Impact of Promoter Polymorphisms on the Transcriptional Regulation of the Organic Cation Transporter OCT1 (SLC22A1)
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture and Transfection
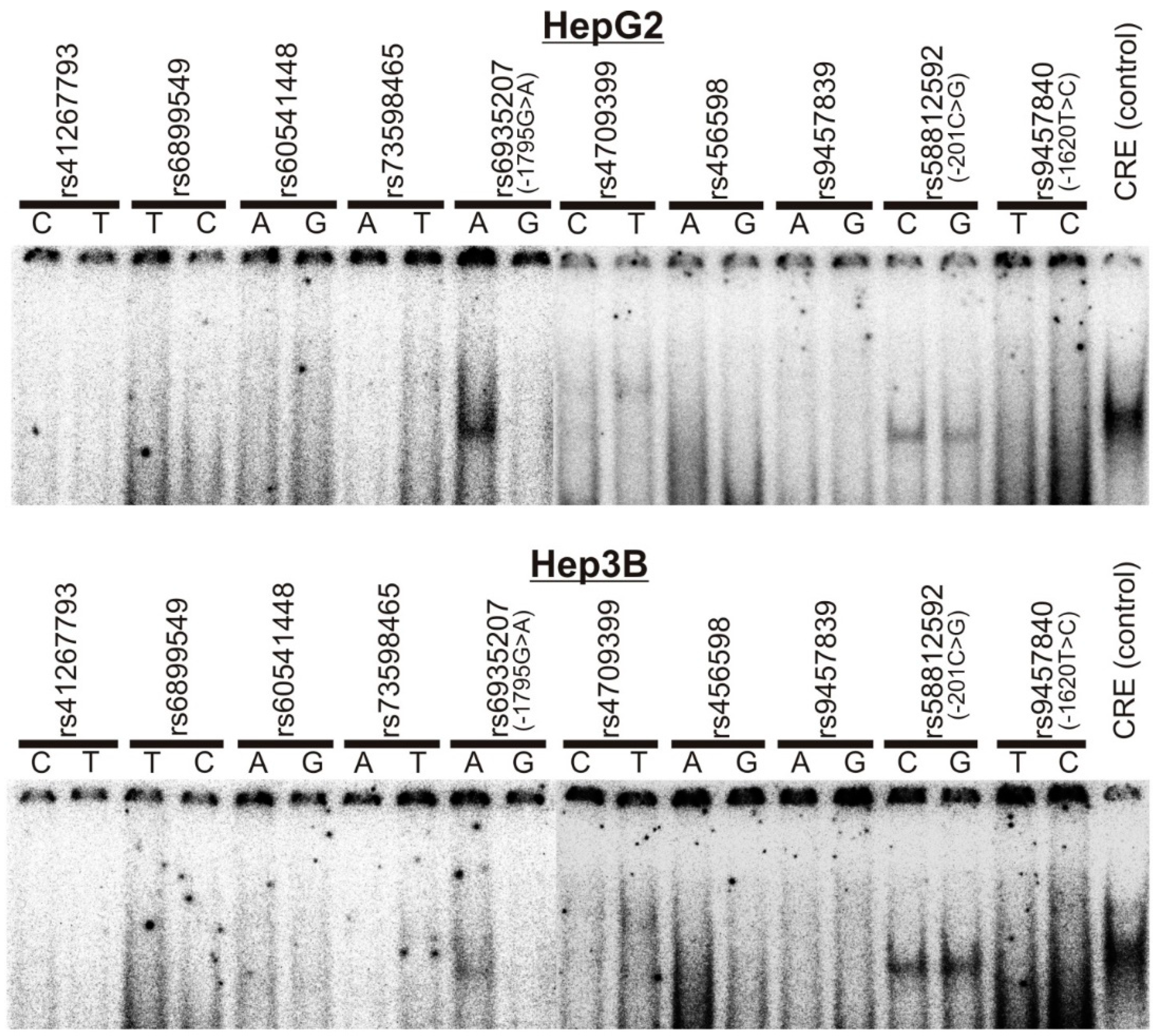
2.2. Electrophoretic Mobility Shift Assays (EMSA)
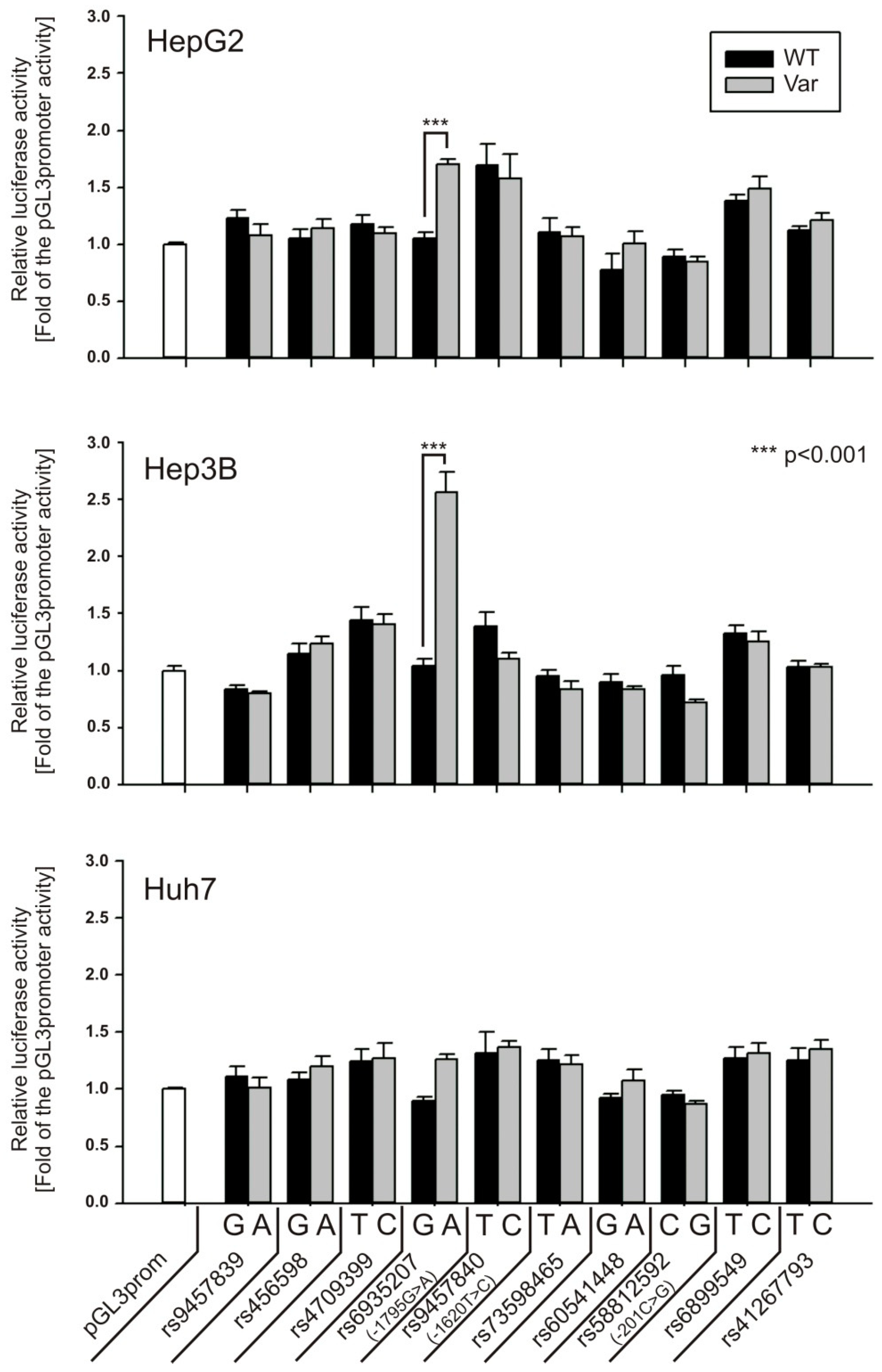
2.3. Generation of the Luciferase Reporter Plasmids
3. Results
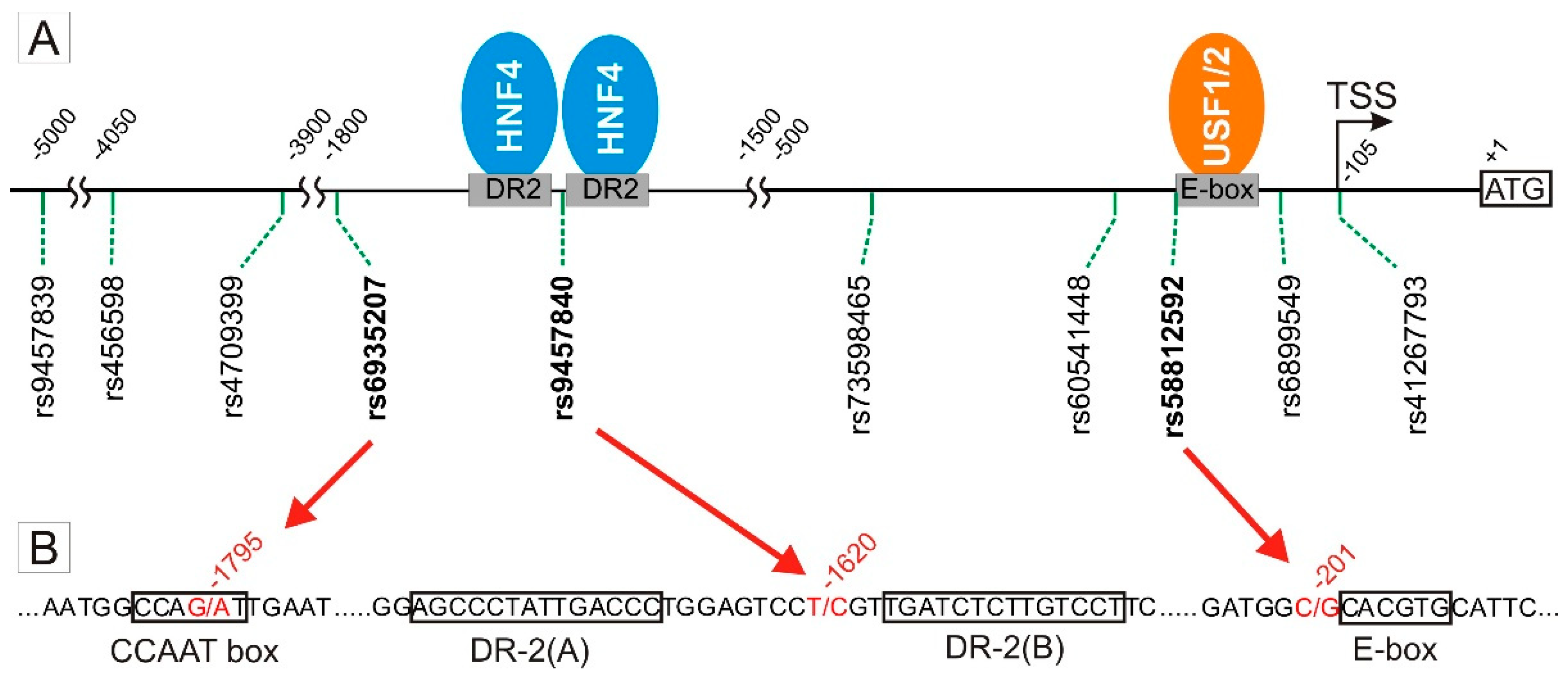
3.1. Screening of SNPs in the OCT1 Promoter for Effects on Transcription Factor Binding and Promoter Activity
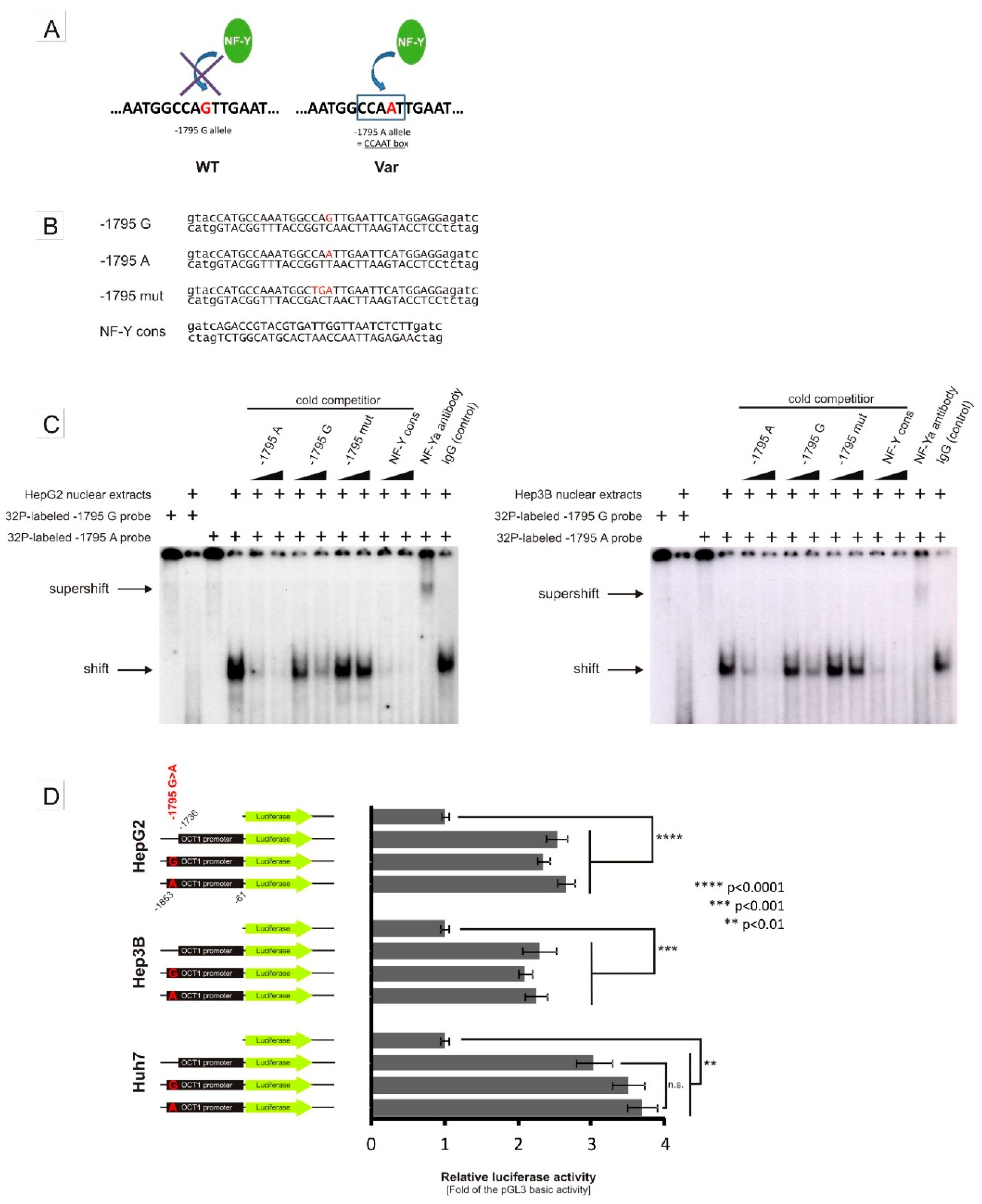
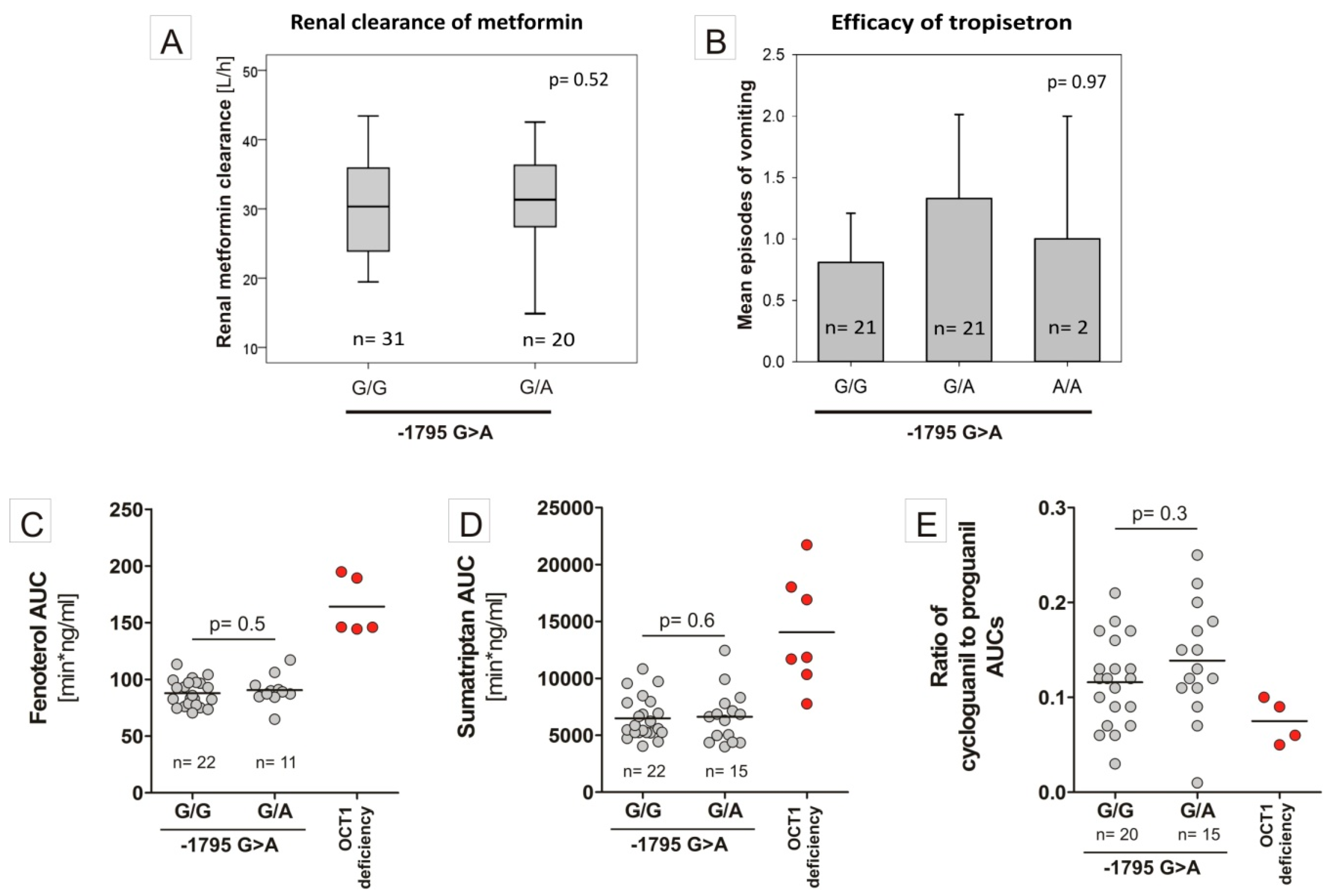
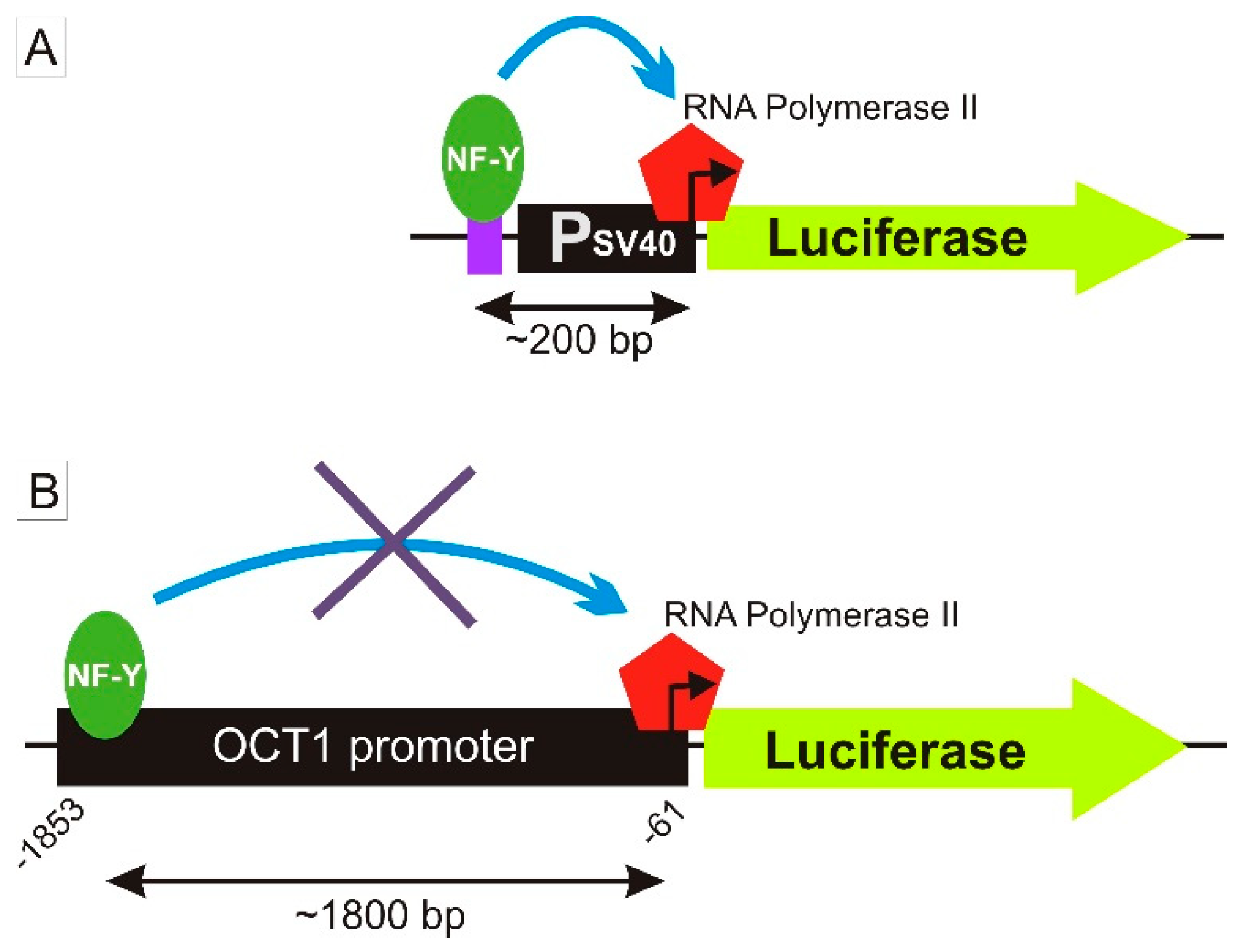
3.2. Specific Binding of NF-Y to the A-Allele of the -1795G>A (rs6935207) Polymorphism
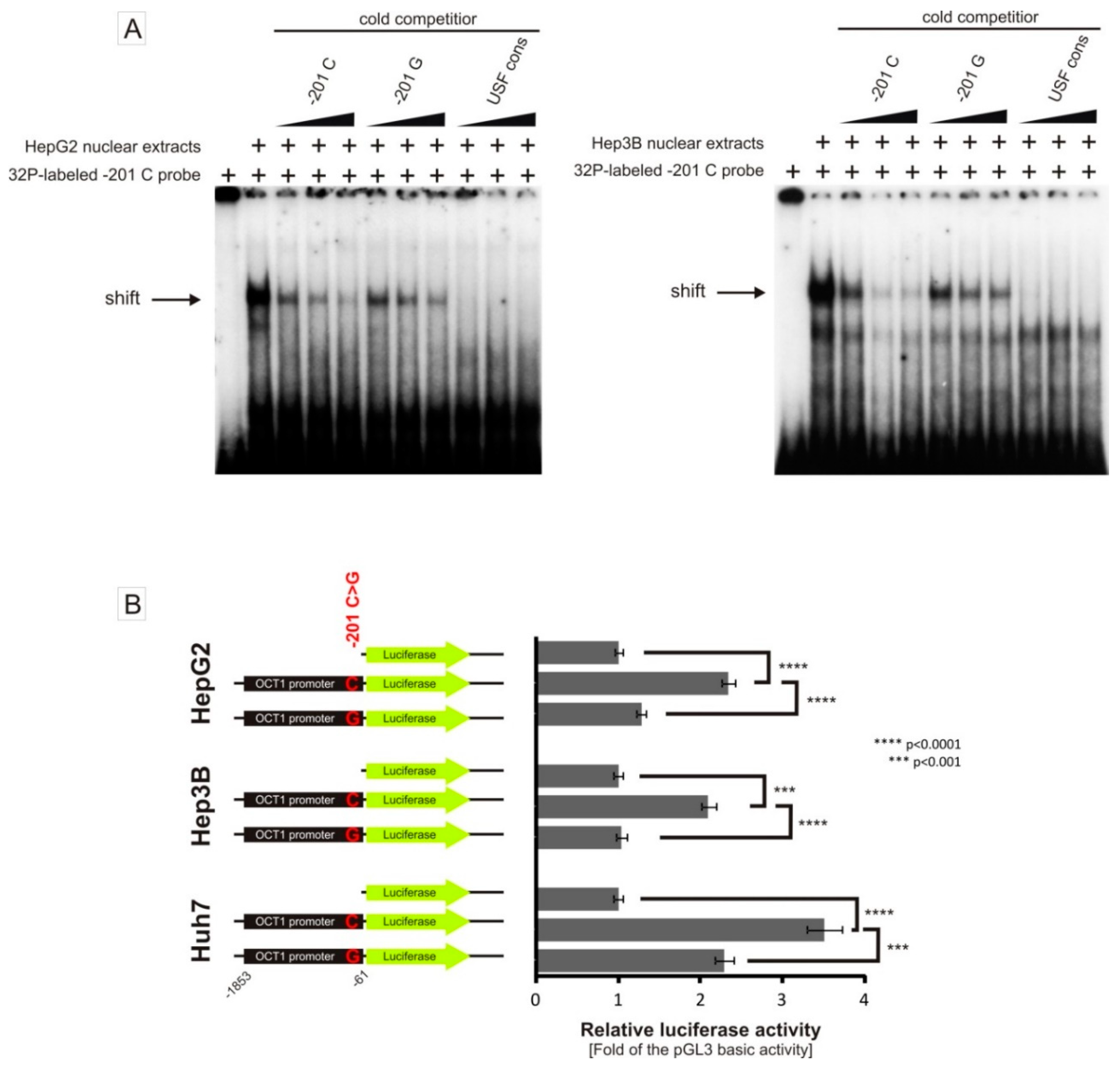
3.3. Allele-Dependent Binding of USF1/2 to the −201C>G (rs58812592) Polymorphism
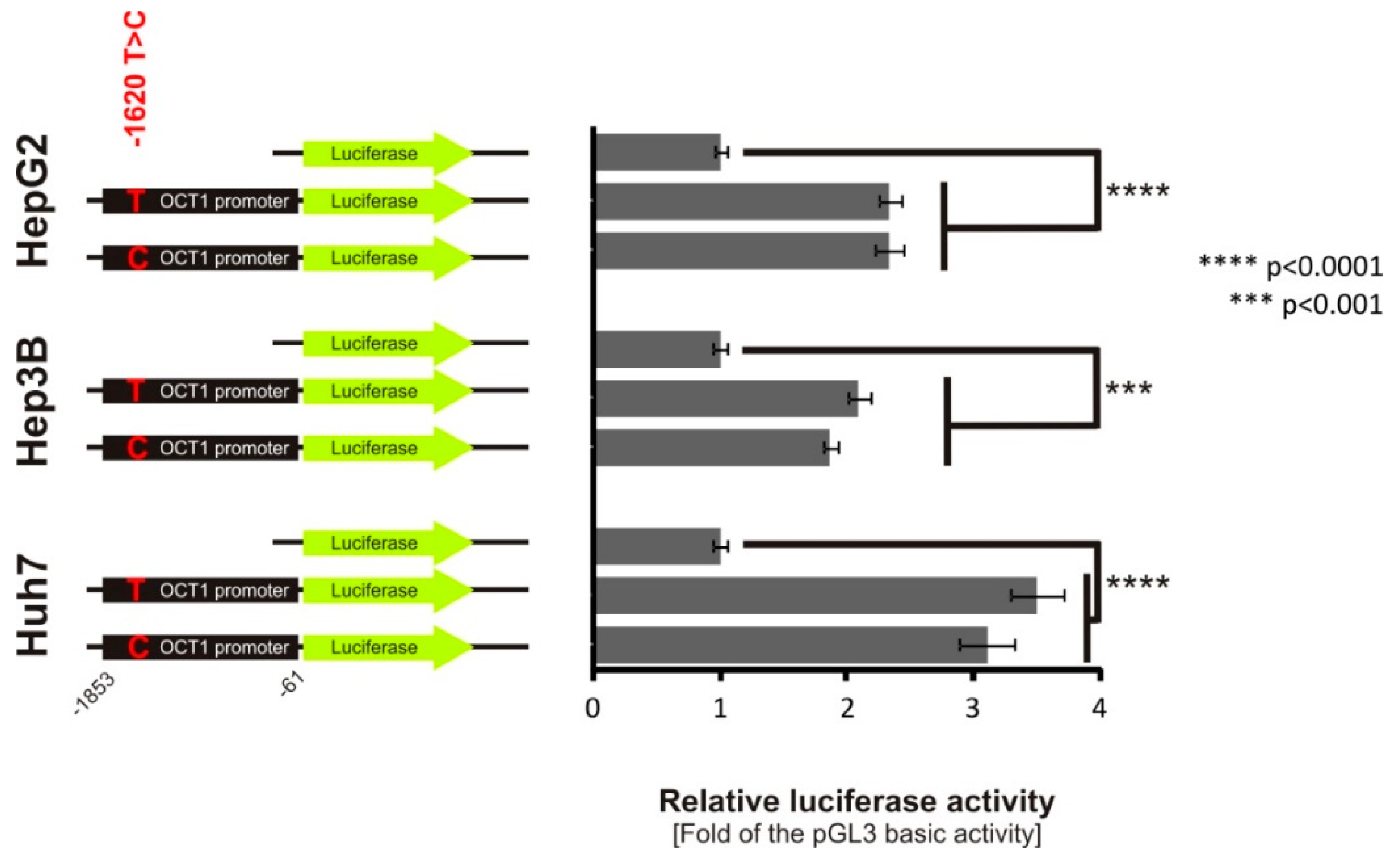
3.4. Lack of Effect of -1620T>C on HNF4α Binding and OCT1 Promoter Activity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nies, A.T.; Koepsell, H.; Winter, S.; Burk, O.; Klein, K.; Kerb, R.; Zanger, U.M.; Keppler, D.; Schwab, M.; Schaeffeler, E. Expression of organic cation transporters OCT1 (SLC22A1) and OCT3 (SLC22A3) is affected by genetic factors and cholestasis in human liver. Hepatology 2009, 50, 1227–1240. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, O.; Ohtsuki, S.; Kawakami, H.; Inoue, T.; Liehner, S.; Saito, A.; Sakamoto, A.; Ishiguro, N.; Matsumaru, T.; Terasaki, T.; et al. Absolute quantification and differential expression of drug transporters, cytochrome p450 enzymes, and UDP-glucuronosyltransferases in cultured primary human hepatocytes. Drug Metab. Dispos. 2012, 40, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Prasad, B.; Salphati, L.; Chu, X.; Gupta, A.; Hop, C.E.; Evers, R.; Unadkat, J.D. Interspecies variability in expression of hepatobiliary transporters across human, dog, monkey, and rat as determined by quantitative proteomics. Drug Metab. Dispos. 2015, 43, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Tzvetkov, M.V.; Saadatmand, A.R.; Lotsch, J.; Tegeder, I.; Stingl, J.C.; Brockmoller, J. Genetically polymorphic oct1: Another piece in the puzzle of the variable pharmacokinetics and pharmacodynamics of the opioidergic drug tramadol. Clin. Pharmacol. Ther. 2011, 90, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Tzvetkov, M.V.; Vormfelde, S.V.; Balen, D.; Meineke, I.; Schmidt, T.; Sehrt, D.; Sabolic, I.; Koepsell, H.; Brockmoller, J. The effects of genetic polymorphisms in the organic cation