Relationship between Body Composition and Cardiac Autonomic Regulation in a Large Population of Italian Olympic Athletes
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol
2.1.1. Body Composition
2.1.2. Autonomic Evaluation and Exercise Stress Test
2.1.3. Subjective Somatic Stress Symptoms
2.2. Statistics
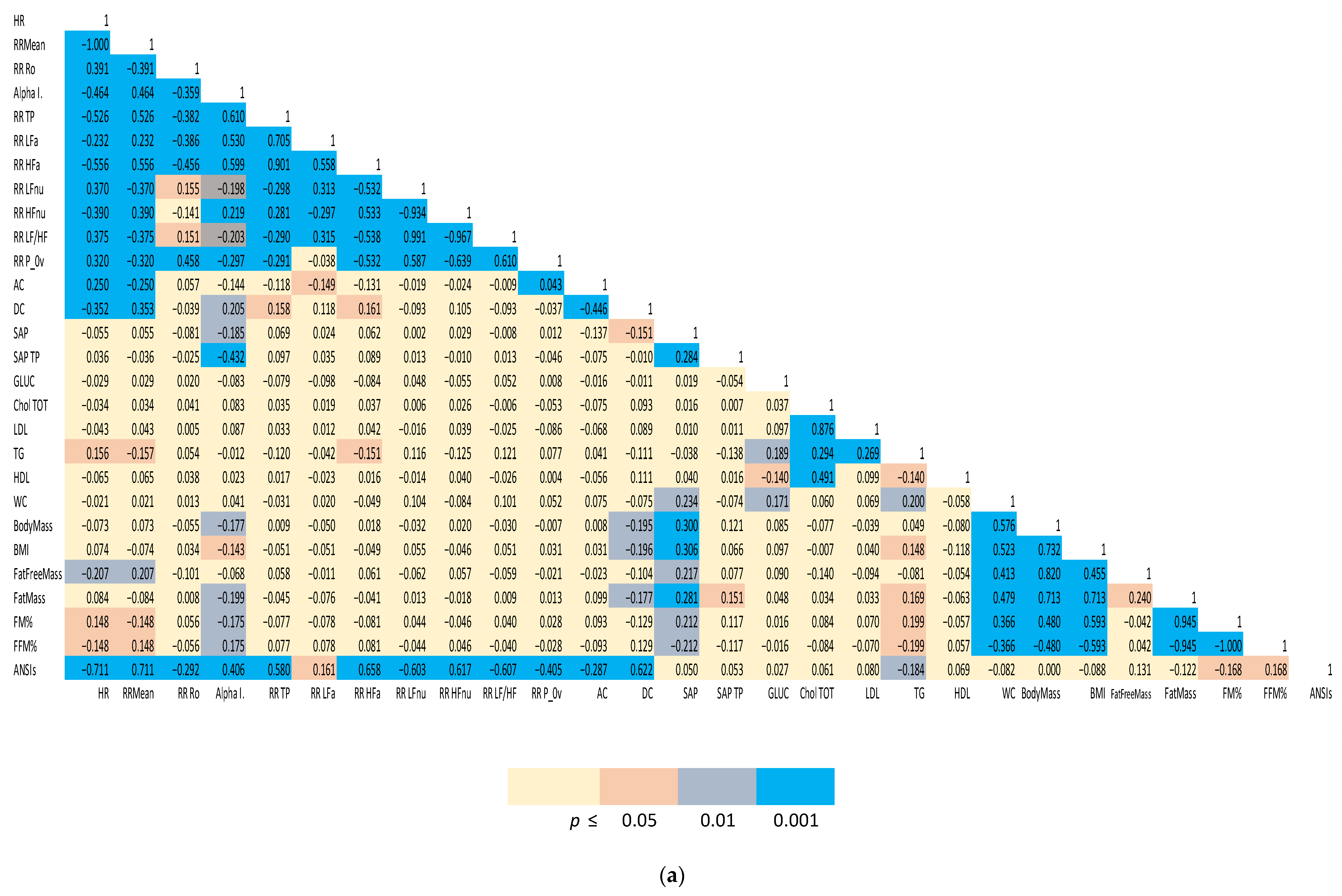
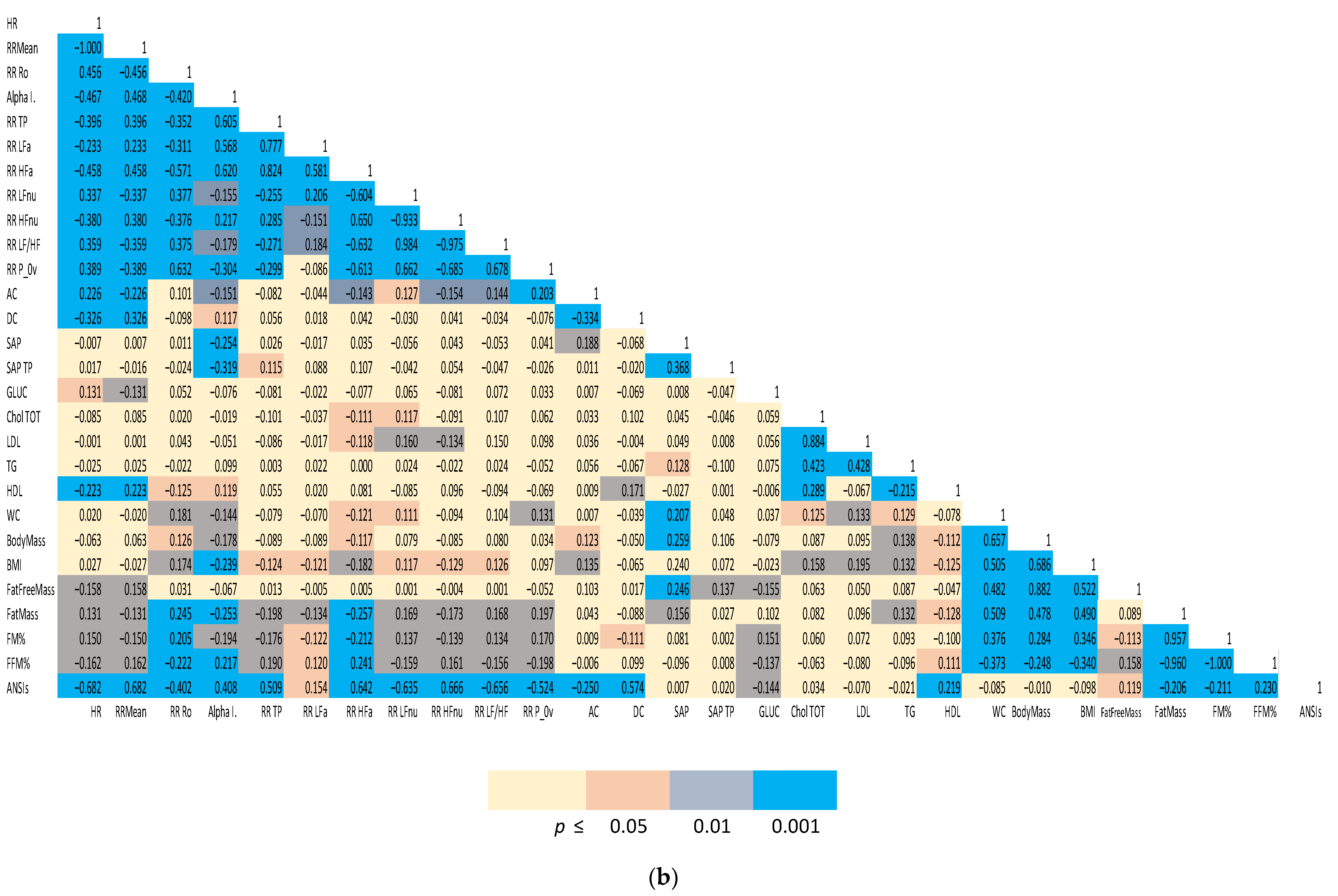
3. Results
4. Discussion
4.1. ANSIs as a Proxy of Cardiac Autonomic Regulation in Athletes
4.2. Body Composition in Olympic Athletes
4.3. Relationship between Body Composition and Autonomic Cardiac Regulation
4.4. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Ferretti, G. Energetics of Muscular Exercise; Springer Nature: Heidelberg, Germany, 2015; ISBN 978-3-319-05636-4. [Google Scholar]
- Pagani, M.; Lucini, D. Can autonomic monitoring predict results in distance runners? Am. J. Physiol. Circ. Physiol. 2009, 296, H1721–H1722. [Google Scholar] [CrossRef] [PubMed]
- Meyer, N.L.; Sundgot-Borgen, J.; Lohman, T.G.; Ackland, T.; Stewart, A.; Maughan, R.J.; Smith, S.; Müller, W. Body composition for health and performance: A survey of body composition assessment practice carried out by the Ad Hoc Research Working Group on Body Composition, Health and Performance under the auspices of the IOC Medical Commission. Br. J. Sports Med. 2013, 47, 1044–1053. [Google Scholar] [CrossRef] [PubMed]
- Kujala, U.M.; Vaara, J.P.; Kainulainen, H.; Vasankari, T.; Vaara, E.; Kyröläinen, H. Associations of Aerobic Fitness and Maximal Muscular Strength With Metabolites in Young Men. JAMA Netw. Open 2019, 2, e198265. [Google Scholar] [CrossRef]
- Ahn, A.C.; Tewari, M.; Poon, C.-S.; Phillips, R.S. The Limits of Reductionism in Medicine: Could Systems Biology Offer an Alternative? PLoS Med. 2006, 3, e208. [Google Scholar] [CrossRef] [PubMed]
- Otte, F.W.; Davids, K.; Millar, S.-K.; Klatt, S. When and How to Provide Feedback and Instructions to Athletes?—How Sport Psychology and Pedagogy Insights Can Improve Coaching Interventions to Enhance Self-Regulation in Training. Front. Psychol. 2020, 11, 1444. [Google Scholar] [CrossRef] [PubMed]
- Yook, J.S.; Rakwal, R.; Shibato, J.; Takahashi, K.; Koizumi, H.; Shima, T.; Ikemoto, M.J.; Oharomari, L.K.; McEwen, B.S.; Soya, H. Leptin in hippocampus mediates benefits of mild exercise by an antioxidant on neurogenesis and memory. Proc. Natl. Acad. Sci. USA 2019, 116, 10988–10993. [Google Scholar] [CrossRef]
- Iellamo, F.D.; Pigozzi, F.; Spataro, A.; Di Salvo, V.; Fagnani, F.; Roselli, A.; Rizzo, M.; Malacarne, M.; Pagani, M.; Lucini, D. Autonomic and psychological adaptations in Olympic rowers. J. Sports Med. Phys. Fit. 2006, 46, 598–604. [Google Scholar]
