Interferon-Inducible Protein-10 as a Marker to Detect Latent Tuberculosis Infection in Patients with Inflammatory Rheumatic Diseases
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Performing and Assessing TST
2.3. Whole Blood Stimulation
2.4. IFN-Gamma Level Quantification
2.5. IP-10 Level Quantification
2.6. Statistical Analysis
3. Results
3.1. Level of IP-10
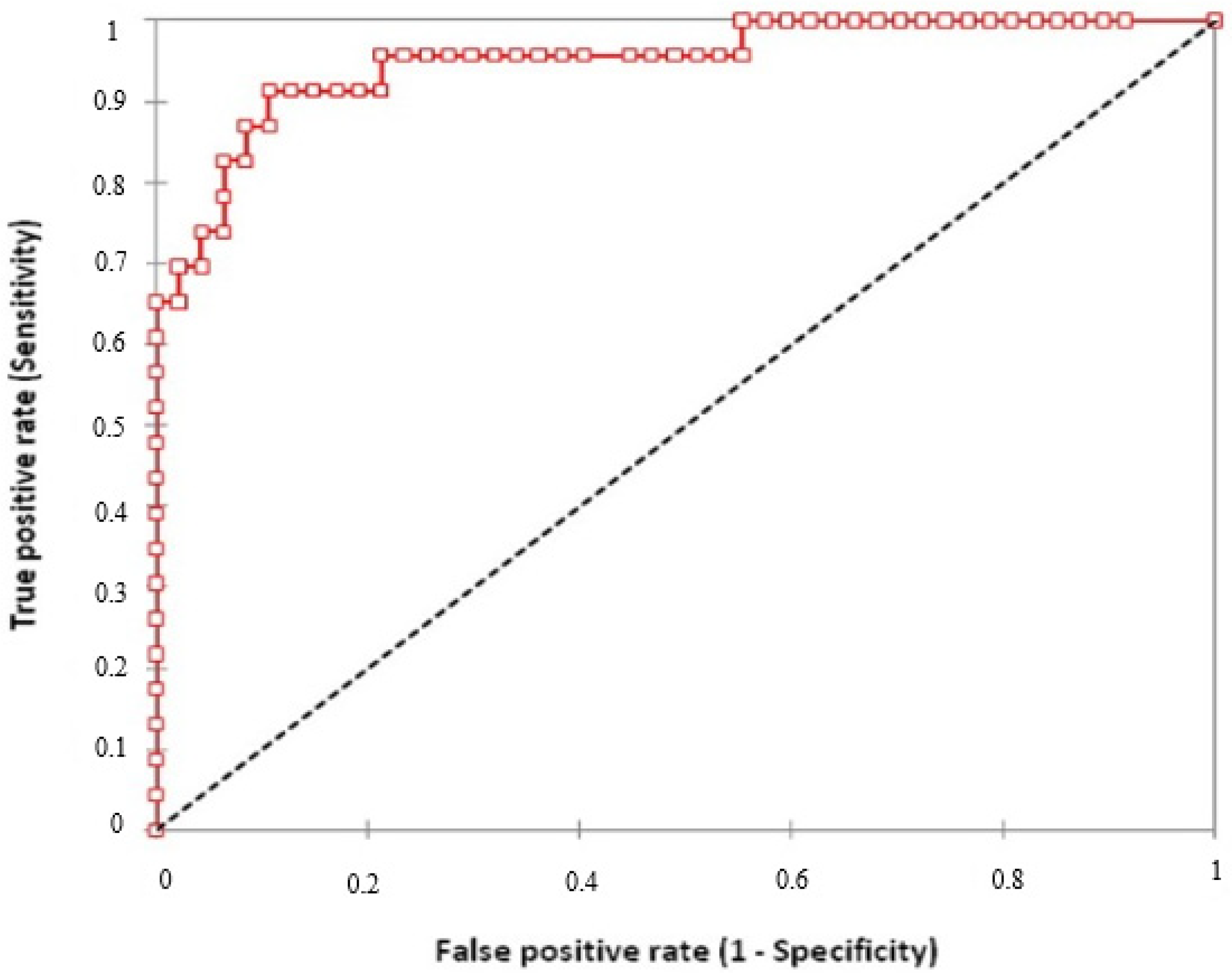
3.2. Cut-Off Point Determination for IP-10
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chiang, C.-Y.; Van Weezenbeek, C.; Mori, T.; Enarson, D.A. Challenges to the Global Control of Tuberculosis: Global Control of TB. Respirology 2013, 18, 596–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cantini, F.; Nannini, C.; Niccoli, L.; Petrone, L.; Ippolito, G.; Goletti, D. Risk of Tuberculosis Reactivation in Patients with Rheumatoid Arthritis, Ankylosing Spondylitis, and Psoriatic Arthritis Receiving Non-Anti-TNF-Targeted Biologics. Mediat. Inflamm. 2017, 2017, 8909834. [Google Scholar] [CrossRef] [PubMed]
- Saidenberg-Kermanac’h, N.; Semerano, L.; Naccache, J.M.; Brauner, M.; Falgarone, G.; Dumont-Fischer, D.; Guillot, X.; Valeyre, D.; Boissier, M.-C. Screening for Latent Tuberculosis in Anti-TNF-α Candidate Patients in a High Tuberculosis Incidence Setting. Int. J. Tuberc. Lung Dis. 2012, 16, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Cantini, F.; Nannini, C.; Niccoli, L.; Iannone, F.; Delogu, G.; Garlaschi, G.; Sanduzzi, A.; Matucci, A.; Prignano, F.; Conversano, M.; et al. Guidance for the Management of Patients with Latent Tuberculosis Infection Requiring Biologic Therapy in Rheumatology and Dermatology Clinical Practice. Autoimmun. Rev. 2015, 14, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Brock, I.; Weldingh, K.; Lillebaek, T.; Follmann, F.; Andersen, P. Comparison of Tuberculin Skin Test and New Specific Blood Test in Tuberculosis Contacts. Am. J. Respir. Crit. Care Med. 2004, 170, 65–69. [Google Scholar] [CrossRef]
- Martins, M.V.B.S.; Lima, M.C.B.S.; Duppre, N.C.; Matos, H.J.; Spencer, J.S.; Brennan, P.J.; Sarno, E.N.; Fonseca, L.; Pereira, G.M.B.; Pessolani, M.C.V. The Level of PPD-Specific IFN-γ-Producing CD4+ T Cells in the Blood Predicts the in Vivo Response to PPD. Tuberculosis 2007, 87, 202–211. [Google Scholar] [CrossRef]
