Theoretical and Experimental Study of the Friction Behavior of Halogen-Free Ionic Liquids in Elastohydrodynamic Regime
Abstract
:1. Introduction
2. Theory
2.1. Friction in Elastohydrodynamic Contacts
2.2. Calculating the Barus and Roelands Pressure Viscosity Coefficient (α)
3. Experimental Details
3.1. Lubricants
3.2. Tribological Experiments
3.3. Viscosity of Lubricants
4. Results and Discussion
4.1. Viscosities and Pressure Viscosity Coefficients of Lubricants
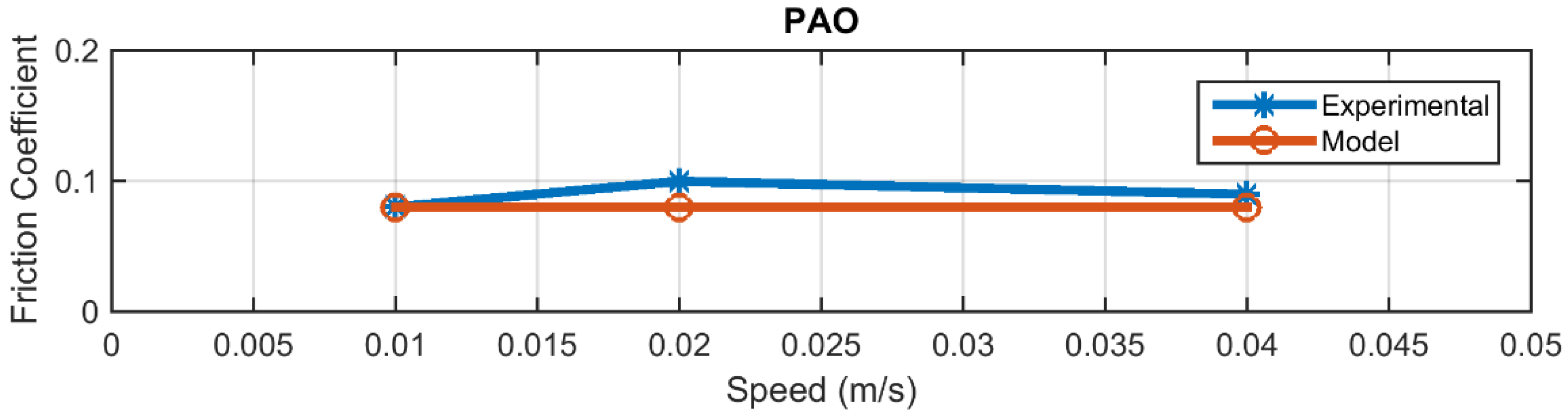
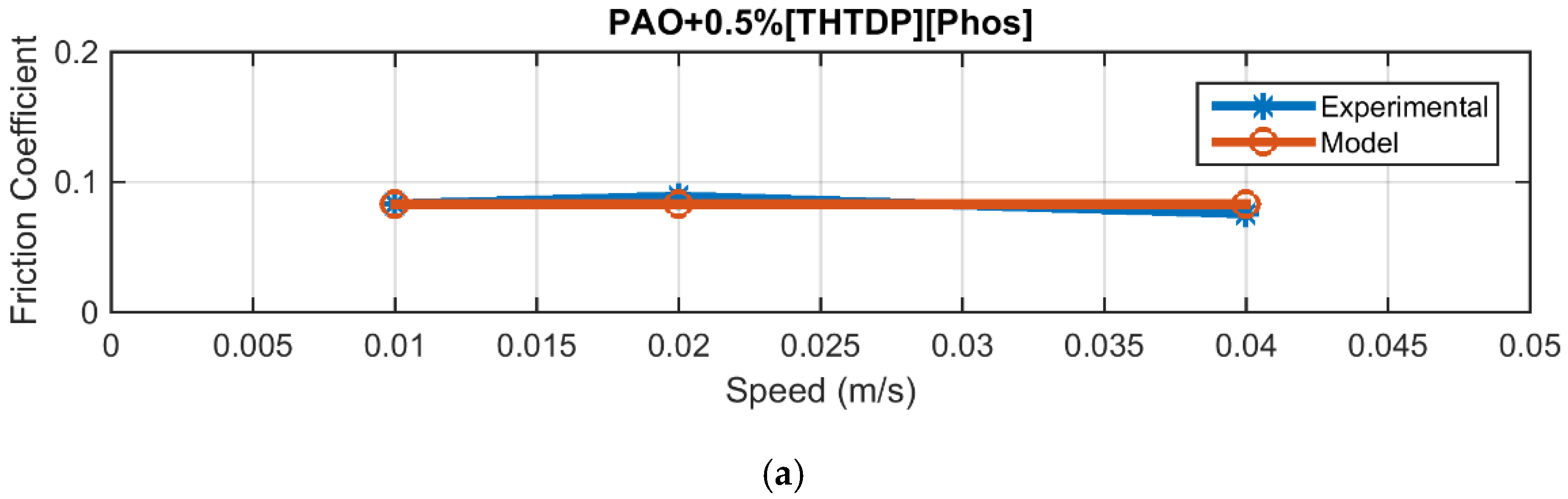
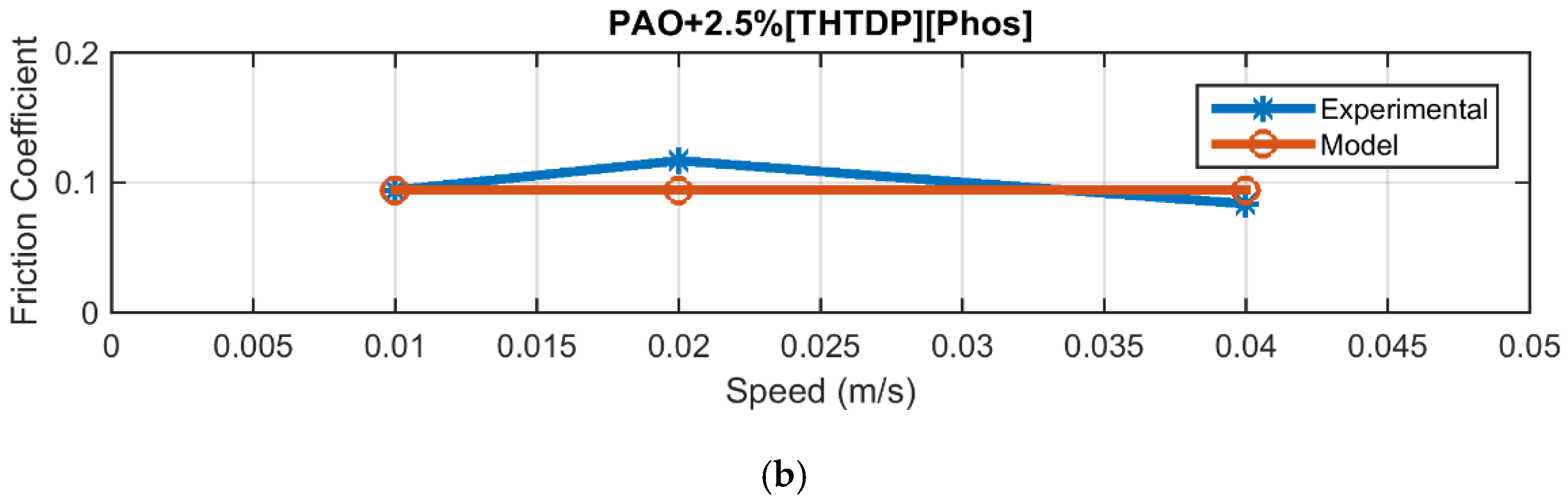
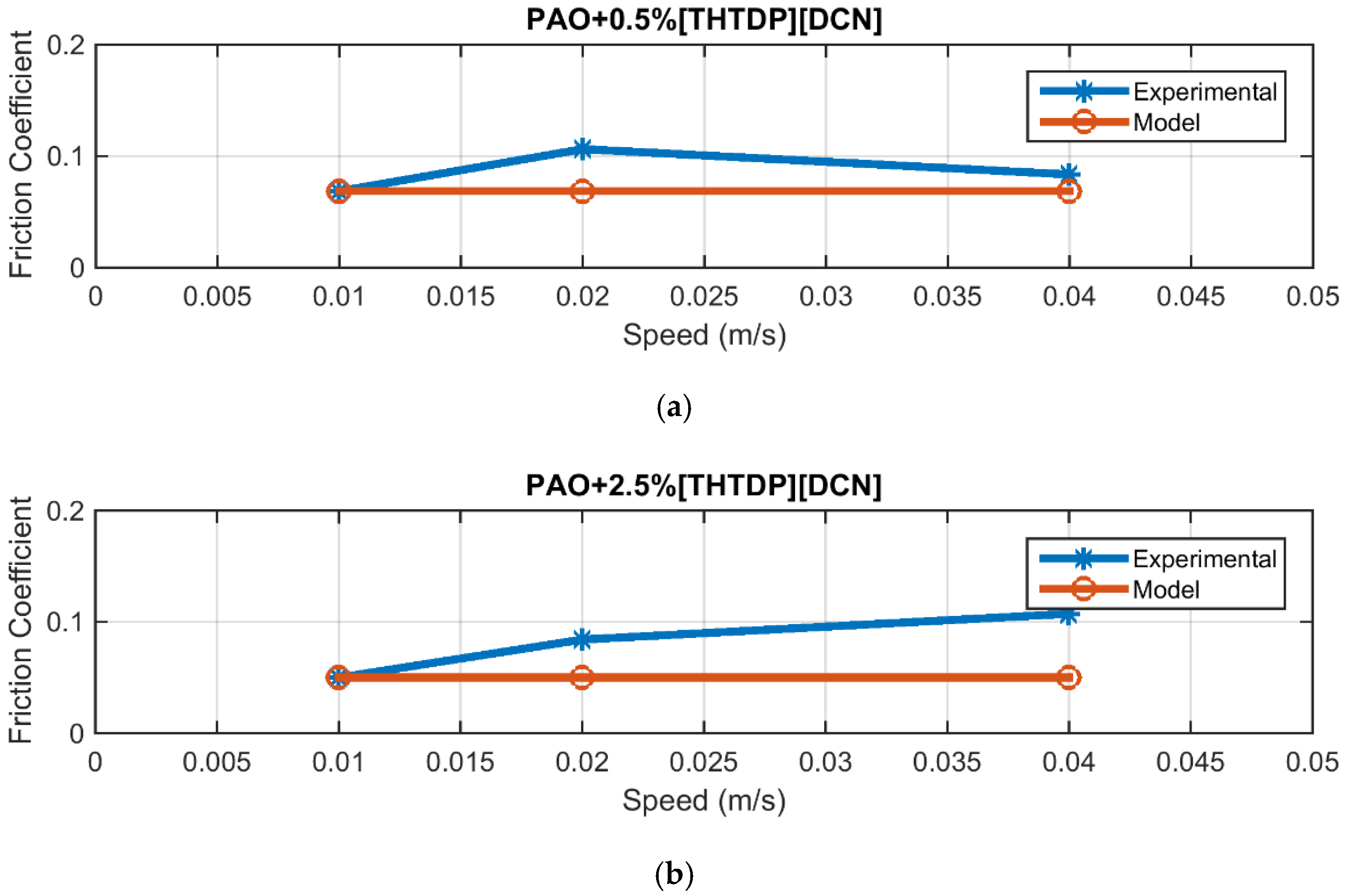
4.2. Friction
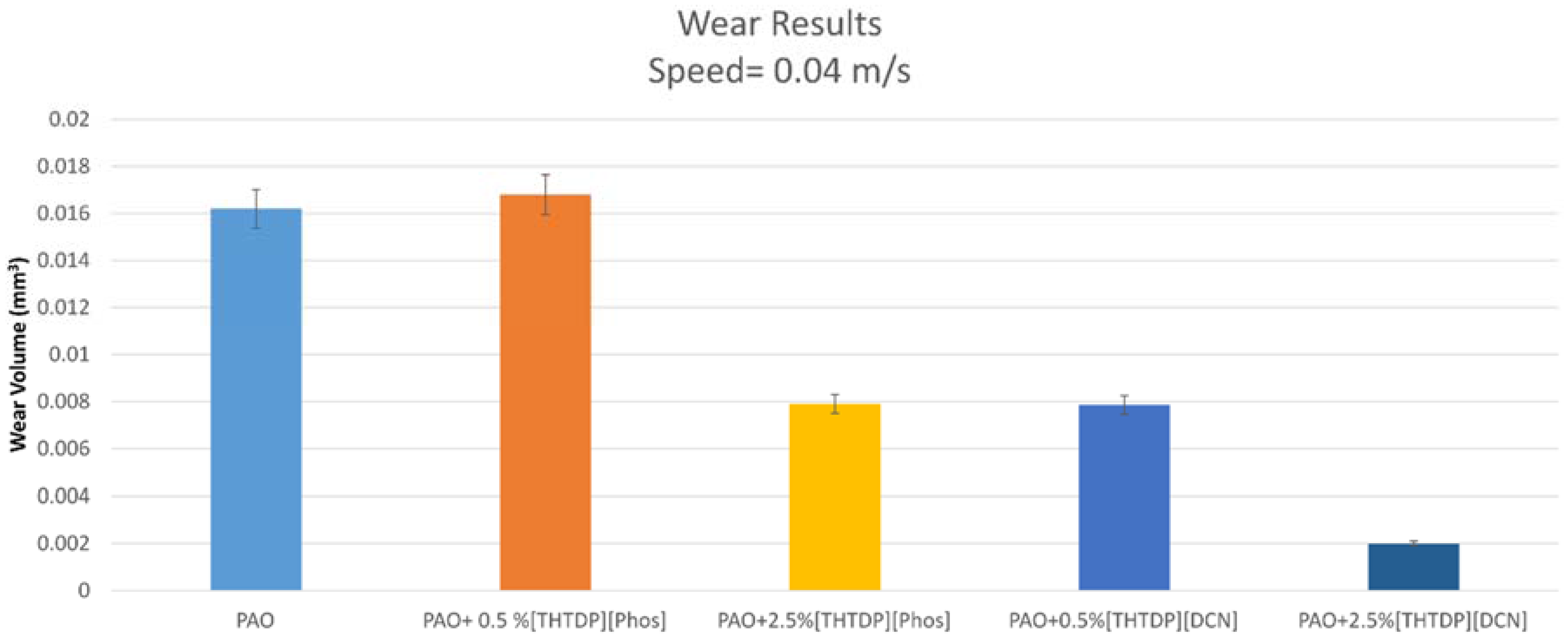
4.3. Wear
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix
| a | Cylinder Diameter (meters) |
| α | Barus Pressure-Viscosity Coefficient (GPa−1) |
| α* | Bloks Reciprocal Isoviscous Pressure Coefficeint (GPa−1) |
| η | Viscosity (cP) |
| p | Pressure (MPa) |
| Z | Roeland’s Pressure-Viscosity Index |
| h | Film Thickness (m) |
| U | Speed (m/s) |
| E* | Young’s Reduced Modulus (GPa) |
| R | Reduced Radius of Curvature (m) |
| λ | Film Parameter |
| σ | Surface Roughness (µm) |
| τ | Shear Stress (N/m2) |
| µ | Coefficient of Friction |
| G | Shear Modulus (Gpa) |
References
- Sripada, P.K.; Sharma, R.V.; Dalai, A.K. Comparative study of tribological properties of trimethylolpropane-based biolubricants derived from methyl oleate and canola biodiesel. Ind. Crops Prod. 2013, 50, 95–103. [Google Scholar] [CrossRef]
- Holmberg, K.; Andersson, P.; Erdemir, A. Global energy consumption due to friction in passenger cars. Tribol. Int. 2012, 47, 221–234. [Google Scholar] [CrossRef]
