Serological Response and Relationship with Gender-Sensitive Variables among Healthcare Workers after SARS-CoV-2 Vaccination
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Vaccination and Blood Specimen Collection
2.3. Antibody Measurement and Interpretation
2.4. Statistical Analysis
3. Results
3.1. Anti-S IgG at T1 (1 Month after Second Injection)
3.2. Anti-S IgG at T2 (5 Months after Second Injection)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Walls, A.C.; Park, Y.J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292.e6. [Google Scholar] [CrossRef] [PubMed]
- Antia, A.; Ahmed, H.; Handel, A.; Carlson, N.E.; Amanna, I.J.; Antia, R.; Slifka, M. Heterogeneity and longevity of antibody memory to viruses and vaccines. PLOS Biol. 2018, 16, e2006601. [Google Scholar] [CrossRef]
- Sethuraman, N.; Jeremiah, S.S.; Ryo, A. Interpreting Diagnostic Tests for SARS-CoV-2. JAMA 2020, 323, 2249–2251. [Google Scholar] [CrossRef]
- de Giorgi, V.; West, K.A.; Henning, A.N.; Chen, L.N.; Holbrook, M.R.; Gross, R.; Liang, J.; Postnikova, E.; Trenbeath, J.; Pogue, S.; et al. Naturally Acquired SARS-CoV-2 Immunity Persists for Up to 11 Months Following Infection. J. Infect. Dis. 2021, 224, 1294–1304. [Google Scholar] [CrossRef]
- Campo, F.; Venuti, A.; Pimpinelli, F.; Abril, E.; Blandino, G.; Conti, L.; De Virgilio, A.; De Marco, F.; Di Noia, V.; Di Domenico, E.G.; et al. Antibody Persistence 6 Months Post-Vaccination with BNT162b2 among Health Care Workers. Vaccines 2021, 9, 1125. [Google Scholar] [CrossRef] [PubMed]
