Identification of Circulating Exosomal microRNAs Associated with Radioiodine Refractory in Papillary Thyroid Carcinoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Lines and Patient Samples
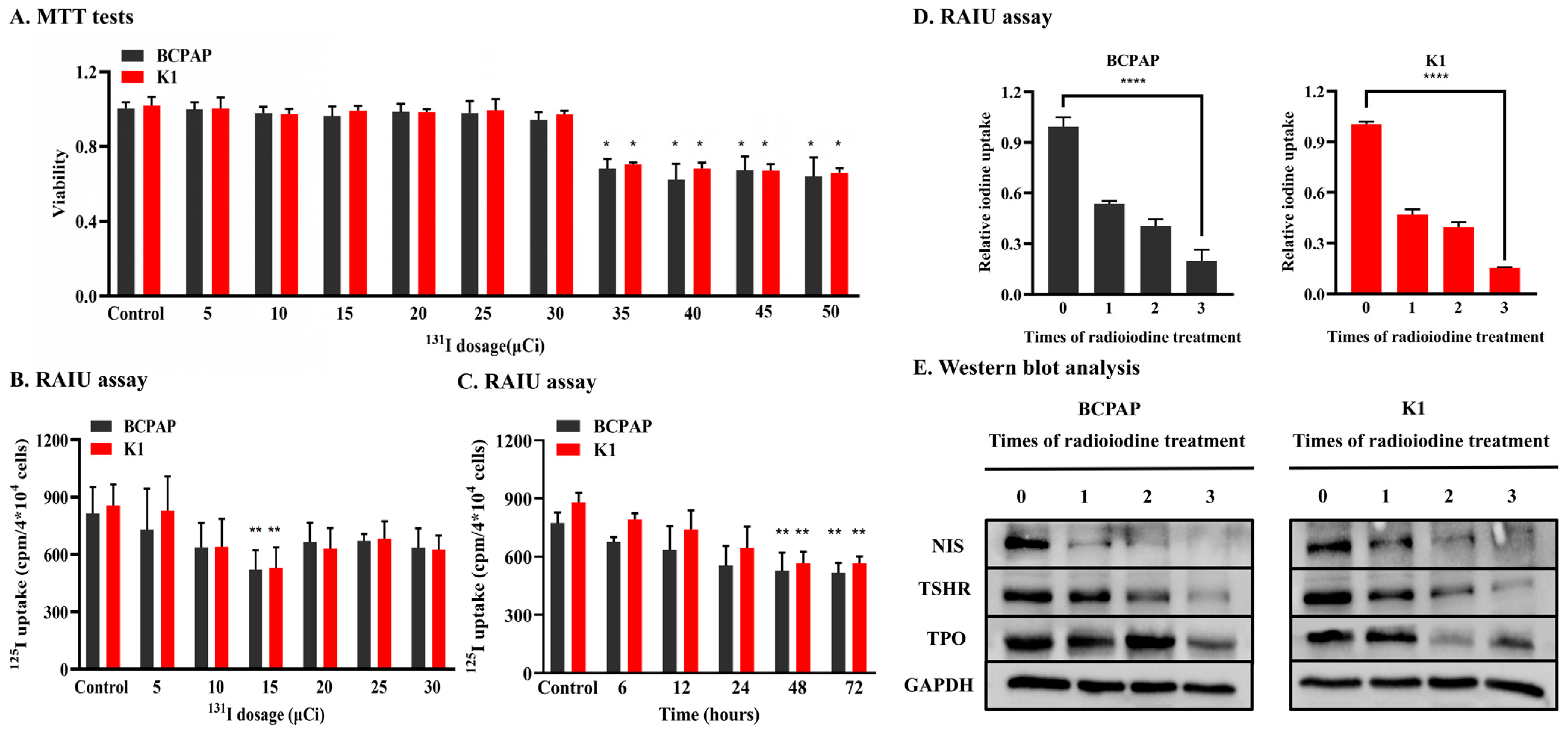
2.2. Dedifferentiation of PTC Cell Lines by 131I Pretreatment
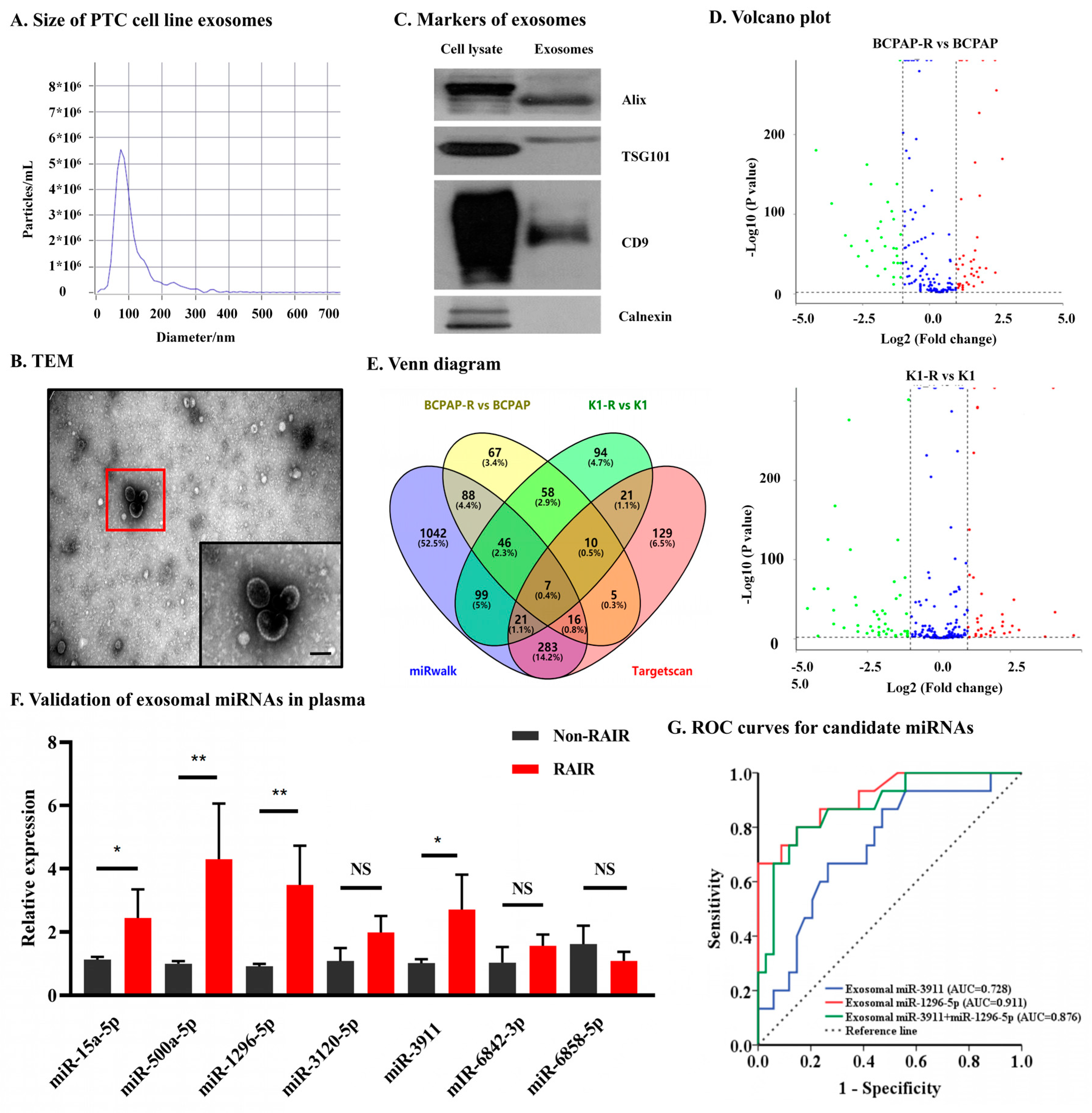
2.3. Isolation and Identification of Exosomes
2.4. Total RNA Extraction and RNA Analyses
2.5. qRT-PCR of miRNAs
2.6. Fluorescence In Situ Hybridization (FISH)
2.7. The miRNA Transfections
2.8. Luciferase Reporter Analysis
2.9. Statistical Analysis
3. Results
3.1. Establishment of RAIR PTC Cell Lines
3.2. Identification and Verification of Candidate Exosomal miRNAs Associated with RAIR PTC
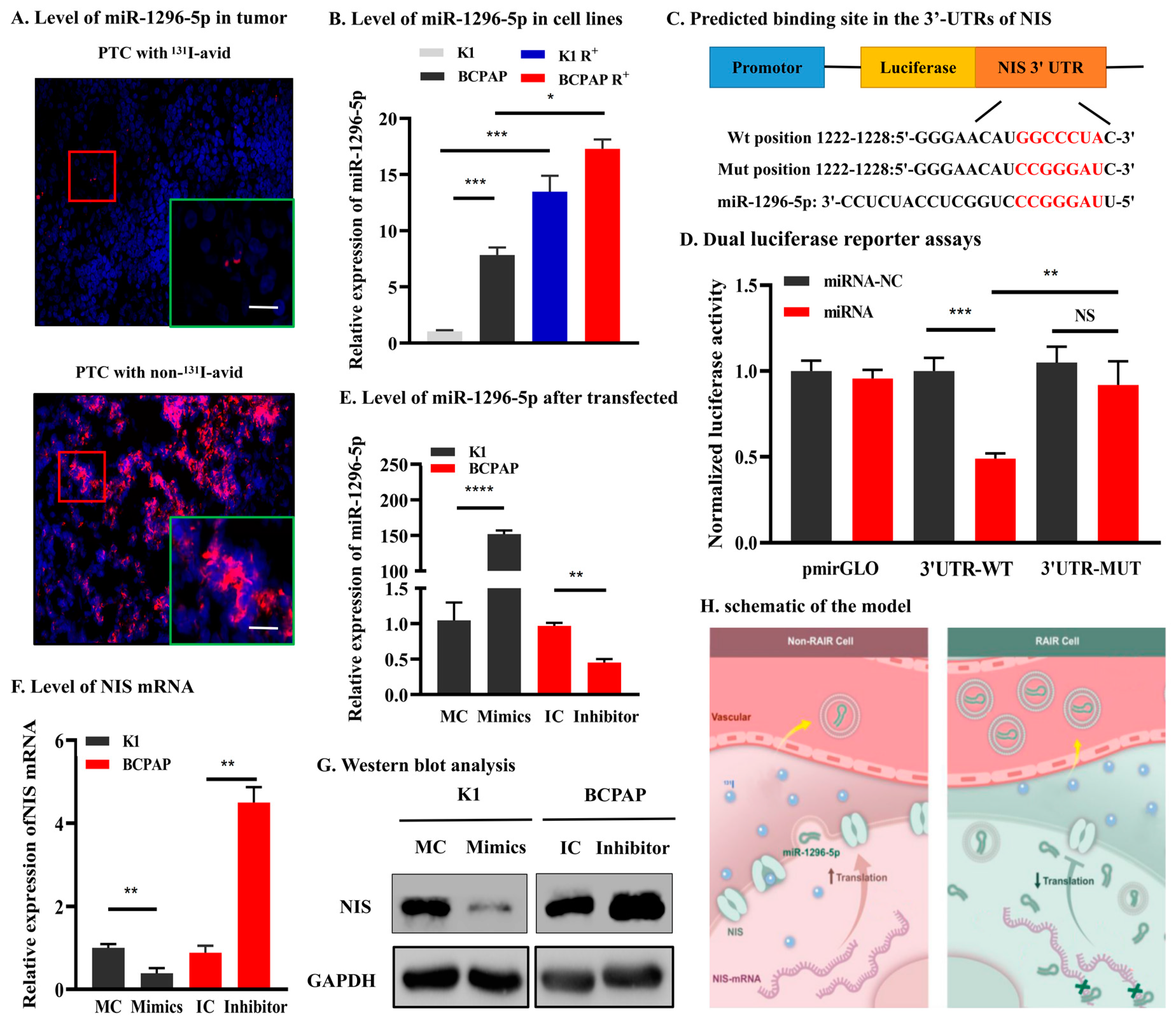
3.3. Circulating Exosomal miR-1296-5p Is a Good Biomarker and an Independent Risk Factor for RAIR PTC
3.4. miR-1296-5p Directly Targets the Downstream Gene of NIS
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Toumi, M.A.; DiGennaro, M.C.; Vahdat, V.; Jalali, M.S.; Gazelle, G.S.; Chhatwal, J.; Kelz, R.R.; Lubitz, C.C. Trends in Thyroid Surgery and Guideline-Concordant Care in the United States, 2007–2018. Thyroid 2021, 31, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Haugen, B.R. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: What is new and what has changed? Cancer 2017, 123, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Koehler, V.F.; Berg, E.; Adam, P.; Weber, G.-L.; Pfestroff, A.; Luster, M.; Kutsch, J.M.; Lapa, C.; Sandner, B.; Rayes, N.; et al. Real-World Efficacy and Safety of Multi-Tyrosine Kinase Inhibitors in Radioiodine Refractory Thyroid Cancer. Thyroid 2021, 31, 1531–1541. [Google Scholar] [CrossRef] [PubMed]
- Brose, M.S.; Panaseykin, Y.; Konda, B.; de la Fouchardiere, C.; Hughes, B.G.M.; Gianoukakis, A.G.; Park, Y.J.; Romanov, I.; Krzyzanowska, M.K.; Leboulleux, S.; et al. A Randomized Study of Lenvatinib 18 mg vs 24 mg in Patients With Radioiodine-Refractory Differentiated Thyroid Cancer. J. Clin. Endocrinol. Metab. 2022, 107, 776–787. [Google Scholar] [CrossRef] [PubMed]
