Sex-Related Differences in Pharmacological Response to CNS Drugs: A Narrative Review
Abstract
:1. Introduction
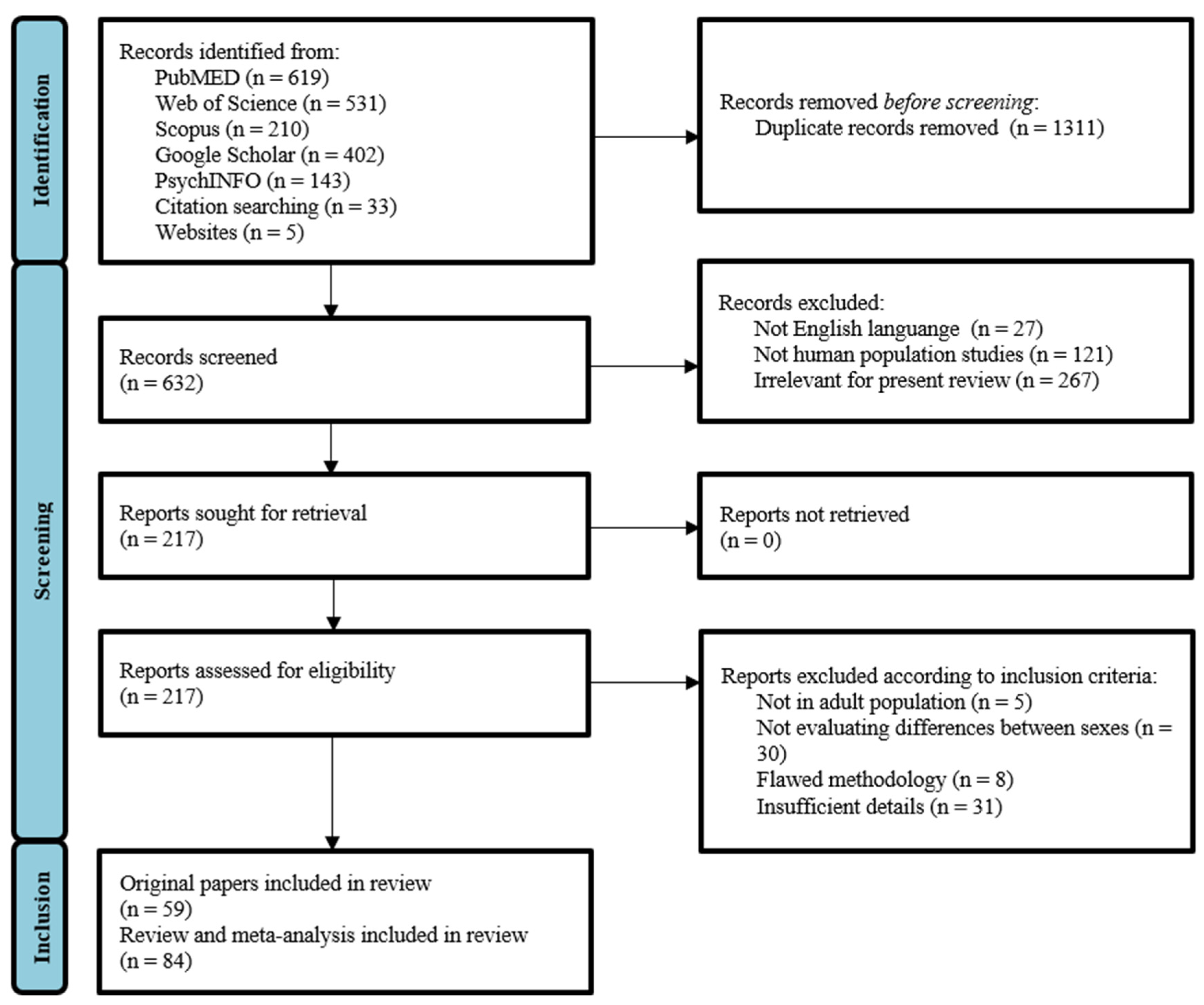
2. Materials and Methods
2.1. Literature Search Strategy
2.2. Selection Criteria
- -
- Human studies (clinical trials, experimental studies, case reports, reviews, meta-analysis);
- -
- Studies in adult patients (subjects > 18 years);
- -
- Studies that investigated sex differences.
- -
- Investigated animal populations;
- -
- Failed to analyze outcomes and differentiate between men and women;
- -
- Were methodologically flawed;
- -
- Provided insufficient details or irrelevant outcomes.
