Stenting in Brain Hemodynamic Injury of Carotid Origin Caused by Type A Aortic Dissection: Local Experience and Systematic Literature Review
Abstract
1. Introduction
- −
- An extension of the dissection to the supra-aortic branches, with carotid or vertebral occlusion, by compression of the true lumen, or thrombo-embolic mechanism arising from the false lumen;
- −
2. Materials and Methods
2.1. Ethics
2.2. Local Center Patients’ Selection
2.3. Literature Review
2.4. Data Extraction and Expression of Results
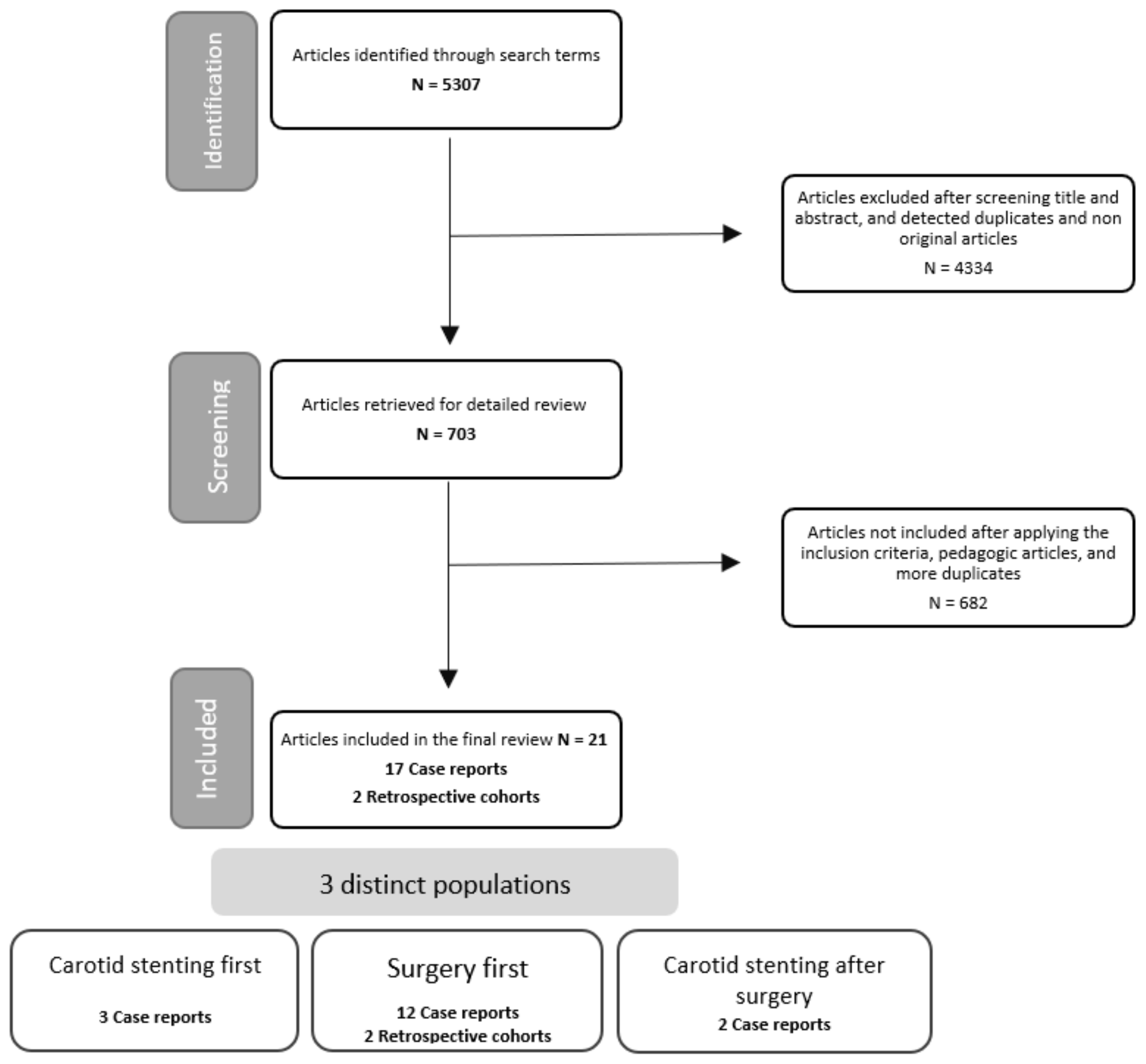
3. Results
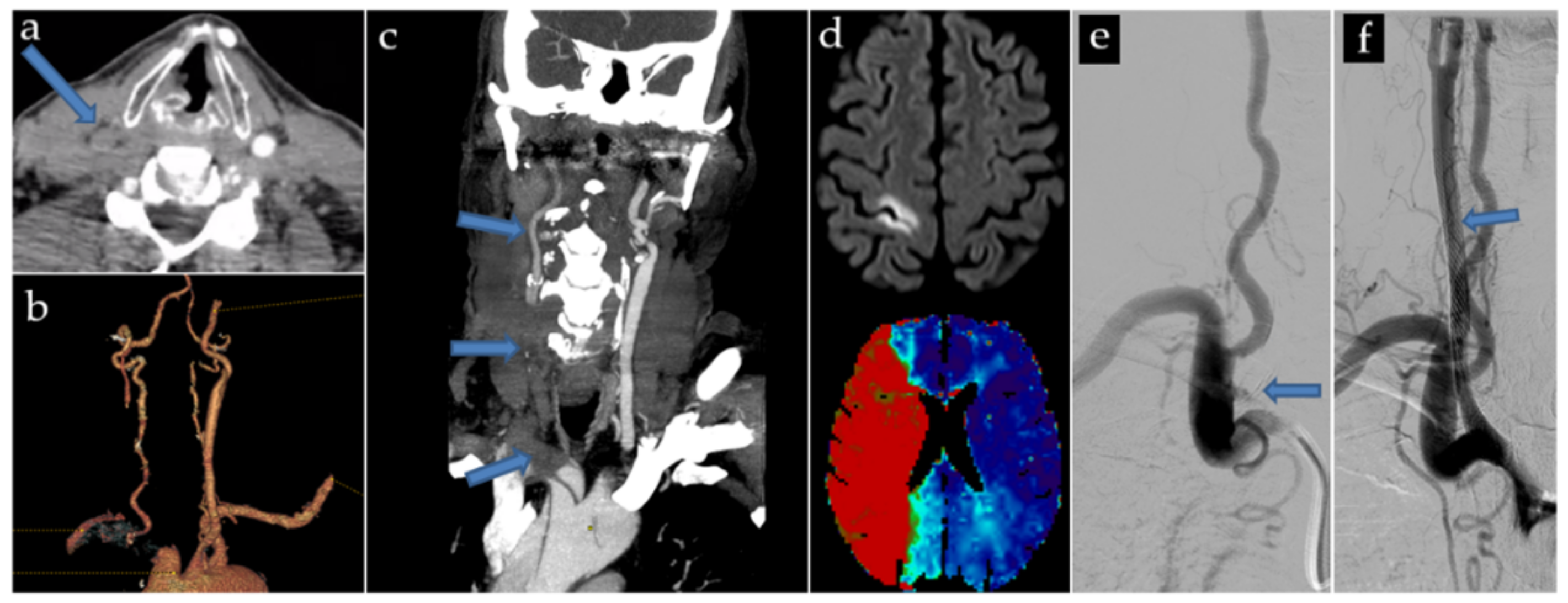
3.1. Local Center Patients’ Selection (n = 9)
3.2. Literature Review (n = 73)
| Study | Year | Study Type | Number of Patients Included | First Treatment |
|---|---|---|---|---|
| Schönholz et al. [15] | 2008 | Case report | 1 | Surgery |
| Chahine et al. [16] | 2018 | Case report | 2 | Surgery |
| Morihara et al. [17] | 2016 | Case report | 1 | Surgery |
| Matsumoto et al. [18] | 2016 | Case report | 2 | Surgery |
| Amr et al. [13] | 2016 | Case report | 2 | Surgery |
| Hong et al. [19] | 2005 | Case report | 1 | Surgery |
| Karawabuki et al. [20] | 2006 | Case report | 1 | Surgery |
| Kim et al. [29] | 2006 | Case report | 1 | Surgery |
| Ueyama et al. [22] | 2007 | Case report | 1 | Surgery |
| Roseborough et al. [11] | 2006 | Case report | 1 | Surgery then stenting |
| Sakaguchi et al. [23] | 2005 | Case report | 1 | Surgery |
| Usui et al. [24] | 2021 | Case report | 1 | Surgery |
| Fukuhara et al. [25] | 2021 | Case report | 1 | Surgery |
| Funakoshi et al. [26] | 2020 | Case report | 2 | Stenting then surgery |
| Heran et al. [27] | 2019 | Case report | 1 | Stenting then surgery |
| Popovic et al. [14] | 2016 | Case report | 1 | Stenting then surgery |
| Casana et al. [28] | 2011 | Case report | 1 | Surgery then stenting |
| Fichadaya et al. [12] | 2022 | Cohort | 10 | Surgery |
| Morimoto et al. [30] | 2011 | Cohort | 41 | Surgery |
3.3. Aortic and Carotid Surgery (ACS) Group (n = 72)
3.4. Carotid Stenting (CS) before Aortic Surgery (AS) Group (n = 8)
3.5. Carotid Stenting (CS) after Aortic Surgery (AS) Group (n = 7)
- −
- A 78 yo patient presented with a distal A2 embolus, too distant to be accessible by thrombectomy. Three 9 × 30 mm Carotid Wallstents were used, covering the entirety of the brachiocephalic trunk and right common carotid artery. After deployment of the first two Carotid Wallstents, an intra-stent thrombosis occurred, and was immediately and successfully treated by aspiration, and was not recurrent. Antiplatelet treatment was started the next day.
