CmPn/CmP Signaling Networks in the Maintenance of the Blood Vessel Barrier
Abstract
1. Introduction
2. Key Factors Influencing Blood Vessel Permeability
2.1. EC Junctions
2.2. Inflammation and BBB Integrity
2.3. Angiogenic Factors and BBB Integrity
3. Angiogenic Impacts of Steroids on Maintenance of Vasculature
3.1. Steroids Can Influence the BBB Integrity
3.2. Steroids Might Play Major Roles for Compromised BBB
3.3. Steroids as Potential Therapeutics
4. Impacts of Sex Steroids on Maintenance of Vasculature
4.1. Neovascularization
4.2. Downstream Angiogenic Factors of PRG Signaling
5. Angiogenic Impacts of PRG on the Neurovascular Unit (NVU)
5.1. Neuroprotective Impact of PRGs on the NVU
5.2. Neuroprotective Effects of PRG and Its Derivatives on the BBB against Thrombin and Matrix Metalloproteinase (MMP)
5.3. Neuroprotective Effects of PRG on BBB against Inflammatory Pathway
5.4. Actions of PRG and Its Derivatives as Neurosteroids
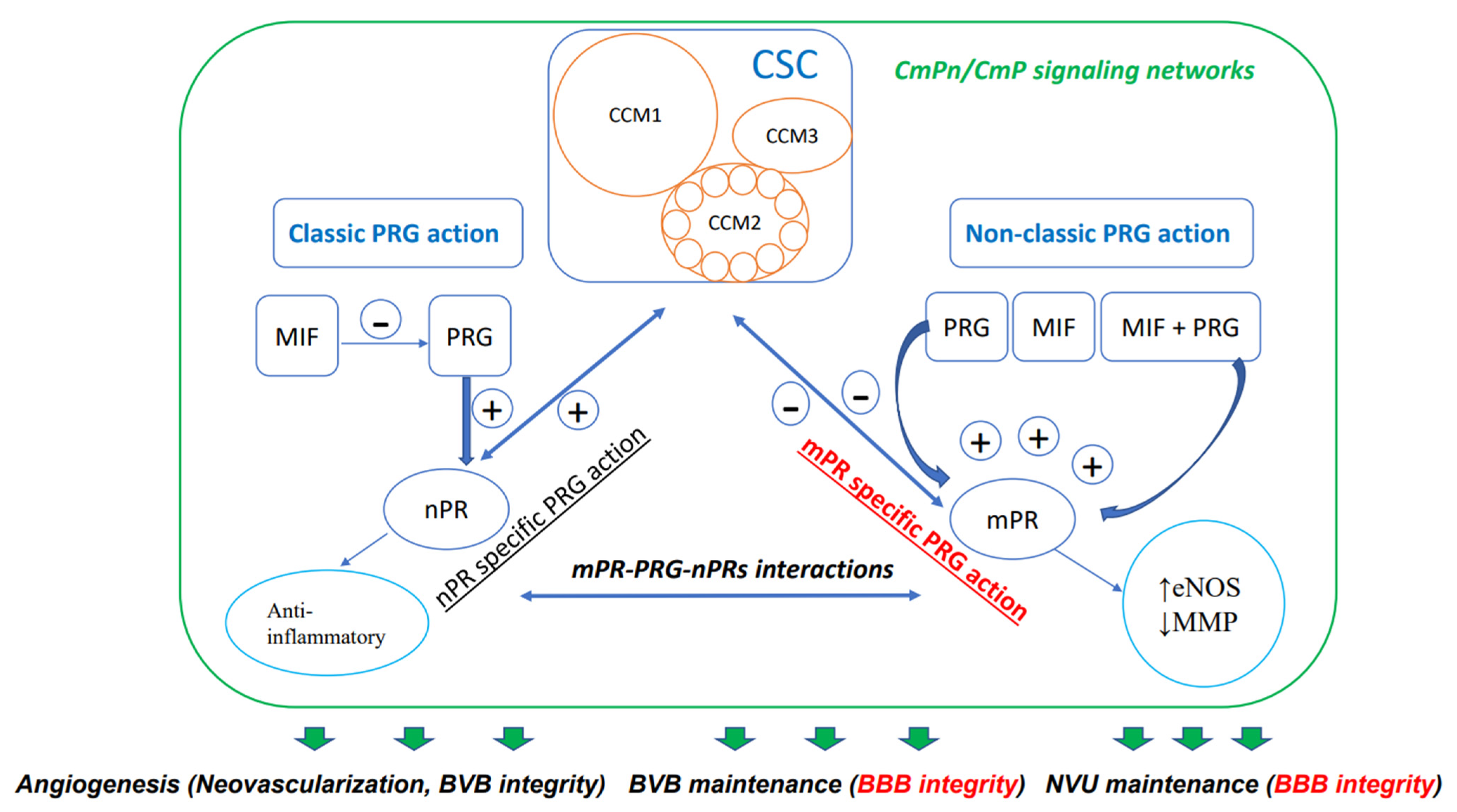
6. Impacts of CmPn/CmP Networks on the BBB
6.1. The Impact of PRG-Mediated Signaling on Endothelial Cell (EC) Function in the Vasculature
6.2. CmPn/CmP Signaling Ntworks Maintain the BBB Integrity
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 5α-DHP | 5α-dihydroprogestrone |
| AJs | adherens junctions |
| ANG | angiopoietin |
| AP | 3,5-tetrahydroxyprogesterone |
| BBB | blood–brain barrier |
| BDNF | brain-derived neurotrophic factor |
| bFGF | basic fibroblast growth factor |
| CCM | cerebral cavernous malformations |
| CmP | CSC-mPRs-PRG |
| CmPn | CSC-mPRs-PRG-nPRs |
| CNS | central nervous system |
| CSC | CCM signaling complex |
| CSF | cerebrospinal fluid |
| EC | endothelial cells |
| PCs | pericytes |
| BMs | basement membranes |
| AE | astrocyte endfeet |
| eNOS | endothelial nitric oxide synthase |
| EST | estrogen |
| fCCM | familial CCM |
| GC | glucocorticoid |
| GRs | glucocorticoid receptors |
| HIF1a | Hypoxia-inducible factor 1a |
| HRT | hormone replacement therapy |
| HUVEC | human umbilical vein endothelial cells |
| ICAM-1 | intercellular cell adhesion molecule 1 |
| JAMs | junction adhesion molecules |
| KO | knockout |
| LPS | lipopolysaccharide |
| MAPKs | mitogen-activated protein kinases |
| MEFs | mouse embryonic fibroblasts |
| MIF | mifepristone |
| MMPs | matrix metalloproteinases |
| mPRs | membrane PRG receptors |
| MR | mineralocorticoid receptor |
| MRI | magnetic resonance imaging |
| NF-κB | nuclear factor kappa B |
| NFAT | nuclear factor of activated T cells |
| NO | nitric oxide |
| NOS | nitric oxide synthase |
| NVU | neurovascular unit |
| nPR–s | nuclear PRG receptors |
| PD-ECGF | platelet-derived EC growth factor |
| PI3Ks | phosphoinositide 3-kinases |
| PRG | progesterone |
| PGRMC1,2 | progesterone receptor membrane component 1,2 |
| siRNA | short interfering RNA |
| Th | T helper |
| TJs | tight junctions |
| TBI | post-traumatic brain injury |
| VCAM-1 | vascular cell adhesion molecule 1 |
| VEGF | vascular endothelial growth factor |
| WT | wildtype |
References
- Padarti, A.; Zhang, J. Recent advances in cerebral cavernous malformation research. Vessel Plus 2018, 2, 21. [Google Scholar] [CrossRef]
- Zhang, J. Molecular biology of cerebral cavernous malformation. In Cavernous Malformations of the Nervous System; Rigamonti, D., Ed.; Cambridge University Press: Cambridge, UK, 2011; pp. 31–40. [Google Scholar] [CrossRef]
- Zhang, J.; Clatterbuck, R.E.; Rigamonti, D.; Dietz, H.C. Cloning of the murine Krit1 cDNA reveals novel mammalian 5′ coding exons. Genomics 2000, 70, 392–395. [Google Scholar] [CrossRef]
- Laberge-le Couteulx, S.; Jung, H.H.; Labauge, P.; Houtteville, J.P.; Lescoat, C.; Cecillon, M.; Marechal, E.; Joutel, A.; Bach, J.F.; Tournier-Lasserve, E. Truncating mutations in CCM1, encoding KRIT1, cause hereditary cavernous angiomas. Nat. Genet. 1999, 23, 189–193. [Google Scholar] [CrossRef]
- Jiang, X.; Padarti, A.; Qu, Y.; Sheng, S.; Abou-Fadel, J.; Badr, A.; Zhang, J. Alternatively spliced isoforms reveal a novel type of PTB domain in CCM2 protein. Sci. Rep. 2019, 9, 15808. [Google Scholar] [CrossRef]
- Liquori, C.L.; Berg, M.J.; Siegel, A.M.; Huang, E.; Zawistowski, J.S.; Stoffer, T.; Verlaan, D.; Balogun, F.; Hughes, L.; Leedom, T.P.; et al. Mutations in a gene encoding a novel protein containing a phosphotyrosine-binding domain cause type 2 cerebral cavernous malformations. Am. J. Hum. Genet. 2003, 73, 1459–1464. [Google Scholar] [CrossRef]
- Bergametti, F.; Denier, C.; Labauge, P.; Arnoult, M.; Boetto, S.; Clanet, M.; Coubes, P.; Echenne, B.; Ibrahim, R.; Irthum, B.; et al. Mutations within the programmed cell death 10 gene cause cerebral cavernous malformations. Am. J. Hum. Genet. 2005, 76, 42–51. [Google Scholar] [CrossRef]
- Abou-Fadel, J.; Jiang, X.; Padarti, A.; Goswami, D.G.; Smith, M.; Grajeda, B.; Bhalli, M.; Le, A.; Walker, W.E.; Zhang, J. mPR-Specific Actions Influence Maintenance of the Blood-Brain Barrier (BBB). Int. J. Mol. Sci. 2022, 23, 9684. [Google Scholar] [CrossRef]
- Abou-Fadel, J.; Smith, M.; Falahati, K.; Zhang, J. Comparative omics of CCM signaling complex (CSC). Chin. Neurosurg. J. 2020, 6, 4. [Google Scholar] [CrossRef]
- Abou-Fadel, J.; Vasquez, M.; Grajeda, B.; Ellis, C.; Zhang, J. Systems-wide analysis unravels the new roles of CCM signal complex (CSC). Heliyon 2019, 5, e02899. [Google Scholar] [CrossRef]
- Abou-Fadel, J.; Zhang, J. Systems Wide Analysis of CCM Signaling Complex Alterations in CCM-Deficient Models Using Omics Approaches. Methods Mol. Biol. 2020, 2152, 325–344. [Google Scholar] [CrossRef]
- Yan, L.; Moriarty, R.A.; Stroka, K.M. Recent progress and new challenges in modeling of human pluripotent stem cell-derived blood-brain barrier. Theranostics 2021, 11, 10148–10170. [Google Scholar] [CrossRef]
- Daneman, R.; Prat, A. The blood-brain barrier. Cold Spring Harb. Perspect. Biol. 2015, 7, a020412. [Google Scholar] [CrossRef]
- Correale, J.; Villa, A. Cellular elements of the blood-brain barrier. Neurochem. Res. 2009, 34, 2067–2077. [Google Scholar] [CrossRef]
- Liebner, S.; Czupalla, C.J.; Wolburg, H. Current concepts of blood-brain barrier development. Int. J. Dev. Biol. 2011, 55, 467–476. [Google Scholar] [CrossRef]
- Cardoso, F.L.; Brites, D.; Brito, M.A. Looking at the blood-brain barrier: Molecular anatomy and possible investigation approaches. Brain Res. Rev. 2010, 64, 328–363. [Google Scholar] [CrossRef]
