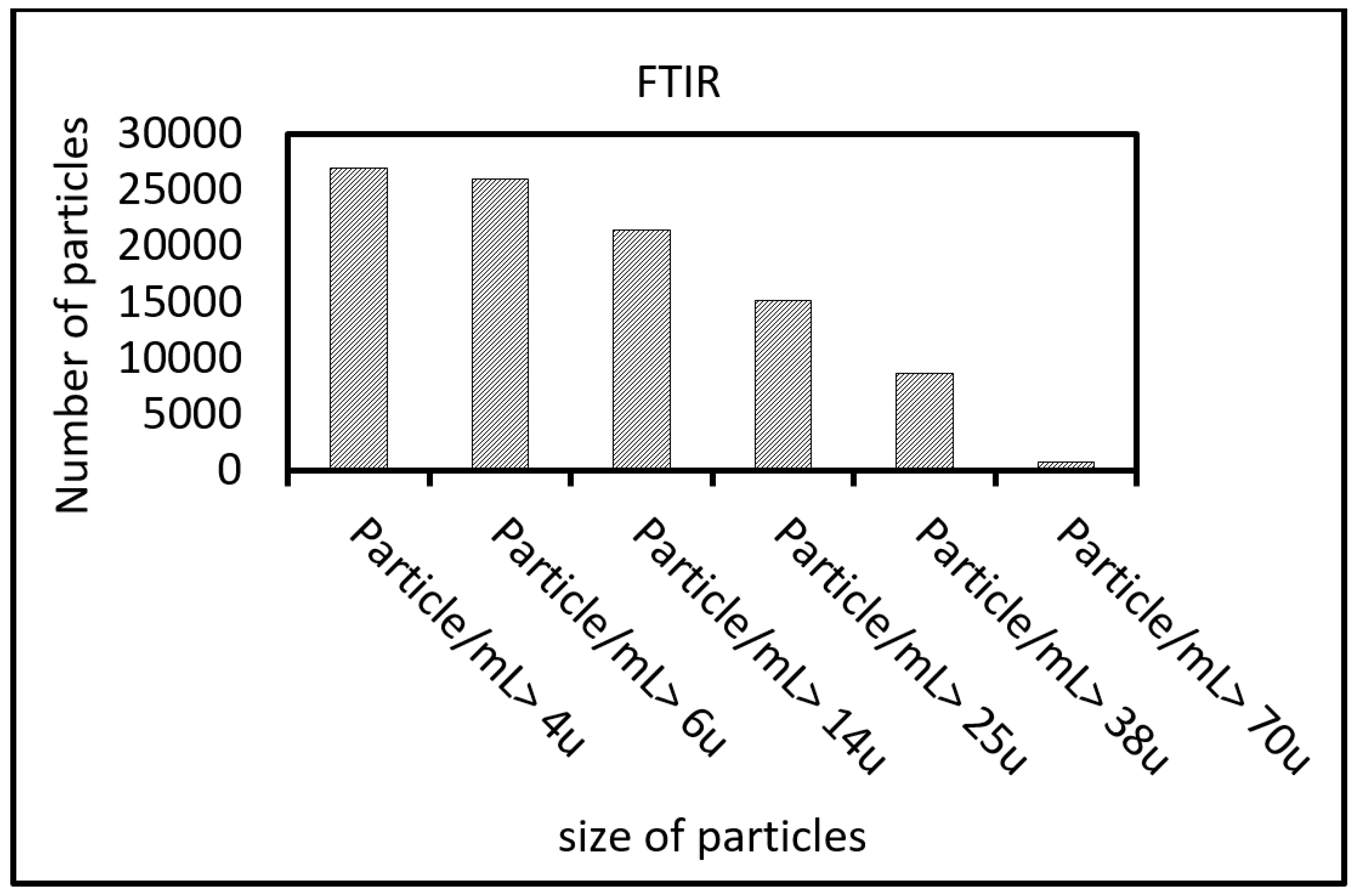
2.3.3. FTIR
2.3.4. Viscosity
2.3.5. RPVOT
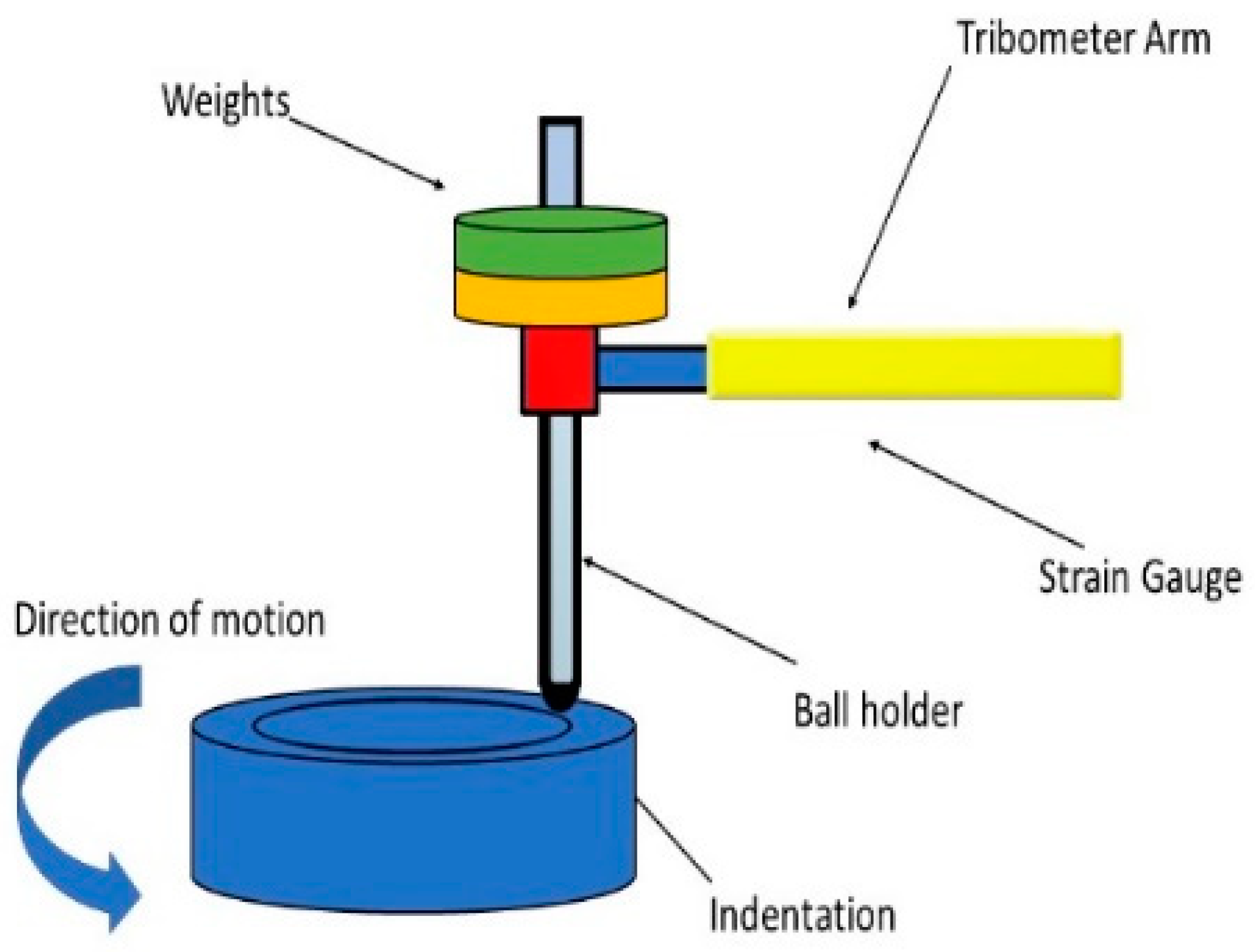
2.3.6. Ball-on-Disk Test
2.4. Statistical Analysis
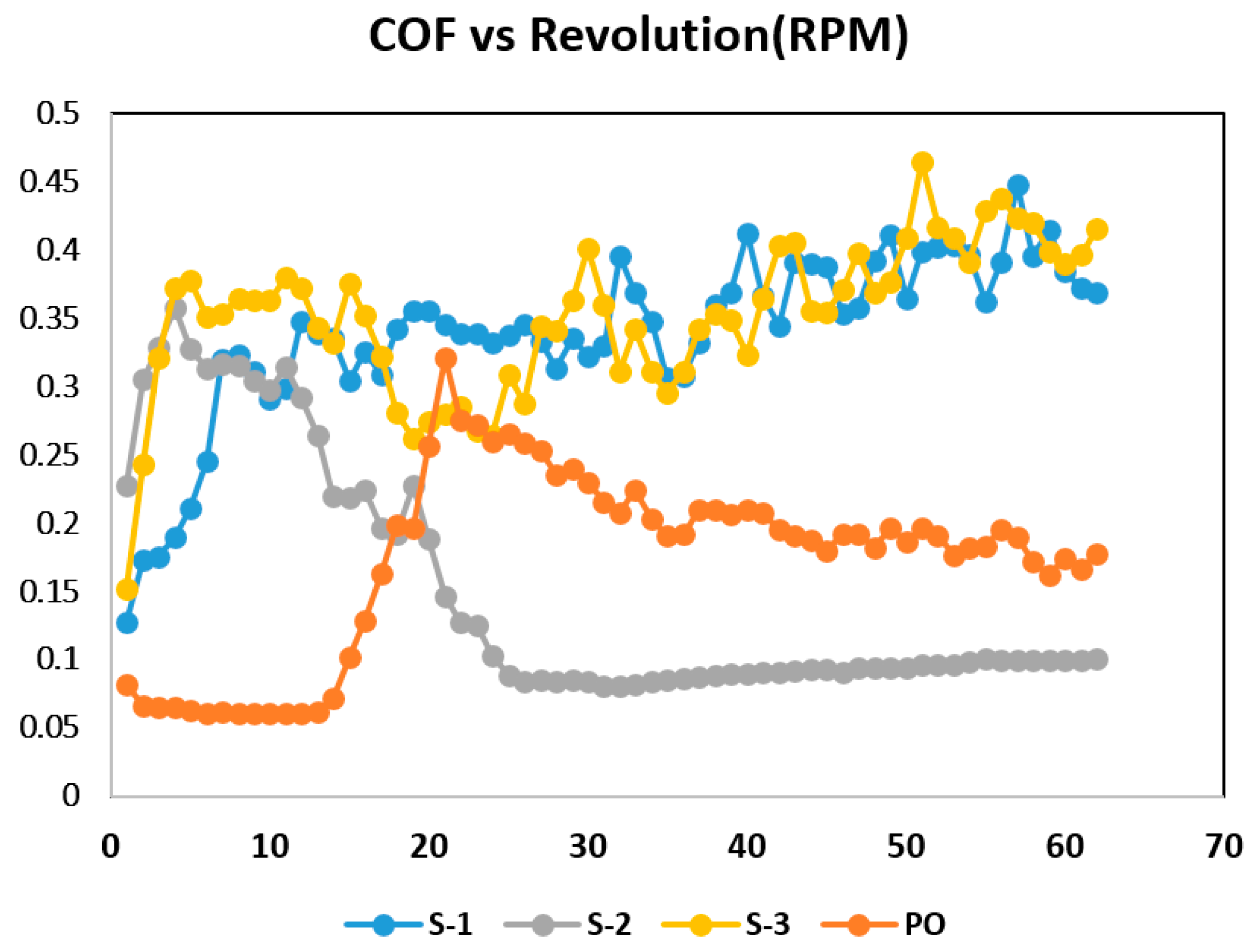
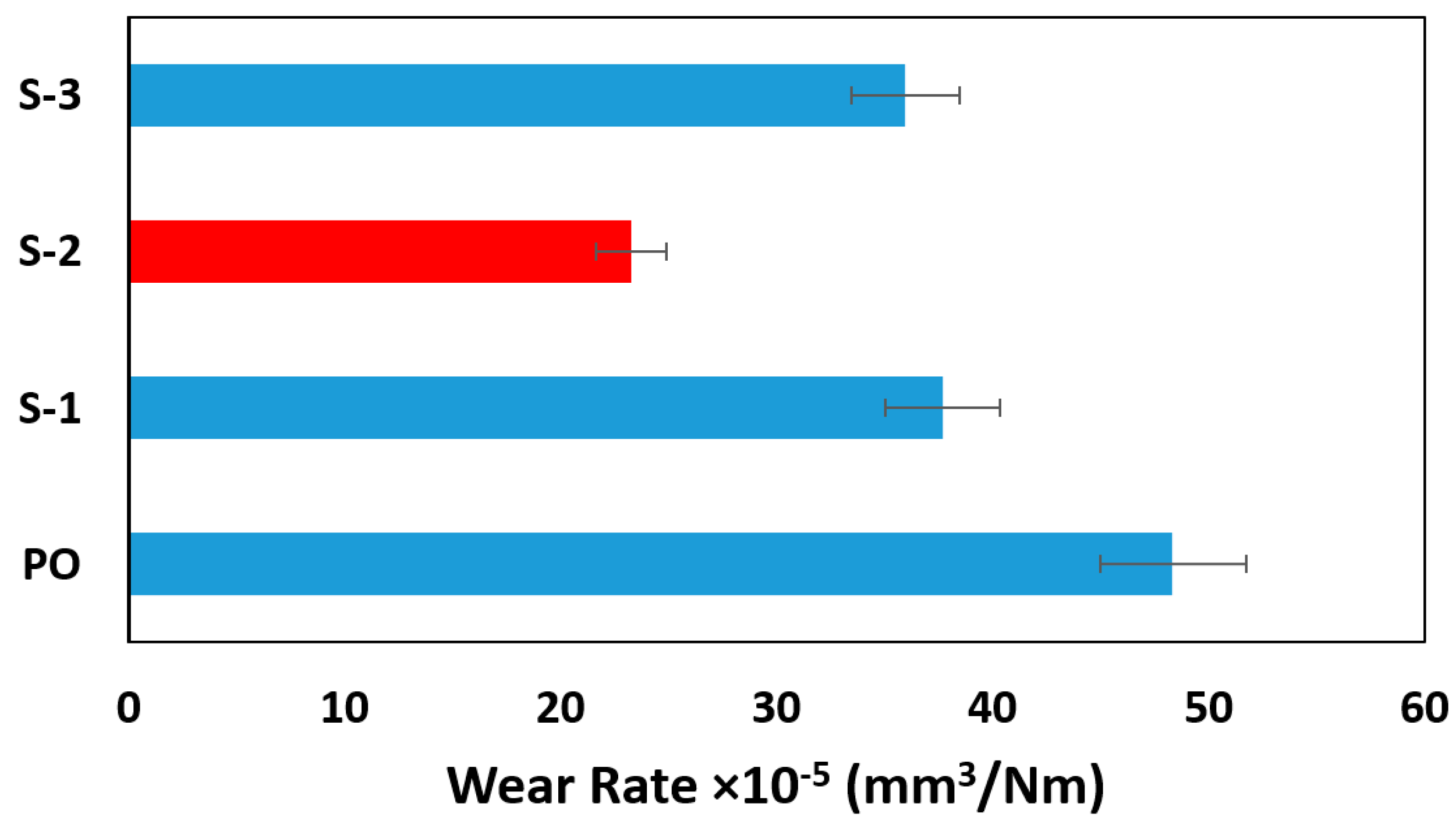
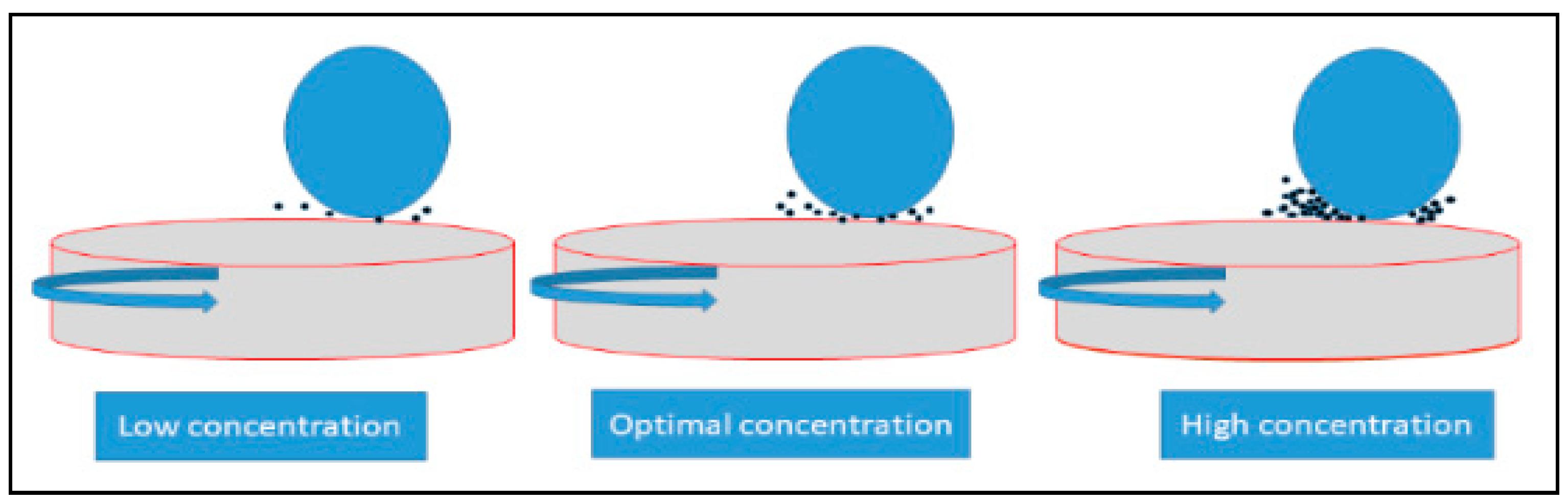
3. Results and Discussion
3.1. Reduced Graphene Oxide Characterization
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Holmberg, K.; Erdemir, A. Influence of tribology on global energy consumption, costs and emissions. Friction 2017, 5, 263–284. [Google Scholar] [CrossRef]
- Hutchings, I.; Shipway, P. Tribology: Friction and Wear of Engineering Materials; Butterworth-Heinemann: Oxford, UK, 2017. [Google Scholar]
- Padgurskas, J.; Rukuiza, R.; Prosyčevas, I.; Kreivaitis, R. Tribological properties of lubricant additives of fe, cu and co nanoparticles. Tribol. Int. 2013, 60, 224–232. [Google Scholar] [CrossRef]
- Cho, D.-H.; Kim, J.-S.; Kwon, S.-H.; Lee, C.; Lee, Y.-Z. Evaluation of hexagonal boron nitride nano-sheets as a lubricant additive in water. Wear 2013, 302, 981–986. [Google Scholar] [CrossRef]