- Andersen, P.; Munk, M.E.; Pollock, J.M.; Doherty, T.M. Specific Immune-Based Diagnosis of Tuberculosis. Lancet 2000, 356, 1099–1104. [Google Scholar] [CrossRef]
- Pai, M.; Zwerling, A.; Menzies, D. Systematic Review: T-Cell–Based Assays for the Diagnosis of Latent Tuberculosis Infection: An Update. Ann. Intern. Med. 2008, 149, 177. [Google Scholar] [CrossRef]
- Jeong, D.H.; Kang, J.; Jung, Y.J.; Yoo, B.; Lee, C.-K.; Kim, Y.-G.; Hong, S.; Shim, T.S.; Jo, K.-W. Comparison of Latent Tuberculosis Infection Screening Strategies before Tumor Necrosis Factor Inhibitor Treatment in Inflammatory Arthritis: IGRA-Alone versus Combination of TST and IGRA. PLoS ONE 2018, 13, e0198756. [Google Scholar] [CrossRef]
- Diel, R.; Goletti, D.; Ferrara, G.; Bothamley, G.; Cirillo, D.; Kampmann, B.; Lange, C.; Losi, M.; Markova, R.; Migliori, G.B.; et al. Interferon- Release Assays for the Diagnosis of Latent Mycobacterium Tuberculosis Infection: A Systematic Review and Meta-Analysis. Eur. Respir. J. 2011, 37, 88–99. [Google Scholar] [CrossRef] [Green Version]
- Bartalesi, F.; Vicidomini, S.; Goletti, D.; Fiorelli, C.; Fiori, G.; Melchiorre, D.; Tortoli, E.; Mantella, A.; Benucci, M.; Girardi, E.; et al. QuantiFERON-TB Gold and the TST Are Both Useful for Latent Tuberculosis Infection Screening in Autoimmune Diseases. Eur. Respir. J. 2009, 33, 586–593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruhwald, M.; Bodmer, T.; Maier, C.; Jepsen, M.; Haaland, M.B.; Eugen-Olsen, J.; Ravn, P.; on behalf of TBNET. Evaluating the Potential of IP-10 and MCP-2 as Biomarkers for the Diagnosis of Tuberculosis. Eur. Respir. J. 2008, 32, 1607–1615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sokal, J.E. Measurement of Delayed Skin-Test Responses. N. Engl. J. Med. 1975, 293, 501–502. [Google Scholar] [CrossRef]
- Republic of Turkey Ministry of Health. Directorate General of the Public Health, Tuberculosis Diagnosis and Treatment Guide, Tuberculosis Guideline in Patients Receiving Anti-TNF Treatment, Ankara, May 2019. Available online: https://hsgm.saglik.gov.tr/depo/birimler/tuberkuloz_db/haberler/Tuberkuloz_Tani_Ve_Tedavi_Rehberi_/Tuberkuloz_Tani_ve_Tedavi_Rehberi.pdf (accessed on 16 June 2022).
