Abstract
Nanotechnology, the art of engineering structures on a molecular level, offers the opportunity to implement new strategies for the diagnosis and management of pregnancy-related disorders. This review aims to summarize the current state of nanotechnology in obstetrics and cancer in pregnancy, focusing on existing and potential applications, and provides insights on safety and future directions. A systematic and comprehensive literature assessment was performed, querying the following databases: PubMed/Medline, Scopus, and Endbase. The databases were searched from their inception to 22 March 2022. Five independent reviewers screened the items and extracted those which were more pertinent within the scope of this review. Although nanotechnology has been on the bench for many years, most of the studies in obstetrics are preclinical. Ongoing research spans from the development of diagnostic tools, including optimized strategies to selectively confine contrast agents in the maternal bloodstream and approaches to improve diagnostics tests to be used in obstetrics, to the synthesis of innovative delivery nanosystems for therapeutic interventions. Using nanotechnology to achieve spatial and temporal control over the delivery of therapeutic agents (e.g., commonly used drugs, more recently defined formulations, or gene therapy-based approaches) offers significant advantages, including the possibility to target specific cells/tissues of interest (e.g., the maternal bloodstream, uterus wall, or fetal compartment). This characteristic of nanotechnology-driven therapy reduces side effects and the amount of therapeutic agent used. However, nanotoxicology appears to be a significant obstacle to adopting these technologies in clinical therapeutic praxis. Further research is needed in order to improve these techniques, as they have tremendous potential to improve the accuracy of the tests applied in clinical praxis. This review showed the increasing interest in nanotechnology applications in obstetrics disorders and pregnancy-related pathologies to improve the diagnostic algorithms, monitor pregnancy-related diseases, and implement new treatment strategies.
1. Introduction
Over the previous decades, significant advances in obstetrics have led to a reduction in mortality and morbidity associated with pregnancy complications [1,2]. Fundamental advances in neonatology have also ameliorated pregnancy outcomes and improved the survival rate of newborns delivered at earlier gestational ages, although not without an economic impact [1,3,4]. Fetal growth restriction, preterm birth, and cancer management during pregnancy still require research advancements to improve the quality of life of mothers and newborns, and possibly to reduce the costs of the postnatal management of newborns resulting from particularly problematic pregnancies.
For instance, the global average rate of preterm delivery was estimated in 2014 to be around 10.6% (9.0–12.0%) [5], representing a considerable concern in terms of morbidity (immediate and long-term sequels) and mortality [6,7]. Hypertensive disorders of pregnancy and preeclampsia are also potentially harmful pathologies [8,9], with a prevalence varying from 5% in low-risk to 20% in high-risk pregnancies [10]. Fetal growth restriction, which complicates approximately 10% of all pregnancies, alongside preeclampsia, is a contributing factor to preterm deliveries [11]. Gestational diabetes mellitus and pregestational diabetes mellitus significantly impact pregnancy, maternal, and offspring outcomes (e.g., favoring pregnancy complications, abnormal fetal growth trajectories, or long-term sequels) [12,13,14].
Furthermore, with the progressive aging of the female population and the childbearing delay [15], a significant increase in the incidence of pregnancy-associated breast (and other) cancers has been observed [16,17]. This increase boosts the demand for targeted and safe technologies that can also be used during pregnancy to improve the accuracy of diagnosis and the treatment of neoplastic pathologies.
The advancement of precision medicine strategies to manage these pathologies relies on a cornerstone that requires a more comprehensive understanding of the pathogeneses of these conditions, which is expected to lead to the implementation of current strategies and the development of new ones, with diagnostic and therapeutic applications [18,19]. As the science of engineering structures on a molecular level, nanotechnology offers a significant opportunity to achieve this goal [18,20].
Conceptually introduced in the 1950s, nanotechnology refers to the use of nanoscopic particles and devices with the potential to specifically integrate electronic, optical, fluid, and mechanical functions. In the biological field, these approaches are primarily applied in diagnostics and therapy [21,22]. The main purpose of nanotechnologies in the medical field is to improve the biodistribution, specificity, and targeting of bioactive molecules, in order to induce a desired therapeutic outcome while reducing potentially threatening side effects [22].
This review proposes a survey of the state of knowledge on the application of nanotechnology to obstetrics, focusing on its current and potential applications and safety. We will discuss the most recent discoveries in the early diagnosis, prevention, and treatment of maternal–fetal pathologies. Moreover, we will also highlight the issues associated with managing pregnancy-related malignancies, which represent a growing issue of our times, and provide insights on the role nanotechnology may play in developing intervention options.
2. Materials and Methods
A systematic and comprehensive literature assessment was performed, querying the following databases: PubMed/ Medline, Scopus, and Endbase. The databases were searched from their inception to 22 March 2022. Five authors independently screened the items and extracted those which were more pertinent within the scope of this review. The screened items which fall within the scope of this review should be peer-reviewed full-text articles discussing nanotechnology in the obstetric field. The following words were used in combination to identify the literature: pregnancy, pregnancies, gestation, pregnant, maternal–fetal, mother–fetus, mother fetus, obstetric*, tumor*, neoplasm*, neoplasia, neoplasias, cancer*, malignant, malignancy, malignancies, nanotechnology, nanomedicine, nanoparticle*, nanotherapeutic*, nanoformulation*, liposome*, micelle*, exosome*, nano theranostic*, nanofluidics, drug delivery, precision medicine, nano-obstetric*, and nanoobstetric*. Details about the queries are shown in Supplemental Table S1. In total, 11,982 items were found, with 7613 remaining upon the removal of duplicates. After the manual screening of the titles and abstracts, 674 items were considered relevant for this review, and were assessed for their full text. Selected articles included in this review are based on their relevance and scientific merit. The scientific merit assessment was based on full-text publication in peer-reviewed journals, excluding eventually retracted items. Meanwhile, relevance was based on the following principles: pragmatism to include the most valuable articles to give a comprehensive overview starting from literature reviews, pluralism to have as many perspectives as possible, and contestation to discuss conflicting data and debate arguments. A bibliometric investigation was also performed using data extracted from the Scopus database and saved in BibTeX format. The bibliometric analysis was carried out using the bibliometrix package (version 4.0.0) and R (version 4.2.1; R Core Team-2022. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria https://www.R-project.org/, accessed on 23 June 2022) [23,24]. The Scopus database was chosen because the references of each item were available, and it was possible to calculate a local citation ranking. We also assessed country collaborations, the keyword frequency over time, and word co-occurrences in titles and abstracts.
3. Nanotechnologies and Pregnancy
The use of drugs during pregnancy has increased significantly over time, probably due to the greater confidence that modern pharmacology has allowed us to acquire regarding the use of specific medication classes [25,26,27]. In spite of the greater awareness of medication use in pregnancy and lactation, information about indications and contraindications is not always available, especially in the case of newly introduced active principles or formulations. This paucity of information somewhat depends on the fact that trials seeking to ascertain drug effects on the fetus are performed solely in particular cases (e.g., treatments targeted explicitly for pregnancy or fetal in utero therapy), and pregnant women are willingly excluded in most studies due to ethical issues [28].
From this perspective, nanotechnology has only recently been applied to pregnancy and lactation, resulting in a new branch of medicine known as “nano-obstetrics”. Nanomedicine-based therapies are currently gaining great interest for what concerns the treatment of conditions affecting the mother, the placenta, and the fetus, and to improve the prognosis for both mothers and newborns [29]. A common fear of potentially damaging gametes, embryos, and fetuses, or even negatively impacting a woman’s reproductive potential, affects the development of innovative therapeutics in reproductive medicine and obstetrics. Modern delivery systems may provide alternative targeted intervention strategies by precisely targeting the source of the disease while minimizing short- and long-term consequences for the mother and the progeny [18,30,31].
Despite the evident success of nanotechnologies in the last few decades and the growing interest in the development of strategies which are able to temporally and spatially deliver drugs and retain them at the targeted site, pregnancy still represents a challenge for the application of these novel medications [31]. On the one hand, as introduced above, there is often a lack of data regarding the possible adverse effects on the mother and fetus, as for classical drugs [28,31]. Consequently, only phase IV observations allow us subsequently to draw definitive conclusions relating to any secondary effects in the female gender or specific conditions such as pregnancy. On the other hand, specific design and chemical features make nanoparticles capable of overcoming physiological barriers, such as the blood–placental barrier [32], and potentially to be active also on the fetus.
4. The Placental Barrier, Therapeutic Perspectives of Nanoparticles, and Nanotoxicology
During pregnancy, the blood–placental barrier regulates the transport of oxygen, nutrients, and residual products between the maternal and fetal bloodstreams. It also prevents fetal exposure to possibly harmful molecules from the maternal circulation. Therefore, the essential role that the blood–placental barrier plays in supporting the interaction between the mother and the fetus represents an interesting opportunity for the delivery of nanotherapeutics to treat various pathologies (e.g., targeting a drug specifically to the maternal compartment only, without entering the fetal compartment) [33].
During its intrauterine development, the fetus is vulnerable to chemicals and other substances that can impair its development [34]. A major limitation for the application of nanomedicine in the field of maternal and fetal health is the lack of established and reliable in vitro and ex vivo models which are available to adequately simulate the pregnant patient scenario [29]. Experimental models of the placenta have been developed to address the function of the placental barrier, which can be classified into in vivo, ex vivo, and in vitro models (Table 1) [35,36,37]. As expected, any of these models presents some strengths and some limitations. In vivo animal models can be costly and limited because of species differences [36,38]. Ex vivo explants and perfused placentae are optimal models of the placental interface but are unfortunately burdened with a short viability [36]. The static in vitro models based on cell cultures frequently use cancer cell lines with no variability, unlike in vivo models [35,36]. Recently, a better understanding has been promised by implementing new study models such as the placenta on-a-chip models, or organoids [35,36,37] (Table 1). The effect of nanoparticles on cell viability and biological barrier integrity has also been studied in many different epithelial barriers, such as the lungs, intestines, kidneys, and skin. All of this information can be helpful to better understand the interaction of nanoparticles with maternal and fetal tissues.

Table 1.
Experimental models of the placenta.