- Lee, J.; Cho, S.; Hwang, Y.; Lee, C.; Kim, S.H. Enhancement of lubrication properties of nano-oil by controlling the amount of fullerene nanoparticle additives. Tribol. Lett. 2007, 28, 203–208. [Google Scholar] [CrossRef]
- Mauter, M.S.; Elimelech, M. Environmental applications of carbon-based nanomaterials. Environ. Sci. Technol. 2008, 42, 5843–5859. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The rise of grapheme. In Nanoscience and Technology: A Collection of Reviews from Nature Journals; World Scientific: Singapore, 2010; pp. 11–19. [Google Scholar]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of grapheme. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer grapheme. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer grapheme. Science 2008, 321, 385–388. [Google Scholar] [CrossRef]
- Orlita, M.; Faugeras, C.; Plochocka, P.; Neugebauer, P.; Martinez, G.; Maude, D.K.; Barra, A.-L.; Sprinkle, M.; Berger, C.; De Heer, W.A.; et al. Approaching the dirac point in high-mobility multilayer epitaxial grapheme. Phys. Rev. Lett. 2008, 101, 267601. [Google Scholar] [CrossRef]
- Service, R.F. Materials science. Carbon sheets an atomthick give rise to graphene dreams. Science 2009, 324, 875–877. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lee, S.-M.; Oh, Y.-S.; Yang, Y.-H.; Lim, Y.S.; Yoon, D.H.; Lee, C.; Kim, J.-Y.; Ruoff, R.S. Unoxidized graphene/alumina nanocomposite: Fracture-and wear-resistance effects of graphene on alumina matrix. Sci. Rep. 2014, 4, 5176. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.-S.; Ren, W.; Gao, L.; Liu, B.; Jiang, C.; Cheng, H.-M. Synthesis of high-quality graphene with a pre-determined number of layers. Carbon 2009, 47, 493–499. [Google Scholar] [CrossRef]
- Zhong, Y.L.; Tian, Z.; Simon, G.P.; Li, D. Scalable production of graphene via wet chemistry: Progress and challenges. Mater. Today 2015, 18, 73–78. [Google Scholar] [CrossRef]
- Lu, G.; Ocola, L.E.; Chen, J. Gas detection using low-temperature reduced graphene oxide sheets. Appl. Phys. Lett. 2009, 94, 083111. [Google Scholar] [CrossRef]
- Wang, S.J.; Geng, Y.; Zheng, Q.; Kim, J.-K. Fabrication of highly conducting and transparent graphene films. Carbon 2010, 48, 1815–1823. [Google Scholar] [CrossRef]