- Cellestis. QuantiFERON s-TB Gold Package Insert. Available online: https://www.quantiferon.com/products/quantiferon-tb-gold/package-inserts/ (accessed on 16 June 2022).
- RayBiotech. RayBio Human IP-10 ELISA Kit User Manual. Available online: https://www.raybiotech.com/human-ip-10-elisa/ (accessed on 16 June 2022).
- Ponce de Leon, D. Attenuated Response to Purified Protein Derivative in Patients with Rheumatoid Arthritis: Study in a Population with a High Prevalence of Tuberculosis. Ann. Rheum. Dis. 2005, 64, 1360–1361. [Google Scholar] [CrossRef] [PubMed]
- Ucan, E.; Sevinc, C.; Abadoglu, O.; Arpaz, S.; Ellidokuz, H. Interpretation of tuberculin test results standards of our country and new needs. J. Toraks 2000, 1, 25–29. Available online: https://turkthoracj.org/en/interpretation-of-tuberculin-test-results-standards-of-our-country-and-new-needs-1317/ (accessed on 16 June 2022).
- Smith, R.; Cattamanchi, A.; Steingart, K.R.; Denkinger, C.; Dheda, K.; Winthrop, K.L.; Pai, M. Interferon-Gamma Release Assays for Diagnosis of Latent Tuberculosis Infection: Evidence in Immune-Mediated Inflammatory Disorders. Curr. Opin. Rheumatol. 2011, 23, 377–384. [Google Scholar] [CrossRef]
- Cobanoglu, N.; Ozcelik, U.; Kalyoncu, U.; Ozen, S.; Kiraz, S.; Gurcan, N.; Kaplan, M.; Dogru, D.; Yalcin, E.; Pekcan, S.; et al. Interferon-Gamma Assays for the Diagnosis of Tuberculosis Infection before Using Tumour Necrosis Factor-Alpha Blockers. Int. J. Tuberc. Lung Dis. 2007, 11, 1177–1182. [Google Scholar]
- Ferrara, G.; Losi, M.; Meacci, M.; Meccugni, B.; Piro, R.; Roversi, P.; Bergamini, B.M.; D’Amico, R.; Marchegiano, P.; Rumpianesi, F.; et al. Routine Hospital Use of a New Commercial Whole Blood Interferon-Gamma Assay for the Diagnosis of Tuberculosis Infection. Am. J. Respir. Crit. Care Med. 2005, 172, 631–635. [Google Scholar] [CrossRef] [Green Version]
- Kobashi, Y.; Mouri, K.; Obase, Y.; Fukuda, M.; Miyashita, N.; Oka, M. Clinical Evaluation of QuantiFERON TB-2G Test for Immunocompromised Patients. Eur. Respir. J. 2007, 30, 945–950. [Google Scholar] [CrossRef] [Green Version]
- Ruhwald, M.; Bjerregaard-Andersen, M.; Rabna, P.; Kofoed, K.; Eugen-Olsen, J.; Ravn, P. CXCL10/IP-10 Release Is Induced by Incubation of Whole Blood from Tuberculosis Patients with ESAT-6, CFP10 and TB7.7. Microbes Infect. 2007, 9, 806–812. [Google Scholar] [CrossRef]
- Kabeer, B.S.A.; Raja, A.; Raman, B.; Thangaraj, S.; Leportier, M.; Ippolito, G.; Girardi, E.; Lagrange, P.H.; Goletti, D. IP-10 Response to RD1 Antigens Might Be a Useful Biomarker for Monitoring Tuberculosis Therapy. BMC Infect. Dis. 2011, 11, 135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aabye, M.G.; Ruhwald, M.; PrayGod, G.; Jeremiah, K.; Faurholt-Jepsen, M.; Faurholt-Jepsen, D.; Range, N.; Friis, H.; Changalucha, J.; Andersen, A.B.; et al. Potential of Interferon-γ-Inducible Protein 10 in