- Pagani, M.; Guaraldi, P.; Baschieri, F.; Lucini, D.; Cortelli, P. Interpreting Heart Rate Variability in Sleep: Why, When, and How? In Autonomic Nervous System and Sleep Order and Disorder; Chokroverty, S., Cortelli, P., Eds.; Springer: Cham, Switzerland, 2021; p. 345. ISBN 9783030622633. [Google Scholar]
- Williamson, J. Autonomic responses to exercise: Where is central command? Auton. Neurosci. Basic Clin. 2015, 188, 3–4. [Google Scholar] [CrossRef]
- Billman, G.E. Homeostasis: The Underappreciated and Far Too Often Ignored Central Organizing Principle of Physiology. Front. Physiol. 2020, 11, 200. [Google Scholar] [CrossRef]
- Mitchell, J.H.; Haskell, W.; Snell, P.; Van Camp, S.P. Task Force 8: Classification of Sports. J. Am. Coll. Cardiol. 2005, 45, 1364–1367. [Google Scholar] [CrossRef]
- Vatner, S.F.; Pagani, M. Cardiovascular adjustments to exercise: Hemodynamics and mechanisms. Prog. Cardiovasc. Dis. 1976, 19, 91–108. [Google Scholar] [CrossRef]
- Oggionni, G.; Spataro, A.; Pelliccia, A.; Malacarne, M.; Pagani, M.; Lucini, D. Left ventricular hypertrophy in world class elite athletes is associated with signs of improved cardiac autonomic regulation. Eur. J. Prev. Cardiol. 2019, 28, 1118–1124. [Google Scholar] [CrossRef] [PubMed]
- Malik, M.; John Camm, A.; Thomas Bigger, J.; Breithardt, G.; Cerutti, S.; Cohen, R.J.; Coumel, P.; Fallen, E.L.; Kennedy, H.L.; Kleiger, R.E.; et al. Heart Rate Variability: Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, heart rate variability—Standards of measurement, physiological interpretation, and clinical use. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef]
- Plews, D.J.; Laursen, P.B.; Stanley, J.; Kilding, A.E.; Buchheit, M. Training Adaptation and Heart Rate Variability in Elite Endurance Athletes: Opening the Door to Effective Monitoring. Sports Med. 2013, 43, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Dobbs, W.C.; Fedewa, M.V.; MacDonald, H.V.; Holmes, C.J.; Cicone, Z.S.; Plews, D.J.; Esco, M.R. The Accuracy of Acquiring Heart Rate Variability from Portable Devices: A Systematic Review and Meta-Analysis. Sports Med. 2019, 49, 417–435. [Google Scholar] [CrossRef] [PubMed]
- Karemaker, J.M. Heart rate variability: Why do spectral analysis? Heart 1997, 77, 99–101. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Akselrod, S.; Gordon, D.; Madwed, J.B.; Snidman, N.C.; Shannon, D.C.; Cohen, R.J. Hemodynamic regulation: Investigation by spectral analysis. Am. J. Physiol. Circ. Physiol. 1985, 249, H867–H875. [Google Scholar] [CrossRef]
- Pagani, M.; Lombardi, F.; Guzzetti, S.; Rimoldi, O.; Furlan, R.; Pizzinelli, P.; Sandrone, G.; Malfatto, G.; Dell’Orto, S.; Piccaluga, E. Power spectral analysis of heart rate and arterial pressure variabilities as a marker of sympatho-vagal interaction in man and conscious dog. Circ. Res. 1986, 59, 178–193. [Google Scholar] [CrossRef]
