Abstract
This article presents the effect of laser alloying with boron on the surface layer of iron alloys: steel and grey cast iron. The general goal of this review is to specify the main differences that can be expected after this treatment of selected iron-based alloys. Boron as an alloying element is first characterized. The effects of laser alloying are described in comparison to diffusion processing. The next section describes the effect of laser alloying with boron on the microstructure, hardness, and wear resistance of the surface layer of selected iron alloys. As a result of the conducted analysis, the most significant differences in the outcomes of laser alloying with boron, which may occur during the processing of various iron alloys, are as follows: the presence of graphite in the surface layer in the case of grey cast iron treatment and a clearly visible transition zone between the alloyed zone and the hardened zone during the treatment of grey cast iron as opposed to steel; variable depths of the modified surface layer and varied grain size in the alloy zone depending on the thermophysical properties of the material being treated.
1. Introduction
1.1. Boron as an Alloying Element
Boron is an important additive to metal alloys, especially iron alloys. It is used as a component of the base material and as a surface layer modifier.
This element is used in steel as a micro-additive, for example, in low-alloy welded steels, alloyed tool steels, and heat-resistant superalloys [1]. Boron, in concentrations of a few thousandths of a percent, primarily enhances the hardening properties of heat-treated steels. The hardenability is improved (for example, with boron amount of 3–30 ppm) by its segregation at austenite grains boundaries (this inhibits ferrite and perlite creation), and in this way it promotes the creation (during hardening treatment) of a martensite phase [1,2,3,4] (it has been estimated that higher amounts of >30 ppm boron as a micro-additive cause segregation and precipitation processes that can reduce hardenability and lead to embrittlement [4]). Thus, a small addition of boron (as a minor additive) improves strength and impact resistance, but it has also been found that if the amount is larger, the impact resistance decreases [5]. In general, micro-additives form dispersive precipitates that inhibit austenite grain growth and recrystallization during heat and plastic processing, and they also act as heat transfer zones during welding. This lowers the brittle-to-metal transition temperature and provides additional strengthening to the steel [1].
Additionally, it was also found that the micro-addition of boron in dual-phase steel increases the yield strength, tensile strength and corrosion resistance [6]. Furthermore, boron also reacts with carbon to form boron carbide, or with nitrogen to form boron nitride. These precipitates increase hardenability [2]. In [7], it was found that boron increases the homogeneity and hardness of steel by transforming the structure from a combination containing martensite and bainite to lath martensite (mainly). The studies of Fe-1.5%Mo alloys with different boron contents presented in [8] showed that with increasing boron content, the yield strength increases and ductility decreases. It was also observed in [9] that the hardness and strength of Fe-5 Cr-1.5B-0.45C alloys depend on the presence of boron carbide.
In turn, the studies presented in [10] have shown, for example, that steel containing less than 1% of boron is characterized by increased hardenability and fracture resistance compared to steel without boron. Boron generally forms borides and borocarbides, thereby influencing the microstructure. In the case of Fe-Cr-B-C alloys [11], a hard boron carbide network can be observed in the microstructure, which increases the wear resistance. In [12], it was also found that with increasing boron content in the Fe-Cr-C-Mo-xB alloy, the size of primary M7C3 carbides becomes finer and the hardness increases to over 1000 HV (from almost 700 HV) [12].
Boron, in addition to acting as an alloying element in iron alloys, can also be implemented only into the surface layer of the metal part. For example, in the research presented in [13], boron was introduced into the surface layer by TIG welding. Hard boride phases were observed. This microstructure resulted in a significant improvement in hardness. As a result of another surface treatment, laser cladding, boron has been observed to form metal borides such as M2B or M3B2, in which an iron atom may be replaced by a chromium or manganese atom. These phases increase the hardness and wear resistance of the material [14,15]. It was found that iron coatings with 6% of boron are characterized by the highest hardness and the best wear resistance [14]. Other research has shown that the addition of B (but introduced in B4C) effectively improved the hardness (4 times), fatigue, adhesiveness, and oxidative wear resistance of 316 stainless steel coatings. As a result, Fe23 (B3C3), Cr23 (B1.5C4.5) were formed [16]. Boron could be introduced into the surface layer as an ingredient of Fe-based alloy, as was the aspect of research on laser cladding of 45 steel presented in [14]. As a result, Fe2B (among other phases) was found in the surface layer. Iron borides like FeB and Fe2B were also found after laser alloying with boron and nitrogen on 45 steels [17].
1.2. Laser Alloying vs. Diffusive Method
There are many surface treatments that allow for the implementation of elements into the surface layer of metal parts and that modify, in this way, their tribological properties at various conditions (for example, high temperature, oxidative environment).
Nevertheless, the most common and known methods are chemical heat treatments, where the purpose of these methods is to saturate the surface layer with an element using diffusion.
A diffusion modification creates a complex microstructure within the diffusion layer. Such treatment, for example with boron, increases hardness, wear resistance, and corrosion resistance [18,19]. The microstructure is therefore composed of both types of iron borides (FeB and Fe2B) with a needle-like morphology. FeB appears near the surface, with Fe2B beneath it. The hardness of Fe2B borides can be 1400–1800 HV, while FeB can be higher, even from 1800 to 2400 HV. The borided layer has been found to have improved surface fatigue strength, resistance to abrasive wear, fretting and erosion, corrosion, and corrosion fatigue. Such treatment has also been found to increase the heat resistance (up to 800 °C in the atmosphere).
Although diffusion boronizing generally improves the mechanical properties of the surface layer, it should be noted that forming FeB can originate cracking and spalling. The presence of brittle FeB and the needle-like microstructure of the layer can increase embrittlement. The layer under borides is not hardened [20,21].
The boronizing diffusion process causes the formation of a boride layer (in case of grey cast iron) of approximately 0.08 mm after 4 h (but it needs to be noted that diffusion processes can take longer, even several dozen hours) of saturation at a temperature of 950 °C [22]. Conducting the diffusion process in a furnace makes it possible to modify the surface layer on the whole treated machine part. And many parts can be processed at once. Element carrier media used in the process need to be utilized [22]. Diffusion-borided surface layers resist wear at higher temperatures. For example, in high-entropy alloys, after boronization at 900 °C for 6 h, the 50 μm borided layer is stable, and B2O3 and SiO2 form on the surface, providing wear resistance up to 600 °C. It was stated that their presence can be attributed to the good lubricating The hardness increased over 6 times [23]. It was found that after boronizing, high-entropy alloys were characterized by a lower friction coefficient in various conditions of friction, as well as in dry or in liquid [24].
Another method that allows the formation of a surface layer with boron is, for example, magnetron sputtering [25,26]. Although it is quite a popular method, its main problems are the relatively slow deposition rate of some metals, that some gaseous impurities may also be present, and a need for vacuum systems. The next method is boriding carried out by treating the steel surface with atomic beams of boron [27], but this is a new method that is not fully tested. Another way to obtain boron-enriched layers is to use hybrid treatments like surface mechanical attrition treatment (SMAT) and surface thermochemical treatment [28]. This hybrid method is characterized by complexity and difficulty in controlling the whole process.
