Surface Film Adsorption and Lubricity of Soybean Oil In-Water Emulsion and Triblock Copolymer Aqueous Solution: A Comparative Study
Abstract
:1. Introduction
2. Experiments
3. Results and Discussion
3.1. Surface Film Adsorption
3.2. FTIR Molecular Analysis of the Film Residues on the Worn Surfaces and the Lubricants
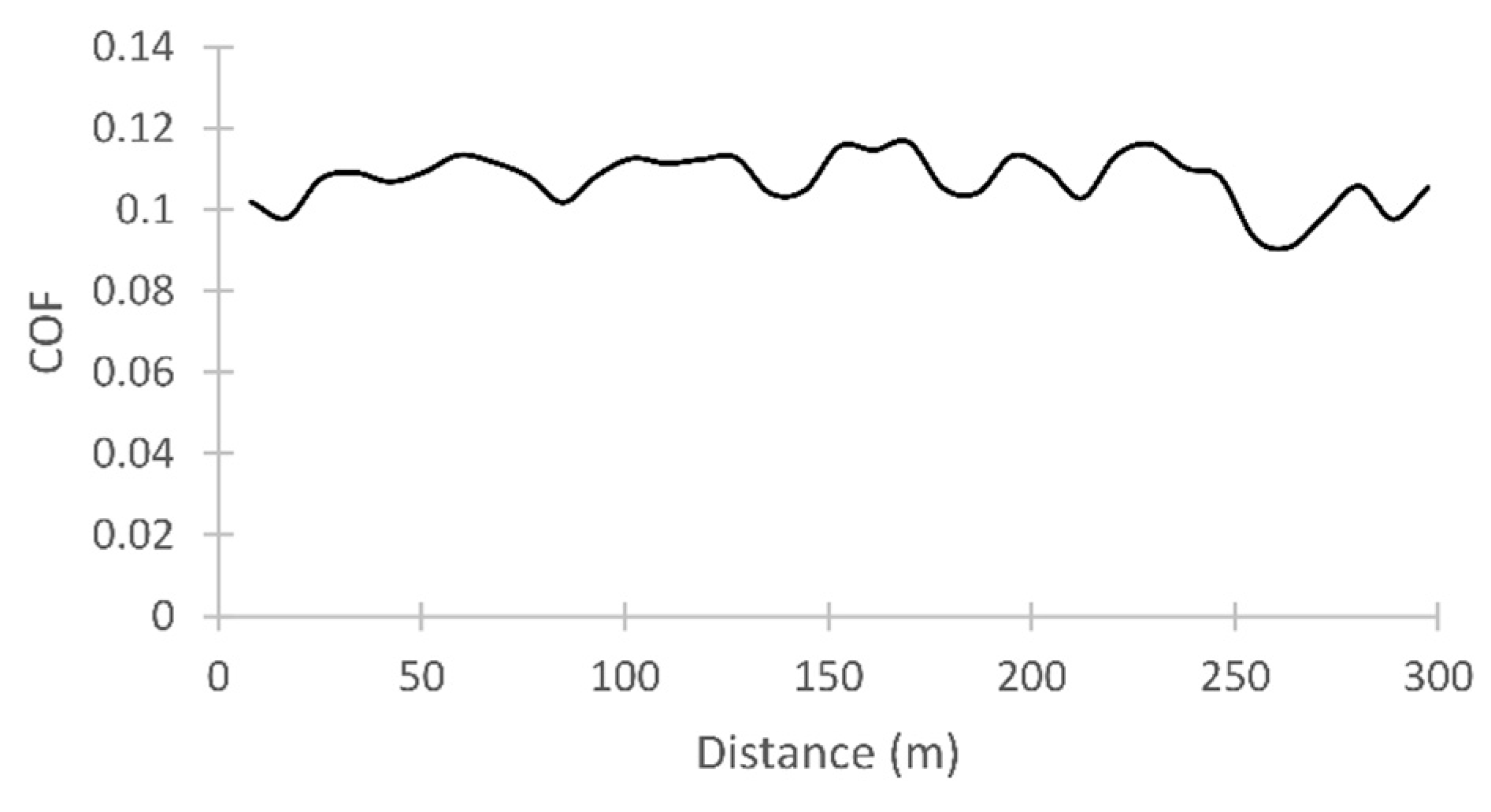
3.3. Friction
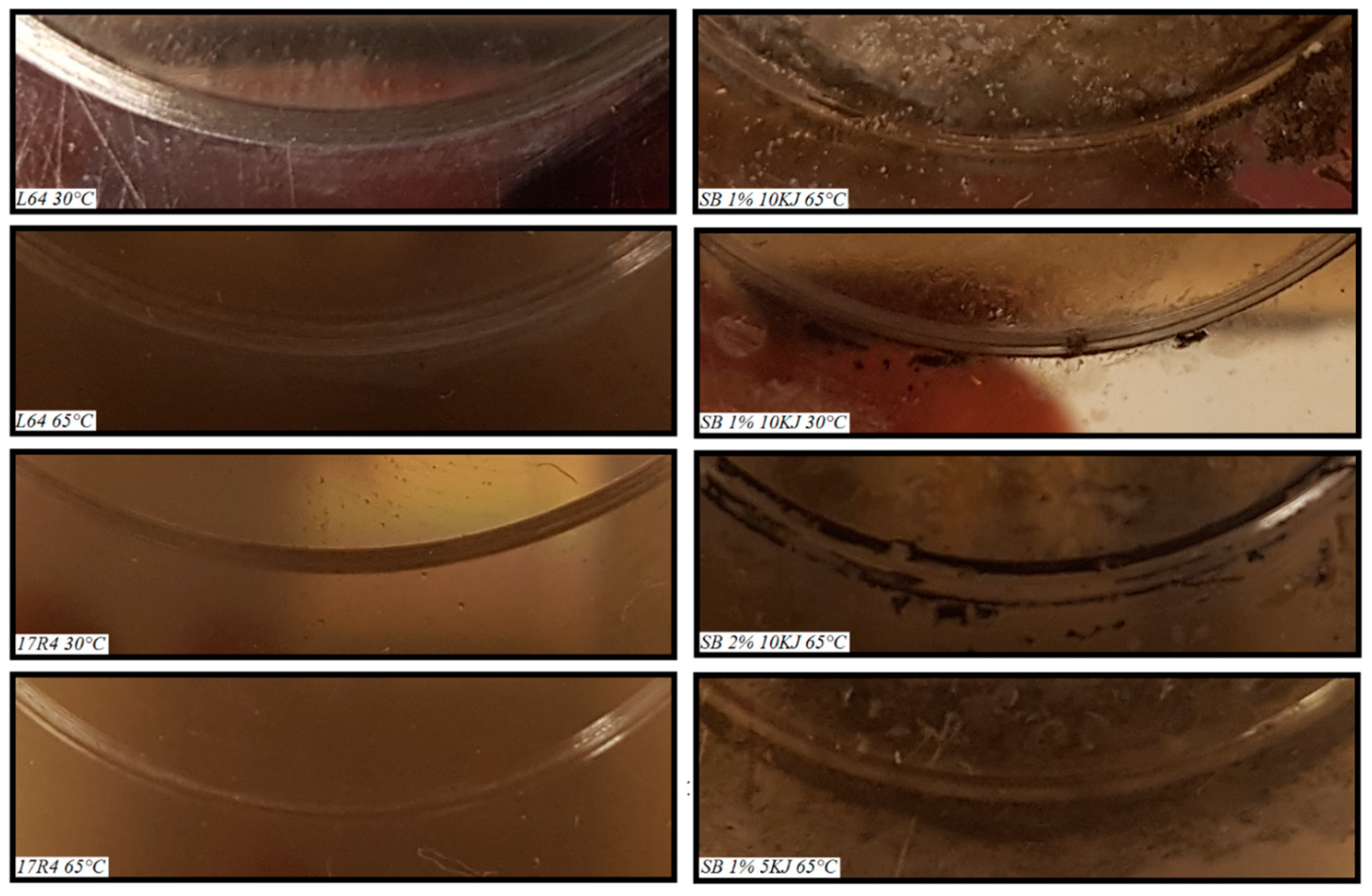
3.4. Worn Track and Ball Scar Analysis
3.5. Surface Cleanliness
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Menezes, P.L.; Reeves, C.J.; Lovell, M.R. Fundamentals of lubrication. In Tribology for Scientists and Engineers; Menezes, P.L., Ingole, S.P., Nosonovsky, M., Kailas, S.V., Lovell, M.R., Eds.; Springer: New York, USA, 2013; pp. 295–296. [Google Scholar]
- Cambiella, A.; Benito, J.M.; Pazos, C.; Coca, J.; Ratoi, M.; Spikes, H.A. The effect of emulsifier concentration on the lubricating properties of oil-in-water emulsions. Tribol. Lett. 2006, 22, 53–65. [Google Scholar] [CrossRef]
- Tieu, A.K.; Kosasih, P.B.; Godbole, A. A thermal analysis of strip-rolling in mixed-film lubrication with o/w emulsions. Tribol. Int. 2006, 39, 1591–1600. [Google Scholar] [CrossRef]
- Tieu, A.K.; Kosasih, P.B. Experimental and numerical study of o/w emulsion lubricated strip rolling in mixed film regime. Tribol. Lett. 2007, 25, 23–32. [Google Scholar] [CrossRef]
- Wang, A.; Chen, L.; Jiang, D.; Zeng, H.; Yan, Z. Vegetable oil-based ionic liquid microemulsion biolubricants: Effect of integrated surfactants. Ind. Crops Prod. 2014, 62, 515–521. [Google Scholar] [CrossRef]
- Jain, A.; Bisht, R.P.S. Metalworking emulsions from industrial vegetable oils. J. Synth. Lubr. 2008, 25, 87–94. [Google Scholar] [CrossRef]
- Kosasih, B.; Novareza, O.; Tieu, K.; Zhu, H. Friction and anti-wear property of aqueous tri-block copolymer solutions in metal forming. Int. J. Surf. Sci. Eng. 2014, 8, 109–123. [Google Scholar] [CrossRef]
- Kosasih, B.; Novareza, O.; Zhu, H.; Taheri, R.; Lin, B.; Tieu, A.K. Thickness and scratch resistance of adsorbed film formed by triblock symmetrical copolymer solutions. Lubr. Sci. 2016, 28, 299–315. [Google Scholar] [CrossRef]