- Becerril, H.A.; Mao, J.; Liu, Z.; Stoltenberg, R.M.; Bao, Z.; Chen, Y. Evaluation of solution-processed reduced graphene oxide films as transparent conductors. ACS Nano 2008, 2, 463–470. [Google Scholar] [CrossRef]
- Yang, D.; Velamakanni, A.; Bozoklu, G.; Park, S.; Stoller, M.; Piner, R.D.; Stankovich, S.; Jung, I.; Field, D.A.; Ventrice, C.A., Jr.; et al. Chemical analysis of graphene oxide films after heat and chemical treatments by x-ray photoelectron and micro-raman spectroscopy. Carbon 2009, 47, 145–152. [Google Scholar] [CrossRef]
- Gómez-Navarro, C.; Weitz, R.T.; Bittner, A.M.; Scolari, M.; Mews, A.; Burghard, M.; Kern, K. Electronic transport properties of individual chemically reduced graphene oxide sheets. Nanoletters 2007, 7, 3499–3503. [Google Scholar] [CrossRef]
- Lee, S.W.; Mattevi, C.; Chhowalla, M.; Sankaran, R.M. Plasma-assisted reduction of graphene oxide at low temperature and atmospheric pressure for flexible conductor ap-plications. J. Phys. Chem. Lett. 2012, 3, 772–777. [Google Scholar] [CrossRef]
- Cheng, M.; Yang, R.; Zhang, L.; Shi, Z.; Yang, W.; Wang, D.; Xie, G.; Shi, D.; Zhang, G. Restoration of graphene from graphene oxide by defect repair. Carbon 2012, 50, 2581–2587. [Google Scholar] [CrossRef]