- Iellamo, F.; Legramante, J.M.; Pigozzi, F.; Spataro, A.; Norbiato, G.; Lucini, D.; Pagani, M. Conversion From Vagal to Sympathetic Predominance With Strenuous Training in High-Performance World Class Athletes. Circulation 2002, 105, 2719–2724. [Google Scholar] [CrossRef]
- McGlory, C.; Van Vliet, S.; Stokes, T.; Mittendorfer, B.; Phillips, S.M. The impact of exercise and nutrition on the regulation of skeletal muscle mass. J. Physiol. 2019, 597, 1251–1258. [Google Scholar] [CrossRef]
- Pavlov, V.A.; Tracey, K.J. The vagus nerve and the inflammatory reflex—linking immunity and metabolism. Nat. Rev. Endocrinol. 2012, 8, 743–754. [Google Scholar] [CrossRef] [PubMed]
- Solaro, N.; Pagani, M.; Lucini, D. Altered Cardiac Autonomic Regulation in Overweight and Obese Subjects: The Role of Age-and-Gender-Adjusted Statistical Indicators of Heart Rate Variability and Cardiac Baroreflex. Front. Physiol. 2021, 11, 567312. [Google Scholar] [CrossRef] [PubMed]
- Lucini, D.; Di Fede, G.; Parati, G.; Pagani, M. Impact of Chronic Psychosocial Stress on Autonomic Cardiovascular Regulation in Otherwise Healthy Subjects. Hypertension 2005, 46, 1201–1206. [Google Scholar] [CrossRef]
- Hess, W.R. The Central Control of the Activity of Internal Organs. In Nobel Lecture Physiology or Medicine 1942–1962; Elsevier Publishing Company: Boston, MA, USA, 1949. [Google Scholar]
- Webber, J.; MacDonald, I. Metabolic actions of catecholamines in man. Baillieres. Clin. Endocrinol. Metab. 1993, 7, 393–413. [Google Scholar] [CrossRef]
- Yao, G.; Kang, L.; Li, J.; Long, Y.; Wei, H.; Ferreira, C.A.; Jeffery, J.J.; Lin, Y.; Cai, W.; Wang, X. Effective weight control via an implanted self-powered vagus nerve stimulation device. Nat. Commun. 2018, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Smoljo, T.; Stanić, I.; Sila, S.; Kovačić, U.; Crnošija, L.; Junaković, A.; Adamec, I.; Hojsak, I.; Skorić, M.K.; Habek, M. The Relationship between Autonomic Regulation of Cardiovascular Function and Body Composition. J. Obes. Metab. Syndr. 2021, 29, 188–197. [Google Scholar] [CrossRef]
- Casadei, B.; Cochrane, S.; Johnsoton, J.; Conway, J.; Sleight, P. Pitfalls in the interpretation of spectral analysis of the heart rate variability during exercise in humans. Acta Physiol. Scand. 1995, 153, 125–131. [Google Scholar] [CrossRef]
- Sala, R.; Malacarne, M.; Tosi, F.; Benzi, M.; Solaro, N.; Tamorri, S.; Spataro, A.; Pagani, M.; Lucini, D. May a unitary autonomic index help assess autonomic cardiac regulation in elite athletes? Preliminary observations on the national Italian Olympic committee team. J. Sports Med. Phys. Fit. 2017, 57, 1702–1710. [Google Scholar] [CrossRef]