- Lee, S.; Spencer, N.D. Poly(l-lysine)-graft-poly(ethylene glycol): A versatile aqueous lubricant additive for tribosystems involving thermoplastics. Lubr. Sci. 2008, 20, 21–34. [Google Scholar] [CrossRef]
- Alexandridis, P.; Hatton, T.A. Poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) block copolymer surfactants in aqueous solutions and at interfaces: Thermodynamics, structure, dynamics, and modeling. Colloids Surf. A Physicochem. Eng. Asp. 1995, 96, 1–46. [Google Scholar] [CrossRef]
- Pfister, D.P.; Xia, Y.; Larock, R.C. Recent advances in vegetable oil-based polyurethanes. ChemSusChem 2011, 4, 703–717. [Google Scholar] [CrossRef] [PubMed]
- Odi-Owei, S. Tribological properties of some vegetable oils and fats. Lubr. Eng. 1998, 45, 685–690. [Google Scholar]
- Joseph, P.V.; Sharma, D.K. Improvement of thermooxidative stability of non-edible vegetable oils of indian origin for biodegradable lubricant application. Lubr. Sci. 2010, 22, 149–161. [Google Scholar] [CrossRef]
- Siniawski, M.T.; Saniei, N.; Adhikari, B.; Doezema, L.A. Influence of fatty acid composition on the tribological performance of two vegetable-based lubricants. J. Synth. Lubr. 2007, 24, 101–110. [Google Scholar] [CrossRef]
- Joseph, P.V.; Saxena, D.; Sharma, D.K. Study of some non-edible vegetable oils of indian origin for lubricant application. J. Synth. Lubr. 2007, 24, 181–197. [Google Scholar] [CrossRef]
- Biresaw, G.; Adhvaryu, A.; Erhan, S.Z. Friction properties of vegetable oils. J. Am. Oil Chem. Soc. 2003, 80, 698–704. [Google Scholar] [CrossRef]
- Quinchia, L.A.; Delgado, M.A.; Reddyhoff, T.; Gallegos, C.; Spikes, H.A. Tribological studies of potential vegetable oil-based lubricants containing environmentally friendly viscosity modifiers. Tribol. Int. 2014, 69, 110–117. [Google Scholar] [CrossRef]
- Padmini, R.; Krishna, P.V.; Rao, G.K.M. Effectiveness of vegetable oil based nanofluids as potential cutting fluids in turning aisi 1040 steel. Tribol. Int. 2016, 94, 490–501. [Google Scholar] [CrossRef]
