Tribocatalysis Induced Carbon-Based Tribofilms—An Emerging Tribological Approach for Sustainable Lubrications
Abstract
1. Introduction
2. Carbon-Based Thin Films and Their Lubrication Performance
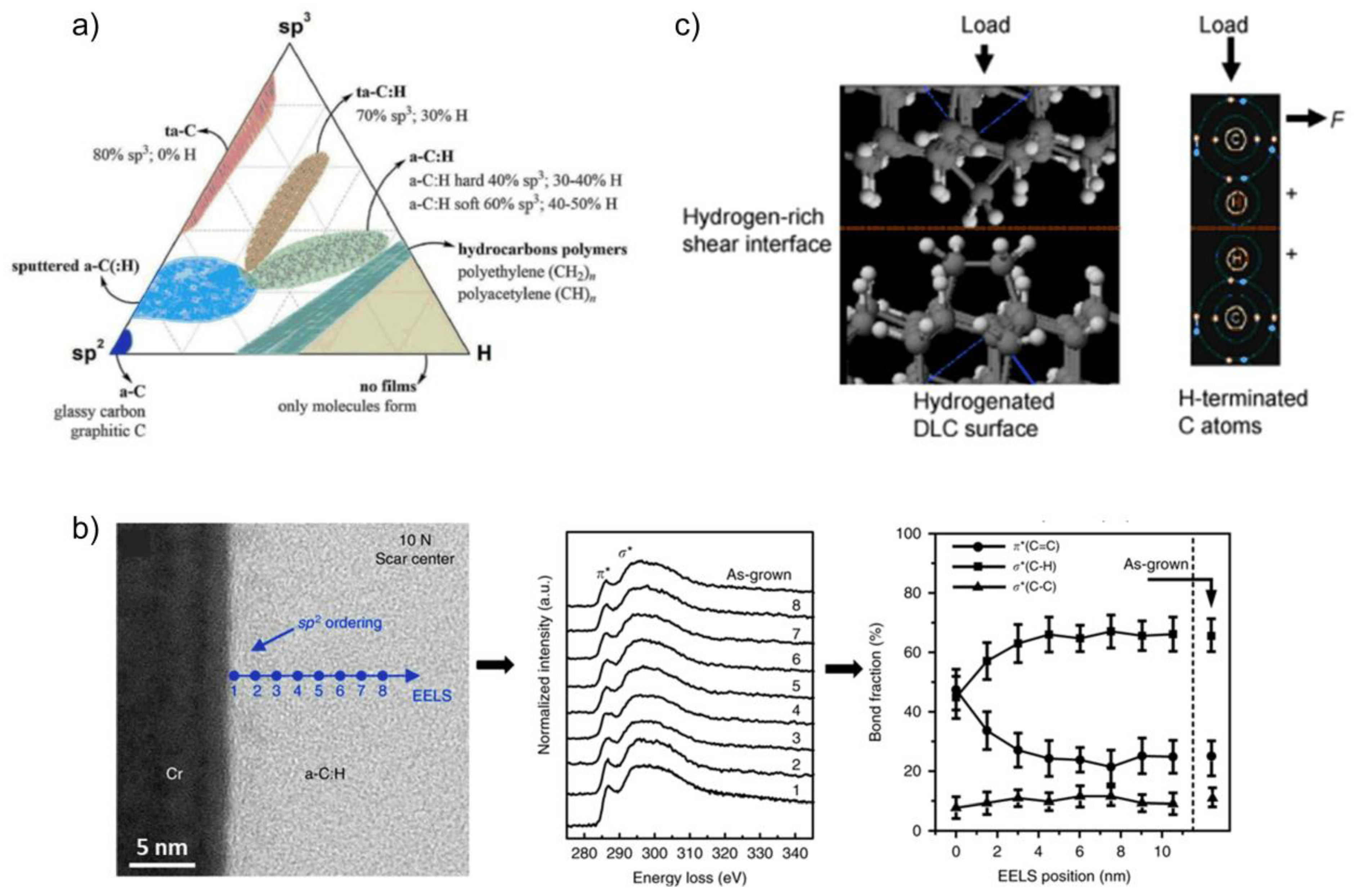
2.1. Diamond-Like Carbon Films
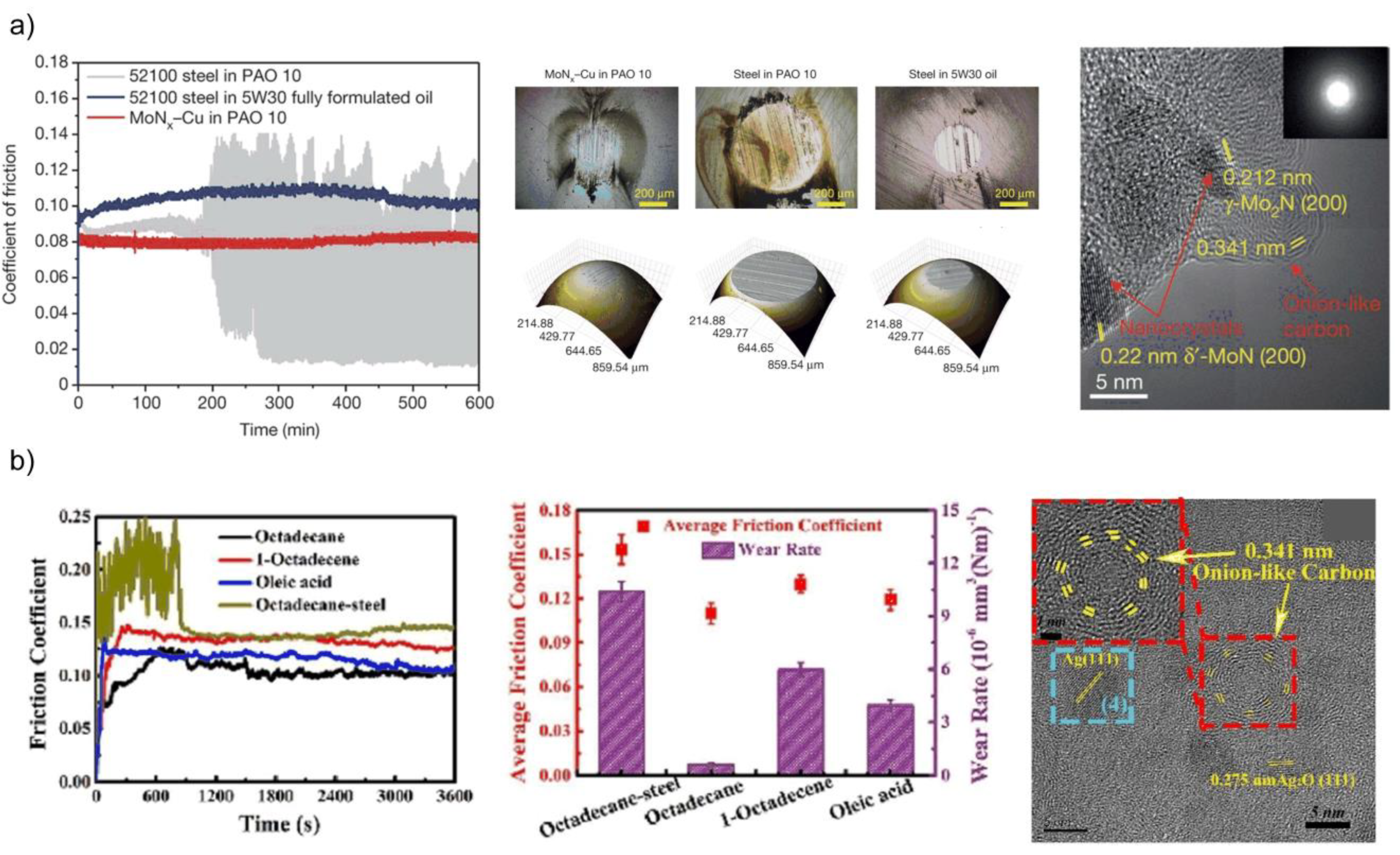
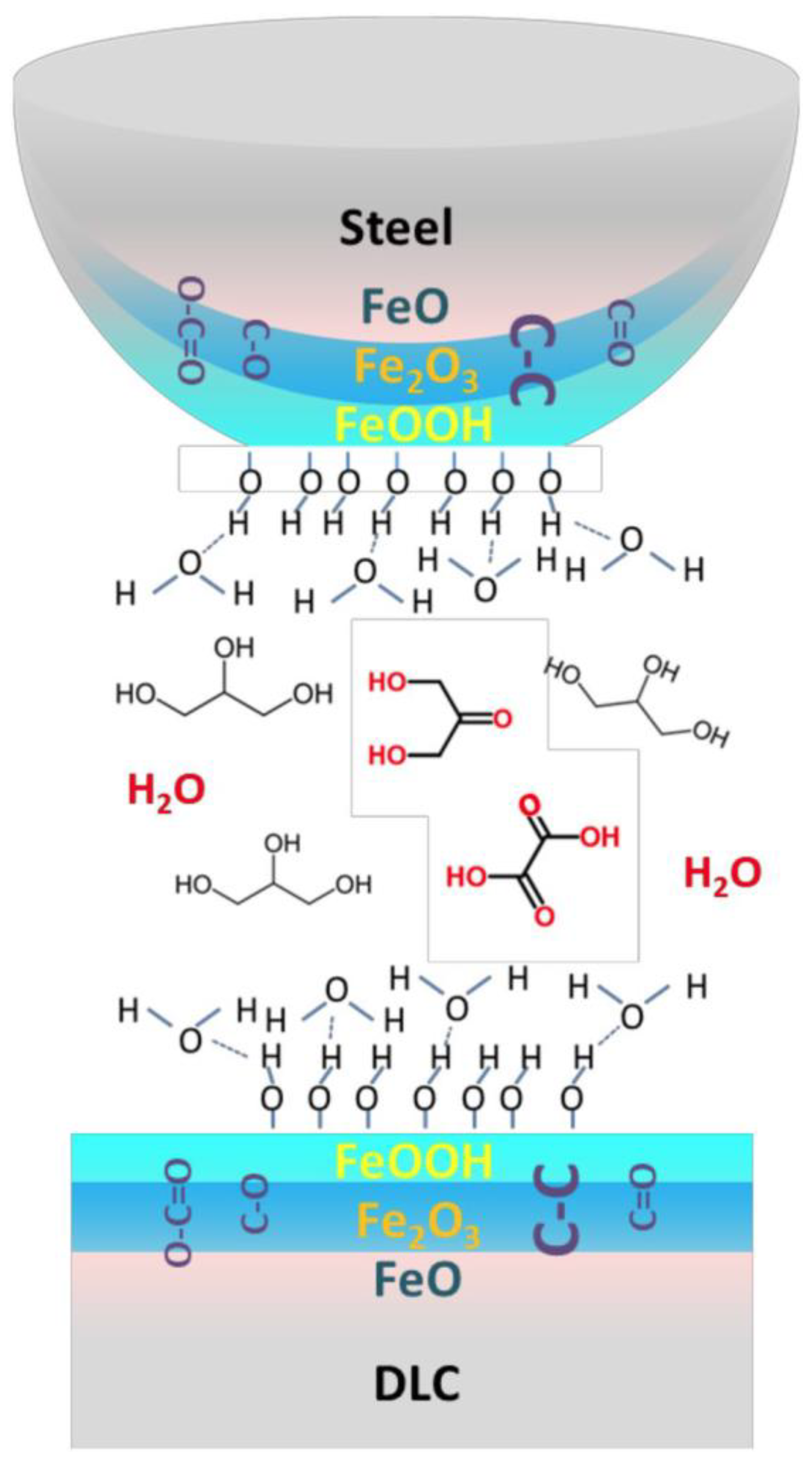
2.2. Carbon-Based Tribofilms Produced by Catalytic Coatings
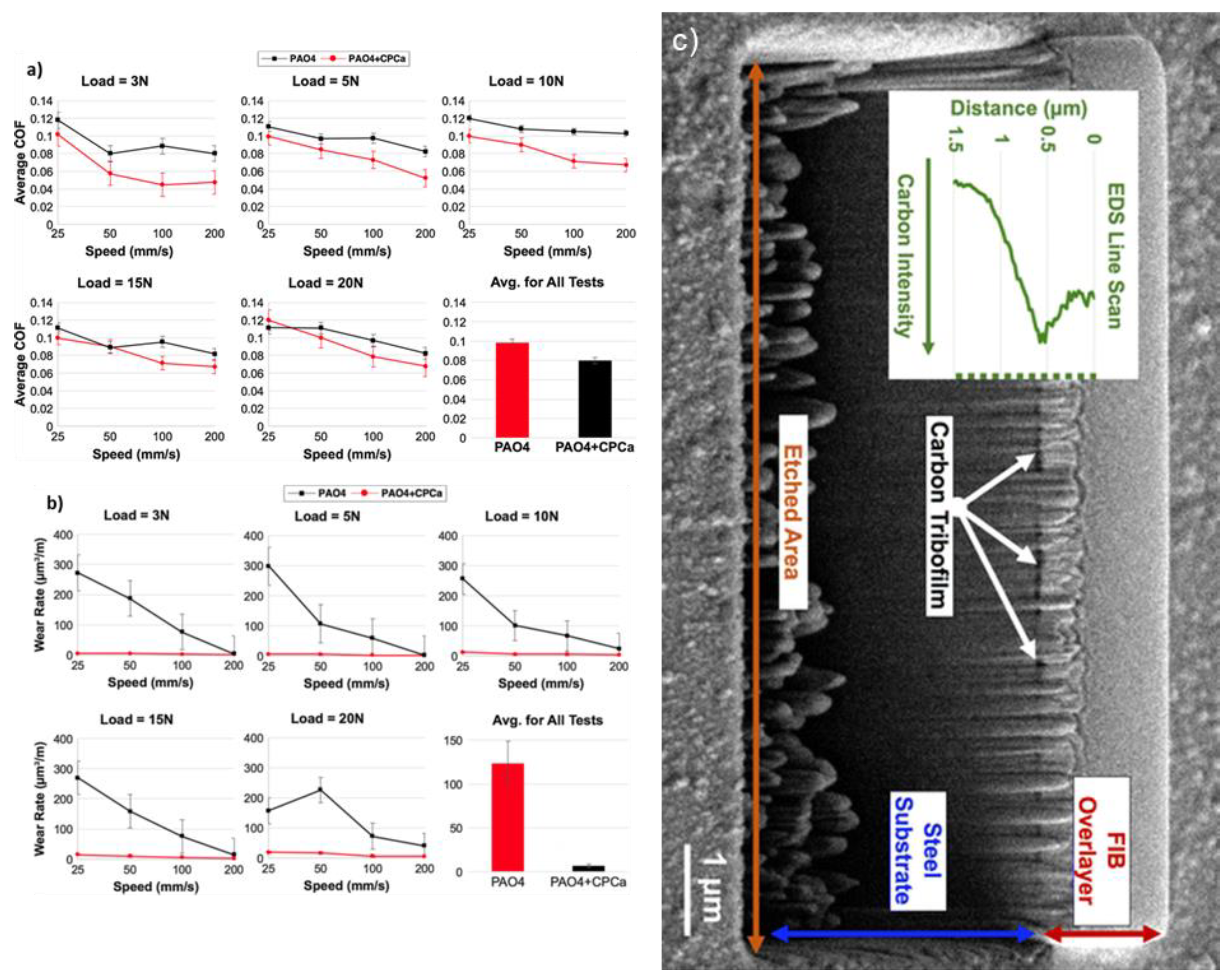
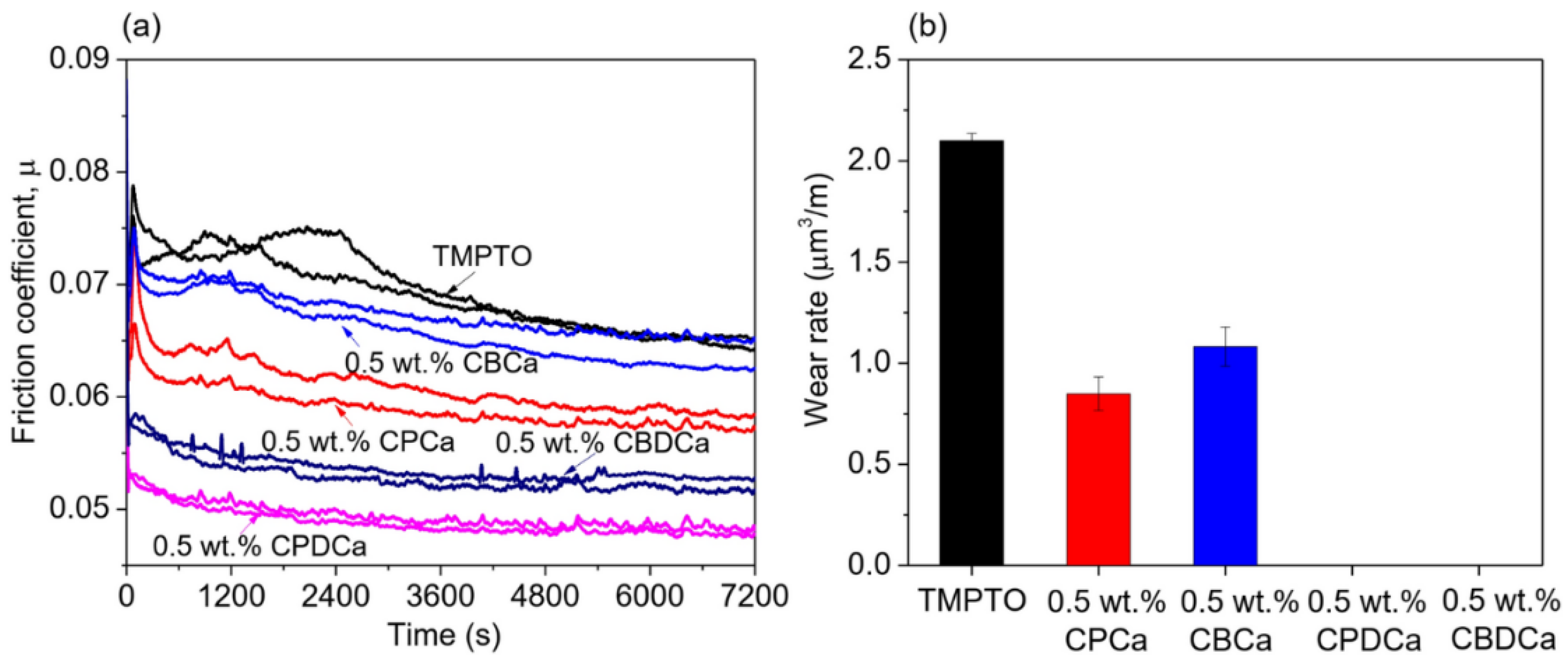
2.3. Carbon-Based Tribofilms Produced by Organic Additives
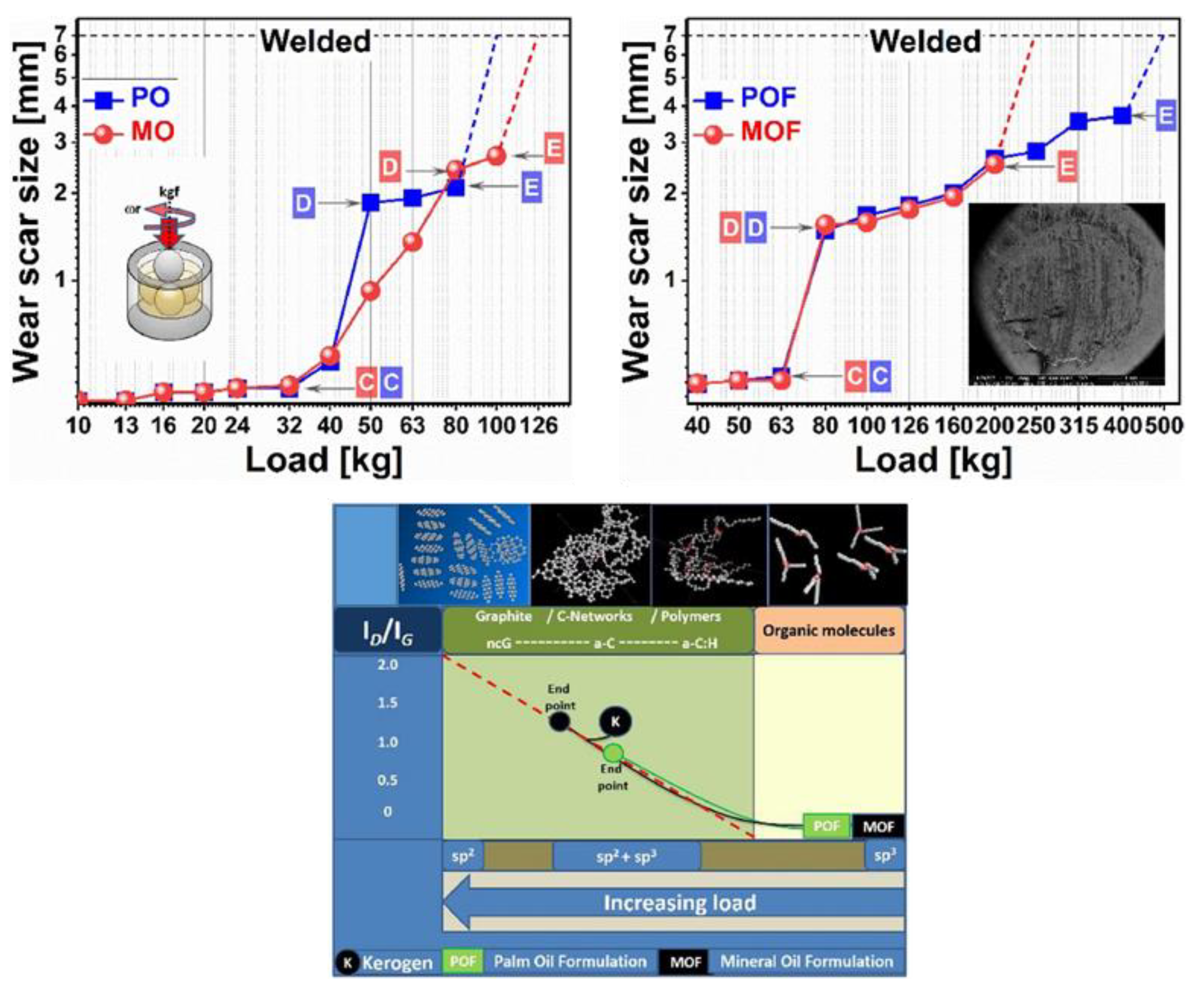
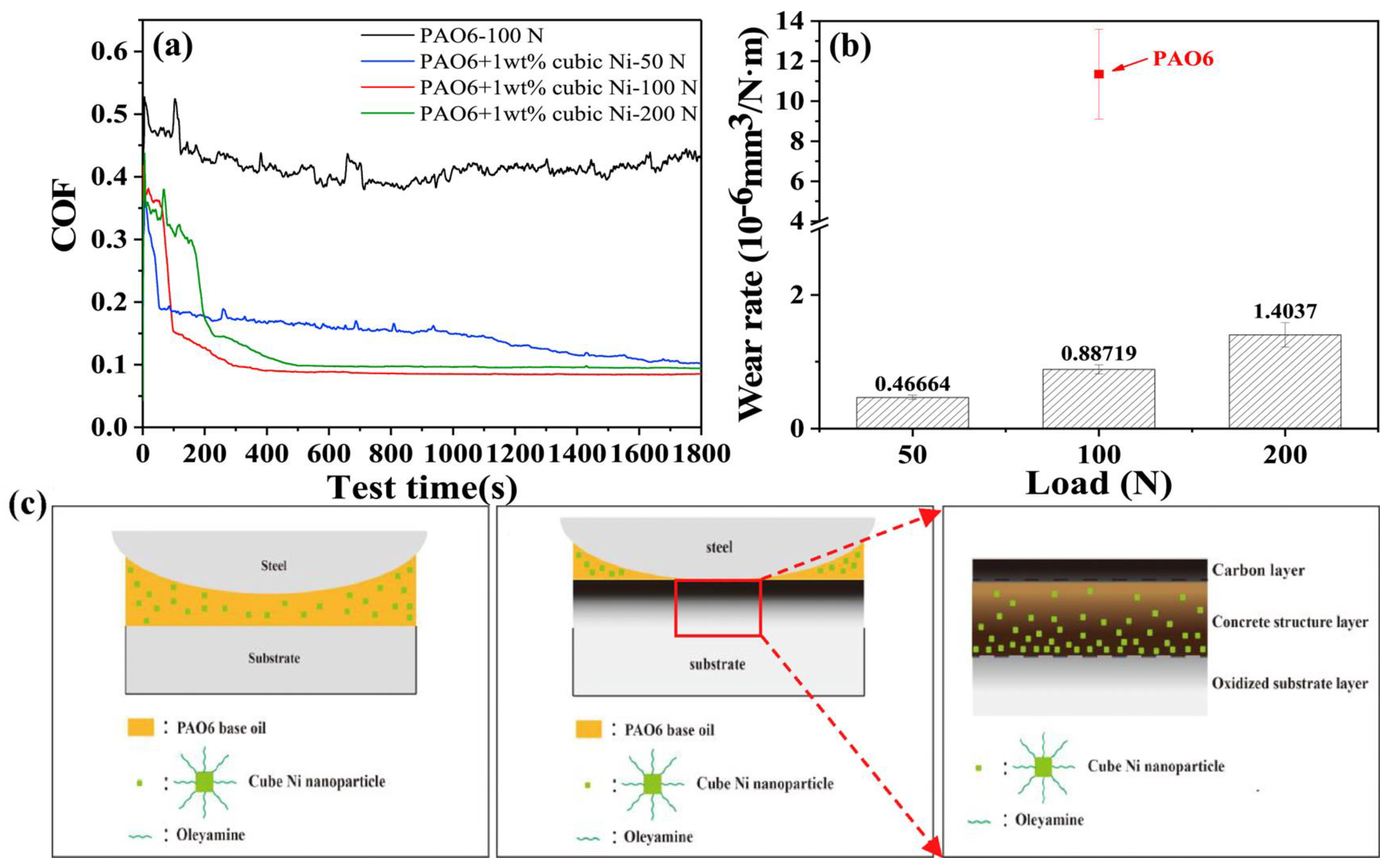
2.4. Carbon-Based Tribofilms Produced by Catalyst Additives
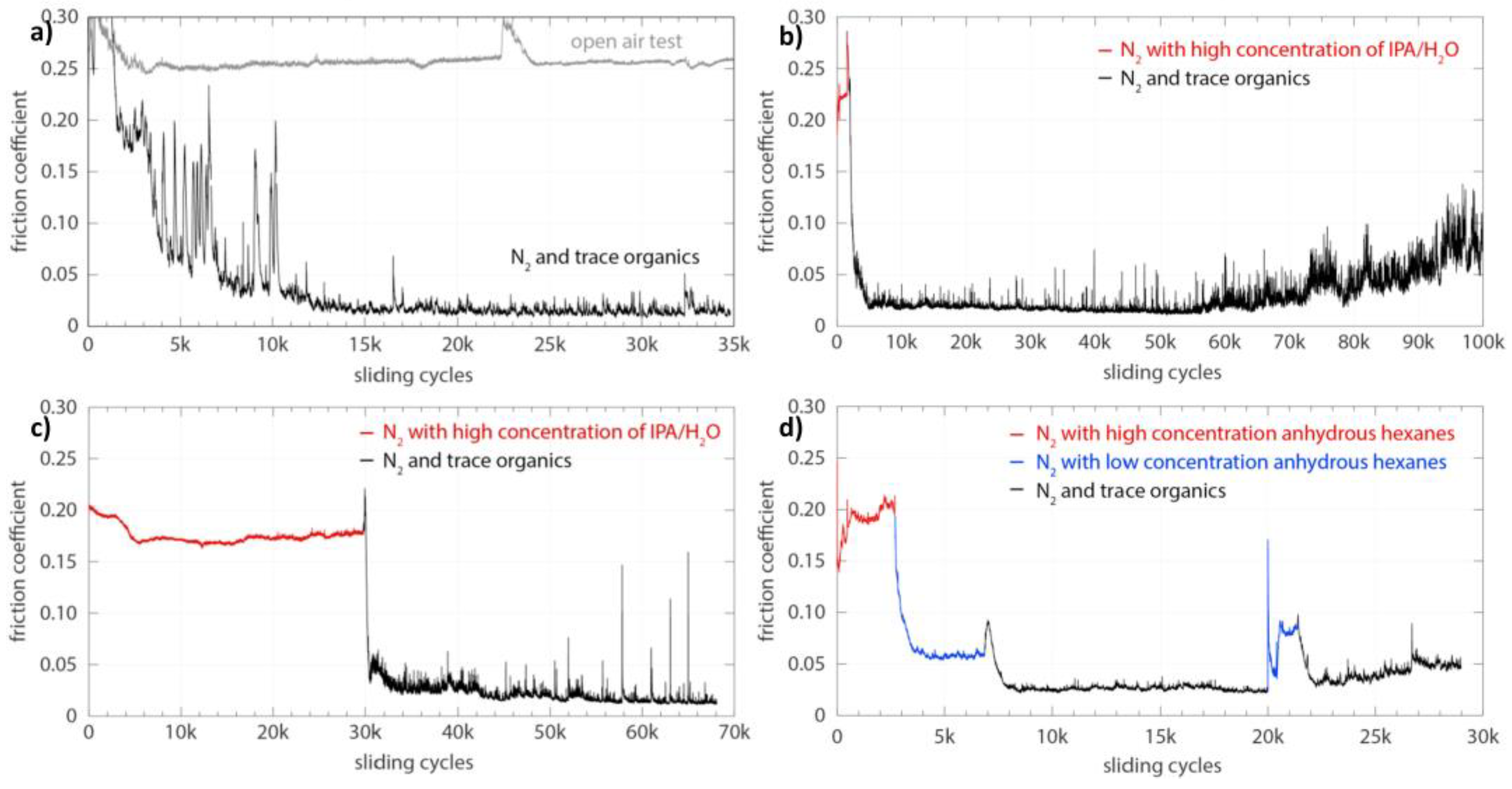
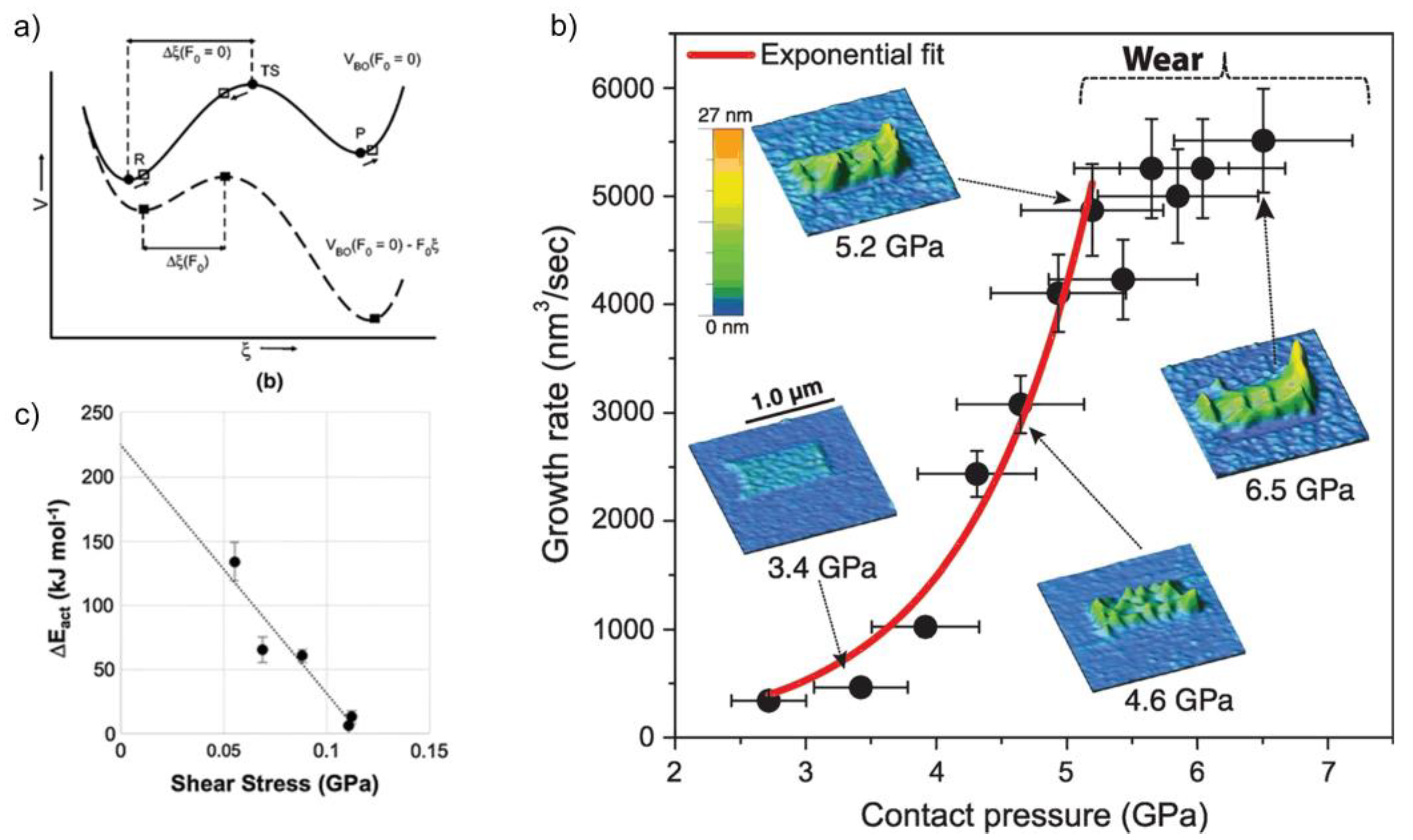
3. Tribochemistry and Mechanistic of the Formation of Carbon-Based Tribofilms
4. Characterizations of Carbon-Based Tribofilms by Analytical Techniques
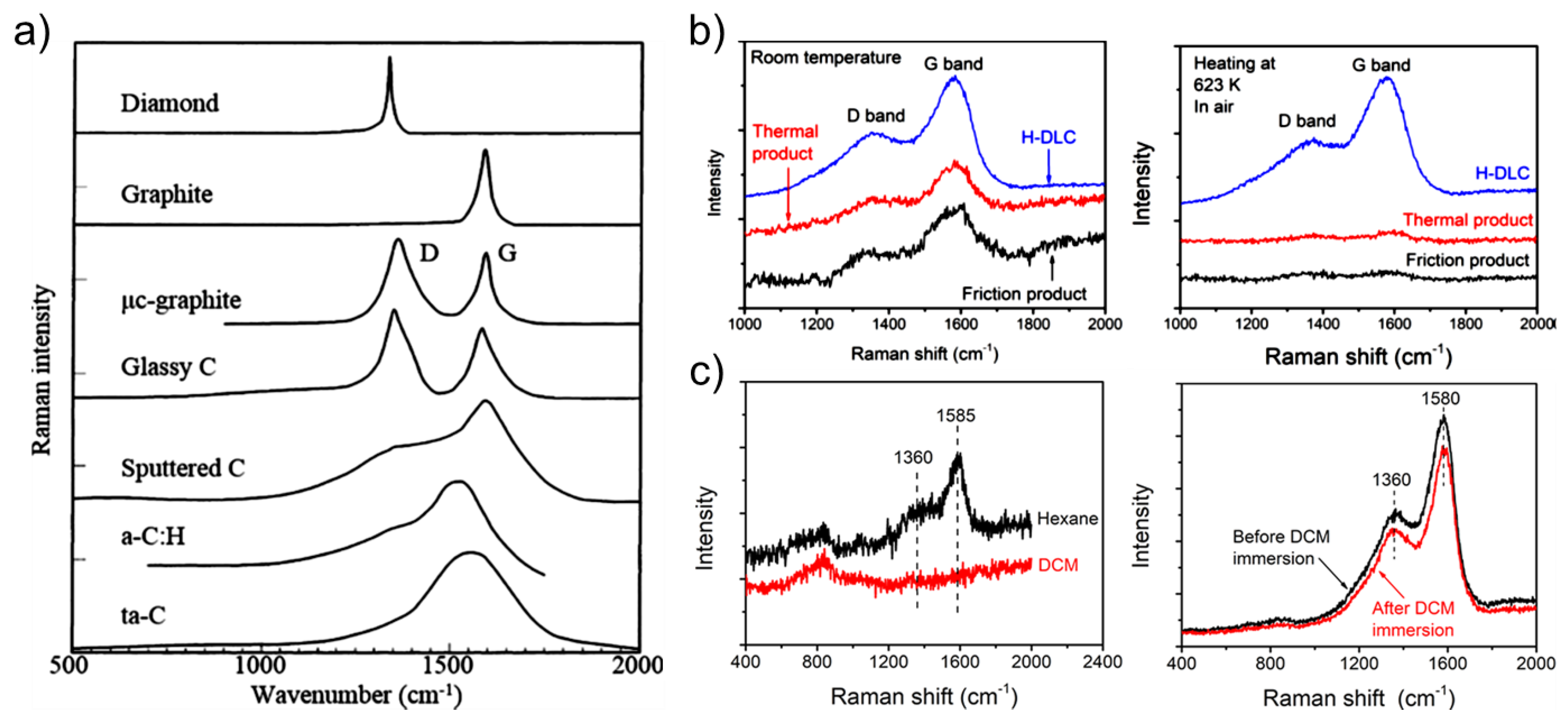
4.1. Raman Spectroscopy
4.2. X-ray Photoelectron Spectroscopy (XPS)
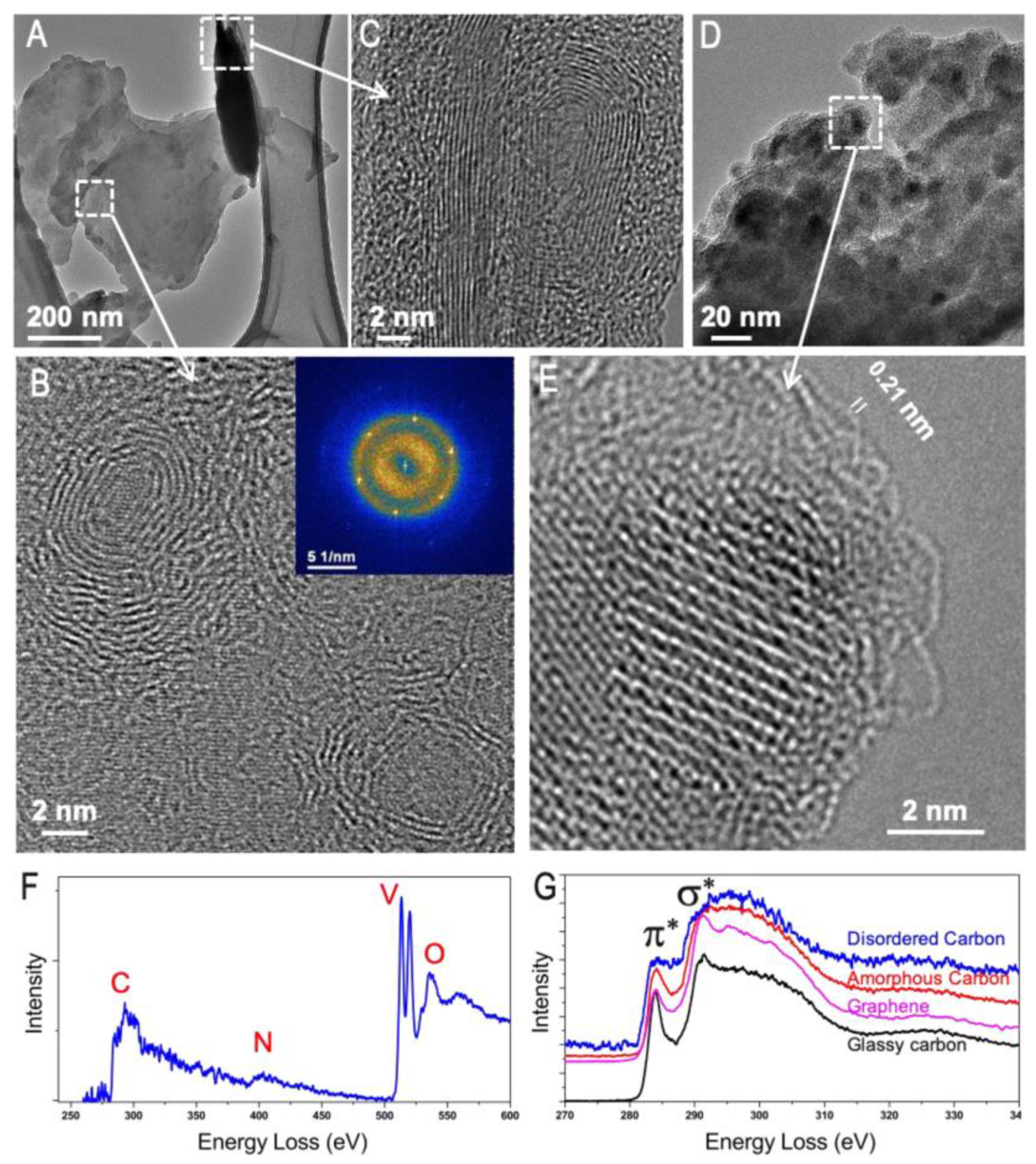
4.3. Scanning/Transmission Electron Microscope (S/TEM) and Electron Energy Loss Spectroscopy (EELS) Combination
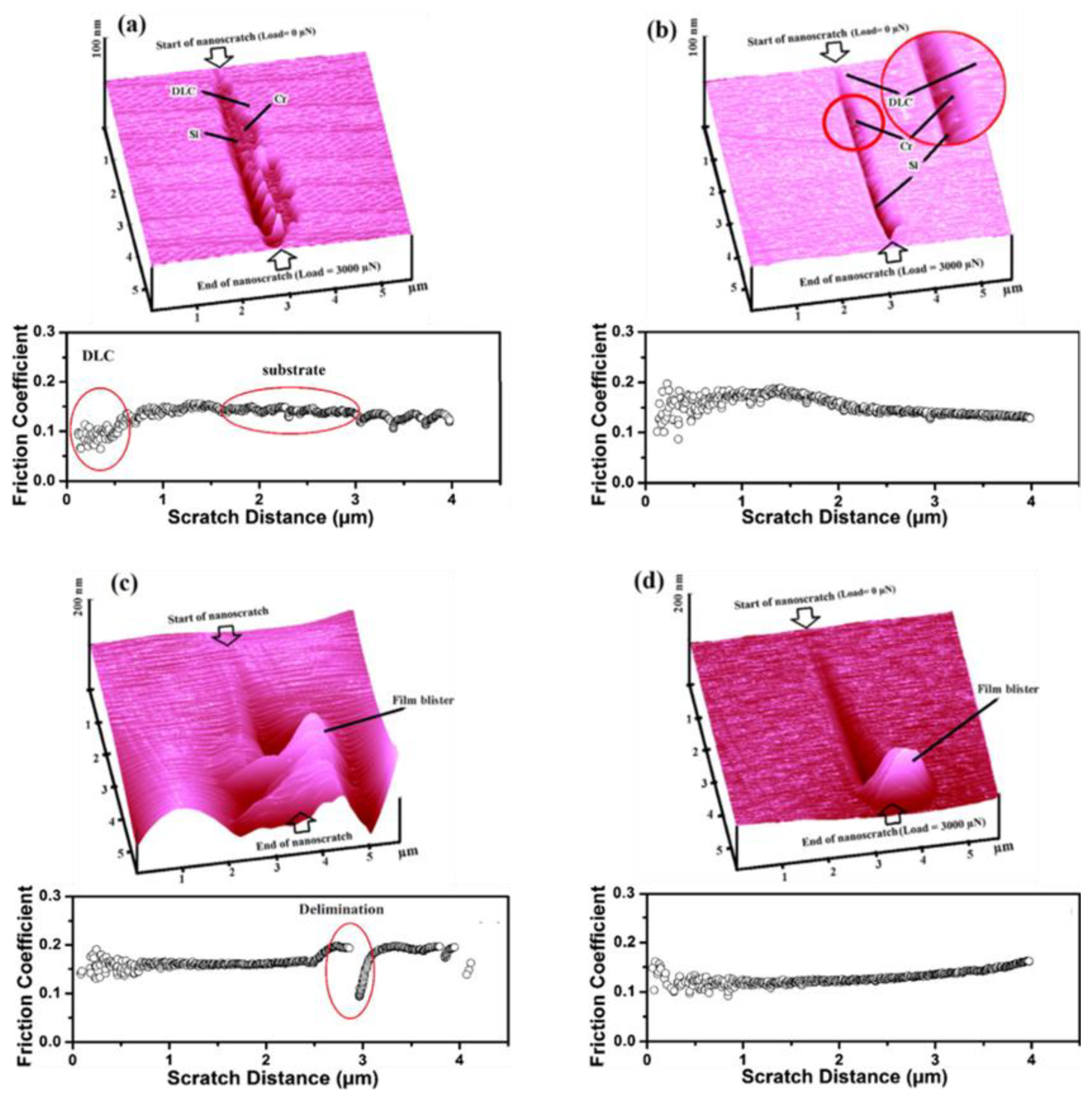
4.4. Mechanical Analysis
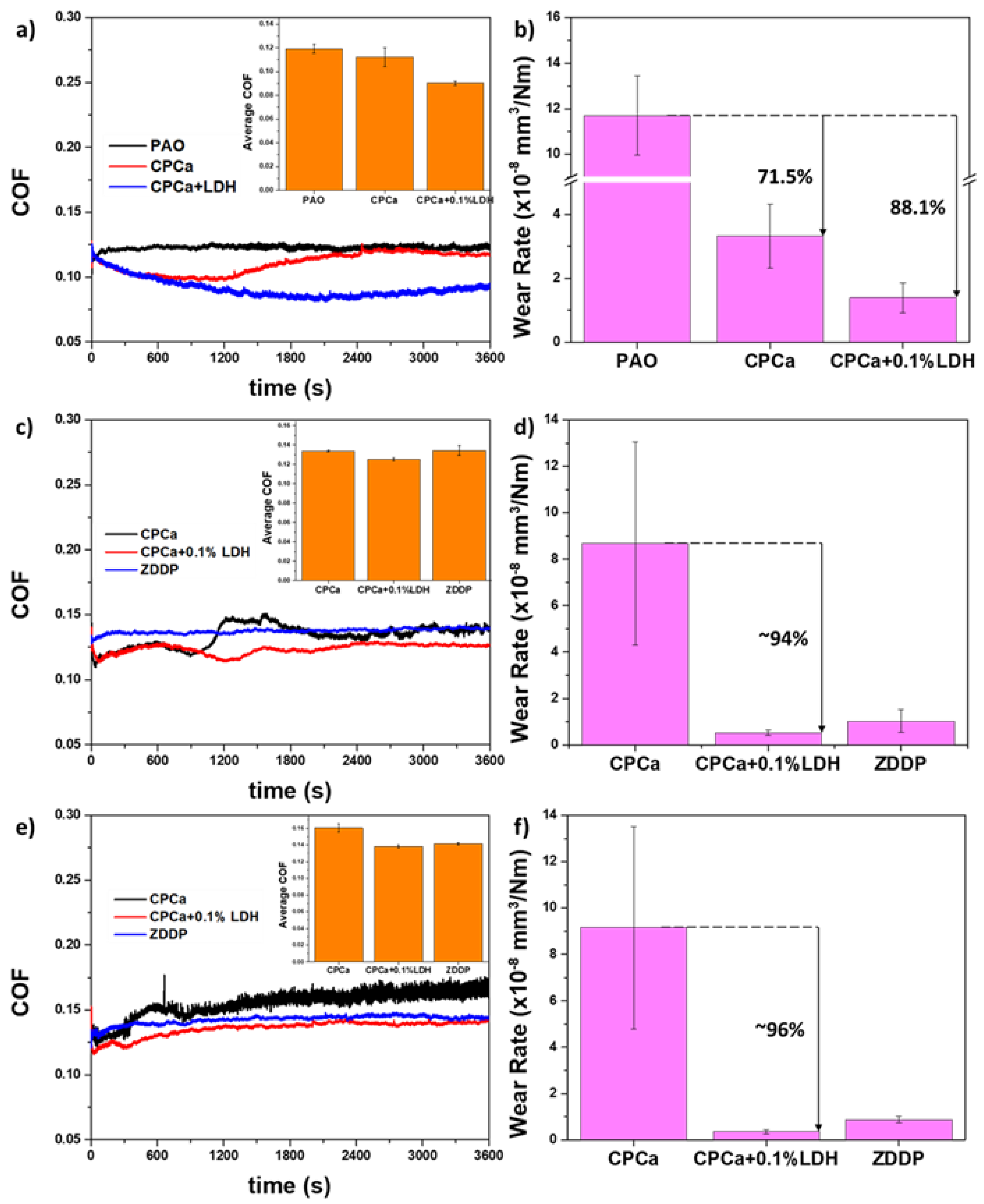
5. Compatibility of Carbon Tribofilms with Other Lubricant Additives
6. Outlooks
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Holmberg, K.; Erdemir, A. Influence of tribology on global energy consumption, costs and emissions. Friction 2017, 5, 263–284. [Google Scholar] [CrossRef]
- Holmberg, K.; Andersson, P.; Nylund, N.-O.; Mäkelä, K.; Erdemir, A. Global energy consumption due to friction in trucks and buses. Tribol. Int. 2014, 78, 94–114. [Google Scholar] [CrossRef]
- Verma, S.; Mishra, S.; Gaur, A.; Chowdhury, S.; Mohapatra, S.; Dwivedi, G.; Verma, P. A comprehensive review on energy storage in hybrid electric vehicle. J. Traffic Transp. Eng 2021, 8, 621–637. [Google Scholar] [CrossRef]
- Lešnik, L.; Kegl, B.; Torres-Jiménez, E.; Cruz-Peragón, F. Why we should invest further in the development of internal combustion engines for road applications. Oil & Gas Science and Technology–Revue d’IFP Energies nouvelles 2020, 75, 56. [Google Scholar]
- Martinez, J.S.; Hissel, D.; Pera, M.-C.; Amiet, M. Practical control structure and energy management of a testbed hybrid electric vehicle. IEEE Trans. Veh. 2011, 60, 4139–4152. [Google Scholar] [CrossRef]
- Tie, S.F.; Tan, C.W. A review of energy sources and energy management system in electric vehicles. Renew. Sust. Energy Rev. 2013, 20, 82–102. [Google Scholar] [CrossRef]
- Leach, F.; Kalghatgi, G.; Stone, R.; Miles, P. The scope for improving the efficiency and environmental impact of internal combustion engines. Transp. Eng. 2020, 1, 100005. [Google Scholar] [CrossRef]
- Kohlhauser, B.; Ripoll, M.R.; Riedl, H.; Koller, C.; Koutna, N.; Amsüss, A.; Hutter, H.; Ramirez, G.; Gachot, C.; Erdemir, A. How to get noWear?–A new take on the design of in-situ formed high performing low-friction tribofilms. Mater. Des. 2020, 190, 108519. [Google Scholar] [CrossRef]
- Xu, X.; Su, F.; Li, Z. Microstructure and tribological behaviors of MoN-Cu nanocomposite coatings sliding against Si3N4 ball under dry and oil-lubricated conditions. Wear 2019, 434, 202994. [Google Scholar] [CrossRef]
- Yu, H.; Xu, Y.; Shi, P.; Wang, H.; Wei, M.; Zhao, K.; Xu, B. Microstructure, mechanical properties and tribological behavior of tribofilm generated from natural serpentine mineral powders as lubricant additive. Wear 2013, 297, 802–810. [Google Scholar] [CrossRef]
- Charlier, J.-C.; Michenaud, J.-P. Energetics of multilayered carbon tubules. Phys. Rev. Lett. 1993, 70, 1858. [Google Scholar] [CrossRef]
- Zhang, R.; Ning, Z.; Zhang, Y.; Zheng, Q.; Chen, Q.; Xie, H.; Zhang, Q.; Qian, W.; Wei, F. Superlubricity in centimetres-long double-walled carbon nanotubes under ambient conditions. Nat. Nanotechnol. 2013, 8, 912–916. [Google Scholar] [CrossRef]
- Kinoshita, H.; Nishina, Y.; Alias, A.A.; Fujii, M. Tribological properties of monolayer graphene oxide sheets as water-based lubricant additives. Carbon 2014, 66, 720–723. [Google Scholar] [CrossRef]
- Wu, P.; Li, X.; Zhang, C.; Chen, X.; Lin, S.; Sun, H.; Lin, C.-T.; Zhu, H.; Luo, J. Self-assembled graphene film as low friction solid lubricant in macroscale contact. ACS Appl. Mater. Interfaces 2017, 9, 21554–21562. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xu, X.; Zhou, Y.; Lee, K.-R.; Wang, A. Insights into friction dependence of carbon nanoparticles as oil-based lubricant additive at amorphous carbon interface. Carbon 2019, 150, 465–474. [Google Scholar] [CrossRef]
- Tang, J.; Chen, S.; Jia, Y.; Ma, Y.; Xie, H.; Quan, X.; Ding, Q. Carbon dots as an additive for improving performance in water-based lubricants for amorphous carbon (aC) coatings. Carbon 2020, 156, 272–281. [Google Scholar] [CrossRef]
- Chen, X.; Li, J. Superlubricity of carbon nanostructures. Carbon 2020, 158, 1–23. [Google Scholar] [CrossRef]