- Manno, E.M.; Atkinson, J.L.; Fulgham, J.R.; Wijdicks, E.F. Emerging medical and surgical management strategies in the evaluation and treatment of intracerebral hemorrhage. Mayo Clin. Proc. 2005, 80, 420–433. [Google Scholar] [CrossRef]
- Gomes, J.A.; Manno, E. New developments in the treatment of intracerebral hemorrhage. Neurol. Clin. 2013, 31, 721–735. [Google Scholar] [CrossRef]
- Ballabh, P. Pathogenesis and prevention of intraventricular hemorrhage. Clin. Perinatol. 2014, 41, 47–67. [Google Scholar] [CrossRef]
- Zhao, Z.; Nelson, A.R.; Betsholtz, C.; Zlokovic, B.V. Establishment and Dysfunction of the Blood-Brain Barrier. Cell 2015, 163, 1064–1078. [Google Scholar] [CrossRef]
- Vandenbroucke, E.; Mehta, D.; Minshall, R.; Malik, A.B. Regulation of endothelial junctional permeability. Ann. N. Y. Acad. Sci. 2008, 1123, 134–145. [Google Scholar] [CrossRef]
- Ono, S.; Egawa, G.; Kabashima, K. Regulation of blood vascular permeability in the skin. Inflamm. Regen. 2017, 37, 11. [Google Scholar] [CrossRef]
- Abbott, N.J.; Patabendige, A.A.; Dolman, D.E.; Yusof, S.R.; Begley, D.J. Structure and function of the blood-brain barrier. Neurobiol. Dis. 2010, 37, 13–25. [Google Scholar] [CrossRef]
- Profaci, C.P.; Munji, R.N.; Pulido, R.S.; Daneman, R. The blood-brain barrier in health and disease: Important unanswered questions. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef]
- Greene, C.; Hanley, N.; Campbell, M. Claudin-5: Gatekeeper of neurological function. Fluids Barriers CNS 2019, 16, 3. [Google Scholar] [CrossRef]
- Batra, S.; Lin, D.; Recinos, P.F.; Zhang, J.; Rigamonti, D. Cavernous malformations: Natural history, diagnosis and treatment. Nat. Rev. Neurol. 2009, 5, 659–670. [Google Scholar] [CrossRef]
- Witt, K.A.; Sandoval, K.E. Steroids and the blood-brain barrier: Therapeutic implications. Adv. Pharmacol. 2014, 71, 361–390. [Google Scholar] [CrossRef]
- Hawkins, B.T.; Davis, T.P. The blood-brain barrier/neurovascular unit in health and disease. Pharmacol. Rev. 2005, 57, 173–185. [Google Scholar] [CrossRef]
- Salvador, E.; Shityakov, S.; Forster, C. Glucocorticoids and endothelial cell barrier function. Cell Tissue Res. 2014, 355, 597–605. [Google Scholar] [CrossRef]
- Friden, P.M. Receptor-mediated transport of therapeutics across the blood-brain barrier. Neurosurgery 1994, 35, 294–298, discussion 298. [Google Scholar] [CrossRef]
- Roberts, R.L.; Fine, R.E.; Sandra, A. Receptor-mediated endocytosis of transferrin at the blood-brain barrier. J. Cell Sci. 1993, 104 Pt 2, 521–532. [Google Scholar] [CrossRef]
- Pulgar, V.M. Transcytosis to Cross the Blood Brain Barrier, New Advancements and Challenges. Front. Neurosci. 2018, 12, 1019. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Liu, L.; Ma, F.; Wong, C.W.; Guo, X.E.; Chacko, J.V.; Farhoodi, H.P.; Zhang, S.X.; Zimak, J.; Segaliny, A.; et al. Elucidation of Exosome Migration across the Blood-Brain Barrier Model In Vitro. Cell. Mol. Bioeng. 2016, 9, 509–529. [Google Scholar] [CrossRef] [PubMed]
- Hersh, D.S.; Wadajkar, A.S.; Roberts, N.; Perez, J.G.; Connolly, N.P.; Frenkel, V.; Winkles, J.A.; Woodworth, G.F.; Kim, A.J. Evolving Drug Delivery Strategies to Overcome the Blood Brain Barrier. Curr. Pharm. Des. 2016, 22, 1177–1193. [Google Scholar] [CrossRef] [PubMed]
- Lajoie, J.M.; Shusta, E.V. Targeting receptor-mediated transport for delivery of biologics across the blood-brain barrier. Annu Rev. Pharmacol. Toxicol. 2015, 55, 613–631. [Google Scholar] [CrossRef]
- Stanimirovic, D.B.; Sandhu, J.K.; Costain, W.J. Emerging Technologies for Delivery of Biotherapeutics and Gene Therapy Across the Blood-Brain Barrier. BioDrugs 2018, 32, 547–559. [Google Scholar] [CrossRef]
- Gomes, P.A.; Bodo, C.; Nogueras-Ortiz, C.; Samiotaki, M.; Chen, M.; Soares-Cunha, C.; Silva, J.M.; Coimbra, B.; Stamatakis, G.; Santos, L.; et al. A novel isolation method for spontaneously released extracellular vesicles from brain tissue and its implications for stress-driven brain pathology. Cell Commun. Signal. 2023, 21, 35. [Google Scholar] [CrossRef]
- Serafini, U.; Masala, C. Corticosteroids and autoimmune diseases. Recenti Prog. Med. 1976, 61, 684–710. [Google Scholar]
- McWilliams, D.F.; Thankaraj, D.; Jones-Diette, J.; Morgan, R.; Ifesemen, O.S.; Shenker, N.G.; Walsh, D.A. The efficacy of systemic glucocorticosteroids for pain in rheumatoid arthritis: A systematic literature review and meta-analysis. Rheumatology 2021, 61, 76–89. [Google Scholar] [CrossRef]
- Euers, L.; Abughazaleh, S.; Glassner, K.; Gajula, P.; Jones-Pauley, M.; Ezeana, C.; Puppala, M.; Wang, L.; Wong, S.; Oglat, A.; et al. Risk Factors for and Frequency of CT Scans, Steroid Use, and Repeat Visits in Inflammatory Bowel Disease Patients Seen at a Single-Center Emergency Department: A Retrospective Cohort Study. J. Clin. Med. 2021, 10, 2679. [Google Scholar] [CrossRef]
- Forster, C.; Burek, M.; Romero, I.A.; Weksler, B.; Couraud, P.O.; Drenckhahn, D. Differential effects of hydrocortisone and TNFalpha on tight junction proteins in an in vitro model of the human blood-brain barrier. J. Physiol. 2008, 586, 1937–1949. [Google Scholar] [CrossRef]
- Abou-Fadel, J.; Bhalli, M.; Grajeda, B.; Zhang, J. CmP Signaling Network Leads to Identification of Prognostic Biomarkers for Triple-Negative Breast Cancer in Caucasian Women. Genet. Test. Mol. Biomarkers 2022, 26, 198–219. [Google Scholar] [CrossRef] [PubMed]
- Abou-Fadel, J.; Grajeda, B.; Jiang, X.; Cailing-De La, O.A.; Flores, E.; Padarti, A.; Bhalli, M.; Le, A.; Zhang, J. CmP signaling network unveils novel biomarkers for triple negative breast cancer in African American women. Cancer Biomark. 2022, 34, 607–636. [Google Scholar] [CrossRef] [PubMed]
- Abou-Fadel, J.; Jiang, X.; Grajeda, B.; Padarti, A.; Ellis, C.C.; Flores, E.; Cailing-De La, O.A.; Zhang, J. CCM signaling complex (CSC) couples both classic and non-classic Progesterone receptor signaling. Cell Commun. Signal. 2022, 20, 120. [Google Scholar] [CrossRef] [PubMed]
- Abou-Fadel, J.; Jiang, X.; Padarti, A.; Goswami, D.; Smith, M.; Grajeda, B.; Walker, W.; Zhang, J. CCM signaling complex (CSC) is a master regulator governing homeostasis of progestins and their mediated signaling cascades. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zhang, J.; Abou-Fadel, J. Calm the raging hormone—A new therapeutic strategy involving progesterone-signaling for hemorrhagic CCMs. Vessel Plus 2021, 5, 23. [Google Scholar] [CrossRef]