- Burton, G.; Goo, C.-S.; Zhang, Y.; Jun, M.B.G. Use of vegetable oil in water emulsion achieved through ultrasonic atomization as cutting fluids in micro-milling. J. Manuf. Process. 2014, 16, 405–413. [Google Scholar] [CrossRef]
- Li, Y.; Rojas, O.J.; Hinestroza, J.P. Boundary lubrication of peo-ppo-peo triblock copolymer physisorbed on polypropylene, polyethylene, and cellulose surfaces. Ind. Eng. Chem. Res. 2012, 51, 2931–2940. [Google Scholar] [CrossRef]
- Zhang, C.; Liu, J.; Zhang, C.; Liu, S. Friction reducing and anti-wear property of metallic friction pairs under lubrication of aqueous solutions with polyether added. Wear 2012, 292–293, 11–16. [Google Scholar] [CrossRef]
- Nakahara, T.; Makino, T.; Kyogoku, K. Observations of liquid droplet behavior and oil film formation in o/w type emulsion lubrication. J. Tribol. 1988, 110, 348–353. [Google Scholar] [CrossRef]
- Nakanishi, H.; Saiki, K.; Hirayama, T.; Matsuoka, T. Variation of oil introduction behaviour during oil-in-water emulsion rolling. Mater. Trans. 2013, 54, 1408–1415. [Google Scholar] [CrossRef]
- Barker, D.C.; Johnstona, G.J.; Spikesa, H.A.; Bunemannb, T.F. Ehd film formation and starvation of oil-in-water emulsions. Tribol. Trans. 1993, 36, 565–572. [Google Scholar] [CrossRef]
- Fox, N.J.; Stachowiak, G.W. Boundary lubrication properties of oxidized sunflower oil. J. Soc. Tribol. Lubr. Eng. 2003, 59, 15–20. [Google Scholar]
- Boyde, S. Green lubricants. Environmental benefits and impacts of lubrication. Green Chem. 2002, 4, 293–307. [Google Scholar] [CrossRef]
- Adhvaryu, A.; Erhan, S.Z.; Perez, J.M. Tribological studies of thermally and chemically modified vegetable oils for use as environmentally friendly lubricants. Wear 2004, 257, 359–367. [Google Scholar] [CrossRef]
- Biresaw, G. Adsorption of amphiphiles at an oil–water vs. An oil–metal interface. J. Am. Oil Chem. Soc. 2005, 82, 285–292. [Google Scholar] [CrossRef]
- Das, L.M.; Bora, D.K.; Pradhan, S.; Naik, M.K.; Naik, S.N. Long-term storage stability of biodiesel produced from karanja oil. Fuel 2009, 88, 2315–2318. [Google Scholar] [CrossRef]