- Azman, S.S.N.; Zulkifli, N.W.M.; Masjuki, H.; Gulzar, M.; Zahid, R. Study of tribological properties of lubricating oil blend added with graphene nanoplatelets. J. Mater. Res. 2016, 31, 1932–1938. [Google Scholar] [CrossRef]
- Lin, J.; Wang, L.; Chen, G. Modification of graphene platelets and their tribological properties as a lubricant additive. Tribol. Lett. 2011, 41, 209–215. [Google Scholar] [CrossRef]
- Berman, D.; Erdemir, A.; Sumant, A.V. Graphene: A new emerging lubricant. Mater. Today 2014, 17, 31–42. [Google Scholar] [CrossRef]
- Eswaraiah, V.; Sankaranarayanan, V.; Ramaprabhu, S. Graphene-based engine oil nanofluids for tribological applications. ACS Appl. Mater. Interfaces. 2011, 3, 4221–4227. [Google Scholar] [CrossRef]
- Al Mahmud, K.; Kalam, M.A.; Masjuki, H.H.; Mobarak, H.; Zulkifli, N. An updated overview of diamond-like carbon coating in tribology. Crit. Rev. Solid State Mater. Sci. 2015, 40, 90–118. [Google Scholar] [CrossRef]
- Vetter, J. 60 years of DLC coatings: Historical highlights and technical review of cathodic arc processes to synthesize various DLC types, and their evolution for industrial applications. Surf. Coat. Technol. 2014, 257, 213–240. [Google Scholar] [CrossRef]
- Whitener Jr, K.E.; Sheehan, P.E. Graphene synthesis. Diam. Relat. Mater. 2014, 46, 25–34. [Google Scholar] [CrossRef]
- Taylor, C. Automobile engine tribology—Design considerations for efficiency and durability. Wear 1998, 221, 1–8. [Google Scholar] [CrossRef]
- Berman, D.; Erdemir, A. Achieving Ultralow Friction and Wear by Tribocatalysis: Enabled by In-Operando Formation of Nanocarbon Films. ACS Nano 2021, 15, 18865–18879. [Google Scholar] [CrossRef]
- Spikes, H. Low-and zero-sulphated ash, phosphorus and sulphur anti-wear additives for engine oils. Lubr. Sci. 2008, 20, 103–136. [Google Scholar] [CrossRef]
- Wang, J.; Li, Z.; Zhao, H.; Ren, S.; Wang, C.; Huang, X.; Zheng, L.; Ren, T. Synergistic effects between sulfur-and phosphorus-free organic molybdenums and ZDDP as lubricating additives in PAO 6. Tribol. Int. 2022, 165, 107324. [Google Scholar] [CrossRef]
- Wong, V.W.; Tung, S.C. Overview of automotive engine friction and reduction trends–Effects of surface, material, and lubricant-additive technologies. Friction 2016, 4, 1–28. [Google Scholar] [CrossRef]
- Macian, V.; Tormos, B.; Ruiz, S.; Miro, G. Low viscosity engine oils: Study of wear effects and oil key parameters in a heavy duty engine fleet test. Tribol. Int. 2016, 94, 240–248. [Google Scholar] [CrossRef]
- Tormos, B.; Ramírez, L.; Johansson, J.; Björling, M.; Larsson, R. Fuel consumption and friction benefits of low viscosity engine oils for heavy duty applications. Tribol. Int. 2017, 110, 23–34. [Google Scholar] [CrossRef]
- Erdemir, A.; Donnet, C. Tribology of diamond-like carbon films: Recent progress and future prospects. J. Phys. D 2006, 39, R311. [Google Scholar] [CrossRef]
- Biswas, S. Some mechanisms of tribofilm formation in metal/metal and ceramic/metal sliding interactions. Wear 2000, 245, 178–189. [Google Scholar] [CrossRef]
- Johnson, B.; Wu, H.; Desanker, M.; Pickens, D.; Chung, Y.-W.; Wang, Q.J. Direct formation of lubricious and wear-protective carbon films from phosphorus-and sulfur-free oil-soluble additives. Tribol. Lett. 2018, 66, 2. [Google Scholar] [CrossRef]
- Rouhani, M.; Hobley, J.; Ou, H.-H.; Lee, J.-T.; Metla, S.B.S.; Jeng, Y.-R. A new gateway to ecofriendly self-healing amorphous carbon tribofilms from ancient oils. Appl. Mater. Today 2022, 29, 101616. [Google Scholar] [CrossRef]
- Erdemir, A.; Ramirez, G.; Eryilmaz, O.L.; Narayanan, B.; Liao, Y.; Kamath, G.; Sankaranarayanan, S.K. Carbon-based tribofilms from lubricating oils. Nature 2016, 536, 67–71. [Google Scholar] [CrossRef]
- Ma, Q.; Khan, A.M.; Wang, Q.J. Dependence of Tribological Performance and Tribopolymerization on the Surface Binding Strength of Selected Cycloalkane-Carboxylic Acid Additives. Tribol. Lett. 2020, 68, 1–10. [Google Scholar] [CrossRef]
- Fu, X.; Cao, L.; Wan, Y.; Li, R. Superlubricity achieved with TiN coatings via the in situ formation of a carbon-based film at the sliding interfaces. Ceram. Int. 2021, 47, 33917–33921. [Google Scholar] [CrossRef]
- Khan, A.M.; Wu, H.; Ma, Q.; Chung, Y.-W.; Wang, Q.J. Relating tribological performance and tribofilm formation to the adsorption strength of surface-active precursors. Tribol. Lett. 2020, 68, 1–9. [Google Scholar] [CrossRef]
- Wu, H.; Khan, A.M.; Johnson, B.; Sasikumar, K.; Chung, Y.-W.; Wang, Q.J. Formation and nature of carbon-containing tribofilms. ACS Appl. Mater. Interfaces 2019, 11, 16139–16146. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, Y.; Yang, G.; Gao, C.; Song, N.; Zhang, S.; Zhang, P. In-situ formed carbon based composite tribo-film with ultra-high load bearing capacity. Tribol. Int. 2020, 152, 106577. [Google Scholar] [CrossRef]
