DPYD, TYMS and MTHFR Genes Polymorphism Frequencies in a Series of Turkish Colorectal Cancer Patients
Abstract
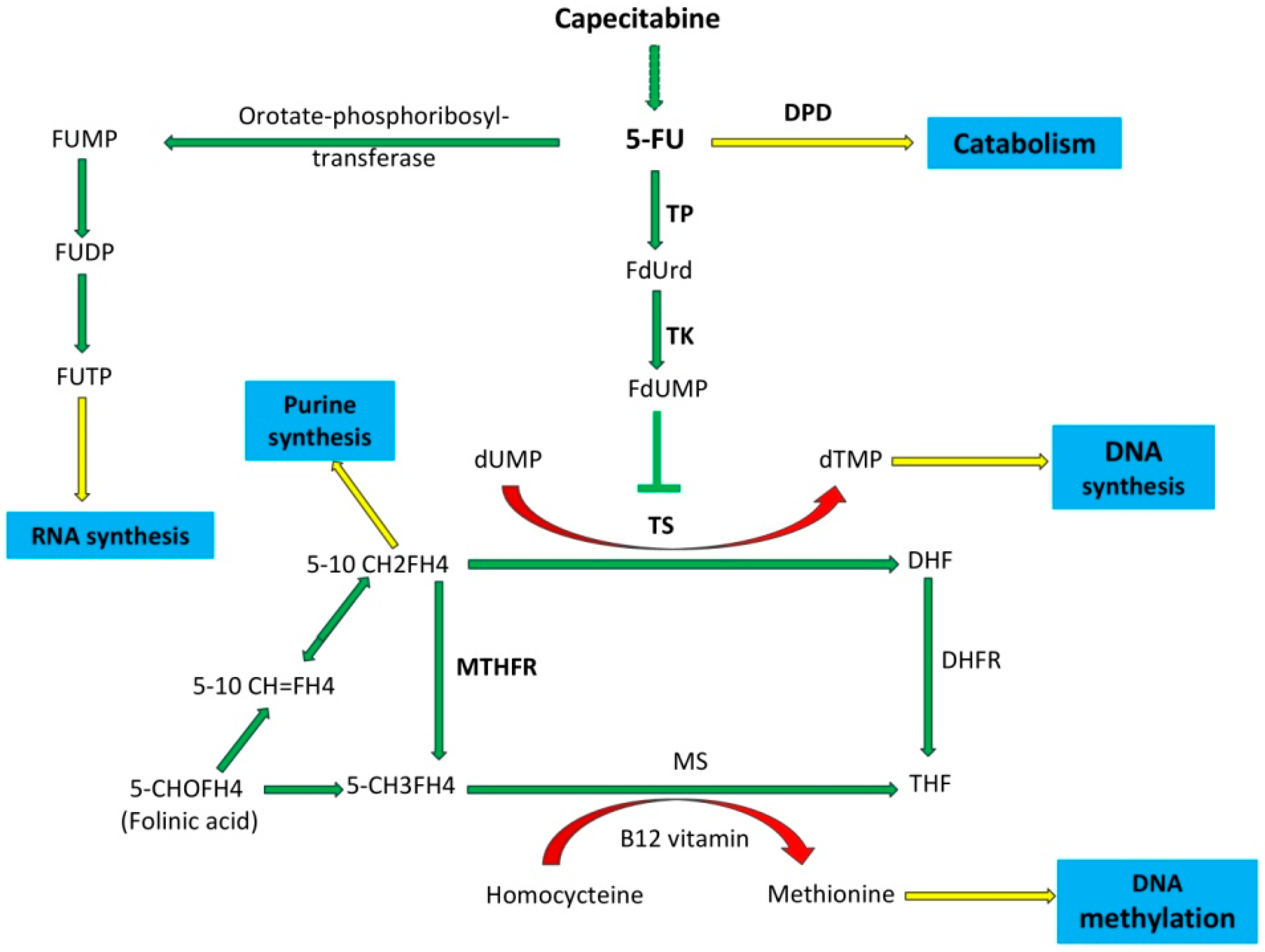
1. Introduction
2. Materials and Methods
2.1. Study Population and Setting
2.2. Demographic Data
2.3. Toxicity Evaluation
2.4. Genotyping
2.5. Statistical Analysis
3. Results
3.1. Patients’ Toxicity Types and Grades
3.2. Patient Genotypes
3.3. Impact of Gene Polymorphisms on Toxicity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Cremolini, C.; Del Re, M.; Antoniotti, C.; Lonardi, S.; Bergamo, F.; Loupakis, F.; Borelli, B.; Marmorino, F.; Citi, V.; Cortesi, E.; et al. DPYD and UGT1A1 genotyping to predict adverse events during first-line FOLFIRI or FOLFOXIRI plus bevacizumab in metastatic colorectal cancer. Oncotarget 2018, 9, 7859–7866. [Google Scholar] [CrossRef] [PubMed]
- Amstutz, U.; Froehlich, T.K.; Largiader, C.R. Dihydropyrimidine dehydrogenase gene as a major predictor of severe 5-fluorouracil toxicity. Pharmacogenomics 2011, 12, 1321–1336. [Google Scholar] [CrossRef] [PubMed]
- Ciccolini, J.; Gross, E.; Dahan, L.; Lacarelle, B.; Mercier, C. Routine dihydropyrimidine dehydrogenase testing for anticipating 5-fluorouracil-related severe toxicities: Hype or hope? Clin. Colorectal Cancer 2010, 9, 224–228. [Google Scholar] [CrossRef]
- Thomas, F.; Motsinger-Reif, A.A.; Hoskins, J.M.; Dvorak, A.; Roy, S.; Alyasiri, A.; Myerson, R.J.; Fleshman, J.W.; Tan, B.R.; McLeod, H.L. Methylenetetrahydrofolate reductase genetic polymorphisms and toxicity to 5-FU-based chemoradiation in rectal cancer. Br. J. Cancer 2011, 105, 1654–1662. [Google Scholar] [CrossRef]
- Afzal, S.; Jensen, S.A.; Vainer, B.; Vogel, U.; Matsen, J.P.; Sorensen, J.B.; Andersen, P.K.; Poulsen, H.E. MTHFR polymorphisms and 5-FU-based adjuvant chemotherapy in colorectal cancer. Ann. Oncol. 2009, 20, 1660–1666. [Google Scholar] [CrossRef] [PubMed]
- Cohen, V.; Panet-Raymond, V.; Sabbaghian, N.; Morin, I.; Batist, G.; Rozen, R. Methylenetetrahydrofolate reductase polymorphism in advanced colorectal cancer: A novel genomic predictor of clinical response to fluoropyrimidine-based chemotherapy. Clin. Cancer Res. 2003, 9, 1611–1615. [Google Scholar] [PubMed]