2.3. Selection Process
2.4. Data Extraction
3. Opioids
4. Antidepressants
5. Anticonvulsants
6. Antipsychotics
| Pharmacological Class/Drug | Effectiveness | Comments and Conclusion | Reference | |
|---|---|---|---|---|
| Results | Consistency | |||
| Opioids | F > M | C | Mixed µ-k-opioid agonist-antagonists and pure µ-agonists appear to be slightly more effective in women judging by the consumption of opioids in the two sexes. | [43,44,45,46,47,48,49,50,51] |
| Morphine | F > M | B | The majority of studies indicate that immediate postoperative analgesia is less effective in women since they experience a slower speed of onset. Contrarywise, PCA shows that female patients self-administer significantly less morphine than males. | [12,25,27,28,29,30,31,32,33,34,35] |
| TCAs | M > F | C | Males report an increased efficacy of pro-noradrenergic drugs probably due to their lower tolerance to sexual dysfunctions associated with SSRIs. | [55,56,73,74,75,76,77,78,79] |
| SSRIs | F > M | C | Women respond better to pro-serotoninergic drugs because anticholinergic ADRs associated with TCAs might be less desirable to these patients. | [55,56,73,74,75,76,77,78,79] |
| Anticonvulsants | M = F | B | There are few studies available, which either do not address gender dissimilarities, or their findings are not statistically significant. | [101,102] |
| Antipsychotics | F > M | C | Antipsychotic response seems to be higher in females, but this may simply indicate that, compared to males, they are at an earlier stage of illness. | [124,125,126,127,128,129,130] |
| Pharmacological Class/Drug | ADRs’ Frequency/Intensity | Comments and Conclusion | Reference | ||
|---|---|---|---|---|---|
| Results | Consistency | Type of ADRs | |||
| Morphine | F > M | A | Nausea, vomiting, respiratory depression | Gastrointestinal and respiratory ADRs are considerably more frequent in women. There are hints that cardiovascular ADRs are also influenced by sex, but the available data are scarce. | [36,37,38,39,40,41,42] |
| TCAs | F > M | B | Dry mouth, constipation, sedation, sweating, and tremor | Pharmacokinetic studies revealed that women have higher plasma levels of TCAs than men, therefore being more sensitive to side effects. | [56,78,79] |
| SSRIs | M > F | B | Sexual dysfunction | SSRIs deteriorate the sexual function precisely through: impairment in desire and arousal, inhibition of orgasm, delayed ejaculation, and male impotence. | [90,91,92] |
| Anticonvulsants | - | I | Sex-hormone-related ADRs | Generally, AEDs can lead to changes within sex hormones’ metabolism. However, since these drugs have a multitude of mechanisms of action, a general conclusion over sex-related differences cannot be drawn. | [101,112,113,114,115,116] |
| Valproic acid | F > M | B | Polycystic ovary syndrome, hyperinsulinism, hyperandrogenism, hypothalamic amenorrhea | Valproic acid has been incriminated in producing gender-related side effects, especially among women, increasing the incidence of the mentioned ADRs, apart from the acknowledged effects on offsprings. | [116] |
| CarbamazepinePhenytoinPhenobarbital | F > M | B | Alteration in bone metabolism | They increase the levels of sex-hormone-binding-globulin and decrease the levels of total serum testosterone, free androgen index, dehydroepiandosterone sulfate, and estradiol. | [112,113] |
| Antipsychotics | F > M | B | metabolic dysfunctions, cardiovascular disorders, hyperprolactinemia | Females exhibit lower fasting plasma glucose levels, elevated waist circumference and waist-to-hip ratio, prolonged QTc interval, and reduced bone density due to hyperprolactinemia. | [124,134,135,136,137] |
| M > F | C | acute dystonic reactions, tardive dystonia, akathisia | Males are generally more prone to developing extrapyramidal side effects. | [122] | |
| M = F | B | Sexual dysfunction | ADRs are due to dopaminergic antagonists (females) or drugs having α1-antiadrenergic/anticholinergic properties (males). | [138,139,140,141,142,143] | |
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Whitley, H.P.; Lindsey, W. Sex-Based Differences in Drug Activity. Am. Fam. Physician 2009, 80, 1254–1258. [Google Scholar] [PubMed]
- Beery, A.K.; Zucker, I. Sex Bias in Neuroscience and Biomedical Research. Neurosci. Biobehav. Rev. 2011, 35, 565–572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parekh, A.; Fadiran, E.O.; Uhl, K.; Throckmorton, D.C. Adverse Effects in Women: Implications for Drug Development and Regulatory Policies. Expert Rev. Clin. Pharmacol. 2011, 4, 453–466. [Google Scholar] [CrossRef] [PubMed]
- Fadiran, E.; Zhang, L. Effects of Sex Differences in the Pharmacokinetics of Drugs and Their Impact on the Safety of Medicines in Women. Med. Women 2015, 41–68. [Google Scholar] [CrossRef]
- Soldin, O.P.; Mattison, D.R. Sex Differences in Pharmacokinetics and Pharmacodynamics. Clin. Pharmacokinet. 2009, 48, 143–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gandhi, M.; Aweeka, F.; Greenblatt, R.M.; Blaschke, T.F. Sex Differences in Pharmacokinetics and Pharmacodynamics. Annu. Rev. Pharmacol. Toxicol. 2004, 44, 499–523. [Google Scholar] [CrossRef] [PubMed]
- Madla, C.M.; Gavins, F.K.H.; Merchant, H.A.; Orlu, M.; Murdan, S.; Basit, A.W. Let’s Talk about Sex: Differences in Drug Therapy in Males and Females. Adv. Drug Deliv. Rev. 2021, 175, 113804. [Google Scholar] [CrossRef]
- Krecic-Shepard, M.E.; Barnas, C.R.; Slimko, J.; Jones, M.P.; Schwartz, J.B. Gender-Specific Effects on Verapamil Pharmacokinetics and Pharmacodynamics in Humans. J. Clin. Pharmacol. 2000, 40, 219–230. [Google Scholar] [CrossRef]