transporters OCT1, OCT2, and OCT3 on the renal clearance of metformin. Clin. Pharmacol. Ther. 2009, 86, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Tzvetkov, M.V.; Saadatmand, A.R.; Bokelmann, K.; Meineke, I.; Kaiser, R.; Brockmoller, J. Effects of OCT1 polymorphisms on the cellular uptake, plasma concentrations and efficacy of the 5-HT(3) antagonists tropisetron and ondansetron. Pharmacogenomics J. 2012, 12, 22–29. [Google Scholar] [CrossRef]
- Tzvetkov, M.V.; dos Santos Pereira, J.N.; Meineke, I.; Saadatmand, A.R.; Stingl, J.C.; Brockmoller, J. Morphine is a substrate of the organic cation transporter OCT1 and polymorphisms in OCT1 gene affect morphine pharmacokinetics after codeine administration. Biochem. Pharmacol. 2013, 86, 666–678. [Google Scholar] [CrossRef]
- Matthaei, J.; Kuron, D.; Faltraco, F.; Knoch, T.; Dos Santos Pereira, J.N.; Abu Abed, M.; Prukop, T.; Brockmoller, J.; Tzvetkov, M.V. OCT1 mediates hepatic uptake of sumatriptan and loss-of-function OCT1 polymorphisms affect sumatriptan pharmacokinetics. Clin. Pharmacol. Ther. 2016, 99, 633–641. [Google Scholar] [CrossRef]
- Tzvetkov, M.V.; Matthaei, J.; Pojar, S.; Faltraco, F.; Vogler, S.; Prukop, T.; Seitz, T.; Brockmoller, J. Increased systemic exposure and stronger cardiovascular and metabolic adverse reactions to fenoterol in individuals with heritable oct1 deficiency. Clin. Pharmacol. Ther. 2018, 103, 868–878. [Google Scholar] [CrossRef]
- Sundelin, E.; Gormsen, L.C.; Jensen, J.B.; Vendelbo, M.H.; Jakobsen, S.; Munk, O.L.; Christensen, M.; Brosen, K.; Frokiaer, J.; Jessen, N. Genetic polymorphisms in organic cation transporter 1 attenuates hepatic metformin exposure in humans. Clin. Pharmacol. Ther. 2017, 102, 841–848. [Google Scholar] [CrossRef]
- Matthaei, J.; Seitz, T.; Jensen, O.; Tann, A.; Prukop, T.; Tadjerpisheh, S.; Brockmoller, J.; Tzvetkov, M.V. Oct1 deficiency affects hepatocellular concentrations and pharmacokinetics of cycloguanil, the active metabolite of the antimalarial drug proguanil. Clin. Pharmacol. Ther. 2018. [Google Scholar] [CrossRef] [PubMed]
- Seitz, T.; Stalmann, R.; Dalila, N.; Chen, J.; Pojar, S.; Dos Santos Pereira, J.N.; Kratzner, R.; Brockmoller, J.; Tzvetkov, M.V. Global genetic analyses reveal strong inter-ethnic variability in the loss of activity of the organic cation transporter oct1. Genome Med. 2015, 7, 56. [Google Scholar] [CrossRef] [PubMed]
- Saadatmand, A.R.; Tadjerpisheh, S.; Brockmoller, J.; Tzvetkov, M.V. The prototypic pharmacogenetic drug debrisoquine is a substrate of the genetically polymorphic organic cation transporter OCT1. Biochem. Pharmacol. 2012, 83, 1427–1434. [Google Scholar] [CrossRef] [PubMed]