An alternative method to diffusion thermochemical treatment is laser alloying. It involves introducing an element (or a mixture of elements) into the surface layer of a machine part using laser beam heating. In this process, the laser beam simultaneously fuses the coating (the paste with the alloying element) with a thin layer of the coated material (e.g., steel). These materials are then mixed in a liquid state and rapidly cooled. As a result, the formed and coagulated alloy differs from the steel and paste with the component. Based on the Fe-B system, it can be seen that the solubility of boron in the iron crystal lattice is very low. Higher boron content leads to the formation of stable and hard intermetallic boride phases FeB and Fe2B. For FeB boride, the boron content is 16.23% wt., and for Fe2B, 8.83% wt. [18]. During diffusion boronizing (that is equilibrium process), such borides form in the surface layer. The structure of such a layer is relatively easy to identify. During laser heat treatment, after melting the iron alloy with boron addition, the layer is cooled rapidly, leading to the formation of non-equilibrium structures. The structure of the entire layer is more complex than that formed after diffusion boronizing. For example, in addition to iron borides, the presence of boron-supersaturated solid solutions can also be expected. As our own research showed [29], the microstructure of the alloyed zone after laser treatment with boron highly depends on the cooling rate (various cooling rates of the alloyed area cannot be applied in the case of diffusive methods). Although α-Fe, Fe2B, and needle-like Fe3C phases were detected in all microstructures cooled at different rates, stronger boride and cementite peaks were detected in X-ray diffraction at a cooling rate of 8 × 103 °C, indicating their higher content in this microstructure than in the microstructure cooled eight times longer. This can be explained by the greater number of iron boride crystallization nuclei during the faster cooling. Overall, ultrafine crystalline microstructures were observed in the layer cooled at the faster rate, in contrast to the layer cooled at the slower rate, where larger, polygonal iron boride phases were found. Hardness measurements confirmed the difference between the microstructures described above. Cooling of the microstructure at the higher rate was characterized by a value of 1300 HV0.1, and that at the lower rate was characterized by 1100 HV0.1.
As described above, the microstructure as well as the properties of the surface layer after the laser treatment with boron are not just the result of the saturation of the base material with this element; the final effect also depends on the laser processing conditions like the cooling rate, which is dependent on the laser beam parameters such as its power, the scanning rate, and its diameter. These basic parameters determine the laser beam power density, its interaction time, and finally its fluence. For example, the higher the value of the laser beam power density and the longer its interaction time, the higher the temperature in the surface layer is, so more intense changes in the surface layer can be expected. If a low value of the laser beam power density and a short interaction time are employed, changes in the solid state can be expected, such as stress relief, annealing or tempering. If the laser beam power density is higher than the hardened form, a solid state can be achieved. So, the effect of the martensite microstructure is high hardness and compressive stresses that are converted, for example, to an increase in wear resistance. If the laser beam power density is even higher (or/and the interaction time is longer), the temperature may be high enough to cause melting. Then, as a consequence of immediate cooling, many crystallization nuclei appear, causing a fine-grained microstructure, and as a result of hardening, martensite and other supersaturated phases are formed. Such a microstructure is characterized by higher hardness and wear resistance in comparison to hardening from the solid state. Additionally, the remelted microstructure is more resistant to corrosion. But a remelted microstructure could also be varied and controlled by the laser beam parameters. By appropriately selecting the power density value and its interaction time on the surface, as mentioned above, the cooling rate of the surface layer can be influenced. For example, a high power density value and a short exposure time will result in rapid cooling, which results in a fine-crystalline microstructure with supersaturated solutions and high hardness, translating into greater resistance to tribological wear, which was the subject of one study [30]. Of course, as a result of higher stresses, cracks may appear. In turn, a relatively low laser beam power density and a longer interaction time will result in a slower cooling time, resulting in a microstructure that is not so fine and crystalline, as in the case of a fast cooling rate. Additionally, any increase in the laser beam power density or/and the interaction time will result in a deeper modified layer. This is particularly important in the case of alloying, as a larger melted area will reduce its content. In the case of grey iron if long interaction time (or/and high laser beam density) is applied, more graphite is diluted in the solution. Consequently, less graphite phase is present in the surface layer in this case. In turn, an application of very high laser beam density (and/or long interaction time) evaporation can be expected.
Generally, laser alloying causes a more uniform and finer microstructure than the base material [31]. Usually, the laser alloyed layer, among others, martensite, cementite, retained austenite, and, in case of grey cast iron, also graphite [32]. As opposed to diffusive methods it is also possible to reach an amorphous structure [33]. Metallic glass in this case has been achieved on cast iron by adding special combination of three alloying elements: boron, silicon and cobalt (and applying fast cooling rate, as is possible in case of laser treatment, prevent forming crystal structure) in various proportions: 40-30-30, 50-25-25, 20-40-40. Boron was used to obtain heat resistance, cobalt to obtain corrosion resistance, and silicon as a catalyst for the formation of the glassy state. In 5 out of 27 samples, an amorphous structure was obtained. The XRD and TEM observation (in the research presented in [34]) showed that a laser beam scanning speed of approximately 80 m/min (with a cooling rate of 105 °C/s on the treated surface) caused a large volume fraction of amorphous phase and some crystalline dendrites in the layer. The process of amorphous structure creation using laser treatment is also characterized in [35,36].
Generally, even though there is no amorphous structure, the layer created after laser alloying is more uniform and homogenous than the needle-shaped microstructure of borides in the case of, for example, diffusive boronizing. Laser alloying allows for control over the amount of element added to the surface layer, which, for example, allows for avoiding the formation of FeB when alloying with boron. Laser alloying also allows for a consistent distribution of boron from the surface throughout the modified layer. And it has also been observed that the depth of the boronized layer can be greater than that achieved by the diffusion method [31]. Usually, the hardness of the alloyed layer is over 1000 HV, but it depends on the kind of alloying element.
Under the alloyed zone, after laser alloying of the surface layer, except for the zone that was melted during the heating, the heat-affected zone (HAZ), as opposed to the effects of diffusive methods, can be observed. HAZ is characterized by the presence of martensite. So, in this case, the surface layer is additionally reinforced. Also, the hardness decrease from the surface to the core material direction is gentle. As a consequence of gentle changes in the internal stresses, the cross-section of the surface layer of treated material should also be changed, which is not without significance in the case of durability and reliability of machine parts [31]. An appropriate gradient of boron in the surface layer of 45 steel achieved by laser cladding was the aspect studied in [37]. It was found that chemical gradient optimization effectively improved interlayer stress concentration. It reduced defects, like cracks.
As a result, laser alloying increases the wear resistance of treated components. In the research presented in articles [38,39], lower mass loss of treated parts was noted after tribological tests. For instance, an 18-fold lower mass loss of alloyed samples with silicon carbide was noted, and a 24-fold lower mass loss of samples alloyed with tungsten carbide (than untreated samples) was observed [39]. Also, a better resistance to other types of wear, like fatigue [40] and thermal, was noted [41]. Moreover, a 2-fold increase in the elasticity modulus, 15% decrease in the creep parameter, and 2.5-fold decrease in the plastic work were noted in the case of a laser alloyed layer compared to the base material [42].
The next issue to consider (besides comparing microstructure and properties) is the economic issue. From an economic point of view, diffusive chemical heat treatment is generally a cost-effective treatment, as it is simple and uses conventional methods and materials [18,19]. It is excellent when the whole machine part needs to be treated, or when many parts need to be treated at once. But from another perspective, laser alloying allows for treating the surface layer very quickly (then, of course, the energy consumption is high but only for a very short time) and only in the area where it needs to be changed (area exposed to wear, for example), so that expensive elements could be saved. There is no need to utilize the substances that carry the alloying substance as in the case of the diffusive method. Therefore, in many cases, laser alloying is a useful alternative method to the diffusive method.
Nevertheless, there is a need to mention the limitations of laser alloying, especially with elements that are prone to forming very hard, and usually brittle, phases. Cracks in the case of diffusion boronizing are commonly observed, particularly when the FeB phase is formed, because this one is harder and more brittle than Fe2B. It is appropriate to avoid the formation of FeB boride, and in the case of laser processing, by limiting the amount of boron introduced and controlling the depth of the layer by appropriate selection of laser beam parameters. Nevertheless, considering the case of the laser treatment process, an additional risk during their formation is cracks due to fast cooling. The excessive stresses in the surface layer can lead to the formation of discontinuities in the layer. To avoid such an effect, one can control the conditions for creating a microstructure using appropriately selected values of laser beam parameters that favour the formation of a continuous structure. Another solution is to heat the component to be laser processed. Another problem with laser treatment occurs when creating a raster, which is used when it is necessary to create larger surfaces modified by laser processing. Overlapping modified paths interact with each other. Specifically, the modified path being formed heats the previous path, partially causing tempering processes within it. This results in a heterogeneous microstructure and, consequently, hardness. The solution to this problem is to increase the diameter of the laser beam if possible.