- Tzanakis, I.; Hadfield, M.; Thomas, B.; Noya, S.M.; Henshaw, I.; Austen, S. Future perspectives on sustainable tribology. Renew. Sustain. Energy Rev. 2012, 16, 4126–4140. [Google Scholar] [CrossRef]
- Zhou, F.; Liang, Y.; Liu, W. Ionic liquid lubricants: Designed chemistry for engineering applications. Chem. Soc. Rev. 2009, 38, 2590–2599. [Google Scholar] [CrossRef] [PubMed]
- Bermúdez, M.D.; Jiménez, A.E.; Sanes, J.; Carrión, F.J. Ionic liquids as advanced lubricant fluids. Molecules 2009, 14, 2888–2908. [Google Scholar] [CrossRef] [PubMed]
- Minami, I. Ionic liquids in tribology. Molecules 2009, 14, 2286–2305. [Google Scholar] [CrossRef] [PubMed]
- Ye, C.; Liu, W.; Chen, Y.; Yu, L. Room-temperature ionic liquids: A novel versatile lubricant. Chem. Commun. (Camb.) 2001. [Google Scholar] [CrossRef]
- Totolin, V.; Minami, I.; Gabler, C.; Dörr, N. Halogen-free borate ionic liquids as novel lubricants for tribological applications. Tribol. Int. 2013, 67, 191–198. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D.; Iglesias, P.; Carrión, F.J.; Martínez-Nicolás, G. 1-N-alkyl -3-methylimidazolium ionic liquids as neat lubricants and lubricant additives in steel-aluminium contacts. Wear 2006, 260, 766–782. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D. Ionic liquids as lubricants of titanium-steel contact. Tribol. Lett. 2009, 33, 111–126. [Google Scholar] [CrossRef]
- Grace, J.; Vysochanska, S.; Lodge, J.; Iglesias, P. Ionic Liquids as Additives of Coffee Bean Oil in Steel-Steel Contacts. Lubricants 2015, 3, 637–649. [Google Scholar] [CrossRef]