- −
- A 74 yo patient presented a sylvian M2 embolus at the end of the procedure, successfully treated by thrombectomy immediately after stenting with three Carotid Wallstent (7 × 40 mm, 9 × 50 mm and 5 × 30 mm) and a Smart Control 14 × 40 mm. Antiplatelet treatment and preventive low molecular weight Heparin were started the next day.
- −
- A 62 yo patient presented multiple distal emboli not accessible to thrombectomy. Antiplatelet treatment and curative low molecular weight Heparin were started 12 h post-procedure.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sukockienė, E.; Laučkaitė, K.; Jankauskas, A.; Mickevičienė, D.; Jurkevičienė, G.; Vaitkus, A.; Stankevičius, E.; Petrikonis, K.; Rastenytė, D. Crucial role of carotid ultrasound for the rapid diagnosis of hyperacute aortic dissection complicated by cerebral infarction: A case report and literature review. Medicina 2016, 52, 378–388. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, P.R.; Wolfson, A.B.; Leckey, R.D.; Burke, J.L. Diagnosis of acute thoracic aortic dissection in the emergency department. Am. J. Emerg. Med. 2000, 18, 46–50. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, J.B.; Lansman, S.L.; Kai, M.; Tang, G.H.L.; Malekan, R.; Spielvogel, D. Malperfusion in Type A Dissection: Consider Reperfusion First. Semin. Thorac. Cardiovasc. Surg. 2017, 29, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Ma, W.; Wen, D.; Duan, W.; Zheng, M. Computed tomography angiography findings predict the risk factors for preoperative acute ischaemic stroke in patients with acute type A aortic dissection. Eur. J. Cardio-Thorac. Surg. 2020, 57, 912–919. [Google Scholar] [CrossRef]
- Chemtob, R.A.; Fuglsang, S.; Geirsson, A.; Ahlsson, A.; Olsson, C.; Gunn, J.; Ahmad, K.; Hansson, E.C.; Pan, E.; O Arnadottir, L.; et al. Stroke in acute type A aortic dissection: The Nordic Consortium for Acute Type A Aortic Dissection (NORCAAD). Eur. J. Cardio-Thorac. Surg. 2020, 58, 1027–1034. [Google Scholar] [CrossRef]
- Okita, Y.; Okada, K. Treatment strategies for malperfusion syndrome secondary to acute aortic dissection. J. Card Surg. 2021, 36, 1745–1752. [Google Scholar] [CrossRef]
- Czerny, M.; Schoenhoff, F.; Etz, C.; Englberger, L.; Khaladj, N.; Zierer, A.; Weigang, E.; Hoffmann, I.; Blettner, M.; Carrel, T.P. The Impact of Pre-Operative Malperfusion on Outcome in Acute Type A Aortic Dissection. J. Am. Coll. Cardiol. 2015, 65, 2628–2635. [Google Scholar] [CrossRef]
