Evaluating Translational Methods for Personalized Medicine—A Scoping Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Questions
- Which preclinical models are currently used to provide validity data prior to therapeutic clinical trials of PM in oncology and brain disorders and what are the pros and cons of the applied methods?
- Are the current preclinical models predictive for the outcome of PM trials?
2.2. Study Identification
2.3. Study Selection and Eligibility Criteria
2.4. Charting the Data
2.5. Consultation Exercise
2.6. Patient and Public Involvement
3. Results
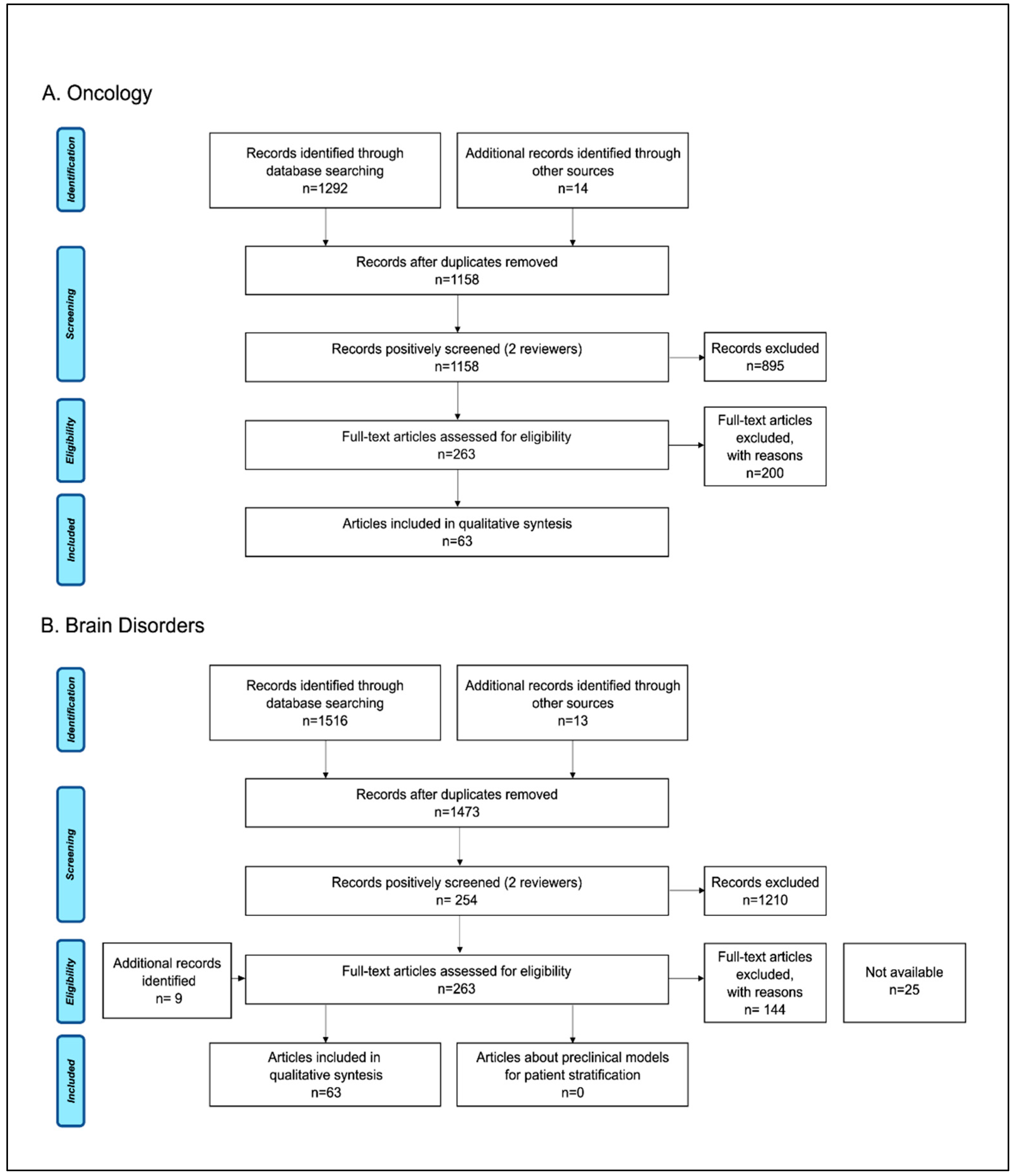
3.1. Study Selection and General Characteristics of Reports
3.2. In Vivo Models for PM
3.3. In Vitro Models for PM