The next aspect of the laser alloying that needs to be taken into account is that even when performed under the same parameters and conditions, it causes different effects when applied to different materials, even if the alloys are made of the same base, such as iron. Certain differences between alloys should be considered prior to their processing. Consequently, the overall aim of the following review is to identify the main differences that can be expected after laser alloying selected iron-based alloys with boron. The goal of this review article is to highlight one of the problems related to laser processing. Its effects depend not only on the conditions of the laser processing itself but also to a large extent on the type of base material.
In this comparison, steel and grey cast iron are considered, since they are the most common iron alloys used in practice. For example, cast steel, despite having better mechanical properties than grey cast irons, has limited application due to poor casting properties, such as the need to use very large risers, sometimes larger than the casting itself. Another iron alloy is white cast iron, but its application is very limited because of its brittleness. As a consequence, surface heat treatments, including laser treatment, of cast steel and white cast iron are not commonly performed.
Laser heat treatment is also used in cases of other metal alloys like aluminum or titanium alloys, or high-entropy alloys (which are already being tested for laser cladding with boron [43]), but it is not as common as in for iron alloys, especially steel and grey cast iron. This is due to their greatly limited application in comparison to iron alloys.
In many cases, surface treatment of steel and cast iron components is necessary. In many cases, only a localized change in the properties of the surface layer is necessary, which is where laser heat treatment is ideally suited.
Many examples of the need for local treatment can be found, for example, in the automotive industry. For instance, the crankshaft journals (which may be made of steel or ductile iron) should be characterized by high hardness and resistance to frictional wear. The situation is similar in the case of the camshaft, where such properties must be provided not only to the journals but also to the cam noses. The surface of the brake discs or drum discs (made of flake cast iron) should be resistant to abrasive wear. Laser heat treatment can also be used in this case. The surface of pistons, piston rings, and cylinders should also be strengthened by surface treatment.
Another area of application is the agricultural industry. Many components of machines, such as those working in the ground, that are made of cast iron or steel, are also susceptible to wear, not across their entire surface, but only in parts. These include coulter flaps and plough shares.
2. The Effect of Laser Alloying with Boron of Selected Iron Alloys
2.1. Microstructure of the Surface Layer
2.1.1. Steel
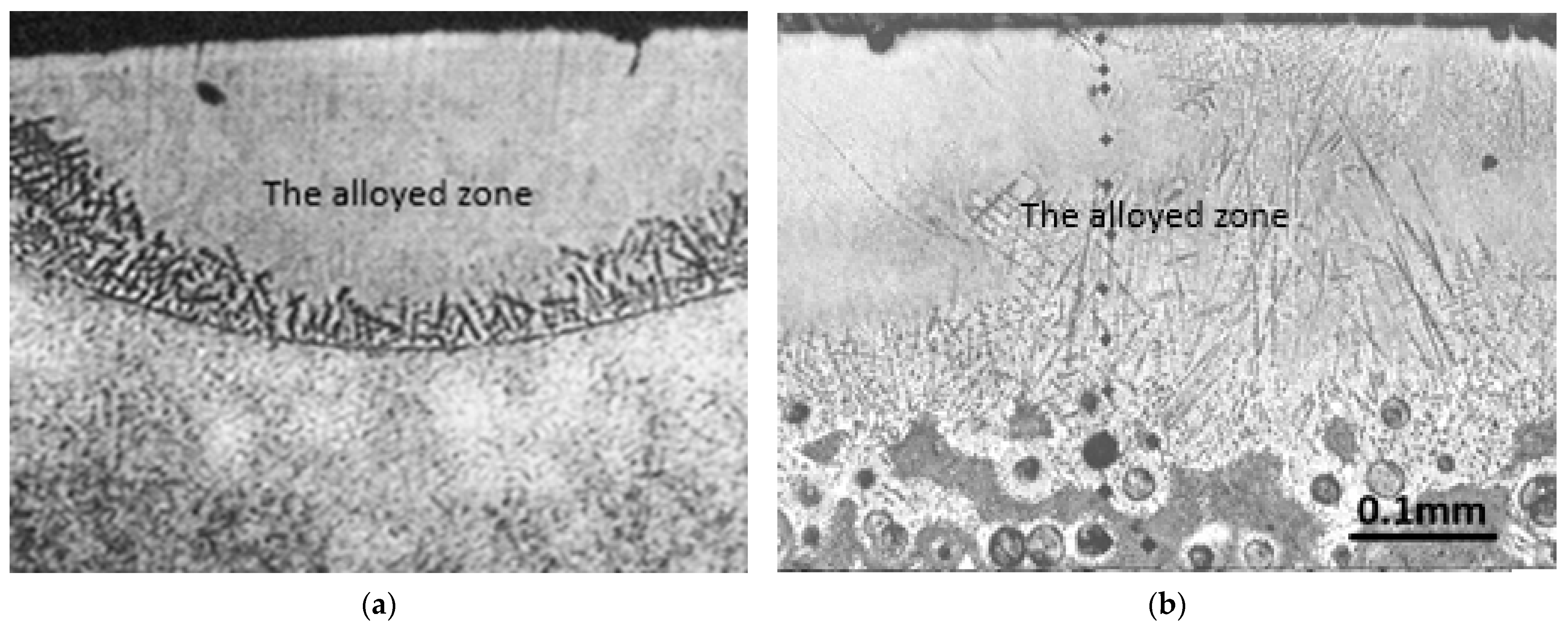
The effects of laser alloying of steel on the surface layer can be distinguished as the alloyed zone (with boron) and the heat-affected zone under it [31]. The microstructure of the alloyed zone was found to be fine with martensite formation [44] and other hard phases [45]. The alloyed zone was also found to be quite homogeneous (especially in the area near the surface). A clear dendritic microstructure was visible at the border of the heat-affected (not melted) zone. The alloyed zone can be seen as the first one from the surface in Figure 1a, with a homogenous structure (particularly in comparison to the base material) and dendrites nucleating during the solidification at the boundary with the non-melted material. Under this border, the heat-affected zone can be noticed. The heat-affected zone contains a hardened zone. If steel is hardened before the laser treatment, then additionally a tempered zone can be seen under the hardened zone, as in the case of laser alloying bearing steel with boron [31]. Laser alloying steel with boron results in high compressive stresses [44].
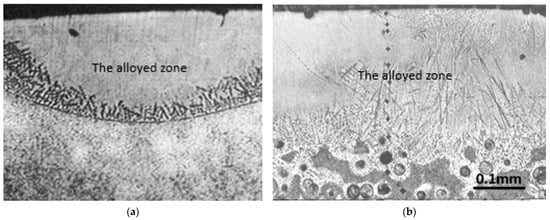
Figure 1.
Microstructure of the surface layer after laser alloying steel with boron (a) and nodular cast iron (b).
The microstructure of the alloyed zone can contain various phases. FeB, Fe2B, and martensite could be found most commonly. Borides are formed due to the presence of boron. As was proved [31], it is possible to introduce 13% at. of boron in the alloyed zone of steel (detected by the AES method). Thus, it is enough to form the eutectic structure of (α + Fe2B). Fe2B as well as FeB have been found in the layer after laser alloying carbon steel with boron [46]. Due to the presence of borides in the alloyed zone, an increase in corrosion resistance was stated [44]. It is a result of the formation of a passivation layer. It decreases the general corrosion, pitting corrosion, and resistance to degradation to hydrogen.
In turn, in the case of laser alloying with boron of 18Ni-300 maraging steel, the alloyed zone showed borides such as FeB, Fe2B, and Fe3B (taking into account that other metals are also present, complex borides like MB, M2B, and M3B could occur, where M could also be Ni, Co, Mo, Ti). Microstructure observation revealed a primary γ-Fe phase and fine lamellar boride-based eutectics in the microstructure of the alloyed zone. It was also observed that dendrites of a solid solution of Fe have been surrounded by a boride shell. It was noticed that the higher the laser operating velocity, the finer the microstructure was. The laser operating velocity also influenced boron content (boron was found in the range from 3 to 6% wt.) [47]. Laser alloying of 41Cr4 steel with boron resulted in a martensite structure with Fe2B and also in metastable Fe3B [19].