- Shu, Y.; Sheardown, S.A.; Brown, C.; Owen, R.P.; Zhang, S.; Castro, R.A.; Ianculescu, A.G.; Yue, L.; Lo, J.C.; Burchard, E.G.; et al. Effect of genetic variation in the organic cation transporter 1 (OCT1) on metformin action. J. Clin. Investig. 2007, 117, 1422–1431. [Google Scholar] [CrossRef] [PubMed]
- Arimany-Nardi, C.; Minuesa, G.; Keller, T.; Erkizia, I.; Koepsell, H.; Martinez-Picado, J.; Pastor-Anglada, M. Role of human organic cation transporter 1 (hOCT1) polymorphisms in lamivudine (3TC) uptake and drug-drug interactions. Front. Pharmacol. 2016, 7, 175. [Google Scholar] [CrossRef] [PubMed]
- Arimany-Nardi, C.; Montraveta, A.; Lee-Verges, E.; Puente, X.S.; Koepsell, H.; Campo, E.; Colomer, D.; Pastor-Anglada, M. Human organic cation transporter 1 (hOCT1) as a mediator of bendamustine uptake and cytotoxicity in chronic lymphocytic leukemia (CLL) cells. Pharmacogenomics J. 2015, 15, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Zamek-Gliszczynski, M.J.; Giacomini, K.M.; Zhang, L. Emerging clinical importance of hepatic organic cation transporter 1 (OCT1) in drug pharmacokinetics, dynamics, pharmacogenetic variability, and drug interactions. Clin. Pharmacol. Ther. 2018, 103, 758–760. [Google Scholar] [CrossRef] [PubMed]
- Tzvetkov, M.V. OCT1 pharmacogenetics in pain management: Is a clinical application within reach? Pharmacogenomics 2017, 18, 1515–1523. [Google Scholar] [CrossRef]
- Yee, S.W.; Brackman, D.J.; Ennis, E.A.; Sugiyama, Y.; Kamdem, L.K.; Blanchard, R.; Galetin, A.; Zhang, L.; Giacomini, K.M. Influence of transporter polymorphisms on drug disposition and response: A perspective from the international transporter consortium. Clin. Pharmacol. Ther. 2018. [Google Scholar] [CrossRef]
- Nies, A.T.; Schaeffeler, E.; van der Kuip, H.; Cascorbi, I.; Bruhn, O.; Kneba, M.; Pott, C.; Hofmann, U.; Volk, C.; Hu, S.; et al. Cellular uptake of imatinib into leukemic cells is independent of human organic cation transporter 1 (OCT1). Clin Cancer Res. 2014, 20, 985–994. [Google Scholar] [CrossRef]
- Tzvetkov, M.V.; Seitz, T.; Bokelmann, K.; Mueller, T.; Brockmoller, J.; Koepsell, H. Does the haplotype Met408-Del420, which was apparently predictive for imatinib efficacy, really exist and how strongly may it affect oct1 activity? Blood 2014, 123, 1427–1429. [Google Scholar] [CrossRef] [PubMed]