- Geirsson, A.; Szeto, W.Y.; Pochettino, A.; McGarvey, M.L.; Keane, M.G.; Woo, Y.J.; Augoustides, J.G.; Bavaria, J.E. Significance of malperfusion syndromes prior to contemporary surgical repair for acute type A dissection: Outcomes and need for additional revascularizations. Eur. J. Cardio-Thorac. Surg. 2007, 32, 255–262. [Google Scholar] [CrossRef]
- Kreibich, M.; Desai, N.D.; Bavaria, J.E.; Szeto, W.Y.; Vallabhajosyula, P.; Beyersdorf, F.; Czerny, M.; Siepe, M.; Rylski, B.; Itagaki, R.; et al. Common carotid artery true lumen flow impairment in patients with type A aortic dissection. Eur. J. Cardio-Thorac. Surg. 2021, 59, 490–496. [Google Scholar] [CrossRef]
- Munir, W.; Chong, J.H.; Harky, A.; Bashir, M.; Adams, B. Type A aortic dissection: Involvement of carotid artery and impact on cerebral malperfusion. Asian Cardiovasc Thorac Ann. 2021, 29, 635–642. [Google Scholar] [CrossRef]
- Roseborough, G.S.; Murphy, K.P.; Barker, P.B.; Sussman, M. Correction of symptomatic cerebral malperfusion due to acute type I aortic dissection by transcarotid stenting of the innominate and carotid arteries. J. Vasc. Surg. 2006, 44, 1091–1096. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fichadiya, A.; Menon, B.K.; Gregory, A.J.; Teleg, E.; Appoo, J.J. Neuroanatomy and severity of stroke in patients with type A aortic dissection. J. Card. Surg. 2022, 37, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Amr, G.; Boulouis, G.; Bricout, N.; Modine, T.; Fayad, G.; Aguettaz, P.; Koussa, M. Stroke Presentation of Acute Type A Aortic Dissection with 100% Perfusion-Weighted Imaging–Diffusion-Weighted Imaging Mismatch: A Call for Urgent Action. J. Stroke Cerebrovasc. Dis. 2016, 25, 1280–1283. [Google Scholar] [CrossRef] [PubMed]
- Popovic, R.; Radovinovic-Tasic, S.; Rusovic, S.; Lepic, T.; Ilic, R.; Raicevic, R.; Obradovic, D. Urgent carotid stenting before cardiac surgery in a young male patient with acute ischemic stroke caused by aortic and carotid dissection. VSP 2016, 73, 674–678. [Google Scholar] [CrossRef]
- Schönholz, C.; Ikonomidis, J.S.; Hannegan, C.; Mendaro, E. Bailout Percutaneous External Shunt to Restore Carotid Flow in a Patient With Acute Type A Aortic Dissection and Carotid Occlusion. J. Endovasc. Ther. 2008, 15, 639–642. [Google Scholar] [CrossRef]
- Chahine, J.; Thapa, B.; Gajulapalli, R.D.; Kadri, A. Acute Aortic Dissection Presenting with a Headache: An Easily Missed Life-threatening Emergency. Cureus 2018, 10, e3531. [Google Scholar] [CrossRef]