- D’Errico, G.; Paduano, L.; Khan, A. Temperature and concentration effects on supramolecular aggregation and phase behavior for poly(propylene oxide)-b-poly(ethylene oxide)-b-poly(propylene oxide) copolymers of different composition in aqueous mixtures, 1. J. Colloid Interface Sci. 2004, 279, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Alexandridis, P. Poly(ethylene oxide)/poly(propylene oxide) block copolymer surfactants. Curr. Opin. Colloid Interface Sci. 1997, 2, 478–489. [Google Scholar] [CrossRef]
- Kiss, É.; Erdélyi, K.; Szendrö, I.; Vargha-Butler, E.I. Adsorption and wetting properties of pluronic block copolymers on hydrophobic surfaces studied by optical waveguide lightmode spectroscopy and dynamic tensiometric method. J. Adhes. 2004, 80, 815–829. [Google Scholar] [CrossRef]
- Novareza, O. Tribology of aqueous Copolymer Lubricants for Metal Forming Applications. Ph.D. Thesis, University of Wollongong, Wollongong, Australia, 2014. [Google Scholar]
- Taheri, R.; Kosasih, B.; Zhu, H.; Tieu, A.K. Phase behaviour and lubricity of aqueous PEO-PPO-PEO and ppo-peo-ppo triblock copolymer solutions. Tribol. Trans. 2016, 1–9. [Google Scholar] [CrossRef]
- Shar, J.A.; Obey, T.M.; Cosgrove, T. Adsorption studies of polyethers part ii: Adsorption onto hydrophilic surfaces. Colloids Surf. A Physicochem. Eng. Asp. 1999, 150, 15–23. [Google Scholar] [CrossRef]
- Shar, J.A.; Obey, T.M.; Cosgrove, T. Adsorption studies of polyethers part i. Adsorption onto hydrophobic surfaces. Colloids Surf. A Physicochem. Eng. Asp. 1998, 136, 21–33. [Google Scholar] [CrossRef]
- Lee, S.; Iten, R.; Muller, M.; Spencer, N.D. Influence of molecular architecture on the adsorption of poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) on pdms surfaces and implications for aqueous lubrication. Macromolecules 2004, 37, 8349–8356. [Google Scholar] [CrossRef]
- Brandani, P.; Stroeve, P. Adsorption and desorption of PEO-PPO-PEO triblock copolymers on a self-assembled hydrophobic surface. Macromolecules 2003, 36, 9492–9501. [Google Scholar] [CrossRef]