- Cromer, D.; Juno, J.A.; Khoury, D.; Reynaldi, A.; Wheatley, A.K.; Kent, S.J.; Davenport, M.P. Prospects for durable immune control of SARS-CoV-2 and prevention of reinfection. Nat. Rev. Immunol. 2021, 21, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Mehta, P.; Paul, A.; Anu, S.; Cherian, S.; Shenoy, V.; Nalianda, K.K.; Joseph, S.; Poulose, A.; Shenoy, P. Postvaccination antibody titres predict protection against COVID-19 in patients with autoimmune diseases: Survival analysis in a prospective cohort. Ann. Rheum. Dis. 2022, 81, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Levin, E.G.; Lustig, Y.; Cohen, C.; Fluss, R.; Indenbaum, V.; Amit, S.; Doolman, R.; Asraf, K.; Mendelson, E.; Ziv, A.; et al. Waning Immune Humoral Response to BNT162b2 COVID-19 Vaccine over 6 Months. N. Engl. J. Med. 2021, 385, e84. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Balena, A.; Tuccinardi, D.; Tozzi, R.; Risi, R.; Masi, D.; Caputi, A.; Rossetti, R.; Spoltore, M.E.; Filippi, V.; et al. Central obesity, smoking habit, and hypertension are associated with lower antibody titres in response to COVID-19 mRNA vaccine. Diabetes/Metab. Res. Rev. 2022, 38, e3465. [Google Scholar] [CrossRef]