- Morihara, R.; Yamashita, T.; Deguchi, K.; Tsunoda, K.; Manabe, Y.; Takahashi, Y.; Yunoki, T.; Sato, K.; Nakano, Y.; Kono, S.; et al. Successful Delayed Aortic Surgery for a Patient with Ischemic Stroke Secondary to Aortic Dissection. Int. Med. 2017, 56, 2343–2346. [Google Scholar] [CrossRef][Green Version]
- Matsumoto, H.; Yoshida, Y.; Hirata, Y. Usefulness of cervical magnetic resonance imaging for detecting type A acute aortic dissection with acute stroke symptoms. Magn. Reson. Imaging 2016, 34, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Hong, K.-S.; Park, S.-Y.; Whang, S.-I.; Seo, S.-Y.; Lee, D.-H.; Kim, H.-J.; Cho, J.-Y.; Cho, Y.-J.; Jang, W.-I.; Kim, C.Y. Intravenous Recombinant Tissue Plasminogen Activator Thrombolysis in a Patient with Acute Ischemic Stroke Secondary to Aortic Dissection. J. Clin. Neurol. 2009, 5, 49. [Google Scholar] [CrossRef]
- Kawarabuki, K.; Sakakibara, T.; Hirai, M.; Shirasu, M.; Kohara, I.; Tanaka, H.; Oyamada, M.; Takamatsu, T.; Murayama, Y.; Yamaki, T. Acute Aortic Dissection Presenting as a Neurologic Disorder. J. Stroke Cerebrovasc. Dis. 2006, 15, 26–29. [Google Scholar] [CrossRef]
- Kim, S.H.; Song, S.; Kim, S.-P.; Lee, J.; Lee, H.C.; Kim, E.S. Hybrid technique to correct cerebral malperfusion following repair of a type a aortic dissection. Korean J. Thorac. Cardiovasc. Surg. 2014, 47, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Ueyama, K.; Otaki, K.; Koyama, M.; Kamiyama, H. Urgent simultaneous revascularization of the carotid artery and ascending aortic replacement for type A acute aortic dissection with cerebral malperfusion. Gen. Thorac. Cardiovasc. Surg. 2007, 55, 284–286. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, G.; Komiya, T.; Tamura, N.; Obata, S.; Masuyama, S.; Kimura, C.; Kobayashi, T. Cerebral malperfusion in acute type A dissection: Direct innominate artery cannulation. J. Thorac. Cardiovasc. Surg. 2005, 129, 1190–1191. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Usui, T.; Suzuki, K.; Niinami, H.; Sakai, S. Aortic dissection diagnosed on stroke computed tomography protocol: A case report. J. Med. Case Rep. 2021, 15, 299. [Google Scholar] [CrossRef] [PubMed]
- Fukuhara, S.; Norton, E.L.; Chaudhary, N.; Burris, N.; Shiomi, S.; Kim, K.M.; Patel, H.J.; Deeb, G.M.; Yang, B. Type A Aortic Dissection With Cerebral Malperfusion: New Insights. Ann. Thorac. Surg. 2021, 112, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Funakoshi, Y.; Imamura, H.; Tokunaga, S.; Murakami, Y.; Tani, S.; Adachi, H.; Ohara, N.; Kono, T.; Fukumitsu, R.; Sunohara, T.; et al. Carotid artery stenting before surgery for carotid artery occlusion associated with acute type A aortic dissection: Two case reports. Interv. Neuroradiol. 2020, 26, 814–820. [Google Scholar] [CrossRef]