3.4. In Silico Models
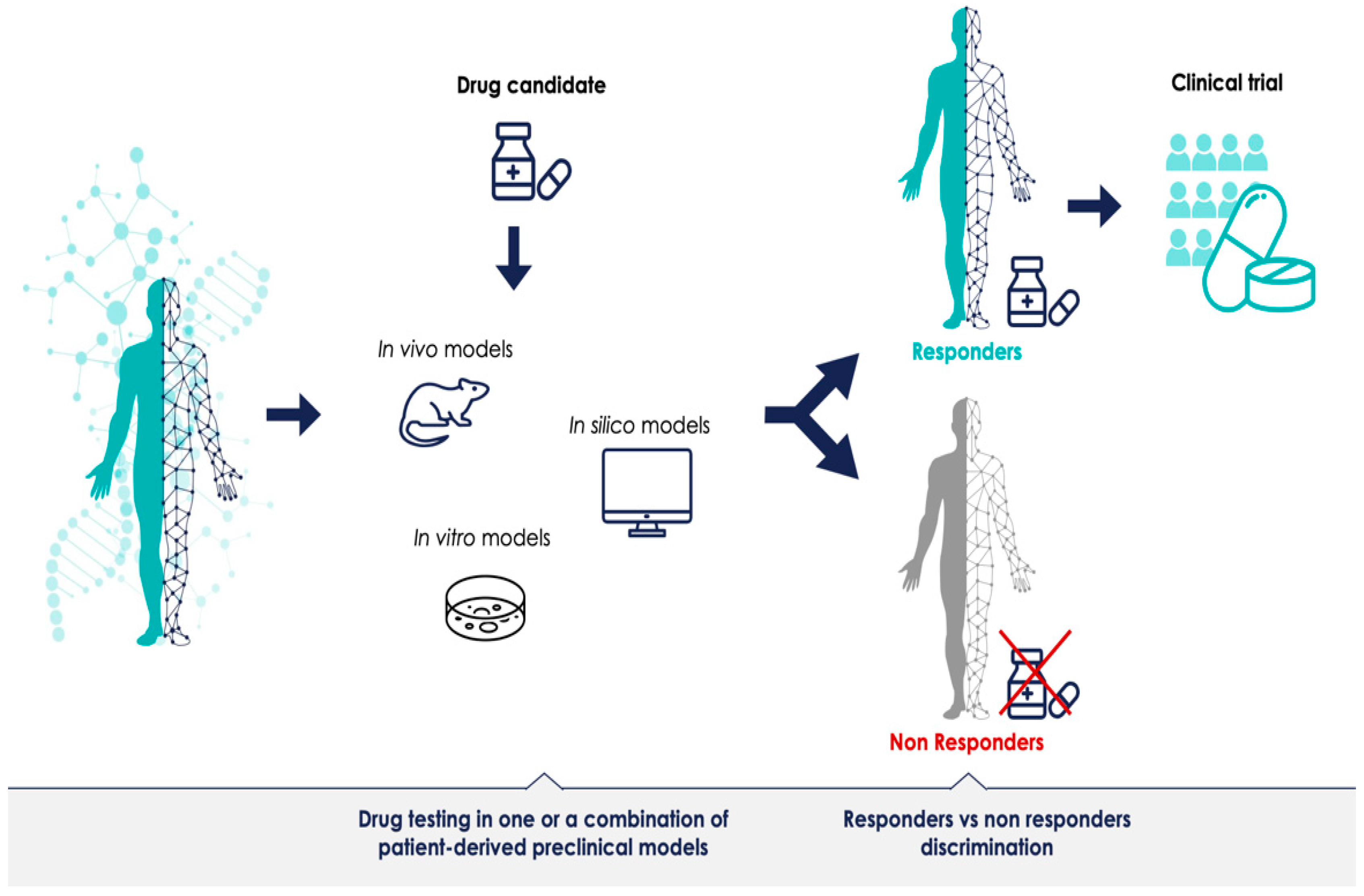
3.5. Are the Current Preclinical Models Predictive for PM Trials?
3.6. Main Gaps Identified
- The first gap is a lack of clinically relevant experimental models for personalized medicine. Despite technical advances and more sophisticated preclinical models, to date, there are knowledge gaps in biology and an inability to recapitulate human phenotypes for many diseases, which is a challenge for translation and prediction of preclinical data to human PM clinical trials. There is also an apparent deficit in validating preclinical methods for clinical relevance; in other words, how well the model represents the phenotype of disease or clustering of patients.
