Smoking Obstructive Sleep Apnea: Arguments for a Distinctive Phenotype and a Personalized Intervention
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Study Variables
2.3. Statistical Analyses
3. Results
3.1. Demographic and Anthropometric Characteristics
3.2. Analysis of SOSA-Related Symptoms
3.3. Analysis of Comorbidities in SOSA
3.4. Analysis of the NRP Study in SOSA
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Huttunen, R.; Heikkinen, T.; Syrjänen, J. Smoking and the outcome of infection. J. Intern. Med. 2010, 269, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Li, K.-J.; Chen, Z.-L.; Huang, Y.; Zhang, R.; Luan, X.-Q.; Lei, T.-T.; Chen, L. Dysbiosis of lower respiratory tract microbiome are associated with inflammation and microbial function variety. Respir. Res. 2019, 20, 272. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, J.; Malvezzi, M.; Negri, E.; La Vecchia, C.; Boffetta, P. Risk factors for lung cancer worldwide. Eur. Respir. J. 2016, 48, 889–902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hsu, W.-Y.; Chiu, N.-Y.; Chang, C.-C.; Chang, T.-G.; Lane, H.-Y. The association between cigarette smoking and obstructive sleep apnea. Tob. Induc. Dis. 2019, 17, 27. [Google Scholar] [CrossRef]
- Franklin, K.A.; Lindberg, E. Obstructive sleep apnea is a common disorder in the population—a review on the epidemiology of sleep apnea. J. Thorac. Dis. 2015, 7, 1311–1322. [Google Scholar] [CrossRef] [PubMed]
- Pezzuto, A.; Carico, E. Effectiveness of smoking cessation in smokers with COPD and nocturnal oxygen desaturation: Functional analysis. Clin. Respir. J. 2020, 14, 29–34. [Google Scholar] [CrossRef]
- Collop, N.A.; Anderson, W.M.; Boehlecke, B.; Claman, D.; Goldberg, R.; Gottlieb, D.J.; Hudgel, D.; Sateia, M.; Schwab, R. Clinical guidelines for the use of unattended portable monitors in the diagnosis of obstructive sleep apnea in adult patients. Portable monitoring task force of the American academy of sleep medicine. J. Clin. Sleep Med. 2007, 3, 737–747. [Google Scholar]
- Epstein, L.J.; Kristo, D.; Strollo, P.J., Jr.; Friedman, N.; Malhotra, A.; Patil, S.P.; Ramar, K.; Rogers, R.; Schwab, R.J.; Weaver, E.M.; et al. Clinical Guideline for the Evaluation, Management and Long-term Care of Obstructive Sleep Apnea in Adults. J. Clin. Sleep Med. 2009, 5, 263–276. [Google Scholar]
- Ruehland, B.W.R.; Rochford, B.P.D.; O’Donoghue, F.J.; Pierce, R.J.; Singh, B.P.; Thornton, A.T. The New AASM Criteria for Scoring Hypopneas: Impact on the Apnea Hypopnea Index. Sleep 2009, 32, 150–157. [Google Scholar] [CrossRef] [Green Version]
- Johns, M.; Hocking, B. Daytime Sleepiness and Sleep Habits of Australian Workers. Sleep 1997, 20, 844–847. [Google Scholar] [CrossRef]
- Berry, R.B.; Gamaldo, C.E.; Harding, S.M.; Lloyd, R.M.; Marcus, C.L.; Vaughn, B.V.; For the American Academy of Sleep Medicine. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; Version 2.0.3; American Academy of Sleep Medicine: Darien, IL, USA, 2014; Available online: https://aasm.org/resources/pdf/scoring-manual-preface.pdf (accessed on 30 December 2020).