- Heran, M.K.S.; Balaji, N.; Cook, R.C. Novel Percutaneous Treatment of Cerebral Malperfusion Before Surgery for Acute Type A Dissection. Ann. Thorac. Surg. 2019, 108, e15–e17. [Google Scholar] [CrossRef]
- Casana, R.; Tolva, V.; Majnardi, A.R.; Bianchi, P.G.; Addobati, L.; Bertoni, G.B.; Cireni, L.V.; Silani, V. Endovascular Management of Symptomatic Cerebral Malperfusion Due to Carotid Dissection After Type A Aortic Dissection Repair. Vasc Endovasc. Surg. 2011, 45, 641–645. [Google Scholar] [CrossRef]
- Sik Kim, Y.; Chernyshev, O.Y.; Alexandrov, A.V. Nonpulsatile Cerebral Perfusion in Patient With Acute Neurological Deficits. Stroke 2006, 37, 1562–1564. [Google Scholar] [CrossRef]
- Morimoto, N.; Okada, K.; Okita, Y. Lack of neurologic improvement after aortic repair for acute type A aortic dissection complicated by cerebral malperfusion: Predictors and association with survival. J. Thorac. Cardiovasc. Surg. 2011, 142, 1540–1544. [Google Scholar] [CrossRef]
- Igarashi, T.; Takahashi, S.; Takase, S.; Yokoyama, H. Intraoperative thrombectomy for occluded carotid arteries in patients with acute aortic dissection: Report of two cases. Surg. Today 2014, 44, 1177–1179. [Google Scholar] [CrossRef]
- Fabre, O.; Guesnier, L.; Renaut, C.; Gautier, L.; Geronimi, H.; Jasaitis, L.; Strauch, K. Prise en charge actuelle des dissections aortiques de type A. Traitement chirurgical et traitement des syndromes de malperfusion. Ann. De Cardiol. Et D’angéiologie 2005, 54, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Lentini, S.; Tancredi, F.; Benedetto, F.; Gaeta, R. Type A aortic dissection involving the carotid arteries: Carotid stenting during open aortic arch surgery. Interact Cardiovasc. Thorac. Surg. 2009, 8, 157–159. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, D.; Lewis, E.; Spinosa, D.; Tang, D.; Ryan, L. Retrograde Carotid Stenting Using Newly Released Venous Stent for Cerebral Malperfusion in Type A Aortic Dissection. J. Endovasc. 2022, 29, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Wang, Y.; Chen, Y.; Jiao, L. Open retrograde endovascular stenting for left common carotid artery dissection secondary to surgical repair of acute aortic dissection: A case report and review of the literature. Ann. Vasc Surg. 2015, 29, e11–e15. [Google Scholar] [CrossRef]