- Aragon, A.A.; Schoenfeld, B.J.; Wildman, R.; Kleiner, S.; VanDusseldorp, T.; Taylor, L.; Earnest, C.P.; Arciero, P.J.; Wilborn, C.; Kalman, D.S.; et al. International society of sports nutrition position stand: Diets and body composition. J. Int. Soc. Sports Nutr. 2017, 14, 1–19. [Google Scholar] [CrossRef]
- Ackland, T.; Lohman, T.G.; Sundgot-Borgen, J.; Maughan, R.J.; Meyer, N.L.; Stewart, A.; Müller, W.; Ackland, W.P.T.R. Current Status of Body Composition Assessment in Sport. Sports Med. 2012, 42, 227–249. [Google Scholar] [CrossRef]
- Farley, A.; Slater, G.J.; Hind, K. Short-Term Precision Error of Body Composition Assessment Methods in Resistance-Trained Male Athletes. Int. J. Sport Nutr. Exerc. Metab. 2021, 31, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Kerkhof, P.L.M.; Peace, R.A.; Handly, N. Ratiology and a Complementary Class of Metrics for Cardiovascular Investigations. Physiology 2019, 34, 250–263. [Google Scholar] [CrossRef]
- Lucini, D.; Sala, R.; Spataro, A.; Malacarne, M.; Benzi, M.; Tamorri, S.; Pagani, M. Can the use of a single integrated unitary autonomic index provide early clues for eventual eligibility for olympic games? Eur. J. Appl. Physiol. 2018, 118, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Bull. World Health Organ. 2007, 85, 867–872. [Google Scholar] [CrossRef]
- Badilini, F.; Pagani, M.; Porta, A. Heartscope: A software tool addressing autonomic nervous system regulation. Comput. Cardiol. 2005, 32, 259–262. [Google Scholar] [CrossRef]
- Toninelli, G.; Vigo, C.; Vaglio, M.; Porta, A.; Lucini, D.; Badilini, F.; Pagani, M.; York, N.; Hospital, L.S. DynaScope: A Software Tool for the Analysis of Heart Rate Variability During Exercise. Comput. Cardiol. 2012, 39, 181–184. [Google Scholar]
- Bauer, A.; Kantelhardt, J.W.; Barthel, P.; Schneider, R.; Mäkikallio, T.; Ulm, K.; Hnatkova, K.; Schömig, A.; Huikuri, H.; Bunde, A.; et al. Deceleration capacity of heart rate as a predictor of mortality after myocardial infarction: Cohort study. Lancet 2006, 367, 1674–1681. [Google Scholar] [CrossRef]
- Porta, A.; Guzzetti, S.; Montano, N.; Furlan, R.; Pagani, M.; Malliani, A.; Cerutti, S. Entropy, entropy rate, and pattern classification as tools to typify complexity in short heart period variability series. IEEE Trans. Biomed. Eng. 2001, 48, 1282–1291. [Google Scholar] [CrossRef]
- Lucini, D.; Vigo, C.; Tosi, F.; Toninelli, G.; Badilini, F.; Pagani, M. Assessing autonomic response to repeated bouts of exercise below and above respiratory threshold: Insight from dynamic analysis of RR variability. Eur. J. Appl. Physiol. 2014, 114, 1269–1279. [Google Scholar] [CrossRef]
- Jouven, X.; Empana, J.-P.; Schwartz, P.J.; Desnos, M.; Courbon, D.; Ducimetière, P. Heart-Rate Profile during Exercise as a Predictor of Sudden Death. N. Engl. J. Med. 2005, 352, 1951–1958. [Google Scholar] [CrossRef]