- Kulkas, A.; Tiihonen, P.; Julkunen, P.; Mervaala, E.; Töyräs, J. Novel parameters indicate significant differences in severity of obstructive sleep apnea with patients having similar apnea–hypopnea index. Med. Biol. Eng. Comput. 2013, 51, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Leppänen, T.; Töyräs, J.; Mervaala, E.; Penzel, T.; Kulkas, A. Severity of individual obstruction events increases with age in patients with obstructive sleep apnea. Sleep Med. 2017, 37, 32–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fietze, I.; Laharnar, N.; Obst, A.; Ewert, R.; Felix, S.B.; Garcia, C.; Gläser, S.; Glos, M.; Schmidt, C.O.; Stubbe, B.; et al. Prevalence and association analysis of obstructive sleep apnea with gender and age differences-Results of SHIP-Trend. J. Sleep Res. 2019, 28, e12770. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, L.; Trisko, B.M.; Lima-Rosa, F.L.; Jackson, J.; Lund-Palau, H.; Yamaguchi, M.; Breen, E.C. Cigarette smoke directly impairs skeletal muscle function through capillary regression and altered myofibre calcium kinetics in mice. J. Physiol. 2018, 596, 2901–2916. [Google Scholar] [CrossRef]
- Graff-Iversen, S.; Hewitt, S.; Forsén, L.; Grøtvedt, L.; Ariansen, I. Associations of tobacco smoking with body mass distribution; a population-based study of 65,875 men and women in midlife. BMC Public Health 2019, 19, 1439. [Google Scholar] [CrossRef]