- Bonelli, F.; Blocki, F.A.; Bunnell, T.; Chu, E.; De La O, A.; Grenache, D.G.; Marzucchi, G.; Montomoli, E.; Okoye, L.; Pallavicini, L.; et al. Evaluation of the automated LIAISON ® SARS-CoV-2 TrimericS IgG assay for the detection of circulating antibodies. Clin. Chem. Lab. Med. 2021, 59, 1463–1467. [Google Scholar] [CrossRef] [PubMed]
- Pani, A.; Cento, V.; Vismara, C.; Campisi, D.; Di Ruscio, F.; Romandini, A.; Senatore, M.; Schenardi, P.A.; Gagliardi, O.M.; Giroldi, S.; et al. Results of the RENAISSANCE Study: REsponse to BNT162b2 COVID-19 vacciNe—short- And long-term Immune reSponSe evAluatioN in health Care workErs. Mayo Clin. Proc. 2021, 96, 2966–2979. [Google Scholar] [CrossRef] [PubMed]
- Mariani, M.; Acquila, M.; Tripodi, G.; Spiazzi, R.; Castagnola, E. Antibodies against Receptor Binding Domain of SARS-CoV-2 spike protein induced by BNT162b2 vaccine: Results from a pragmatic, real-life study. J. Infect. Public Health 2021, 14, 1560–1562. [Google Scholar] [CrossRef] [PubMed]
- Padoan, A.; Dall’Olmo, L.; Della Rocca, F.; Barbaro, F.; Cosma, C.; Basso, D.; Cattelan, A.; Cianci, V.; Plebani, M. Antibody response to first and second dose of BNT162b2 in a cohort of characterized healthcare workers. Clin. Chim. Acta 2021, 519, 60–63. [Google Scholar] [CrossRef]
- Brisotto, G.; Muraro, E.; Montico, M.; Corso, C.; Evangelista, C.; Casarotto, M.; Caffau, C.; Vettori, R.; Cozzi, M.R.; Zanussi, S.; et al. IgG antibodies against SARS-CoV-2 decay but persist 4 months after vaccination in a cohort of healthcare workers. Clin. Chim. Acta 2021, 523, 476–482. [Google Scholar] [CrossRef]