- Thompson, B. Exploratory and Confirmatory Factor Analysis: Understanding Concepts and Applications; APA Books: Washington, DC, USA, 2004. [Google Scholar]
- Lucini, D.; Solaro, N.; Pagani, M. Autonomic Differentiation Map: A Novel Statistical Tool for Interpretation of Heart Rate Variability. Front. Physiol. 2018, 9, 401. [Google Scholar] [CrossRef] [PubMed]
- Koenker, R. Quantile Regression; Cambridge University Press: New York, NY, USA, 2005. [Google Scholar]
- Pagani, M.; Sala, R.; Malacarne, M.; Lucini, D. Benchmarking Heart Rate Variability to Overcome Sex-Related Bias. Adv. Exp. Med. Biol. 2018, 1065, 191–205. [Google Scholar] [CrossRef] [PubMed]
- Lucini, D.; Marchetti, I.; Spataro, A.; Malacarne, M.; Benzi, M.; Tamorri, S.; Sala, R.; Pagani, M. Heart rate variability to monitor performance in elite athletes: Criticalities and avoidable pitfalls. Int. J. Cardiol. 2017, 240, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Malliani, A.; Pagani, M.; Lombardi, F.; Cerutti, S. Cardiovascular neural regulation explored in the frequency domain. Circulation 1991, 84, 482–492. [Google Scholar] [CrossRef]
- Reinert, B.L.; Pohlman, R.; Hartzler, L. Correlation of Air Displacement Plethysmography with Alternative Body Fat Meas-urement Techniques in Men and Women. Int. J. Exerc. Sci. 2012, 5, 367–378. [Google Scholar]
- Santos, D.A.; Dawson, J.A.; Matias, C.N.; Rocha, P.M.; Minderico, C.S.; Allison, D.B.; Sardinha, L.B.; Silva, A.M. Reference Values for Body Composition and Anthropometric Measurements in Athletes. PLoS ONE 2014, 9, e97846. [Google Scholar] [CrossRef]
- Bentzur, K.M.; Kravitz, L.; Lockner, D.W. Evaluation of the BOD POD for Estimating Percent Body Fat in Collegiate Track and Field Female Athletes: A Comparison of Four Methods. J. Strength Cond. Res. 2008, 22, 1985–1991. [Google Scholar] [CrossRef]
- Castizo-Olier, J.; Irurtia, A.; Jemni, M.; Carrasco-Marginet, M.; Fernández-García, R.; Rodríguez, F.A. Bioelectrical impedance vector analysis (BIVA) in sport and exercise: Systematic review and future perspectives. PLoS ONE 2018, 13, e0197957. [Google Scholar] [CrossRef]
- Tian, Y.; Huang, C.; He, Z.; Hong, P.; Zhao, J. Autonomic function responses to training: Correlation with body composition changes. Physiol. Behav. 2015, 151, 308–313. [Google Scholar] [CrossRef]
- Mancia, G.; Grassi, G. The Autonomic Nervous System and Hypertension. Circ. Res. 2014, 114, 1804–1814. [Google Scholar] [CrossRef]