- Dibble, K. Chemical Cleaning of Metals. Available online: http://nzic.org.nz/ChemProcesses/metals/8H.pdf (accessed on 15 December 2016).
- Liu, K. Soybeans: Chemistry, Technology and Utilization; Chapman & Hall: New York, NY, USA, 1997. [Google Scholar]
- Martin, S.J.; Granstaff, V.E.; Frye, G.C. Characterization of a quartz crystal microbalance with simultaneous mass and liquid loading. Anal. Chem. 1991, 63, 2272–2281. [Google Scholar] [CrossRef]
- Kanazawa, K.K.; Gordon, J.G.I. The oscillation frequency of a quartz resonator in contact with a liquid. Anal. Chim. Acta 1985, 175, 99–105. [Google Scholar] [CrossRef]
- Lynn, J.L.J. Detergents and detergency. In Bailey’s Industrial Oil and Fat Products, 6th ed.; Shahidi, F., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2005; pp. 164–166. [Google Scholar]
- Stalgren, J.J.R.; Claesson, P.M.; Warnheim, T. Adsorption of liposomes and emulsions studied with a quartz crystal microbalance. Adv. Colloid Interface Sci. 2001, 89–90, 383–394. [Google Scholar] [CrossRef]
- Abismail, B.; Canselier, J.P.; Wilhelm, A.M.; Delmas, H.; Gourdon, C. Emulsification by ultrasound: Drop size distribution and stability. Ultrason. Sonochem. 1999, 6, 75–83. [Google Scholar] [CrossRef]
- Atkins, P.; de Paula, J. Physical Chemistry, 8th ed.; W. H. Freeman and Company: New York, NY, USA, 2006. [Google Scholar]
- Connell, S.D.A.; Allen, S.; Roberts, C.J.; Davies, J.; Davies, M.C.; Tendler, S.J.B.; Williams, P.M. Investigating the interfacial properties of single-liquid nanodroplets by atomic force microscopy. Langmuir 2002, 19, 1719–1728. [Google Scholar] [CrossRef]
- Kenbeck, D.; Bunemann, T.F. Organic friction modifiers. In Lubricant Additives: Chemistry and Applications, 2nd ed.; Rudnick, L.R., Ed.; CRC Press: New York, NY, 2009; pp. 201–202. [Google Scholar]
- Adhvaryu, A.; Erhan, S.Z. Epoxidized soybean oil as a potential source of high-temperature lubricants. Ind. Crops Prod. 2002, 15, 247–254. [Google Scholar] [CrossRef]
- Beltzer, M.; Jahanmir, S. Effect of additive molecular structure on friction. Lubr. Sci. 1988, 1, 3–26. [Google Scholar] [CrossRef]