- Tré-Hardy, M.; Cupaiolo, R.; Wilmet, A.; Beukinga, I.; Blairon, L. Waning antibodies in SARS-CoV-2 naïve vaccinees: Results of a three-month interim analysis of ongoing immunogenicity and efficacy surveillance of the mRNA-1273 vaccine in healthcare workers. J. Infect. 2021, 83, 381–412. [Google Scholar] [CrossRef]
- Boyarsky, B.J.; Werbel, W.A.; Avery, R.K.; Tobian, A.A.; Massie, A.B.; Segev, D.L.; Garonzik-Wang, J.M. Antibody Response to 2-Dose SARS-CoV-2 mRNA Vaccine Series in Solid Organ Transplant Recipients. JAMA 2021, 325, 2204–2206. [Google Scholar] [CrossRef]
- Mandl, P.; Tobudic, S.; Haslacher, H.; Karonitsch, T.; Mrak, D.; Nothnagl, T.; Perkmann, T.; Radner, H.; Sautner, J.; Simader, E.; et al. Response to SARS-CoV-2 vaccination in systemic autoimmune rheumatic disease depends on immunosuppressive regimen: A matched, prospective cohort study. Ann. Rheum. Dis. 2022, 81, 1017–1022. [Google Scholar] [CrossRef]
- Salvagno, G.L.; Henry, B.M.; Pighi, L.; De Nitto, S.; Gianfilippi, G.L.; Lippi, G. Three-month analysis of total humoral response to Pfizer BNT162b2 mRNA COVID-19 vaccination in healthcare workers. J. Infect. 2021, 83, e4–e5. [Google Scholar] [CrossRef]