- Zucker, I.; Prendergast, B.J. Sex Differences in Pharmacokinetics Predict Adverse Drug Reactions in Women. Biol. Sex Differ. 2020, 11, 32. [Google Scholar] [CrossRef]
- De Vries, S.T.; Denig, P.; Ekhart, C.; Burgers, J.S.; Kleefstra, N.; Mol, P.G.M.; van Puijenbroek, E.P. Sex Differences in Adverse Drug Reactions Reported to the National Pharmacovigilance Centre in the Netherlands: An Explorative Observational Study. Br. J. Clin. Pharmacol. 2019, 85, 1507–1515. [Google Scholar] [CrossRef] [Green Version]
- Hendriksen, L.C.; van der Linden, P.D.; Lagro-Janssen, A.L.M.; van den Bemt, P.M.L.A.; Siiskonen, S.J.; Teichert, M.; Kuiper, J.G.; Herings, R.M.C.; Stricker, B.H.; Visser, L.E. Sex Differences Associated with Adverse Drug Reactions Resulting in Hospital Admissions. Biol. Sex Differ. 2021, 12, 34. [Google Scholar] [CrossRef] [PubMed]
- Periasamy, S.; Poovathai, R.; Pondiyadanar, S. Influences of Gender on Postoperative Morphine Consumption. J. Clin. Diagn. Res. JCDR 2014, 8, GC04–GC07. [Google Scholar] [CrossRef] [PubMed]
- Mogil, J.S. Sex Differences in Pain and Pain Inhibition: Multiple Explanations of a Controversial Phenomenon. Nat. Rev. Neurosci. 2012, 13, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Fillingim, R.B.; King, C.D.; Ribeiro-dasilva, M.C.; Rahim-williams, B.; Riley, J.L.; Dentistry, C. Sex, Gender, and Pain: A Review of Recent Clinical and Experimental Findings. J. Pain 2009, 10, 447–485. [Google Scholar] [CrossRef] [Green Version]
- Riley, J.L.; Robinson, M.E.; Wise, E.A.; Myers, C.D.; Fillingim, R.B. Sex Differences in the Perception of Noxious Experimental Stimuli: A Meta-Analysis. Pain 1998, 74, 181–187. [Google Scholar] [CrossRef]
- Bartley, E.J.; Fillingim, R.B. Sex Differences in Pain: A Brief Review of Clinical and Experimental Findings. Br. J. Anaesth. 2013, 111, 52–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Racine, M.; Tousignant-Laflamme, Y.; Kloda, L.A.; Dion, D.; Dupuis, G.; Choinire, M. A Systematic Literature Review of 10 Years of Research on Sex/Gender and Experimental Pain Perception—Part 1: Are There Really Differences between Women and Men? Pain 2012, 153, 602–618. [Google Scholar] [CrossRef]
- Unruh, A.M. Gender Variations in Clinical Pain Experience. Pain 1996, 65, 123–167. [Google Scholar] [CrossRef]
- Berkley, K. Sex Differences in Pain. J. Am. Dent. Assoc. 1997, 143, 764–765. [Google Scholar] [CrossRef]
- Fillingim, R.B.; Maixner, W. Gender Differences in the Responses to Noxious Stimuli. Pain Forum 1995, 4, 209–221. [Google Scholar] [CrossRef]
- Packiasabapathy, S.; Sadhasivam, S. Gender, Genetics, and Analgesia: Understanding the Differences in Response to Pain Relief. J. Pain Res. 2018, 11, 2729–2739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Back, S.E.; Payne, R.L.; Wahlquist, A.H.; Carter, R.E.; Stroud, Z.; Haynes, L.; Hillhouse, M.; Brady, K.T.; Ling, W. Comparative Profiles of Men and Women with Opioid Dependence: Results from a National Multisite Effectiveness Trial. Am. J. Drug Alcohol Abuse 2011, 37, 313–323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greenspan, J.D.; Craft, R.M.; LeResche, L.; Arendt-Nielsen, L.; Berkley, K.J.; Fillingim, R.B.; Gold, M.S.; Holdcroft, A.; Lautenbacher, S.; Mayer, E.A.; et al. Studying Sex and Gender Differences in Pain and Analgesia: A Consensus Report. Pain 2007, 132, 26–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kest, B.; Sarton, E.; Dahan, A. Gender differences in opioid-mediated analgesia: Animal and human studies. Anesthesiology 2000, 93, 539–547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cicero, T.J.; Ennis, T.; Ogden, J.; Meyer, E.R. Gender Differences in the Reinforcing Properties of Morphine. Pharmacol. Biochem. Behav. 2000, 65, 91–96. [Google Scholar] [CrossRef]
- Craft, R.M. Sex Differences in Opioid Analgesia: “From Mouse to Man”. Clin. J. Pain 2003, 19, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Chia, Y.Y.; Chow, L.H.; Hung, C.C.; Liu, K.; Ger, L.P.; Wang, P.N. Gender and Pain upon Movement Are Associated with the Requirements for Postoperative Patient-Controlled Iv Analgesia: A Prospective Survey of 2298 Chinese Patients. Can. J. Anesth. 2002, 49, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Tsui, S.L.; Tong, W.N.; Irwin, M.; Ng, K.F.J.; Lo, J.R.; Chan, W.S.; Yang, J. The Efficacy, Applicability and Side-Effects of Postoperative Intravenous Patient-Controlled Morphine Analgesia: An Audit of 1233 Chinese Patients. Anaesth. Intensive Care 1996, 24, 658–664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burns, J.W.; Hodsman, N.B.A.; McLintock, T.T.C.; Gillies, G.W.A.; Kenny, G.N.C.; McArdle, C.S. The Influence of Patient Characteristics on the Requirements for Postoperative Analgesia. Anaesthesia 1989, 44, 2–6. [Google Scholar] [CrossRef]
- Sarton, E.; Olofsen, E.; Romberg, R.; Den Hartigh, J.; Kest, B.; Nieuwenhuijs, D.; Burm, A.; Teppema, L.; Dahan, A. Sex Differences in Morphine Analgesia: An Experimental Study in Healthy Volunteers. Anesthesiology 2000, 93, 1245–1254. [Google Scholar] [CrossRef]
- Aubrun, F.; Salvi, N.; Coriat, P.; Riou, B. Sex- and Age-Related Differences in Morphine Requirements for Postoperative Pain Relief. Anesthesiology 2005, 103, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Cepeda, M.S.; Carr, D.B. Women Experience More Pain and Require More Morphine Than Men to Achieve a Similar Degree of Analgesia. Anesth. Analg. 2003, 97, 1464–1468. [Google Scholar] [CrossRef] [PubMed]