- Jahanmir, S.; Beltzer, M. Effect of additive molecular structure on friction coefficient and adsorption. J. Tribol. 1986, 108, 109–116. [Google Scholar] [CrossRef]
- Rossrucker, T.; Fessenbecker, A. Sulfur carriers. In Lubricant Additives: Chemistry and Applications, 2nd ed.; Rudnick, L.R., Ed.; CRC Press: New York, NY, USA, 2009; pp. 267–269. [Google Scholar]
- Phillips, W.D. Ashless phosphorus-containing lubricating oil additives. In Lubricant Additives: Chemistry and Applications, 2nd ed.; Heinemann, H., Ed.; CRC Press: New York, NY, USA, 2009; pp. 78–79. [Google Scholar]
- Liu, X.; Wu, D.; Turgman-Cohen, S.; Genzer, J.; Theyson, T.W.; Rojas, O.J. Adsorption of a nonionic symmetric triblock copolymer on surfaces with different hydrophobicity. Langmuir 2010, 26, 9565–9574. [Google Scholar] [CrossRef] [PubMed]
- Viitala, T.; Liang, H.; Gupta, M.; Zwinger, T.; Yliperttula, M.; Bunker, A. Fluid dynamics modeling for synchronizing surface plasmon resonance and quartz crystal microbalance as tools for biomolecular and targeted drug delivery studies. J. Colloid Interface Sci. 2012, 378, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Caragheorgheopol, A.; Schlick, S. Hydration in the various phases of the triblock copolymers eo13 po30 eo13 (pluronic l64) and eo6po34eo6 (pluronic l62), based on electron spin resonance spectra of cationic spin probes. Macromolecules 1998, 31, 7736–7745. [Google Scholar] [CrossRef]
- Cohn, D.; Lando, G.; Sosnik, A.; Garty, S.; Levi, A. PEO-PPO-PEO-based poly(ether ester urethane)s as degradable reverse thermo-responsive multiblock copolymers. Biomaterials 2006, 27, 1718–1727. [Google Scholar] [CrossRef] [PubMed]
- Green, R.J.; Tasker, S.; Davies, J.; Davies, M.C.; Roberts, C.J.; Tendler, S.J.B. Adsorption of PEO-PPO-PEO triblock copolymers at the solid/liquid interface: A surface plasmon resonance study. Langmuir 1997, 13, 6510–6515. [Google Scholar] [CrossRef]