- Ballal, M.S.; Yadav, M.P.; Moon, E.S.; Roesch, F.; Kumari, M.S.; Agarwal, S.; Tripathi, M.; Sahoo, R.K.; Mangu, B.S.; Tupalli, A.; et al. Novel Fibroblast Activation Protein Inhibitor-Based Targeted Theranostics for Radioiodine-Refractory Differentiated Thyroid Cancer Patients: A Pilot Study. Thyroid 2022, 32, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.S.; Zhang, X.; Wang, C.; Liu, Y.Q.; Guan, W.M.; Liang, J. Long-Term Results of a Phase II Trial of Apatinib for Progressive Radioiodine Refractory Differentiated Thyroid Cancer. J. Clin. Endocrinol. Metab. 2021, 106, e3027–e3036. [Google Scholar] [CrossRef]
- Miller, K.C.; Chintakuntlawar, A.V. Molecular-Driven Therapy in Advanced Thyroid Cancer. Curr. Treat. Options. Oncol. 2021, 22, 24. [Google Scholar] [CrossRef]
- Mishra, P.; Laha, D.; Grant, R.; Nilubol, N. Advances in Biomarker-Driven Targeted Therapies in Thyroid Cancer. Cancers 2021, 13, 6194. [Google Scholar] [CrossRef]
- Cunha, L.L.; Domingues, G.A.B.; Morari, E.C.; Soares, F.A.; Vassallo, J.; Ward, L.S. The immune landscape of the microenvironment of thyroid cancer is closely related to differentiation status. Cancer Cell Int. 2021, 21, 387. [Google Scholar] [CrossRef]
- Moretti, S.; Menicali, E.; Nucci, N.; Guzzetti, M.; Morelli, S.; Puxeddu, E. Therapy of endocrine disease Immunotherapy of advanced thyroid cancer: From bench to bedside. Eur. J. Endocrinol. 2020, 183, R41–R55. [Google Scholar] [CrossRef]
- Valerio, L.; Giani, C.; Agate, L.; Molinaro, E.; Viola, D.; Bottici, V.; Matrone, A.; Puleo, L.; Lorusso, L.; Cappagli, V.; et al. Prevalence and Risk Factors of Developing Fistula or Organ Perforation in Patients Treated with Lenvatinib for Radioiodine-Refractory Thyroid Cancer. Eur. Thyroid. J. 2021, 10, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.J.; Chang, Y.H.; Chen, Y.H.; Wu, C.W.; Wang, P.W.; Hsiao, P.J. Low Dose of Lenvatinib Treatment for Patients of Radioiodine-Refractory Differentiated Thyroid Carcinoma-A Real-World Experience. Cancer Manag. Res. 2021, 13, 7139–7148. