Computer-Aided Histopathological Characterisation of Endometriosis Lesions
Abstract
:1. Introduction
2. Material and Methods
2.1. Sample and Pain Collection
2.2. Immunodetection of Epithelial and Stromal Cells in Excised Lesion Tissue
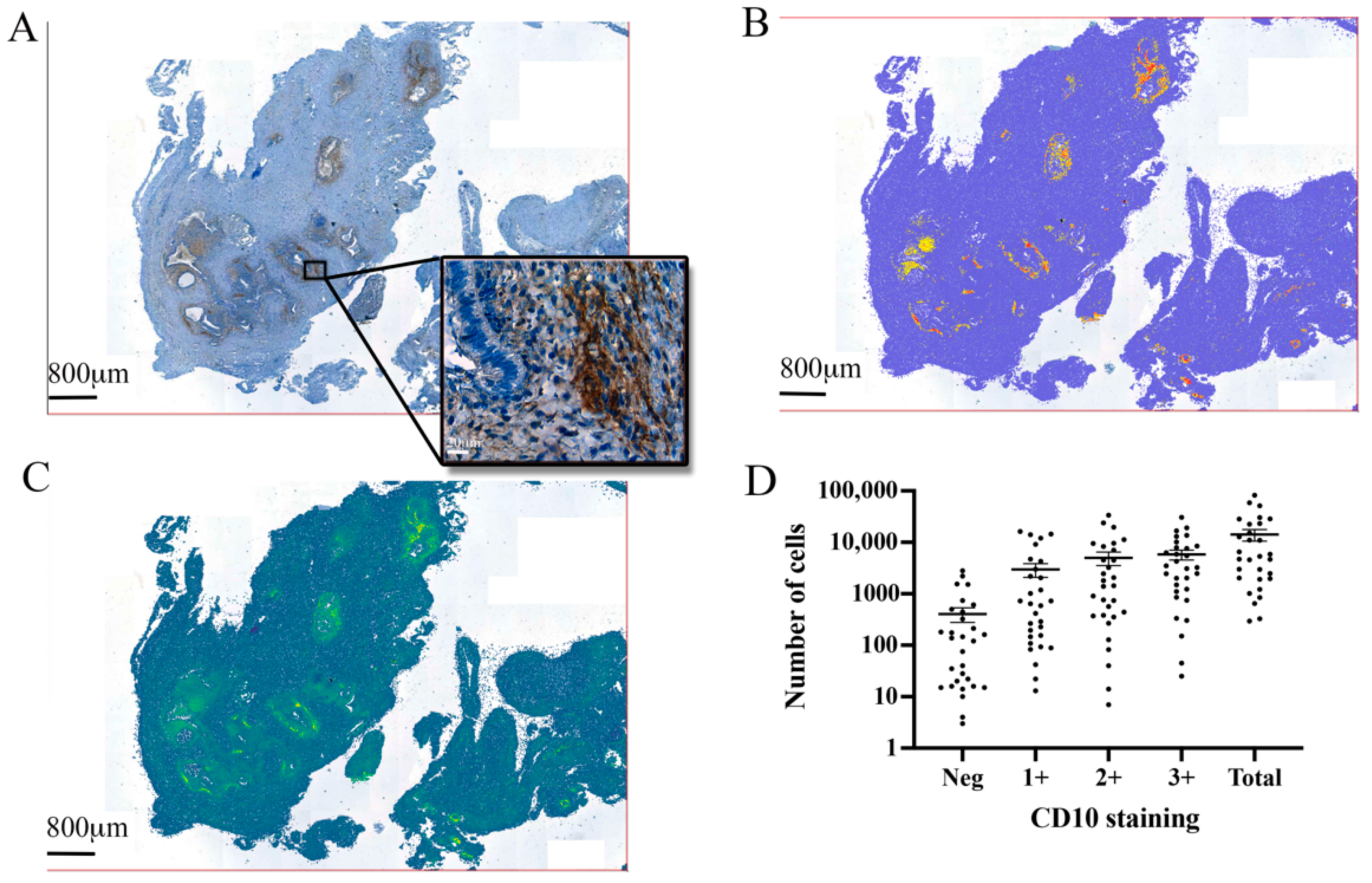
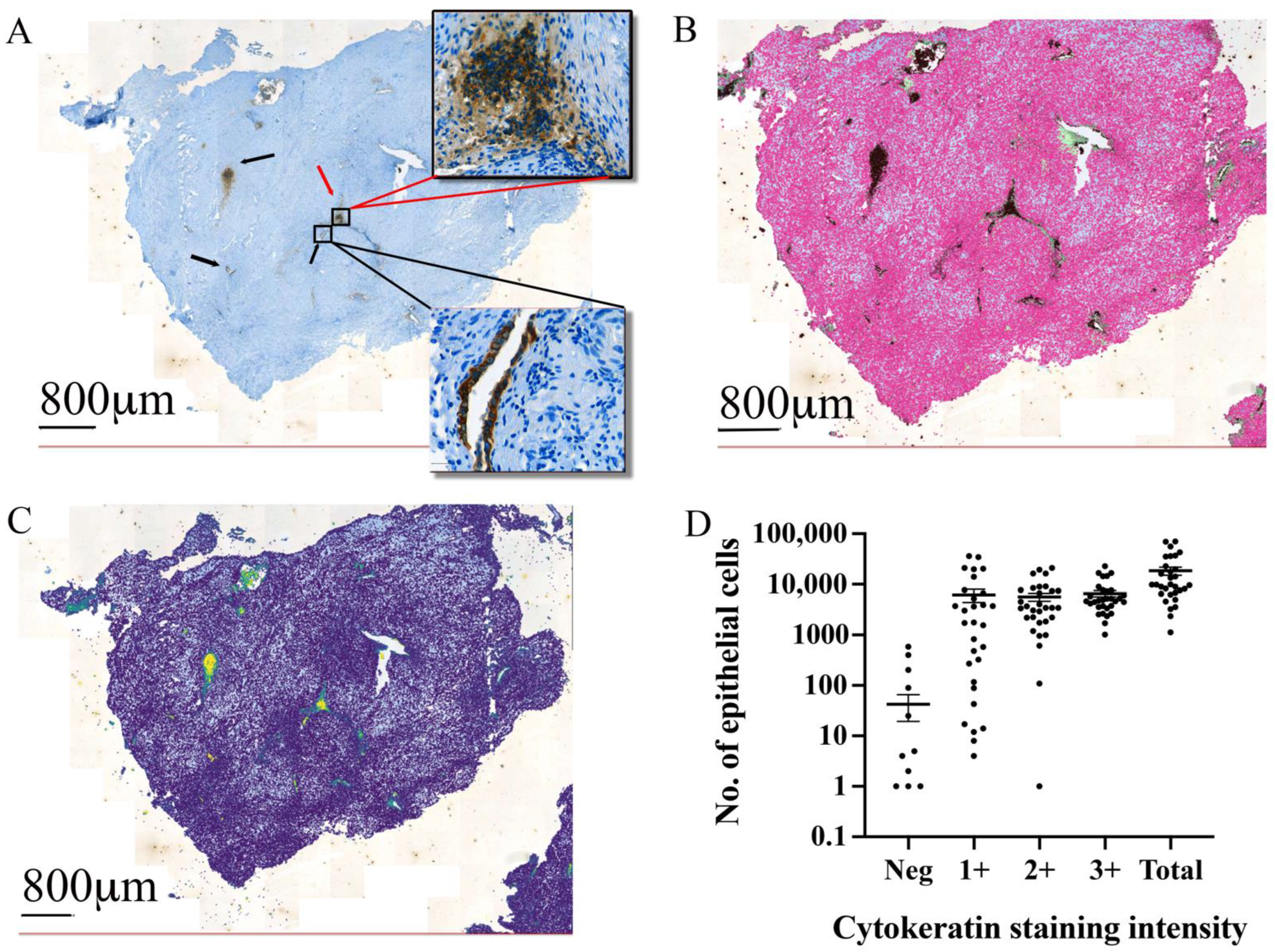
2.3. Tissue Section Digitisation and Automated Cell Detection
2.4. Statistical Analysis
3. Results
3.1. Patient Data
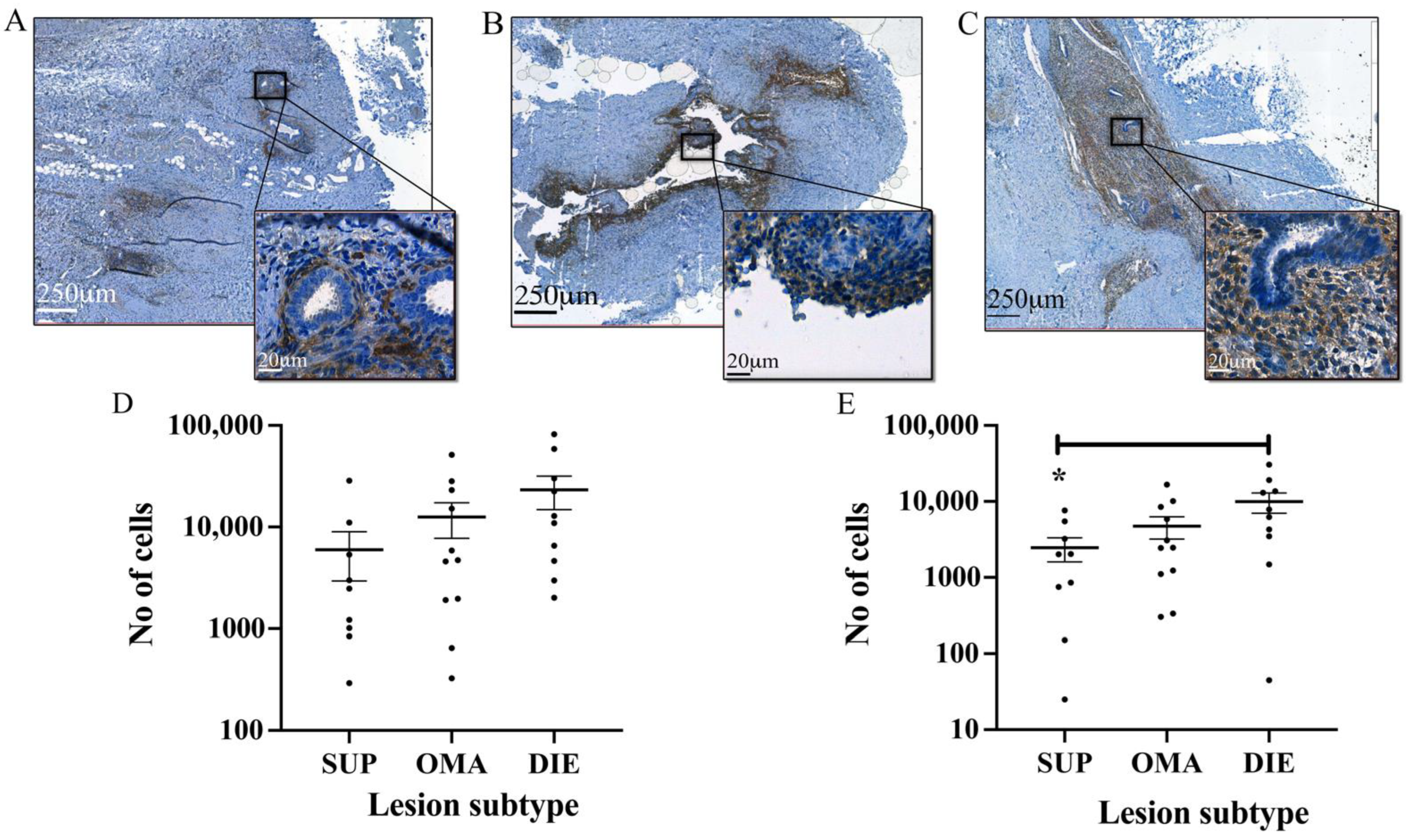
3.2. Automated Endometrial Stromal Cell Detection and Quantification in Excised Tissue
3.3. Automated Epithelial Cell Detection and Quantification in Excised Tissue
3.4. Comparison between the Epithelial and Stromal Contents in Lesions
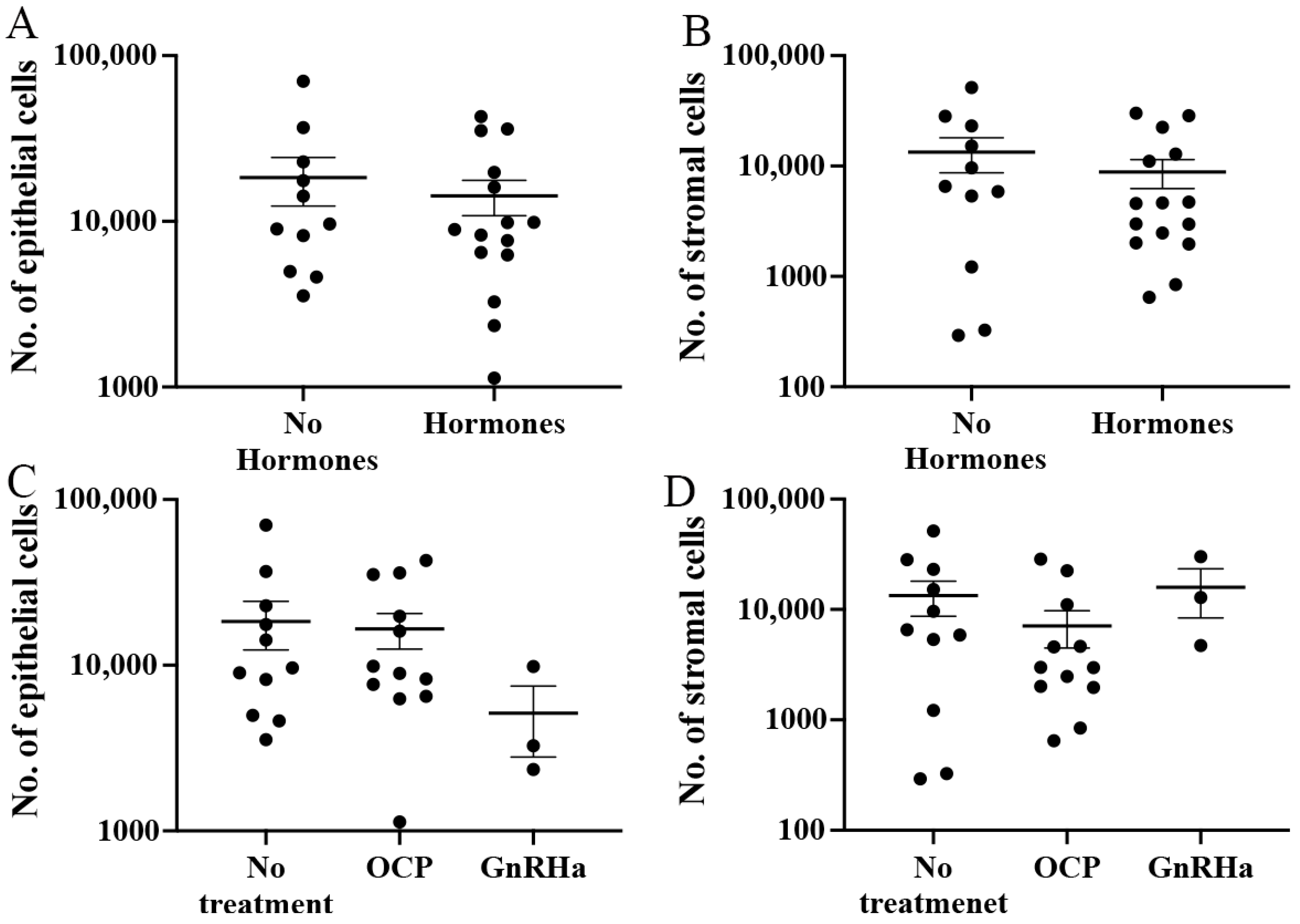
3.5. Influence of Hormonal Treatment on Endometriotic Lesion Cells and Their Ratio
3.6. Association between Automated Cell Counts, Stage of Disease, and Patient Symptoms
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Tomassetti, C.; D’Hooghe, T. Endometriosis and infertility: Insights into the causal link and management strategies. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 51, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Ballard, K.D.; Seaman, H.E.; de Vries, C.S.; Wright, J.T. Can symptomatology help in the diagnosis of endometriosis? Findings from a national case-control study—Part 1. Bjog 2008, 115, 1382–1391. [Google Scholar] [CrossRef] [PubMed]