- Ferrara, P.; Gianfredi, V.; Tomaselli, V.; Polosa, R. The Effect of Smoking on Humoral Response to COVID-19 Vaccines: A Systematic Review of Epidemiological Studies. Vaccines 2022, 10, 303. [Google Scholar] [CrossRef]
- Sahni, S.; Gupta, G.; Sarda, R.; Pandey, S.; Pandey, R.; Sinha, S. Impact of metabolic and cardiovascular disease on COVID-19 mortality: A systematic review and meta-analysis. Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 102308. [Google Scholar] [CrossRef] [PubMed]
- Dan, J.M.; Mateus, J.; Kato, Y.; Hastie, K.M.; Yu, E.D.; Faliti, C.E.; Grifoni, A.; Ramirez, S.I.; Haupt, S.; Frazier, A.; et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science 2021, 371, eabf4063. [Google Scholar] [CrossRef] [PubMed]
- Notarte, K.I.; Guerrero-Arguero, I.; Jacqueline, I.; Velasco, J.V.; Ver, A.T.; de Oliveira, M.H.S.; Catahay, J.A.; Khan, S.R.; Pastrana, A.; Juszczyk, G.; et al. Characterization of the significant decline in humoral immune response six months post-SARS-CoV-2 mRNA vaccination: A systematic review. J. Med. Virol. 2022, 94, 2939–2961. [Google Scholar] [CrossRef] [PubMed]
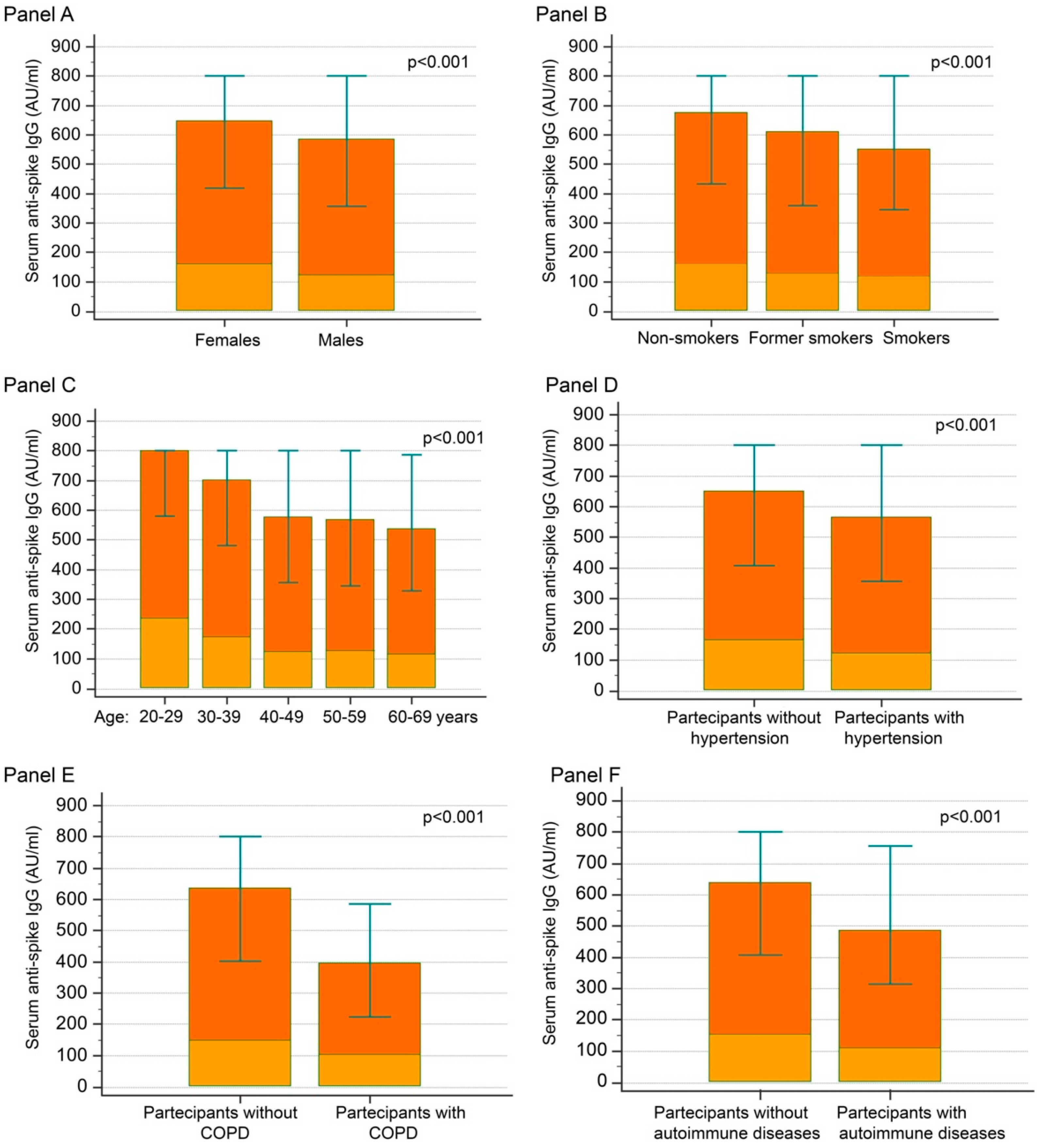
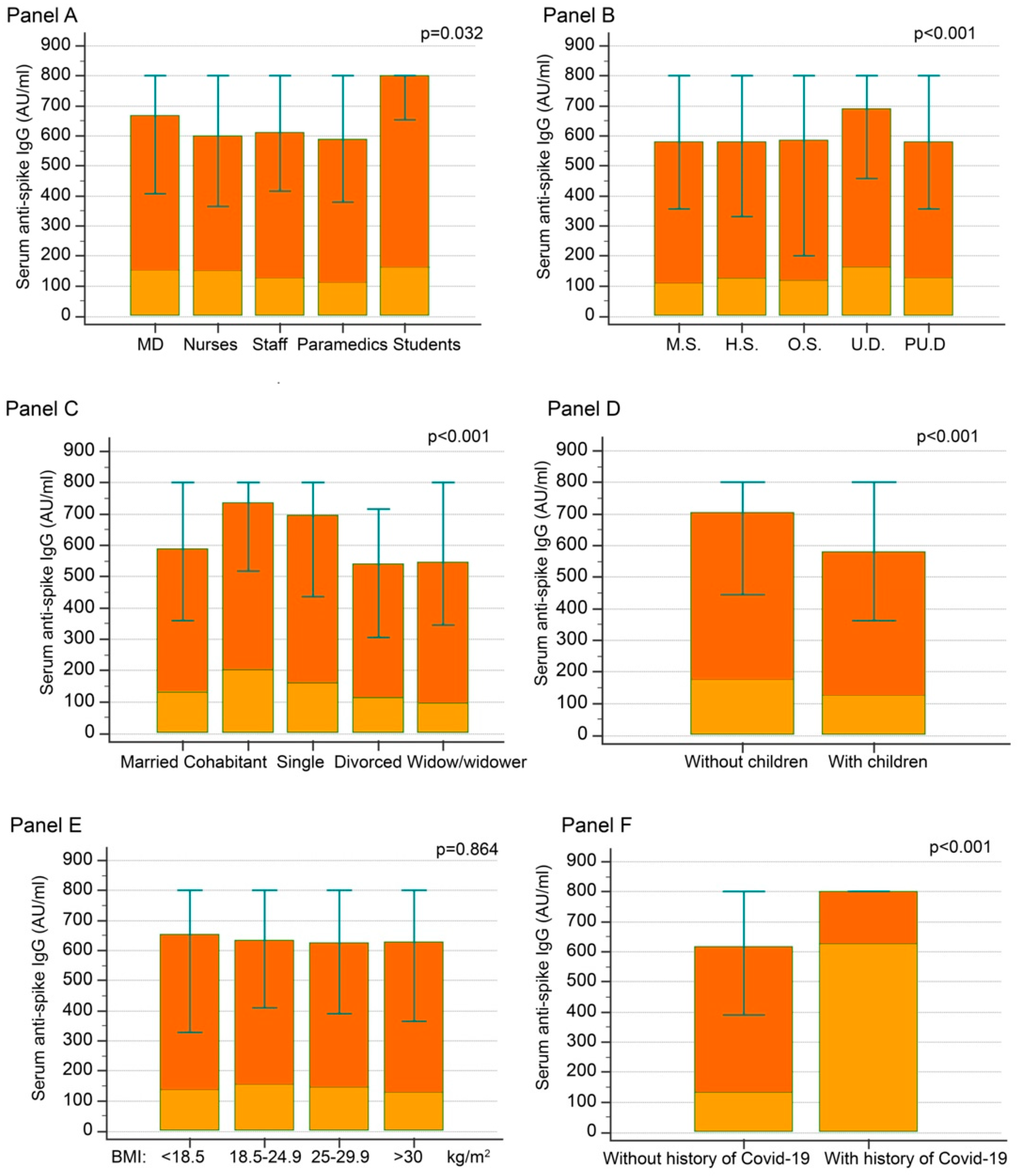
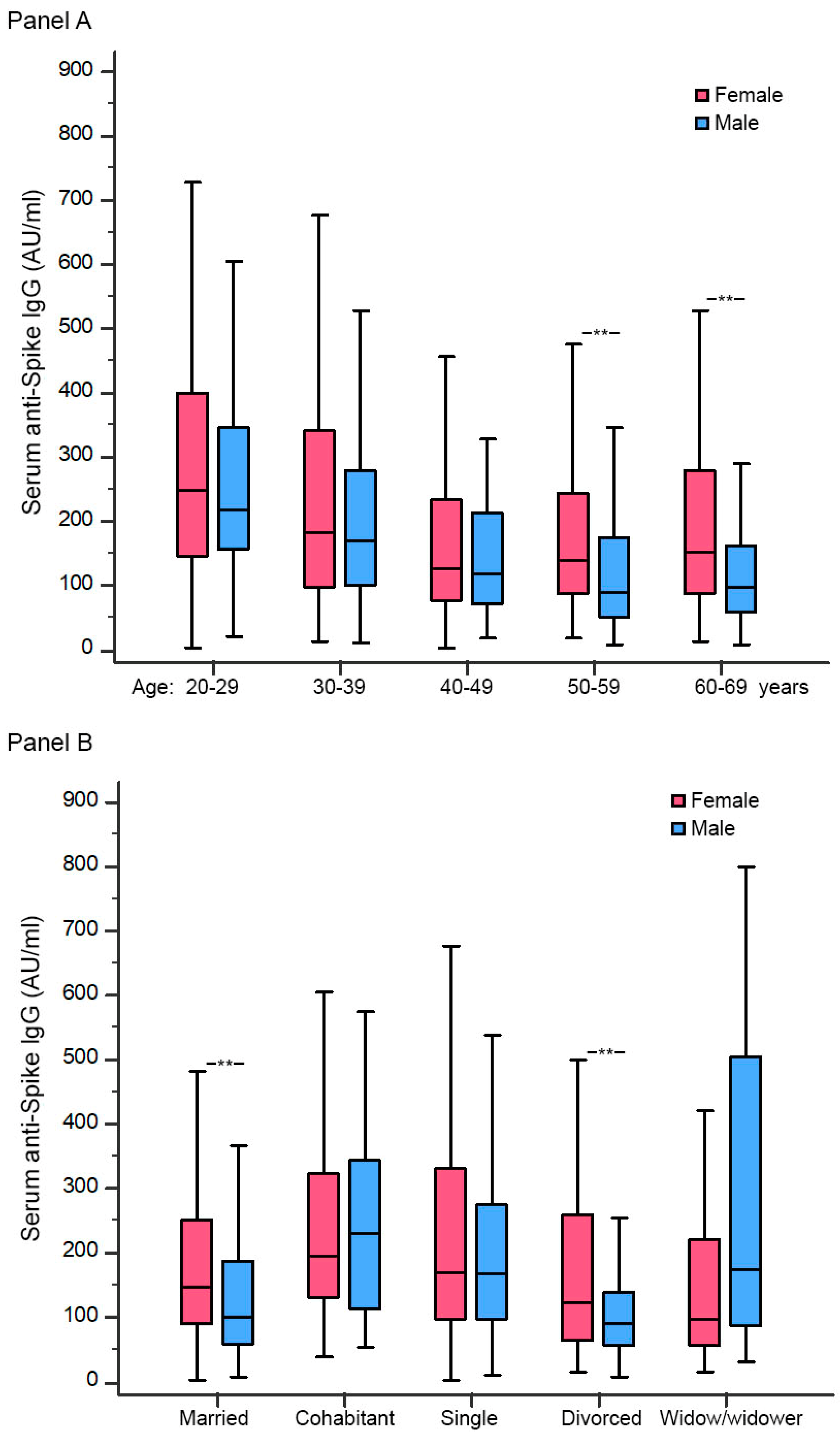
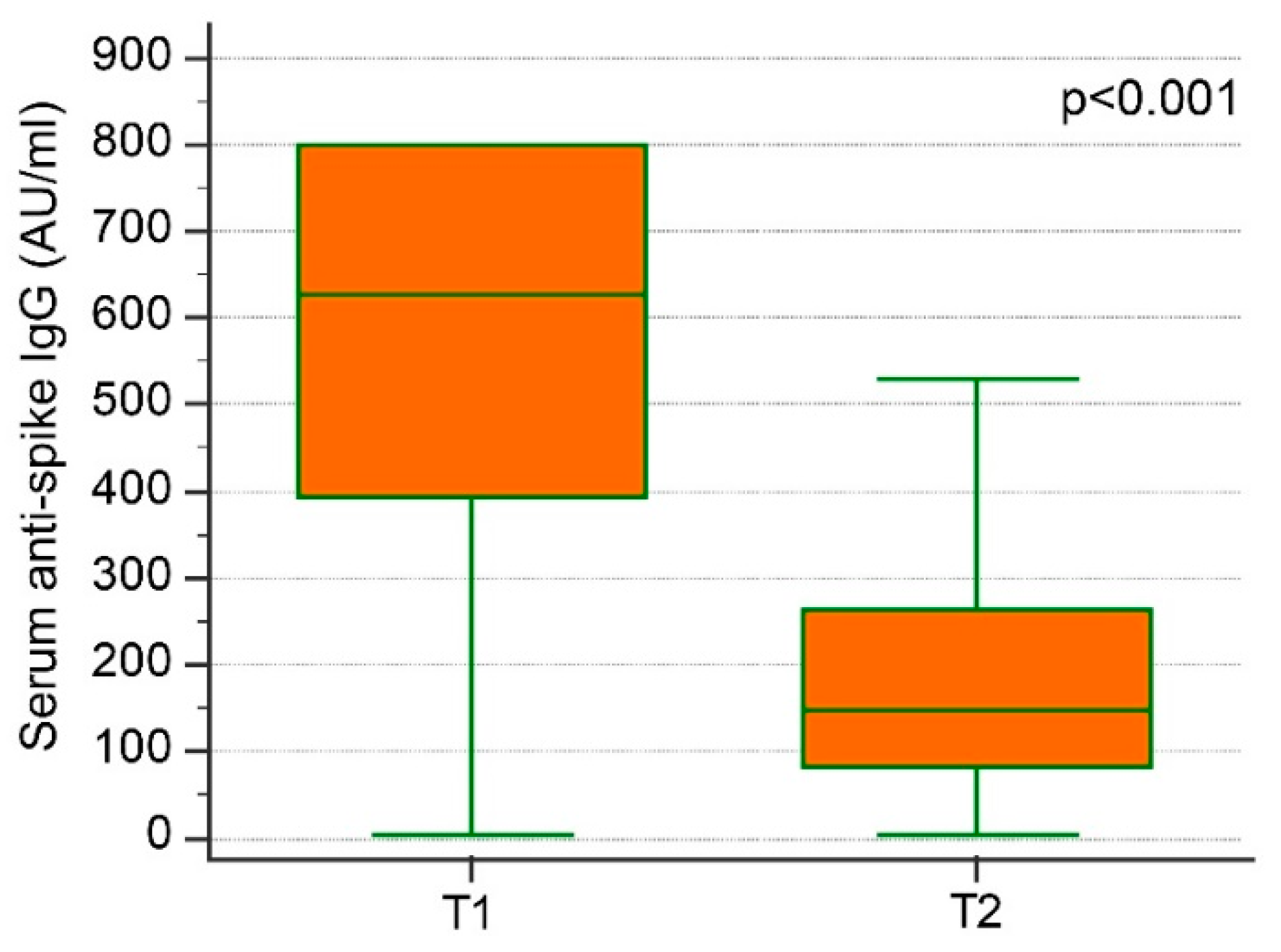
| Characteristics | Anti-S IgG (≥800 AU) | Anti-S IgG (<800 AU) | p | |
|---|---|---|---|---|
| Age (years) | 45.9 ± 13.3 | 41.9 ± 13.6 | 47.9 ± 12.8 | <0.001 |