- Kim, J.; Shim, K.W.; Yoon, Y.S.; Lee, S.Y.; Kim, S.S.; Oh, S.W. Cigarette Smoking Increases Abdominal and Visceral Obesity but Not Overall Fatness: An Observational Study. PLoS ONE 2012, 7, e45815. [Google Scholar] [CrossRef] [Green Version]
- Carreras-Torres, R.; Johansson, M.; Haycock, P.C.; Relton, C.L.; Smith, G.D.; Brennan, P.; Martin, R.M. Role of obesity in smoking behaviour: Mendelian randomisation study in UK Biobank. BMJ 2018, 361, k1767. [Google Scholar] [CrossRef] [Green Version]
- Lv, J.; Chen, W.; Sun, D.; Li, S.; Millwood, I.Y.; Smith, M.; Guo, Y.; Bian, Z.; Yu, C.; Zhou, H.; et al. Gender-Specific Association between Tobacco Smoking and Central Obesity among 0.5 Million Chinese People: The China Kadoorie Biobank Study. PLoS ONE 2015, 10, e0124586. [Google Scholar] [CrossRef]
- Lanas, F.; Bazzano, L.; Rubinstein, A.; Calandrelli, M.; Chen, C.-S.; Elorriaga, N.; Gutierrez, L.; Manfredi, J.A.; Seron, P.; Mores, N.; et al. Prevalence, Distributions and Determinants of Obesity and Central Obesity in the Southern Cone of America. PLoS ONE 2016, 11, e0163727. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.; Xu, H.; Qian, Y.; Liu, Y.; Zou, J.; Yi, H.; Guan, J.; Yin, S. Abdominal Obesity Is More Strongly Correlated with Obstructive Sleep Apnea than General Obesity in China: Results from Two Separated Observational and Longitudinal Studies. Obes. Surg. 2019, 29, 2535–2547. [Google Scholar] [CrossRef]