- Yu, H.; Liu, X.; Zheng, Z.; Qiao, D.; Feng, D.; Gong, Z.; Dong, G. In Situ Graphene Formation Induced by Tribochemical Reaction for Sustainable Lubrication. ACS Sustain. Chem. Eng. 2023, 11, 2238–2248. [Google Scholar] [CrossRef]
- Schmellenmeier, H. Die Beeinflussung von festen Oberflachen durch eine ionisierte. Exp. Tech. Phys 1953, 1, 49–68. [Google Scholar]
- Hauert, R. An overview on the tribological behavior of diamond-like carbon in technical and medical applications. Tribol. Int. 2004, 37, 991–1003. [Google Scholar] [CrossRef]
- Field, S.; Jarratt, M.; Teer, D. Tribological properties of graphite-like and diamond-like carbon coatings. Tribol. Int. 2004, 37, 949–956. [Google Scholar] [CrossRef]
- Fein, R.; Kreuz, K. Chemistry of boundary lubrication of steel by hydrocarbons. ASLE Trans. 1965, 8, 29–38. [Google Scholar] [CrossRef]
- Yeon, J.; He, X.; Martini, A.; Kim, S.H. Mechanochemistry at solid surfaces: Polymerization of adsorbed molecules by mechanical shear at tribological interfaces. ACS Appl. Mater. Interfaces 2017, 9, 3142–3148. [Google Scholar] [CrossRef]
- Donnet, C.; Erdemir, A. Tribology of Diamond-Like Carbon Films: Fundamentals and Applications; Springer Science & Business Media: Berlin, Germany, 2007. [Google Scholar] [CrossRef]
- Kano, M. Super low friction of DLC applied to engine cam follower lubricated with ester-containing oil. Tribol. Int. 2006, 39, 1682–1685. [Google Scholar] [CrossRef]
- Artini, C.; Muolo, M.; Passerone, A. Diamond–metal interfaces in cutting tools: A review. J. Mater. Sci. 2012, 47, 3252–3264. [Google Scholar] [CrossRef]
- Sullivan, J.; Friedmann, T.; Hjort, K. Diamond and amorphous carbon MEMS. MRS Bull. 2001, 26, 309–311. [Google Scholar] [CrossRef]
- Erdemir, A. The role of hydrogen in tribological properties of diamond-like carbon films. Surf. Coat. Technol. 2001, 146, 292–297. [Google Scholar] [CrossRef]
- Robertson, J. Diamond-like amorphous carbon. Mater. Sci. Eng. R Rep. 2002, 37, 129–281. [Google Scholar] [CrossRef]
- da Cruz, M.G.A.; Budnyak, T.M.; Rodrigues, B.V.; Budnyk, S.; Slabon, A. Biocoatings and additives as promising candidates for ultralow friction systems. Green Chem Lett Rev 2021, 14, 358–381. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, C.; Kato, T.; Yang, X.-a.; Wu, S.; Wang, R.; Nosaka, M.; Luo, J. Evolution of tribo-induced interfacial nanostructures governing superlubricity in aC: H and aC: H: Si films. Nat. Commun. 2017, 8, 1–13. [Google Scholar] [CrossRef]
- Erdemir, A.; Eryilmaz, O. Achieving superlubricity in DLC films by controlling bulk, surface, and tribochemistry. Friction 2014, 2, 140–155. [Google Scholar] [CrossRef]
- Hoffman, E.E.; Marks, L.D. Graphitic carbon films across systems. Tribol. Lett. 2016, 63, 1–21. [Google Scholar] [CrossRef]
- Erdemir, A.; Eryilmaz, O.; Fenske, G. Synthesis of diamondlike carbon films with superlow friction and wear properties. J. Vac. Sci. Technol. 2000, 18, 1987–1992. [Google Scholar] [CrossRef]
- Rusanov, A.; Nevshupa, R.; Fontaine, J.; Martin, J.-M.; Le Mogne, T.; Elinson, V.; Lyamin, A.; Roman, E. Probing the tribochemical degradation of hydrogenated amorphous carbon using mechanically stimulated gas emission spectroscopy. Carbon 2015, 81, 788–799. [Google Scholar] [CrossRef]
- Al-Azizi, A.A.; Eryilmaz, O.; Erdemir, A.; Kim, S.H. Surface structure of hydrogenated diamond-like carbon: Origin of run-in behavior prior to superlubricious interfacial shear. Langmuir 2015, 31, 1711–1721. [Google Scholar] [CrossRef]
- Robertson, J. Diamond-like carbon films, properties and applications. Compr. Hard Mater. 2014, 101–139. [Google Scholar]
- Shahsavari, F.; Ehteshamzadeh, M.; Amin, M.H.; Barlow, A.J. A comparative study of surface morphology, mechanical and tribological properties of DLC films deposited on Cr and Ni nanolayers. Ceram. Int. 2020, 46, 5077–5085. [Google Scholar] [CrossRef]
- Tamulevičius, S.; Meškinis, Š.; Tamulevičius, T.; Rubahn, H.-G. Diamond like carbon nanocomposites with embedded metallic nanoparticles. Rep. Prog. Phys. 2018, 81, 024501. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Chen, C.-H. The effect of thermal and plastic mismatch on stress distribution in diamond like carbon film under different interlayer/substrate system. Diam. Relat. Mater. 2008, 17, 1534–1540. [Google Scholar] [CrossRef]
- Zhou, K. Carbon Nanomaterials: Modeling, Design, and Applications; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Wei, C.; Wang, Y.-S.; Tai, F.-C. The role of metal interlayer on thermal stress, film structure, wettability and hydrogen content for diamond like carbon films on different substrate. Diam. Relat. Mater. 2009, 18, 407–412. [Google Scholar] [CrossRef]
- Peng, X.; Barber, Z.; Clyne, T. Surface roughness of diamond-like carbon films prepared using various techniques. Surf. Coat. Technol. 2001, 138, 23–32. [Google Scholar] [CrossRef]
- Puchi-Cabrera, E.; Staia, M.; Ochoa-Pérez, E.; Teer, D.; Santana-Méndez, Y.; La Barbera-Sosa, J.; Chicot, D.; Lesage, J. Fatigue behavior of a 316L stainless steel coated with a DLC film deposited by PVD magnetron sputter ion plating. Mater. Sci. Eng. A 2010, 527, 498–508. [Google Scholar] [CrossRef]