- The second gap is the lack of standards for methods, validation procedures, and the lack of quality assessment systems. The fact is that preclinical models are often not robust enough for translation. Some of the hurdles for model validation are that this type of work is not academically rewarded, it is time consuming, and it is expensive.
- The third gap is the lack of accurate reporting and the lack of reporting negative results, which then further leads to a lack of systematic reviews and meta-analyses on methods, and these are important tools for evidence-based medicine. Access to preclinical data supporting clinical trials is challenging. There is a publication bias toward positive experiments, and methods are often not reported in sufficient detail to attempt reproducibility of results.
- The fourth gap relates to regulation, and the lack of harmonized guidelines for evaluating the relevance and robustness of preclinical evidence.
- The last gap we identified is the lack of involvement between preclinical and clinical research, and the need for a better definition for patient engagement.
4. Discussion
4.1. Principal Findings
4.2. Limitations of the Scope
4.3. Challenges of Preclinical Research in PM
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- EU. 2015/C 421/03 Council conclusions on personalised medicine for patients. Off. J. Eur. Union 2015, 421, 2–5. [Google Scholar]
- European Commission. 2019 Report on the Statistics on the Use of Animals for Scientific Purposes in the Member States of the European Union in 2015–2017; CELEX: Philadelphia, PA, USA, 2020. [Google Scholar]
- Nestler, E.J.; Hyman, S.E. Animal models of neuropsychiatric disorders. Nat. Neurosci 2010, 13, 1161–1169. [Google Scholar] [CrossRef] [PubMed]
- Day, C.P.; Merlino, G.; Van Dyke, T. Preclinical mouse cancer models: A maze of opportunities and challenges. Cell 2015, 163, 39–53. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, D.; De Deyn, P.P. Animal models in the drug discovery pipeline for Alzheimer’s disease. Br. J. Pharmacol. 2011, 164, 1285–1300. [Google Scholar] [CrossRef] [PubMed]
- Crawley, J.N. Translational animal models of autism and neurodevelopmental disorders. Dialogues Clin. Neurosci. 2012, 14, 293–305. [Google Scholar] [CrossRef]
- Mak, I.W.; Evaniew, N.; Ghert, M. Lost in translation: Animal models and clinical trials in cancer treatment. Am. J. Transl. Res. 2014, 6, 114–118. [Google Scholar]
- Aartsma-Rus, A.; van Putten, M. The use of genetically humanized animal models for personalized medicine approaches. Dis. Model. Mech. 2019, 13, dmm041673. [Google Scholar] [CrossRef]
- Li, H.; Auwerx, J. Mouse Systems Genetics as a Prelude to Precision Medicine. Trends Genet. 2020, 36, 259–272. [Google Scholar] [CrossRef]
- Bhimani, J.; Ball, K.; Stebbing, J. Patient-derived xenograft models-the future of personalised cancer treatment. Br. J. Cancer 2020, 122, 601–602. [Google Scholar] [CrossRef]
- Byrne, A.T.; Alferez, D.G.; Amant, F.; Annibali, D.; Arribas, J.; Biankin, A.V.; Bruna, A.; Budinska, E.; Caldas, C.; Chang, D.K.; et al. Interrogating open issues in cancer precision medicine with patient-derived xenografts. Nat. Rev. Cancer 2017, 17, 254–268. [Google Scholar] [CrossRef]
- Clohessy, J.G.; Pandolfi, P.P. Mouse hospital and co-clinical trial project--from bench to bedside. Nat. Rev. Clin. Oncol. 2015, 12, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Cavero, I.; Guillon, J.M.; Holzgrefe, H.H. Human organotypic bioconstructs from organ-on-chip devices for human-predictive biological insights on drug candidates. Expert Opin. Drug. Saf. 2019, 18, 651–677. [Google Scholar] [CrossRef] [PubMed]