- Isono, S. Obesity and obstructive sleep apnoea: Mechanisms for increased collapsibility of the passive pharyngeal airway. Respirology 2011, 17, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, C.D.; Wang, S.H.; Manuel, A.R.; Keenan, B.T.; McIntyre, A.G.; Schwab, R.J.; Stradling, J.R. Relationships between MRI fat distributions and sleep apnea and obesity hypoventilation syndrome in very obese patients. Sleep Breath. 2018, 22, 673–681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roşulescu, R. The Role of Cortisol in Neurocognitive Performance in Obstructive Sleep Apnea Patients. Acta Endocrinol. 2016, 12, 375–377. [Google Scholar] [CrossRef]
- Ghiciuc, C.M.; Dima-Cozma, L.C.; Bercea, R.M.; Lupusoru, C.E.; Mihaescu, T.; Cozma, S.; Patacchioli, F.R. Imbalance in the diurnal salivary testosterone/cortisol ratio in men with severe obstructive sleep apnea: An observational study. Braz. J. Otorhinolaryngol. 2016, 82, 529–535. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Svartberg, J.; Von Mühlen, D.; Sundsfjord, J.; Jorde, R. Waist Circumference and Testosterone Levels in Community Dwelling Men. The Tromsø Study. Eur. J. Epidemiol. 2003, 19, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.; Colodner, R.; Masalha, R.; Haimov, I. The Relationship Between Tobacco Smoking, Cortisol Secretion, and Sleep Continuity. Subst. Use Misuse 2019, 54, 1705–1714. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.; Katz, E.G.; Huxley, R. Associations between gender, age and waist circumference. Eur. J. Clin. Nutr. 2009, 64, 6–15. [Google Scholar] [CrossRef] [Green Version]