- Hainsworth, S.V.; Uhure, N. Diamond like carbon coatings for tribology: Production techniques, characterisation methods and applications. Int. Mater. Rev. 2007, 52, 153–174. [Google Scholar] [CrossRef]
- Aouadi, S.M.; Gu, J.; Berman, D. Self-healing ceramic coatings that operate in extreme environments: A review. J. Vac. Sci. Technol. 2020, 38, 050802. [Google Scholar] [CrossRef]
- Shirani, A.; Gu, J.; Wei, B.; Lee, J.; Aouadi, S.M.; Berman, D. Tribologically enhanced self-healing of niobium oxide surfaces. Surf. Coat. Technol. 2019, 364, 273–278. [Google Scholar] [CrossRef]
- Shirani, A.; Li, Y.; Eryilmaz, O.L.; Berman, D. Tribocatalytically-activated formation of protective friction and wear reducing carbon coatings from alkane environment. Sci. Rep. 2021, 11, 1–9. [Google Scholar] [CrossRef]
- Xu, X.; Li, Q.; Su, F.; Sun, J.; Li, W. In-situ formation of onion-like carbon film by tribo-induced catalytic degradation of hydrocarbon: Effect of lubrication condition and load. Chem. Eng. J. 2023, 459, 141566. [Google Scholar] [CrossRef]
- Xu, X.; Xu, Z.; Sun, J.; Tang, G.; Su, F. In situ Synthesizing Carbon-Based Film by Tribo-Induced Catalytic Degradation of Poly-α-Olefin Oil for Reducing Friction and Wear. Langmuir 2020, 36, 10555–10564. [Google Scholar] [CrossRef] [PubMed]
- Curry, J.F.; Babuska, T.F.; Furnish, T.A.; Lu, P.; Adams, D.P.; Kustas, A.B.; Nation, B.L.; Dugger, M.T.; Chandross, M.; Clark, B.G. Achieving ultralow wear with stable nanocrystalline metals. Adv. Mater. 2018, 30, 1802026. [Google Scholar] [CrossRef] [PubMed]
- Bhaskaran, H.; Gotsmann, B.; Sebastian, A.; Drechsler, U.; Lantz, M.A.; Despont, M.; Jaroenapibal, P.; Carpick, R.W.; Chen, Y.; Sridharan, K. Ultralow nanoscale wear through atom-by-atom attrition in silicon-containing diamond-like carbon. Nat. Nanotechnol. 2010, 5, 181–185. [Google Scholar] [CrossRef]
- Argibay, N.; Babuska, T.; Curry, J.; Dugger, M.; Lu, P.; Adams, D.; Nation, B.; Doyle, B.; Pham, M.; Pimentel, A. In-situ tribochemical formation of self-lubricating diamond-like carbon films. Carbon 2018, 138, 61–68. [Google Scholar] [CrossRef]
- Homma, Y. Gold nanoparticles as the catalyst of single-walled carbon nanotube synthesis. Catalysts 2014, 4, 38–48. [Google Scholar] [CrossRef]
- Sattler, J.J.; Ruiz-Martinez, J.; Santillan-Jimenez, E.; Weckhuysen, B.M. Catalytic dehydrogenation of light alkanes on metals and metal oxides. Chem. Rev. 2014, 114, 10613–10653. [Google Scholar] [CrossRef]
- Qi, Y.; Yang, J.; Rappe, A.M. Theoretical modeling of tribochemical reaction on Pt and Au contacts: Mechanical load and catalysis. ACS Appl. Mater. Interfaces 2016, 8, 7529–7535. [Google Scholar] [CrossRef]
- Jones, M.R.; DelRio, F.W.; Beechem, T.E.; McDonald, A.E.; Babuska, T.F.; Dugger, M.T.; Chandross, M.; Argibay, N.; Curry, J.F. Stress-and Time-Dependent Formation of Self-Lubricating In Situ Carbon (SLIC) Films on Catalytically-Active Noble Alloys. JOM 2021, 73, 3658–3667. [Google Scholar] [CrossRef]
- Shirani, A.; Li, Y.; Smith, J.; Curry, J.; Lu, P.; Wilson, M.; Chandross, M.; Argibay, N.; Berman, D. Mechanochemically driven formation of protective carbon films from ethanol environment. Mater. Today Chem. 2022, 26, 101112. [Google Scholar] [CrossRef]
- DelRio, F.W.; Mangolini, F.; Edwards, C.E.; Babuska, T.F.; Adams, D.P.; Lu, P.; Curry, J.F. Revealing the structure-property relationships of amorphous carbon tribofilms on platinum-gold surfaces. Wear 2023, 522, 204690. [Google Scholar] [CrossRef]
- Ta, T.D.; Tieu, A.K.; Tran, B.H. Influences of Iron and Iron Oxides on Ultra-thin Carbon-based Tribofilm Lubrication. Tribol. Int. 2022, 173, 107665. [Google Scholar] [CrossRef]
- Zheng, J.; Zhang, L.; Du, Z.; Zhang, C.; Li, H. Tribopolymerization of n-butyl acrylate on the steel–steel rubbing surface. Tribol. Int. 2008, 41, 769–777. [Google Scholar] [CrossRef]
- He, X.; Kim, S.H. Surface Chemistry Dependence of Mechanochemical Reaction of Adsorbed Molecules An Experimental Study on Tribopolymerization of α-Pinene on Metal, Metal Oxide, and Carbon Surfaces. Langmuir 2018, 34, 2432–2440. [Google Scholar] [CrossRef]
- Khajeh, A.; He, X.; Yeon, J.; Kim, S.H.; Martini, A. Mechanochemical association reaction of interfacial molecules driven by shear. Langmuir 2018, 34, 5971–5977. [Google Scholar] [CrossRef]
- Fuadi, Z.; Adachi, K.; Muhammad, T. Formation of carbon-based tribofilm under palm methyl ester. Tribol. Lett. 2018, 66, 1–11. [Google Scholar] [CrossRef]
- Schneider, M.P. Plant-oil-based lubricants and hydraulic fluids. J. Sci. Food Agric. 2006, 86, 1769–1780. [Google Scholar] [CrossRef]
- Zulkifli, N.W.M.; Kalam, M.; Masjuki, H.H.; Shahabuddin, M.; Yunus, R. Wear prevention characteristics of a palm oil-based TMP (trimethylolpropane) ester as an engine lubricant. Energy 2013, 54, 167–173. [Google Scholar] [CrossRef]