- Hazra, K.; Rafiee, J.; Rafiee, M.; Mathur, A.; Roy, S.; McLauhglin, J.; Koratkar, N.; Misra, D. Thinning of multilayer graphene to monolayer graphene in a plasma environment. Nanotechnology 2010, 22, 025704. [Google Scholar] [CrossRef] [PubMed]
- Eda, G.; Chhowalla, M. Chemically derived graphene oxide: Towards large-area thin-film electronics and optoelectronics. Adv. Mater. 2010, 22, 2392–2415. [Google Scholar] [CrossRef] [PubMed]
- Zaaba, N.; Foo, K.; Hashim, U.; Tan, S.; Liu, W.-W.; Voon, C. Synthesis of graphene oxide using modified hummers method: Solvent influence. Procedia Eng. 2017, 184, 469–477. [Google Scholar] [CrossRef]
- Hafiz, S.M.; Ritikos, R.; Whitcher, T.J.; Razib, N.M.; Bien, D.C.S.; Chanlek, N.; Nakajima, H.; Saisopa, T.; Songsiriritthigul, P.; Huang, N.M.; et al. A practical carbon dioxide gas sensor using room-temperature hydrogen plasma reduced graphene oxide. Sens. Actuators B Chem. 2014, 193, 692–700. [Google Scholar] [CrossRef]
- Shahriary, L.; Athawale, A.A. Graphene oxide synthesized by using modified hummers approach. Int. J. Renew. Energy Environ. Eng. 2014, 2, 58–63. [Google Scholar]