2.1.2. Grey Cast Iron
Firstly, the role of the graphite phase in cast iron during laser processing also needs to be considered. First of all, the graphite phase exists on the surface, absorbing the laser beam much more than the metal matrix. Graphite is also characterized by different thermophysical properties than steel and the metal matrix of cast iron. For example, the specific heat is one-third higher, and thermal conductivity parallel to the plane of graphite layers can be over 140 times higher. So, the interaction of the laser beam with a metallic material containing additional non-metallic phases is extremely complex. An additional aspect is the morphology of graphite. Due to the various shapes of graphite in cast iron, various effects can be expected. For example, flakes are more prone to dilution in liquid metal alloy during laser melting (including laser alloying) than nodules. Also, size is important. A small graphite phase will dissolve faster than a large one. The effect of more intense dissolution in the case of flakes or smaller graphite precipitates is a greater saturation of the liquid metal during remelting, and consequently greater carbon enrichment in the newly formed phases after solidification. Thus, a high carbon content can result in a larger amount of primary cementite or a larger amount of retained austenite as a consequence of incomplete martensitic transformation.
After laser alloying of cast iron, as in the case of steel, in the surface layer, the alloyed zone (melted with boron) and hardened zone form the visible solid state. Additionally, a clearly visible transition zone is marked between them (as a result of the structural diversity due to dissolving carbon from the graphite into the matrix in its vicinity). In the alloyed zone (except α-Fe), newly formed hard phases like boride irons—Fe2B and cementite—Fe3C can be found. The homogeneity of the microstructure of the alloyed zone has been proven by stable element distribution [29]. Especially in the case of nodular iron, graphite nodules can be noticed in the alloyed layer. They are especially present near the surface. This means that nodules are ‘pushed’ by solidifying crystals on the border of the transition zone. It was noted that more undiluted graphite is present in zones formed with higher cooling rates than in zones formed with lower rates. This is due to the shorter time for the possible dilution of graphite in the liquid matrix during the treatment. This is an important aspect if graphite (as a solid lubricant) is needed for applications in surfaces that will be used in high-friction conditions.
Nevertheless, the content of graphite depends on the laser treatment conditions. For example, if a shorter integration time of the laser beam was applied, the amount of graphite nodules would be higher in the alloyed zone. So, this means that during the melting process, more graphite nodules are diluted in the liquid matrix when the interaction is longer. In the case of the cooling rate of approximately 103 °C/s, it was found that iron borides with a polygonal shape were observed. Additionally, needle-like primary cementite phases have also been found. On the other hand, if the cooling rate was 8·103 °C/s, the microstructure of the alloyed zone would be ultra-fine, contrary to the alloy zone cooled more slowly [29].
As was mentioned above, contrary to the structure of the steels’ surface layer after laser alloying in the case of grey cast iron, especially in nodular cast iron, the occurrence of a transition zone is clearly visible (Figure 1b). It contains both melted and not melted grains. For example, it is characterized by graphite nodules in a double shell; the first one is made of ledeburite and the second one of martensite. Under the transition zone in grey cast iron is a hardened zone. In this case, this zone is much more complex, contrary to steel’s hardened zone. It is due to the more complex microstructure of grey cast iron (containing graphite) than steel. For example, in the case of nodular cast iron, it contains graphite nodules in a martensite shell in the matrix made of ferrite and martensite [29].
2.2. Depth of Modified Layer
In the case of carbon steel, the depth of the alloyed zone that was formed in this steel was 0.2–0.3 mm [46,48]. The depth of the alloyed zone of high-carbon bearing steel was from 0.14 to 0.4 mm [29], and in the case of maraging steel the depth was 0.08–0.18 mm [47]. Nodular cast iron had the smallest depth, which was noted to range from 0.19 to 0.7 mm in the research presented in [29]: 0.19–0.32 mm [47]: 0.1–0.5 mm and [49] 0.3–0.4 mm with eutectic microstructure and 0.6–0.7 mm with hypoeutectic microstructure, respectively.
These various depths of alloyed zone are not only affected by various laser treatment conditions, like values of laser beam parameters, but also by the steel depth of the alloyed zone (and the whole modified surface layer), which can vary for grey cast iron even if the same conditions of treatment are provided.
This is due to their strong dependence on the thermophysical properties of the treated alloy. Consequently, in the case of treating different alloys, the depths of the modified layer will vary (and even the result of modification will be different).
The thermophysical properties of steel are different from those of cast iron. Also, different types of steel or cast iron possess a wide range of those properties. For example, 450 to 545 J/kg·K is the specific heat for grey cast iron, and 30 to 60 W/m·K is the thermal conductivity [47]. Examples of values of these properties are presented in Table 1.

Table 1.
The comparison of approximate values of thermophysical properties of chosen iron alloys.
Based on the thermophysical properties of treated material and laser treatment conditions, it is possible to predict the temperature changes in the section from the treated surface to the core direction and the melted zone depth by using the following equation cited in [47]:
where
- T(y,z,t) is the temperature [K] at (y,z); y, z are coordinates [m];
- T0 is the ambient temperature [K];
- A is the absorptivity [%];
- P is the laser beam power [W];
- λ is the base material thermal conductivity [W·m−1·K−1];
- v is the laser beam velocity [m·s−1];
- t is the interaction time [s];
- t0 is the variable, and it represents the time required for heat to diffuse over a distance equal to the beam radius on the sample surface, and it can be calculated by using the equation:
- α is the base material thermal diffusivity [m2·s−1];
- z0 is a variable, and it measures the distance over which heat can diffuse during the laser beam interaction time.
If the interaction time t is much longer than t0 time (like usually is during the treatment), the value of z0 can be calculated using the following equation:
Consequently, the maximum temperature can be evaluated by the following equation:
where
- ρ is the base material density [kg·m−3];
- Cp is the base material specific heat [J·kg−1·K−1].
