Potential Synthetic Biolubricant as an Alternative to Bovine Serum
Abstract
:1. Introduction
2. Results
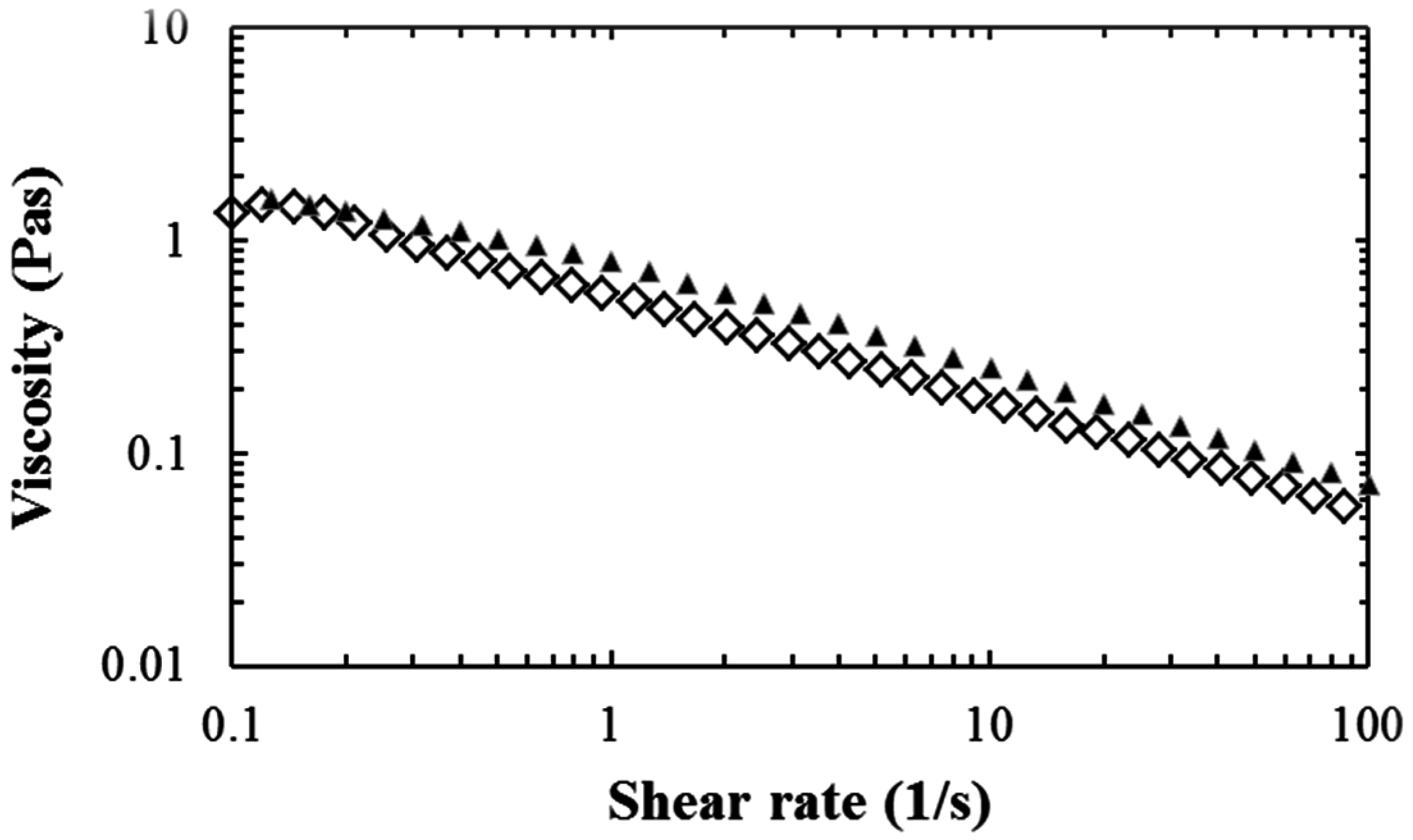
2.1. Viscosity Results
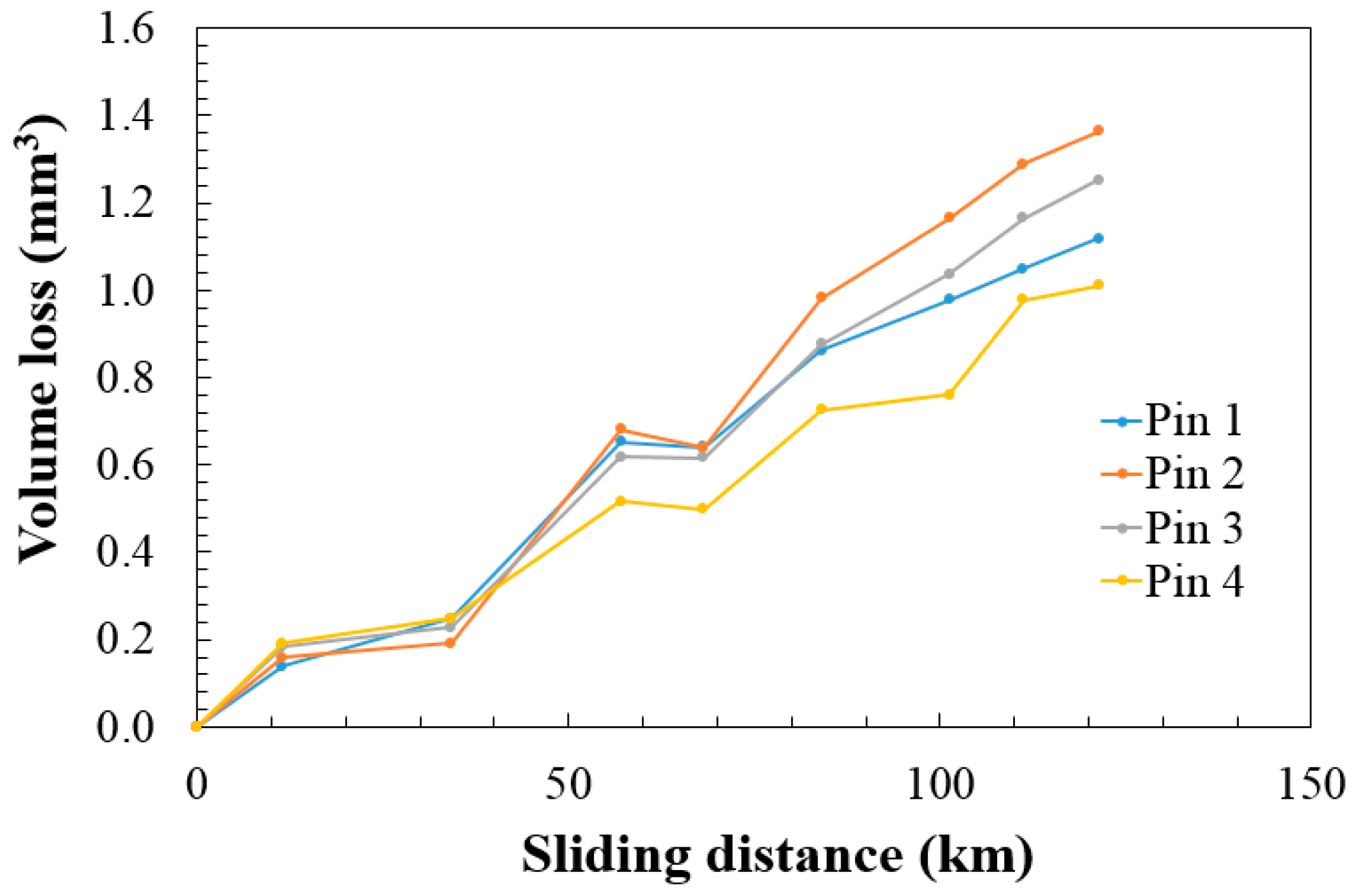
2.2. Wear Results
2.3. Surface Roughness Analysis
2.3.1. UHMWPE Pins
2.3.2. Stainless Steel Plates
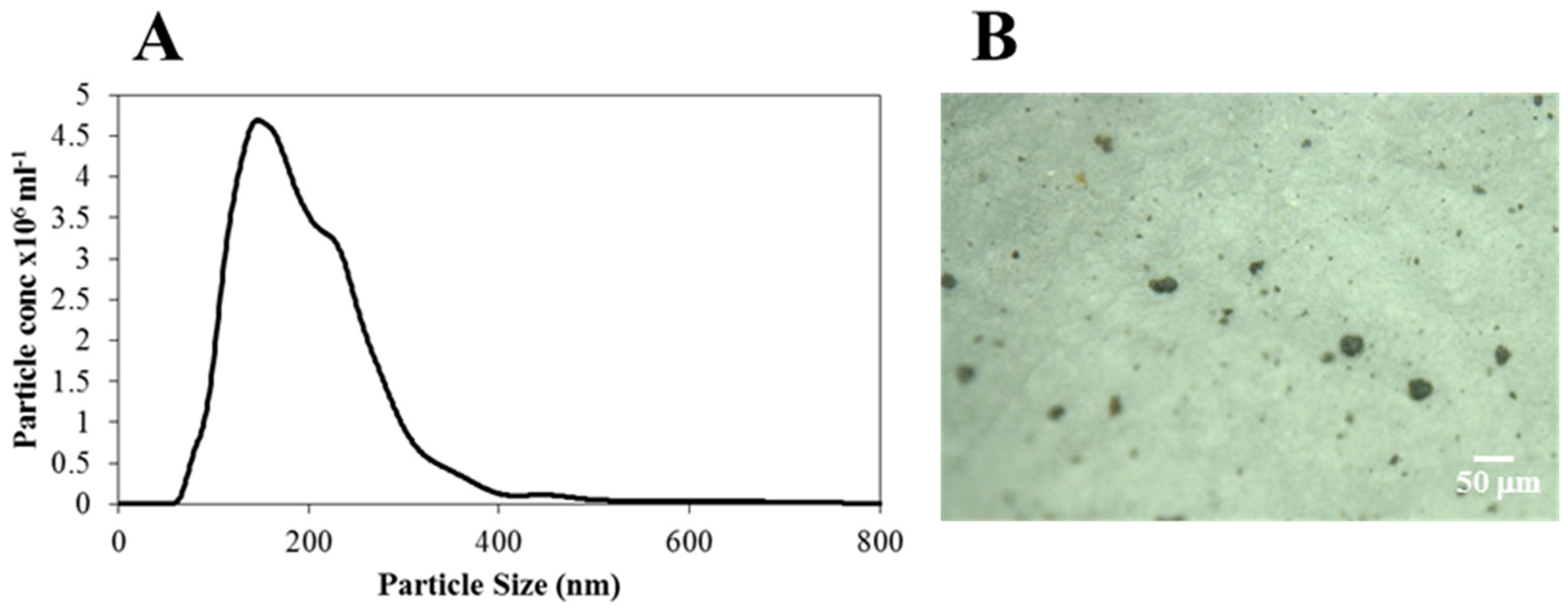
2.4. Wear Particle Analysis
3. Discussion
3.1. Viscosity Results
3.2. Wear Results
3.3. Surface Roughness Analysis