- Fong, E.L.S.; Toh, T.B.; Yu, H.; Chow, E.K.-H. 3D Culture as a Clinically Relevant Model for Personalized Medicine. SLAS Technol. Transl. Life Sci. Innov. 2017, 22, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.R.; Corrochano, S.; Gasco, S.; Tibbit, C.; Thompson, D.; Maduro, C.; Ali, Z.; Fratta, P.; Arozena, A.A.; Cunningham, T.J.; et al. Uses for humanised mouse models in precision medicine for neurodegenerative disease. Mamm. Genome 2019, 30, 173–191. [Google Scholar] [CrossRef] [PubMed]
- Haggarty, S.J.; Silva, M.C.; Cross, A.; Brandon, N.J.; Perlis, R.H. Advancing drug discovery for neuropsychiatric disorders using patient-specific stem cell models. Mol. Cell Neurosci. 2016, 73, 104–115. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Song, L.; Bejoy, J.; Zhao, J.; Kanekiyo, T.; Bu, G.; Zhou, Y.; Li, Y. Modeling Neurodegenerative Microenvironment Using Cortical Organoids Derived from Human Stem Cells. Tissue Eng. Part A 2018, 24, 1125–1137. [Google Scholar] [CrossRef] [PubMed]
- McCammon, J.M.; Sive, H. Addressing the Genetics of Human Mental Health Disorders in Model Organisms. Annu. Rev. Genom. Hum. Genet. 2015, 16, 173–197. [Google Scholar] [CrossRef]
- Driehuis, E.; Kretzschmar, K.; Clevers, H. Establishment of patient-derived cancer organoids for drug-screening applications. Nat. Protoc. 2020, 15, 3380–3409. [Google Scholar] [CrossRef]
- Chakravarty, K.; Antontsev, V.; Bundey, Y.; Varshney, J. Driving success in personalized medicine through AI-enabled computational modeling. Drug. Discov. Today 2021, 26, 1459–1465. [Google Scholar] [CrossRef]
- Chase, J.G.; Preiser, J.C.; Dickson, J.L.; Pironet, A.; Chiew, Y.S.; Pretty, C.G.; Shaw, G.M.; Benyo, B.; Moeller, K.; Safaei, S.; et al. Next-generation, personalised, model-based critical care medicine: A state-of-the art review of in silico virtual patient models, methods, and cohorts, and how to validation them. Biomed. Eng. Online 2018, 17, 24. [Google Scholar] [CrossRef]
- Torres Moral, T.; Sanchez-Niubo, A.; Monistrol-Mula, A.; Gerardi, C.; Banzi, R.; Garcia, P.; Demotes-Mainard, J.; Haro, J.M.; the PERMIT Group. Methods for Stratification and Validation Cohorts: A Scoping Review. J. Pers. Med. 2022, 12, 688. [Google Scholar] [CrossRef] [PubMed]
- Glaab, E.; Rauschenberger, A.; Banzi, R.; Gerardi, C.; Garcia, P.; Demotes, J. Biomarker discovery studies for patient stratification using machine learning analysis of omics data: A scoping review. BMJ Open 2021, 11, e053674. [Google Scholar] [CrossRef]
- Superchi, C.; Brion Bouvier, F.; Gerardi, C.; Carmona, M.; San Miguel, L.; Sánchez-Gómez, L.M.; Imaz-Iglesia, I.; Garcia, P.; Demotes, J.; Banzi, R.; et al. Study designs for clinical trials applied to personalised medicine: A scoping review. BMJ Open 2022, 12, e052926. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.; Colquhoun, H.; Kastner, M.; Levac, D.; Ng, C.; Sharpe, J.P.; Wilson, K.; et al. A scoping review on the conduct and reporting of scoping reviews. BMC Med. Res. Methodol. 2016, 16, 15. [Google Scholar] [CrossRef]
- Peters, M.D.; Godfrey, C.M.; Khalil, H.; McInerney, P.; Parker, D.; Soares, C.B. Guidance for conducting systematic scoping reviews. Int J. Evid. Based Healthc. 2015, 13, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Munn, Z.; Peters, M.D.J.; Stern, C.; Tufanaru, C.; McArthur, A.; Aromataris, E. Systematic review or scoping review? Guidance for authors when choosing between a systematic or scoping review approach. BMC Med. Res. Methodol. 2018, 18, 143. [Google Scholar] [CrossRef]
- Banzi, R.; Gerardi, C.; Fratelli, M.; Garcia, P.; Torres, T.; Abad, J.M.H.; Niubo, A.S.; Glaab, E.; Oldoni, E.; Bietrix, F.; et al. Methodological approaches for personalised medicine: Protocol for a series of scoping reviews. Zenodo 2020. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Paez, A. Gray literature: An important resource in systematic reviews. J. Evid. Based Med. 2017, 10, 233–240. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—a web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef] [PubMed]