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-S.; Yang, H.; Ding, Y.; Cheng, Y.-Z.; Shi, F.; Tan, J.; Deng, Z.-Y.; Chen, Z.-D.; Wang, R.-F.; Ji, Q.-H.; et al. Donafenib in Progressive Locally Advanced or Metastatic Radioactive Iodine-Refractory Differentiated Thyroid Cancer: Results of a Randomized, Multicenter Phase II Trial. Thyroid 2021, 31, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Pillay, P.; Moodley, K.; Vatish, M.; Moodley, J. Exosomal MicroRNAs in Pregnancy Provides Insight into a Possible Cure for Cancer. Int. J. Mol. Sci. 2020, 21, 5384. [Google Scholar] [CrossRef]
- Giassafaki, L.N.; Siqueira, S.; Panteris, E.; Psatha, K.; Chatzopoulou, F.; Aivaliotis, M.; Tzimagiorgis, G.; Müllertz, A.; Fatouros, D.G.; Vizirianakis, I.S. Towards analyzing the potential of exosomes to deliver microRNA therapeutics. J. Cell Physiol. 2021, 236, 1529–1544. [Google Scholar] [CrossRef]
- Katsila, T.; Kardamakis, D. The Role of microRNAs in Gliomas-Therapeutic Implications. Curr. Mol. Pharmacol. 2021, 14, 1004–1012. [Google Scholar] [CrossRef]
- Feng, F.; Wang, H.; Fu, H.; Wu, S.; Ye, Z.; Chen, S.; Li, J. Dedifferentiation of differentiated thyroid carcinoma cell line FTC-133 is enhanced by 131I pretreatment. Nucl. Med. Biol. 2011, 38, 1053–1058. [Google Scholar] [CrossRef]
- Ratajczak, M.; Gawel, D.; Godlewska, M. Novel Inhibitor-Based Therapies for Thyroid Cancer-An Update. Int. J. Mol. Sci. 2021, 22, 11829. [Google Scholar] [CrossRef]
- Fernandez-Mendez, C.; Santisteban, P. A Critical Balance Between PAX8 and the Hippo Mediator TAZ Determines Sodium/Iodide Symporter Expression and Function. Thyroid 2022, 32, 315–325. [Google Scholar] [CrossRef]