3.4. Wear Paricle Analysis
3.5. Limitation
4. Materials and Methods
4.1. Materials
4.2. Test Lubricant
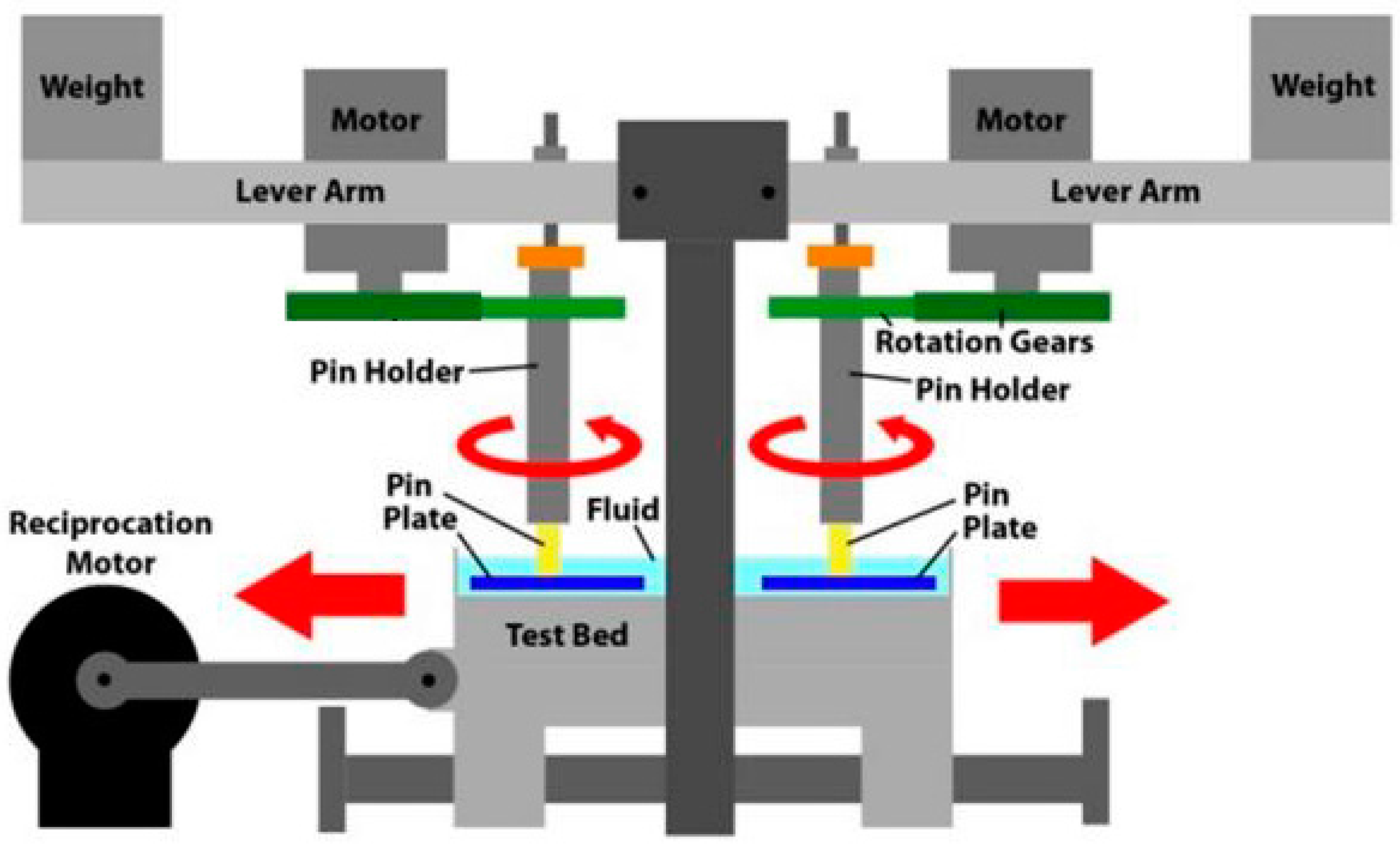
4.3. Apparatus
4.3.1. Pin-on-Plate Wear Test Machine
4.3.2. Surface Roughness Measurements
4.4. Statistical Methods
4.5. Wear Particle Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ISO | International Organisation for Standardisation |
| UHMWPE | ultra-high molecular weight polyethylene |
| NJR | National Joint Registry |
| ASTM | American Society for Testing and Materials |
Appendix A
- At each weighing interval the test bath was removed.
- A 20 mL syringe was used to extract the test fluid from the test and control baths, this fluid was placed into labelled specimen jars.
- The plates were then removed from the test bath and the pins from the pin holders.
- The pins and plates were cleaned in acetone and weighed on a balance with a sensitivity of 10 μg.
- The test bath was also cleaned between tests.
- Each test piece was weighed three times; a mean of these three values was calculated for each specimen.
- After the cleaning, drying, and weighing procedure was complete, the plates were re-located in the correct position in the bath.
- The bath was then fixed into place on the test rig and the pins fixed into the pin holders.
- 20 mL of new lubricant was added to each bath and the test rig was re-started.