- Song, Z.; Liang, Y.; Fan, M.; Zhou, F.; Liu, W. Ionic liquids from amino acids: Fully green fluid lubricants for various surface contacts. RSC Adv. 2014, 4, 19396–19402. [Google Scholar] [CrossRef]
- Espinosa, T.; Jimenez, M.; Sanes, J.; Jimenez, A.E.; Iglesias, M.; Bermudez, M.D. Ultra-low friction with a protic ionic liquid boundary film at the water-lubricated sapphire-stainless steel interface. Tribol. Lett. 2014, 53, 1–9. [Google Scholar] [CrossRef]
- Espinosa, T.; Sanes, J.; Jiménez, A.E.; Bermúdez, M.D. Protic ammonium carboxylate ionic liquid lubricants of OFHC copper. Wear 2013, 303, 495–509. [Google Scholar] [CrossRef]
- Kondo, Y.; Koyama, T.; Sasaki, S. Tribological Properties of Ionic Liquids. In Ionic Liquids—New Aspects for the Future; Intech: Rijeka, Croatia, 2013. [Google Scholar]
- Jacod, B. Friction in Elasto-Hydrodynamic Lubrication. Ph.D. Thesis, University of Twente, Twente, The Netherlands, 2002. [Google Scholar]
- Stachowiak, G.; Batchelor, A.W. Engineering Tribology, 3rd ed.; Butterworth-Heinemann: Burlington, MA, USA, 2005. [Google Scholar]
- Spikes, H.; Jie, Z. History, Origins and Prediction of Elastohydrodynamic Friction. Tribol. Lett. 2014, 56, 1–25. [Google Scholar] [CrossRef]
- Otero, J.E.; Morgado, P.L.; Sánchez-Peñuela, J.B.; Sanz, J.L.M.; Munoz-Guijosa, J.M.; Lantada, A.D.; Yustos, H.L. Elastohydrodynamic Models for Predicting Friction in Point Contacts Lubricated with Polyalphaolefins. In Proceedings of EUCOMES 08 SE—27; Ceccarelli, M., Ed.; Springer: Dordrecht, The Netherlands, 2009; pp. 219–227. [Google Scholar]