- Comer, S.D.; Cooper, Z.D.; Kowalczyk, W.J.; Sullivan, M.A.; Evans, S.M.; Bisaga, A.M.; Vosburg, S.K. Evaluation of Potential Sex Differences in the Subjective and Analgesic Effects of Morphine in Normal, Healthy Volunteers. Psychopharmacology 2010, 208, 45–55. [Google Scholar] [CrossRef] [Green Version]
- Fillingim, R.B.; Ness, T.J.; Glover, T.L.; Campbell, C.M.; Hastie, B.A.; Price, D.D.; Staud, R. Morphine Responses and Experimental Pain: Sex Differences in Side Effects and Cardiovascular Responses but Not Analgesia. J. Pain 2005, 6, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Macintyre, P.E.; Jarvis, D.A. Age Is the Best Predictor of Postoperative Morphine Requirements. Pain 1996, 64, 357–364. [Google Scholar] [CrossRef]
- Cepeda, M.S.; Farrar, J.T.; Baumgarten, M.; Boston, R.; Carr, D.B.; Strom, B.L. Side Effects of Opioids during Short-Term Administration: Effect of Age, Gender, and Race. Clin. Pharmacol. Ther. 2003, 74, 102–112. [Google Scholar] [CrossRef]
- Stadler, M.; Bardiau, F.; Seidel, L.; Albert, A. Difference in Risk Factors for Postoperative Nausea and Vomiting. J. Am. Soc. Anesthesiol. 2003, 98, 46–52. [Google Scholar] [CrossRef]
- Myles, P.S.; McLeod, A.D.M.; Hunt, J.O.; Fletcher, H. Sex Differences in Speed of Emergence and Quality of Recovery after Anaesthesia: Cohort Study. Br. Med. J. 2001, 322, 710–711. [Google Scholar] [CrossRef] [Green Version]
- Zun, L.S.; Downey, L.V.A.; Gossman, W.; Rosenbaum, J.; Sussman, G. Gender Differences in Narcotic-Induced Emesis in the ED. Am. J. Emerg. Med. 2002, 20, 151–154. [Google Scholar] [CrossRef]
- Sarton, E.; Teppema, L.; Dahan, A. Sex Differences in Morphine-Induced Ventilatory Depression Reside within the Peripheral Chemoreflex Loop. J. Am. Soc. Anesthesiol. 1999, 90, 1329–1338. [Google Scholar] [CrossRef]
- Dahan, A.; Sarton, E.; Teppema, L.; Olievier, G. Sex-Related Differences in the Influence of Morphine on Ventilatory Control in Humans. Anesthesiology 1998, 88, 903–913. [Google Scholar] [CrossRef] [PubMed]
- Zacny, J.P. Morphine Responses in Humans: A Retrospective Analysis of Sex Differences. Drug Alcohol Depend. 2001, 63, 23–28. [Google Scholar] [CrossRef]
- Fillingim, R.B.; Gear, R.W. Sex Differences in Opioid Analgesia: Clinical and Experimental Findings. Eur. J. Pain 2004, 8, 413–425. [Google Scholar] [CrossRef]
- Niesters, M.; Dahan, A.; Kest, B.; Zacny, J.; Stijnen, T.; Aarts, L.; Sarton, E. Do Sex Differences Exist in Opioid Analgesia ? A Systematic Review and Meta-Analysis of Human Experimental and Clinical Studies. Pain 2010, 151, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Averitt, D.L.; Eidson, L.N.; Doyle, H.H.; Murphy, A.Z. Neuronal and Glial Factors Contributing to Sex Differences in Opioid Modulation of Pain. Neuropsychopharmacology 2019, 44, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.W.; Ho, I. Sex Differences in Opioid Analgesia and Addiction: Interactions among Opioid Receptors and Estrogen Receptors. Mol. Pain 2013, 9, 45. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gear, R.; Miaskowski, C.; Gordon, N. Kappa-Opioids Produce Significantly Greater Analgesia in Women than in Men. Nat. Med. 1996, 2, 1248–1250. [Google Scholar] [CrossRef]
- Fillingim, R.B.; Ness, T.J.; Glover, T.L.; Campbell, C.M.; Price, D.D.; Staud, R. Experimental Pain Models Reveal No Sex Differences in Pentazocine Analgesia in Humans. Anesthesiology 2004, 100, 1263–1270. [Google Scholar] [CrossRef]
- Mogil, J.S.; Wilson, S.G.; Chesler, E.J.; Rankin, A.L.; Nemmani, K.V.S.; Lariviere, W.R.; Groce, M.K.; Wallace, M.R.; Kaplan, L.; Staud, R.; et al. The Melanocortin-1 Receptor Gene Mediates Female-Specific Mechanisms of Analgesia in Mice and Humans. Proc. Natl. Acad. Sci. USA 2003, 100, 4867–4872. [Google Scholar] [CrossRef] [Green Version]
- Olofsen, E.; Romberg, R.; Bijl, H.; Mooren, R.; Engbers, F.; Kest, B.; Dahan, A. Alfentanil and Placebo Analgesia. Anesthesiology 2005, 103, 130–139. [Google Scholar] [CrossRef]
- Romberg, R.; Olofsen, E.; Sarton, E.; Den Hartigh, J.; Taschner, P.E.M.; Dahan, A. Pharmacokinetic-Pharmacodynamic Modeling of Morphine-6-Glucuronide-Induced Analgesia in Healthy Volunteers: Absence of Sex Differences. Anesthesiology 2004, 100, 120–133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dahan, A.; Kest, B.; Waxman, A.R.; Sarton, E. Sex-Specific Responses to Opiates: Animal and Human Studies. Anesth. Analg. 2008, 107, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Koons, A.L.; Greenberg, M.R.; Cannon, R.D.; Beauchamp, G.A. Women and the Experience of Pain and Opioid Use Disorder: A Literature-Based Commentary. Clin. Ther. 2018, 40, 190–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marcus, S.M.; Young, E.A.; Kerber, K.B.; Kornstein, S.; Farabaugh, A.H.; Mitchell, J.; Wisniewski, S.R.; Balasubramani, G.K.; Trivedi, M.H.; Rush, A.J. Gender Differences in Depression: Findings from the STAR*D Study. J. Affect. Disord. 2005, 87, 141–150. [Google Scholar] [CrossRef]
- LeGates, T.A.; Kvarta, M.D.; Thompson, S.M. Sex Differences in Antidepressant Efficacy. Neuropsychopharmacology 2019, 44, 140–154. [Google Scholar] [CrossRef] [Green Version]
- Keers, R.; Aitchison, K.J. Gender Differences in Antidepressant Drug Response. Int. Rev. Psychiatry 2010, 22, 485–500. [Google Scholar] [CrossRef]
- Marazziti, D.; Baroni, S.; Picchetti, M.; Piccinni, A.; Carlini, M.; Vatteroni, E.; Falaschi, V.; Lombardi, A.; Dell’osso, L. Pharmacokinetics and Pharmacodinamics of Psychotropic Drugs: Effect of Sex. CNS Spectr. 2013, 18, 118–127. [Google Scholar] [CrossRef]