- Afzal, S.; Gusella, M.; Vainer, B.; Vogel, U.B.; Andersen, J.T.; Broedbaek, K.; Petersen, M.; Jimenez-Solem, E.; Bertolaso, L.; Barile, C.; et al. Combinations of polymorphisms in genes involved in the 5-Fluorouracil metabolism pathway are associated with gastrointestinal toxicity in chemotherapy-treated colorectal cancer patients. Clin. Cancer Res. 2011, 17, 3822–3829. [Google Scholar] [CrossRef] [PubMed]
- Schwab, M.; Zanger, U.M.; Marx, C.; Schaeffeler, E.; Klein, K.; Dippon, J.; Kerb, R.; Blievernicht, J.; Fischer, J.; Hofmann, U.; et al. Role of genetic and nongenetic factors for fluorouracil treatment-related severe toxicity: A prospective clinical trial by the German 5-FU Toxicity Study Group. J. Clin. Oncol. 2008, 26, 2131–2138. [Google Scholar] [CrossRef] [PubMed]
- Houtsma, D.; Guchelaar, H.J.; Gelderblom, H. Pharmacogenetics in oncology: A promising field. Curr. Pharm. Des. 2010, 16, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Greene, F.L.; Page, D.L.; Fleming, I.D.; Fritz, A.; Balch, C.M.; Haller, D.G.; Morrow, M. (Eds.) Cancer Staging Manual. In American Joint Committee on Cancer (AJCC); Digestive System; Lippincott Raven Publishers: Philadelphia, PA, USA, 2002; pp. 113–124. [Google Scholar]
- Bradic, M.; Costa, J.; Chelo, I.M. Genotyping with Sequenom. Meth. Mol. Biol. 2011, 772, 193–210. [Google Scholar] [CrossRef]
- Sherry, S.T.; Ward, M.-H.; Kholodov, M.; Baker, J.; Smigielski, E.M.; Sirotkin, K. dbSNP: The NCBI database of genetic variation. Nucleic Acids Res. 2001, 29, 308–311. [Google Scholar] [CrossRef] [PubMed]
- Van Kuilenburg, A.B.; De Abreu, R.A.; van Gennip, A.H. Pharmacogenetic and clinical aspects of dihydropyrimidine dehydrogenase deficiency. Ann. Clin. Biochem. 2003, 40, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Maitland, M.L.; Vasisht, K.; Ratain, M.J. TPMT, UGT1A1 and DPYD: Genotyping to ensure safer cancer therapy? Trends Pharmacol. Sci. 2006, 27, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Sulzyc-Bielicka, V.; Binczak-Kuleta, A.; Pioch, W.; Kladny, J.; Gziut, K.; Bielicki, D.; Ciechanowicz, A. 5-Fluorouracil toxicity-attributable IVS14 + 1G > A mutation of the dihydropyrimidine dehydrogenase gene in Polish colorectal cancer patients. Pharmacol. Rep. 2008, 60, 238–242. [Google Scholar] [PubMed]