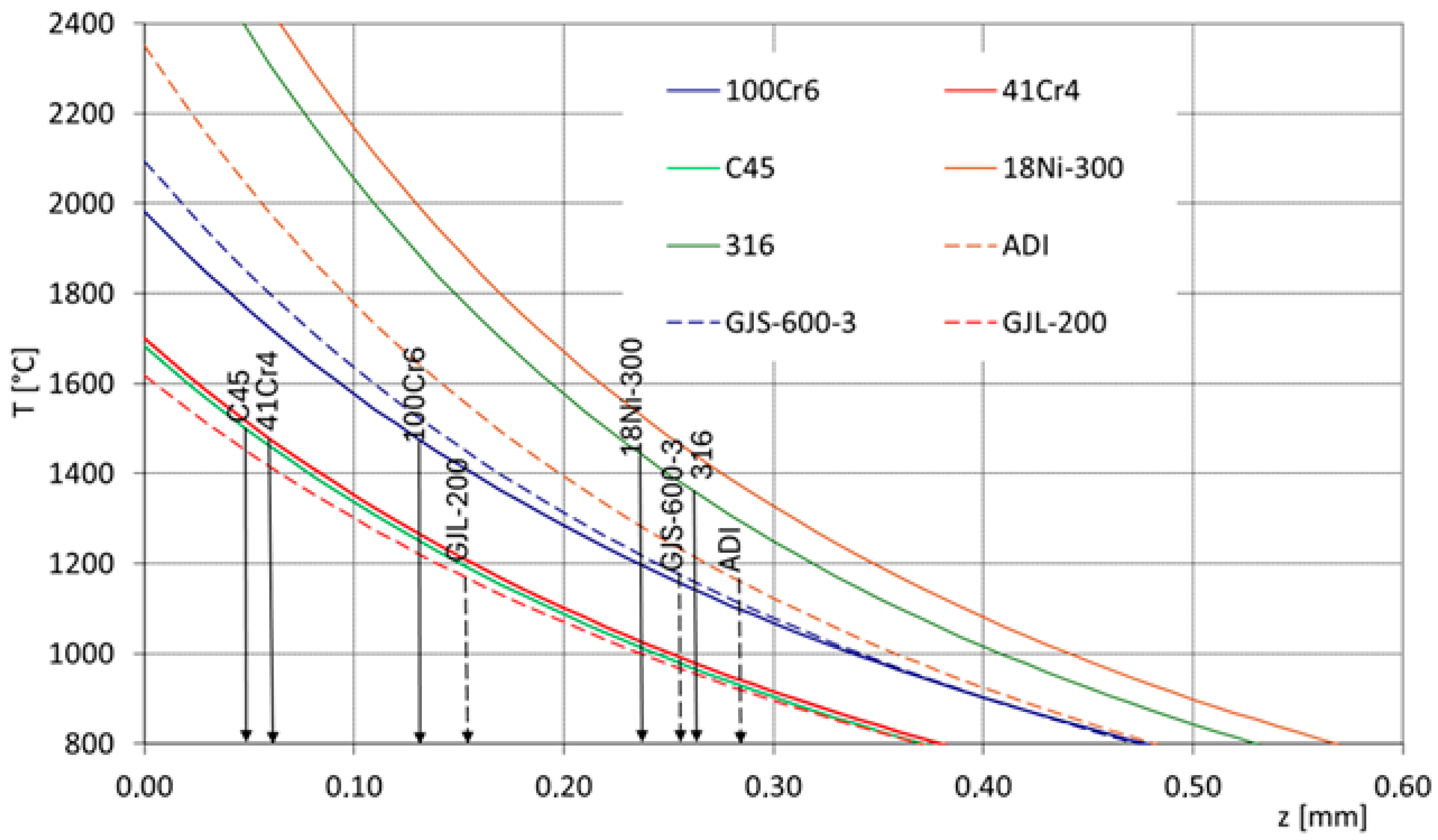
Material specific heat, thermal conductivity, density, and thermal diffusivity influence on temperature achieved in the surface layer as well as its cooling rate. In Figure 2, the counted changes in the temperature from the surface to the core during laser heating are presented (all of them were counted for the same laser treatment conditions: laser beam power 700 W, laser beam radius 1.25 mm, and laser beam interaction time 0.1 s). The lines represent the changes in the selected iron alloys and represent various thermophysical properties: C45 carbon steel, 41Cr4 and 100Cr6 alloyed steels, 316 austenitic steel, 18Ni-300 maraging steel, GJL-200 flake cast iron, GJS-600-3 nodular cast iron, and ADI austempered cast iron. Considering this set of iron alloys, high-carbon bearing steel is characterized by the lowest specific heat (390 J/kg × K), and the highest was found for ADI cast iron (550 J/kg × K). The highest thermal conductivity is represented by flake cast iron (54 W/m × K), and the lowest is represented by maraging steel (14–16 W/m × K). The highest densities are observed in nickel steels—maraging steel and austenitic steel (8 g/cm3)—while the lowest is found in flake cast iron (7.1 g/cm3). It can be noted that in all these cases, a varied temperature distribution can be expected during laser alloying. The highest surface layer temperatures can be achieved in austenitic and maraging steels, whereas the lowest are observed in flake cast iron, as well as in C45 carbon steel and 41Cr4 low-alloy steel. These differences result in varying depths of the alloyed zones. Considering the approximate melting temperature ranges (around 1500 °C for C45, 41Cr4, and 100Cr6 steels, around 1400 °C for 316 and 18Ni-300 steels, and around 1170 °C for cast irons), it can be observed that a thinner alloyed zone will form in C45 carbon steel and 41Cr4 low-alloy steel, whereas the largest zone will occur in nodular cast iron, ADI, and 316 and 18Ni-300 steels. The difference may even be more than five-fold. Therefore, to properly plan the depth of the alloyed zone, it is also necessary to consider the thermophysical properties of the base material. This is also important when selecting the appropriate amount of alloying element to achieve the desired concentration in the surface layer. Additionally, this is significant when creating the so-called raster in the surface layer and calculating the appropriate distances between laser-scanned traces. Another critical consideration is cooling rates. It can be expected that if there are varying temperature values in the surface layer, the cooling rate values will also differ.
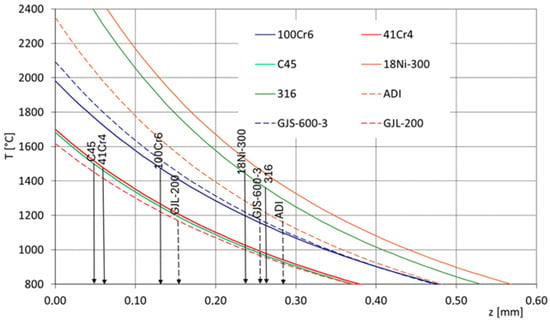
Figure 2.
Temperature distributions in the surface layer during laser alloying of various ferrous alloys.
Similarly to the case of temperature, warming rate and cooling rate can also be calculated by the following equation:
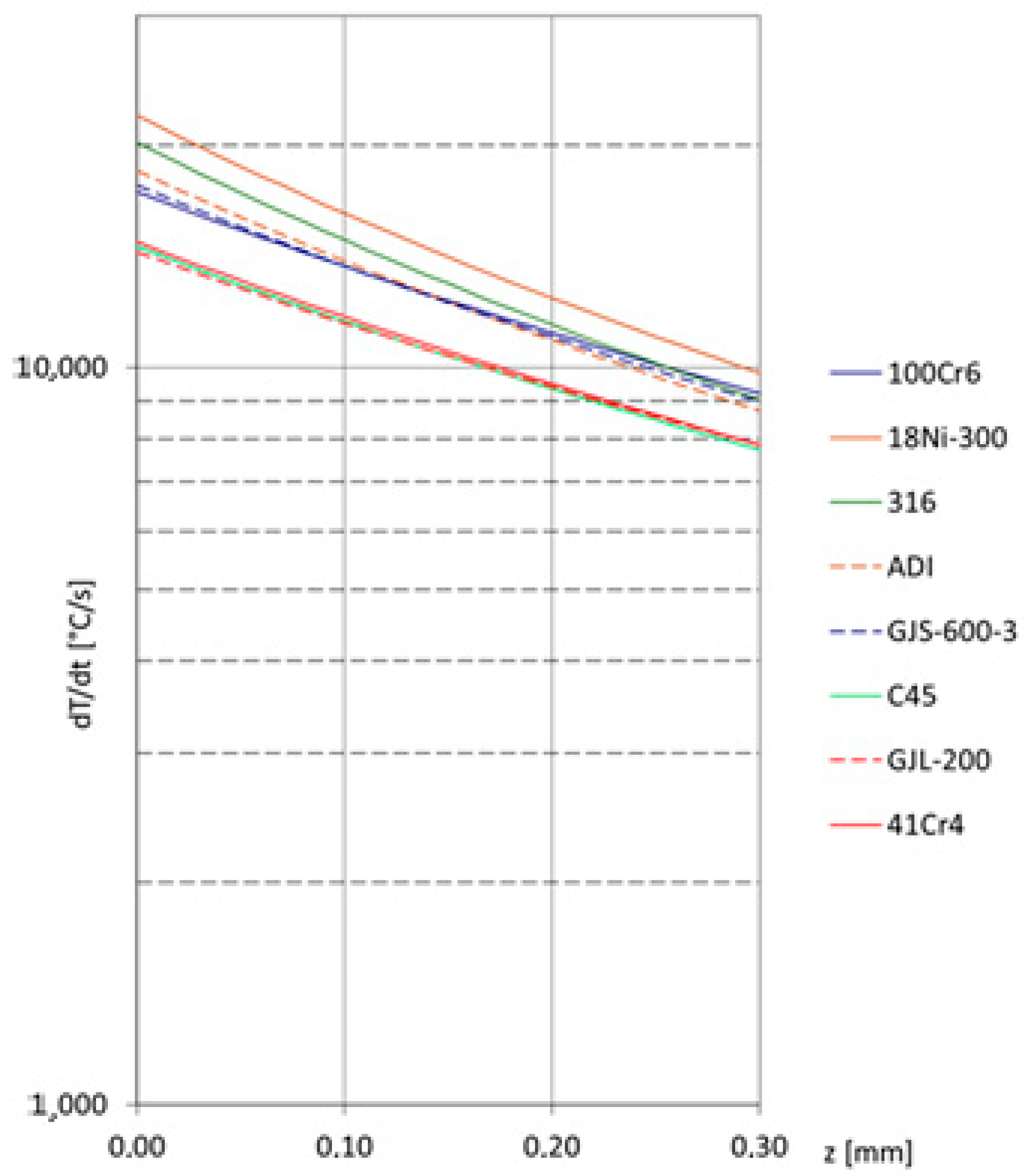
Heating is presented in the first part within square brackets, and cooling in the second part. The cooling rate distributions in the surface layer during laser alloying of various iron alloys are shown in Figure 3. The same laser treatment was applied as in the case of counting the temperature distributions presented in Figure 2.