- Fletcher, A.; Read, M.L.; Thornton, C.E.M.; Larner, D.P.; Poole, V.L.; Brookes, K.; Nieto, H.R.; Alshahrani, M.; Thompson, R.J.; Lavery, G.G.; et al. Targeting Novel Sodium Iodide Symporter Interactors ADP-Ribosylation Factor 4 and Valosin-Containing Protein Enhances Radioiodine Uptake. Cancer Res. 2020, 80, 102–115. [Google Scholar] [CrossRef]
- Chen, J.Y.; Wang, J.J.; Lee, H.C.; Chi, C.W.; Lee, C.H.; Hsu, Y.C. Combination of peroxisome proliferator-activated receptor gamma and retinoid X receptor agonists induces sodium/iodide symporter expression and inhibits cell growth of human thyroid cancer cells. J. Chin. Med. Assoc. 2020, 83, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Hicks, H.M.; McKenna, L.R.; Espinoza, V.L.; Pozdeyev, N.; Pike, L.A.; Sams, S.B.; LaBarbera, D.; Reigan, P.; Raeburn, C.D.; Schweppe, R.E. Inhibition of BRAF and ERK1/2 has synergistic effects on thyroid cancer growth in vitro and in vivo. Mol. Carcinog. 2021, 60, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Spartalis, E.; Kotrotsios, K.; Chrysikos, D.; Spartalis, M.; Paschou, S.A.; Schizas, D.; Tsamakis, K.; Dimitroulis, D.; Troupis, T.; Nikiteas, N. Histone Deacetylase Inhibitors and Papillary Thyroid Cancer. Curr. Pharm. Des. 2021, 27, 2199–2208. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Xu, Z.; Ji, Q.; Ge, M.; Shi, F.; Qin, J.; Wang, F.; Chen, G.; Zhang, Y.; Huang, R.; et al. A Randomized, Phase III Study of Lenvatinib in Chinese Patients with Radioiodine-Refractory Differentiated Thyroid Cancer. Clin. Cancer Res. 2021, 27, 5502–5509. [Google Scholar] [CrossRef]
- Jafri, S.; Yaqub, A. Redifferentiation of BRAF V600E-Mutated Radioiodine Refractory Metastatic Papillary Thyroid Cancer After Treatment With Dabrafenib and Trametinib. Cureus 2021, 13, e17488. [Google Scholar] [CrossRef]
- Thakur, A.; Parra, D.C.; Motallebnejad, P.; Brocchi, M.; Chen, H.J. Exosomes: Small vesicles with big roles in cancer, vaccine development, and therapeutics. Bioact. Mater. 2022, 10, 281–294. [Google Scholar] [CrossRef]