References
- National Joint Registry. National Joint Registry of England and Wales; 7th Annual Report; National Joint Registry: Hertfordshire, UK, 2010. [Google Scholar]
- National Joint Registry. National Joint Registry for England, Wales, Northern Ireland and the Isle of Man; 12th Annual Report; National Joint Registry: Hertfordshire, UK, 2015. [Google Scholar]
- Chakrabarty, G.; Vashishtha, M.; Leeder, D. Polyethylene in knee arthroplasty: A review. J. Clin. Orthop. Trauma 2015, 6, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Revell, P.A. Biological causes of prosthetic joint failure. In Joint Replacement Technology, 2nd ed.; Revell, P.A., Ed.; Woodhead: Cambridge, UK, 2008; pp. 315–348. [Google Scholar]
- Revell, P.A. The combined role of wear particles, macrophages and lymphocytes in the loosening of total joint prostheses. J. R. Soc. Interface 2008, 5, 1263–1278. [Google Scholar] [CrossRef] [PubMed]
- Implants for Surgery. Wear of Total Hip-Joint Prostheses. Loading and Displacement Parameters for Orbital Bearing Type Wear-Testing Machines and Corresponding Environmental Conditions for Test; BS ISO 14242-3:2009; The British Standards Institution: London, UK, 2009.
- Implants for Surgery. Wear of Total Knee-Joint Prostheses. Loading and Displacement Parameters for Wear-Testing Machines with Displacement Control and Corresponding Environmental Conditions for Test; BS ISO 14243-3:2014; British Standards Institution: London, UK, 2014.
- Joyce, T.J. Biopolymer tribology. In Polymer Tribology; Sinha, S.K., Briscoe, B.J., Eds.; Imperial College Press: London, UK, 2009; pp. 227–266. [Google Scholar]
- Harsha, A.P.; Joyce, T.J. Challenges associated with using bovine serum in wear testing orthopaedic biopolymers. Proc. Inst. Mech. Eng. H 2011, 225, 948–958. [Google Scholar] [CrossRef] [PubMed]
- Maskiewicz, V.K.; Williams, P.A.; Prates, S.J.; Bowsher, J.G.; Clarke, I.C. Characterization of protein degradation in serum-based lubricants during simulation wear testing of metal-on-metal hip prostheses. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 94, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.; Besong, A.A.; Tipper, J.L.; Ingham, E.; Wroblewski, B.M.; Stone, M.H.; Fisher, J. Influence of gelatin and bovine serum lubricants on ultra-high molecular weight polyethylene wear debris generated in in vitro simulations. Proc. Inst. Mech. Eng. H 2000, 214, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.; Tipper, J.L.; Ingham, E.; Stone, M.H.; Fisher, J. The influence of phospholipid concentration in protein-containing lubricants on the wear of ultra-high molecular weight polyethylene in artificial hip joints. Proc. Inst. Mech. Eng. H 2001, 215, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Gale, L.R.; Coller, R.; Hargreaves, D.J.; Hills, B.A.; Crawford, R. The role of SAPL as a boundary lubricant in prosthetic joints. Tribol. Int. 2007, 40, 601–606. [Google Scholar] [CrossRef]
- Hua, Z.K.; Su, S.H.; Zhang, J.H. Tribological study on new therapeutic bionic lubricants. Tribol. Lett. 2007, 28, 51–58. [Google Scholar] [CrossRef]
- Scholes, S.C.; Joyce, T.J. In vitro tests of substitute lubricants for wear testing orthopaedic biomaterials. Proc. Inst. Mech. Eng. H 2013, 227, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.; Hunt, B.; Smith, A.; Joyce, T. Wear tests of a potential biolubricant for orthopedic biopolymers. Lubricants 2015, 3, 80–90. [Google Scholar] [CrossRef]