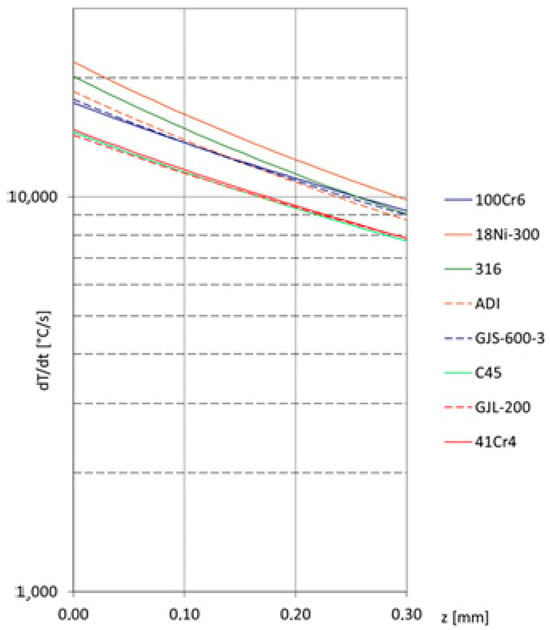
Figure 3.
Cooling rate distributions in the surface layer during laser alloying of selected ferrous alloys.
It can be observed that for C45, 41Cr4, and flake cast iron, the cooling rate will be the lowest. Conversely, the fastest cooling can be expected for high-alloy steels. Since the cooling rate is a key factor in microstructure formation (particularly when metastable phases, highly supersaturated solid solutions, or even amorphous structures are expected during laser alloying), it should also be considered.
2.3. Hardness of the Surface Layer
2.3.1. Steel
Implementation of boron by laser alloying onto the surface layer of steel can result in a hardness of the alloyed zone from 1100 even up to 1600 HV, as presented in [31], and it varies with laser treatment conditions. Considering that the steel being processed was hardened high-carbon steel, the increase was two-fold. However, the hardened part of the heat-affected zone below the alloy zone was strengthened to an average hardness of no more than 1000 HV.
In turn, in the case of C45 construction carbon steel, the hardness of the alloyed zone was achieved at 1200–1300 HV0.1. In the heat-affected zone, the hardness was approximately 600 HV0.1 and decreased to 320 HV0.1 [48]. In the case of 41Cr4 steel, the hardness was raised to 1100–1600 HV [19]. Laser alloying with boron was also performed in the case of 18Ni-300 maraging steel. Hardness even increased to 1770 HV in submicron-sized dendritic boride structure [50].
2.3.2. Grey Cast Iron
The surface layer after laser alloying boron was characterized by a hardness increase over 1000 HV as a result of fine grains, the presence of iron borides, and hardened eutectics. It was found that hardness is correlated with treatment conditions determined by laser beam parameters, like the cooling rate of the alloyed zone. Increasing the cooling rate from 9 × 02 °C/s to 3·103 °C/s causes a hardness increase from 1100 HV0.1 to 1280 HV0.1 [29]. The increase in hardness of the alloy zone can be up to 7 times. Hardness of some areas of the zone can reach 1800 HV0.1.
Additionally, it was also proven that the higher the boron concentration, the harder the expected material. Linear correlation between boron content and the HV0.1 hardness of alloyed zone was noticed. If amount of boron was 7% at, the hardness was 1200 HV0.1, and if 17% at., the hardness was 1600 HV0.1. Also, a stable distribution of hardness in the zone was noticed [49].
2.4. Tribological Properties of the Surface Layer
2.4.1. Steel
Research regarding to the laser alloying with boron of various type of steels proved the increase in wear resistance after this testament. It was found, for example, for 100CrMnSi6-4 bearing steel due to hard phases and fine microstructure [45]. Additionally, it was found that C45 construction carbon steel after laser alloying with boron was characterized by better wear resistance than after diffusion boronizing. The wear intensity index was 1.36 mg/(cm2h) of steel after laser treatment and 1.60 mg/(cm2h) of steel after diffusion treatment [48]. Frictional wear resistance tests were conducted on an Amsler MBT-01 friction tester. The tested steel was as a rotating ring, and the counter-sample was a cemented carbide plate. The tests were conducted under dry friction conditions. The load of F = 147 N was constant. The rotational speed was v = 0.26 m/s.
2.4.2. Grey Cast Iron
The laser alloying of samples with boron made of nodular cast iron caused an 18-fold reduction in their mass loss in comparison to the mass loss of untreated samples [30] after a wear test lasting only 15 min. Wear tests were conducted on an Amsler A135 machine (Kompetenzzentrum Tribologie Mannheim, Mannheim, Germany). The cast iron sample was cuboid-shaped, and the counter-sample was a hardened C45 steel ring. User tests were conducted on each of the samples at a limited speed of 200 rpm for 15 min, with increasing load using paraffin oil lubrication. The distance travelled was 396 m. It is worth noting that also counter samples made of steel cooperating with laser-treated nodular cast iron were characterized by 3-times lower mass loss than counter samples cooperating with untreated nodular cast iron [51]. Also, laser alloying with boron of the journal of crankshaft caused an increase in the resistance to wear [47]. The untreated journals had an average profile depression at the wear point that was 2.5 times greater than that of the laser alloyed journals. It was also found that the modified surface layer did not crack or detach during the test. The wear defects in the surface layer of the untreated journals were deeper than those after laser alloying with boron. Better resistance to wear of laser alloying with boron was also stated in research [48].
3. The Comparison of the Influence of Laser Alloying with Boron on Selected Ferrous Alloys
In Table 2, a comparison of selected steel and grey cast iron alloys is presented in the aspect of laser alloying with boron.

Table 2.
Comparison of the similarities and differences between laser alloying with boron of steel and grey cast iron.
4. Conclusions
This study characterized boron as an element that can be added to a metal alloy to modify its microstructure and properties. In particular, a micro-addition of this element significantly improves hardness and strength. This element is also used as just an additive to the surface layer of metallic components. In this case, it primarily forms hard borides, which increase hardness as well as resistance to wear, corrosion, and temperature. A popular method for introducing this element into the surface layer is the diffusion method. This method allows for the modification of multiple components simultaneously, which is an important advantage when numerous parts and their entire surfaces need processing. However, this requires a lot of time, in addition resulting in the formation of needle-like borides (and very often brittle FeB boride). Such a structure is prone to cracking. An alternative to this treatment is laser alloying.
It can be concluded that this treatment allows for the achievement of a fine, hardened microstructure with fine borides and an appropriate hardness distribution from the surface to the core of the material. This method is faster, enables the processing of only the part of the component that, for example, is most exposed to wear, and does not require a carrier medium with the alloying element, as is necessary in the diffusion process. Nevertheless, the surface layer after laser alloying with only a single element, such as boron, may vary depending on the laser processing conditions, as well as on the material of the component being treated, particularly its chemical composition and thermophysical properties, such as density, thermal conductivity, and specific heat. Consequently, the effects of laser alloying with boron (as well as alloying with other elements) may differ, even if the treatment is performed only in the range of iron alloys.