- Tsai, K.-T.; Shen, T.-C. Challenging carotid intervention after total arch rerouting and hybrid zone 0 elephant trunk repair for a complicated type A aortic dissection. J. Endovasc. 2014, 21, 306–311. [Google Scholar] [CrossRef]
- Cardaioli, P.; Rigatelli, G.; Giordan, M.; Faggian, G.; Chinaglia, M.; Roncon, L. Multiple carotid stenting for extended thoracic aorta dissection after initial aortic surgical repair. Cardiovasc. Revasc. Med. 2007, 8, 213–215. [Google Scholar] [CrossRef]
- Pavkov, I.; Horner, S.; Klein, G.E.; Niederkorn, K. Percutaneous transluminal angioplasty with stenting in extended supra-aortic artery dissection. Croat. Med. J. 2004, 45, 217–219. [Google Scholar]
- Neri, E.; Sani, G.; Massetti, M.; Frati, G.; Buklas, D.; Tassi, R.; Giubbolini, M.; Benvenuti, A.; Sassi, C. Residual dissection of the brachiocephalic arteries: Significance, management, and long-term outcome. J. Thorac. Cardiovasc. Surg. 2004, 128, 303–312. [Google Scholar] [CrossRef]
| Patient | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
|---|---|---|---|---|---|---|---|---|---|
| Treatment | Carotid stenting before aortic surgery | Carotid stenting after aortic surgery | |||||||
| Age | 71 | 71 | 72 | 72 | 78 | 64 | 71 | 74 | 62 |
| Sex | M | M | M | M | F | M | F | F | F |
| Arterial hypertension | yes | No | yes | yes | yes | yes | yes | no | yes |
| Current smoking | No | No | Yes | No | No | Yes | No | No | yes |
| Pre-stroke MRS | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| NIHSS on presentation | 21 | NA | 10 | 3 | NA | 0 | 0 | 0 | 0 |
| ASPECTS score | 7 | 10 | 8 | 9 | 10 | 10 | 8 | NA | NA |
| Supra-aortic branch involved | RCCAand ICA, LCCA and ICA | RCCA and ICA | RCCA and ICA | RCCA | RCCA | LCCA | RCCA | RCCA and ICA, LCCA | RCCA |
| Per-procedure complications | No | No | No | No | Yes (common carotid intra-stent thrombosis treated by aspiration, A2 distal emboli) | No | No | Yes M2 (sylvian emboli) | Yes (distal emboli) |
| New lesion on post-procedure MRI | No | No | No | Yes | No | No | No | Yes (junctionnal infarct) | Yes (diffuse distal ischemic lesions) |
| New neurological deficit post-procedure | No | No | No | No | No | Yes (transient majoration due to reperfusion oedemea) | No | No | No |
| Post-procedure neurologic deficit regression | Partial | Partial | Partial | No | No | Partial | Partial | No | No |
| Survived | Yes | No Mesenteric ischemia (day 2) | Yes | Yes | No Neurologic degradation (day 8) | Yes | Yes | No Cardiac arrest (day 4) | NA |
| MRS at discharge | 2 | - | 4 | NA | - | 1 | 4 | - | NA |
| MRS after 90 days | 1 | - | 3 | 1 | - | 1 | 1 | - | NA |