| Female sex (%) | 63.3 | 66.4 | 61.7 | 0.037 |
| BMI (kg/m2) | 26.1 ± 10.2 | 26.6 ± 12.9 | 25.9 ± 8.3 | 0.258 |
| Age classes: | ||||
| 20–29 years (%) | 18.5 | 29 | 13.4 | |
| 30–39 years (%) | 19.4 | 22.8 | 17.8 | |
| 40–49 years (%) | 15.5 | 12.6 | 17 | <0.001 |
| 50–59 years (%) | 27.1 | 21.6 | 29.8 | |
| 60–69 years (%) | 19.4 | 14 | 21.9 | |
| Medical doctors (%) | 49.8 | 53.8 | 47.6 | |
| Nurses (%) | 32.4 | 30.1 | 33.6 | |
| Administrative staff (%) | 11.6 | 10.3 | 12.3 | 0.069 |
| Paramedics (%) | 5.7 | 4.9 | 5.1 | |
| Students (%) | 0.6 | 1.0 | 0.4 | |
| University degree (%) | 54.2 | 59.3 | 51.5 | |
| Post-university degree (%) | 25.5 | 22.9 | 26.9 | |
| High school (%) | 15.6 | 13.7 | 16.6 | 0.032 |
| Middle school (%) | 3.5 | 2.8 | 3.9 | |
| Other (%) | 1.2 | 1.3 | 1.2 | |
| Single (%) | 40.0 | 45.9 | 36.8 | |
| Married (%) | 41.7 | 35.6 | 45.0 | |
| Cohabitant (%) | 9.3 | 12.3 | 7.7 | <0.001 |
| Divorced (%) | 7.4 | 4.9 | 8.8 | |
| Widow/widower (%) | 1.6 | 1.3 | 1.8 | |
| Having children (%) | 52 | 43 | 57 | <0.001 |
| Smokers (%) | 25.8 | 19.8 | 29.0 | |
| Former smokers (%) | 8.9 | 8 | 9.4 | <0.001 |
| Non-smokers (%) | 65.3 | 72.2 | 61.6 | |
| Hypertension (%) | 18.5 | 14.9 | 20.4 | 0.004 |
| Dyslipidemia (%) | 9.4 | 8.3 | 9.9 | 0.279 |
| T2DM (%) | 2.1 | 1.8 | 2.3 | 0.502 |
| Coronary heart disease (%) | 1.0 | 0.7 | 1.1 | 0.322 |
| COPD (%) | 0.8 | 0.2 | 1.1 | 0.029 |