The main differences that can be anticipated are as follows:
- -
- the variety of newly formed phases containing boron, depending on the chemical composition of the iron alloy (if the base material contains alloying elements, as in the case of maraging or austenitic steel, not only iron borides but also borides with other metals may form);
- -
- the presence of graphite in the surface layer in the case of grey cast iron treatment (particularly nodular graphite cast iron), which is crucial in terms of wear due to friction;
- -
- a clearly visible transition zone (with melted and not-melted grains) between the alloyed zone and the hardened zone during the treatment of grey cast iron;
- -
- a tempered area beneath the hardened zone from the solid state, if the iron alloy was previously hardened;
- -
- variable depths of the modified surface layer depending on the thermophysical properties of the material being treated and the range of its solidification temperatures;
- -
- varied grain size in the alloy zone depending on the thermophysical properties of the material being processed, which affect the cooling rate (ultrafine microstructure in the case of high cooling rates).
The ability to modify the surface layer only in the most wear-prone parts of components is one of the greatest advantages of laser alloying, as it allows for significant savings in expensive alloying elements. This justifies research into the treatment of components made of different materials. The effects of laser beam exposure can be significantly different for different materials, even as described, within alloys of the same metal. It is therefore worthwhile to understand the processes occurring in the surface layers of various alloys (characterized by very different thermophysical property values) under the influence of laser heating. To quantitatively assess resistance to wear, corrosion, friction coefficient, etc., it is appropriate to conduct comparative studies on the same devices after the laser treatment of selected steels, cast irons, and other metal alloys. As was stated, laser boron alloying results in a significant increase in the hardness of alloys (steels and cast irons), partly due to the formation of iron borides. Another goal of further research could be to evaluate the effectiveness of laser boron alloying of various non-ferrous metal alloys in terms of creating the best phases (in terms of wear and corrosion resistance) formed as a result of the reaction of the substrate elements with boron.
Funding
This research was funded by Poznan University of Technology grant number 0414/SBAD/2025.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The author declares no conflict of interest.
References
- Dobrzański, L.A. Metaloznawstwo z Podstawami Nauki o Materiałach; Wydawnictwo Naukowo-Techniczne: Warszawa, Poland, 1998; ISBN 83-204-2255-8. [Google Scholar]
- Lis, J.; Lis, A.; Kolan, C. Investigation of boron segregation in low carbon steel. Metalurgija 2011, 50, 11–14. [Google Scholar]
- Peng, H.; Chen, W.; Chen, H. Effects of boron on continuous cooling transformation and hardenability of 20CrMnTi steel containing tin. Jinshu Rechuli/Heat Treat. Met. 2015, 40, 99–104. [Google Scholar] [CrossRef]
- Ozmen, L.; Cakici, M.; Yucel, O. Effect of boron addition to steel on mechanical properties. In Proceedings of the TMS 2025 154th Annual Meeting & Exhibition, Taoyuan City, Taiwan, 14–15 January 2025; Minerals, Metals and Materials Series. Springer: Cham, Switzerland, 2025; pp. 215–222. [Google Scholar] [CrossRef]
- Yoshimoto, K.; Pasang, T.; Aihara, S.; Kawabata, T. Experiment and numerical modeling of boron effect on weld HAZ hardenability of steels. IOP Conf. Ser. Mater. Sci. Eng. 2020, 758, 012004. [Google Scholar] [CrossRef]
- Kumar, S.; Desai, R. Effect of boron micro-alloying on microstructure and corrosion behavior of dual-phase steel. J. Mater. Eng. Perform. 2019, 28, 2691–2699. [Google Scholar] [CrossRef]
- Wi, D.Y.; Kim, K.S.; Jung, B.I.; Lee, K.A. Prediction of Jominy hardness curves using multiple regression, and effect of alloying elements on the hardenability. Korean J. Mater. Res. 2019, 29, 781–789. [Google Scholar] [CrossRef]
- Menapace, C.; Libardi, S.; D’Incau, M.; Molinari, A. Microstructure and tensile properties of nanostructured ultrafine-grained FeMoB alloys produced by spark plasma sintering of mechanically alloyed powders. J. Mater. Eng. Perform. 2010, 19, 54–60. [Google Scholar] [CrossRef]
- Shi, X.L.; Jiang, Y.H. Analysis of morphological, thermodynamic, and mechanical properties of typical hard phase Fe-5Cr-1.5B-0.45C alloy. J. Alloys Compd. 2020, 816, 152681. [Google Scholar] [CrossRef]
- Sudalaimunthu, G.; Sundaram, J.; Muthukrishnan, G. Characteristic study of boron steel during spheroidization and annealing in a bell-type furnace for heat treatment. AIP Conf. Proc. 2024, 3192, 020061. [Google Scholar] [CrossRef]
- Zhao, G.; Li, Y.; Yao, X.; Wang, Z. Effect of boron on solidification behavior, strength, toughness, and wear resistance of Fe-Cr-B-C alloy. Jinshu Xuebao/Acta Metall. Sin. 2025, 61, 699–716. [Google Scholar] [CrossRef]
- Ding, T.; Li, C.; Jing, W.; Di, X.; Hu, W. Influences of boron on the microstructural characteristics and wear performance of hypereutectic Fe-Cr-C-Mo-xB hardfacing alloy. Surf. Coat. Technol. 2024, 478, 130415. [Google Scholar] [CrossRef]
- Deniz, G.; Sen, S.; Sen, U. Properties of the surface alloyed AISI 1020 steel with boron by TIG welding technique. In Proceedings of the International Conference on Advances in Welding Science and Technology for Construction, Energy and Transportation (AWST), Istanbul, Turkey, 15–16 July 2010; pp. 145–152. [Google Scholar]
- Li, Q.; Zhang, Y.L.; Zhang, L.; Zhang, J.W. Effect of boron on the microstructural evolution and wear resistance of high hardness Fe-based alloy coatings prepared by laser cladding. Surf. Coat. Technol. 2023, 464, 129573. [Google Scholar] [CrossRef]
- Huang, Z.; Xing, J.; Ma, S.; Sun, L. Microstructure and Properties of Fe-B-C Cast Wear-Resistant Alloy. Key Eng. Mater. 2017, 732, 59–68. [Google Scholar] [CrossRef]
- Zhang, D.; Li, H.; Liu, Y.; Jiang, J.; Zhang, Y. Effect of B4C Content on Microstructure and Wear Resistance of Laser-Cladding-Enhanced 316 Stainless Steel Coatings. Coatings 2025, 15, 681. [Google Scholar] [CrossRef]
- Li, H.; Han, G.; Dai, J.; Xu, J. Study on the Preparation and Properties of Laser Boron–Nitrogen Alloying Layer on 45 Steel. J. Mater. Prot. 2022, 4, 98–103. [Google Scholar]
- Bartkowska, A.; Bartkowski, D.; Przestacki, D.; Kieruj, P. Laser processing of diffusion boronized layer produced on Monel® alloy 400–microstructure, microhardness, corrosion and wear resistance tests. Materials 2021, 14, 4831. [Google Scholar] [CrossRef]
- Kulka, M.; Makuch, N.; Pertek, A. Microstructure and properties of laser borided 41Cr4 steel. Opt. Lasers Eng. 2013, 51, 1245–1253. [Google Scholar] [CrossRef]