- Patel, T.; Bahadur, P.; Mata, J. The clouding behaviour of PEO-PPO based triblock copolymers in aqueous ionic surfactant solutions: A new approach for cloud point measurements. J. Colloid Interface Sci. 2010, 345, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, H.; Song, J.; Rojas, O.J.; Hinestroza, J.P. Adsorption and association of a symmetric PEO-PPO-PEO triblock copolymer on polypropylene, polyethylene, and cellulose surfaces. ACS Appl. Mater. Interfaces 2011, 3, 2349–2357. [Google Scholar] [CrossRef] [PubMed]
- Murakami, T.; Sakamoto, H. Lubricating properties of vegetable oils and paraffinic oils with unsaturated fatty acids under high-contact-pressure conditions in four-ball tests. J. Synth. Lubr. 2003, 20, 183–201. [Google Scholar] [CrossRef]
- Knothe, G. Evaluation of ball and disc wear scar data in the hfrr lubricity test. Lubr. Sci. 2008, 20, 35–45. [Google Scholar] [CrossRef]
- Pantoja, S.S.; da Conceição, L.R.V.; da Costa, C.E.F.; Zamian, J.R.; da Rocha Filho, G.N. Oxidative stability of biodiesels produced from vegetable oils having different degrees of unsaturation. Energy Convers. Manag. 2013, 74, 293–298. [Google Scholar] [CrossRef]
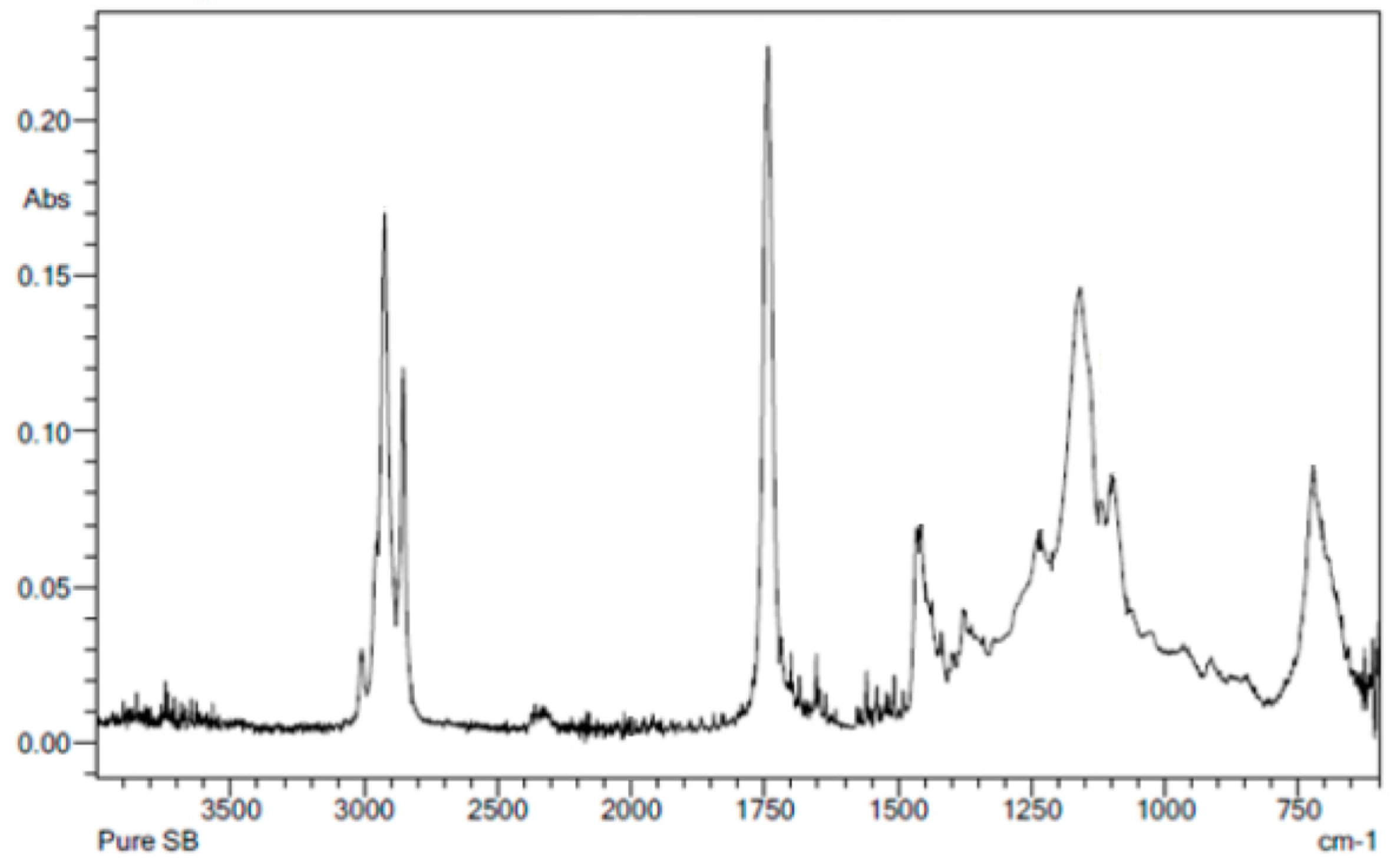

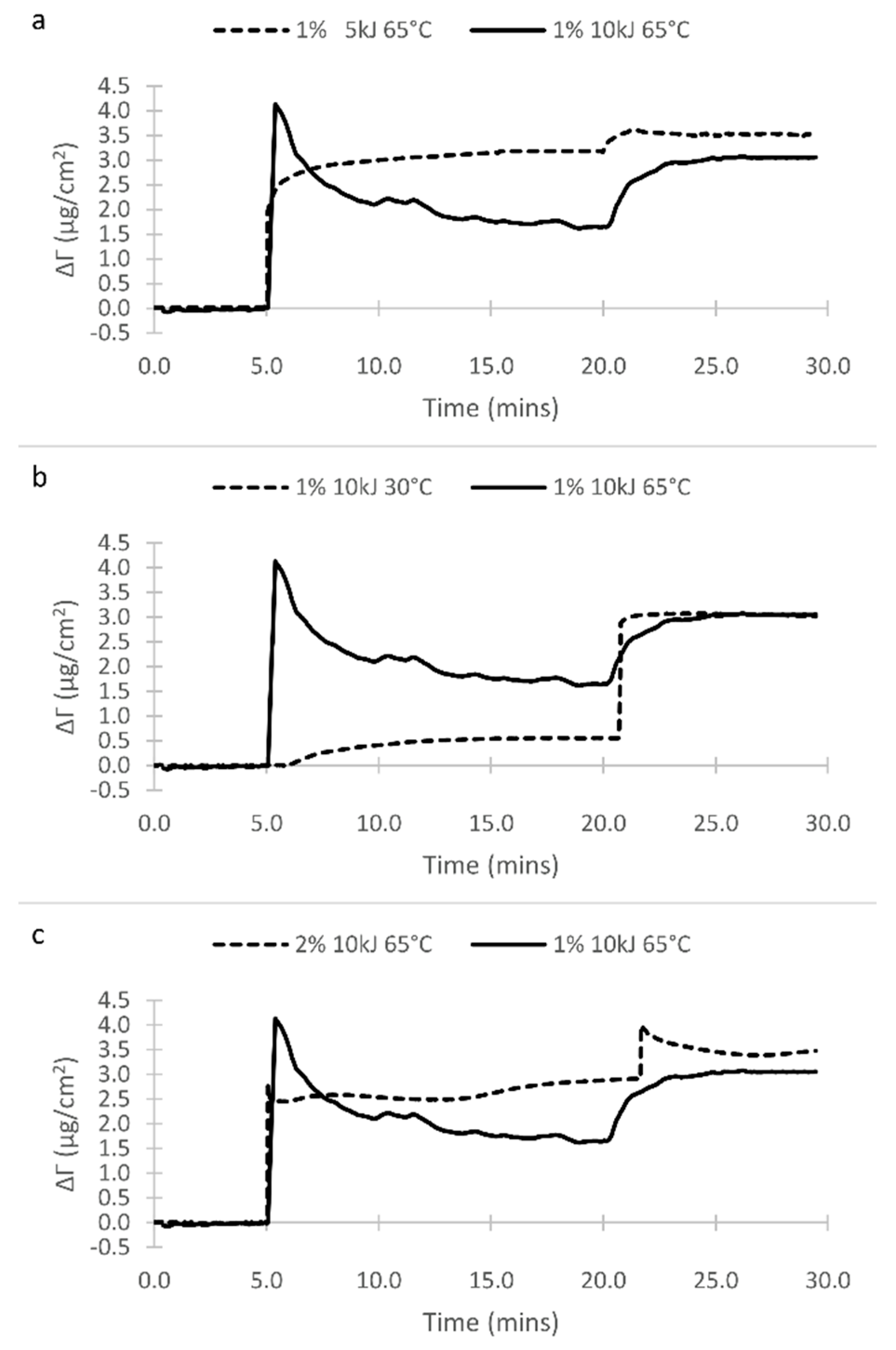
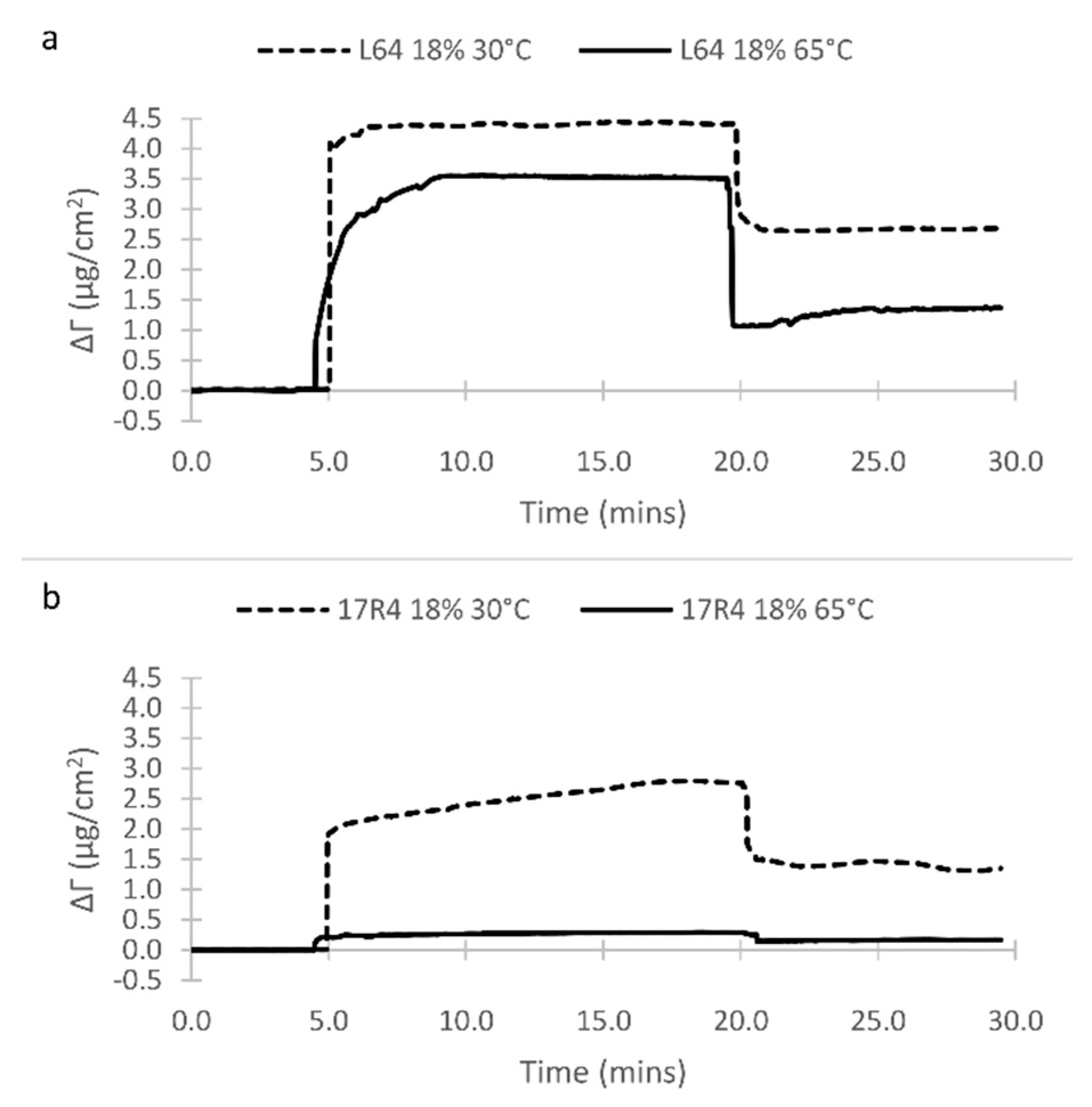

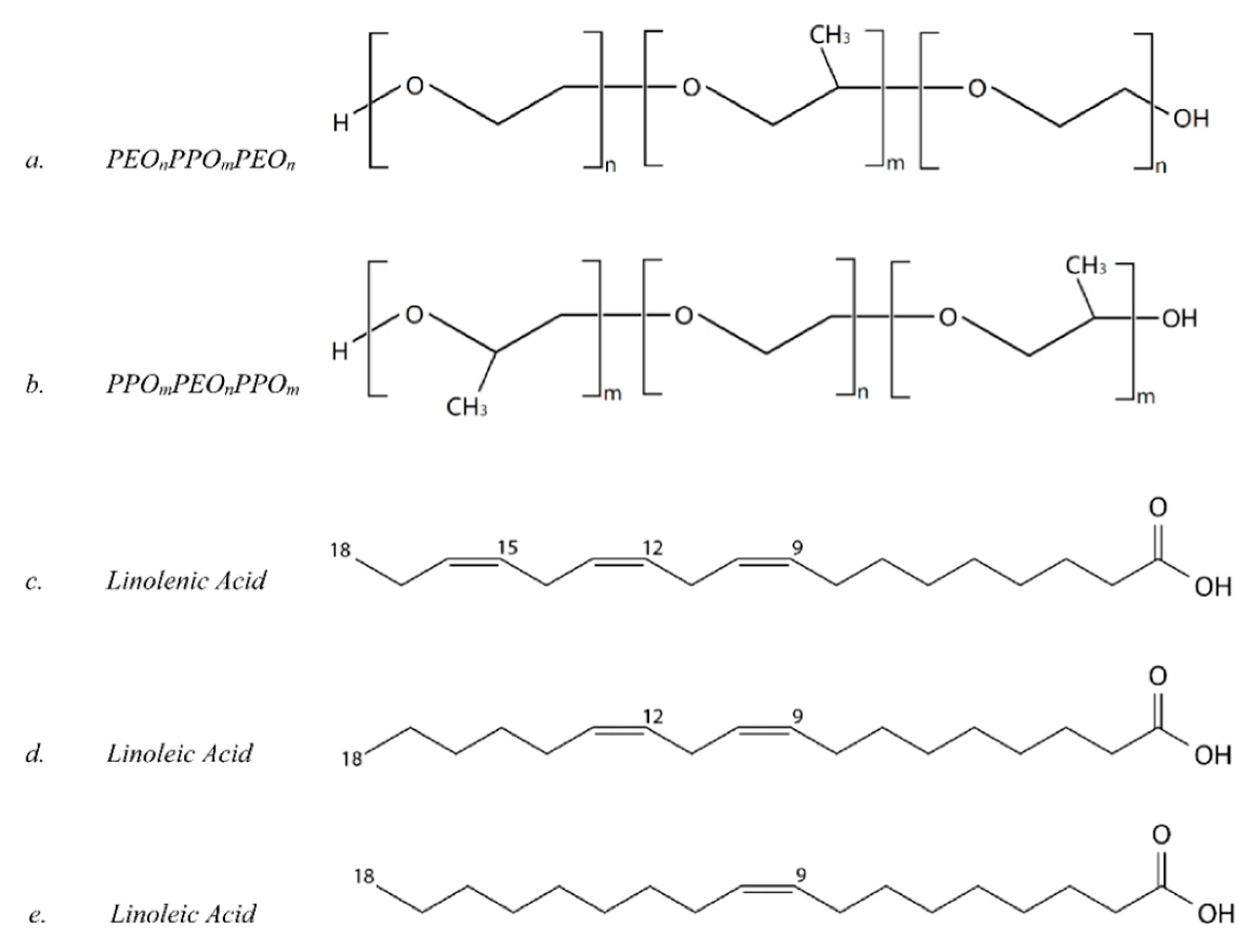





| # | Peak Wavenumber (cm−1) | Element | Functional Group | Pure Oil/Copolymers | Surface * |
|---|---|---|---|---|---|
| 1 | 650–1000 | C–H | Alkanes | All | None |
| 2 | 1100–1200 | C–O | Ester | All | None |
| 3 | 1350–1450 | CH2 | Methylene | 17R4 & L64 | None |
| 4 | 1350–1450 | CH3 | Alkyl | 17R4 & L64 | None |
| 5 | ~1500 | C=C | Alkene | Soybean Oil | None |
| 6 | ~1730 | C=O | Carboxylic Acid | Soybean Oil | None |
| 7 | ~2860 | C–H | Alkanes | All | None |
| 8 | ~2950 | C–H | Alkanes | All | None |
| # | Peak Wavenumber (cm−1) | Element | Functional Group | Solution * | Surface ** |
|---|---|---|---|---|---|
| 1 | 650–1000 | C–H | Alkanes | All | All |
| 2 | ~1100 | C–O | Ester | None | All |
| 3 | ~1200 | C–O | Ester | All | None |
| 4 | 1350–1450 | CH2 | Methylene | 17R4 & L64 | 17R4 & L64 coated |