- Manners, J.; Steinberg, A.; Shutter, L. Early management of acute cerebrovascular accident. Curr. Opin. Crit. Care 2017, 23, 556–560. [Google Scholar] [CrossRef]
- Kuriakose, D.; Xiao, Z. Pathophysiology and Treatment of Stroke: Present Status and Future Perspectives. Int. J. Mol. Sci. 2020, 21, 7609. [Google Scholar] [CrossRef]
- Campbell, B.C.V.; Khatri, P. Stroke. Lancet 2020, 396, 129–142. [Google Scholar] [CrossRef]
- Blanco, M.; Castillo, J. Stroke in 2012: Major advances in the treatment of stroke. Nat. Rev. Neurol. 2013, 9, 68–70. [Google Scholar] [CrossRef]
- Ihezie, S.A.; Mathew, I.E.; McBride, D.W.; Dienel, A.; Blackburn, S.L.; Thankamani Pandit, P.K. Epigenetics in blood-brain barrier disruption. Fluids Barriers CNS 2021, 18, 17. [Google Scholar] [CrossRef]
- Claesson-Welsh, L.; Dejana, E.; McDonald, D.M. Permeability of the Endothelial Barrier: Identifying and Reconciling Controversies. Trends Mol. Med. 2021, 27, 314–331. [Google Scholar] [CrossRef] [PubMed]
- Claesson-Welsh, L. Vascular permeability—The essentials. Upsala J. Med. Sci. 2015, 120, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Nakada, T.; Kwee, I.L. Fluid Dynamics Inside the Brain Barrier: Current Concept of Interstitial Flow, Glymphatic Flow, and Cerebrospinal Fluid Circulation in the Brain. Neuroscientist 2019, 25, 155–166. [Google Scholar] [CrossRef]
- Yang, R.; Chen, M.; Zheng, J.; Li, X.; Zhang, X. The Role of Heparin and Glycocalyx in Blood-Brain Barrier Dysfunction. Front. Immunol. 2021, 12, 754141. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Fang, F.; Gao, W.; Chen, H.; Wen, J.; Wen, X.; Chen, J. The Structure and Function of the Glycocalyx and Its Connection With Blood-Brain Barrier. Front. Cell. Neurosci. 2021, 15, 739699. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Nirwane, A.; Yao, Y. Basement membrane and blood-brain barrier. Stroke Vasc. Neurol. 2019, 4, 78–82. [Google Scholar] [CrossRef] [PubMed]
- McDonald, D.M.; Baluk, P. Imaging of angiogenesis in inflamed airways and tumors: Newly formed blood vessels are not alike and may be wildly abnormal: Parker B. Francis lecture. Chest 2005, 128, 602S–608S. [Google Scholar] [CrossRef]
- Adams, R.H.; Alitalo, K. Molecular regulation of angiogenesis and lymphangiogenesis. Nat. Rev. Mol. Cell Biol. 2007, 8, 464–478. [Google Scholar] [CrossRef]
- Antonetti, D.A.; Lieth, E.; Barber, A.J.; Gardner, T.W. Molecular mechanisms of vascular permeability in diabetic retinopathy. Semin. Ophthalmol. 1999, 14, 240–248. [Google Scholar] [CrossRef]
- Shah, G.N.; Mooradian, A.D. Age-related changes in the blood-brain barrier. Exp. Gerontol. 1997, 32, 501–519. [Google Scholar] [CrossRef]
- Knox, E.G.; Aburto, M.R.; Clarke, G.; Cryan, J.F.; O’Driscoll, C.M. The blood-brain barrier in aging and neurodegeneration. Mol. Psychiatry 2022, 27, 2659–2673. [Google Scholar] [CrossRef] [PubMed]
- Dejana, E.; Orsenigo, F.; Lampugnani, M.G. The role of adherens junctions and VE-cadherin in the control of vascular permeability. J. Cell Sci. 2008, 121, 2115–2122. [Google Scholar] [CrossRef] [PubMed]
- Sandoval, K.E.; Witt, K.A. Blood-brain barrier tight junction permeability and ischemic stroke. Neurobiol. Dis. 2008, 32, 200–219. [Google Scholar] [CrossRef] [PubMed]
- Bazzoni, G.; Dejana, E. Endothelial cell-to-cell junctions: Molecular organization and role in vascular homeostasis. Physiol. Rev. 2004, 84, 869–901. [Google Scholar] [CrossRef]
- Weis, S.M.; Cheresh, D.A. Pathophysiological consequences of VEGF-induced vascular permeability. Nature 2005, 437, 497–504. [Google Scholar] [CrossRef]
- Andriopoulou, P.; Navarro, P.; Zanetti, A.; Lampugnani, M.G.; Dejana, E. Histamine induces tyrosine phosphorylation of endothelial cell-to-cell adherens junctions. Arter. Thromb. Vasc. Biol. 1999, 19, 2286–2297. [Google Scholar] [CrossRef]
- Rosenblum, S.L.; Kosman, D.J. Aberrant Cerebral Iron Trafficking Co-morbid With Chronic Inflammation: Molecular Mechanisms and Pharmacologic Intervention. Front. Neurol. 2022, 13, 855751. [Google Scholar] [CrossRef]
- Huber, J.D.; Witt, K.A.; Hom, S.; Egleton, R.D.; Mark, K.S.; Davis, T.P. Inflammatory pain alters blood-brain barrier permeability and tight junctional protein expression. Am. J. Physiol. Heart Circ. Physiol. 2001, 280, H1241–H1248. [Google Scholar] [CrossRef]
- Vestweber, D. How leukocytes cross the vascular endothelium. Nat. Rev. Immunol. 2015, 15, 692–704. [Google Scholar] [CrossRef]
- McEver, R.P. Selectins: Initiators of leucocyte adhesion and signalling at the vascular wall. Cardiovasc. Res. 2015, 107, 331–339. [Google Scholar] [CrossRef]
- Takeshita, Y.; Ransohoff, R.M. Inflammatory cell trafficking across the blood-brain barrier: Chemokine regulation and in vitro models. Immunol. Rev. 2012, 248, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Olsson, A.K.; Dimberg, A.; Kreuger, J.; Claesson-Welsh, L. VEGF receptor signalling—In control of vascular function. Nat. Rev. Mol. Cell Biol. 2006, 7, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Hoeben, A.; Landuyt, B.; Highley, M.S.; Wildiers, H.; Van Oosterom, A.T.; De Bruijn, E.A. Vascular endothelial growth factor and angiogenesis. Pharmacol. Rev. 2004, 56, 549–580. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.L.; Nam, J.O.; Jean, C.; Lawson, C.; Walsh, C.T.; Goka, E.; Lim, S.T.; Tomar, A.; Tancioni, I.; Uryu, S.; et al. VEGF-induced vascular permeability is mediated by FAK. Dev. Cell 2012, 22, 146–157. [Google Scholar] [CrossRef]
- Kim, I.; Moon, S.O.; Park, S.K.; Chae, S.W.; Koh, G.Y. Angiopoietin-1 reduces VEGF-stimulated leukocyte adhesion to endothelial cells by reducing ICAM-1, VCAM-1, and E-selectin expression. Circ. Res. 2001, 89, 477–479. [Google Scholar] [CrossRef]
- Simoncini, T.; Maffei, S.; Basta, G.; Barsacchi, G.; Genazzani, A.R.; Liao, J.K.; De Caterina, R. Estrogens and glucocorticoids inhibit endothelial vascular cell adhesion molecule-1 expression by different transcriptional mechanisms. Circ. Res. 2000, 87, 19–25. [Google Scholar] [CrossRef]
- Cruz-Topete, D.; Oakley, R.H.; Cidlowski, J.A. Glucocorticoid Signaling and the Aging Heart. Front. Endocrinol. 2020, 11, 347. [Google Scholar] [CrossRef]