- Zeng, H.; Fu, Y.; Shen, L.; Quan, S. MicroRNA signatures in plasma and plasma exosome during window of implantation for implantation failure following in-vitro fertilization and embryo transfer. Reprod. Biol. Endocrinol. 2021, 19, 180. [Google Scholar] [CrossRef]
- Rogucki, M.; Buczynska, A.; Kretowski, A.J.; Poplawska-Kita, A. The Importance of miRNA in the Diagnosis and Prognosis of Papillary Thyroid Cancer. J. Clin. Med. 2021, 10, 4738. [Google Scholar] [CrossRef]
- Alwani, A.; Andreasik, A.; Szatanek, R.; Siedlar, M.; Baj-Krzyworzeka, M. The Role of miRNA in Regulating the Fate of Monocytes in Health and Cancer. Biomolecules 2022, 12, 100. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef]
- Toden, S.; Goel, A. Non-coding RNAs as liquid biopsy biomarkers in cancer. Br. J. Cancer 2022, 126, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Majid, S.; Dar, A.A.; Saini, S.; Chen, Y.; Shahryari, V.; Liu, J.; Zaman, M.S.; Hirata, H.; Yamamura, S.; Ueno, K.; et al. Regulation of minichromosome maintenance gene family by microRNA-1296 and genistein in prostate cancer. Cancer Res. 2010, 70, 2809–2818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.; He, W.; Gao, J.; Luo, J.; Huang, X.; Gao, C. Computational prediction and experimental validation of a novel synthesized pan-PIM inhibitor PI003 and its apoptosis-inducing mechanisms in cervical cancer. Oncotarget 2015, 6, 8019–8035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zang, H.; Li, Y.; Zhang, X.; Huang, G. Circ_0000517 Contributes to Hepatocellular Carcinoma Progression by Upregulating TXNDC5 via Sponging miR-1296-5p. Cancer Manag. Res. 2020, 12, 3457–3468. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Zhao, L.-M.; Bai, H.-Y.; Zhang, C.; Dai, S.-L.; Lv, H.-L.; Shan, B.