- Leenaars, C.H.C.; Kouwenaar, C.; Stafleu, F.R.; Bleich, A.; Ritskes-Hoitinga, M.; De Vries, R.B.M.; Meijboom, F.L.B. Animal to human translation: A systematic scoping review of reported concordance rates. J. Transl. Med. 2019, 17, 223. [Google Scholar] [CrossRef]
- Malaney, P.; Nicosia, S.V.; Davé, V. One mouse, one patient paradigm: New avatars of personalized cancer therapy. Cancer Lett. 2014, 344, 1–12. [Google Scholar] [CrossRef]
- Shi, J.; Li, Y.; Jia, R.; Fan, X. The fidelity of cancer cells in PDX models: Characteristics, mechanism and clinical significance. Int. J. Cancer 2020, 146, 2078–2088. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.T.; Lang, S.H. A systematic review of the validity of patient derived xenograft (PDX) models: The implications for translational research and personalised medicine. PeerJ 2018, 6, e5981. [Google Scholar] [CrossRef]
- Tentler, J.J.; Tan, A.C.; Weekes, C.D.; Jimeno, A.; Leong, S.; Pitts, T.M.; Arcaroli, J.J.; Messersmith, W.A.; Eckhardt, S.G. Patient-derived tumour xenografts as models for oncology drug development. Nat. Rev. Clin. Oncol. 2012, 9, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Saito, R.; Kobayashi, T.; Kashima, S.; Matsumoto, K.; Ogawa, O. Faithful preclinical mouse models for better translation to bedside in the field of immuno-oncology. Int. J. Clin. Oncol. 2020, 25, 831–841. [Google Scholar] [CrossRef]
- Kucherlapati, R. Genetically modified mouse models for biomarker discovery and preclinical drug testing. Clin. Cancer Res. 2012, 18, 625–630. [Google Scholar] [CrossRef][Green Version]
- Just, M.K.; Gram, H.; Theologidis, V.; Jensen, P.H.; Nilsson, K.P.R.; Lindgren, M.; Knudsen, K.; Borghammer, P.; Van Den Berge, N. Alpha-Synuclein Strain Variability in Body-First and Brain-First Synucleinopathies. Front. Aging Neurosci 2022, 14, 907293. [Google Scholar] [CrossRef]
- Van Den Berge, N.; Ulusoy, A. Animal models of brain-first and body-first Parkinson’s disease. Neurobiol. Dis. 2022, 163, 105599. [Google Scholar] [CrossRef]
- Yang, C.; Xia, B.R.; Jin, W.L.; Lou, G. Circulating tumor cells in precision oncology: Clinical applications in liquid biopsy and 3D organoid model. Cancer Cell Int. 2019, 19, 341. [Google Scholar] [CrossRef] [PubMed]
- Powley, I.R.; Patel, M.; Miles, G.; Pringle, H.; Howells, L.; Thomas, A.; Kettleborough, C.; Bryans, J.; Hammonds, T.; MacFarlane, M.; et al. Patient-derived explants (PDEs) as a powerful preclinical platform for anti-cancer drug and biomarker discovery. Br. J. Cancer 2020, 122, 735–744. [Google Scholar] [CrossRef] [PubMed]
- Braunschweig, T.; Chung, J.Y.; Hewitt, S.M. Tissue microarrays: Bridging the gap between research and the clinic. Expert Rev. Proteom. 2005, 2, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Wen, Z.; Christian, K.M.; Song, H.; Ming, G.L. Modeling psychiatric disorders with patient-derived iPSCs. Curr. Opin. Neurobiol. 2016, 36, 118–127. [Google Scholar] [CrossRef]
- Nam, H.Y.; Shim, S.M.; Han, B.G.; Jeon, J.P. Human lymphoblastoid cell lines: A goldmine for the biobankomics era. Pharmacogenomics 2011, 12, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Penney, J.; Ralvenius, W.T.; Tsai, L.H. Modeling Alzheimer’s disease with iPSC-derived brain cells. Mol. Psychiatry 2020, 25, 148–167. [Google Scholar] [CrossRef]
- Silva, M.C.; Haggarty, S.J. Human pluripotent stem cell-derived models and drug screening in CNS precision medicine. Ann. N. Y. Acad. Sci. 2020, 1471, 18–56. [Google Scholar] [CrossRef]