- Kloiber, S.; Ising, M.; Reppermund, S.; Horstmann, S.; Dose, T.; Majer, M.; Zihl, J.; Pfister, H.; Unschuld, P.G.; Holsboer, F.; et al. Overweight and Obesity Affect Treatment Response in Major Depression. Biol. Psychiatry 2007, 62, 321–326. [Google Scholar] [CrossRef]
- Papakostas, G.I.; Petersen, T.; Iosifescu, D.V.; Burns, A.M.; Nierenberg, A.A.; Alpert, J.E.; Rosenbaum, J.F.; Fava, M. Obesity among Outpatients with Major Depressive Disorder. Int. J. Neuropsychopharmacol. 2005, 8, 59–63. [Google Scholar] [CrossRef]
- Khan, A.; Schwartz, K.A.; Kolts, R.L.; Brown, W.A. BMI, Sex, and Antidepressant Response. J. Affect. Disord. 2007, 99, 101–106. [Google Scholar] [CrossRef]
- Bigos, K.L.; Pollock, B.G.; Stankevich, B.A.; Bies, R.R. Sex Differences in the Pharmacokinetics and Pharmacodynamics of Antidepressants: An Updated Review. Gend. Med. 2009, 6, 522–543. [Google Scholar] [CrossRef]
- Smith, K.A.; Fairburn, C.G.; Cowen, P.J. Relapse of Depression after Vapid Depletion of Tryptophan. Lancet 1997, 349, 915–919. [Google Scholar] [CrossRef]
- Maes, M.; Jacobs, M.P.; Suy, E.; Minner, B.; Leclercq, C.; Christiaens, F.; Raus, J. Suppressant Effects of Dexamethasone on the Availability of Plasma L-tryptophan and Tyrosine in Healthy Controls and in Depressed Patients. Acta Psychiatr. Scand. 1990, 81, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Bano, S.; Akhter, S.; Afridi, M.I. Gender Based Response to Fluoxetine Hydrochloride Medication in Endogenous Depression. J. Coll. Physicians Surg. Pak. 2004, 14, 161–165. [Google Scholar] [PubMed]
- Ejsing, T.B.; Linnet, K. Influence of P-Glycoprotein Inhibition on the Distribution of the Tricyclic Antidepressant Nortriptyline over the Blood-Brain Barrier. Hum. Psychopharmacol. 2005, 20, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Uhr, M.; Grauer, M.T.; Holsboer, F. Differential Enhancement of Antidepressant Penetration into the Brain in Mice with Abcb1ab (Mdr1ab) P-Glycoprotein Gene Disruption. Biol. Psychiatry 2003, 54, 840–846. [Google Scholar] [CrossRef]
- Damoiseaux, V.A.; Proost, J.H.; Jiawan, V.C.R.; Melgert, B.N. Sex Differences in the Pharmacokinetics of Antidepressants: Influence of Female Sex Hormones and Oral Contraceptives. Clin. Pharmacokinet. 2014, 53, 509–519. [Google Scholar] [CrossRef]
- Dunlop, B.W.; Davis, P.G. Combination Treatment with Benzodiazepines and SSRIs for Comorbid Anxiety and Depression: A Review. Prim. Care Companion J. Clin. Psychiatry 2008, 10, 222–228. [Google Scholar] [CrossRef]
- Smith, W.T.; Londborg, P.D.; Glaudin, V.; Painter, J.R. Short Term Treatment of Fluoxetine with Clonazepam in the Treatment of Depression: A Double-Blind Study. Am. J. Psychiatry 1998, 155, 1339–1351. [Google Scholar] [CrossRef]
- Greenblatt, D.J.; Preskorn, S.H.; Cotreau, M.M.; Horst, W.D.; Harmatz, J.S. Fluoxetine Impairs Clearance of Alprazolam but Not of Clonazepam. Clin. Pharmacol. Ther. 1992, 52, 479–486. [Google Scholar] [CrossRef]
- Lemberger, L.; Rowe, H.; Bosomworth, J.C.; Tenbarge, J.B.; Bergstrom, R.F. The Effect of Fluoxetine on the Pharmacokinetics and Psychomotor Responses of Diazepam. Clin. Pharmacol. Ther. 1988, 43, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Spina, E.; Scordo, M.G.; D’Arrigo, C. Metabolic Drug Interactions with New Psychotropic Agents. Fundam. Clin. Pharmacol. 2003, 17, 517–538. [Google Scholar] [CrossRef] [PubMed]
- Frank, E.; Carpenter, L. Sex Differences in Recurent Depression: Are There Any That Are Significant? Am. J. Psychiatry 1988, 145, 41–45. [Google Scholar] [PubMed]
- Kim, J.; Kim, S.; Stewart, R.; Kim, S.; Yoon, Y.; Jung, S. Predictors of 12-Week Remission in a Nationwide Cohort of People with Depressive Disorders: The CRESCEND Study. J. Clin. Psychiatry 2011, 55, 391–393. [Google Scholar] [CrossRef]
- Berlanga, C.; Flores-Ramos, M. Different Gender Response to Serotonergic and Noradrenergic Antidepressants. A Comparative Study of the Efficacy of Citalopram and Reboxetine. J. Affect. Disord. 2006, 95, 119–123. [Google Scholar] [CrossRef]
- Trivedi, M.H.; Rush, A.J.; Wisniewski, S.R.; Nierenberg, A.A.; Warden, D.; Ritz, L.; Norquist, G.; Howland, R.H.; Lebowitz, B.; McGrath, P.J.; et al. Evaluation of Outcomes with Citalopram for Depression Using Measurement-Based Care in STAR*D: Implications for Clinical Practice. Am. J. Psychiatry 2006, 163, 28–40. [Google Scholar] [CrossRef]
- Martényi, F.; Dossenbach, M.; Mraz, K.; Metcalfe, S. Gender Differences in the Efficacy of Fluoxetine and Maprotiline in Depressed Patients: A Double-Blind Trial of Antidepressants with Serotonergic or Norepinephrinergic Reuptake Inhibition Profile. Eur. Neuropsychopharmacol. 2001, 11, 227–232. [Google Scholar] [CrossRef]
- Kornstein, S.G.; Schatzberg, A.F.; Thase, M.E.; Yonkers, K.A.; McCullough, J.P.; Keitner, G.I.; Gelenberg, A.J.; Davis, S.M.; Harrison, W.M.; Keller, M.B. Gender Differences in Treatment Response to Sertraline versus Imipramine in Chronic Depression. Am. J. Psychiatry 2000, 157, 1445–1452. [Google Scholar] [CrossRef]
- Baca, E.; Garcia-Garcia, M.; Porras-Chavarino, A. Gender Differences in Treatment Response to Sertraline versus Imipramine in Patients with Nonmelancholic Depressive Disorders. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2004, 28, 57–65. [Google Scholar] [CrossRef]
- Khan, A.; Brodhead, A.E.; Schwartz, K.A.; Kolts, R.L.; Brown, W.A. Sex Differences in Antidepressant Response in Recent Antidepressant Clinical Trials. J. Clin. Psychopharmacol. 2005, 25, 318–324. [Google Scholar] [CrossRef]