- Shao, C.; Qi, H.; Fang, Q.; Tu, J.; Li, Q.; Wang, L. Smoking history and its relationship with comorbidities in patients with obstructive sleep apnea. Tob. Induc. Dis. 2020, 18, 56. [Google Scholar] [CrossRef]
- Owens, R.L.; Malhotra, A. Sleep-disordered breathing and COPD: The overlap syndrome. Respir. Care 2010, 55, 1333–1346. [Google Scholar]
- Purani, H.; Friedrichsen, S.; Allen, A.M. Sleep quality in cigarette smokers: Associations with smoking-related outcomes and exercise. Addict. Behav. 2019, 90, 71–76. [Google Scholar] [CrossRef]
- Casasola, G.G.; Tashkin, N.P.; Álvarez-Sala, J.L.; Marques, A.; Sánchez-Alarcos, J.M.; Espinos, D. Cigarette Smoking Behavior and Respiratory Alterations During Sleep in a Healthy Population. Sleep Breath. 2002, 6, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Toraldo, D.M.; Minelli, M.; De Nuccio, F.; Nicolardi, G. Chronic obstructive pulmonary disease phenotype desaturator with hypoxic vascular remodelling and pulmonary hypertension obtained by cluster analysis. Multidiscip. Respir. Med. 2012, 7, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonsignore, M.R.; Saaresranta, T.; Riha, R.L. Sex differences in obstructive sleep apnoea. Eur. Respir. Rev. 2019, 28, 190030. [Google Scholar] [PubMed]
- Peppard, P.E.; Young, T.; Palta, M.; Dempsey, J.; Skatrud, J. Longitudinal Study of Moderate Weight Change and Sleep-Disordered Breathing. JAMA 2000, 284, 3015–3021. [Google Scholar] [CrossRef] [Green Version]