- Felinski, E.A.; Cox, A.E.; Phillips, B.E.; Antonetti, D.A. Glucocorticoids induce transactivation of tight junction genes occludin and claudin-5 in retinal endothelial cells via a novel cis-element. Exp. Eye Res. 2008, 86, 867–878. [Google Scholar] [CrossRef]
- Harke, N.; Leers, J.; Kietz, S.; Drenckhahn, D.; Forster, C. Glucocorticoids regulate the human occludin gene through a single imperfect palindromic glucocorticoid response element. Mol. Cell. Endocrinol. 2008, 295, 39–47. [Google Scholar] [CrossRef]
- Forster, C.; Waschke, J.; Burek, M.; Leers, J.; Drenckhahn, D. Glucocorticoid effects on mouse microvascular endothelial barrier permeability are brain specific. J. Physiol. 2006, 573, 413–425. [Google Scholar] [CrossRef]
- Forster, C.; Silwedel, C.; Golenhofen, N.; Burek, M.; Kietz, S.; Mankertz, J.; Drenckhahn, D. Occludin as direct target for glucocorticoid-induced improvement of blood-brain barrier properties in a murine in vitro system. J. Physiol. 2005, 565, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Westcott, R.; Chung, N.; Ghosh, A.; Ferguson, L.; Bingaman, W.; Najm, I.M.; Ghosh, C. Glucocorticoid Receptor beta Isoform Predominates in the Human Dysplastic Brain Region and Is Modulated by Age, Sex, and Antiseizure Medication. Int. J. Mol. Sci. 2022, 23, 4940. [Google Scholar] [CrossRef]
- Zhu, Q.; Zhao, C.; Wang, Y.; Mu, L.; Li, X.; Qi, Y.; Yang, J.; Ma, C. Soluble Vascular Cell Adhesion Molecule-1 as an Inflammation-Related Biomarker of Coronary Slow Flow. J. Clin. Med. 2023, 12, 543. [Google Scholar] [CrossRef]
- Pulito-Cueto, V.; Remuzgo-Martinez, S.; Genre, F.; Atienza-Mateo, B.; Mora-Cuesta, V.M.; Iturbe-Fernandez, D.; Lera-Gomez, L.; Sebastian Mora-Gil, M.; Prieto-Pena, D.; Portilla, V.; et al. Elevated VCAM-1, MCP-1 and ADMA serum levels related to pulmonary fibrosis of interstitial lung disease associated with rheumatoid arthritis. Front. Mol. Biosci. 2022, 9, 1056121. [Google Scholar] [CrossRef]
- Kong, D.H.; Kim, Y.K.; Kim, M.R.; Jang, J.H.; Lee, S. Emerging Roles of Vascular Cell Adhesion Molecule-1 (VCAM-1) in Immunological Disorders and Cancer. Int. J. Mol. Sci. 2018, 19, 1057. [Google Scholar] [CrossRef] [PubMed]
- Ungvari, Z.; Labinskyy, N.; Mukhopadhyay, P.; Pinto, J.T.; Bagi, Z.; Ballabh, P.; Zhang, C.; Pacher, P.; Csiszar, A. Resveratrol attenuates mitochondrial oxidative stress in coronary arterial endothelial cells. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H1876–H1881. [Google Scholar] [CrossRef] [PubMed]
- Hafezi-Moghadam, A.; Simoncini, T.; Yang, Z.; Limbourg, F.P.; Plumier, J.C.; Rebsamen, M.C.; Hsieh, C.M.; Chui, D.S.; Thomas, K.L.; Prorock, A.J.; et al. Acute cardiovascular protective effects of corticosteroids are mediated by non-transcriptional activation of endothelial nitric oxide synthase. Nat. Med. 2002, 8, 473–479. [Google Scholar] [CrossRef]
- Baum, M.; Moe, O.W. Glucocorticoid-mediated hypertension: Does the vascular smooth muscle hold all the answers? J. Am. Soc. Nephrol. 2008, 19, 1251–1253. [Google Scholar] [CrossRef]
- Goodwin, J.E.; Zhang, J.; Geller, D.S. A critical role for vascular smooth muscle in acute glucocorticoid-induced hypertension. J. Am. Soc. Nephrol. 2008, 19, 1291–1299. [Google Scholar] [CrossRef]
- Oakley, R.H.; Sar, M.; Cidlowski, J.A. The human glucocorticoid receptor beta isoform. Expression, biochemical properties, and putative function. J. Biol. Chem. 1996, 271, 9550–9559. [Google Scholar] [CrossRef]
- Ramos-Ramirez, P.; Tliba, O. Glucocorticoid Receptor beta (GRbeta): Beyond Its Dominant-Negative Function. Int. J. Mol. Sci. 2021, 22, 3649. [Google Scholar] [CrossRef] [PubMed]
- Hinds, T.D.; Peck, B.; Shek, E.; Stroup, S.; Hinson, J.; Arthur, S.; Marino, J.S. Overexpression of Glucocorticoid Receptor beta Enhances Myogenesis and Reduces Catabolic Gene Expression. Int. J. Mol. Sci. 2016, 17, 232. [Google Scholar] [CrossRef]
- Wintzer, S.; Heckmann, J.G.; Huttner, H.B.; Schwab, S. Dexamethasone in Patients with Spontaneous Intracerebral Hemorrhage: An Updated Meta-Analysis. Cerebrovasc. Dis. 2020, 49, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Sundboll, J.; Horvath-Puho, E.; Schmidt, M.; Dekkers, O.M.; Christiansen, C.F.; Pedersen, L.; Botker, H.E.; Sorensen, H.T. Preadmission Use of Glucocorticoids and 30-Day Mortality After Stroke. Stroke 2016, 47, 829–835. [Google Scholar] [CrossRef]
- Maggioli, E.; McArthur, S.; Mauro, C.; Kieswich, J.; Kusters, D.H.M.; Reutelingsperger, C.P.M.; Yaqoob, M.; Solito, E. Estrogen protects the blood-brain barrier from inflammation-induced disruption and increased lymphocyte trafficking. Brain Behav. Immun. 2016, 51, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Lenert, M.E.; Avona, A.; Garner, K.M.; Barron, L.R.; Burton, M.D. Sensory Neurons, Neuroimmunity, and Pain Modulation by Sex Hormones. Endocrinology 2021, 162. [Google Scholar] [CrossRef]
- Kuruca, S.E.; Karadenizli, S.; Akgun-Dar, K.; Kapucu, A.; Kaptan, Z.; Uzum, G. The effects of 17beta-estradiol on blood brain barrier integrity in the absence of the estrogen receptor alpha; an in-vitro model. Acta Histochem. 2017, 119, 638–647. [Google Scholar] [CrossRef]
- Rettberg, J.R.; Yao, J.; Brinton, R.D. Estrogen: A master regulator of bioenergetic systems in the brain and body. Front. Neuroendocrinol. 2014, 35, 8–30. [Google Scholar] [CrossRef]
- Sohrabji, F.; Bake, S.; Lewis, D.K. Age-related changes in brain support cells: Implications for stroke severity. Neurochem. Int. 2013, 63, 291–301. [Google Scholar] [CrossRef]
- Selvamani, A.; Sohrabji, F. Reproductive age modulates the impact of focal ischemia on the forebrain as well as the effects of estrogen treatment in female rats. Neurobiol. Aging 2010, 31, 1618–1628. [Google Scholar] [CrossRef]
- Brinton, R.D. Investigative models for determining hormone therapy-induced outcomes in brain: Evidence in support of a healthy cell bias of estrogen action. Ann. N. Y. Acad. Sci. 2005, 1052, 57–74. [Google Scholar] [CrossRef] [PubMed]
- Leon, R.L.; Li, X.; Huber, J.D.; Rosen, C.L. Worsened outcome from middle cerebral artery occlusion in aged rats receiving 17beta-estradiol. Endocrinology 2012, 153, 3386–3393. [Google Scholar] [CrossRef] [PubMed]
- Viscoli, C.M.; Brass, L.M.; Kernan, W.N.; Sarrel, P.M.; Suissa, S.; Horwitz, R.I. A clinical trial of estrogen-replacement therapy after ischemic stroke. N. Engl. J. Med. 2001, 345, 1243–1249. [Google Scholar] [CrossRef] [PubMed]