- Mattison, L.K.; Soong, R.; Diasio, R.B. Implications of dihydropyrimidine dehydrogenase on 5-fluorouracil pharmacogenetics and pharmacogenomics. Pharmacogenomics 2002, 3, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Baskin, Y.; Amirfallah, A.; Unal, O.U.; Calibasi, G.; Oztop, I. Dihydropyrimidine dehydrogenase 85T>C mutation is associated with ocular toxicity of 5-fluorouracil: A case report. Am. J. Ther. 2015, 22, e36–e39. [Google Scholar] [CrossRef]
- Seck, K.; Riemer, S.; Kates, R.; Ullrich, T.; Lutz, V.; Harbeck, N.; Schmitt, M.; Kiechle, M.; Diasio, R.; Gross, E. Analysis of the DPYD gene implicated in 5-fluorouracil catabolism in a cohort of Caucasian individuals. Clin. Cancer Res. 2005, 11, 5886–5892. [Google Scholar] [CrossRef]
- He, Y.F.; Wei, W.; Zhang, X.; Li, Y.H.; Li, S.; Wang, F.H.; Lin, X.B.; Li, Z.M.; Zhang, D.S.; Huang, H.Q.; et al. Analysis of the DPYD gene implicated in 5-fluorouracil catabolism in Chinese cancer patients. J. Clin. Pharm. Ther. 2008, 33, 307–314. [Google Scholar] [CrossRef]
- Loganayagam, A.; Arenas Hernandez, M.; Corrigan, A.; Fairbanks, L.; Lewis, C.M.; Harper, P.; Maisey, N.; Ross, P.; Sanderson, J.D.; Marinaki, A.M. Pharmacogenetic variants in the DPYD, TYMS, CDA and MTHFR genes are clinically significant predictors of fluoropyrimidine toxicity. Br. J. Cancer 2013, 108, 2505–2515. [Google Scholar] [CrossRef]
- Lima, A.; Azevedo, R.; Sousa, H.; Seabra, V.; Medeiros, R. Current approaches for TYMS polymorphisms and their importance in molecular epidemiology and pharmacogenetics. Pharmacogenomics 2013, 14, 1337–1351. [Google Scholar] [CrossRef]
- De Mattia, E.; Toffoli, G. C677T and A1298C MTHFR polymorphisms, a challenge for antifolate and fluoropyrimidine-based therapy personalisation. Eur. J. Cancer 2009, 45, 1333–1351. [Google Scholar] [CrossRef] [PubMed]
- Martin, Y.N.; Salavaggione, O.E.; Eckloff, B.W.; Wieben, E.D.; Schaid, D.J.; Weinshilboum, R.M. Human methylenetetrahydrofolate reductase pharmacogenomics: Gene resequencing and functional genomics. Pharmacogenet. Genom. 2006, 16, 265–277. [Google Scholar] [CrossRef]
- Frosst, P.; Blom, H.J.; Milos, R.; Goyette, P.; Sheppard, C.A.; Matthews, R.G.; Boers, G.J.; den Heijer, M.; Kluijtmans, L.A.; van den Heuvel, L.P.; et al. A candidate genetic risk factor for vascular disease: A common mutation in methylenetetrahydrofolate reductase. Nat. Genet. 1995, 10, 111–113. [Google Scholar] [CrossRef]