Nanoparticles can transit through the ordinary placental trans-cellular transport mechanisms such as pinocytosis, active transport, facilitated diffusion, and passive diffusion. The exact pathway is likely to be dependent on the particle size and the surface chemistry [36,39]. For example, gold nanoparticles could cross the placenta, arriving at the fetal circulation employing endocytosis, whether clathrin-mediated or caveolin-mediated [40]. Meanwhile, polystyrene nanoparticles were found to cross the placenta through passive diffusion [41]. Furthermore, polyethylene glycol-coated liposomes were shown to be mostly impermeable to the placental barrier [36,42]. Additionally, the CNKGLRNK peptide-coated liposomes specifically target the placental interface [43]. They do not pass into the fetal circulation, allowing therapies to be delivered right at the placental interface (i.e., the endothelium of the uterine spiral arteries and placental labyrinth) [43]. Various effects have been observed to depend on the chemical nature of the nanomaterial, nanoparticle size, surface coatings, and concentration, as well as on transepithelial electrical resistance and paracellular permeability [44]. Specific examples supporting the supposition that the modifications generated by specific nanomaterials on the placental barrier could lead to severe fetal growth impairment and development consequences are listed below.
High concentrations of polystyrene nanoparticles reduced in vitro the cell viability of choriocarcinoma cells (BeWo cell line) [45]. The authors attributed the effect to a previously known positive association between a high polystyrene dosage and a pro-inflammatory effect [45,46]. The in vivo administration of cobalt and chromium 80 nm nanoparticles resulted in neurodevelopmental abnormalities, and increased DNA damage in the fetal hippocampus [47]. Maternal–fetal oxygen transfer and the production of human chorionic gonadotropin were not modified by polyamidoamine dendrimer exposure in an ex vivo placenta perfusion experiment [48].
Another example is represented by silica nanoparticles, which reduced BeWo cell viability at high concentrations, possibly due to lipid peroxidation [44,49]. The application of transepithelial electrical resistance (TEER) could provide helpful information concerning the integrity of the epithelial cell layer, taking into account that its values before and after silica nanoparticle exposition did not change at low concentrations, suggesting that the barrier function remained undamaged [44,49]. Measuring TEER across a barrier is a non-destructive real-time method to assess barrier integrity [50]. In mice, an experiment showed that silica nanoparticles had significant adverse effects on the placental barrier, such as spiral artery impairment, blood flow reduction, and apoptotic cell death in spongiotrophoblasts [51].
Some authors excluded any short-term direct embryotoxic or teratogenic effects of quantum dots, whereas they observed that their long-term accumulation in the maternal organism might increase the risk of adverse effects on embryo development [52]. Furthermore, other authors excluded toxic silver accumulation in embryos/fetuses after the intravenous injection of silver nanoparticles in pregnant mice, despite a notable silver accumulation in the maternal liver, spleen, and visceral yolk sac [53]. This accumulation has been subsequently observed to affect embryonic growth and to delay physical and cognitive development in the offspring, likely through the induction of epigenetic changes in the embryo and the abnormal development of the placenta [54,55]. Consequently, perinatal exposure to silver nanoparticles should be limited or prevented in any case [56,57].
Nanoparticle permeability through the placental barrier may be affected by the disruption of tight junctions, which can compromise its physiological regulatory processes. Wang and coworkers demonstrated in an in vivo animal model that zirconium dioxide nanoparticles translocated through the placental barrier and accumulated in the fetal brain [58]. This process was mediated by receptor-based endocytosis and the tight-junction breakdown in the maternal and fetal blood placental barrier [58].
Different organic, inorganic, or hybrid nanoparticles have been previously tested (Table 2) [36,37,44,45,47,48,49,51,58]. Shojaei and coworkers reviewed the literature in detail on different placental models and the fetal risk assessment, considering various organic and inorganic nanoparticles [36]. Inorganic nanoparticles have been demonstrated to easily cross the blood–placental barrier and induce several toxicological effects [59]. In contrast, organic nanoparticles can be more selective in their target potential and show less toxicological effects [18,59]. However, additional studies are still required in order to broaden their application in the obstetric field [18,59].

Table 2.
Types of nanoparticles.
Surface-functionalized nanoparticles can prevent transplacental passage and promote placental-specific drug delivery, thus enhancing medication safety and efficacy. Optimal results have been achieved, for instance, by combining nanoparticles with specific proteins which are exclusively expressed in the placenta, such as the placental chondroitin sulfate A-binding peptide or oxytocin receptor [60,61,62,63,64,65]. Among surface-functionalized nanoparticles, Zhang and coworkers proposed the use of placenta-specific exosomes as potential carriers for the placental targeted therapy of pregnancy complications [62]. The exosomes are extracellular vesicles with an endosomal origin physiologically produced by cells which are yet to be wholly characterized [66]. Their isolation, too, presents significant challenges, and the optimal methodology has yet to be developed [66]. Delorme-Axford and coworkers, in an ex-vivo study using human trophoblasts in primary cell cultures, found placental-derived trophoblastic exosomes to induce in non-placental cell defense against viral infections such as human cytomegalovirus by delivering exosomal miRNA (chromosome 19 miRNA cluster, C19MC) [67]. In two different time-series studies, the quantity and characteristics of exosomes through normal pregnancies were analyzed using maternal peripheral blood samples [68,69]. They found that placental-derived exosomes increase during the first trimester of pregnancy, and then gradually declined until delivery [68,69,70]. The same research group also found a differential release and function of exosomes in gestational diabetes mellitus [70,71]. Moreover, their therapeutic use is promising because they were found to be well tolerated after repeated treatments [66]. Furthermore, they are efficient at entering other cells with minimal immune clearance, and can be engineered to target specific cell types [66].
Other surface-functionalized nanoparticles can prevent transplacental passage and limit the exposure to the mother compartment. For example, doxorubicin is an anticancer agent that crosses the placenta and can harm the fetus. Soininen and coworkers demonstrated that polyethylene glycol-coated liposomes encapsulating doxorubicin exhibited, both in vitro (BeWo cells) and ex vivo (perfused a placental model), a lower placental permeability than the pH-sensitive liposomal formulation of doxorubicin and free doxorubicin [42].
Although the characteristics of nanoparticles could be predictive of their maternal, placental, or fetal uptake, the achievement of a comprehensive understanding of nanoparticle uptake, accumulation, and translocation, as well as of how their size, shape, surface chemistry, and charge affect biodistribution and therapeutic efficacy through predictive placental transfer models, is absolutely mandatory in order to determine how the timing and route of nanoparticle administration impact their distribution, effectiveness, and safety [72].
5. Point-of-Care Testing and Other Applications in Diagnostics
Along with therapeutic nanoparticles, novel nanotechnologies have been designed to improve diagnostic accuracy. Point-of-care testing is a medical concept to describe diagnostic testing at or near the point of care, which is the place and time of the patient care. This kind of advanced testing can be used in remote locations or outpatient facilities, reducing the costs of traditional medical laboratories, specialized operators, and complex equipment. One of the most prominent examples in obstetrics is the introduction of the home pregnancy test in the 1970s [73]. Through the years, the test has been modified, opening the way to the paper-based diagnostic technologies [74,75]. Generally, paper-based diagnostics, including lateral flow assays and microfluidic paper-based analytical devices, are affordable, user-friendly, rapid, robust, and scalable for manufacturing [74]. These diagnostics are optimal for the improvement of clinical pathways in remote settings and resource-limited areas. Nanotechnology was proposed to strengthen test performance in lateral flow assays or microfluidic paper-based analytical devices [74]. As previously mentioned, the introduction of a point-of-care test for pregnancy has modified the clinical management of early pregnancy, allowing the wide spread of the prompt detection of early pregnancy and the better planning of the following management. Moreover, using a β-HCG point-of-care test while assessing fertile women with low abdominal pain in outpatient facilities allows us, with low resources, to rapidly exclude from the possible causes of pain a life-threatening situation such as extra-uterine pregnancy. The widespread use of these types of test has changed clinical management, and has the potential to change clinical pathways further. Nanotechnology has recently been applied to human chorionic gonadotropin testing to improve detection sensitivity and widen the application of these tests in clinical management [76,77,78,79]. For example, Cai and coworkers assessed the possibility of using a nanotechnology-based test to detect β-hCG levels in peripheral blood and in uterine cervix secretions quantitatively [76]. In addition to the accuracy of the test, they proved that the cervical β-hCG/serum β-hCG ratio was predictive of spontaneous miscarriage and ectopic pregnancy diagnosis [76]. These advances can be essential in order to improve and simplify the diagnostic algorithms, which are standardized step-by-step procedures for reaching a diagnosis or management decision.
Nanotechnology-based approaches were also used to retrieve and isolate trophoblasts from the human cervix during pregnancy, allowing an early attempt at prenatal diagnosis starting from 5 weeks of gestation [80,81]. In particular, Bolnick and coworkers collected a cervical specimen with a brushing system for liquid-based cytology [80]. The cells were marked with an antibody specific for trophoblastic cells (anti-HLA-G) [80]. A secondary antibody conjugated with magnetic nanoparticles was used for the immunomagnetic isolation of the trophoblastic cells [80]. Other applications in the field of diagnostics include cell-free fetal DNA isolation for prenatal screenings [82,83,84,85].
Nanoparticles have been designed to improve imaging techniques which are traditionally considered off-limits during pregnancy. For example, the classical contrast medium of magnetic resonance imaging, gadolinium, is blamed for possible teratogenic and chromosomal damages. In order to overcome this issue, the use of superparamagnetic iron oxide nanoparticle ferumoxytol has been tested, with encouraging results and no impact at the maternal–fetal interface in pregnant rhesus macaques [86]. Moreover, liposomal gadolinium has also been proposed as a promising tool in the obstetrics field [87]. In particular, Shetty and coworkers found, in a mouse model, that liposomal gadolinium nanoparticles were confined to the maternal compartment without passing to the fetal compartment [88]. In addition, Badachhape and coworkers, in a mouse model, were able to visualize and study the retroplacental clear space through gestation using liposomal gadolinium nanoparticles [89]. Finally, microbubble contrast-enhanced ultrasound agents are also promising for the in vivo study of placental pathologies [90]. Even if microbubbles’ content and composition can vary, they are typically formed of an inert gas core stabilized by an outer shell assembled with lipids, carbohydrates, proteins, or polymers [90]. In particular, the advantage of microbubble contrast agents is their use in the study of the microcirculation (which is not accessible with ordinary Doppler-based studies) using an easy-to-use and cost-effective instrument such as the diagnostic ultrasound. Furthermore, it has no tissue toxicity, like gadolinium; meanwhile, it allows an adequate microcirculation study that can be useful in the imaging of the differential diagnosis of the malignant lesion without exposing the pregnant women to radiation or tissue toxicity.