- Chapron, C.; Marcellin, L.; Borghese, B.; Santulli, P. Rethinking mechanisms, diagnosis and management of endometriosis. Nat. Rev. Endocrinol. 2019, 15, 666–682. [Google Scholar] [CrossRef] [PubMed]
- Becker, C.M.; Bokor, A.; Heikinheimo, O.; Horne, A.; Jansen, F.; Kiesel, L.; King, K.; Kvaskoff, M.; Nap, A.; Petersen, K.; et al. ESHRE guideline: Endometriosis. Hum. Reprod. Open 2022, 2022, hoac009. [Google Scholar] [CrossRef] [PubMed]
- Donnez, J.; Nisolle, M.; Casanas-Roux, F.; Bassil, S.; Anaf, V. Surgery: Rectovaginal septum, endometriosis or adenomyosis: Laparoscopic management in a series of 231 patients. Hum. Reprod. 1995, 10, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Chapron, C.; Souza, C.; Borghese, B.; Lafay-Pillet, M.-C.; Santulli, P.; Bijaoui, G.; Goffinet, F.; de Ziegler, D. Oral contraceptives and endometriosis: The past use of oral contraceptives for treating severe primary dysmenorrhea is associated with endometriosis, especially deep infiltrating endometriosis. Hum. Reprod. 2011, 26, 2028–2035. [Google Scholar] [CrossRef]
- Bankhead, P.; Loughrey, M.B.; Fernández, J.A.; Dombrowski, Y.; McArt, D.G.; Dunne, P.D.; McQuaid, S.; Gray, R.T.; Murray, L.J.; Coleman, H.G.; et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 2017, 7, 16878. [Google Scholar] [CrossRef]
- Rowlands, I.J.; Abbott, J.A.; Montgomery, G.W.; Hockey, R.; Rogers, P.; Mishra, G.D. Prevalence and incidence of endometriosis in Australian women: A data linkage cohort study. Bjog 2020, 128, 657–665. [Google Scholar] [CrossRef]