- Neul, C.; Baker, S.D.; Sparreboom, A.; Schaeffeler, E.; Laufer, S.; Schwab, M.; Nies, A.T. Evaluation of organic cation transporter 1 (OCT1, SLC22A1) as transporter for sorafenib. In Proceedings of the AACR 107th Annual Meeting 2016, New Orleans, LA, USA, 16–20 April 2016. [Google Scholar]
- Neul, C.; Schaeffeler, E.; Laufer, S.; Schwab, M.; Nies, A.T. Cellular uptake of sorafenib is independent of major human organic cation and organic anion uptake transporters of the hepatocyte. In Proceedings of the 17th Jahreskongress für Klinische Pharmakologie, Cologne, Germany, 1–2 October 2015. [Google Scholar]
- O’Brien, V.P.; Bokelmann, K.; Ramirez, J.; Jobst, K.; Ratain, M.J.; Brockmoller, J.; Tzvetkov, M.V. Hepatocyte nuclear factor 1 regulates the expression of the organic cation transporter 1 via binding to an evolutionary conserved region in intron 1 of the oct1 gene. J. Pharmacol. Exp. Ther. 2013, 347, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Fattah, S.; Shinde, A.B.; Matic, M.; Baes, M.; van Schaik, R.H.N.; Allegaert, K.; Parmentier, C.; Richert, L.; Augustijns, P.; Annaert, P. Inter-subject variability in OCT1 activity in 27 batches of cryopreserved human hepatocytes and association with oct1 mrna expression and genotype. Pharm. Res. 2017, 34, 1309–1319. [Google Scholar] [CrossRef] [PubMed]
- Schaeffeler, E.; Hellerbrand, C.; Nies, A.T.; Winter, S.; Kruck, S.; Hofmann, U.; van der Kuip, H.; Zanger, U.M.; Koepsell, H.; Schwab, M. DNA methylation is associated with downregulation of the organic cation transporter OCT1 (SLC22A1) in human hepatocellular carcinoma. Genome Med. 2011, 3, 82. [Google Scholar] [CrossRef] [PubMed]
- Hesselson, S.E.; Matsson, P.; Shima, J.E.; Fukushima, H.; Yee, S.W.; Kobayashi, Y.; Gow, J.M.; Ha, C.; Ma, B.; Poon, A.; et al. Genetic variation in the proximal promoter of ABC and SLC superfamilies: Liver and kidney specific expression and promoter activity predict variation. PLoS ONE 2009, 4, e6942. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Hong, C.; Chen, E.C.; Yee, S.W.; Xu, L.; Almof, E.U.; Wen, C.; Fujii, K.; Johns, S.J.; Stryke, D.; et al. Genetic and epigenetic regulation of the organic cation transporter 3, SLC22A3. Pharmacogenomics J. 2013, 13, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, K.; Terada, T.; Motohashi, H.; Asaka, J.; Aoki, M.; Katsura, T.; Kamba, T.; Ogawa, O.; Inui, K. Analysis of regulatory polymorphisms in organic ion transporter genes (SLC22A) in the kidney. J. Hum. Genet. 2008, 53, 607–614. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.Y.; Cho, S.K.; Kim, T.H.; Kim, K.H.; Jang, G.H.; Kim, C.O.; Park, E.M.; Cho, J.Y.; Jang, I.J.; Choi, J.H. Functional characterization of MATE2-K genetic variants and their effects on metformin pharmacokinetics. Pharmacogenet. Genom. 2013, 23, 365–373. [Google Scholar] [CrossRef]