- Bu, R.; Gutierrez, M.I.; Al-Rasheed, M.; Belgaumi, A.; Bhatia, K. Variable drug metabolism genes in Arab population. Pharmacogenom. J. 2004, 4, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Toffoli, G.; Veronesi, A.; Boiocchi, M.; Crivellari, D. MTHFR gene polymorphism and severe toxicity during adjuvant treatment of early breast cancer with cyclophosphamide, methotrexate, and fluorouracil (CMF). Ann. Oncol. 2000, 11, 373–374. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.W.; Gao, C.M.; Wu, J.Z.; Sun, X.F.; Wang, L.; Feng, J.F. Relationship of methylenetetrahydrofolate reductase C677T polymorphism and chemosensitivity to 5-fluorouracil in gastric carcinoma. Ai Zheng = Aizheng = Chin. J. Cancer 2004, 23, 958–962. (In Chinese) [Google Scholar]
- Nahid, N.A.; Apu, M.N.H.; Islam, M.R.; Shabnaz, S.; Chowdhury, S.M.; Ahmed, M.U.; Nahar, Z.; Islam, M.S.; Islam, M.S.; Hasnat, A. DPYD*2A and MTHFR C677T predict toxicity and efficacy, respectively, in patients on chemotherapy with 5-fluorouracil for colorectal cancer. Cancer Chemother. Pharmacol. 2018, 81, 119–129. [Google Scholar] [CrossRef]
- Capitain, O.; Boisdron-Celle, M.; Poirier, A.L.; Abadie-Lacourtoisie, S.; Morel, A.; Gamelin, E. The influence of fluorouracil outcome parameters on tolerance and efficacy in patients with advanced colorectal cancer. Pharmacogenom. J. 2008, 8, 256–267. [Google Scholar] [CrossRef]
| Demographic Details | Patients % | |
|---|---|---|
| Gender n (%) | Male | 67.1 |
| Female | 32.9 | |
| Age | Range | 20–81 |
| Mean | 58.88 | |
| Primary Tumor Site | Colon | 67.1 |
| Rectum | 32.9 | |
| Histopathological Type | Adenocarcinoma | 91 |
| Other | 9 | |
| Stage of Tumor | I | 0 |
| II | 9.4 | |
| III | 30.6 | |
| IV | 60 |
| Toxicity Types | Grade of Toxicities, % | |||
|---|---|---|---|---|
| Grade 1 | Grade 2 | Grade 3 | Grade 4 | |
| Gastrointestinal toxicity | 5.9 | 5.9 | 1.2 | - |
| Hematopoietic toxicity | 16.5 | 9.4 | 21.2 | 7.1 |
| Hair and skin toxicity | 1.2 | - | 2.4 | - |
| Hand-foot Syndrome | 9.4 | 7.1 | 4.7 | - |
| Polymorphism | SNP no | Amino Acid Change | Genotype Frequency % | Allelic Frequency | |||
|---|---|---|---|---|---|---|---|
| wt/wt 1 | wt/mut 2 | mut/mut | wt | mut | |||
| DPYD*2A (IVS14+1G>A) | rs3918290 | Exon skipping- Exon 14 | GG (98.8) | AG (1.2) | AA (0) | 0.99 | 0.01 |