- McKinnon, B.D.; Bertschi, D.; Bersinger, N.A.; Mueller, M.D. Inflammation and nerve fiber interaction in endometriotic pain. Trends Endocrinol. Metab. 2015, 26, 1–10. [Google Scholar] [CrossRef]
- Saunders, P.T.K.; Horne, A.W. Endometriosis: Etiology, pathobiology, and therapeutic prospects. Cell 2021, 184, 2807–2824. [Google Scholar] [CrossRef]
- Anglesio, M.S.; Papadopoulos, N.; Ayhan, A.; Nazeran, T.M.; Noe, M.; Horlings, H.M.; Lum, A.; Jones, S.; Senz, J.; Seckin, T.; et al. Cancer-Associated Mutations in Endometriosis without Cancer. N. Engl. J. Med. 2017, 376, 1835–1848. [Google Scholar] [CrossRef] [PubMed]
- Lac, V.; Nazeran, T.M.; Tessier-Cloutier, B.; Aguirre-Hernandez, R.; Albert, A.; Lum, A.; Khattra, J.; Praetorius, T.; Mason, M.; Chiu, D.; et al. Oncogenic mutations in histologically normal endometrium: The new normal? J. Pathol. 2019, 249, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Grandi, G.; Mueller, M.; Bersinger, N.; Papadia, A.; Nirgianakis, K.; Cagnacci, A.; McKinnon, B. Progestin suppressed inflammation and cell viability of tumor necrosis factor-alpha-stimulated endometriotic stromal cells. Am. J. Reprod. Immunol. 2016, 76, 292–298. [Google Scholar] [CrossRef]
- Grandi, G.; Mueller, M.; Bersinger, N.A.; Cagnacci, A.; Volpe, A.; McKinnon, B. Does dienogest influence the inflammatory response of endometriotic cells? A systematic review. Inflamm. Res. 2016, 65, 183–192. [Google Scholar] [CrossRef]