- Aboulkheyr Es, H.; Montazeri, L.; Aref, A.R.; Vosough, M.; Baharvand, H. Personalized Cancer Medicine: An Organoid Approach. Trends Biotechnol. 2018, 36, 358–371. [Google Scholar] [CrossRef]
- Bartfeld, S.; Clevers, H. Stem cell-derived organoids and their application for medical research and patient treatment. J. Mol. Med. 2017, 95, 729–738. [Google Scholar] [CrossRef]
- Phan, N.; Hong, J.J.; Tofig, B.; Mapua, M.; Elashoff, D.; Moatamed, N.A.; Huang, J.; Memarzadeh, S.; Damoiseaux, R.; Soragni, A. A simple high-throughput approach identifies actionable drug sensitivities in patient-derived tumor organoids. Commun. Biol. 2019, 2, 78. [Google Scholar] [CrossRef]
- Granat, L.M.; Kambhampati, O.; Klosek, S.; Niedzwecki, B.; Parsa, K.; Zhang, D. The promises and challenges of patient-derived tumor organoids in drug development and precision oncology. Animal. Model. Exp. Med. 2019, 2, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Kondo, J.; Inoue, M. Application of Cancer Organoid Model for Drug Screening and Personalized Therapy. Cells 2019, 8, 470. [Google Scholar] [CrossRef] [PubMed]
- Weeber, F.; Ooft, S.N.; Dijkstra, K.K.; Voest, E.E. Tumor Organoids as a Pre-clinical Cancer Model for Drug Discovery. Cell Chem. Biol. 2017, 24, 1092–1100. [Google Scholar] [CrossRef] [PubMed]
- Sidhaye, J.; Knoblich, J.A. Brain organoids: An ensemble of bioassays to investigate human neurodevelopment and disease. Cell Death Differ. 2021, 28, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.O.; Price, D.J. Building brains in a dish: Prospects for growing cerebral organoids from stem cells. Neuroscience 2016, 334, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Meng, X.; Liu, Y.; Song, D.; Jiang, C.; Cai, J. Applications of brain organoids in neurodevelopment and neurological diseases. J. Biomed. Sci. 2021, 28, 30. [Google Scholar] [CrossRef]
- Rossetti, A.C.; Koch, P.; Ladewig, J. Drug discovery in psychopharmacology: From 2D models to cerebral organoids. Dialogues Clin. Neurosci 2019, 21, 203–224. [Google Scholar] [CrossRef]
- Sontheimer-Phelps, A.; Hassell, B.A.; Ingber, D.E. Modelling cancer in microfluidic human organs-on-chips. Nat. Rev. Cancer 2019, 19, 65–81. [Google Scholar] [CrossRef]
- Kankala, R.K.; Wang, S.B.; Chen, A.Z. Microengineered Organ-on-a-chip Platforms towards Personalized Medicine. Curr. Pharm. Des. 2018, 24, 5354–5366. [Google Scholar] [CrossRef]
- Skardal, A.; Shupe, T.; Atala, A. Organoid-on-a-chip and body-on-a-chip systems for drug screening and disease modeling. Drug Discov. Today 2016, 21, 1399–1411. [Google Scholar] [CrossRef]
- Shang, M.; Soon, R.H.; Lim, C.T.; Khoo, B.L.; Han, J. Microfluidic modelling of the tumor microenvironment for anti-cancer drug development. Lab. Chip. 2019, 19, 369–386. [Google Scholar] [CrossRef] [PubMed]
- Haring, A.P.; Sontheimer, H.; Johnson, B.N. Microphysiological Human Brain and Neural Systems-on-a-Chip: Potential Alternatives to Small Animal Models and Emerging Platforms for Drug Discovery and Personalized Medicine. Stem. Cell Rev. Rep. 2017, 13, 381–406. [Google Scholar] [CrossRef] [PubMed]
- Marx, U.; Akabane, T.; Andersson, T.B.; Baker, E.; Beilmann, M.; Beken, S.; Brendler-Schwaab, S.; Cirit, M.; David, R.; Dehne, E.M.; et al. Biology-inspired microphysiological systems to advance patient benefit and animal welfare in drug development. Altex 2020, 37, 365–394. [Google Scholar] [CrossRef] [PubMed]
- Kamel Boulos, M.N.; Zhang, P. Digital Twins: From Personalised Medicine to Precision Public Health. J. Pers. Med. 2021, 11, 745. [Google Scholar] [CrossRef]
- Klinghammer, K.; Walther, W.; Hoffmann, J. Choosing wisely-Preclinical test models in the era of precision medicine. Cancer Treat. Rev. 2017, 55, 36–45. [Google Scholar] [CrossRef]