- Pande, A.C.; Birkett, M.; Fechner-Bates, S.; Haskett, R.F.; Greden, J.F. Fluoxetine versus Phenelzine in Atypical Depression. Biol. Psychiatry 1996, 40, 1017–1020. [Google Scholar] [CrossRef]
- Thase, M.E.; Entsuah, R.; Cantillon, M.; Kornstein, S.G. Relative Antidepressant Efficacy of Venlafaxine and SSRIs: Sex-Age Interactions. J. Women’s Health 2005, 14, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Wohlfarth, T.; Storosum, J.G.; Elferink, A.J.A.; Van Zwieten, B.J.; Fouwels, A.; Van Den Brink, W. Response to Tricyclic Antidepressants: Independent of Gender? Am. J. Psychiatry 2004, 161, 370–372. [Google Scholar] [CrossRef] [PubMed]
- Quitkin, F.M.; Stewart, J.W.; McGrath, P.J. Are There Differences between Women’s and Men’s Antidepressant Responses? Prim. Care Companion J. Clin. Psychiatry 2002, 4, 205. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, M.G.; Steyerberg, E.W.; Stage, K.B.; Passchier, J.; Kragh-soerensen, P. Are Gender Differences Important for the Clinical Effects of Antidepressants? Am. J. Psychiatry 2003, 160, 1643–1650. [Google Scholar] [CrossRef] [Green Version]
- Parker, G.; Parker, K.; Austin, M.P.; Mitchell, P.; Brotchie, H. Gender Differences in Response to Differing Antidepressant Drug Classes: Two Negative Studies. Psychol. Med. 2003, 33, 1473–1477. [Google Scholar] [CrossRef]
- Marsh, W.; Deligiannidis, K. Sex-Related Differences in Antidepressant Response: When to Adjust Treatment. Curr. Psychiatr. 2010, 9, 25–30. [Google Scholar]
- Coppen, A.; Bailey, J. Enhancement of the Antidepressant Action of FLuoxetine by Folic Acid: A Randomised, Placebo Controlled Trial. J. Affect. Disord. 2000, 60, 121–130. [Google Scholar] [CrossRef]
- Altshuler, L.L.; Bauer, M.; Frye, M.A.; Gitlin, M.J.; Mintz, J.; Szuba, M.P.; Leight, K.L.; Whybrow, P.C. Does Thyroid Supplementation Accelerate Tricyclic Antidepressant Response? A Review and Meta-Analysis of the Literature. Am. J. Psychiatry 2001, 158, 1617–1622. [Google Scholar] [CrossRef]
- Zemishlany, Z.; Weizman, A. The Impact of Mental Illness on Sexual Dysfunction. Adv. Psychosom. Med. 2008, 29, 89–106. [Google Scholar] [CrossRef]
- Clayton, A.; Keller, A.; McGarvey, E.L. Burden of Phase-Specific Sexual Dysfunction with SSRIs. J. Affect. Disord. 2006, 91, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, J.M. SSRI Antidepressant Medications: Adverse Effects and Tolerability. Prim. Care Companion J. Clin. Psychiatry 2001, 3, 22. [Google Scholar] [CrossRef] [PubMed]
- Nash, J.; Nutt, D. Antidepressants. Psychiatry 2007, 6, 289–294. [Google Scholar] [CrossRef]
- Montgomery, S.A.; Judge, R. Treatment of Depression with Associated Anxiety: Comparison of Tricyclic Antidepressant and Selective Serotonin Reuptake Inhibitors. Acta Psychiatr. Scand. Suppl. 2000, 101, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Peretti, S.; Judge, R.; Hindmarch, I. Safety and Tolerability Considerations: Tricyclic Antidepressants vs. Selective Serotonin Reuptake Inhibitors. Acta Psychiatr. Scand. 2000, 101, 17–25. [Google Scholar] [CrossRef]
- Samba Reddy, D. Sex Differences in the Anticonvulsant Activity of Neurosteroids. J. Neurosci. Res. 2017, 95, 661–670. [Google Scholar] [CrossRef] [Green Version]
- Verrotti, A.; Greco, R.; Giannuzzi, R.; Chiarelli, F.; Latini, G. Old and New Antiepileptic Drugs for the Treatment of Idiopathic Generalized Epilepsies. Curr. Clin. Pharmacol. 2008, 2, 249–259. [Google Scholar] [CrossRef]
- Herzog, A.G. Hormonal Therapies: Progesterone. Neurotherapeutics 2009, 6, 383–391. [Google Scholar] [CrossRef] [Green Version]
- Younus, I.; Reddy, D.S. Seizure Facilitating Activity of the Oral Contraceptive Ethinyl Estradiol. Epilepsy Res. 2016, 121, 29–32. [Google Scholar] [CrossRef] [Green Version]
- Herzog, A.G. Differential Impact of Antiepileptic Drugs on the Effects of Contraceptive Methods on Seizures: Interim Findings of the Epilepsy Birth Control Registry. Seizure 2015, 28, 71–75. [Google Scholar] [CrossRef] [Green Version]
- Perucca, E.; Battino, D.; Tomson, T. Gender Issues in Antiepileptic Drug Treatment. Neurobiol. Dis. 2014, 72, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Johannessen Landmark, C.; Johannessen, S.I.; Tomson, T. Host Factors Affecting Antiepileptic Drug Delivery-Pharmacokinetic Variability. Adv. Drug Deliv. Rev. 2012, 64, 896–910. [Google Scholar] [CrossRef] [PubMed]
- Herzog, A.G.; Blum, A.S.; Farina, E.L.; Maestri, X.E.; Newman, J.; Garcia, E.; Krishnamurthy, K.B.; Hoch, D.B.; Replansky, S.; Fowler, K.M.; et al. Valproate and Lamotrigine Level Variation with Menstrual Cycle Phase and Oral Contraceptive Use. Neurology 2009, 72, 911–914. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, M.; Guillebaud, J. Contraception for Women Taking Antiepileptic Drugs. J. Fam. Plan. Reprod. Health Care 2011, 37, 60. [Google Scholar] [CrossRef] [Green Version]
- Patsalos, P.N. Drug Interactions with the Newer Antiepileptic Drugs (AEDs)—Part 2: Pharmacokinetic and Pharmacodynamic Interactions between AEDs and Drugs Used to Treat Non-Epilepsy Disorders. Clin. Pharmacokinet. 2013, 52, 1045–1061. [Google Scholar] [CrossRef]
- Boruban, M.C. Tamoxifen Inhibits Cytochrome P450 2C9. J. Chemother. 2006, 18, 421–424. [Google Scholar] [CrossRef]
- Cialis 2.5 mg, 5 mg, 10 mg & 20 mg Film-Coated Tablets—Summary of Product Characteristics (SmPC)—(Emc). Available online: https://www.medicines.org.uk/emc/medicine/11363#INTERACTIONS (accessed on 20 January 2021).