- Grandi, G.; Mueller, M.D.; Papadia, A.; Kocbek, V.; Bersinger, N.A.; Petraglia, F.; Cagnacci, A.; McKinnon, B. Inflammation influences steroid hormone receptors targeted by progestins in endometrial stromal cells from women with endometriosis. J. Reprod. Immunol. 2016, 117, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Weng, H.; Liu, F.; Hu, S.; Li, L.; Wang, Y. GnRH agonists induce endometrial epithelial cell apoptosis via GRP78 down-regulation. J. Transl. Med. 2014, 12, 306. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, B.; Bersinger, N.A.; Wotzkow, C.; Mueller, M.D. Endometriosis-associated nerve fibers, peritoneal fluid cytokine concentrations, and pain in endometriotic lesions from different locations. Fertil. Steril. 2012, 97, 373–380. [Google Scholar] [CrossRef]
- Mechsner, S.; Kaiser, A.; Kopf, A.; Gericke, C.; Ebert, A.; Bartley, J. A pilot study to evaluate the clinical relevance of endometriosis-associated nerve fibers in peritoneal endometriotic lesions. Fertil. Steril. 2009, 92, 1856–1861. [Google Scholar] [CrossRef]
- Tran, L.V.; Tokushige, N.; Berbic, M.; Markham, R.; Fraser, I.S. Macrophages and nerve fibres in peritoneal endometriosis. Hum. Reprod. 2009, 24, 835–841. [Google Scholar] [CrossRef]
- Greaves, E.; Temp, J.; Esnal-Zufiurre, A.; Mechsner, S.; Horne, A.W.; Saunders, P.T. Estradiol is a critical mediator of macrophage-nerve cross talk in peritoneal endometriosis. Am. J. Pathol. 2015, 185, 2286–2297. [Google Scholar] [CrossRef]