- Bortel, E.; Charbonnier, B.; Heuberger, R. Development of a synthetic synovial fluid for tribological testing. Lubricants 2015, 3, 664. [Google Scholar] [CrossRef]
- Ahlroos, T.; Saikko, V. Wear of prosthetic joint materials in various lubricants. Wear 1997, 211, 113–119. [Google Scholar] [CrossRef]
- Smith, A.M.; Fleming, L.; Wudebwe, U.; Bowen, J.; Grover, L.M. Development of a synovial fluid analogue with bio-relevant rheology for wear testing of orthopaedic implants. J. Mech. Behav. Biomed. Mater. 2014, 32, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.M.; Shelton, R.M.; Perrie, Y.; Harris, J.J. An initial evaluation of gellan gum as a material for tissue engineering applications. J. Biomater. Appl. 2007, 22, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, M.H.; Conway, B.R.; Smith, A.M. Evaluation of gellan gum fluid gels as modified release oral liquids. Int. J. Pharm. 2014, 475, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, M.H.; Conway, B.R.; Smith, A.J. Development of mucoadhesive sprayable gellan gum fluid gels. Int. J. Pharm. 2015, 488, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Garrec, D.A.; Norton, I.T. Understanding fluid gel formation and properties. J. Food Eng. 2012, 112, 175–182. [Google Scholar] [CrossRef]
- Farrés, I.F.; Norton, I.T. The influence of co-solutes on tribology of agar fluid gels. Food Hydrocoll. 2015, 45, 186–195. [Google Scholar] [CrossRef]
- Implants for Surgery, Partial and Total Hip Joint Prostheses. Articulating Surfaces Made of Metallic, Ceramic and Plastics Materials; BS EN ISO 7206-2; British Standards Institution: London, UK, 2011.
- Scholes, S.C.; Unsworth, A. Comparison of friction and lubrication of different hip prostheses. Proc. Inst. Mech. Eng. H 2000, 214, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Joyce, T.J.; Monk, D.; Scholes, S.C.; Unsworth, A. A multi-directional wear screening device and preliminary results of UHMWPE articulating against stainless steel. Biomed. Mater. Eng. 2000, 10, 241–249. [Google Scholar] [PubMed]
- Joyce, T.J. Wear tests of orthopaedic biopolymers with the biolubricant augmented by a visco-supplement. Proc. Inst. Mech. Eng. J. 2009, 223, 297–302. [Google Scholar] [CrossRef]
- Hall, R.M.; Unsworth, A.; Siney, P.; Wroblewski, B.M. Wear in retrieved Charnley acetabular sockets. Proc. Inst. Mech. Eng. H 1996, 210, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Sychterz, C.J.; Moon, K.H.; Hashimoto, Y.; Terefenko, K.M.; Engh, C.A.; Bauer, T.W. Wear polyethylene cups in total hip arthroplasty—A study of specimens retrieved post mortem. J. Bone Jt. Surg.-Am. Vol. 1996, 78, 1193–1200. [Google Scholar] [CrossRef]
- Harsha, A.P.; Joyce, T.J. Comparative wear tests of ultra-high molecular weight polyethylene and cross-linked polyethylene. Proc. Inst. Mech. Eng. H 2013, 227, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Hunt, B.; Joyce, T. A tribological assessment of ultra high molecular weight polyethylene types GUR 1020 and GUR 1050 for orthopedic applications. Lubricants 2016, 4, 25. [Google Scholar] [CrossRef]
- Elfick, A.P.D.; Hall, R.M.; Pinder, I.M.; Unsworth, A. The influence of femoral head surface roughness on the wear of ultrahigh molecular weight polyethylene sockets in cementless total hip replacement. J. Biomed. Mater. Res. 1999, 48, 712–718. [Google Scholar] [CrossRef]