- Nishikawa, H.; Handa, K.; Kaneta, M. Behavior of EHL Films in Reciprocating Motion. Trans. Jpn. Soc. Mech. Eng. Ser. C 1992, 58, 1911–1918. [Google Scholar] [CrossRef]
- Pensado, A.S.; Comuñas, M.J.P.; Fernández, J. The pressure-viscosity coefficient of several ionic liquids. Tribol. Lett. 2008, 31, 107–118. [Google Scholar] [CrossRef]
- Harris, K.R.; Woolf, L.A.; Kanakubo, M. Temperature and Pressure Dependence of the Viscosity of the Ionic Liquid 1-Butyl-3-methylimidazolium Hexafluorophosphate. J. Chem. Eng. Data 2005, 50, 1777–1782. [Google Scholar] [CrossRef]
- Harris, K.R.; Kanakubo, M.; Woolf, L.A. Temperature and pressure dependence of the viscosity of the ionic liquids 1-hexyl-3-methylimidazolium hexafluorophosphate and 1-butyl-3-methylimidazolium BiS(trifluoromethylsulfonyl)imide. J. Chem. Eng. Data 2007, 52, 1080–1085. [Google Scholar] [CrossRef]
- Harris, L.K.R.; Kanakubo, M.; Woolf, A.; Data, J.C.E. Temperature and Pressure Dependence of the Viscosity of the Ionic Liquid 1-Butyl-3-methylimidazolium Tetrafluoroborate: Viscosity and Density Relationships in Ionic Liquids. J. Chem. Eng. Data 2007, 52, 2425–2430. [Google Scholar] [CrossRef]
- Fein, R. High Pressure Viscosity and EHL Pressure-Viscosity Coefficients. In Tribology Data Handbook; CRC Press: Boca Raton, USA, 1997. [Google Scholar]
- Roelands, C. Correlational Aspects of the Viscosity-Temperature Pressure Relationship of Lubricating Oils. Ph.D. Thesis, Delft Institute of Technology, Groningen, The Netherlands, 1966. [Google Scholar]