- Murphy, C.M.; Rohsenow, D.J.; Johnson, K.C.; Wing, R.R. Smoking and weight loss among smokers with overweight and obesity in Look AHEAD. Health Psychol. 2018, 37, 399–406. [Google Scholar] [CrossRef]
- Tønnesen, P.; Carrozzi, L.; Fagerstrom, K.O.; Gratziou, C.; Jiménez-Ruiz, C.; Nardini, S.; Viegi, G.; Lazzaro, C.; Campell, I.A.; Dagli, E.; et al. Smoking cessation in patients with respiratory diseases: A high priority, integral component of therapy. Eur. Respir. J. 2006, 29, 390–417. [Google Scholar] [CrossRef] [Green Version]
- Hudgel, D.W.; Patel, S.R.; Ahasic, A.M.; Bartlett, S.J.; Bessesen, D.H.; Coaker, M.A.; Fiander, P.M.; Grunstein, R.R.; Gurubhagavatula, I.; Kapur, V.K.; et al. The Role of Weight Management in the Treatment of Adult Obstructive Sleep Apnea. An Official American Thoracic Society Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2018, 198, e70–e87. [Google Scholar] [CrossRef]



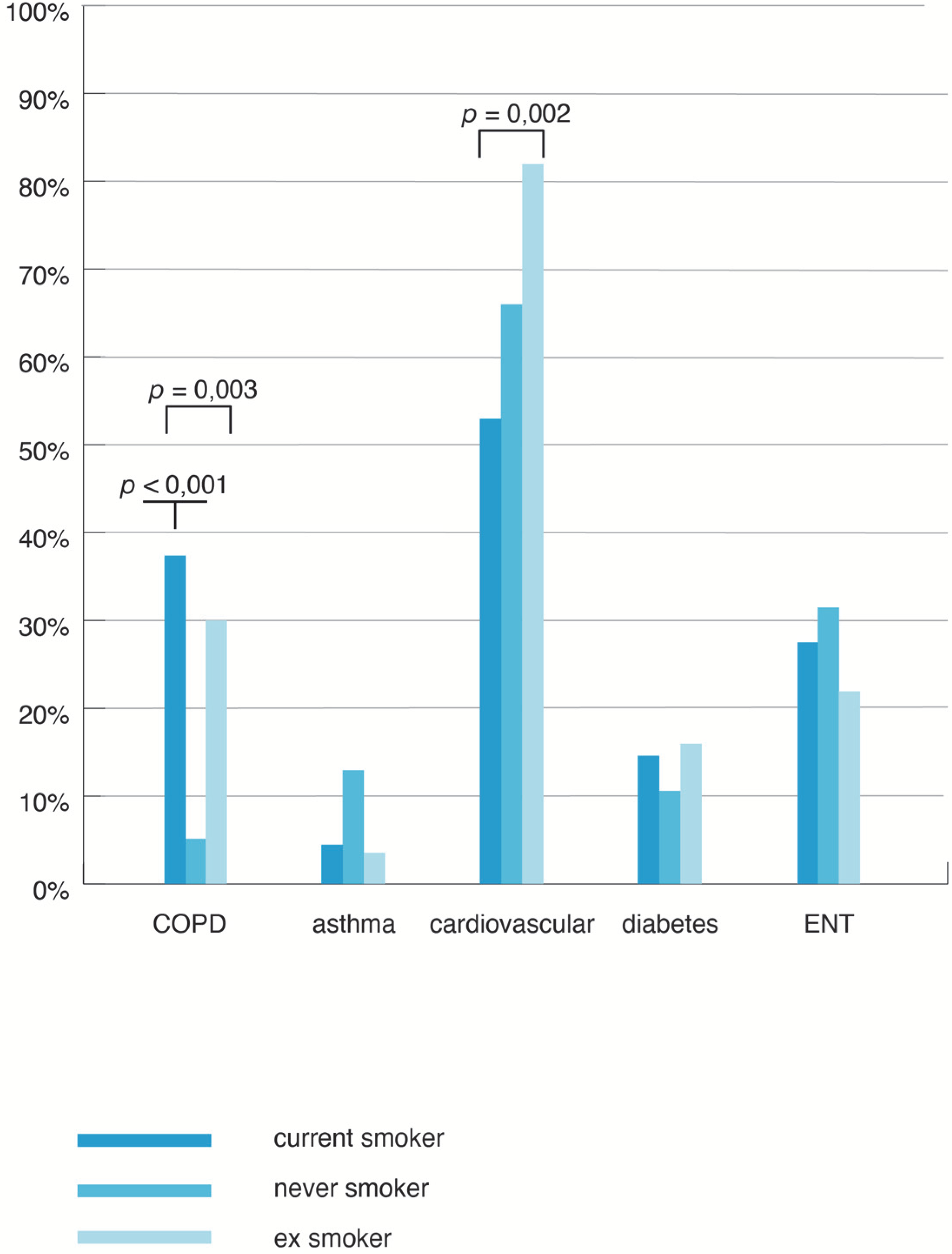

| CS (n = 81) | NS (n = 66) | ES (n = 57) | Total | p | |
|---|---|---|---|---|---|
| Age (years) | |||||
| Average | 50.06 ± 9.23 | 52.26 ± 12.47 | 57.47 ± 8.52 | 52.84 ± 10.61 | 0.00006 |
| Median | 52 | 56 | 59 | 56 | |
| Gender (M/F) | 68/13 | 44/22 | 48/9 | 160/44 | 0.018 |
| % Men | 83.95% | 66.67% | 84.21% | 78.43% | |
| BMI (kg/m2) | |||||
| Average | 36.17 ± 6.97 | 34.35 ± 6,82 | 34.84 ± 6.12 | 35.21 ± 6.71 | 0.31 |
| Median | 35 | 34 | 34 | 34 | |
| Waist circumference (cm) | |||||
| Average | 113.34 ± 10.59 | 109.53 ± 10.99 | 111.93 ± 8.58 | 111.71 ± 10.29 | 0.13 |
| Median | 112 | 108 | 111 | 110 | |
| Neck circumference (cm) | |||||