- Forster, R.; Sarginson, A.; Velichkova, A.; Hogg, C.; Dorning, A.; Horne, A.W.; Saunders, P.T.K.; Greaves, E. Macrophage-derived insulin-like growth factor-1 is a key neurotrophic and nerve-sensitizing factor in pain associated with endometriosis. FASEB J. 2019, 33, 11210–11222. [Google Scholar] [CrossRef] [PubMed]
- Redwine, D.B. Was Sampson wrong? Fertil. Steril. 2002, 78, 686–693. [Google Scholar] [CrossRef]
- Colgrave, E.M.; Bittinger, S.; Healey, M.; Dior, U.P.; Rogers, P.A.W.; Keast, J.R.; Girling, J.E.; Holdsworth-Carson, S.J. Superficial peritoneal endometriotic lesions are histologically diverse and rarely demonstrate menstrual cycle synchronicity with matched eutopic endometrium. Hum. Reprod. 2020, 35, 2701–2714. [Google Scholar] [CrossRef] [PubMed]
- Colgrave, E.M.; Keast, J.R.; Bittinger, S.; Healey, M.; Rogers, P.A.W.; Holdsworth-Carson, S.J.; Girling, J.E. Comparing endometriotic lesions with eutopic endometrium: Time to shift focus? Hum. Reprod. 2021, 36, 2814–2823. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, B.; Mueller, M.; Montgomery, G. Progesterone Resistance in Endometriosis: An Acquired Property? Trends Endocrinol. Metab. 2018, 29, 535–548. [Google Scholar] [CrossRef]
- Škegro, B.; Bjedov, S.; Mikuš, M.; Mustač, F.; Lešin, J.; Matijević, V.; Ćorić, M.; Elveđi Gašparović, V.; Medić, F.; Sokol Karadjole, V. Endometriosis, Pain and Mental Health. Psychiatr. Danub. 2021, 33, 632–636. [Google Scholar]
- Guo, S.-W. Recurrence of endometriosis and its control. Hum. Reprod. Update 2009, 15, 441–461. [Google Scholar] [CrossRef]