- Gould, S.E.; Junttila, M.R.; de Sauvage, F.J. Translational value of mouse models in oncology drug development. Nat. Med. 2015, 21, 431–439. [Google Scholar] [CrossRef]
- Lieu, C.H.; Tan, A.C.; Leong, S.; Diamond, J.R.; Eckhardt, S.G. From bench to bedside: Lessons learned in translating preclinical studies in cancer drug development. J. Natl. Cancer Inst. 2013, 105, 1441–1456. [Google Scholar] [CrossRef]
- Liu, Z.; Delavan, B.; Roberts, R.; Tong, W. Lessons Learned from Two Decades of Anticancer Drugs. Trends Pharmacol. Sci. 2017, 38, 852–872. [Google Scholar] [CrossRef]
- Seyhan, A.A. Lost in translation: The valley of death across preclinical and clinical divide–identification of problems and overcoming obstacles. Transl. Med. Commun. 2019, 4, 18. [Google Scholar] [CrossRef]
- Godlee, F. We need better animal research, better reported. BMJ 2018, 360, k124. [Google Scholar] [CrossRef]
- Begley, C.G.; Ioannidis, J.P. Reproducibility in science: Improving the standard for basic and preclinical research. Circ. Res. 2015, 116, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Collins, F.S.; Tabak, L.A. Policy: NIH plans to enhance reproducibility. Nature 2014, 505, 612–613. [Google Scholar] [CrossRef] [PubMed]
- Perrin, S. Preclinical research: Make mouse studies work. Nature 2014, 507, 423–425. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, J.P. Acknowledging and Overcoming Nonreproducibility in Basic and Preclinical Research. JAMA 2017, 317, 1019–1020. [Google Scholar] [CrossRef]
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. PLoS Biol. 2010, 8, e1000412. [Google Scholar] [CrossRef]
- Du Sert, N.P.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; Emerson, M.; et al. Reporting animal research: Explanation and elaboration for the ARRIVE guidelines 2.0. PLoS Biol. 2020, 18, e3000411. [Google Scholar] [CrossRef]
- Brown, K.M.; Xue, A.; Mittal, A.; Samra, J.S.; Smith, R.; Hugh, T.J. Patient-derived xenograft models of colorectal cancer in pre-clinical research: A systematic review. Oncotarget 2016, 7, 66212–66225. [Google Scholar] [CrossRef]
- Koga, Y.; Ochiai, A. Systematic Review of Patient-Derived Xenograft Models for Preclinical Studies of Anti-Cancer Drugs in Solid Tumors. Cells 2019, 8, 418. [Google Scholar] [CrossRef]
- 46th ESAO Congress 3–7 September 2019 Hannover, Germany Abstracts. Int. J. Artif. Organs 2019, 42, 386–474. [CrossRef]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef]
- Landis, S.C.; Amara, S.G.; Asadullah, K.; Austin, C.P.; Blumenstein, R.; Bradley, E.W.; Crystal, R.G.; Darnell, R.B.; Ferrante, R.J.; Fillit, H.; et al. A call for transparent reporting to optimize the predictive value of preclinical research. Nature 2012, 490, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Olson, C.M.; Rennie, D.; Cook, D.; Dickersin, K.; Flanagin, A.; Hogan, J.W.; Zhu, Q.; Reiling, J.; Pace, B. Publication Bias in Editorial Decision Making. JAMA 2002, 287, 2825–2828. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.P.; Boyd, E.A.; Holroyd-Leduc, J.M.; Bacchetti, P.; Bero, L.A. Predictors of publication: Characteristics of submitted manuscripts associated with acceptance at major biomedical journals. Med. J. Aust. 2006, 184, 621–626. [Google Scholar] [CrossRef]
- Okike, K.; Kocher, M.S.; Mehlman, C.T.; Heckman, J.D.; Bhandari, M. Publication bias in orthopaedic research: An analysis of scientific factors associated with publication in the Journal of Bone and Joint Surgery (American Volume). J. Bone Joint Surg Am. 2008, 90, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Pratte, M.; Ganeshamoorthy, S.; Carlisle, B.; Kimmelman, J. How well are Phase 2 cancer trial publications supported by preclinical efficacy evidence? Int. J. Cancer 2019, 145, 3370–3375. [Google Scholar] [CrossRef]