6. Preterm Birth
The use of nanotechnology has been proposed for the treatment of preterm labor. This technology was suggested to improve the accuracy and cost-effectiveness of preterm delivery diagnosis by implementing a fibronectin test using magnetic nanoparticles coated with anti-fibronectin antibodies [91]. Furthermore, it was also applied to improve drug treatment. In the current practice, the use of drugs to reduce uterine contractions and prevent preterm birth is limited due to the systemic or fetal effects of currently available medications such as ritodrine or indomethacin [7,92]. Nanotechnology-based approaches have been developed to prevent the drug’s passage in the fetal bloodstream, and to target its localization at the uterine-wall level [60,61,62,63,64,65]. To this end, liposomes coated with antibodies or specific receptor antagonists were developed (e.g., placental chondroitin sulfate A-binding peptide, oxytocin receptor antagonist, or oxytocin receptor antibody) [60,61,62,63,64,65]. Moreover, nanotechnology was also used to improve the effectiveness of preterm-delivery-preventive drugs via the vaginal administration of progesterone, significantly reducing the prevalence of preterm birth in a mouse model and lengthening the time-to-delivery outcome by 39% [93]. In addition to the implementation of new devices for the administration of already known drugs, nanotechnology has also been exploited to optimize the delivery of innovative medicines, in order to counteract the mechanisms of inflammation known to be involved in preterm labor and offspring complications (e.g., improving the delivery of N-acetylcysteine) [94,95].
7. Preeclampsia and Fetal Growth Restriction
Both preeclampsia and fetal growth restriction are associated with aging and dysfunctional placentae [96]. In this field, nanotechnology was also applied with different intents: (1) to improve diagnostic management and (2) to implement treatment strategies. New nanotechnology-based point-of-care tests are being developed to improve the assessment of preeclampsia development risk [97,98,99]. For instance, recent advancements have been made in the development of electrochemical immunosensors for the early clinical diagnosis of preeclampsia within the 18–20th gestational weeks [100]. In particular, the aim would be to establish a high-specificity immuno-diagnostic platform which is able to detect and analyze multiple molecules simultaneously (at the picomolar resolution), which could be highly predictive for preeclampsia onset to anticipate and improve its further treatment. There is also a tremendous effort in the development of nanotechnology-based approaches to target the placental tissue in order to prevent or mild the consequences of preeclampsia and fetal growth restriction [101,102,103,104,105,106,107]. Another field of interest was the development of new disease-specific models [108]. In a recent article, Yu and coworkers precisely silenced a long non-coding RNA (lncRNA H19) in the placental tissue of a mouse model, obtaining in vivo the occurrence of pre-eclampsia-like symptoms [108]. This result is extremely important, and opens the path to future discoveries. First, the previous models of pre-eclampsia were mainly established by the systemic administration of drugs or surgery, hence inducing unwanted systemic toxicity and limiting the possible understanding of pre-eclampsia. Pre-eclampsia is supposed to be an obstetric pathology of placental origin [96]. Removing the placenta will bring back normal homeostasis in the mother, healing pre-eclampsia. This model lets us start pre-eclampsia from the placenta and not from a systemic interference, allowing us to understand the disease better. This result also means the possible development of new therapeutic strategies. In addition, this kind of model will allow the testing of possible etiopathogenic pathways in vivo, corroborating an eventual causative hypothesis.
8. Diabetes Mellitus
Nanotechnology has been proposed for the implementation of highly accurate tests for the diagnosis and monitoring of diabetes mellitus [109]. Recently, a nanotechnology-based point-of-care test was developed with the intent to monitor glycated albumin in gestational diabetic pregnancies [110]. In addition, different approaches (e.g., Cerium nanoparticles or zinc oxide resveratrol encapsulated in Chitosan) were also developed to treat gestational diabetes and its comorbidities [109,111,112,113]. For example, Du and coworkers found, in a streptozocin-induced diabetes rat model, that zinc oxide resveratrol encapsulated in Chitosan improves the diabetic biological signs, including a reduction in insulin resistance [112]. Moreover, recently in the same diabetes animal model, it was found that selenium nanoparticles could mitigate diabetic nephropathy during pregnancy [114].
9. In Utero Gene Therapy
In 2019, a viral-vector-based gene therapy for spinal muscular atrophy that delivers a functional copy of the survival of motor neuron gene cDNA was approved by the FDA. Since then, the interest in this field has grown [115], and nanoparticles have been exploited as carriers of nucleic acids in utero during embryonic or fetal life [116,117,118,119]. Although no human studies have been carried out until now for in utero gene therapy, many in vitro and in vivo studies have been performed [120]. In utero gene therapy is a potential game-changer for monogenic diseases because the treatment can prevent disease inception, avoiding early damage to the tissues. Another advantage is the prevention of immune system reactions to the gene therapy approach, limiting or overcoming its effectiveness, as it occurs in post-natal genetic therapy. Other potential benefits are the capacity to cross the blood–brain barrier or deliver the treatment with a high vector-to-target-cell ratio [120].
The broad container of gene therapy comprises endogenous gene editing, gene replacement or augmentation, and the use of antisense oligonucleotides to regulate protein expression [120]. However, these techniques bring some risks, such as insertional mutagenesis (observed in post-natal treatment) or the threat of germline transfer. In particular, insertional mutagenesis results from off-target gene insertion with the disruption of another biological pathway. An example of insertional mutagenesis was the development of T-cell leukemia several years after gene therapy for X-linked severe combined immunodeficiency in four patients [121]. In these four cases, the retroviral vector had inserted within the LMO-2 locus, which is known to be involved in T-cell leukemia development [120,121]. However, the potential is high, and preclinical studies focus on various pathologies such as spinal muscular atrophy, thalassemia, neuronopathic Gaucher disease, congenital retinal blindness, and cystic fibrosis [120].
10. Assisted Reproductive Technology
Another fascinating application field of nanomedicine related to pregnancy is assisted reproductive technology. With the progressive delay of first pregnancies due to contemporary social and working habits, modern society is now experiencing its highest infertility rate [15,122]. Novel techniques have been consequently developed to improve the conception rate in couples utilizing assisted reproductive technology. Exploring nanotechnology in non-human models is stimulating, as they make it possible to optimize newly developed protocols using nanomaterials against the impairments still faced by reproductive medicine [123,124]. For example, Abreu and coworkers used a non-invasive nanotechnology-based system to find the embryos with the highest chances of successful implantation among a pool of morphologically identical viable embryos [124]. In a preliminary study, they used an immunosensor to detect the embryo beta-HCG production, allowing improved embryo selection before implantation (the detection vs. no detection of beta-HCG was found to be a favorable prognostic factor for the establishment of a pregnancy) [124].
11. Cancer in Pregnancy
The increase of pregnancy-related cancer prevalence is a critical issue, partly due to the progressive delay of first pregnancies in high-income countries for work and social reasons [15]. Pregnant cancer patients represent a major concern in the modern maternal and fetal health field [125,126]. However, nowadays, many intervention options exist which can dramatically change patients’ prognosis, and consequently positively improve the therapeutic outcomes [19,127]. In most cases, patients require a personalized and integrated treatment which is necessarily drawn within a multidisciplinary setting in order to prevent the iatrogenic pregnancy complications which could be caused by cancer treatment, such as preterm delivery or impaired fetal growth [125].
The most frequent malignancy diagnosed during pregnancy is breast cancer [125,126]. Nanomedicine improvements can allow the safe administration of antiblastic agents during pregnancy, avoid improper therapeutic delays, and enable highly effective and safe treatment for the mother and the offspring [126]. An example is represented by the worldwide commercially available nanoformulations of paclitaxel, which are adopted against breast cancer due to their recognized favorable dosing regimens and low side effect profiles. However, transplacental transport and resultant fetal exposure to this nanoformulation remains an issue of debate, and ongoing studies are still drawing the future development of rational and safe treatment strategies for pregnancy-associated breast cancer and other perinatal diseases [128,129]. Additional formulations exist, such as polylactic glycolic-acid-encapsulated paclitaxel associated with Vitamin D3, liposomal doxorubicin, nanosomal docetaxel lipid suspension, or pegylated liposomal doxorubicin [42,126,130,131]. Recently, Ramaswamy and coworkers found that using, in a combination chemotherapy, the nanosomal docetaxel lipid suspension during pregnancy in a pregnant woman with fourth-stage breast cancer disease was safe and effective [126]. Liposome nanoparticles were also used to carry other drugs (e.g., NSAIDs or indomethacin) and limit their access to the fetal compartment [132]. Although rarer than breast cancer, other cancer types can also be diagnosed during pregnancy, such as hematological, skin, or ovarian cancer [125]. Recently, a review clearly outlined how nanotechnology has ushered in a new era of treatment in many different types of cancers [133]. Nanotechnology has allowed the improvement of pharmacokinetics, biocompatibility, and tumor targeting; the overcoming drug resistance; and the reduction of systemic toxicity [133].
In addition, several monoclonal antibodies have been developed that target the Her2 protein, expressed in several breast cancer patients, and with a notoriously poor prognosis [134]. The efficacy of these drugs in both the neoadjuvant and adjuvant setting has been one of the greatest successes of the last few decades against this frequent and life-threatening disease. Monoclonal antibodies represent a prime example of the most efficient modalities of targeted drug delivery we have today against malignancies, and probably represent the future of personalized medicine in the oncological field. Unfortunately, there is insufficient evidence to justify their use during pregnancy, which is why their administration is generally delayed in the puerperium. However, we can imagine that in the future, we will likely be able to offer monoclonal antibodies in formulations which are capable of sparing the fetus while battling the pregnant woman’s neoplastic disease. In fact, targeted liposomal carriers are an emerging field of research, and are capable of targeting a compartment or tissue specifically [63,64,65,132]. For example, Refuerzo and coworkers demonstrated in a mouse model that delivering indomethacin within multilamellar liposomes prevents the drug from passing the placental barrier, significantly reducing fetal exposure [132].