- Huang, E.-P.; Huang, E.; Yu, S.-C.; Chen, Y.-H.; Lee, J.-S.; Fang, J.-N. In situ Raman spectroscopy on kerogen at high temperatures and high pressures. Phys Chem Miner 2010, 37, 593–600. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Y.; Liu, H.; Zeng, F.; Guo, P.; Jiang, W. Study on the adsorption, diffusion and permeation selectivity of shale gas in organics. Energies 2017, 10, 142. [Google Scholar] [CrossRef]
- Reinhardt, M.; Goetz, W.; Duda, J.-P.; Heim, C.; Reitner, J.; Thiel, V. Organic signatures in Pleistocene cherts from Lake Magadi (Kenya)–implications for early Earth hydrothermal deposits. Biogeosciences 2019, 16, 2443–2465. [Google Scholar] [CrossRef]
- Dai, W.; Kheireddin, B.; Gao, H.; Liang, H. Roles of nanoparticles in oil lubrication. Tribol. Int. 2016, 102, 88–98. [Google Scholar] [CrossRef]
- Lee, K.; Hwang, Y.; Cheong, S.; Choi, Y.; Kwon, L.; Lee, J.; Kim, S.H. Understanding the role of nanoparticles in nano-oil lubrication. Tribol. Lett. 2009, 35, 127–131. [Google Scholar] [CrossRef]
- Chou, R.; Battez, A.H.; Cabello, J.; Viesca, J.; Osorio, A.; Sagastume, A. Tribological behavior of polyalphaolefin with the addition of nickel nanoparticles. Tribol. Int. 2010, 43, 2327–2332. [Google Scholar] [CrossRef]
- Williams, G.R.; O'Hare, D. Towards understanding, control and application of layered double hydroxide chemistry. J. Mater. Chem. 2006, 16, 3065–3074. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y. Superlubricity achieved with two-dimensional nano-additives to liquid lubricants. Friction 2020, 8, 1007–1024. [Google Scholar] [CrossRef]
- Rives, V. Layered Double Hydroxides: Present and Future; Nova Publishers: Hauppauge, NY, USA, 2001. [Google Scholar]
- Wang, H.; Wang, Y.; Liu, Y.; Zhao, J.; Li, J.; Wang, Q.; Luo, J. Tribological behavior of layered double hydroxides with various chemical compositions and morphologies as grease additives. Friction 2021, 9, 952–962. [Google Scholar] [CrossRef]
- Xiao, H.; Liu, S. 2D nanomaterials as lubricant additive: A review. Mater. Des. 2017, 135, 319–332. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Liu, W.; Liu, Y.; Wang, K.; Li, J.; Ma, T.; Eryilmaz, O.L.; Shi, Y.; Erdemir, A. Superlubricity of polyalkylene glycol aqueous solutions enabled by ultrathin layered double hydroxide nanosheets. ACS Appl. Mater. Interfaces 2019, 11, 20249–20256. [Google Scholar] [CrossRef]
- Zhao, M.Q.; Zhang, Q.; Jia, X.L.; Huang, J.Q.; Zhang, Y.H.; Wei, F. Hierarchical composites of single/double-walled carbon nanotubes interlinked flakes from direct carbon deposition on layered double hydroxides. Adv. Funct. Mater. 2010, 20, 677–685. [Google Scholar] [CrossRef]
- Zhang, L.; Li, F.; Xiang, X.; Wei, M.; Evans, D.G. Ni-based supported catalysts from layered double hydroxides: Tunable microstructure and controlled property for the synthesis of carbon nanotubes. Chem. Eng. J. 2009, 155, 474–482. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, M.Q.; Tang, D.M.; Li, F.; Huang, J.Q.; Liu, B.; Zhu, W.C.; Zhang, Y.H.; Wei, F. Carbon-nanotube-array double helices. Angew. Chem. 2010, 122, 3724–3727. [Google Scholar] [CrossRef]
- Zhao, M.-Q.; Liu, X.-F.; Zhang, Q.; Tian, G.-L.; Huang, J.-Q.; Zhu, W.; Wei, F. Graphene/single-walled carbon nanotube hybrids: One-step catalytic growth and applications for high-rate Li–S batteries. ACS Nano 2012, 6, 10759–10769. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Guo, F.; Sheng, H.; Xia, K.; Liu, W.; Wen, J.; Shi, Y.; Erdemir, A.; Luo, J. Catalytically active oil-based lubricant additives enabled by calcining Ni–Al layered double hydroxides. J. Phys. Chem. Lett. 2019, 11, 113–120. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Liu, W.; Wang, R.; Wen, J.; Sheng, H.; Peng, J.; Erdemir, A.; Luo, J. Tribological behavior of NiAl-layered double hydroxide nanoplatelets as oil-based lubricant additives. ACS Appl. Mater. Interfaces 2017, 9, 30891–30899. [Google Scholar] [CrossRef]
- Huynh, K.K.; Pham, S.T.; Tieu, A.K.; Collins, S.M.; Lu, C.; Wan, S. Tribo-induced catalytically active oxide surfaces enabling the formation of the durable and high-performance carbon-based tribofilms. Tribol. Int. 2023, 184, 108476. [Google Scholar] [CrossRef]
- Wang, H.; Liu, Y.; Chen, Z.; Wu, B.; Xu, S.; Luo, J. Layered double hydroxide nanoplatelets with excellent tribological properties under high contact pressure as water-based lubricant additives. Sci. Rep. 2016, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, J.C. Moving sources of heat and the temperature at sliding contacts. Proc. Roy. Soc. New South Wales 1942, 76, 203. [Google Scholar] [CrossRef]
- Kajdas, C.; Kulczycki, A.; Ozimina, D. A new concept of the mechanism of tribocatalytic reactions induced by mechanical forces. Tribol. Int. 2017, 107, 144–151. [Google Scholar] [CrossRef]
- Eyring, H. Viscosity, plasticity, and diffusion as examples of absolute reaction rates. J. Chem. Phys. 1936, 4, 283–291. [Google Scholar] [CrossRef]
- Spikes, H. Stress-augmented thermal activation: Tribology feels the force. Friction 2018, 6, 1–31. [Google Scholar] [CrossRef]