| Asthma (%) | 5.7 | 5.2 | 6.0 | 0.527 |
| History of VTE (%) | 1.1 | 0.8 | 1.3 | 0.349 |
| Heart failure (%) | 0.2 | 0.3 | 0.1 | 0.249 |
| Chronic hepatic disease (%) | 0.2 | 0.2 | 0.2 | 0.954 |
| Chronic kidney disease (%) | 0.3 | 0.0 | 0.5 | 0.072 |
| Neoplastic disease (%) | 0.6 | 0.5 | 0.6 | 0.743 |
| Atrial fibrillation (%) | 0.7 | 0.2 | 1.1 | 0.039 |
| Autoimmune diseases (%) | 5.2 | 3.3 | 6.2 | 0.008 |
| Hematologic diseases (%) | 0.8 | 0.7 | 0.9 | 0.616 |
| History of COVID-19 (%) | 6.0 | 14.3 | 1.8 | <0.001 |
| O.R. | 95% CI | p | ||
|---|---|---|---|---|
| Age decades | 0.804 | 0.764 | 0.845 | <0.001 |
| Smoking habit | 0.581 | 0.452 | 0.748 | <0.001 |
| Autoimmune diseases | 0.552 | 0.327 | 0.930 | 0.026 |
| History of COVID-19 (anti-N IgG and/or self-reported) | 7.771 | 4.672 | 12.92 | <0.001 |
| OR | 95% CI | p | ||
|---|---|---|---|---|
| Male sex | 1.384 | 1.052 | 1.820 | 0.021 |
| History of COVID-19 | 0.218 | 0.078 | 0.610 | <0.001 |
| Marital status: cohabitant | 0.449 | 0.246 | 0.822 | 0.005 |
| Age decades | 1.171 | 1.080 | 1.271 | <0.001 |
| Hypertension | 1.405 | 1.015 | 1.946 | 0.042 |
| Smoking habit | 1.376 | 1.021 | 1.855 | 0.038 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cangemi, R.; Di Franco, M.; Angeloni, A.; Zicari, A.; Cardinale, V.; Visentini, M.; Antonelli, G.; Napoli, A.; Anastasi, E.; Romiti, G.F.; et al. Serological Response and Relationship with Gender-Sensitive Variables among Healthcare Workers after SARS-CoV-2 Vaccination. J. Pers. Med. 2022, 12, 994. https://doi.org/10.3390/jpm12060994