- Pertek-Owsiana, A. Kształtowanie Struktury i Właściwości Warstw Borków Żelaza Otrzymywanych w Procesie Borowania Gazowego; WPP Rozprawy; Publishing House of Poznan University of Technology: Poznań, Poland, 2001; p. 365. [Google Scholar]
- Paczkowska, M. Właściwości warstw powierzchniowych elementów stalowych borowanych laserowo i dyfuzyjnie. J. Res. Appl. Agric. Eng. 2004, 49, 43–48. [Google Scholar]
- Paczkowska, M. The influence of the laser and diffusion boronizing on the surface layer of nodular iron. J. Res. Appl. Agric. Eng. 2017, 62, 158–162. [Google Scholar]
- Yang, R.; Gui, X.; Yang, H.; Qiao, J. Tribological behavior of boronized Fe40Mn20Cr20Ni20 high-entropy alloys at high temperature. Surf. Coat. Technol. 2023, 467, 129395. [Google Scholar] [CrossRef]
- Ma, W.; Wan, Q.; Jia, B.; Li, S. Mechanical and tribological properties of in-situ prepared boride hard coatings on the surface of three high-entropy alloys. Ceram. Int. 2025, 51, 1756–1765. [Google Scholar] [CrossRef]
- Audronis, M.; Kelly, P.J.; Leyland, A.; Matthews, A. A New Approach to the Deposition of Elemental Boron and Boron-Based Coatings by Pulsed Magnetron Sputtering of Loosely Packed Boron Powder Targets. Plasma Process. Polym. 2007, 4, S160–S165. [Google Scholar] [CrossRef]
- Oks, E.; Anders, A.; Nikolaev, A.; Yushkov, Y. Sputtering of pure boron using a magnetron without a radio-frequency supply. Rev. Sci. Instrum. 2017, 88, 043506. [Google Scholar] [CrossRef]
- Migal, Y.F.; Sidashov, A.V. Boriding of steel surface with beams of boron atoms. MATEC Web Conf. 2018, 226, 03004. [Google Scholar] [CrossRef]
- Sreejith, G.; Sunny, T.; Sahu, J.; Sasikumar, C. Low Temperature Boronizing of Surface Nanostructured Ni-Cr-Mo Steel Using SMAT. Mater. Sci. Forum 2015, 830–831, 663–666. [Google Scholar] [CrossRef]
- Paczkowska, M.; Ratuszek, W.; Waligóra, W. Microstructure of laser boronized nodular iron. Surf. Coat. Technol. 2010, 205, 2542–2545. [Google Scholar] [CrossRef]
- Paczkowska, M.; Wojciechowski, Ł. Badania zużycia adhezyjnego elementów z żeliwa sferoidalnego borowanych laserowo. Tribologia 2007, 3–4, 71–83. [Google Scholar]
- Paczkowska, M.; Kinal, G.; Rewolińska, A.; Wojciechowski, Ł. Evaluation of the surface layer microstructure of high carbon alloy steel after laser modification. J. Res. Appl. Agric. Eng. 2016, 61, 61–65. [Google Scholar]
- Grum, J.; Šturm, R. Comparison of measured and calculated thickness of martensite and ledeburite shells around graphite nodules in the hardened layer of nodular iron after laser surface remelting. Appl. Surf. Sci. 2002, 187, 116–123. [Google Scholar] [CrossRef]
- Riahi, M. Surface treatment of cast iron by adding different alloying elements to form a metallic glass structure layer using an industrial carbon dioxide laser. J. Mech. Work. Technol. 1996, 58, 3–12. [Google Scholar] [CrossRef][Green Version]
- Li, R.; Yuan, W.; Yue, H.; Zhu, Y. Study on microstructure and properties of Fe-based amorphous composite coating by high-speed laser cladding. Opt. Laser Technol. 2022, 146, 107574. [Google Scholar] [CrossRef]
- Pauly, S.; Löber, L.; Petters, R.; Stoica, M.; Scudino, S.; Kühn, U.; Eckert, J. Processing metallic glasses by selective laser melting. Mater. Today 2013, 16, 37–41. [Google Scholar] [CrossRef]
- Żrodowski, Ł.; Wysocki, B.; Błyskun, P.; Wróblewski, R.; Święszkowski, W. Additive Manufacturing of Amorphous Alloys via Laser Melting. Mechanik 2017, 1, 50–55. [Google Scholar] [CrossRef][Green Version]
- Wang, Q.; Li, Q.; Zhang, J.W. Effect of Boron Gradient Design on the Microstructure and Properties of Laser-Clad Fe-Based Coatings. Mater. Lett. 2025, 398, 138932. [Google Scholar] [CrossRef]
- Chen, C.; Feng, A.; Liu, B.; Wei, Y.; Song, X. Effect of quench-tempering and laser quenching on the microstructure and properties of high-chromium cast iron. J. Mater. Res. Technol. 2022, 19, 2759–2773. [Google Scholar] [CrossRef]
- Feldshtein, E.; Devojno, O.; Patalas-Maliszewska, J.; Kardapolava, M.; Kasiakova, I. Effect of processing parameters on the formation of alloyed surface layer and its properties on the efficiency of laser alloying of grey cast iron with tungsten and silicon carbides. Materials 2023, 16, 6230. [Google Scholar] [CrossRef]
- Che, Z.; Zhou, T.; Zhang, P.; Zhang, H.; Yang, W.; Zhou, H.; Ren, L. Influences of single laser tracks’ space on the rolling fatigue contact of gray cast iron. Opt. Laser Technol. 2015, 72, 15–24. [Google Scholar] [CrossRef]
- Tong, X.; Zhou, H.; Zhang, W.; Zhang, Z.; Ren, L. Thermal fatigue behavior of gray cast iron with striated biomimetic non-smooth surface. J. Mater. Process. Technol. 2008, 206, 473–480. [Google Scholar] [CrossRef]
- Paczkowska, M. Influence of silicon and cobalt laser alloying on the microstructure and nanomechanical properties of the gray cast iron surface layer. J. Res. Appl. Agric. Eng. 2022, 67, 13–20. [Google Scholar] [CrossRef]
- Jia, Y.; Wu, H.; Zhang, Y.; Wang, H. Enhancement of Surface Hardness and Self-Lubrication of CoCrFeNiMn High Entropy Alloy through Laser Boriding. Mater. Charact. 2025, 224, 115064. [Google Scholar] [CrossRef]
- Lunarska, E.; Stabryła, J.; Nikiforow, K.; Starczewski, L. Boron alloying and laser treatment to improve corrosion and hydrogen resistance of 25G steel. Mater. Sci. 2005, 21, 123–129. [Google Scholar] [CrossRef]
- Piasecki, A.; Kotkowiak, M.; Kulka, M. The effect of CaF2 and BaF2 solid lubricants on wear resistance of laser borided 100CrMnSi6-4 bearing steel. Arch. Mater. Sci. Eng. 2017, 86, 15–23. [Google Scholar] [CrossRef]
- Marimoto, J.; Ozaki, T.; Katoh, Y.; Tsukamoto, M. Laser boronizing of carbon steel with direct diode laser. In Proceedings of the 26th International Congress on Laser Materials Processing, Laser Microprocessing and Nanomanufacturin, Orlando, FL, USA, 29 October–1 November 2007; pp. 25–30. [Google Scholar]
- Paczkowska, M. Kształtowanie Odporności na Zużycie Tribologiczne Elementów Maszyn z Żeliwa Przez Laserową Obróbkę Cieplną (LOC); Wydawnictwo PP: Poznań, Poland, 2016. [Google Scholar]
- Kinal, G.; Bartkowska, A. Możliwości kształtowania odporności na procesy zużywania laserowo borowanych warstw powierzchniowych elementów stalowych oraz żeliwnych. Tribologia 2014, 3, 97–107. [Google Scholar]
- Paczkowska, M. The Possibility of Selected Surface Layer Modification of Nodular Iron Engine Parts by Laser Boronizing. Veh. Eng. 2014, 1, 64–69. [Google Scholar]
- Skamat, J.; Bucelis, K.; Cernasjus, O. Laser boronizing of additively manufactured 18Ni-300 Maraging Steel part surface. Materials 2022, 15, 1345. [Google Scholar] [CrossRef]
- Paczkowska, M.; Wojciechowski, Ł. Analiza zużycia stalowych pierścieni współpracujących z żeliwem sferoidalnym poddanym laserowej obróbce cieplnej. Tribologia 2015, 5, 99–107. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).