| 5 | 1350–1450 | CH3 | Alkyl | 17R4 & L64 | 17R4 & L64 coated |
| 6 | ~1500 | C=C | Alkene | None | Soybean oil coated |
| 7 | ~1635 | C=C | Alkene | All | None |
| 8 | ~1730 | C=O | Carboxylic Acid | Soybean oil | Soybean oil coated |
| 9 | ~2810 | C–H | Alkanes | All | None |
| 10 | ~2860 | C–H | Alkanes | None | 17R4 & L64 coated |
| 11 | ~2900 | C–H | Alkanes | All | All |
| 12 | ~3330 | O–H | - | All | None |
| 13 | ~3610 | O–H | - | None | Soybean oil coated |
| 14 | ~3730 | O–H | - | None | Soybean oil coated |
| 15 | ~3860 | O–H | - | None | Soybean oil coated |
| 16 | ~3750 | O–H | - | None | 17R4 coated |
| # | Concentration (%) | Preparation Energy (kJ) | T (°C) | Disc Scar Width (mm) | Ball Scar Area (mm2) | Ball Mass Loss (µg) | COF |
|---|---|---|---|---|---|---|---|
| L64 | 18 | N/A | 30 | 0.89 | 0.87 | 0.24 | 0.360 |
| L64 | 18 | N/A | 65 | 1.23 | 1.69 | 0.94 | 0.342 |
| 17R4 | 18 | N/A | 30 | 0.88 | 0.60 | 0.11 | 0.205 |
| 17R4 | 18 | N/A | 65 | 1.15 | 1.21 | 0.47 | 0.156 |
| Soybean | 1 | 10 | 30 | 0.85 | 0.68 | 0.15 | 0.393 |
| Soybean | 1 | 10 | 65 | 0.34 | Non Visible | Non Visible | 0.107 |
| Soybean | 1 | 5 | 65 | 0.35 | Non Visible | Non Visible | 0.110 |
| Soybean | 2 | 10 | 65 | 0.39 | Non Visible | Non Visible | 0.127 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taheri, R.; Kosasih, B.; Zhu, H.; Tieu, A.K. Surface Film Adsorption and Lubricity of Soybean Oil In-Water Emulsion and Triblock Copolymer Aqueous Solution: A Comparative Study. Lubricants 2017, 5, 1. https://doi.org/10.3390/lubricants5010001
Taheri R, Kosasih B, Zhu H, Tieu AK. Surface Film Adsorption and Lubricity of Soybean Oil In-Water Emulsion and Triblock Copolymer Aqueous Solution: A Comparative Study. Lubricants. 2017; 5(1):1. https://doi.org/10.3390/lubricants5010001
Chicago/Turabian StyleTaheri, Reza, Buyung Kosasih, Hongtao Zhu, and Anh Kiet Tieu. 2017. "Surface Film Adsorption and Lubricity of Soybean Oil In-Water Emulsion and Triblock Copolymer Aqueous Solution: A Comparative Study" Lubricants 5, no. 1: 1. https://doi.org/10.3390/lubricants5010001