-E. The tumor-suppressive function of miR-1296-5p by targeting EGFR and CDK6 in gastric cancer. Biosci. Rep. 2019, 39, BSR20181556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shan, X.; Wen, W.; Zhu, D.; Yan, T.; Cheng, W.; Huang, Z.; Zhang, L.; Zhang, H.; Wang, T.; Zhu, W.; et al. miR 1296-5p Inhibits the Migration and Invasion of Gastric Cancer Cells by Repressing ERBB2 Expression. PLoS ONE 2017, 12, e0170298. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Hu, K.; Chao, Y.; Wang, X. MicroRNA-1296-5p suppresses the proliferation, migration, and invasion of human osteosarcoma cells by targeting NOTCH2. J. Cell Biochem. 2020, 121, 2038–2046. [Google Scholar] [CrossRef] [PubMed]
- Fallahi, P.; Ferrari, S.M.; Elia, G.; Ragusa, F.; Patrizio, A.; Paparo, S.R.; Marone, G.; Galdiero, M.R.; Guglielmi, G.; Foddis, R.; et al. Primary cell cultures for the personalized therapy in aggressive thyroid cancer of follicular origin. Semin. Cancer Biol. 2022, 79, 203–216. [Google Scholar] [CrossRef]
| Exosomal miRNAs | Log2. FC (B) | p-Value | Log2. FC (K) | p-Value | Description |
|---|---|---|---|---|---|
| miR-15a-5p | −6.5031 | <0.001 | −4.2368 | 0.002 | Down |
| miR-500a-5p | −2.9256 | <0.001 | −5.8217 | <0.001 | Down |
| miR-1296-5p | 6.1642 | <0.001 | 3.0664 | 0.007 | Up |
| miR-3120-5p | 5.1564 | <0.001 | 3.7183 | 0.002 | Up |
| miR-3911 | 6.7649 | <0.001 | 3.5621 | 0.002 | Up |
| miR-6842-3p | 4.2663 | <0.001 | 6.5256 | <0.001 | Up |
| miR-6858-5p | 5.2635 | <0.001 | 2.7625 | 0.002 | Up |
| Clinical Characteristics | 131I-avid (n = 48) | Non-131I-avid (n = 21) | p-Value |
|---|---|---|---|
| Age (mean ± SD, years) | 45.2 ± 12.1 | 47.7 ± 18.3 | 0.514 |
| ≥55/<55 | 9 (18.8%)/39 (81.2%) | 6 (28.6%)/15 (71.4%) | 0.363 |
| Gender (male/female) | 16 (33.3%)/32 (66.7%) | 9 (42.9%)/12(57.1%) | 0.449 |
| BMI (mean ± SD, kg/m2) | 23.0 ± 3.0 | 23.3 ± 4.5 | 0.722 |
| Nodular goiter (yes/no) | 27 (56.3%)/21 (43.7%) | 8 (38.1%)/13 (61.9%) | 0.165 |