- Leeuwen, H. The determination of the pressure–viscosity coefficient of a lubricant through an accurate film thickness formula and accurate film thickness measurements. J. Eng. Tribol. 2009, 223, 1143–1163. [Google Scholar] [CrossRef]
- Moes, H. Lubrication and Beyond; Utwente Lect. Notes; Twente University Press: Enschede, The Netherlands, 2000; Volume 366. [Google Scholar]
- Qu, J.; Truhan, J.J. An efficient method for accurately determining wear volumes of sliders with non-flat wear scars and compound curvatures. Wear 2006, 261, 848–855. [Google Scholar] [CrossRef]
- Somers, A.; Howlett, P.; MacFarlane, D.; Forsyth, M. A Review of Ionic Liquid Lubricants. Lubricants 2013, 1, 3–21. [Google Scholar] [CrossRef]
- Jiménez, A.E.; Bermúdez, M.D.; Iglesias, P. Lubrication of Inconel 600 with ionic liquids at high temperature. Tribol. Int. 2009, 42, 1744–1751. [Google Scholar] [CrossRef]
- Iglesias, P.; Bermúdez, M.D.; Carrión, F.J.; Martínez-Nicolás, G. Friction and wear of aluminium-steel contacts lubricated with ordered fluids-neutral and ionic liquid crystals as oil additives. Wear 2004, 256, 386–392. [Google Scholar] [CrossRef]
- Palacio, M.; Bhushan, B. A review of ionic liquids for green molecular lubrication in nanotechnology. Tribol. Lett. 2010, 40, 247–268. [Google Scholar] [CrossRef]

| Code | Structure | IUPAC Name | |
|---|---|---|---|
| Cation | Anion | ||
| [THTDP][DCN] | 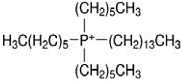 | 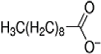 | Trihexyltetradecylphosphonium decanoate |
| [THTDP][Phos] | 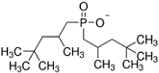 | Trihexyltetradecylphosphonium bis(2,4,4-trimethylpentyl)phosphinate | |
| Speed (m/s) | Roughness (µm) | Film Parameter (λ) |