- Tomson, T.; Landmark, C.J.; Battino, D. Antiepileptic Drug Treatment in Pregnancy: Changes in Drug Disposition and Their Clinical Implications. Epilepsia 2013, 54, 405–414. [Google Scholar] [CrossRef]
- Reimers, A.; Helde, G.; Bråthen, G.; Brodtkorb, E. Lamotrigine and Its N2-Glucuronide during Pregnancy: The Significance of Renal Clearance and Estradiol. Epilepsy Res. 2011, 94, 198–205. [Google Scholar] [CrossRef]
- Petrenaite, V.; Sabers, A.; Hansen-Schwartz, J. Seizure Deterioration in Women Treated with Oxcarbazepine during Pregnancy. Epilepsy Res. 2009, 84, 245–249. [Google Scholar] [CrossRef]
- Tomson, T.; Palm, R.; Källén, K.; Ben-Menachem, E.; Söderfeldt, B.; Danielsson, B.; Johansson, R.; Luef, G.; Öhman, I. Pharmacokinetics of Levetiracetam during Pregnancy, Delivery, in the Neonatal Period, and Lactation. Epilepsia 2007, 48, 1111–1116. [Google Scholar] [CrossRef]
- Verrotti, A.; D’Egidio, C.; Mohn, A.; Coppola, G.; Parisi, P.; Chiarelli, F. Antiepileptic Drugs, Sex Hormones, and PCOS. Epilepsia 2011, 52, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Brodie, M.J.; Mintzer, S.; Pack, A.M.; Gidal, B.E.; Vecht, C.J.; Schmidt, D. Enzyme Induction with Antiepileptic Drugs: Cause for Concern? Epilepsia 2013, 54, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Isojärvi, J.I.; Taubøll, E.; Herzog, A.G. Effect of antiepileptic drugs on reproductive endocrine function in individuals with epilepsy. CNS Drugs 2005, 19, 207–223. [Google Scholar] [CrossRef] [PubMed]
- Herzog, A.G.; Drislane, F.W.; Schomer, D.L.; Pennell, P.B.; Bromfield, E.B.; Dworetzky, B.A.; Farina, E.L.; Frye, C.A. Differential Effects of Antiepileptic Drugs on Neuroactive Steroids in Men with Epilepsy. Epilepsia 2006, 47, 1945–1948. [Google Scholar] [CrossRef]
- Bauer, J.; Isojärvi, J.I.T.; Herzog, A.G.; Reuber, M.; Polson, D.; Taubøll, E.; Genton, P.; Van der Ven, H.; Roesing, B.; Luef, G.J.; et al. Reproductive Dysfunction in Women with Epilepsy: Recommendations for Evaluation and Management. J. Neurol. Neurosurg. Psychiatry 2002, 73, 121–125. [Google Scholar] [CrossRef]
- Rubin, L.H.; Haas, G.L.; Keshavan, M.S.; Sweeney, J.A.; Maki, P.M. Sex Difference in Cognitive Response to Antipsychotic Treatment in First Episode Schizophrenia. Neuropsychopharmacology 2008, 33, 290–297. [Google Scholar] [CrossRef]
- Usall, J.; Araya, S.; Ochoa, S.; Busquets, E.; Gost, A.; Márquez, M. Gender Differences in a Sample of Schizophrenic Outpatients. Compr. Psychiatry 2001, 42, 301–305. [Google Scholar] [CrossRef]
- Häfner, H. Gender Differences in Schizophrenia. Psychoneuroendocrinology 2003, 28, 17–54. [Google Scholar] [CrossRef]
- Seeman, M.V. Gender Differences in the Prescribing of Antipsychotic Drugs. Am. J. Psychiatry 2004, 161, 1324–1333. [Google Scholar] [CrossRef]
- Crawford, M.B.; DeLisi, L.E. Issues Related to Sex Differences in Antipsychotic Treatment. Curr. Opin. Psychiatry 2016, 29, 211–217. [Google Scholar] [CrossRef]
- Smith, S. Gender Differences in Antipsychotic Prescribing. Int. Rev. Psychiatry 2010, 22, 472–484. [Google Scholar] [CrossRef] [PubMed]
- Zyprexa—Summary of Product Characteristics. Available online: https://www.ema.europa.eu/en/documents/product-information/zyprexa-epar-product-information_en.pdf (accessed on 1 March 2021).