12. Bibliometric Analysis
A total of 4031 items were available in the Scopus database. The annual growth rate was 7.73%, the average number of citations per item was 26.55, the average number of citations per item per year was 3.30, and the total number of references cited by the 4031 included items was 230,112. Among the top five sources, two were obstetrics and gynecological journals (Placenta and American Journal of Obstetrics and Gynecology), and three were more generalist journals (International Journal of Molecular Sciences, Plos One, and Scientific Reports). Figure 1A,B illustrates the countries’ collaboration networks. The network shows active collaborations between high-income and low-income countries where some obstetrical pathologies’ prevalence is high, as are their detrimental consequences (e.g., Sub-Saharan Africa) (Figure 1B). Many efforts have been established in low-income countries to reduce maternal mortality and achieve the WHO Sustainable Development Goals [135]. Despite significant structural changes, still more work is needed in order to reduce maternal mortality due to pre-eclampsia and other obstetric complications [135]. The collaborations between high-income and low-income countries are essential in nanotechnology because of the possible advantages of the development of highly accurate and affordable point-of-care tests that can be cost-effectively implemented in a low-resource setting. Figure 2 shows the relative frequency of some keywords over the years. Figure 2A shows how the reference to humans has grown over time, as has the reference to controlled studies and clinical trials. However, the latter has slowed down over the last decade. At the same time, the reference to animal models is the most frequent, and has increased further in the last decade. Figure 2B shows the zenith of pharmacological safety and efficacy keyword frequency around 2010. Only since the end of the last decade has there emerged an increased reference to pre-eclampsia and diabetes in pregnancy (Figure 2B). Figure 2C shows the growing literature concerning drug delivery systems up to 2010; meanwhile, interest in exosomes has increased since then. Figure 3A shows the co-occurrence of words in the item titles showing five major clusters: one focused on drug delivery, one focused on fetal exposure, one focused on placental vesicles, one focused specifically on placental exosomes, and one focused on diabetes. Figure 3B shows the co-occurrence of words in the abstracts where two clusters were found, and one was focused on disease treatment. Among the top five most cited articles among the 4031 analyzed items in the Scopus database that were already discussed in this narrative review, three were about exosomes [68,69,70], one was about placental barrier capacity [32], and one was about metal nanoparticle toxicity [51].
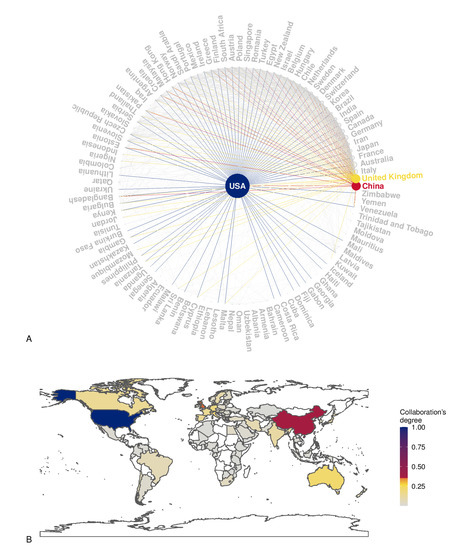
Figure 1.
Panel (A): The plot shows the country collaboration network based on Scopus data. The vertex size is proportional to the number of collaborations, and the networks of the three most connected countries are highlighted. Panel (B): Spatial visualization of the degree of collaboration; blue and red colors correspond to the countries with more collaborations.
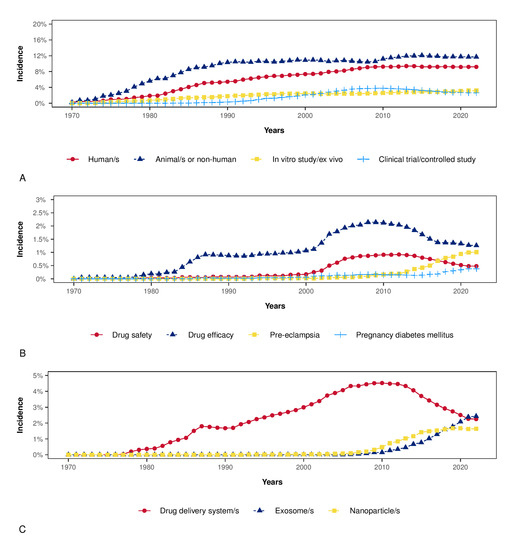
Figure 2.
These plots show the annual incidence of a selected group of keywords among the top 100 (based on Scopus data). Panel (A): Keywords comprising the main type of studies. Panel (B): Keywords comprising the main topics in obstetrics. Panel (C): Trends in the annual incidence of the keyword “exosome” in comparison to “drug delivery system” and “nanoparticles”.
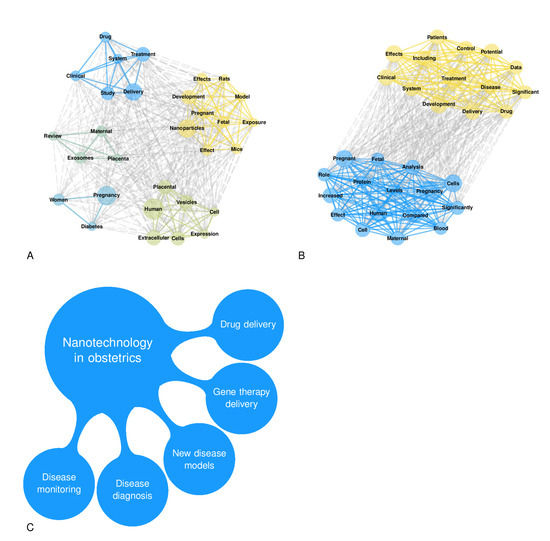
Figure 3.
Panel (A): This network plot shows the title word co-occurrences from the Scopus database, considering the top 30 most frequent words. Panel (B): This network plot shows the abstract word co-occurrences from the Scopus database, considering the top 30 most frequent words. Panel (C): Main applications of nanotechnology in obstetrics.
13. Limitations
Despite this field’s tremendous interest and promises, nanotoxicity is the primary hindrance. Before using nanotechnology in the therapeutic management of pregnancy complications, more evidence should be collected in this field. However, it should be highlighted that this point is less limiting in diagnostic applications where there is no direct contact between the nanotechnology device and the pregnant patient, nor is there a risk of long-term adverse effects. A promising and emerging topic is the use of engineered exosomes as carriers. However, there are some limitations for the widespread adoption of exosomes. First, isolation technology should be improved. Second, their physiologic processes still need to be cleared entirely.
14. Implications for Clinicians and Policy-Makers/Healthcare Providers
The interest in nanotechnology is high because of the expected benefits to global society. The studies included in this review support these expectations, and we foresee growing support from the stakeholders for the expansion of research into the development of nanotechnology to improve healthcare in general and during pregnancy. Consequently, this increases exposure to nanomaterials, which also have potential toxic effects. For the foreseeable future, a universal set of guidelines for the nanotoxicity assessment of nanomaterials used in pregnancy by regulatory agencies will become mandatory in order to allow pregnant women to safely reap the benefits of nanotechnology-enabled products while assisting in the implementation of exposure controls to ensure maternal and fetal safety [136].
15. Unanswered Questions and Future Research
Nanotechnology in obstetrics is an evolving field, and the main suggested applications are shown in Figure 3C. In our opinion, the main impending questions to be answered in the future are presented in Table 3. In particular, we believe that the leading two open questions are the following: whether nanotechnology will allow for the early diagnosis (as soon as five weeks of gestation) of the major obstetrics pathologies and prenatal screening, and whether nanotechnology-based therapies will safely and specifically target the maternal compartment, the placenta, or the fetal compartment while minimizing therapy side effects.

Table 3.
Open questions.
16. Conclusions
In summary, this review showed the increasing interest in the application of nanotechnology-based strategies in obstetrics, such as improving the diagnostic algorithms, monitoring pregnancy-related diseases, and implementing new treatment approaches. However, their clinical application is yet to achieve maturity, despite the growing interest in the literature in the targeting of specific pregnancy pathologies in recent years. An emergent and promising approach is the use of exosomes to specifically target the therapeutic agents and avoid the toxicity of inorganic nanocarriers. Although the bibliometric analysis revealed a broad collaboration between high-income and low-income countries, in the future, the greater involvement of low-income countries would be desirable. Nanotechnology-based point-of-care tests have the potential to spread in low-resource settings, as beneficial instruments to improve the management of high-risk pregnancies and help to achieve the WHO Sustainable Development Goals Agenda.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/jpm12081324/s1. Table S1: Summary of database queries.
Author Contributions
Substantial contributions to the conception and design or acquisition of the data, or to analysis and interpretation of the data, S.B., B.C., L.S., J.A.D.Ñ., C.C., A.F., D.C., A.C. and A.P.L.; Drafting the article or revising it critically for important intellectual content, S.B., B.C., L.S., J.A.D.Ñ., C.C., A.F., D.C., A.C. and A.P.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The present study is exempt from ethical approval, as this meta-analysis only involves anonymous data which have already been published.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data were extracted from previously published studies, which are publicly available.