| DPYD*9A 85T>C | rs1801265 | Cys29Arg- Exon 2 | TT (63.5) | TC (29.4) | CC (7.1) | 0.78 | 0.22 |
| DPYD*13 1679T>G/T | rs55886062 | I560S- Exon 13 | AA (100) | AC (0) | CC (0) | 1 | 0 |
| DPYD 2846A>T | rs67376798 | D949V- Exon 22 | AA (100) | AT (0) | TT (0) | 1 | 0 |
| TYMS 1494 del TTAAAG | rs34489327 | 6 bp deletion-3′-UTR region | (-;-) (36.5) | (-;TTAAAG) (38.8) | (TTAAAG;TTAAAG) (24.7) | 0.56 | 0.44 |
| MTHFR 677C>T | rs1801133 | A222V- Exon 4 | CC (47.1) | CT (43.5) | TT (9.4) | 0.69 | 0.31 |
| MTHFR 1298A>C | rs1801131 | E429A- Exon 7 | AA (89.4) | AC (8.2) | CC (2.4) | 0.91 | 0.09 |
| MTHFR 677C>T | Toxicity (n) | Risk Ratio | 95% CI | p Value | ||||
|---|---|---|---|---|---|---|---|---|
| + | − | |||||||
| Het (CT) + Hom (TT) | 45 1 | 0 | 1.18 | (1.03–1.34) | 0.007 | |||
| Wild Type (CC) | 34 | 6 | ||||||
| MTHFR 1298A>C | Hematopoietic Toxicity (n) | Risk Ratio | 95% CI | p Value | ||||
|---|---|---|---|---|---|---|---|---|
| + | − | |||||||
| Het (AC) + Hom (CC) | 9 1 | 0 | 1.86 | (1.50–2.32) | 0.008 | |||
| Wild Type (AA) | 37 | 32 | ||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amirfallah, A.; Calibasi Kocal, G.; Unal, O.U.; Ellidokuz, H.; Oztop, I.; Basbinar, Y. DPYD, TYMS and MTHFR Genes Polymorphism Frequencies in a Series of Turkish Colorectal Cancer Patients. J. Pers. Med. 2018, 8, 45. https://doi.org/10.3390/jpm8040045
Amirfallah A, Calibasi Kocal G, Unal OU, Ellidokuz H, Oztop I, Basbinar Y. DPYD, TYMS and MTHFR Genes Polymorphism Frequencies in a Series of Turkish Colorectal Cancer Patients. Journal of Personalized Medicine. 2018; 8(4):45. https://doi.org/10.3390/jpm8040045
Chicago/Turabian StyleAmirfallah, Arsalan, Gizem Calibasi Kocal, Olcun Umit Unal, Hulya Ellidokuz, Ilhan Oztop, and Yasemin Basbinar. 2018. "DPYD, TYMS and MTHFR Genes Polymorphism Frequencies in a Series of Turkish Colorectal Cancer Patients" Journal of Personalized Medicine 8, no. 4: 45. https://doi.org/10.3390/jpm8040045
APA StyleAmirfallah, A., Calibasi Kocal, G., Unal, O. U., Ellidokuz, H., Oztop, I., & Basbinar, Y. (2018). DPYD, TYMS and MTHFR Genes Polymorphism Frequencies in a Series of Turkish Colorectal Cancer Patients. Journal of Personalized Medicine, 8(4), 45. https://doi.org/10.3390/jpm8040045