- Sainz, J.; Prieto, C.; Crespo-facorro, B. Sex Differences in Gene Expression Related to Antipsychotic Induced Weight Gain. PLoS ONE 2019, 14, e0215477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lange, B.; Mueller, J.K.; Leweke, F.M.; Bumb, J.M. How Gender Affects the Pharmacotherapeutic Approach to Treating Psychosis—A Systematic Review. Expert Opin. Pharmacother. 2017, 18, 351–362. [Google Scholar] [CrossRef] [PubMed]
- Seeman, M.V. Men and Women Respond Differently to Antipsychotic Drugs. Neuropharmacology 2020, 163, 107631. [Google Scholar] [CrossRef]
- Usall, J.; Suarez, D.; Haro, J.M. Gender Differences in Response to Antipsychotic Treatment in Outpatients with Schizophrenia. Psychiatry Res. 2007, 153, 225–231. [Google Scholar] [CrossRef]
- Salokangas, R.K.R. Gender and the Use of Neuroleptics in Schizophrenia. Schizophr. Res. 2004, 66, 41–49. [Google Scholar] [CrossRef]
- Melkersson, K.I.; Hulting, A.L.; Rane, A.J. Dose Requirement and Prolactin Elevation of Antipsychotics in Male and Female Patients with Schizophrenia or Related Psychoses. Br. J. Clin. Pharmacol. 2001, 51, 317–324. [Google Scholar] [CrossRef] [Green Version]
- Walther, S.; Moggi, F.; Horn, H.; Moskvitin, K.; Abderhalden, C.; Maier, N.; Strik, W.; Müller, T.J. Rapid Tranquilization of Severely Agitated Patients with Schizophrenia Spectrum Disorders: A Naturalistic, Rater-Blinded, Randomized, Controlled Study with Oral Haloperidol, Risperidone, and Olanzapine. J. Clin. Psychopharmacol. 2014, 34, 124–128. [Google Scholar] [CrossRef] [Green Version]
- Haslemo, T.; Eikeseth, P.H.; Tanum, L.; Molden, E.; Refsum, H. The Effect of Variable Cigarette Consumption on the Interaction with Clozapine and Olanzapine. Eur. J. Clin. Pharmacol. 2006, 62, 1049–1053. [Google Scholar] [CrossRef] [Green Version]
- Zullino, D.F.; Delessert, D.; Eap, C.B.; Preisig, M.; Baumann, P. Tobacco and Cannabis Smoking Cessation Can Lead to Intoxication with Clozapine or Olanzapine. Int. Clin. Psychopharmacol. 2002, 17, 141–143. [Google Scholar] [CrossRef]
- Lowe, E.J.; Ackman, M.L. Impact of Tobacco Smoking Cessation on Stable Clozapine or Olanzapine Treatment. Ann. Pharmacother. 2010, 44, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Kraal, A.Z.; Ward, K.M.; Ellingrod, V.L. Sex Differences in Antipsychotic Related Metabolic Functioning in Schizophrenia Spectrum Disorders. Psychopharmacol. Bull. 2017, 47, 8–21. [Google Scholar] [PubMed]
- Russell, J.M.; Mackell, J.A. Bodyweight Gain Associated with Atypical Antipsychotics: Epidemiology and Therapeutic Implications. CNS Drugs 2001, 15, 537–551. [Google Scholar] [CrossRef] [PubMed]
- Homel, P.; Casey, D.; Allison, D.B. Changes in Body Mass Index for Individuals with and without Schizophrenia, 1987-1996. Schizophr. Res. 2002, 55, 277–284. [Google Scholar] [CrossRef]
- Stroup, T.S.; Gray, N. Management of Common Adverse Effects of Antipsychotic Medications. World Psychiatry 2018, 17, 341–356. [Google Scholar] [CrossRef] [PubMed]
- Aichhorn, W.; Whitworth, A.B.; Weiss, E.M.; Marksteiner, J. Second-Generation Antipsychotics: Is There Evidence for Sex Differences in Pharmacokinetic and Adverse Effect Profiles? Drug Saf. 2006, 29, 587–598. [Google Scholar] [CrossRef]
- Klibanski, A.; Neer, R.; Beitins, I.; Ridgway, C. Decreased Bone Density in Hyperprolactinemic Women. N. Engl. J. Med. 1980, 303, 1511–1514. [Google Scholar] [CrossRef]
- Hamner, M.B.; Arana, G.W. Hyperprolactinaemia in Antipsychotic-Treated Patients: Guidelines for Avoidance and Management. CNS Drugs 1998, 10, 209–222. [Google Scholar] [CrossRef]
- Debusk, R.; Drory, Y.; Goldstein, I.; Jackson, G.; Kaul, S.; Kimmel, S.E.; Kostis, J.B.; Kloner, R.A.; Lakin, M.; Meston, C.M.; et al. Management of Sexual Dysfunction in Patients with Cardiovascular Disease: Recommendations of the Princeton Consensus Panel. Am. J. Cardiol. 2000, 86, 62–68. [Google Scholar] [CrossRef]
- Knegtering, H.; Van Der Moolen, A.E.G.M.; Castelein, S.; Kluiter, H.; Van Den Bosch, R.J. What Are the Effects of Antipsychotics on Sexual Dysfunctions and Endocrine Functioning? Psychoneuroendocrinology 2003, 28, 109–123. [Google Scholar] [CrossRef]
- Mitchell, J.E.; Popkin, M.K. Antipsychotic Drug Therapy and Sexual Dysfunction in Men. Am. J. Psychiatry 1982, 139, 633–637. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romanescu, M.; Buda, V.; Lombrea, A.; Andor, M.; Ledeti, I.; Suciu, M.; Danciu, C.; Dehelean, C.A.; Dehelean, L. Sex-Related Differences in Pharmacological Response to CNS Drugs: A Narrative Review. J. Pers. Med. 2022, 12, 907. https://doi.org/10.3390/jpm12060907
Romanescu M, Buda V, Lombrea A, Andor M, Ledeti I, Suciu M, Danciu C, Dehelean CA, Dehelean L. Sex-Related Differences in Pharmacological Response to CNS Drugs: A Narrative Review. Journal of Personalized Medicine. 2022; 12(6):907. https://doi.org/10.3390/jpm12060907
Chicago/Turabian StyleRomanescu, Mirabela, Valentina Buda, Adelina Lombrea, Minodora Andor, Ionut Ledeti, Maria Suciu, Corina Danciu, Cristina Adriana Dehelean, and Liana Dehelean. 2022. "Sex-Related Differences in Pharmacological Response to CNS Drugs: A Narrative Review" Journal of Personalized Medicine 12, no. 6: 907. https://doi.org/10.3390/jpm12060907
APA StyleRomanescu, M., Buda, V., Lombrea, A., Andor, M., Ledeti, I., Suciu, M., Danciu, C., Dehelean, C. A., & Dehelean, L. (2022). Sex-Related Differences in Pharmacological Response to CNS Drugs: A Narrative Review. Journal of Personalized Medicine, 12(6), 907. https://doi.org/10.3390/jpm12060907