Acknowledgments
The authors would like to thank the whole staff who collaborated on the article collection, selection, and reading, and in the paper writing and reviewing.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pearlman, S.A. Advancements in neonatology through quality improvement. J Perinatol. 2022, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Adane, A.A.; Bailey, H.D.; Marriott, R.; Farrant, B.M.; White, S.W.; Stanley, F.J.; Shepherd, C.C.J. Disparities between Aboriginal and non-Aboriginal perinatal mortality rates in Western Australia from 1980 to 2015. Paediatr. Perinat. Epidemiol. 2019, 33, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Allen, K.M.; Smith, B.; Iliev, I.; Evans, J.; Werthammer, J. Short term cost of care for the surviving periviable neonate. J. Neonatal Perinat. Med. 2017, 10, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.A.; Petrou, S.; Dritsaki, M.; Johnson, S.J.; Manktelow, B.; Draper, E.S.; Smith, L.K.; Seaton, S.E.; Marlow, N.; Dorling, J.; et al. Economic costs associated with moderate and late preterm birth: A prospective population-based study. BJOG 2015, 122, 1495–1505. [Google Scholar] [CrossRef] [PubMed]
- Chawanpaiboon, S.; Vogel, J.P.; Moller, A.-B.; Lumbiganon, P.; Petzold, M.; Hogan, D.; Landoulsi, S.; Jampathong, N.; Kongwattanakul, K.; Laopaiboon, M.; et al. Global, regional, and national estimates of levels of preterm birth in 2014: A systematic review and modelling analysis. Lancet Glob. Health 2019, 7, e37–e46. [Google Scholar] [CrossRef]
- Liu, L.; Oza, S.; Hogan, D.; Perin, J.; Rudan, I.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, regional, and national causes of child mortality in 2000-13, with projections to inform post-2015 priorities: An updated systematic analysis. Lancet 2015, 385, 430–440. [Google Scholar] [CrossRef]
- Driul, L.; Londero, A.P.; Adorati-Menegato, A.; Vogrig, E.; Bertozzi, S.; Fachechi, G.; Forzano, L.; Cacciaguerra, G.; Perin, E.; Miceli, A.; et al. Therapy side-effects and predictive factors for preterm delivery in patients undergoing tocolysis with atosiban or ritodrine for threatened preterm labour. J. Obstet. Gynaecol. 2014, 34, 684–689. [Google Scholar] [CrossRef]
- Fruscalzo, A.; Londero, A.P.; Orsaria, M.; Marzinotto, S.; Driul, L.; Di Loreto, C.; Mariuzzi, L. Placental fibronectin staining is unaffected in pregnancies complicated by late-onset intrauterine growth restriction and small for gestational age fetuses. Int. J. Gynaecol. Obstet. 2020, 148, 253–260. [Google Scholar] [CrossRef]
- Xodo, S.; Cecchini, F.; Celante, L.; Novak, A.; Rossetti, E.; Baccarini, G.; Londero, A.P.; Driul, L. Preeclampsia and low sodium: A retrospective cohort analysis and literature review. Pregnancy Hypertens. 2021, 23, 169–173. [Google Scholar] [CrossRef]
- Visentin, S.; Londero, A.P.; Camerin, M.; Grisan, E.; Cosmi, E. A possible new approach in the prediction of late gestational hypertension: The role of the fetal aortic intima-media thickness. Medicine 2017, 96, e5515. [Google Scholar] [CrossRef]
- Visentin, S.; Londero, A.P.; Grumolato, F.; Trevisanuto, D.; Zanardo, V.; Ambrosini, G.; Cosmi, E. Timing of delivery and neonatal outcomes for small-for-gestational-age fetuses. J. Ultrasound Med. 2014, 33, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- Fruscalzo, A.; Cividino, A.; Rossetti, E.; Maurigh, A.; Londero, A.P.; Driul, L. First trimester PAPP-A serum levels and long-term metabolic outcome of mothers and their offspring. Sci. Rep. 2020, 10, 5131. [Google Scholar] [CrossRef] [PubMed]
- Fruscalzo, A.; Londero, A.P.; Driul, L.; Henze, A.; Tonutti, L.; Ceraudo, M.; Zanotti, G.; Berni, R.; Schweigert, F.J.; Raila, J. First trimester concentrations of the TTR-RBP4-retinol complex components as early markers of insulin-treated gestational diabetes mellitus. Clin. Chem. Lab. Med. 2015, 53, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Xodo, S.; Londero, A.P.; D’Agostin, M.; Novak, A.; Galasso, S.; Pittini, C.; Baccarini, G.; Grimaldi, F.; Driul, L. Is Glycated Hemoglobin A1c Level Associated with Adverse Pregnancy Outcomes of Women Affected by Pre-Gestational Diabetes? Medicina 2021, 57, 461. [Google Scholar] [CrossRef]
- Londero, A.P.; Rossetti, E.; Pittini, C.; Cagnacci, A.; Driul, L. Maternal age and the risk of adverse pregnancy outcomes: A retrospective cohort study. BMC Pregnancy Childbirth 2019, 19, 261. [Google Scholar] [CrossRef]
- Johansson, A.L.; Fredriksson, I.; Mellemkjaer, L.; Stensheim, H.; Lähteenmäki, P.; Winther, J.F.; Ullenhag, G.J.; Lundberg, F.E. Cancer survival in women diagnosed with pregnancy-associated cancer: An overview using nationwide registry data in Sweden 1970–2018. Eur. J. Cancer 2021, 155, 106–115. [Google Scholar] [CrossRef]
- Wolters, V.; Heimovaara, J.; Maggen, C.; Cardonick, E.; Boere, I.; Lenaerts, L.; Amant, F. Management of pregnancy in women with cancer. Int. J. Gynecol. Cancer 2021, 31, 314–322. [Google Scholar] [CrossRef]
- Kannan, R.M.; Kannan, S. Emerging nanomedicine approaches in obstetrics. Am. J. Obstet. Gynecol. 2017, 216, 201–203. [Google Scholar] [CrossRef][Green Version]
- Tesarova, P.; Pavlista, D.; Parizek, A. Is It Possible to Personalize the Diagnosis and Treatment of Breast Cancer during Pregnancy? J. Pers. Med. 2020, 11, 18. [Google Scholar] [CrossRef]
- Patel, S.P.; Patel, P.B.; Parekh, B.B. Application of nanotechnology in cancers prevention, early detection and treatment. J. Cancer Res. Ther. 2014, 10, 479–486. [Google Scholar]
- Barkalina, N.; Charalambous, C.; Jones, C.; Coward, K. Nanotechnology in reproductive medicine: Emerging applications of nanomaterials. Nanomedicine 2014, 10, 921–938. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, B.B.S.; Lasham, A.; Shelling, A.N.; Al-Kassas, R. Nanoparticle therapeutics: Technologies and methods for overcoming cancer. Eur. J. Pharm. Biopharm. 2015, 97, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Aria, M.; Cuccurullo, C. Bibliometrix: An R-tool for comprehensive science mapping analysis. J. Informetr. 2017, 11, 959–975. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022. [Google Scholar]
- Glover, D.D.; Amonkar, M.; Rybeck, B.F.; Tracy, T.S. Prescription, over-the-counter, and herbal medicine use in a rural, obstetric population. Am. J. Obstet. Gynecol. 2003, 188, 1039–1045. [Google Scholar] [CrossRef]
- Lee, E.; Maneno, M.K.; Smith, L.; Weiss, S.R.; Zuckerman, I.H.; Wutoh, A.K.; Xue, Z. National patterns of medication use during pregnancy. Pharmacoepidemiol. Drug Saf. 2006, 15, 537–545. [Google Scholar] [CrossRef]
- Refuerzo, J.S.; Blackwell, S.C.; Sokol, R.J.; Lajeunesse, L.; Firchau, K.; Kruger, M.; Sorokin, Y. Use of over-the-counter medications and herbal remedies in pregnancy. Am. J. Perinatol. 2005, 22, 321–324. [Google Scholar] [CrossRef]
- Joshi, M.D. Drug delivery during pregnancy: How can nanomedicine be used? Ther. Deliv. 2017, 8, 1023–1025. [Google Scholar] [CrossRef]
- Irvin-Choy, N.S.; Nelson, K.M.; Gleghorn, J.P.; Day, E.S. Design of nanomaterials for applications in maternal/fetal medicine. J. Mater. Chem. B 2020, 8, 6548–6561. [Google Scholar] [CrossRef]
- Pritchard, N.; Kaitu’u-Lino, T.; Harris, L.; Tong, S.; Hannan, N. Nanoparticles in pregnancy: The next frontier in reproductive therapeutics. Hum. Reprod. Update 2021, 27, 280–304. [Google Scholar] [CrossRef]
- Keelan, J.A.; Leong, J.W.; Ho, D.; Iyer, K.S. Therapeutic and safety considerations of nanoparticle-mediated drug delivery in pregnancy. Nanomedicine 2015, 10, 2229–2247. [Google Scholar] [CrossRef]
- Wick, P.; Malek, A.; Manser, P.; Meili, D.; Maeder-Althaus, X.; Diener, L.; Diener, P.-A.; Zisch, A.; Krug, H.F.; Von Mandach, U. Barrier capacity of human placenta for nanosized materials. Environ. Health Perspect. 2010, 118, 432–436. [Google Scholar] [CrossRef] [PubMed]
- de Araújo, T.E.; Milián, I.C.B.; de Souza, G.; da Silva, R.J.; Rosini, A.M.; Guirelli, P.M.; Franco, P.S.; Barbosa, B.F.; Ferro, E.A.V.; da Costa, I.N. Experimental models of maternal-fetal interface and their potential use for nanotechnology applications. Cell Biol. Int. 2020, 44, 36–50. [Google Scholar] [CrossRef] [PubMed]
- Caporale, N.; Leemans, M.; Birgersson, L.; Germain, P.-L.; Cheroni, C.; Borbély, G.; Engdahl, E.; Lindh, C.; Bressan, R.B.; Cavallo, F.; et al. From cohorts to molecules: Adverse impacts of endocrine disrupting mixtures. Science 2022, 375, eabe8244. [Google Scholar] [CrossRef] [PubMed]
- Fliedel, L.; Alhareth, K.; Mignet, N.; Fournier, T.; Andrieux, K. Placental Models for Evaluation of Nanocarriers as Drug Delivery Systems for Pregnancy Associated Disorders. Biomedicines 2022, 10, 936. [Google Scholar] [CrossRef]