- Ribas-Arino, J.; Marx, D. Covalent mechanochemistry: Theoretical concepts and computational tools with applications to molecular nanomechanics. Chem. Rev. 2012, 112, 5412–5487. [Google Scholar] [CrossRef] [PubMed]
- Gosvami, N.; Bares, J.; Mangolini, F.; Konicek, A.; Yablon, D.; Carpick, R. Mechanisms of antiwear tribofilm growth revealed in situ by single-asperity sliding contacts. Science 2015, 348, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Spikes, H. On the mechanism of ZDDP antiwear film formation. Tribol. Lett. 2016, 63, 1–15. [Google Scholar] [CrossRef]
- Bhushan, B.; Fuchs, H.; Tomitori, M. Applied Scanning Probe Methods VIII: Scanning Probe Microscopy Techniques; Springer Science & Business Media: Berlin, Germany, 2007. [Google Scholar]
- Pham, S.T.; Wan, S.; Tieu, K.A.; Ma, M.; Zhu, H.; Nguyen, H.H.; Mitchell, D.R.; Nancarrow, M.J. Unusual Competitive and Synergistic Effects of Graphite Nanoplates in Engine Oil on the Tribofilm Formation. Adv. Mater. Interfaces 2019, 6, 1901081. [Google Scholar] [CrossRef]
- Fursey, A. Oxide films on mild steel. Nature 1965, 207, 747–748. [Google Scholar] [CrossRef]
- Hu, E.; Hu, X.; Liu, T.; Fang, L.; Dearn, K.D.; Xu, H. The role of soot particles in the tribological behavior of engine lubricating oils. Wear 2013, 304, 152–161. [Google Scholar] [CrossRef]
- Huynh, K.K.; Tieu, K.A.; Pham, S.T. Synergistic and Competitive Effects between Zinc Dialkyldithiophosphates and Modern Generation of Additives in Engine Oil. Lubricants 2021, 9, 35. [Google Scholar] [CrossRef]
- Li, Y.-S.; Jang, S.; Khan, A.M.; Martin, T.V.; Ogrinc, A.L.; Wang, Q.J.; Martini, A.; Chung, Y.-W.; Kim, S.H. Possible origin of D-and G-band features in Raman spectra of tribofilms. Tribol. Lett. 2023, 71, 57. [Google Scholar] [CrossRef]
- Takabayashi, S.; Takahagi, T. Surface oxidation process of a diamond-like carbon film analyzed by difference X-ray photoelectron spectroscopy. Surf Interface Anal 2015, 47, 345–349. [Google Scholar] [CrossRef]
- Guo, C.; Pei, Z.; Fan, D.; Gong, J.; Sun, C. Microstructure and tribomechanical properties of (Cr, N)-DLC/DLC multilayer films deposited by a combination of filtered and direct cathodic vacuum arcs. Diam. Relat. Mater. 2015, 60, 66–74. [Google Scholar] [CrossRef]
- Takabayashi, S.; Okamoto, K.; Nakatani, T.; Sakaue, H.; Takahagi, T. Surface Analysis of Carbon–Hydrogen Bonds in Diamondlike Carbon Films by X-ray Photoelectron Spectroscopy. Jpn. J. Appl. Phys. 2009, 48, 092304. [Google Scholar] [CrossRef]
- Egerton, R.F. Electron energy-loss spectroscopy in the TEM. Rep. Prog. Phys. 2008, 72, 016502. [Google Scholar] [CrossRef]
- Hsu, C.S.; Robinson, P.R. Practical Advances in Petroleum Processing; Springer Science & Business Media: Berlin, Germany, 2007; Volume 1. [Google Scholar]
- Ramirez, G.; Eryilmaz, O.L.; Fatti, G.; Righi, M.C.; Wen, J.; Erdemir, A. Tribochemical conversion of methane to graphene and other carbon nanostructures: Implications for friction and wear. ACS Appl. Nano Mater. 2020, 3, 8060–8067. [Google Scholar] [CrossRef]
- Kajdas, C.; Hiratsuka, K. Tribochemistry, tribocatalysis, and the negative-ion-radical action mechanism. P I MECH ENG J-J ENG 2009, 223, 827–848. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, L.; Zhou, C. Review of chemical vapor deposition of graphene and related applications. Acc. Chem. Res. 2013, 46, 2329–2339. [Google Scholar] [CrossRef]
- Franz, R.; Mitterer, C. Vanadium containing self-adaptive low-friction hard coatings for high-temperature applications: A review. Surf. Coat. Technol. 2013, 228, 1–13. [Google Scholar] [CrossRef]
- Oliver, W.C.; Pharr, G.M. An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments. J. Mater. Res. 1992, 7, 1564–1583. [Google Scholar] [CrossRef]
- Kartsonakis, I.; Balaskas, A.; Koumoulos, E.; Charitidis, C.; Kordas, G. Incorporation of ceramic nanocontainers into epoxy coatings for the corrosion protection of hot dip galvanized steel. Corros. Sci. 2012, 57, 30–41. [Google Scholar] [CrossRef]
- Wei, C.; Yen, J.-Y. Effect of film thickness and interlayer on the adhesion strength of diamond like carbon films on different substrates. Diam. Relat. Mater. 2007, 16, 1325–1330. [Google Scholar] [CrossRef]
- Barenblatt, G.I. The Mathematical Theory of Equilibrium Cracks in Brittle Fracture. In Advances in Applied Mechanics; Elsevier: Amsterdam, The Netherlands, 1962; Volume 7, pp. 55–129. [Google Scholar]
- Windischmann, H. An intrinsic stress scaling law for polycrystalline thin films prepared by ion beam sputtering. J. Appl. Phys. 1987, 62, 1800–1807. [Google Scholar] [CrossRef]
- Davis, C. A simple model for the formation of compressive stress in thin films by ion bombardment. Thin Solid Films 1993, 226, 30–34. [Google Scholar] [CrossRef]
- Machlin, E. Materials Science in Microelectronics: The Relationship between Thin Film Processing and Structure; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Brenner, A.; Senderoff, S. Calculation of stress in electrodeposits from the curvature of a plated strip. J. Res. Natl. Bur. Stand 1949, 42, 105–123. [Google Scholar] [CrossRef]