- Nirgianakis, K.; Ma, L.; McKinnon, B.; Mueller, M.D. Recurrence Patterns after Surgery in Patients with Different Endometriosis Subtypes: A Long-Term Hospital-Based Cohort Study. J. Clin. Med. 2020, 9, 496. [Google Scholar] [CrossRef]
- Zakhari, A.; Delpero, E.; McKeown, S.; Tomlinson, G.; Bougie, O.; Murji, A. Endometriosis recurrence following post-operative hormonal suppression: A systematic review and meta-analysis. Hum. Reprod. Update 2021, 27, 96–107. [Google Scholar] [CrossRef]
- van der Laak, J.; Litjens, G.; Ciompi, F. Deep learning in histopathology: The path to the clinic. Nat. Med. 2021, 27, 775–784. [Google Scholar] [CrossRef]


| SUP (Mean ± SEM) | OVA (Mean ± SEM) | DIE (Mean ± SEM) | * Total (Mean ± SEM) | p | |
|---|---|---|---|---|---|
| n | 7 | 9 | 9 | 26 | |
| Age (years) | 30.29 ± 1.46 | 33.04 ± 1.25 | 34.78 ± 2.06 | 32.68 ± 0.91 | 0.1523 |
| BMI (kg/m2) | 21.99 ± 1.40 | 21.53 ± 1.40 | 22.38 ± 1.09 | 21.93 ± 0.68 | 0.8902 |
| Menstrual pain | 4.60 ± 1.75 | 4.83 ± 1.66 | 8.00 ± 0.77 | 5.82 ± 0.85 | 0.2511 |
| Abdominal pain | 1.57 ± 0.84 | 3.57 ± 0.94 | 2.00 ± 0.82 | 2.50 ± 0.50 | 0.2576 |
| Dsypareunia | 0.86 ±0.70 | 3.71 ± 1.30 | 2.29 ± 0.99 | 2.23 ± 0.60 | 0.175 |
| rAFS Mild (I–II) | rAFS Severe (III–IV) | p Value | |||
|---|---|---|---|---|---|
| Mean | SEM | Mean | SEM | ||
| Epithelial cells | 24,096 | 8195 | 16,454 | 3659 | 0.3368 |
| Stromal cells | 10,207 | 3702 | 15,336 | 4504 | 0.5267 |
| Total cells | 34,303 | 9483 | 31,791 | 7882 | 0.8642 |
| Menstrual Pain | Abdominal Pain | Dyspareunia | |||||||