- Park, H.-Y.; Jung, W.-S.; Kim, J.; Hwang, H.; Lim, K. Twelve Weeks of Aerobic Exercise at the Lactate Threshold Improves Autonomic Nervous System Function, Body Composition, and Aerobic Performance in Women with Obesity. J. Obes. Metab. Syndr. 2020, 29, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Benedini, S.; Dozio, E.; Invernizzi, P.L.; Vianello, E.; Banfi, G.; Terruzzi, I.; Luzi, L.; Romanelli, M.M.C. Irisin: A Potential Link between Physical Exercise and Metabolism—An Observational Study in Differently Trained Subjects, from Elite Athletes to Sedentary People. J. Diabetes Res. 2017, 2017, 1039161. [Google Scholar] [CrossRef] [PubMed]
- Joyner, M.J.; Green, D.J. Exercise protects the cardiovascular system: Effects beyond traditional risk factors. J. Physiol. 2009, 587, 5551–5558. [Google Scholar] [CrossRef] [PubMed]

| Variables | Units | Definition |
|---|---|---|
| Alpha Index | ms/mmHg | Alpha index is a frequency domain index of cardiac baroreflex |
| BRS | ms/mmHg | Cardiac baroreflex sensitivity |
| HR | beat/min | Heart rate, i.e., number of cardiac beats per minute |
| RR mean | ms | RR interval obtained from a single lead ECG tachogram |
| RR TP | ms2 | RR total variance obtained from a single lead ECG tachogram |
| RR LFa | ms2 | Absolute power (a) of low frequency (LF) autoregressive spectral component of RR variability (V) |
| RR HFa | ms2 | Absolute power (a) of high frequency (HF) spectral component of RRV |
| RR LFnu | nu | Normalized power (nu) of low frequency (LF) spectral component of RRV |
| RR HFnu | nu | Normalized power (nu) of high frequency (HF) spectral component of RRV |
| RR LF/HF | . | Numerical ratio between LF and HF spectral powers from tachogram |
| ΔLFnu | . | Difference in LF power in nu between stand and rest |
| RR Ro | . | Tachogram index of self-similarity (range 0–1) |
| RR P_0v | . | Frequency of tachogram three beat pattern, no variations |
| RR P_2uv | . | Frequency of tachogram three beat pattern, unlike variations |
| AC | msc | Acceleration capacity index of the tachogram |
| DC | ms | Deceleration capacity index of the tachogram |
| SAP | mmHg | Systolic arterial pressure (resting) |
| DAP | mmHg | Diastolic arterial pressure (resting) |
| SAP Mean | mmHg | Mean values of continuous systolic arterial pressure recordings during rest |
| SAP TP | mmHg2 | Systolic arterial pressure variance (resting) |
| SAP LFa | mmHg2 | Absolute power (a) of low frequency (LF) autoregressive spectral component of SAP variability |
| SAP HFa | mmHg2 | Absolute power (a) of high frequency (LF) autoregressive spectral component of SAP variability |
| BMI | Kg/m2 | Body mass index |
| WC | cm | Waist circumference |
| BSA | m2 | Body surface area |
| FM% | % | Fat mass percent (BOD POD) |
| FFM% | % | Fat free mass percent (BOD POD) |
| Fat Mass | kg | Measured fat mass (BOD POD) |
| FatFreeMass | kg | Measured fat free mass (BOD POD) |
| Body Mass | kg | Measured body mass (BOD POD) |
| Body Volume | L | Volume of overall body |
| Chol TOT | mg/dL | Fasting total cholesterol (plasma) |