- Beal, C.C.; Faucher, M.A. Stroke and pregnancy: An integrative review with implications for neuroscience nurses. J. Neurosci. Nurs. 2015, 47, 76–84, quiz E71. [Google Scholar] [CrossRef]
- Carbillon, L. Pregnancy is an essential spontaneous screening stress test for the risk of early stroke in women. Stroke 2008, 39, e138. [Google Scholar] [CrossRef]
- Caso, V.; Falorni, A.; Bushnell, C.D.; Acciarresi, M.; Remohi, J.; Sprigg, N.; Gerli, S. Pregnancy, Hormonal Treatments for Infertility, Contraception, and Menopause in Women After Ischemic Stroke: A Consensus Document. Stroke 2017, 48, 501–506. [Google Scholar] [CrossRef]
- Cheng, C.A.; Lee, J.T.; Lin, H.C.; Lin, H.C.; Chung, C.H.; Lin, F.H.; Tsao, C.H.; Wu, Y.F.; Chien, W.C.; Chiu, H.W. Pregnancy increases stroke risk up to 1 year postpartum and reduces long-term risk. QJM 2017, 110, 355–360. [Google Scholar] [CrossRef]
- Feske, S.K. Stroke in pregnancy. Semin. Neurol. 2007, 27, 442–452. [Google Scholar] [CrossRef]
- James, A.H.; Bushnell, C.D.; Jamison, M.G.; Myers, E.R. Incidence and risk factors for stroke in pregnancy and the puerperium. Obstet. Gynecol. 2005, 106, 509–516. [Google Scholar] [CrossRef]
- Jeng, J.S.; Tang, S.C.; Yip, P.K. Stroke in women of reproductive age: Comparison between stroke related and unrelated to pregnancy. J. Neurol. Sci. 2004, 221, 25–29. [Google Scholar] [CrossRef]
- Sanders, B.D.; Davis, M.G.; Holley, S.L.; Phillippi, J.C. Pregnancy-Associated Stroke. J. Midwifery Womens Health 2018, 63, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Ban, L.; Abdul Sultan, A.; Stephansson, O.; Tata, L.J.; Sprigg, N.; Nelson-Piercy, C.; Bath, P.M.; Ludvigsson, J.F. The incidence of first stroke in and around pregnancy: A population-based cohort study from Sweden. Eur. Stroke J. 2017, 2, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Camargo, E.C.; Feske, S.K.; Singhal, A.B. Stroke in Pregnancy: An Update. Neurol. Clin. 2019, 37, 131–148. [Google Scholar] [CrossRef] [PubMed]
- Leffert, L.R.; Clancy, C.R.; Bateman, B.T.; Cox, M.; Schulte, P.J.; Smith, E.E.; Fonarow, G.C.; Schwamm, L.H.; Kuklina, E.V.; George, M.G. Patient Characteristics and Outcomes After Hemorrhagic Stroke in Pregnancy. Circ. Cardiovasc. Qual. Outcomes 2015, 8, S170–S178. [Google Scholar] [CrossRef]
- Liew, B.S.; Ghani, A.A.; You, X. Stroke in pregnancy. Med. J. Malaysia 2019, 74, 246–249. [Google Scholar] [PubMed]
- Liu, X.J.; Wang, S.; Zhao, Y.L.; Zhang, D.; Zhao, J.Z. A single-center study of hemorrhagic stroke caused by cerebrovascular disease during pregnancy and puerperium in China. Int. J. Gynaecol. Obstet. 2011, 113, 82–83. [Google Scholar] [CrossRef]
- Miller, E.C.; Sundheim, K.M.; Willey, J.Z.; Boehme, A.K.; Agalliu, D.; Marshall, R.S. The Impact of Pregnancy on Hemorrhagic Stroke in Young Women. Cerebrovasc. Dis. 2018, 46, 10–15. [Google Scholar] [CrossRef]
- Ulrich, N.D.; Lapeyre, E.R.; Moore, R.C. Hemorrhagic Stroke Resulting From Venous Malformation at 20 Weeks of Pregnancy. Ochsner J. 2016, 16, 542–544. [Google Scholar]
- Yoshida, K.; Takahashi, J.C.; Takenobu, Y.; Suzuki, N.; Ogawa, A.; Miyamoto, S. Strokes Associated With Pregnancy and Puerperium: A Nationwide Study by the Japan Stroke Society. Stroke 2017, 48, 276–282. [Google Scholar] [CrossRef]
- Witiw, C.D.; Abou-Hamden, A.; Kulkarni, A.V.; Silvaggio, J.A.; Schneider, C.; Wallace, M.C. Cerebral cavernous malformations and pregnancy: Hemorrhage risk and influence on obstetrical management. Neurosurgery 2012, 71, 626–630, discussion 631. [Google Scholar] [CrossRef]
- Zuurbier, S.M.; Santos, A.N.; Flemming, K.D.; Schmidt, B.; Jabbarli, R.; Lanzino, G.; Sure, U.; Dammann, P. Female Hormone Therapy and Risk of Intracranial Hemorrhage From Cerebral Cavernous Malformations: A Multicenter Observational Cohort Study. Neurology 2023, 100, e1673–e1679. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Nakase, H.; Nakagawa, I.; Nishimura, F.; Motoyama, Y.; Park, Y.S. Cavernous malformations in pregnancy. Neurol. Med. Chir. 2013, 53, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Renteria, M.; Belkin, O.; Aickareth, J.; Jang, D.; Hawwar, M.; Zhang, J. Zinc’s Association with the CmPn/CmP Signaling Network in Breast Cancer Tumorigenesis. Biomolecules 2022, 12, 1672. [Google Scholar] [CrossRef]
- Renteria, M.; Belkin, O.; Jang, D.; Aickareth, J.; Bhalli, M.; Zhang, J. CmPn signaling networks in the tumorigenesis of breast cancer. Front. Endocrinol. 2022, 13, 1013892. [Google Scholar] [CrossRef] [PubMed]
- Karteris, E.; Zervou, S.; Pang, Y.; Dong, J.; Hillhouse, E.W.; Randeva, H.S.; Thomas, P. Progesterone signaling in human myometrium through two novel membrane G protein-coupled receptors: Potential role in functional progesterone withdrawal at term. Mol. Endocrinol. 2006, 20, 1519–1534. [Google Scholar] [CrossRef]
- Flemming, K.D.; Kumar, S.; Brown, R.D., Jr.; Lanzino, G. Predictors of Initial Presentation with Hemorrhage in Patients with Cavernous Malformations. World Neurosurg. 2020, 133, e767–e773. [Google Scholar] [CrossRef]
- Zahreddine, R.; Davezac, M.; Smirnova, N.; Buscato, M.; Lhuillier, E.; Lupieri, A.; Solinhac, R.; Vinel, A.; Vessieres, E.; Henrion, D.; et al. Tamoxifen Accelerates Endothelial Healing by Targeting ERalpha in Smooth Muscle Cells. Circ. Res. 2020, 127, 1473–1487. [Google Scholar] [CrossRef]
- Zahreddine, R.; Davezac, M.; Buscato, M.; Smirnova, N.; Laffargue, M.; Henrion, D.; Adlanmerini, M.; Lenfant, F.; Arnal, J.F.; Fontaine, C. A historical view of estrogen effect on arterial endothelial healing: From animal models to medical implication. Atherosclerosis 2021, 338, 30–38. [Google Scholar] [CrossRef]
- Takagi, H. Diagnosis and management of cavernous hemangioma of the liver. Semin. Surg. Oncol. 1985, 1, 12–22. [Google Scholar] [CrossRef]
- Dourmishev, L.A.; Dourmishev, A.L. Craniofacial cavernous hemangioma: Succesful treatment with methylprednisolone. Acta Dermatovenerol. Alp. Pannonica Adriat. 2005, 14, 49–52. [Google Scholar]