| Average | 44.44 ± 3.39 | 43.26 ± 3.78 | 44.23 ± 3.03 | 44 ± 3.45 | 0.07 |
| Median | 44 | 42 | 44 | 44 |
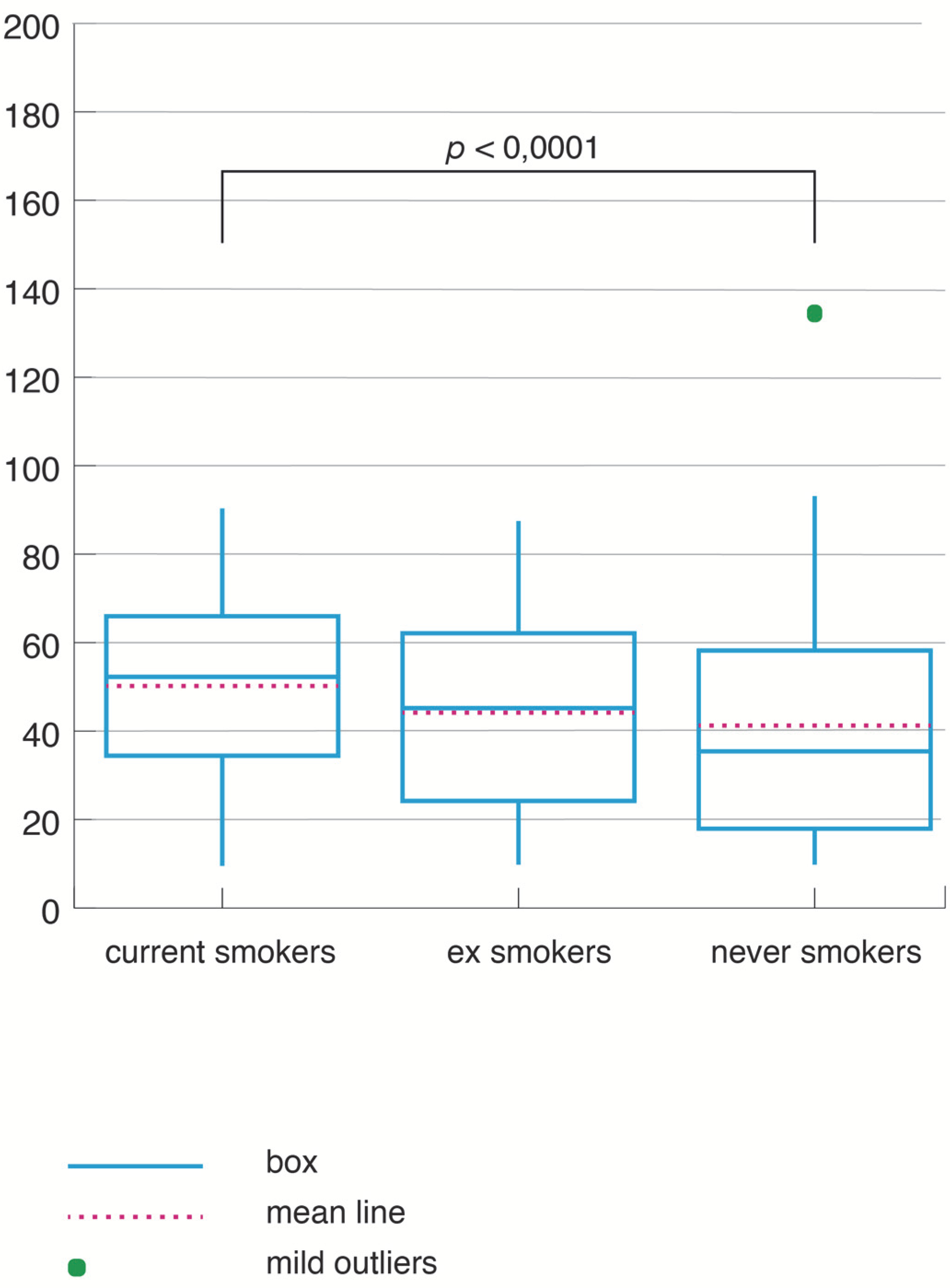
| Number of Pack-Years | ||||
|---|---|---|---|---|
| Anthropometric Characteristics | Coefficient | SE | CI | p |
| Waist circumference | 0.08 * | 0.04 | 0.159–2.068 | 0.04 |
| 0.12 ** | 0.05 | 0.02–0.22 | 0.04 | |
| 0.09 *** | 0.05 | −0.007–0.20 | 0.06 | |
| Neck circumference | 0.03 * | 0.02 | 0.004–0.065 | 0.02 |
| 0.02 ** | 0.016 | −0.01–0.05 | 0.22 | |
| 0.019 *** | 0.02 | −0.01–0.05 | 0.23 | |
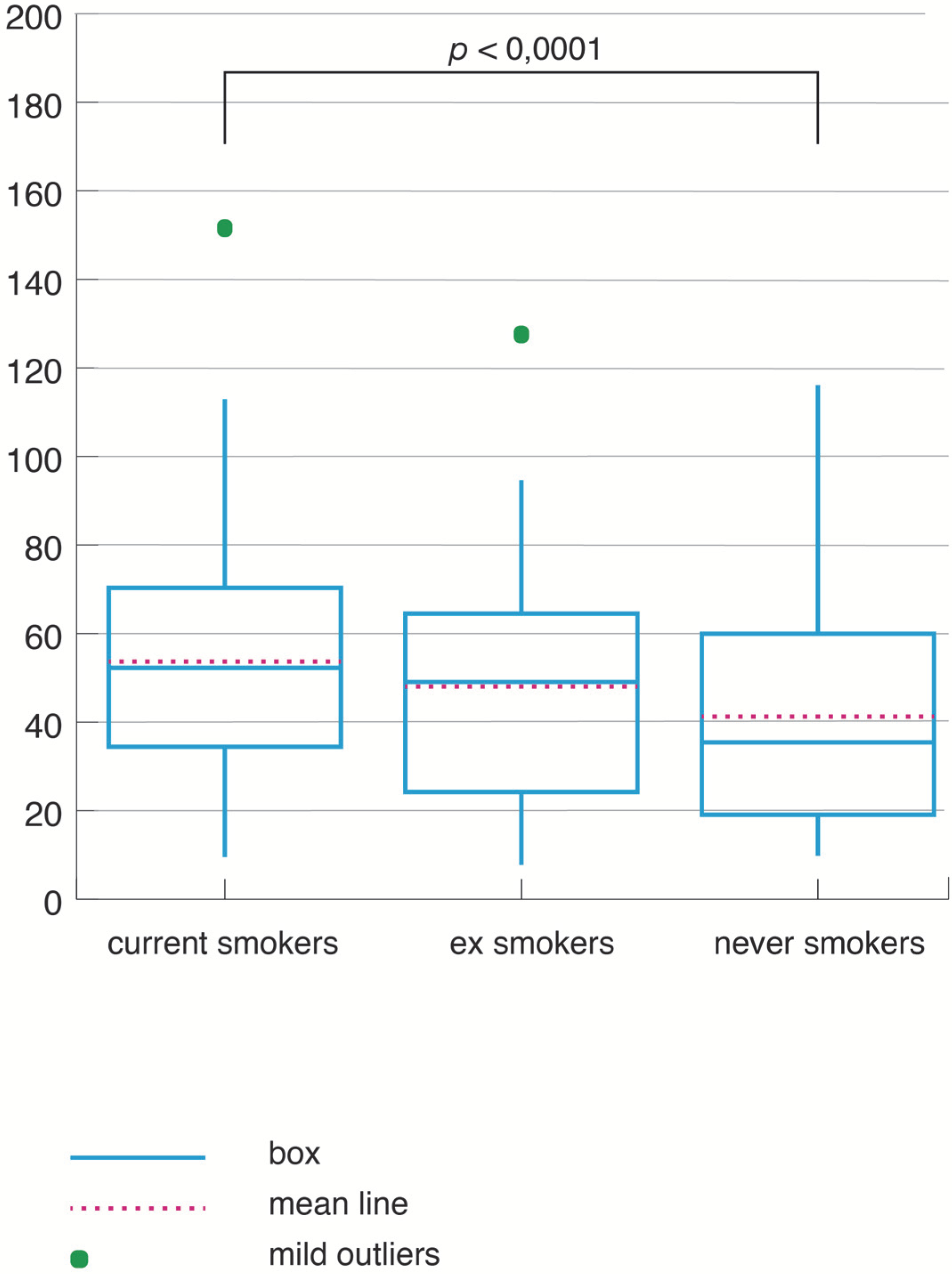
| Sleep Characteristics | CS | NS | ES | Total | p |
|---|---|---|---|---|---|
| AHI | |||||
| Average ± SD | 50.44 ± 22.17 | 41.48 ± 27.19 | 44.58 ± 21.38 | 45.91 ± 23.91 | 0.03 |
| Median | 53 | 35 | 45 | 45 | |
| Average SpO2 | |||||
| Average ± SD | 90.81 ± 3.97 | 91.52 ± 4.69 | 91.44 ± 3.12 | 91.22 ± 4.01 | 0.12 |
| Median | 92 | 93 | 92 | 92 | |
| Minimal SPO2 | |||||
| Average ± SD | 72.84 ± 8.23 | 73.32 ± 7.67 | 72.72 ± 7.01 | 72.96 ± 7.69 | 0.83 |
| Median | 72 | 73 | 72 | 72.5 | |
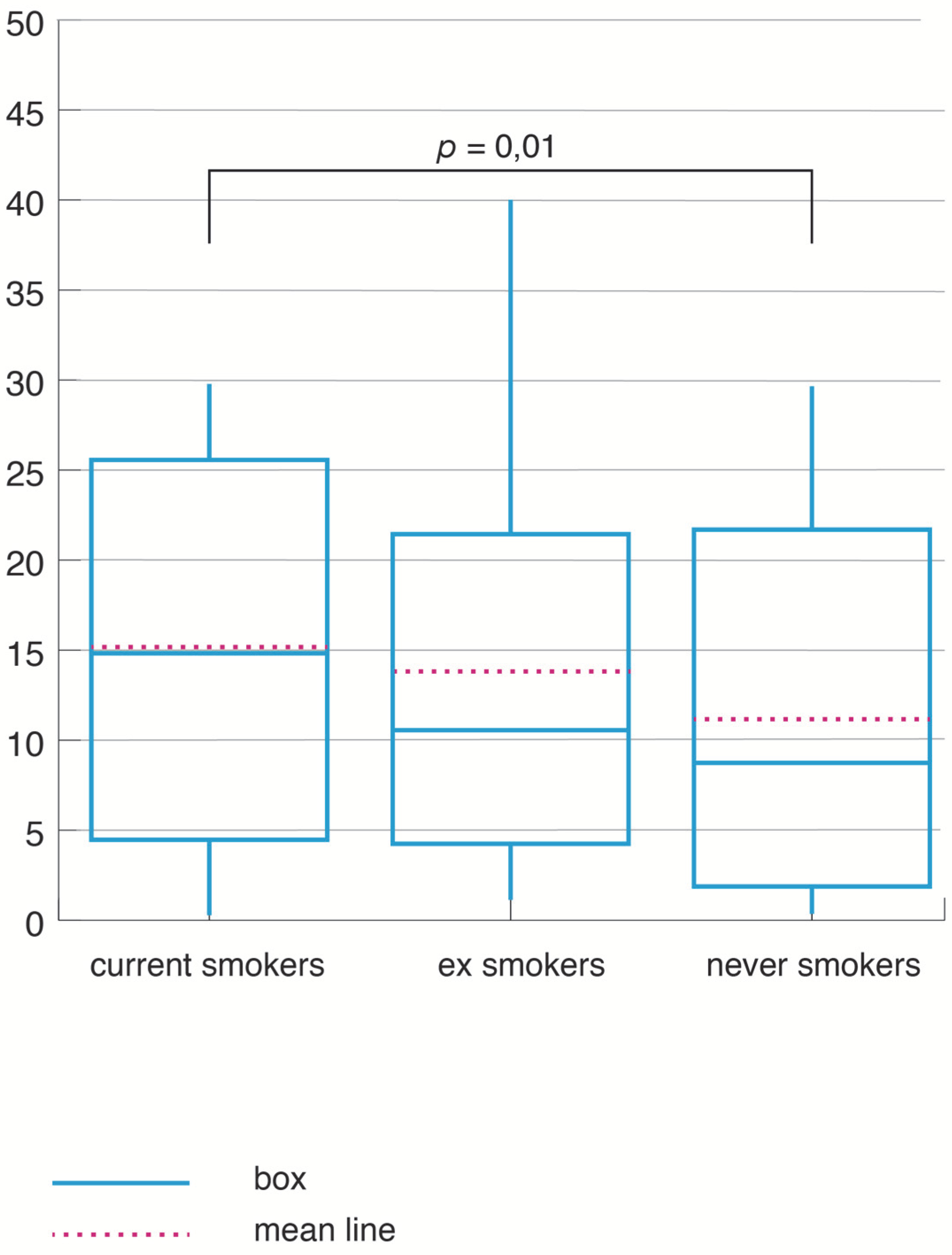
| TS_SpO290 | |||||
| Average ± SD | 15.07 ± 10.81 | 11.27 ± 10.15 | 13.71 ± 10.04 | 13.47 ± 10.46 | 0.057 |
| Median | 15 | 8.5 | 11 | 11 | |
| ODI | |||||
| Average ± SD | 53.43 ± 27.59 | 41.5 ± 26.50 | 47.96 ± 25.38 | 48.04 ± 26.98 | 0.02 |
| Median | 52 | 34.5 | 49 | 46 |
| Severity of OSA | ||||
|---|---|---|---|---|
| Coefficient | SE | CI | p | |