- Shahsavari, F.; Ehteshamzadeh, M.; Naimi-Jamal, M.R.; Irannejad, A. Nanoindentation and nanoscratch behaviors of DLC films growth on different thickness of Cr nanolayers. Diam. Relat. Mater. 2016, 70, 76–82. [Google Scholar] [CrossRef]
- Fischer-Cripps, A.C. Time-Dependent Nanoindentation. In Nanoindentation; Springer: Berlin/Heidelberg, Germany, 2011; pp. 125–145. [Google Scholar]
- Hay, J.; O’Hern, M.; Oliver, W. Tie Importance of Contact Radius for Substrate-Independent Property Measurement of Thin Films. MRS Online Proc. Libr. 1998, 522, 27–32. [Google Scholar] [CrossRef]
- Podgornik, B.; Sedlaček, M.; Vižintin, J. Compatibility of DLC coatings with formulated oils. Tribol. Int. 2008, 41, 564–570. [Google Scholar] [CrossRef]
- Al Mahmud, K.; Varman, M.; Kalam, M.; Masjuki, H.H.; Mobarak, H.; Zulkifli, N. Tribological characteristics of amorphous hydrogenated (aC: H) and tetrahedral (ta-C) diamond-like carbon coating at different test temperatures in the presence of commercial lubricating oil. Surf. Coat. Technol. 2014, 245, 133–147. [Google Scholar] [CrossRef]
- Podgornik, B.; Vižintin, J. Action of oil additives when used in DLC coated contacts. Tribol. Mater. Surf. Interfaces 2010, 4, 186–190. [Google Scholar] [CrossRef]
- Kalin, M.; Roman, E.; Ožbolt, L.; Vižintin, J. Metal-doped (Ti, WC) diamond-like-carbon coatings: Reactions with extreme-pressure oil additives under tribological and static conditions. Thin Solid Films 2010, 518, 4336–4344. [Google Scholar] [CrossRef]
- Kržan, B.; Novotny-Farkas, F.; Vižintin, J. Tribological behavior of tungsten-doped DLC coating under oil lubrication. Tribol. Int. 2009, 42, 229–235. [Google Scholar] [CrossRef]
- Mistry, K.K.; Morina, A.; Neville, A. A tribochemical evaluation of a WC–DLC coating in EP lubrication conditions. Wear 2011, 271, 1739–1744. [Google Scholar] [CrossRef]
- Haque, T.; Morina, A.; Neville, A.; Kapadia, R.; Arrawsmith, S. Study of the ZDDP Antiwear Tribofilm Formed on the DLC Coating Using AFM and XPS Techniques. In Automotive Lubricant Testing and Advanced Additive Development; ASTM International: West Conshohocken, PA, USA, 2008. [Google Scholar] [CrossRef]
- Topolovec-Miklozic, K.; Lockwood, F.; Spikes, H. Behaviour of boundary lubricating additives on DLC coatings. Wear 2008, 265, 1893–1901. [Google Scholar] [CrossRef]
- Vengudusamy, B.; Green, J.H.; Lamb, G.D.; Spikes, H.A. Durability of ZDDP tribofilms formed in DLC/DLC contacts. Tribol. Lett. 2013, 51, 469–478. [Google Scholar] [CrossRef]
- Tasdemir, H.A.; Tokoroyama, T.; Kousaka, H.; Umehara, N.; Mabuchi, Y. Influence of zinc dialkyldithiophosphate tribofilm formation on the tribological performance of self-mated diamond-like carbon contacts under boundary lubrication. Thin Solid Films 2014, 562, 389–397. [Google Scholar] [CrossRef]
- Vengudusamy, B.; Green, J.H.; Lamb, G.D.; Spikes, H.A. Influence of hydrogen and tungsten concentration on the tribological properties of DLC/DLC contacts with ZDDP. Wear 2013, 298, 109–119. [Google Scholar] [CrossRef]
- Qu, J.; Meyer III, H.M.; Cai, Z.-B.; Ma, C.; Luo, H. Characterization of ZDDP and ionic liquid tribofilms on non-metallic coatings providing insights of tribofilm formation mechanisms. Wear 2015, 332, 1273–1285. [Google Scholar] [CrossRef]
- Yue, W.; Liu, C.; Fu, Z.; Wang, C.; Huang, H.; Liu, J. Effects of molybdenum dithiocarbamate and zinc dialkyl dithiophosphate additives on tribological behaviors of hydrogenated diamond-like carbon coatings. Mater. Des. 2014, 64, 601–607. [Google Scholar] [CrossRef]
- Ren, S.; Zheng, S.; Pu, J.; Lu, Z.; Zhang, G. Study of tribological mechanisms of carbon-based coatings in antiwear additive containing lubricants under high temperature. RSC Adv. 2015, 5, 66426–66437. [Google Scholar] [CrossRef]
- Salinas Ruiz, V.R.; Kuwahara, T.; Galipaud, J.; Masenelli-Varlot, K.; Hassine, M.B.; Héau, C.; Stoll, M.; Mayrhofer, L.; Moras, G.; Martin, J.M. Interplay of mechanics and chemistry governs wear of diamond-like carbon coatings interacting with ZDDP-additivated lubricants. Nat. Commun. 2021, 12, 4550. [Google Scholar] [CrossRef]
- Kano, M.; Yasuda, Y.; Okamoto, Y.; Mabuchi, Y.; Hamada, T.; Ueno, T.; Ye, J.; Konishi, S.; Takeshima, S.; Martin, J. Ultralow friction of DLC in presence of glycerol mono-oleate (GNO). Tribol. Lett. 2005, 18, 245–251. [Google Scholar] [CrossRef]
- Dobrenizki, L.; Tremmel, S.; Wartzack, S.; Hoffmann, D.C.; Brögelmann, T.; Bobzin, K.; Bagcivan, N.; Musayev, Y.; Hosenfeldt, T. Efficiency improvement in automobile bucket tappet/camshaft contacts by DLC coatings–Influence of engine oil, temperature and camshaft speed. Surf. Coat. Technol. 2016, 308, 360–373. [Google Scholar] [CrossRef]
- Zahid, R.; Bhutta, M.U.; Mufti, R.A.; Abdullah, M.U.; Masjuki, H.H.; Varman, M.; Kalam, M.A.; Ali, M.A.; Aslam, J.; Akhtar, K. Friction and Wear Performance Evaluation of Bio-Lubricants and DLC Coatings on Cam/Tappet Interface of Internal Combustion Engines. Materials 2021, 14, 7206. [Google Scholar] [CrossRef] [PubMed]