- Sitton, A.; Ameye, J.; Kauffman, R.E. Residue analysis on RPVOT test samples for single and multiple antioxidants chemistry for turbine lubricants. J. ASTM Int. 2006, 3, 1–15. [Google Scholar] [CrossRef]


| Sample Identification | Concentration |
|---|---|
| Pure Oil | No additive (99.9% pure) |
| Sample-1(S-1) | 0.01% w/w nano additive |
| Sample-2(S-2) | 0.05% w/w nano additive |
| Sample-3(S-3) | 0.1% w/w nano additive |
| Test Parameters | Unit Value |
|---|---|
| Load | 30 N |
| Test duration | 20 min |
| Speed | 0.01–150 RPM |
| Ramp type | Logarithmic |
| Radius | 10 mm |
| Revolution | 312 Revolution |
| Ball material | SS440C |
| Ball diameter | 6 mm |
| Samples | RPVOT in min. |
|---|---|
| P.O. | 27.1 |
| S-1 | 26.1 |
| S-2 | 25.8 |
| S-3 | 25.7 |
| Samples | Viscosity at 40 °C | Viscosity at 100 °C | Viscosity Index |
|---|---|---|---|
| PO | 41.5 mm2/s | 6.3 mm2/s | 98 |
| S-1 | 41.2 mm2/s | 6.4 mm2/s | 104 |
| S-2 | 41.2 mm2/s | 6.4 mm2/s | 104 |
| S-3 | 41.2 mm2/s | 6.38 mm2/s | 103 |
| Samples | Sa (µm) | Sz (µm) |