Cangemi R, Di Franco M, Angeloni A, Zicari A, Cardinale V, Visentini M, Antonelli G, Napoli A, Anastasi E, Romiti GF, et al. Serological Response and Relationship with Gender-Sensitive Variables among Healthcare Workers after SARS-CoV-2 Vaccination. Journal of Personalized Medicine. 2022; 12(6):994. https://doi.org/10.3390/jpm12060994
Chicago/Turabian StyleCangemi, Roberto, Manuela Di Franco, Antonio Angeloni, Alessandra Zicari, Vincenzo Cardinale, Marcella Visentini, Guido Antonelli, Anna Napoli, Emanuela Anastasi, Giulio Francesco Romiti, and et al. 2022. "Serological Response and Relationship with Gender-Sensitive Variables among Healthcare Workers after SARS-CoV-2 Vaccination" Journal of Personalized Medicine 12, no. 6: 994. https://doi.org/10.3390/jpm12060994
APA StyleCangemi, R., Di Franco, M., Angeloni, A., Zicari, A., Cardinale, V., Visentini, M., Antonelli, G., Napoli, A., Anastasi, E., Romiti, G. F., d’Alba, F., Alvaro, D., Polimeni, A., Basili, S., & SAPIENZAVAX Collaborators. (2022). Serological Response and Relationship with Gender-Sensitive Variables among Healthcare Workers after SARS-CoV-2 Vaccination. Journal of Personalized Medicine, 12(6), 994. https://doi.org/10.3390/jpm12060994