| HDL | mg/dL | Fasting high-density cholesterol (plasma) |
| LDL | mg/dL | Fasting low-density cholesterol (plasma) |
| TG | mg/dL | Fasting triglyceride (plasma) |
| GLUC | mg/dL | Fasting glucose (plasma) |
| Stress perception | . | Perception of stress |
| Fatigue perception | . | Perception of fatigue |
| 4SQ | . | Subjective stress-related somatic symptoms questionnaire |
| ANSIs | % | Percentile ranked Autonomic Nervous System Index for sports |
| Variables | Females | Males | All | Sig (2-Tailed) |
|---|---|---|---|---|
| n | 230 | 353 | 583 | (F vs. M) |
| Age (years) | 23.00 ± 4.00 | 24.00 ± 4.00 | 23.00 ± 4.00 | 0.842 |
| Weight (kg) | 62.00 ± 6.00 | 79.00 ± 7.00 | 72.00 ± 9.00 | <0.001 |
| Height (m) | 1.70 ± 0.05 | 1.83 ± 0.07 | 1.78 ± 0.08 | <0.001 |
| SAP (mmHg) | 111.00 ± 6.00 | 125.00 ± 7.00 | 119.00 ± 9.00 | <0.001 |
| DAP (mmHg) | 62.00 ± 4.00 | 65.00 ± 5.00 | 64.00 ± 4.00 | <0.001 |
| WC (cm) | 75.00 ± 5.00 | 84.00 ± 4.00 | 81.00 ± 5.50 | <0.001 |
| BMI (kg/m2) | 21.30 ± 1.23 | 23.29 ± 1.49 | 22.63 ± 1.80 | <0.001 |
| Variables | Females | Males | All | Sig (2-Tailed) |
|---|---|---|---|---|
| FM% (%) | 19.35 ± 3.55 | 11.10 ± 3.00 | 13.50 ± 4.90 | <0.001 |
| FFM% (%) | 80.65 ± 3.55 | 88.80 ± 3.00 | 86.40 ± 5.00 | <0.001 |
| Fat Mass (kg) | 11.91 ± 3.10 | 8.65 ± 2.94 | 10.05 ± 3.35 | <0.001 |
| FatFreeMass (kg) | 50.51 ± 4.47 | 69.43 ± 6.90 | 61.80 ± 9.67 | <0.001 |
| Body Mass (kg) | 62.57 ± 6.08 | 78.46 ± 7.77 | 72.60 ± 9.30 | <0.001 |
| Body Volume (L) | 59.13 ± 5.80 | 73.40 ± 7.35 | 68.44 ± 8.72 | <0.001 |
| BSA (m2) | 1.72 ± 0.11 | 2.00 ± 0.13 | 1.89 ± 0.16 | <0.001 |
| Variables | Females | Males | All | Sig (2-Tailed) |
|---|---|---|---|---|
| Chol TOT (mg/dL) | 182 ± 22 | 168 ± 20 | 173 ± 22 | <0.001 |
| HDL (mg/dL) | 74 ± 10 | 61 ± 9 | 66 ± 10 | <0.001 |
| LDL (mg/dL) | 93 ± 18 | 91 ± 18 | 92 ± 18 | 0.989 |
| TG (mg/dL) | 64 ± 13 | 64 ± 17 | 64 ± 15 | 0.624 |
| GLUC (mg/dL) | 92 ± 4 | 95 ± 4 | 94 ± 4 | <0.001 |
| Stress perception (.) | 2 ± 2 | 2 ± 2 | 2 ± 2 | 0.120 |
| Fatigue perception (.) | 3 ± 2 | 3 ± 2 | 3 ± 2 | 0.387 |
| 4SQ (.) | 17 ± 14 | 12 ± 11 | 14 ± 12 | 0.006 |
| Variables | Females | Males | All | Sig (2-Tailed) |
|---|---|---|---|---|
| HR (bpm) | 59.70 ± 7.23 | 57.73 ± 6.37 | 58.28 ± 6.57 | 0.029 |
| RR mean (ms) | 1005.02 ± 120.85 | 1039.38 ± 112.94 | 1029.57 ± 116.14 | 0.029 |
| RR TP (ms2) | 3672.69 ± 1902.73 | 3356.49 ± 1610.03 | 3493.89 ± 1716.71 | 0.374 |
| RR LFa (ms2) | 638.86 ± 339.92 | 918.18 ± 505.07 | 784.14 ± 446.78 | <0.001 |
| RR HFa (ms2) | 1484.47 ± 1008.77 | 918.61 ± 615.45 | 1120.38 ± 761.38 | <0.001 |
| RR LFnu (nu) | 30.94 ± 12.62 | 42.86 ± 15.20 | 38.43 ± 14.59 | <0.001 |
| RR HFnu (nu) | 62.94 ± 14.45 | 50.75 ± 15.39 | 56.24 ± 15.37 | <0.001 |