|---|---|---|
| PO | 1.386 ± 0.0693 | 15.19 ± 0.0035 |
| S-1 | 2.018 ± 0.1009 | 25.67 ± 0.0050 |
| S-2 | 0.6091 ± 0.0305 | 11.89 ± 0.0015 |
| S-3 | 1.995 ± 0.0998 | 37.55 ± 0.0050 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patel, J.; Kiani, A. Effects of Reduced Graphene Oxide (rGO) at Different Concentrations on Tribological Properties of Liquid Base Lubricants. Lubricants 2019, 7, 11. https://doi.org/10.3390/lubricants7020011
Patel J, Kiani A. Effects of Reduced Graphene Oxide (rGO) at Different Concentrations on Tribological Properties of Liquid Base Lubricants. Lubricants. 2019; 7(2):11. https://doi.org/10.3390/lubricants7020011
Chicago/Turabian StylePatel, Jankhan, and Amirkianoosh Kiani. 2019. "Effects of Reduced Graphene Oxide (rGO) at Different Concentrations on Tribological Properties of Liquid Base Lubricants" Lubricants 7, no. 2: 11. https://doi.org/10.3390/lubricants7020011
APA StylePatel, J., & Kiani, A. (2019). Effects of Reduced Graphene Oxide (rGO) at Different Concentrations on Tribological Properties of Liquid Base Lubricants. Lubricants, 7(2), 11. https://doi.org/10.3390/lubricants7020011