- Simič, R.; Kalin, M. Adsorption mechanisms for fatty acids on DLC and steel studied by AFM and tribological experiments. Appl. Surf. Sci. 2013, 283, 460–470. [Google Scholar] [CrossRef]
- De Barros Bouchet, M.I.; Martin, J.M.; Avila, J.; Kano, M.; Yoshida, K.; Tsuruda, T.; Bai, S.; Higuchi, Y.; Ozawa, N.; Kubo, M. Diamond-like carbon coating under oleic acid lubrication: Evidence for graphene oxide formation in superlow friction. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Naganuma, Y.; Kano, M. Effect of Degree of Unsaturation in Vegetable Oils on Friction Properties of DLC Coatings. Tribol. Online 2021, 16, 210–215. [Google Scholar] [CrossRef]
- Tang, J.; Ding, Q.; Zhang, G.; Hu, L. The influence of total acid number of ester oil in tribological behavior of DLC contacts. Tribol. Trans. 2015, 58, 849–858. [Google Scholar] [CrossRef]
- Matta, C.; Joly-Pottuz, L.; Bouchet, M.D.B.; Martin, J.; Kano, M.; Zhang, Q.; Goddard III, W. Superlubricity and tribochemistry of polyhydric alcohols. Phys. Rev. B 2008, 78, 085436. [Google Scholar] [CrossRef]
- Sun, S.; Li, J.; Li, J.; Luo, J. Enhanced superlubricity on aC films by lubrication with 3-hydroxypropionic acid. Carbon 2022, 199, 161–169. [Google Scholar] [CrossRef]
- Long, Y.; Bouchet, M.-I.D.B.; Lubrecht, T.; Onodera, T.; Martin, J.M. Superlubricity of glycerol by self-sustained chemical polishing. Sci. Rep. 2019, 9, 6286. [Google Scholar] [CrossRef]
- Choi, Y.; Lee, C.; Hwang, Y.; Park, M.; Lee, J.; Choi, C.; Jung, M. Tribological behavior of copper nanoparticles as additives in oil. Curr Appl Phys 2009, 9, e124–e127. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, S.; Sun, D.; Yang, G.; Gao, C.; Zhou, C.; Zhang, C.; Zhang, P. Wide adaptability of Cu nano-additives to the hardness and composition of DLC coatings in DLC/PAO solid-liquid composite lubricating system. Tribol. Int. 2019, 138, 184–195. [Google Scholar] [CrossRef]
- Tang, G.; Su, F.; Xu, X.; Chu, P.K. 2D black phosphorus dotted with silver nanoparticles: An excellent lubricant additive for tribological applications. Chem. Eng. J. 2020, 392, 123631. [Google Scholar] [CrossRef]
- Wang, W.; Xie, G.; Luo, J. Black phosphorus as a new lubricant. Friction 2018, 6, 116–142. [Google Scholar] [CrossRef]
- Wang, W.; Xie, G.; Luo, J. Superlubricity of black phosphorus as lubricant additive. ACS Appl. Mater. Interfaces 2018, 10, 43203–43210. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.W.; Zhou, Y.; Bhattacharya, P.; Erck, R.; Qu, J.; Bays, J.T.; Cosimbescu, L. Probing the molecular design of hyper-branched aryl polyesters towards lubricant applications. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef]
- Han, K.; Zhang, Y.; Song, N.; Yu, L.; Zhang, P.; Zhang, Z.; Qian, L.; Zhang, S. The Current Situation and Future Direction of Nanoparticles Lubricant Additives in China. Lubricants 2022, 10, 312. [Google Scholar] [CrossRef]
- Pham, S.T.; Tieu, A.K.; Sencadas, V.; Lei, W.; Liu, D.; Wan, S.; Hao, J. Smart-Responsive Colloidal Capsules as an Emerging Tool to Design a Multifunctional Lubricant Additive. ACS Appl. Mater. Interfaces 2021, 13, 7714–7724. [Google Scholar] [CrossRef]




| Coating Systems | ID/IG | sp2/sp3 | sp3/(sp2 +sp3) (%) | [H]total (%) | Thermal Stress (MPa) |
|---|---|---|---|---|---|
| DLC-Si | 0.712 | 0.95 | 51.2 | 48.0 | 16.87 |
| DLC-(20 nm Cr)-Si | 0.669 | 1.64 | 37.9 | 42.2 | 36.50 |
| DLC-(20 nm Al)-Si | 0.673 | 2.01 | 33.2 | 40.0 | 55.30 |
| DLC-(40 nm Cr)-Si | 0.664 | 2.17 | 31.5 | 39.1 | 48.64 |
| DLC-(40 nm Al)-Si | 0.296 | 2.65 | 27.4 | 36.9 | 75.95 |
| DLC-Steel | 0.939 | 1.07 | 48.3 | 46.7 | 179.70 |
| DLC-(20 nm Cr)-Steel | 1.553 | 0.50 | 66.6 | 53.1 | 178.10 |
| DLC-(20 nm Al)-Steel | 1.361 | 0.80 | 55.6 | 49.5 | 115.00 |
| DLC-(40 nm Cr)-Steel | 1.488 | 0.48 | 67.6 | 56.0 | 177.10 |
| DLC-(40 nm Al)-Steel | 1.604 | 0.64 | 61.0 | 51.3 | 111.30 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huynh, K.K.; Pham, S.T.; Tieu, K.A.; Wan, S. Tribocatalysis Induced Carbon-Based Tribofilms—An Emerging Tribological Approach for Sustainable Lubrications. Lubricants 2023, 11, 327. https://doi.org/10.3390/lubricants11080327
Huynh KK, Pham ST, Tieu KA, Wan S. Tribocatalysis Induced Carbon-Based Tribofilms—An Emerging Tribological Approach for Sustainable Lubrications. Lubricants. 2023; 11(8):327. https://doi.org/10.3390/lubricants11080327
Chicago/Turabian StyleHuynh, Khai K., Sang T. Pham, Kiet A. Tieu, and Shanhong Wan. 2023. "Tribocatalysis Induced Carbon-Based Tribofilms—An Emerging Tribological Approach for Sustainable Lubrications" Lubricants 11, no. 8: 327. https://doi.org/10.3390/lubricants11080327
APA StyleHuynh, K. K., Pham, S. T., Tieu, K. A., & Wan, S. (2023). Tribocatalysis Induced Carbon-Based Tribofilms—An Emerging Tribological Approach for Sustainable Lubrications. Lubricants, 11(8), 327. https://doi.org/10.3390/lubricants11080327