- Shojaei, S.; Ali, M.S.; Suresh, M.; Upreti, T.; Mogourian, V.; Helewa, M.; Labouta, H.I. Dynamic placenta-on-a-chip model for fetal risk assessment of nanoparticles intended to treat pregnancy-associated diseases. Biochim. Biophys. Acta Mol. Basis Dis. 2021, 1867, 166131. [Google Scholar] [CrossRef]
- Tutar, R.; Çelebi-Saltik, B. Modeling of Artificial 3D Human Placenta. Cells Tissues Organs 2021, 1–10. [Google Scholar] [CrossRef]
- Viola, L.; Marzinotto, S.; Bertacchi, M.; Londero, A.P.; Orsaria, M.; Bertozzi, S.; Driul, L.; Di Loreto, C.; Studer, M.; Mariuzzi, L.; et al. COUP-TFI deletion affects angiogenesis and apoptosis related gene expression in mouse placenta: Results of an explorative study. Clin. Exp. Obstet. Gynecol. 2022, 49, 1. [Google Scholar] [CrossRef]
- Al-Enazy, S.; Ali, S.; Albekairi, N.; El-Tawil, M.; Rytting, E. Placental control of drug delivery. Adv. Drug Deliv. Rev. 2017, 116, 63–72. [Google Scholar] [CrossRef]
- Rattanapinyopituk, K.; Shimada, A.; Morita, T.; Sakurai, M.; Asano, A.; Hasegawa, T.; Inoue, K.; Takano, H. Demonstration of the clathrin- and caveolin-mediated endocytosis at the maternal-fetal barrier in mouse placenta after intravenous administration of gold nanoparticles. J. Vet. Med. Sci. 2014, 76, 377–387. [Google Scholar] [CrossRef]
- Kloet, S.K.; Walczak, A.P.; Louisse, J.; van den Berg, H.H.; Bouwmeester, H.; Tromp, P.; Fokkink, R.G.; Rietjens, I.M. Translocation of positively and negatively charged polystyrene nanoparticles in an in vitro placental model. Toxicol. In Vitro 2015, 29, 1701–1710. [Google Scholar] [CrossRef]
- Soininen, S.K.; Repo, J.K.; Karttunen, V.; Auriola, S.; Vähäkangas, K.H.; Ruponen, M. Human placental cell and tissue uptake of doxorubicin and its liposomal formulations. Toxicol. Lett. 2015, 239, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Cureton, N.; Korotkova, I.; Baker, B.; Greenwood, S.; Wareing, M.; Kotamraju, V.R.; Teesalu, T.; Cellesi, F.; Tirelli, N.; Ruoslahti, E.; et al. Selective Targeting of a Novel Vasodilator to the Uterine Vasculature to Treat Impaired Uteroplacental Perfusion in Pregnancy. Theranostics 2017, 7, 3715–3731. [Google Scholar] [CrossRef]
- Ali, S.; Rytting, E. Influences of nanomaterials on the barrier function of epithelial cells. Adv. Exp. Med. Biol. 2014, 811, 45–54. [Google Scholar]
- Rytting, E.; Cartwright, L.; Poulsen, M.S.; Nielsen, H.M.; Pojana, G.; Knudsen, L.E.; Saunders, M. In vitro placental model optimization for nanoparticle transport studies. Int. J. Nanomed. 2012, 7, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Wilson, M.R.; MacNee, W.; Stone, V.; Donaldson, K. Size-dependent proinflammatory effects of ultrafine polystyrene particles: A role for surface area and oxidative stress in the enhanced activity of ultrafines. Toxicol. Appl. Pharmacol. 2001, 175, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, S.J.; Crompton, L.A.; Sood, A.; Saunders, M.; Boyle, N.T.; Buckley, A.; Minogue, A.M.; McComish, S.F.; Jiménez-Moreno, N.; Cordero-Llana, O.; et al. Nanoparticle-induced neuronal toxicity across placental barriers is mediated by autophagy and dependent on astrocytes. Nat. Nanotechnol. 2018, 13, 427–433. [Google Scholar] [CrossRef]
- Menjoge, A.R.; Rinderknecht, A.L.; Navath, R.S.; Faridnia, M.; Kim, C.J.; Romero, R.; Miller, R.K.; Kannan, R.M. Transfer of PAMAM dendrimers across human placenta: Prospects of its use as drug carrier during pregnancy. J. Control. Release 2011, 150, 326–338. [Google Scholar] [CrossRef]
- Poulsen, M.S.; Mose, T.; Maroun, L.L.; Mathiesen, L.; Knudsen, L.E.; Rytting, E. Kinetics of silica nanoparticles in the human placenta. Nanotoxicology 2015, 9 (Suppl 1), 79–86. [Google Scholar] [CrossRef]
- Odijk, M.; van der Meer, A.D.; Levner, D.; Kim, H.J.; van der Helm, M.W.; Segerink, L.I.; Frimat, J.-P.; Hamilton, G.A.; Ingber, D.E.; Berg, A.V.D. Measuring direct current trans-epithelial electrical resistance in organ-on-a-chip microsystems. Lab Chip 2015, 15, 745–752. [Google Scholar] [CrossRef]
- Yamashita, K.; Yoshioka, Y.; Higashisaka, K.; Mimura, K.; Morishita, Y.; Nozaki, M.; Yoshida, T.; Ogura, T.; Nabeshi, H.; Nagano, K.; et al. Silica and titanium dioxide nanoparticles cause pregnancy complications in mice. Nat. Nanotechnol. 2011, 6, 321–328. [Google Scholar] [CrossRef]
- Zalgeviciene, V.; Kulvietis, V.; Bulotiene, D.; Didziapetriene, J.; Rotomskis, R. The effect of nanoparticles in rats during critical periods of pregnancy. Medicina 2012, 48, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Austin, C.A.; Hinkley, G.K.; Mishra, A.R.; Zhang, Q.; Umbreit, T.H.; Betz, M.W.; Wildt, B.E.; Casey, B.J.; Francke-Carroll, S.; Hussain, S.M.; et al. Distribution and accumulation of 10 nm silver nanoparticles in maternal tissues and visceral yolk sac of pregnant mice, and a potential effect on embryo growth. Nanotoxicology 2016, 10, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Ema, M.; Okuda, H.; Gamo, M.; Honda, K. A review of reproductive and developmental toxicity of silver nanoparticles in laboratory animals. Reprod. Toxicol. 2017, 67, 149–164. [Google Scholar] [CrossRef]
- Zhang, X.F.; Park, J.H.; Choi, Y.J.; Kang, M.H.; Gurunathan, S.; Kim, J.H. Silver nanoparticles cause complications in pregnant mice. Int. J. Nanomed. 2015, 10, 7057–7071. [Google Scholar]
- Salim, E.I.; Abdel-Halim, K.Y.; Abu-Risha, S.E.; Abdel-Latif, A.S. Induction of 8-hydroxydeoxyguanosine and ultrastructure alterations by silver nanoparticles attributing to placental transfer in pregnant rats and fetuses. Hum. Exp. Toxicol. 2019, 38, 734–745. [Google Scholar] [CrossRef] [PubMed]
- Campagnolo, L.; Massimiani, M.; Vecchione, L.; Piccirilli, D.; Toschi, N.; Magrini, A.; Bonanno, E.; Scimeca, M.; Castagnozzi, L.; Buonanno, G.; et al. Silver nanoparticles inhaled during pregnancy reach and affect the placenta and the foetus. Nanotoxicology 2017, 11, 687–698. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, C.; Liu, X.; Huang, F.; Wang, Z.; Yan, B. Oral intake of ZrO2 nanoparticles by pregnant mice results in nanoparticles’ deposition in fetal brains. Ecotoxicol. Environ. Saf. 2020, 202, 110884. [Google Scholar] [CrossRef]
- Pereira, K.V.; Giacomeli, R.; Gomes de Gomes, M.; Haas, S.E. The challenge of using nanotherapy during pregnancy: Technological aspects and biomedical implications. Placenta 2020, 100, 75–80. [Google Scholar] [CrossRef]
- Zhang, B.; Chen, Z.; Han, J.; Li, M.; Nayak, N.R.; Fan, X. Comprehensive Evaluation of the Effectiveness and Safety of Placenta-Targeted Drug Delivery Using Three Complementary Methods. J. Vis. Exp. 2018, 139, e58219. [Google Scholar] [CrossRef]
- Zhang, B.; Tan, L.; Yu, Y.; Wang, B.; Chen, Z.; Han, J.; Li, M.; Chen, J.; Xiao, T.; Ambati, B.K.; et al. Placenta-specific drug delivery by trophoblast-targeted nanoparticles in mice. Theranostics 2018, 8, 2765–2781. [Google Scholar] [CrossRef]
- Zhang, B.; Liang, R.; Zheng, M.; Cai, L.; Fan, X. Surface-Functionalized Nanoparticles as Efficient Tools in Targeted Therapy of Pregnancy Complications. Int. J. Mol. Sci. 2019, 20, 3642. [Google Scholar] [CrossRef] [PubMed]
- Refuerzo, J.S.; Leonard, F.; Bulayeva, N.; Gorenstein, D.; Chiossi, G.; Ontiveros, A.; Longo, M.; Godin, B. Uterus-targeted liposomes for preterm labor management: Studies in pregnant mice. Sci. Rep. 2016, 6, 34710. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.; Vaughan, B. In vitro comparison of liposomal drug delivery systems targeting the oxytocin receptor: A potential novel treatment for obstetric complications. Int. J. Nanomed. 2019, 14, 2191–2206. [Google Scholar] [CrossRef]
- Hua, S. Synthesis and in vitro characterization of oxytocin receptor targeted PEGylated immunoliposomes for drug delivery to the uterus. J. Liposome Res. 2019, 29, 357–367. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef] [PubMed]
- Delorme-Axford, E.; Donker, R.B.; Mouillet, J.-F.; Chu, T.; Bayer, A.; Ouyang, Y.; Wang, T.; Stolz, D.B.; Sarkar, S.N.; Morelli, A.E.; et al. Human placental trophoblasts confer viral resistance to recipient cells. Proc. Natl. Acad. Sci. USA 2013, 110, 12048–12053. [Google Scholar] [CrossRef]
- Salomon, C.; Torres, M.J.; Kobayashi, M.; Scholz-Romero, K.; Sobrevia, L.; Dobierzewska, A.; Illanes, S.E.; Mitchell, M.D.; Rice, G.E. A gestational profile of placental exosomes in maternal plasma and their effects on endothelial cell migration. PLoS ONE 2014, 9, e98667. [Google Scholar] [CrossRef]