| Age | −0.02 | 0.02 | 0.95–1.01 | 0.33 |
| Gender | −0.03 | 0.45 | 0.40–2.35 | 0.95 |
| Waist circumference | 0.13 | 0.024 | 1.09–1.20 | <0.001 |
| Number of pack years | 1.03 | 0.01 | 1.003–1.06 | 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oțelea, M.R.; Trenchea, M.; Rașcu, A.; Antoniu, S.; Zugravu, C.; Busnatu, Ș.; Simionescu, A.A.; Arghir, O.C. Smoking Obstructive Sleep Apnea: Arguments for a Distinctive Phenotype and a Personalized Intervention. J. Pers. Med. 2022, 12, 293. https://doi.org/10.3390/jpm12020293
Oțelea MR, Trenchea M, Rașcu A, Antoniu S, Zugravu C, Busnatu Ș, Simionescu AA, Arghir OC. Smoking Obstructive Sleep Apnea: Arguments for a Distinctive Phenotype and a Personalized Intervention. Journal of Personalized Medicine. 2022; 12(2):293. https://doi.org/10.3390/jpm12020293
Chicago/Turabian StyleOțelea, Marina Ruxandra, Mihaela Trenchea, Agripina Rașcu, Sabina Antoniu, Corina Zugravu, Ștefan Busnatu, Anca Angela Simionescu, and Oana Cristina Arghir. 2022. "Smoking Obstructive Sleep Apnea: Arguments for a Distinctive Phenotype and a Personalized Intervention" Journal of Personalized Medicine 12, no. 2: 293. https://doi.org/10.3390/jpm12020293
APA StyleOțelea, M. R., Trenchea, M., Rașcu, A., Antoniu, S., Zugravu, C., Busnatu, Ș., Simionescu, A. A., & Arghir, O. C. (2022). Smoking Obstructive Sleep Apnea: Arguments for a Distinctive Phenotype and a Personalized Intervention. Journal of Personalized Medicine, 12(2), 293. https://doi.org/10.3390/jpm12020293