- Tang, A.T.; Sullivan, K.R.; Hong, C.C.; Goddard, L.M.; Mahadevan, A.; Ren, A.; Pardo, H.; Peiper, A.; Griffin, E.; Tanes, C.; et al. Distinct cellular roles for PDCD10 define a gut-brain axis in cerebral cavernous malformation. Sci. Transl. Med. 2019, 11, eaaw3521. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.; Tee, J.W.; Trost, N.; McKelvie, P.; Wang, Y.Y. Anterior visual pathway cavernous malformations. J. Clin. Neurosci. 2015, 22, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Chu, K.; Jeong, S.W.; Park, H.K.; Bae, H.J.; Yoon, B.W. Cerebral cavernous malformations with dynamic and progressive course: Correlation study with vascular endothelial growth factor. Arch Neurol. 2003, 60, 1613–1618. [Google Scholar] [CrossRef] [PubMed]
- Lunsford, L.D.; Khan, A.A.; Niranjan, A.; Kano, H.; Flickinger, J.C.; Kondziolka, D. Stereotactic radiosurgery for symptomatic solitary cerebral cavernous malformations considered high risk for resection. J. Neurosurg. 2010, 113, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Abou-Al-Shaar, H.; Bahatheq, A.; Takroni, R.; Al-Thubaiti, I. Optic chiasmal cavernous angioma: A rare suprasellar vascular malformation. Surg. Neurol. Int. 2016, 7, S523–S526. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.Y.; Sun, H.; Xu, J.G.; Li, Q.Y. Stereotactic radiosurgery of brainstem cavernous malformations: A systematic review and meta-analysis. J. Neurosurg. 2014, 120, 982–987. [Google Scholar] [CrossRef]
- Kim, B.S.; Kim, K.H.; Lee, M.H.; Lee, J.I. Stereotactic Radiosurgery for Brainstem Cavernous Malformations: An Updated Systematic Review and Meta-Analysis. World Neurosurg. 2019, 130, e648–e659. [Google Scholar] [CrossRef]
- Chakhtoura, Z.; Canonico, M.; Gompel, A.; Thalabard, J.C.; Scarabin, P.Y.; Plu-Bureau, G. Progestogen-only contraceptives and the risk of stroke: A meta-analysis. Stroke 2009, 40, 1059–1062. [Google Scholar] [CrossRef]
- Louw-du Toit, R.; Hapgood, J.P.; Africander, D. A direct comparison of the transcriptional activities of progestins used in contraception and menopausal hormone therapy via the mineralocorticoid receptor. Biochem. Biophys. Res. Commun. 2020, 526, 466–471. [Google Scholar] [CrossRef]
- Hapgood, J.P.; Ray, R.M.; Govender, Y.; Avenant, C.; Tomasicchio, M. Differential glucocorticoid receptor-mediated effects on immunomodulatory gene expression by progestin contraceptives: Implications for HIV-1 pathogenesis. Am. J. Reprod. Immunol. 2014, 71, 505–512. [Google Scholar] [CrossRef]
- Mueck, A.O.; Seeger, H. Progestogens and target tissues: Vascular systems. Maturitas 2009, 62, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Mirkin, S.; Wong, B.C.; Archer, D.F. Effect of 17 beta-estradiol, progesterone, synthetic progestins, tibolone, and tibolone metabolites on vascular endothelial growth factor mRNA in breast cancer cells. Fertil. Steril. 2005, 84, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Charnock-Jones, D.S.; Macpherson, A.M.; Archer, D.F.; Leslie, S.; Makkink, W.K.; Sharkey, A.M.; Smith, S.K. The effect of progestins on vascular endothelial growth factor, oestrogen receptor and progesterone receptor immunoreactivity and endothelial cell density in human endometrium. Hum. Reprod. 2000, 15 (Suppl. 3), 85–95. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Xiao, J.; Dai, Z.; Chen, Q. Membrane progesterone receptor alpha (mPRalpha) enhances hypoxia-induced vascular endothelial growth factor secretion and angiogenesis in lung adenocarcinoma through STAT3 signaling. J. Transl. Med. 2022, 20, 72. [Google Scholar] [CrossRef]
- Neubauer, H.; Adam, G.; Seeger, H.; Mueck, A.O.; Solomayer, E.; Wallwiener, D.; Cahill, M.A.; Fehm, T. Membrane-initiated effects of progesterone on proliferation and activation of VEGF in breast cancer cells. Climacteric 2009, 12, 230–239. [Google Scholar] [CrossRef]
- Pang, Y.; Dong, J.; Thomas, P. Progesterone increases nitric oxide synthesis in human vascular endothelial cells through activation of membrane progesterone receptor-alpha. Am. J. Physiol. Endocrinol. Metab. 2015, 308, E899–E911. [Google Scholar] [CrossRef]
- Ribatti, D.; Pezzella, F. Overview on the Different Patterns of Tumor Vascularization. Cells 2021, 10, 639. [Google Scholar] [CrossRef]
- Demir, R.; Yaba, A.; Huppertz, B. Vasculogenesis and angiogenesis in the endometrium during menstrual cycle and implantation. Acta Histochem. 2010, 112, 203–214. [Google Scholar] [CrossRef]
- Yu, P.; Li, S.; Zhang, Z.; Wen, X.; Quan, W.; Tian, Q.; Gao, C.; Su, W.; Zhang, J.; Jiang, R. Progesterone-mediated angiogenic activity of endothelial progenitor cell and angiogenesis in traumatic brain injury rats were antagonized by progesterone receptor antagonist. Cell Prolif 2017, 50, e12362. [Google Scholar] [CrossRef]
- Xia, Z.; Xiao, J.; Chen, Q. Solving the Puzzle: What Is the Role of Progestogens in Neovascularization? Biomolecules 2021, 11, 1686. [Google Scholar] [CrossRef]
- Matsubara, Y.; Matsubara, K. Estrogen and progesterone play pivotal roles in endothelial progenitor cell proliferation. Reprod. Biol. Endocrinol. 2012, 10, 2. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N.; Houck, K.; Jakeman, L.; Leung, D.W. Molecular and biological properties of the vascular endothelial growth factor family of proteins. Endocr. Rev. 1992, 13, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Hyder, S.M.; Stancel, G.M.; Chiappetta, C.; Murthy, L.; Boettger-Tong, H.L.; Makela, S. Uterine expression of vascular endothelial growth factor is increased by estradiol and tamoxifen. Cancer Res. 1996, 56, 3954–3960. [Google Scholar]
- Cullinan-Bove, K.; Koos, R.D. Vascular endothelial growth factor/vascular permeability factor expression in the rat uterus: Rapid stimulation by estrogen correlates with estrogen-induced increases in uterine capillary permeability and growth. Endocrinology 1993, 133, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Jazbutyte, V.; Arias-Loza, P.A.; Hu, K.; Widder, J.; Govindaraj, V.; von Poser-Klein, C.; Bauersachs, J.; Fritzemeier, K.H.; Hegele-Hartung, C.; Neyses, L.; et al. Ligand-dependent activation of ERbeta lowers blood pressure and attenuates cardiac hypertrophy in ovariectomized spontaneously hypertensive rats. Cardiovasc. Res. 2008, 77, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Selles, J.; Polini, N.; Alvarez, C.; Massheimer, V. Progesterone and 17 beta-estradiol acutely stimulate nitric oxide synthase activity in rat aorta and inhibit platelet aggregation. Life Sci. 2001, 69, 815–827. [Google Scholar] [CrossRef]