|---|---|---|---|---|---|---|---|---|---|
| n | r | p | n | r | p | n | r | p | |
| Epithelial cells | 17 | 0.1521 | 0.5600 | 22 | 0.6066 | 0.0028 ** | 22 | −0.1260 | 0.5763 |
| Stromal cells | 17 | 0.1216 | 0.6421 | 22 | 0.5117 | 0.0149 * | 22 | 0.2589 | 0.2447 |
| Total cells | 17 | 0.1683 | 0.5186 | 22 | 0.6782 | 0.0005 *** | 22 | 0.0573 | 0.7999 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McKInnon, B.D.; Nirgianakis, K.; Ma, L.; Wotzkow, C.A.; Steiner, S.; Blank, F.; Mueller, M.D. Computer-Aided Histopathological Characterisation of Endometriosis Lesions. J. Pers. Med. 2022, 12, 1519. https://doi.org/10.3390/jpm12091519
McKInnon BD, Nirgianakis K, Ma L, Wotzkow CA, Steiner S, Blank F, Mueller MD. Computer-Aided Histopathological Characterisation of Endometriosis Lesions. Journal of Personalized Medicine. 2022; 12(9):1519. https://doi.org/10.3390/jpm12091519
Chicago/Turabian StyleMcKInnon, Brett D., Konstantinos Nirgianakis, Lijuan Ma, Carlos Alvarez Wotzkow, Selina Steiner, Fabian Blank, and Michael D. Mueller. 2022. "Computer-Aided Histopathological Characterisation of Endometriosis Lesions" Journal of Personalized Medicine 12, no. 9: 1519. https://doi.org/10.3390/jpm12091519
APA StyleMcKInnon, B. D., Nirgianakis, K., Ma, L., Wotzkow, C. A., Steiner, S., Blank, F., & Mueller, M. D. (2022). Computer-Aided Histopathological Characterisation of Endometriosis Lesions. Journal of Personalized Medicine, 12(9), 1519. https://doi.org/10.3390/jpm12091519