- Choi, J.H.; Yee, S.W.; Ramirez, A.H.; Morrissey, K.M.; Jang, G.H.; Joski, P.J.; Mefford, J.A.; Hesselson, S.E.; Schlessinger, A.; Jenkins, G.; et al. A common 5′-UTR variant in MATE2-K is associated with poor response to metformin. Clin. Pharmacol. Ther. 2011, 90, 674–684. [Google Scholar] [CrossRef]
- Ha Choi, J.; Wah Yee, S.; Kim, M.J.; Nguyen, L.; Ho Lee, J.; Kang, J.O.; Hesselson, S.; Castro, R.A.; Stryke, D.; Johns, S.J.; et al. Identification and characterization of novel polymorphisms in the basal promoter of the human transporter, mate1. Pharmacogenet. Genom. 2009, 19, 770–780. [Google Scholar] [CrossRef]
- Kajiwara, M.; Terada, T.; Asaka, J.; Aoki, M.; Katsura, T.; Ikai, I.; Inui, K. Regulation of basal core promoter activity of human organic cation transporter 1 (OCT1/SLC22A1). Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 295, G1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Saborowski, M.; Kullak-Ublick, G.A.; Eloranta, J.J. The human organic cation transporter-1 gene is transactivated by hepatocyte nuclear factor-4aα. J. Pharmacol. Exp. Ther. 2006, 317, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Tahara, H.; Yee, S.W.; Urban, T.J.; Hesselson, S.; Castro, R.A.; Kawamoto, M.; Stryke, D.; Johns, S.J.; Ferrin, T.E.; Kwok, P.Y.; et al. Functional genetic variation in the basal promoter of the organic cation/carnitine transporters OCTN1 (SLC22A4) and OCTN2 (SLC22A5). J. Pharmacol. Exp. Ther. 2009, 329, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, B.; Li, Y.; Chen, J.; Lou, G.; Chen, M.; Zhou, D. Transcriptional regulation of the human PNRC promoter by nfy in HEPG2 cells. J. Biochem. 2008, 143, 675–683. [Google Scholar] [CrossRef] [PubMed]
- Novak, E.M.; Bydlowski, S.P. NFY transcription factor binds to regulatory element AIC and transactivates the human apolipoprotein A-I promoter in HEPG2 cells. Biochem. Biophys. Res. Commun. 1997, 231, 140–143. [Google Scholar] [CrossRef] [PubMed]
- Pallai, R.; Simpkins, H.; Chen, J.; Parekh, H.K. The CCAAT box binding transcription factor, nuclear factor-Y (NF-Y) regulates transcription of human aldo-keto reductase 1C1 (AKR1C1) gene. Gene 2010, 459, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, Y.; Chen, J.C.; Hsu, L.C.; Yoshida, A. The transcriptional regulation of human aldehyde dehydrogenase I gene. The structural and functional analysis of the promoter. J. Biol. Chem. 1995, 270, 17521–17527. [Google Scholar] [CrossRef]