- Simoncini, T.; Mannella, P.; Fornari, L.; Caruso, A.; Willis, M.Y.; Garibaldi, S.; Baldacci, C.; Genazzani, A.R. Differential signal transduction of progesterone and medroxyprogesterone acetate in human endothelial cells. Endocrinology 2004, 145, 5745–5756. [Google Scholar] [CrossRef]
- Lopez-Ramirez, M.A.; Lai, C.C.; Soliman, S.I.; Hale, P.; Pham, A.; Estrada, E.J.; McCurdy, S.; Girard, R.; Verma, R.; Moore, T.; et al. Astrocytes propel neurovascular dysfunction during cerebral cavernous malformation lesion formation. J. Clin. Investig. 2021, 131. [Google Scholar] [CrossRef]
- Fujimura, M.; Watanabe, M.; Shimizu, H.; Tominaga, T. Expression of matrix metalloproteinases (MMPs) and tissue inhibitor of metalloproteinase (TIMP) in cerebral cavernous malformations: Immunohistochemical analysis of MMP-2, -9 and TIMP-2. Acta Neurochir. 2007, 149, 179–183, discussion 183. [Google Scholar] [CrossRef]
- Choquet, H.; Trapani, E.; Goitre, L.; Trabalzini, L.; Akers, A.; Fontanella, M.; Hart, B.L.; Morrison, L.A.; Pawlikowska, L.; Kim, H.; et al. Cytochrome P450 and matrix metalloproteinase genetic modifiers of disease severity in Cerebral Cavernous Malformation type 1. Free Radic. Biol. Med. 2016, 92, 100–109. [Google Scholar] [CrossRef]
- Xu, L.; Sapolsky, R.M.; Giffard, R.G. Differential sensitivity of murine astrocytes and neurons from different brain regions to injury. Exp. Neurol. 2001, 169, 416–424. [Google Scholar] [CrossRef] [PubMed]
- Pluchino, N.; Cubeddu, A.; Giannini, A.; Merlini, S.; Cela, V.; Angioni, S.; Genazzani, A.R. Progestogens and brain: An update. Maturitas 2009, 62, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Vidal, M.D.; Cervera-Gaviria, M.; Ruelas, R.; Escobar, A.; Morali, G.; Cervantes, M. Progesterone: Protective effects on the cat hippocampal neuronal damage due to acute global cerebral ischemia. Arch. Med. Res. 1998, 29, 117–124. [Google Scholar]
- Roof, R.L.; Duvdevani, R.; Braswell, L.; Stein, D.G. Progesterone facilitates cognitive recovery and reduces secondary neuronal loss caused by cortical contusion injury in male rats. Exp. Neurol. 1994, 129, 64–69. [Google Scholar] [CrossRef]
- Guo, Q.; Sayeed, I.; Baronne, L.M.; Hoffman, S.W.; Guennoun, R.; Stein, D.G. Progesterone administration modulates AQP4 expression and edema after traumatic brain injury in male rats. Exp. Neurol. 2006, 198, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Moorthy, K.; Yadav, U.C.; Siddiqui, M.R.; Mantha, A.K.; Basir, S.F.; Sharma, D.; Cowsik, S.M.; Baquer, N.Z. Effect of hormone replacement therapy in normalizing age related neuronal markers in different age groups of naturally menopausal rats. Biogerontology 2005, 6, 345–356. [Google Scholar] [CrossRef]
- Roof, R.L.; Hoffman, S.W.; Stein, D.G. Progesterone protects against lipid peroxidation following traumatic brain injury in rats. Mol. Chem. Neuropathol. 1997, 31, 1–11. [Google Scholar] [CrossRef]
- Singh, M.; Su, C. Progesterone, brain-derived neurotrophic factor and neuroprotection. Neuroscience 2013, 239, 84–91. [Google Scholar] [CrossRef]
- Su, C.; Cunningham, R.L.; Rybalchenko, N.; Singh, M. Progesterone increases the release of brain-derived neurotrophic factor from glia via progesterone receptor membrane component 1 (Pgrmc1)-dependent ERK5 signaling. Endocrinology 2012, 153, 4389–4400. [Google Scholar] [CrossRef]
- Bali, N.; Arimoto, J.M.; Morgan, T.E.; Finch, C.E. Progesterone antagonism of neurite outgrowth depends on microglial activation via Pgrmc1/S2R. Endocrinology 2013, 154, 2468–2480. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, C.C.; Baudry, M. Progesterone reverses 17beta-estradiol-mediated neuroprotection and BDNF induction in cultured hippocampal slices. Eur. J. Neurosci. 2009, 29, 447–454. [Google Scholar] [CrossRef]
- Nobile, B.; Maimoun, L.; Jaussent, I.D.; Seneque, M.; Dupuis-Maurin, K.; Lefebvre, P.; Courtet, P.; Renard, E.; Guillaume, S. Effects of Hormonal Contraception Use on Cognitive Functions in Patients With Bulimia Nervosa. Front. Psychiatry 2021, 12, 658182. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.D.; Le, C.S.; Zhang, H.M.; Shang, D.S.; Tong, L.S.; Gao, F. Thrombin disrupts vascular endothelial-cadherin and leads to hydrocephalus via protease-activated receptors-1 pathway. CNS Neurosci. Ther. 2019, 25, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Kondo, N.; Ogawa, M.; Wada, H.; Nishikawa, S. Thrombin induces rapid disassembly of claudin-5 from the tight junction of endothelial cells. Exp. Cell Res. 2009, 315, 2879–2887. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, F.; Mogami, H.; Moriuchi, K.; Chigusa, Y.; Mandai, M.; Kondoh, E. Mechanisms of thrombin-Induced myometrial contractions: Potential targets of progesterone. PLoS ONE 2020, 15, e0231944. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.H.; Guo, S.; Lok, J.; Navaratna, D.; Whalen, M.J.; Kim, K.W.; Lo, E.H. Neurovascular matrix metalloproteinases and the blood-brain barrier. Curr. Pharm. Des. 2012, 18, 3645–3648. [Google Scholar] [CrossRef] [PubMed]
- Lischper, M.; Beuck, S.; Thanabalasundaram, G.; Pieper, C.; Galla, H.J. Metalloproteinase mediated occludin cleavage in the cerebral microcapillary endothelium under pathological conditions. Brain Res. 2010, 1326, 114–127. [Google Scholar] [CrossRef]
- Feng, Z.H.; Hao, J.; Ye, L.; Dayao, C.; Yan, N.; Yan, Y.; Chu, L.; Shi, F.D. Overexpression of mu-calpain in the anterior temporal neocortex of patients with intractable epilepsy correlates with clinicopathological characteristics. Seizure 2011, 20, 395–401. [Google Scholar] [CrossRef]
- Ishrat, T.; Sayeed, I.; Atif, F.; Hua, F.; Stein, D.G. Progesterone and allopregnanolone attenuate blood-brain barrier dysfunction following permanent focal ischemia by regulating the expression of matrix metalloproteinases. Exp. Neurol. 2010, 226, 183–190. [Google Scholar] [CrossRef]
- Shynlova, O.; Tsui, P.; Jaffer, S.; Lye, S.J. Integration of endocrine and mechanical signals in the regulation of myometrial functions during pregnancy and labour. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 144 (Suppl. 1), S2–S10. [Google Scholar] [CrossRef]
- Fedotcheva, T.A.; Fedotcheva, N.I.; Shimanovsky, N.L. Progesterone as an Anti-Inflammatory Drug and Immunomodulator: New Aspects in Hormonal Regulation of the Inflammation. Biomolecules 2022, 12, 1299. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.J.; O’Malley, B.W. Molecular mechanisms of action of steroid/thyroid receptor superfamily members. Annu. Rev. Biochem. 1994, 63, 451–486. [Google Scholar] [CrossRef]