| RR LF/HF | 0.48 ± 0.28 | 0.86 ± 0.49 | 0.68 ± 0.41 | <0.001 |
| Alpha Index (ms/mmHg) | 27.80 ± 8.93 | 22.80 ± 7.98 | 24.80 ± 8.51 | <0.001 |
| BRS (ms/mmHg) | 24.70 ± 9.47 | 21.16 ± 8.82 | 22.15 ± 9.03 | 0.007 |
| RR Ro (.) | 0.22 ± 0.06 | 0.23 ± 0.07 | 0.22 ± 0.06 | 0.076 |
| SAP Mean (mmHg) | 110.84 ± 10.12 | 125.58 ± 9.79 | 118.86 ± 11.02 | <0.001 |
| SAP TP (mmHg2) | 13.12 ± 7.64 | 24.64 ± 14.16 | 19.97 ± 11.96 | <0.001 |
| SAP LFa (mmHg2) | 1.81 ± 1.13 | 2.82 ± 1.79 | 2.33 ± 1.48 | <0.001 |
| SAP HFa (mmHg2) | 0.92 ± 0.50 | 1.14 ± 0.68 | 1.03 ± 0.59 | 0.086 |
| Δ LF (nu) | 45.47 ± 13.28 | 39.09 ± 15.60 | 42.65 ± 14.51 | 0.002 |
| ANSIs (%) | 49.81 ± 25.95 | 50.58 ± 24.04 | 50.28 ± 25.00 | 0.692 |
| RRP_0v (.) | 8.80 ± 5.22 | 14.00 ± 7.50 | 11.50 ± 6.34 | <0.001 |
| RRP_2uv (.) | 31.62 ± 10.51 | 26.47 ± 9.98 | 28.38 ± 10.15 | <0.001 |
| AC (ms) | 2.95 ± 0.52 | 3.12 ± 0.59 | 3.05 ± 0.57 | 0.006 |
| DC (ms) | 3.47 ± 0.93 | 2.96 ± 0.96 | 3.16 ± 0.98 | 0.032 |
| Latent Domain | Standardized B Coefficient (*) | t (**) | Significance (**) | ∆Pseudo R2 (***) |
|---|---|---|---|---|
| 1 | −0.545 | −13.170 | <0.001 | 0.171 |
| 2 | 0.064 | 1.533 | 0.126 | 0.004 |
| 3 | −0.130 | −3.080 | 0.002 | 0.012 |
| 4 | 0.254 | 6.251 | <0.001 | 0.049 |
| 5 | 0.029 | 0.584 | 0.559 | 0.001 |
| 6 | 0.615 | 14.247 | <0.001 | 0.204 |
| Model quality measures: Pseudo R2 = 0.397; Mean Absolute Error (MAE) = 14.883 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lucini, D.; Spataro, A.; Giovanelli, L.; Malacarne, M.; Spada, R.; Parati, G.; Solaro, N.; Pagani, M. Relationship between Body Composition and Cardiac Autonomic Regulation in a Large Population of Italian Olympic Athletes. J. Pers. Med. 2022, 12, 1508. https://doi.org/10.3390/jpm12091508
Lucini D, Spataro A, Giovanelli L, Malacarne M, Spada R, Parati G, Solaro N, Pagani M. Relationship between Body Composition and Cardiac Autonomic Regulation in a Large Population of Italian Olympic Athletes. Journal of Personalized Medicine. 2022; 12(9):1508. https://doi.org/10.3390/jpm12091508
Chicago/Turabian StyleLucini, Daniela, Antonio Spataro, Luca Giovanelli, Mara Malacarne, Raffaella Spada, Gianfranco Parati, Nadia Solaro, and Massimo Pagani. 2022. "Relationship between Body Composition and Cardiac Autonomic Regulation in a Large Population of Italian Olympic Athletes" Journal of Personalized Medicine 12, no. 9: 1508. https://doi.org/10.3390/jpm12091508
APA StyleLucini, D., Spataro, A., Giovanelli, L., Malacarne, M., Spada, R., Parati, G., Solaro, N., & Pagani, M. (2022). Relationship between Body Composition and Cardiac Autonomic Regulation in a Large Population of Italian Olympic Athletes. Journal of Personalized Medicine, 12(9), 1508. https://doi.org/10.3390/jpm12091508