- Sarker, S.; Scholz-Romero, K.; Perez, A.; Illanes, S.E.; Mitchell, M.D.; Rice, G.E.; Salomon, C. Placenta-derived exosomes continuously increase in maternal circulation over the first trimester of pregnancy. J. Transl. Med. 2014, 12, 204. [Google Scholar] [CrossRef]
- Mitchell, M.D.; Peiris, H.N.; Kobayashi, M.; Koh, Y.Q.; Duncombe, G.; Illanes, S.E.; Rice, G.E.; Salomon, C. Placental exosomes in normal and complicated pregnancy. Am. J. Obstet. Gynecol. 2015, 213, S173–S181. [Google Scholar] [CrossRef]
- Salomon, C.; Scholz-Romero, K.; Sarker, S.; Sweeney, E.; Kobayashi, M.; Correa, P.; Longo, S.; Duncombe, G.; Mitchell, M.D.; Rice, G.E.; et al. Gestational Diabetes Mellitus Is Associated With Changes in the Concentration and Bioactivity of Placenta-Derived Exosomes in Maternal Circulation Across Gestation. Diabetes 2016, 65, 598–609. [Google Scholar] [CrossRef]
- Muoth, C.; Aengenheister, L.; Kucki, M.; Wick, P.; Buerki-Thurnherr, T. Nanoparticle transport across the placental barrier: Pushing the field forward! Nanomedicine 2016, 11, 941–957. [Google Scholar] [CrossRef] [PubMed]
- Leavitt, S. “A private little revolution”: The home pregnancy test in American culture. Bull. Hist. Med. 2006, 80, 317–345. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Wang, S.; Wang, L.; Li, F.; Pingguan-Murphy, B.; Lu, T.J.; Xu, F. Advances in paper-based point-of-care diagnostics. Biosens. Bioelectron. 2014, 54, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Yetisen, A.K.; Akram, M.S.; Lowe, C.R. Paper-based microfluidic point-of-care diagnostic devices. Lab Chip 2013, 13, 2210–2251. [Google Scholar] [CrossRef]
- Cai, L.; Huang, Y.; Sun, P.; Zheng, W.; Zhou, S.; Huang, P.; Liang, Z. Accurate detection of β-hCG in women’s serum and cervical secretions for predicting early pregnancy viability based on time-resolved luminescent lanthanide nanoprobes. Nanoscale 2020, 12, 6729–6735. [Google Scholar] [CrossRef]
- Chiu, N.F.; Kuo, C.T.; Chen, C.Y. High-affinity carboxyl-graphene oxide-based SPR aptasensor for the detection of hCG protein in clinical serum samples. Int. J. Nanomed. 2019, 14, 4833–4847. [Google Scholar] [CrossRef]
- Kuo, H.T.; Yeh, J.Z.; Wu, P.H.; Jiang, C.M.; Wu, M.C. Application of immunomagnetic particles to enzyme-linked immunosorbent assay (ELISA) for improvement of detection sensitivity of HCG. J. Immunoass. Immunochem. 2012, 33, 377–387. [Google Scholar] [CrossRef]
- Kuo, H.T.; Yeh, J.Z.; Jiang, C.M.; Wu, M.C. Magnetic particle-linked anti hCG β antibody for immunoassay of human chorionic gonadotropin (hCG), potential application to early pregnancy diagnosis. J. Immunol. Methods 2012, 381, 32–40. [Google Scholar] [CrossRef]
- Bolnick, J.M.; Kilburn, B.A.; Bajpayee, S.; Reddy, N.; Jeelani, R.; Crone, B.; Simmerman, N.; Singh, M.; Diamond, M.; Armant, D.R. Trophoblast retrieval and isolation from the cervix (TRIC) for noninvasive prenatal screening at 5 to 20 weeks of gestation. Fertil. Steril. 2014, 102, 135–142. [Google Scholar] [CrossRef]
- Fritz, R.; Kohan-Ghadr, H.R.; Sacher, A.; Bolnick, A.D.; Kilburn, B.A.; Bolnick, J.M.; Diamond, M.; Drewlo, S.; Armant, D.R. Trophoblast retrieval and isolation from the cervix (TRIC) is unaffected by early gestational age or maternal obesity. Prenat. Diagn. 2015, 35, 1218–1222. [Google Scholar] [CrossRef]
- Rahaie, M.; Ostad-Hasanzadeh, B.; Faridbod, F. A Novel Fluorescence Nanobiosensor based on Modified Graphene Quantum dots-HTAB for Early Detection of Fetal Sexuality with Cell Free Fetal DNA. J. Fluoresc. 2021, 31, 1843–1853. [Google Scholar] [CrossRef] [PubMed]
- Calcagno, M.; D’Agata, R.; Breveglieri, G.; Borgatti, M.; Bellassai, N.; Gambari, R.; Spoto, G. Nanoparticle-Enhanced Surface Plasmon Resonance Imaging Enables the Ultrasensitive Detection of Non-Amplified Cell-Free Fetal DNA for Non-Invasive Prenatal Testing. Anal. Chem. 2022, 94, 1118–1125. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Chen, X.; Wang, Q.; Xiao, M.; Zhong, D.; Sun, W.; Zhang, G.; Zhang, Z. Ultrasensitive Monolayer MoS2 Field-Effect Transistor Based DNA Sensors for Screening of Down Syndrome. Nano Lett. 2019, 19, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Zhao, S.; Wan, H.; Liu, Y.; Zhang, F.; Guo, X.; Zeng, W.; Zhang, H.; Zeng, L.; Qu, J.; et al. High-resolution DNA size enrichment using a magnetic nano-platform and application in non-invasive prenatal testing. Analyst 2020, 145, 5733–5739. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, S.M.; Wiepz, G.J.; Schotzko, M.; Simmons, H.A.; Mejia, A.; Ludwig, K.D.; Zhu, A.; Brunner, K.; Hernando, D.; Reeder, S.B.; et al. Impact of ferumoxytol magnetic resonance imaging on the rhesus macaque maternal-fetal interface. Biol. Reprod. 2020, 102, 434–444. [Google Scholar] [CrossRef]
- Gatta, G.; Di Grezia, G.; Cuccurullo, V.; Sardu, C.; Iovino, F.; Comune, R.; Ruggiero, A.; Chirico, M.; La Forgia, D.; Fanizzi, A.; et al. MRI in Pregnancy and Precision Medicine: A Review from Literature. J. Pers. Med. 2021, 12, 9. [Google Scholar] [CrossRef]
- Shetty, A.N.; Pautler, R.; Ghaghada, K.; Rendon, D.; Gao, H.; Starosolski, Z.; Bhavane, R.; Patel, C.; Annapragada, A.; Yallampalli, C.; et al. A liposomal Gd contrast agent does not cross the mouse placental barrier. Sci. Rep. 2016, 6, 27863. [Google Scholar] [CrossRef]
- Badachhape, A.A.; Kumar, A.; Ghaghada, K.B.; Stupin, I.V.; Srivastava, M.; Devkota, L.; Starosolski, Z.; Tanifum, E.A.; George, V.; Fox, K.A.; et al. Pre-clinical magnetic resonance imaging of retroplacental clear space throughout gestation. Placenta 2019, 77, 1–7. [Google Scholar] [CrossRef]
- Roberts, V.H.; Frias, A.E. Contrast-enhanced ultrasound for the assessment of placental development and function. Biotechniques 2020, 69, 392–399. [Google Scholar] [CrossRef]
- Wong, C.H.; Chen, C.P.; Chang, C.C.; Chen, C.Y. Bio-functionalized magnetic nanoparticles for the immunoassay of fetal fibronectin: A feasibility study for the prediction of preterm birth. Sci. Rep. 2017, 7, 42461. [Google Scholar] [CrossRef]
- Paul, J.W.; Smith, R. Preventing preterm birth: New approaches to labour therapeutics using Nanoparticles. Best Pract. Res. Clin. Obstet. Gynaecol. 2018, 52, 48–59. [Google Scholar] [CrossRef] [PubMed]
- Patki, M.; Giusto, K.; Gorasiya, S.; Reznik, S.E.; Patel, K. 17-α Hydroxyprogesterone Nanoemulsifying Preconcentrate-Loaded Vaginal Tablet: A Novel Non-Invasive Approach for the Prevention of Preterm Birth. Pharmaceutics 2019, 11, 335. [Google Scholar] [CrossRef] [PubMed]
- Attias Cohen, S.; Kingma, P.S.; Whitsett, J.A.; Goldbart, R.; Traitel, T.; Kost, J. SP-D loaded PLGA nanoparticles as drug delivery system for prevention and treatment of premature infant’s lung diseases. Int. J. Pharm. 2020, 585, 119387. [Google Scholar] [CrossRef] [PubMed]
- Buhimschi, C.S.; Bahtiyar, M.O.; Zhao, G.; Abdelghany, O.; Schneider, L.; Razeq, S.A.; Dulay, A.T.; Lipkind, H.S.; Mieth, S.; Rogers, L.; et al. Antenatal N-acetylcysteine to improve outcomes of premature infants with intra-amniotic infection and inflammation (Triple I): Randomized clinical trial. Pediatr. Res. 2021, 89, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Londero, A.P.; Orsaria, M.; Marzinotto, S.; Grassi, T.; Fruscalzo, A.; Calcagno, A.; Bertozzi, S.; Nardini, N.; Stella, E.; Lellé, R.J.; et al. Placental aging and oxidation damage in a tissue micro-array model: An immunohistochemistry study. Histochem. Cell Biol. 2016, 146, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Chiu, N.F.; Tai, M.J.; Wu, H.P.; Lin, T.L.; Chen, C.Y. Development of a bioaffinity SPR immunosensor based on functionalized graphene oxide for the detection of pregnancy-associated plasma protein A2 in human plasma. Int. J. Nanomed. 2019, 14, 6735–6748. [Google Scholar] [CrossRef]
- Chiu, N.F.; Tai, M.J.; Nurrohman, D.T.; Lin, T.L.; Wang, Y.H.; Chen, C.Y. Immunoassay-Amplified Responses Using a Functionalized MoS2-Based SPR Biosensor to Detect PAPP-A2 in Maternal Serum Samples to Screen for Fetal Down’s Syndrome. Int. J. Nanomed. 2021, 16, 2715–2733. [Google Scholar] [CrossRef]
- Pham, T.T.T.; Tran, D.P.; Nguyen, M.C.; Amen, M.T.; Winter, M.; Whitehead, C.; Toh, J.; Thierry, B. A simplified point-of-care testing approach for preeclampsia blood biomarkers based on nanoscale field effect transistors. Nanoscale. 2021, 13, 12279–12287. [Google Scholar] [CrossRef]
- Suman, P.; Gandhi, S.; Kumar, P.; Garg, K. Prospects of electrochemical immunosensors for early diagnosis of preeclampsia. Am. J. Reprod. Immunol. 2017, 77, e12584. [Google Scholar] [CrossRef]
- Sibley, C.P. Treating the dysfunctional placenta. J. Endocrinol. 2017, 234, R81–R97. [Google Scholar] [CrossRef]