- Federico, C.A.; Carlisle, B.; Kimmelman, J.; Fergusson, D.A. Late, never or non-existent: The inaccessibility of preclinical evidence for new drugs. Br. J. Pharmacol. 2014, 171, 4247–4254. [Google Scholar] [CrossRef]
- Gurusamy, K.S.; Moher, D.; Loizidou, M.; Ahmed, I.; Avey, M.T.; Barron, C.C.; Davidson, B.; Dwek, M.; Gluud, C.; Jell, G.; et al. Clinical relevance assessment of animal preclinical research (RAA) tool: Development and explanation. PeerJ 2021, 9, e10673. [Google Scholar] [CrossRef]
- Ferreira, G.S.; Veening-Griffioen, D.H.; Boon, W.P.C.; Moors, E.H.M.; van Meer, P.J.K. Levelling the Translational Gap for Animal to Human Efficacy Data. Animals 2020, 10, 1199. [Google Scholar] [CrossRef]
- Bahr-Hosseini, M.; Bikson, M.; Iacoboni, M.; Liebeskind, D.S.; Hinman, J.D.; Carmichael, S.T.; Saver, J.L. PRIMED2 Preclinical Evidence Scoring Tool to Assess Readiness for Translation of Neuroprotection Therapies. Transl. Stroke Res. 2022, 13, 222–227. [Google Scholar] [CrossRef]
- Schäfer-Korting, M.; Zoschke, C. How Qualification of 3D Disease Models Cuts the Gordian Knot in Preclinical Drug Development. Handb. Exp. Pharmacol. 2021, 265, 29–56. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine; Division on Earth and Life Studies; Institute for Laboratory Animal Research; Roundtable on Science and Welfare in Laboratory Animal Use. The National Academies Collection: Reports funded by National Institutes of Health. In Advancing Disease Modeling in Animal-Based Research in Support of Precision Medicine: Proceedings of a Workshop; National Academies Press: Washington, DC, USA, 2018. [Google Scholar] [CrossRef]
- Folke, J.; Ferreira, N.; Brudek, T.; Borghammer, P.; Van Den Berge, N. Passive Immunization in Alpha-Synuclein Preclinical Animal Models. Biomolecules 2022, 12, 168. [Google Scholar] [CrossRef] [PubMed]
| Use Case | Advantages | Disadvantages |
|---|---|---|
| Oncology | PDX 1
| PDX 1
|
GEMM 2
| GEMM 2
| |
| Brain disorders |
|
|
| Use Case | Advantages | Disadvantages |
|---|---|---|
| Oncology | 2D monolayer cell culture
| 2D monolayer cell culture
|
3D tumor cultures
| 3D tumor cultures
| |
Organoids
| Organoids
| |
Organ-on-chips
| Organ-on-chips
| |
| Brain disorders | LCL 1
| LCL 1
|
iPSC 2
| iPSC 2
| |
Organoids
| Organoids
|
| Use Case | Advantages | Disadvantages |
|---|---|---|
| Oncology |
|
|
| Brain disorders |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fosse, V.; Oldoni, E.; Gerardi, C.; Banzi, R.; Fratelli, M.; Bietrix, F.; Ussi, A.; Andreu, A.L.; McCormack, E.; the PERMIT Group. Evaluating Translational Methods for Personalized Medicine—A Scoping Review. J. Pers. Med. 2022, 12, 1177. https://doi.org/10.3390/jpm12071177
Fosse V, Oldoni E, Gerardi C, Banzi R, Fratelli M, Bietrix F, Ussi A, Andreu AL, McCormack E, the PERMIT Group. Evaluating Translational Methods for Personalized Medicine—A Scoping Review. Journal of Personalized Medicine. 2022; 12(7):1177. https://doi.org/10.3390/jpm12071177
Chicago/Turabian StyleFosse, Vibeke, Emanuela Oldoni, Chiara Gerardi, Rita Banzi, Maddalena Fratelli, Florence Bietrix, Anton Ussi, Antonio L. Andreu, Emmet McCormack, and the PERMIT Group. 2022. "Evaluating Translational Methods for Personalized Medicine—A Scoping Review" Journal of Personalized Medicine 12, no. 7: 1177. https://doi.org/10.3390/jpm12071177
APA StyleFosse, V., Oldoni, E., Gerardi, C., Banzi, R., Fratelli, M., Bietrix, F., Ussi, A., Andreu, A. L., McCormack, E., & the PERMIT Group. (2022). Evaluating Translational Methods for Personalized Medicine—A Scoping Review. Journal of Personalized Medicine, 12(7), 1177. https://doi.org/10.3390/jpm12071177