| Sent permeability at follow-up | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | NA |
| Carotid Stenting First | Aortic and Carotid Surgery First |
|---|---|
| Benefits | |
| Local anesthesia | Faster repair of ascending aorta |
| Faster installation (15 min of installation on table) | Correction of systemic hypotension |
| Treatment of distal internal carotid artery dissections | |
| Complete exclusion of the false lumen | |
| Drawbacks | |
| Double antiaggregation post-procedure: postponed surgery | Longer preparation |
| Difficulty in catheterizing the true lumen | General anesthesia |
| Restriction of surgical options: impossibility of total arch replacement when stents cover the origin of supra-aortic branches | Risk of intraoperative hypoperfusion under cardiopulmonary bypass |
| Risk of distal embolization per procedure | Risk of residual dissection and/or cerebral malperfusion if distal repair is impossible |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aita, J.-F.; Agripnidis, T.; Testud, B.; Barral, P.-A.; Jacquier, A.; Reyre, A.; Alnuaimi, A.; Girard, N.; Tradi, F.; Habert, P.; et al. Stenting in Brain Hemodynamic Injury of Carotid Origin Caused by Type A Aortic Dissection: Local Experience and Systematic Literature Review. J. Pers. Med. 2023, 13, 58. https://doi.org/10.3390/jpm13010058
Aita J-F, Agripnidis T, Testud B, Barral P-A, Jacquier A, Reyre A, Alnuaimi A, Girard N, Tradi F, Habert P, et al. Stenting in Brain Hemodynamic Injury of Carotid Origin Caused by Type A Aortic Dissection: Local Experience and Systematic Literature Review. Journal of Personalized Medicine. 2023; 13(1):58. https://doi.org/10.3390/jpm13010058
Chicago/Turabian StyleAita, Jean-François, Thibault Agripnidis, Benoit Testud, Pierre-Antoine Barral, Alexis Jacquier, Anthony Reyre, Ammar Alnuaimi, Nadine Girard, Farouk Tradi, Paul Habert, and et al. 2023. "Stenting in Brain Hemodynamic Injury of Carotid Origin Caused by Type A Aortic Dissection: Local Experience and Systematic Literature Review" Journal of Personalized Medicine 13, no. 1: 58. https://doi.org/10.3390/jpm13010058
APA StyleAita, J.-F., Agripnidis, T., Testud, B., Barral, P.-A., Jacquier, A., Reyre, A., Alnuaimi, A., Girard, N., Tradi, F., Habert, P., Gariboldi, V., Collart, F., Bartoli, A., & Hak, J.-F. (2023). Stenting in Brain Hemodynamic Injury of Carotid Origin Caused by Type A Aortic Dissection: Local Experience and Systematic Literature Review. Journal of Personalized Medicine, 13(1), 58. https://doi.org/10.3390/jpm13010058