- Alfaifi, A.A.; Heyder, R.S.; Bielski, E.R.; Almuqbil, R.M.; Kavdia, M.; Gerk, P.M.; da Rocha, S.R. Megalin-targeting liposomes for placental drug delivery. J. Control. Release 2020, 324, 366–378. [Google Scholar] [CrossRef] [PubMed]
- de Alwis, N.; Binder, N.K.; Beard, S.; Tu’uhevaha, J.; Tong, S.; Brownfoot, F.; Hannan, N.J. Novel approaches to combat preeclampsia: From new drugs to innovative delivery. Placenta 2020, 102, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Jena, M.K.; Sharma, N.R.; Petitt, M.; Maulik, D.; Nayak, N.R. Pathogenesis of Preeclampsia and Therapeutic Approaches Targeting the Placenta. Biomolecules 2020, 10, 953. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, H.; Xue, J.; Chen, P.; Zhou, Q.; Zhang, C. Nanoparticle-Mediated Simultaneous Downregulation of Placental Nrf2 and sFlt1 Improves Maternal and Fetal Outcomes in a Preeclampsia Mouse Model. ACS Biomater. Sci. Eng. 2020, 6, 5866–5873. [Google Scholar] [CrossRef]
- Pepe, G.J.; Albrecht, E.D. Novel Technologies for Target Delivery of Therapeutics to the Placenta during Pregnancy: A Review. Genes 2021, 12, 1255. [Google Scholar] [CrossRef]
- Renshall, L.J.; Beards, F.; Evangelinos, A.; Greenwood, S.L.; Brownbill, P.; Stevens, A.; Sibley, C.P.; Aplin, J.D.; Johnstone, E.D.; Teesalu, T.; et al. Targeted Delivery of Epidermal Growth Factor to the Human Placenta to Treat Fetal Growth Restriction. Pharmaceutics 2021, 13, 1778. [Google Scholar] [CrossRef]
- Yu, Q.; Qiu, Y.; Wang, X.; Tang, J.; Liu, Y.; Mei, L.; Li, M.; Yang, M.; Tang, L.; Gao, H.; et al. Efficient siRNA transfer to knockdown a placenta specific lncRNA using RGD-modified nano-liposome: A new preeclampsia-like mouse model. Int. J. Pharm. 2018, 546, 115–124. [Google Scholar] [CrossRef]
- Barani, M.; Sargazi, S.; Mohammadzadeh, V.; Rahdar, A.; Pandey, S.; Jha, N.K.; Gupta, P.K.; Thakur, V.K. Theranostic Advances of Bionanomaterials against Gestational Diabetes Mellitus: A Preliminary Review. J. Funct. Biomater. 2021, 12, 54. [Google Scholar] [CrossRef]
- Belsare, S.; Coté, G. Development of a colorimetric paper fluidic dipstick assay for measurement of glycated albumin to monitor gestational diabetes at the point-of-care. Talanta 2021, 223, 121728. [Google Scholar] [CrossRef]
- Cheng, X.; Xu, Y.; Jia, Q.; Guo, N.; Wang, Z.; Wang, Y. Novel greener approached synthesis of polyacrylic nanoparticles for therapy and care of gestational diabetes. Drug Deliv. 2020, 27, 1263–1270. [Google Scholar] [CrossRef]
- Du, S.; Lv, Y.; Li, N.; Huang, X.; Liu, X.; Li, H.; Wang, C.; Jia, Y.-F. Biological investigations on therapeutic effect of chitosan encapsulated nano resveratrol against gestational diabetes mellitus rats induced by streptozotocin. Drug Deliv. 2020, 27, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Vafaei-Pour, Z.; Shokrzadeh, M.; Jahani, M.; Shaki, F. Embryo-Protective Effects of Cerium Oxide Nanoparticles against Gestational Diabetes in Mice. Iran. J. Pharm. Res. 2018, 17, 964–975. [Google Scholar] [PubMed]
- Alhazza, I.M.; Ebaid, H.; Omar, M.S.; Hassan, I.; Habila, M.A.; Al-Tamimi, J.; Sheikh, M. Supplementation with selenium nanoparticles alleviates diabetic nephropathy during pregnancy in the diabetic female rats. Environ. Sci. Pollut. Res. Int. 2022, 29, 5517–5525. [Google Scholar] [CrossRef] [PubMed]
- Aslesh, T.; Yokota, T. Restoring SMN Expression: An Overview of the Therapeutic Developments for the Treatment of Spinal Muscular Atrophy. Cells 2022, 11, 417. [Google Scholar] [CrossRef] [PubMed]
- Francia, V.; Schiffelers, R.M.; Cullis, P.R.; Witzigmann, D. The Biomolecular Corona of Lipid Nanoparticles for Gene Therapy. Bioconjug. Chem. 2020, 31, 2046–2059. [Google Scholar] [CrossRef]
- Liang, X.; Liu, L.; Wei, Y.Q.; Gao, G.P.; Wei, X.W. Clinical Evaluations of Toxicity and Efficacy of Nanoparticle-Mediated Gene Therapy. Hum. Gene Ther. 2018, 29, 1227–1234. [Google Scholar] [CrossRef]
- Ochiya, T.; Takahama, Y.; Baba-Toriyama, H.; Tsukamoto, M.; Yasuda, Y.; Kikuchi, H.; Terada, M. Evaluation of cationic liposome suitable for gene transfer into pregnant animals. Biochem. Biophys. Res. Commun. 1999, 258, 358–365. [Google Scholar] [CrossRef]
- Valero, L.; Alhareth, K.; Espinoza Romero, J.; Viricel, W.; Leblond, J.; Chissey, A.; Dhotel, H.; Roques, C.; Campiol Arruda, D.; Escriou, V.; et al. Liposomes as Gene Delivery Vectors for Human Placental Cells. Molecules 2018, 23, 1085. [Google Scholar] [CrossRef]
- Schwab, M.E.; MacKenzie, T.C. Prenatal Gene Therapy. Clin. Obstet. Gynecol. 2021, 64, 876–885. [Google Scholar] [CrossRef]
- Hacein-Bey-Abina, S.; Von Kalle, C.; Schmidt, M.; Le Deist, F.; Wulffraat, N.; McIntyre, E.; Radford, I.; Villeval, J.-L.; Fraser, C.C.; Cavazzana-Calvo, M.; et al. A serious adverse event after successful gene therapy for X-linked severe combined immunodeficiency. N. Engl. J. Med. 2003, 348, 255–256. [Google Scholar] [CrossRef]
- Grant, J.; Hoorens, S.; Sivadasan, S.; Loo, M.V.H.; DaVanzo, J.; Hale, L.; Butz, W. Trends in European fertility: Should Europe try to increase its fertility rate...or just manage the consequences? Int. J. Androl. 2006, 29, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Remião, M.H.; Segatto, N.V.; Pohlmann, A.; Guterres, S.S.; Seixas, F.K.; Collares, T. The Potential of Nanotechnology in Medically Assisted Reproduction. Front. Pharmacol. 2017, 8, 994. [Google Scholar] [CrossRef] [PubMed]
- Abreu, C.M.; Thomas, V.; Knaggs, P.; Bunkheila, A.; Cruz, A.; Teixeira, S.R.; Alpuim, P.; Francis, L.W.; Gebril, A.; Ibrahim, A.; et al. Non-invasive molecular assessment of human embryo development and implantation potential. Biosens. Bioelectron. 2020, 157, 112144. [Google Scholar] [CrossRef] [PubMed]
- Calsteren, K.V.; Heyns, L.; Smet, F.D.; Eycken, L.V.; Gziri, M.M.; Gemert, W.V.; Halaska, M.; Vergote, I.; Ottevanger, N.; Amant, F. Cancer during pregnancy: An analysis of 215 patients emphasizing the obstetrical and the neonatal outcomes. J. Clin. Oncol. 2010, 28, 683–689. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, R.; Joshi, N.; Khan, M.A.; Siddhara, S. Nanosomal docetaxel lipid suspension based chemotherapy in a pregnant MBC patient—A case report. Onco Targets Ther. 2019, 12, 5679–5685. [Google Scholar] [CrossRef]
- Marmé, F.; Schneeweiss, A. Personalized therapy in breast cancer. Onkologie 2012, 35 (Suppl 1), 28–33. [Google Scholar] [CrossRef]
- Ali, S.; Albekairi, N.A.; Al-Enazy, S.; Shah, M.; Patrikeeva, S.; Nanovskaya, T.N.; Ahmed, M.S.; Rytting, E. Formulation effects on paclitaxel transfer and uptake in the human placenta. Nanomedicine 2021, 33, 102354. [Google Scholar] [CrossRef]
- Scialli, A.R.; Waterhouse, T.B.; Desesso, J.M.; Rahman, A.; Goeringer, G.C. Protective effect of liposome encapsulation on paclitaxel developmental toxicity in the rat. Teratology 1997, 56, 305–310. [Google Scholar] [CrossRef]
- Green, A.E.; Rose, P.G. Pegylated liposomal doxorubicin in ovarian cancer. Int. J. Nanomed. 2006, 1, 229–239. [Google Scholar]
- Khodaverdi, S.; Jafari, A.; Movahedzadeh, F.; Madani, F.; Yousefi Avarvand, A.; Falahatkar, S. Evaluating Inhibitory Effects of Paclitaxel and Vitamin D3 Loaded Poly Lactic Glycolic Acid Co-Delivery Nanoparticles on the Breast Cancer Cell Line. Adv. Pharm. Bull. 2020, 10, 30–38. [Google Scholar] [CrossRef]
- Refuerzo, J.S.; Alexander, J.F.; Leonard, F.; Leon, M.; Longo, M.; Godin, B. Liposomes: A nanoscale drug carrying system to prevent indomethacin passage to the fetus in a pregnant mouse model. Am. J. Obstet. Gynecol. 2015, 212, 508. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Zhou, Y.; Liu, L.; Xu, Y.; Chen, Q.; Wang, Y.; Wu, S.; Deng, Y.; Zhang, J.; Shao, A. Nanoparticle-Based Drug Delivery in Cancer Therapy and Its Role in Overcoming Drug Resistance. Front. Mol. Biosci. 2020, 7, 193. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.L.B.; Czerniecki, B.J. Clinical development of immunotherapies for HER2+ breast cancer: A review of HER2-directed monoclonal antibodies and beyond. NPJ Breast Cancer 2020, 6, 10. [Google Scholar] [CrossRef] [PubMed]
- Tamang, S.T.; Dorji, T.; Yoezer, S.; Phuntsho, T.; Dorji, P. Knowledge and understanding of obstetric danger signs among pregnant women attending the antenatal clinic at the National Referral Hospital in Thimphu, Bhutan: A cross-sectional study. BMC Pregnancy Childbirth 2021, 21, 104. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, P.A. Gestational nanomaterial exposures: Microvascular implications during pregnancy, fetal development and adulthood. J. Physiol. 2016, 594, 2161–2173. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).