- Elfick, A.P.D.; Smith, S.L.; Green, S.M.; Unsworth, A. A re-appraisal of wear features of acetabular sockets using atomic force microscopy. Wear 2002, 253, 839–847. [Google Scholar] [CrossRef]
- Flannery, M.; McGloughlin, T.; Jones, E.; Birkinshaw, C. Analysis of wear and friction of total knee replacements. Part I. Wear assessment on a three station wear simulator. Wear 2008, 265, 999–1008. [Google Scholar] [CrossRef]
- Naylor, A.; Talwalkar, S.C.; Trail, I.A.; Joyce, T.J. In vitro wear testing of a CoCr-UHMWPE finger prosthesis with hydroxyapatite coated CoCr Stems. Lubricants 2015, 3, 244–255. [Google Scholar] [CrossRef]
- Edidin, A.A.; Rimnac, C.M.; Goldberg, V.M.; Kurtz, S.M. Mechanical behavior, wear surface morphology, and clinical performance of UHMWPE acetabular components after 10 years of implantation. Wear 2001, 250, 152–158. [Google Scholar] [CrossRef]
- Scholes, S.C.; Kennard, E.; Gangadharan, R.; Weir, D.; Holland, J.; Deehan, D.; Joyce, T.J. Topographical analysis of the femoral components of ex vivo total knee replacements. J. Mater. Sci. Mater. Med. 2013, 24, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Galvin, A.L.; Tipper, J.L.; Ingham, E.; Fisher, J. Nanometre size wear debris generated from crosslinked and non-crosslinked ultra high molecular weight polyethylene in artificial joints. Wear 2005, 259, 977–983. [Google Scholar] [CrossRef]
- Nine, M.J.; Choudhury, D.; Hee, A.C.; Mootanah, R.; Abu Osman, N.A. Wear debris characterization and corresponding biological response: Artificial hip and knee joints. Materials 2014, 7, 980–1016. [Google Scholar] [CrossRef]
- Scholes, S.C.; Unsworth, A. Pin-on-plate studies on the effect of rotation on the wear of metal-on-metal samples. J. Mater. Sci. Mater. Med. 2001, 12, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Scholes, S.C.; Unsworth, A. Wear studies on the likely performance of CFR-PEEK/CoCrMo for use as artificial joint bearing materials. J. Mater. Sci. Mater. Med. 2009, 20, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Scholes, S.C.; Unsworth, A. The wear properties of CFR-PEEK articulating against ceramic assessed on a multidirectional pin-on-plate machine. Proc. Inst. Mech. Eng. H 2007, 221, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Bragdon, C.R.; O’Connor, D.O.; Lowenstein, J.D.; Jasty, M.; Biggs, S.A.; Harris, W.H. A new pin-on-disk wear testing method for simulating wear of polyethylene on cobalt-chrome alloy in total hip arthroplasty. J. Arthroplast. 2001, 16, 658–665. [Google Scholar] [CrossRef] [PubMed]
- Joyce, T.J.; Vandelli, C.; Cartwright, T.; Unsworth, A. A comparison of the wear of cross-linked polyethylene against itself under reciprocating and multi-directional motion with different lubricants. Wear 2001, 250, 206–211. [Google Scholar] [CrossRef]
- Joyce, T.J.; Unsworth, A. Wear studies of all UHMWPE couples under various bio-tribological conditions. J. Appl. Biomater. Biomech. 2004, 2, 29–34. [Google Scholar] [PubMed]
- Saikko, V. A hip wear simulator with 100 test stations. Proc. Inst. Mech. Eng. H 2005, 219, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Geometric Product Specification, Surface Texture (Areal). Part 2: Terms, Definitions and Surface Texture Parameters; BS EN ISO 25178-2; British Standards Institution: London, UK, 2012.