- Ueda, A.; Yoshimura, T. Characterization of cis-acting elements of the gene for macrophage-stimulating protein from the human. The involvement of positive and negative regulatory elements. J. Biol. Chem. 1996, 271, 20265–20272. [Google Scholar] [CrossRef]
- Christensen, M.M.; Brasch-Andersen, C.; Green, H.; Nielsen, F.; Damkier, P.; Beck-Nielsen, H.; Brosen, K. The pharmacogenetics of metformin and its impact on plasma metformin steady-state levels and glycosylated hemoglobin A1c. Pharmacogenet. Genom. 2011, 21, 837–850. [Google Scholar] [CrossRef]
- Kim, M.H.; Shin, H.J.; Lim, S.J.; Park, J.S.; Lee, S.S.; Song, I.S.; Shin, J.G. Inter-individual variability in OCT1 expression and its relationship with OCT1 genotype in liver samples from a korean population. Drug Metab. Pharmacokinet. 2012, 27, 530–535. [Google Scholar] [CrossRef]
- Maffioli, M.; Camos, M.; Gaya, A.; Hernandez-Boluda, J.C.; Alvarez-Larran, A.; Domingo, A.; Granell, M.; Guillem, V.; Vallansot, R.; Costa, D.; et al. Correlation between genetic polymorphisms of the hOCT1 and mdr1 genes and the response to imatinib in patients newly diagnosed with chronic-phase chronic myeloid leukemia. Leuk. Res. 2011, 35, 1014–1019. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Jimenez, C.P.; Gomez-Lechon, M.J.; Castell, J.V.; Jover, R. Underexpressed coactivators PGC1α and SRC1 impair hepatocyte nuclear factor 4α function and promote dedifferentiation in human hepatoma cells. J. Biol. Chem. 2006, 281, 29840–29849. [Google Scholar] [CrossRef] [PubMed]
- Hyrsova, L.; Smutny, T.; Carazo, A.; Moravcik, S.; Mandikova, J.; Trejtnar, F.; Gerbal-Chaloin, S.; Pavek, P. The pregnane x receptor down-regulates organic cation transporter 1 (SLC22A1) in human hepatocytes by competing for (“squelching”) SRC-1 coactivator. Br. J. Pharmacol. 2016, 173, 1703–1715. [Google Scholar] [CrossRef] [PubMed]
- Rulcova, A.; Krausova, L.; Smutny, T.; Vrzal, R.; Dvorak, Z.; Jover, R.; Pavek, P. Glucocorticoid receptor regulates organic cation transporter 1 (OCT1, SLC22A1) expression via hnf4alpha upregulation in primary human hepatocytes. Pharmacol. Rep. 2013, 65, 1322–1335. [Google Scholar] [CrossRef]
- Hyrsova, L.; Smutny, T.; Trejtnar, F.; Pavek, P. Expression of organic cation transporter 1 (OCT1): Unique patterns of indirect regulation by nuclear receptors and hepatospecific gene regulation. Drug Metab. Rev. 2016, 48, 139–158. [Google Scholar] [CrossRef] [PubMed]
| Name | Direction | Sequence |
|---|---|---|
| EMSA Probes * | ||
| rs41267793_T_f | Forward | 5′-ACTTGGACAGCAAACTGATTTCAAACCACTCa-3′ |