|---|---|---|
| 0.01 | 0.10 | 6.29 |
| 0.02 | 0.20 | 6.82 |
| 0.04 | 0.40 | 6.98 |
| Lubricant | Viscosity (cP) | Pressure Viscosity Coefficient α* (GPa−1) | |
|---|---|---|---|
| 40 °C | 100 °C | ||
| PAO | 325.00 | 32.00 | 16.38 |
| PAO + 0.5% [THTDP][Phos] | 330.09 | 32.34 | 16.39 |
| PAO + 2.5% [THTDP][Phos] | 331.99 | 32.13 | 16.55 |
| PAO + 0.5% [THTDP][DCN] | 339.00 | 32.70 | 16.51 |
| PAO + 2.5% [THTDP][DCN] | 343.30 | 30.85 | 17.56 |
| Lubricant | RMS Error |
|---|---|
| PAO | 0.0110 |
| PAO + 0.5% Phos | 0.0048 |
| PAO + 2.5% Phos | 0.0125 |
| PAO + 0.5% DCN | 0.0203 |
| PAO + 2.5% DCN | 0.0333 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janardhanan, K.; Iglesias, P. Theoretical and Experimental Study of the Friction Behavior of Halogen-Free Ionic Liquids in Elastohydrodynamic Regime. Lubricants 2016, 4, 16. https://doi.org/10.3390/lubricants4020016
Janardhanan K, Iglesias P. Theoretical and Experimental Study of the Friction Behavior of Halogen-Free Ionic Liquids in Elastohydrodynamic Regime. Lubricants. 2016; 4(2):16. https://doi.org/10.3390/lubricants4020016
Chicago/Turabian StyleJanardhanan, Karthik, and Patricia Iglesias. 2016. "Theoretical and Experimental Study of the Friction Behavior of Halogen-Free Ionic Liquids in Elastohydrodynamic Regime" Lubricants 4, no. 2: 16. https://doi.org/10.3390/lubricants4020016
APA StyleJanardhanan, K., & Iglesias, P. (2016). Theoretical and Experimental Study of the Friction Behavior of Halogen-Free Ionic Liquids in Elastohydrodynamic Regime. Lubricants, 4(2), 16. https://doi.org/10.3390/lubricants4020016