- Naylor, A.; Talwalkar, S.; Trail, I.; Joyce, T. Evaluating the surface topography of pyrolytic carbon finger prostheses through measurement of various roughness parameters. J. Funct. Biomater. 2016, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Gadelmawla, E.S.; Koura, M.M.; Maksoud, T.M.A.; Elewa, I.M.; Soliman, H.H. Roughness parameters. J. Mater. Process. Technol. 2002, 123, 133–145. [Google Scholar] [CrossRef]
- Sedlaček, M.; Podgornik, B.; Vižintin, J. Correlation between standard roughness parameters skewness and kurtosis and tribological behaviour of contact surfaces. Tribol. Int. 2012, 48, 102–112. [Google Scholar] [CrossRef]



| Station | Wear Factor (×10−6 mm3/Nm) | |
|---|---|---|
| Pins | Plates | |
| 1 | 0.24 | 0.008 |
| 2 | 0.28 | 0.008 |
| 3 | 0.26 | 0.048 |
| 4 | 0.21 | 0.015 |
| Mean | 0.25 | 0.019 |
| Standard deviation | 0.03 | 0.019 |
| Cycles | UHMWPE Pins (Test) | UHMWPE Pins (Control) | |||||
|---|---|---|---|---|---|---|---|
| Mean (95% CI) | t | p | Mean (95% CI) | t | p | ||
| Sa (nm) | 0 | 1293 (1079 to 1508) | 6.11 | <0.05 | 1244 (894 to 1594) | −0.05 | 0.96 |
| 1 × 106 | 582 (353 to 811) | 1156 (783 to 1529) | |||||
| 2 × 106 | 462 (303 to 620) | 1256 (908 to 1604) | |||||
| Ssk | 0 | −0.10 (−0.30 to 0.11) | −5.93 | <0.05 | 0.142 (0.01 to 0.27) | 0.11 | 0.92 |
| 1 × 106 | 1.04 (0.48 to 1.59) | 0.282 (0.12 to 0.45) | |||||
| 2 × 106 | 1.44 (0.97 to 1.91) | 0.129 (−0.09 to 0.34) | |||||
| Sku | 0 | 2.74 (2.48 to 3.00) | −4.34 | <0.05 | 2.370 (2.05 to 2.69) | −0.19 | 0.85 |
| 1 × 106 | 12.18 (2.85 to 21.51) | 2.470 (2.23 to 2.71) | |||||
| 2 × 106 | 7.67 (5.46 to 9.88) | 2.420 (2.06 to 2.78) | |||||
| Cycles | Stainless Steel Plates (Test) | Stainless Steel Plates (Control) | |||||
|---|---|---|---|---|---|---|---|
| Mean (95% CI) | t | p | Mean (95% CI) | t | p | ||
| Sa (nm) | 0 | 14.47 (8.44 to 20.49) | 1.44 | 0.16 | 7.44 (3.44 to 11.44) | −1.88 | 0.08 |
| 1 × 106 | 16.27 (13.54 to 19) | 11.87 (6.15 to 17.59) | |||||
| 2 × 106 | 9.88 (7.51 to 12.25) | 12.32 (9.18 to 15.45) | |||||
| Ssk | 0 | −1.62 (−2.76 to −0.48) | 2.07 | <0.05 | −0.43 (−0.89 to 0.03) | 0.90 | 0.38 |
| 1 × 106 | −3.32 (−3.77 to −2.87) | −1.62 (−2.33 to −0.91) | |||||
| 2 × 106 | −4.15 (−6.26 to −2.05) | −0.85 (−1.63 to −0.06) | |||||
| Sku | 0 | 17.38 (5.22 to 29.54) | −3.56 | <0.05 | 12.91 (4.41 to 30.23) | −0.66 | 0.52 |
| 1 × 106 | 105 (4.20 to 206) | 12.37 (4.73 to 20.10) | |||||
| 2 × 106 | 113 (61.70 to 164.30) | 20.25 (6.96 to 33.54) | |||||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scholes, S.C.; Colledge, C.J.; Naylor, A.; Mahdi, M.H.; Smith, A.M.; Joyce, T.J. Potential Synthetic Biolubricant as an Alternative to Bovine Serum. Lubricants 2016, 4, 38. https://doi.org/10.3390/lubricants4040038
Scholes SC, Colledge CJ, Naylor A, Mahdi MH, Smith AM, Joyce TJ. Potential Synthetic Biolubricant as an Alternative to Bovine Serum. Lubricants. 2016; 4(4):38. https://doi.org/10.3390/lubricants4040038
Chicago/Turabian StyleScholes, Susan C., Coral J. Colledge, Andrew Naylor, Mohammed H. Mahdi, Alan M. Smith, and Thomas J. Joyce. 2016. "Potential Synthetic Biolubricant as an Alternative to Bovine Serum" Lubricants 4, no. 4: 38. https://doi.org/10.3390/lubricants4040038
APA StyleScholes, S. C., Colledge, C. J., Naylor, A., Mahdi, M. H., Smith, A. M., & Joyce, T. J. (2016). Potential Synthetic Biolubricant as an Alternative to Bovine Serum. Lubricants, 4(4), 38. https://doi.org/10.3390/lubricants4040038