| rs41267793_T_r | Reverse | 5′-gatctGAGTGGTTTGAAATCAGTTTGCTGTCCAAGTgtac-3′ |
| rs41267793_C_f | Forward | 5′-ACTTGGACAGCAAACCGATTTCAAACCACTCa-3′ |
| rs41267793_C_r | Reverse | 5′-gatctGAGTGGTTTGAAATCGGTTTGCTGTCCAAGTgtac-3′ |
| rs6899549_T_f | Forward | 5′-AGGGTAAAAGATTATTTCTACTTGGTTGCCTa-3′ |
| rs6899549_T_r | Reverse | 5′-gatctAGGCAACCAAGTAGAAATAATCTTTTACCCTgtac-3′ |
| rs6899549_C_f | Forward | 5′-AGGGTAAAAGATTATCTCTACTTGGTTGCCTa-3′ |
| rs6899549_C_r | Reverse | 5′-gatctAGGCAACCAAGTAGAGATAATCTTTTACCCTgtac-3′ |
| rs58812592_C_f | Forward | 5′-TTGATCAGATGGCCACGTGCATTCTTCCTTTa-3′ |
| rs58812592_C_r | Reverse | 5′-gatctAAAGGAAGAATGCACGTGGCCATCTGATCAAgtac-3′ |
| rs58812592_G_f | Forward | 5′-TTGATCAGATGGGCACGTGCATTCTTCCTTTa-3′ |
| rs58812592_G_r | Reverse | 5′-gatctAAAGGAAGAATGCACGTGCCCATCTGATCAAgtac-3′ |
| rs60541448_G_f | Forward | 5′-CACTGACTCGCTCCCGGGCAAAGCAAACGATa-3′ |
| rs60541448_G_r | Reverse | 5′-gatctATCGTTTGCTTTGCCCGGGAGCGAGTCAGTGgtac-3′ |
| rs60541448_A_f | Forward | 5′-CACTGACTCGCTCCCAGGCAAAGCAAACGATa-3′ |
| rs60541448_A_r | Reverse | 5′-gatctATCGTTTGCTTTGCCTGGGAGCGAGTCAGTGgtac-3′ |
| rs73598465_T_f | Forward | 5′-TATCACAGAACTAATTAGCCGAATACAGTATa-3′ |
| rs73598465_T_r | Reverse | 5′-gatctATACTGTATTCGGCTAATTAGTTCTGTGATAgtac-3′ |
| rs73598465_A_f | Forward | 5′-TATCACAGAACTAATAAGCCGAATACAGTATa-3′ |
| rs73598465_A_r | Reverse | 5′-gatctATACTGTATTCGGCTTATTAGTTCTGTGATAgtac-3′ |
| rs9457840_T_f | Forward | 5′-GGAGCCCTATTGACCCTGGAGTCCTGTTGATCTCTTGTCCTTCa-3′ |
| rs9457840_T_r | Reverse | 5′-gatctGAAGGACAAGAGATCAACAGGACTCCAGGGTCAATAGGGCTCCgtac-3′ |
| rs9457840_C_f | Forward | 5′-GGAGCCCTATTGACCCTGGAGTCCCGTTGATCTCTTGTCCTTCa-3′ |
| rs9457840_C_r | Reverse | 5′-gatctGAAGGACAAGAGATCAACGGGACTCCAGGGTCAATAGGGCTCCgtac-3′ |
| rs6935207_G_f | Forward | 5′-CATGCCAAATGGCCAGTTGAATTCATGGAGGa-3′ |
| rs6935207_G_r | Reverse | 5′-gatctCCTCCATGAATTCAACTGGCCATTTGGCATGgtac-3′ |
| rs6935207_A_f | Forward | 5′-CATGCCAAATGGCCAATTGAATTCATGGAGGa-3′ |
| rs6935207_A_r | Reverse | 5′-gatctCCTCCATGAATTCAATTGGCCATTTGGCATGgtac-3′ |
| rs6935207_mut_f | Forward | 5′-CATGCCAAATGGCTGATTGAATTCATGGAGGa-3′ |
| rs6935207_mut_r | Reverse | 5′-gatctCCTCCATGAATTCAATCAGCCATTTGGCATGgtac-3′ |
| rs4709399_T_f | Forward | 5′-AAACCTCTGTGGTCATGGTGCCTTTGCATGAa-3′ |
| rs4709399_T_r | Reverse | 5′-gatctTCATGCAAAGGCACCATGACCACAGAGGTTTgtac-3′ |
| rs4709399_C_f | Forward | 5′-AAACCTCTGTGGTCACGGTGCCTTTGCATGAa-3′ |
| rs4709399_C_r | Reverse | 5′-gatctTCATGCAAAGGCACCGTGACCACAGAGGTTTgtac-3′ |
| rs456598_G_f | Forward | 5′-GTAAGGCACTTTTTGGATGGTAGGACTGGTTa-3′ |
| rs456598_G_r | Reverse | 5′-gatctAACCAGTCCTACCATCCAAAAAGTGCCTTACgtac-3′ |