- Mani, S. Progestin receptor subtypes in the brain: The known and the unknown. Endocrinology 2008, 149, 2750–2756. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.D.; Wahlstrom, G.; Backstrom, T. The regional brain distribution of the neurosteroids pregnenolone and pregnenolone sulfate following intravenous infusion. J. Steroid Biochem. Mol. Biol. 1997, 62, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Coirini, H.; McEwen, B.S. Regulation of high-affinity GABAA receptors in the dorsal hippocampus by estradiol and progesterone. Brain Res. 1989, 487, 178–183. [Google Scholar] [CrossRef]
- Mendelsohn, M.E.; Karas, R.H. The protective effects of estrogen on the cardiovascular system. N. Engl. J. Med. 1999, 340, 1801–1811. [Google Scholar] [CrossRef]
- Mueck, A.O.; Seeger, H. Estrogens acting as cardiovascular agents: Direct vascular actions. Curr. Med. Chem. Cardiovasc. Hematol. Agents 2004, 2, 35–42. [Google Scholar] [CrossRef]
- Cid, M.C.; Schnaper, H.W.; Kleinman, H.K. Estrogens and the vascular endothelium. Ann. N. Y. Acad. Sci. 2002, 966, 143–157. [Google Scholar] [CrossRef]
- White, W.B. Drospirenone with 17beta-estradiol in the postmenopausal woman with hypertension. Climacteric 2007, 10 (Suppl. 1), 25–31. [Google Scholar] [CrossRef]
- Imthurn, B.; Rosselli, M.; Jaeger, A.W.; Keller, P.J.; Dubey, R.K. Differential effects of hormone-replacement therapy on endogenous nitric oxide (nitrite/nitrate) levels in postmenopausal women substituted with 17 beta-estradiol valerate and cyproterone acetate or medroxyprogesterone acetate. J. Clin. Endocrinol. Metab. 1997, 82, 388–394. [Google Scholar] [CrossRef]
- Leonhardt, S.A.; Boonyaratanakornkit, V.; Edwards, D.P. Progesterone receptor transcription and non-transcription signaling mechanisms. Steroids 2003, 68, 761–770. [Google Scholar] [PubMed]
- Fu, X.D.; Flamini, M.; Sanchez, A.M.; Goglia, L.; Giretti, M.S.; Genazzani, A.R.; Simoncini, T. Progestogens regulate endothelial actin cytoskeleton and cell movement via the actin-binding protein moesin. Mol. Hum. Reprod. 2008, 14, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Boonyaratanakornkit, V.; McGowan, E.; Sherman, L.; Mancini, M.A.; Cheskis, B.J.; Edwards, D.P. The role of extranuclear signaling actions of progesterone receptor in mediating progesterone regulation of gene expression and the cell cycle. Mol. Endocrinol. 2007, 21, 359–375. [Google Scholar] [CrossRef] [PubMed]
- Boonyaratanakornkit, V.; Edwards, D.P. Receptor mechanisms of rapid extranuclear signalling initiated by steroid hormones. Essays Biochem. 2004, 40, 105–120. [Google Scholar]
- Boonyaratanakornkit, V.; Bi, Y.; Rudd, M.; Edwards, D.P. The role and mechanism of progesterone receptor activation of extra-nuclear signaling pathways in regulating gene transcription and cell cycle progression. Steroids 2008, 73, 922–928. [Google Scholar] [CrossRef]
- Abou-Fadel, J.; Qu, Y.; Gonzalez, E.; Smith, M.; Zhang, J. Emerging roles of CCM genes during tumorigenesis with potential application as novel biomarkers across major types of cancers. Oncol. Rep. 2020, 43, 1945–1963. [Google Scholar] [CrossRef]
- Zhang, J. Learn from the past, review the present, and look towards the future. Vessel Plus 2022, 6, 20. [Google Scholar]
| Main Point | Key Findings | Genes Involved | Key References |
|---|---|---|---|
| Key factors influencing on the blood vessel permeability | The blood brain barrier (BBB, the blood vessel barrier) in the brain, is a dynamic and complex interface that tightly controls the molecular exchanges between the blood and central nervous system (CNS) the integrity of both BVB and BBB are determined by EC junctions that are made up of mainly adherens junctions (AJs) and tight junctions (TJs), which can be influenced by steroids. | JAMs, claudins, occludins, VE-cadherin, N-cadherin, VCAM-1, ICAM-1, | [15,16,27,28,73,75] |
| Angiogenic impacts of steroids on maintenance of vasculature | Steroids might play major roles on maintainence of BBB integrity, through their regulation of EC junction components, dysregulation of steroid-mediasted signaling pathway can lead to compromised BBB, therefore, steroids can be potential therapeutic targets. | MRs, GRs, GRα, GRβ, ERs, nPRs, mPRs, eNOS | [78,81,83,99] |
| Impacts of sex steroids on maintenance of vasculature | Sex steroids might play major roles on neovascularization, angiogenic signaling, and maintainence of BBB integrity, therefore, steroids can be potential therapeutic targets. | bFGF, PD-ECGF, HIF1a, Ang, NOS, eNOS, PI3Ks, Akt, MAPKs | [150,151,152,155] |
| Angiogenic impacts of progesterone (PRG) on the Neurovascular Unit (NVU) | PRGs have neuroprotective impact on the Neurovascular Unit (NVU) | BDNF, PGRMC1, Bcl-2, PAR-1, Claudin-5, MMP, NF-κB, IL-10, GABA-A receptor | [147,150,151,153] |
| Impacts of CmPn/CmP signaling networks on the BBB | PRG and its derivatives-mediated signaling pathways have major impacts on EC performance in vasculature. PRG and its derivatives-mediated CmPn/CmP signaling networks maintain the BBB integrity | CCM (1–3) genes, mPRs, nPRs, | [8,46,124,125,198] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gnanasekaran, R.; Aickareth, J.; Hawwar, M.; Sanchez, N.; Croft, J.; Zhang, J. CmPn/CmP Signaling Networks in the Maintenance of the Blood Vessel Barrier. J. Pers. Med. 2023, 13, 751. https://doi.org/10.3390/jpm13050751
Gnanasekaran R, Aickareth J, Hawwar M, Sanchez N, Croft J, Zhang J. CmPn/CmP Signaling Networks in the Maintenance of the Blood Vessel Barrier. Journal of Personalized Medicine. 2023; 13(5):751. https://doi.org/10.3390/jpm13050751
Chicago/Turabian StyleGnanasekaran, Revathi, Justin Aickareth, Majd Hawwar, Nickolas Sanchez, Jacob Croft, and Jun Zhang. 2023. "CmPn/CmP Signaling Networks in the Maintenance of the Blood Vessel Barrier" Journal of Personalized Medicine 13, no. 5: 751. https://doi.org/10.3390/jpm13050751
APA StyleGnanasekaran, R., Aickareth, J., Hawwar, M., Sanchez, N., Croft, J., & Zhang, J. (2023). CmPn/CmP Signaling Networks in the Maintenance of the Blood Vessel Barrier. Journal of Personalized Medicine, 13(5), 751. https://doi.org/10.3390/jpm13050751