Improving Tuberculosis Diagnosis in HIV-Infected Patients. Eur. Respir. J. 2010, 36, 1488–1490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruhwald, M.; Dominguez, J.; Latorre, I.; Losi, M.; Richeldi, L.; Pasticci, M.B.; Mazzolla, R.; Goletti, D.; Butera, O.; Bruchfeld, J.; et al. A Multicentre Evaluation of the Accuracy and Performance of IP-10 for the Diagnosis of Infection with M. tuberculosis. Tuberculosis 2011, 91, 260–267. [Google Scholar] [CrossRef]
- Syed Ahamed Kabeer, B.; Raman, B.; Thomas, A.; Perumal, V.; Raja, A. Role of QuantiFERON-TB Gold, Interferon Gamma Inducible Protein-10 and Tuberculin Skin Test in Active Tuberculosis Diagnosis. PLoS ONE 2010, 5, e9051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mamishi, S.; Mahmoudi, S.; Banar, M.; Hosseinpour Sadeghi, R.; Marjani, M.; Pourakbari, B. Diagnostic Accuracy of Interferon (IFN)-γ Inducible Protein 10 (IP-10) as a Biomarker for the Discrimination of Active and Latent Tuberculosis. Mol. Biol. Rep. 2019, 46, 6263–6269. [Google Scholar] [CrossRef]
- Ghanaie, R.M.; Karimi, A.; Azimi, L.; James, S.; Nasehi, M.; Mishkar, A.P.; Sheikhi, M.; Fallah, F.; Tabatabaei, S.R.; Hoseini-Alfatemi, S.M. Diagnosis of Latent Tuberculosis Infection among Pediatric Household Contacts of Iranian Tuberculosis Cases Using Tuberculin Skin Test, IFN- γ Release Assay and IFN-γ-Induced Protein-10. BMC Pediatr. 2021, 21, 76. [Google Scholar] [CrossRef]
- Murdaca, G.; Negrini, S.; Pellecchio, M.; Greco, M.; Schiavi, C.; Giusti, F.; Puppo, F. Update upon the Infection Risk in Patients Receiving TNF Alpha Inhibitors. Expert Opin. Drug Saf. 2019, 18, 219–229. [Google Scholar] [CrossRef]
- Murdaca, G.; Spanò, F.; Contatore, M.; Guastalla, A.; Penza, E.; Magnani, O.; Puppo, F. Infection Risk Associated with Anti-TNF-α Agents: A Review. Expert Opin. Drug Saf. 2015, 14, 571–582. [Google Scholar] [CrossRef]
- Villar-Hernández, R.; Latorre, I.; Mínguez, S.; Díaz, J.; García-García, E.; Muriel-Moreno, B.; Lacoma, A.; Prat, C.; Olivé, A.; Ruhwald, M.; et al. Use of IFN-γ and IP-10 Detection in the Diagnosis of Latent Tuberculosis Infection in Patients with Inflammatory Rheumatic Diseases. J. Infect. 2017, 75, 315–325. [Google Scholar] [CrossRef]
- Chen, D.-Y.; Shen, G.-H.; Chen, Y.-M.; Chen, H.-H.; Lin, C.-C.; Hsieh, C.-W.; Lan, J.-L. Interferon-Inducible Protein-10 as a Marker to Detect Latent and Active Tuberculosis in Rheumatoid Arthritis. Int. J. Tuberc. Lung Dis. 2011, 15, 192–200. [Google Scholar]
- Ruhwald, M.; Aabye, M.G.; Ravn, P. IP-10 Release Assays in the Diagnosis of Tuberculosis Infection: Current Status and Future Directions. Expert Rev. Mol. Diagn 2012, 12, 175–187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Type of IRD 1 | (n/%) |
|---|---|
| Ankylosing spondylitis, n/% | 45/64.3% |
| Rheumotoid arthritis, n/% | 25/35.7% |
| Mean age, years ± SD | 47 ± 14 |
| Male, n/% | 36/51.4% |