| rs456598_A_f | Forward | 5′-GTAAGGCACTTTTTGAATGGTAGGACTGGTTa-3′ |
| rs456598_A_r | Reverse | 5′-gatctAACCAGTCCTACCATTCAAAAAGTGCCTTACgtac-3′ |
| rs9457839_G_f | Forward | 5′-CCACATGCACTCCTAGGTCTGAAAATGGGGGa-3′ |
| rs9457839_G_r | Reverse | 5′-gatctCCCCCATTTTCAGACCTAGGAGTGCATGTGGgtac-3′ |
| rs9457839_A_f | Forward | 5′-CCACATGCACTCCTAAGTCTGAAAATGGGGGa-3′ |
| rs9457839_A_r | Reverse | 5′-gatctCCCCCATTTTCAGACTTAGGAGTGCATGTGGgtac-3′ |
| GS_NF-Y_f | Forward | 5′-GATCAGACCGTACGTGATTGGTTAATCTCTT-3′ |
| GS_NF-Y_r | Reverse | 5′-GATCAAGAGATTAACCAATCACGTACGGTCT-3′ |
| CRE2_f | Forward | 5′-actggTCCTTGGCTGACGTCAGAGAGAGAG-3′ |
| CRE_r | Reverse | 5′-taCTCTCTCTCTGACGTCAGCCAAGGAgg-3′ |
| Primers used for site-directed mutagenesis | ||
| 588_mut_f | Forward | 5′-GATTTGATCAGATGGGCACGTGCATTCTTCC-3′ |
| 588_mut_r | Reverse | 5′-GGAAGAATGCACGTGCCCATCTGATCAAATC-3′ |
| 840_mut_f | Forward | 5′-TTGACCCTGGAGTCCCGTTGATCTCTTGTCC-3′ |
| 840_mut_r | Reverse | 5′-GGACAAGAGATCAACGGGACTCCAGGGTCAA-3′ |
| Primers used for Sanger sequencing | ||
| Seq_840_f | Forward | 5′-TGCAACCAGTTTGCACAGAGAG-3′ |
| Seq_588_f | Forward | 5′-GCCTCATACCATCACATCTAGA-3′ |
| Seq_pGL3prom_f | Forward | 5′-GAATCGATAGTACTAACATA-3′ |
| Seq_SV40_r | Reverse | 5′-AAGCCTCCTCACTACTTCTG-3′ |
| Primers used for generation of the reporter gene construct | ||
| OCT1-1853bp_promoter_f | Forward | 5′-ATCGCGGTACCTTTTTAAGAAGTCCTTTTAAGT-3′ |
| OCT1-1853bp_promoter_r | Reverse | 5′-AAGAAGGGAAGGACAAGAGATCAAC-3′ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bokelmann, K.; Brockmöller, J.; Tzvetkov, M.V. Impact of Promoter Polymorphisms on the Transcriptional Regulation of the Organic Cation Transporter OCT1 (SLC22A1). J. Pers. Med. 2018, 8, 42. https://doi.org/10.3390/jpm8040042
Bokelmann K, Brockmöller J, Tzvetkov MV. Impact of Promoter Polymorphisms on the Transcriptional Regulation of the Organic Cation Transporter OCT1 (SLC22A1). Journal of Personalized Medicine. 2018; 8(4):42. https://doi.org/10.3390/jpm8040042
Chicago/Turabian StyleBokelmann, Kristin, Jürgen Brockmöller, and Mladen V. Tzvetkov. 2018. "Impact of Promoter Polymorphisms on the Transcriptional Regulation of the Organic Cation Transporter OCT1 (SLC22A1)" Journal of Personalized Medicine 8, no. 4: 42. https://doi.org/10.3390/jpm8040042
APA StyleBokelmann, K., Brockmöller, J., & Tzvetkov, M. V. (2018). Impact of Promoter Polymorphisms on the Transcriptional Regulation of the Organic Cation Transporter OCT1 (SLC22A1). Journal of Personalized Medicine, 8(4), 42. https://doi.org/10.3390/jpm8040042