| Hashimoto’s thyroiditis (yes/no) | 8 (16.7%)/40 (83.3%) | 4 (19.0%)/17 (81.0%) | 0.811 |
| Graves’s disease (yes/no) | 4 (8.3%)/44 (91.7%) | 0 (0.0%)/21 (100.0%) | 0.306 |
| Multifocality (yes/no) | 16 (33.3%)/32 (66.7%) | 8 (38.1%)/13 (61.7%) | 0.702 |
| T stage (T1-T2/T3-T4) | 4 (8.3%)/44 (91.7%) | 1 (4.8%)/20 (95.2%) | 0.553 |
| n stage (N0/N1) | 7 (14.6%)/41 (85.4%) | 1 (4.8%)/20 (95.2%) | 0.420 |
| RAI dose (median, uci) | 100 (100–150) | 100 (100–200) | 0.698 |
| miRNAs | Univariate | Multivariate | ||
|---|---|---|---|---|
| OR (95%CI) | p Value | OR (95%CI) | p Value | |
| miR-1296-5p | 10.233 (3.164–33.091) | <0.001 * | 12.319 (2.853–53.192) | 0.001 * |
| miR-3911 | 1.765 (0.796–3.916) | 0.162 | 1.092 (0.523–2.277) | 0.815 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, G.; Chen, W.; Gong, Y.; Wei, T.; Gong, R.; Zhu, J.; Li, Z.; Lei, J. Identification of Circulating Exosomal microRNAs Associated with Radioiodine Refractory in Papillary Thyroid Carcinoma. J. Pers. Med. 2022, 12, 2017. https://doi.org/10.3390/jpm12122017
Li G, Chen W, Gong Y, Wei T, Gong R, Zhu J, Li Z, Lei J. Identification of Circulating Exosomal microRNAs Associated with Radioiodine Refractory in Papillary Thyroid Carcinoma. Journal of Personalized Medicine. 2022; 12(12):2017. https://doi.org/10.3390/jpm12122017
Chicago/Turabian StyleLi, Genpeng, Wenjie Chen, Yanping Gong, Tao Wei, Rixiang Gong, Jingqiang Zhu, Zhihui Li, and Jianyong Lei. 2022. "Identification of Circulating Exosomal microRNAs Associated with Radioiodine Refractory in Papillary Thyroid Carcinoma" Journal of Personalized Medicine 12, no. 12: 2017. https://doi.org/10.3390/jpm12122017
APA StyleLi, G., Chen, W., Gong, Y., Wei, T., Gong, R., Zhu, J., Li, Z., & Lei, J. (2022). Identification of Circulating Exosomal microRNAs Associated with Radioiodine Refractory in Papillary Thyroid Carcinoma. Journal of Personalized Medicine, 12(12), 2017. https://doi.org/10.3390/jpm12122017