| BCG positivity, n/% | 54/77.1% |
| TST positivity, n/% | 54/77.1% |
| QFT-GIT positivity, n/% | 21/30% |
| n | TST (+) 1 (n = 54) | p | QFT-GIT (+) (n = 21) | p | IP-10 (+) (n = 31) | p | |
|---|---|---|---|---|---|---|---|
| Gender, n (%) | 0.660 | ||||||
| Female | 34 | 23 (67.6) | 8 (23.5) | >0.05 | 9 (26.5) | 0.004 | |
| Male | 36 | 31 (86.1) | 13 (36.1) | data | 22 (61.1) | ||
| Age, years, n (%) | 0.03 - | >0.05 | 0.66 | ||||
| <29 | 8 | 7 (87.5) | 1 (12.5) | 2 (25.0) | - | ||
| 30–49 | 35 | 31 (88.6) | 10 (28.6) | 16 (45.7) | |||
| 50–69 | 22 | 15 (68.2) | 10 (45.5) | data | 11 (50.0) | ||
| >70 | 5 | 1 (20.0) | 0 (0.0) | data | 2 (40.0) | ||
| BCG vaccinated, n (%) | |||||||
| Yes | 54 | 44 (82.4) | 0.230 | 15 (42.9) | 0.190 | 22 (43.1) | 0.160 |
| No | 16 | 10 (64.3) | 6 (29.4) | data | 9 (64.3) | ||
| Diagnosis, n (%) | |||||||
| RA | 25 | 14 (56.0) | 0.020 | 6 (24.0) | 0.410 | 9 (12.9) | 0.290 |
| AS | 45 | 40 (88.8) | 15 (33.3) | 22 (31.4) | |||
| Treatment TNF-α inhibitors, n (%) | |||||||
| Yes | 28 | 20 (71.4) | 0.350 | 5 (25.0) | 0.700 | 9 (12.9) | 0.090 |
| No | 42 | 34 (80.9) | 16 (38.0) | 22 (52.3) | |||
| Steroid, n (%) | |||||||
| Yes | 12 | 8 (75) | 0.45 | 5 (62.5) | 0.25 | 6 (50) | 0.52 |
| No | 58 | 46 (85.2) | 16 (27.6) | 25(43.1) |
| Tests | Test Results | Kappa Value | |||
|---|---|---|---|---|---|
| −/− | +/− | −/+ | +/+ | ||
| TST/QFT-GIT | 16 | 33 | 0 | 21 | 0.24 |
| TST/IP-10 | 12 | 27 | 4 | 27 | 0.19 |
| QFT-GIT/IP-10 | 38 | 1 | 11 | 20 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortakoylu, M.G.; Bahadir, A.; Iliaz, S.; Soy Bugdayci, D.; Uysal, M.A.; PAKER, N.; Tural Onur, S. Interferon-Inducible Protein-10 as a Marker to Detect Latent Tuberculosis Infection in Patients with Inflammatory Rheumatic Diseases. J. Pers. Med. 2022, 12, 1027. https://doi.org/10.3390/jpm12071027
Ortakoylu MG, Bahadir A, Iliaz S, Soy Bugdayci D, Uysal MA, PAKER N, Tural Onur S. Interferon-Inducible Protein-10 as a Marker to Detect Latent Tuberculosis Infection in Patients with Inflammatory Rheumatic Diseases. Journal of Personalized Medicine. 2022; 12(7):1027. https://doi.org/10.3390/jpm12071027
Chicago/Turabian StyleOrtakoylu, Mediha Gonenc, Ayse Bahadir, Sinem Iliaz, Derya Soy Bugdayci, Mehmet Atilla Uysal, Nurdan PAKER, and Seda Tural Onur. 2022. "Interferon-Inducible Protein-10 as a Marker to Detect Latent Tuberculosis Infection in Patients with Inflammatory Rheumatic Diseases" Journal of Personalized Medicine 12, no. 7: 1027. https://doi.org/10.3390/jpm12071027
APA StyleOrtakoylu, M. G., Bahadir, A., Iliaz, S., Soy Bugdayci, D., Uysal, M. A., PAKER, N., & Tural Onur, S. (2022). Interferon-Inducible Protein-10 as a Marker to Detect Latent Tuberculosis Infection in Patients with Inflammatory Rheumatic Diseases. Journal of Personalized Medicine, 12(7), 1027. https://doi.org/10.3390/jpm12071027






