Sexuality, Intimacy, and Reproductive Health after Spinal Cord Injury
Abstract
1. Introduction/Epidemiology
2. Sexual Health after SCI
2.1. Erectile Dysfunction
2.1.1. Erection Physiology
2.1.2. Diagnosis
2.1.3. Treatment
2.2. Anejaculation
2.2.1. Ejaculation Physiology
2.2.2. Treatment
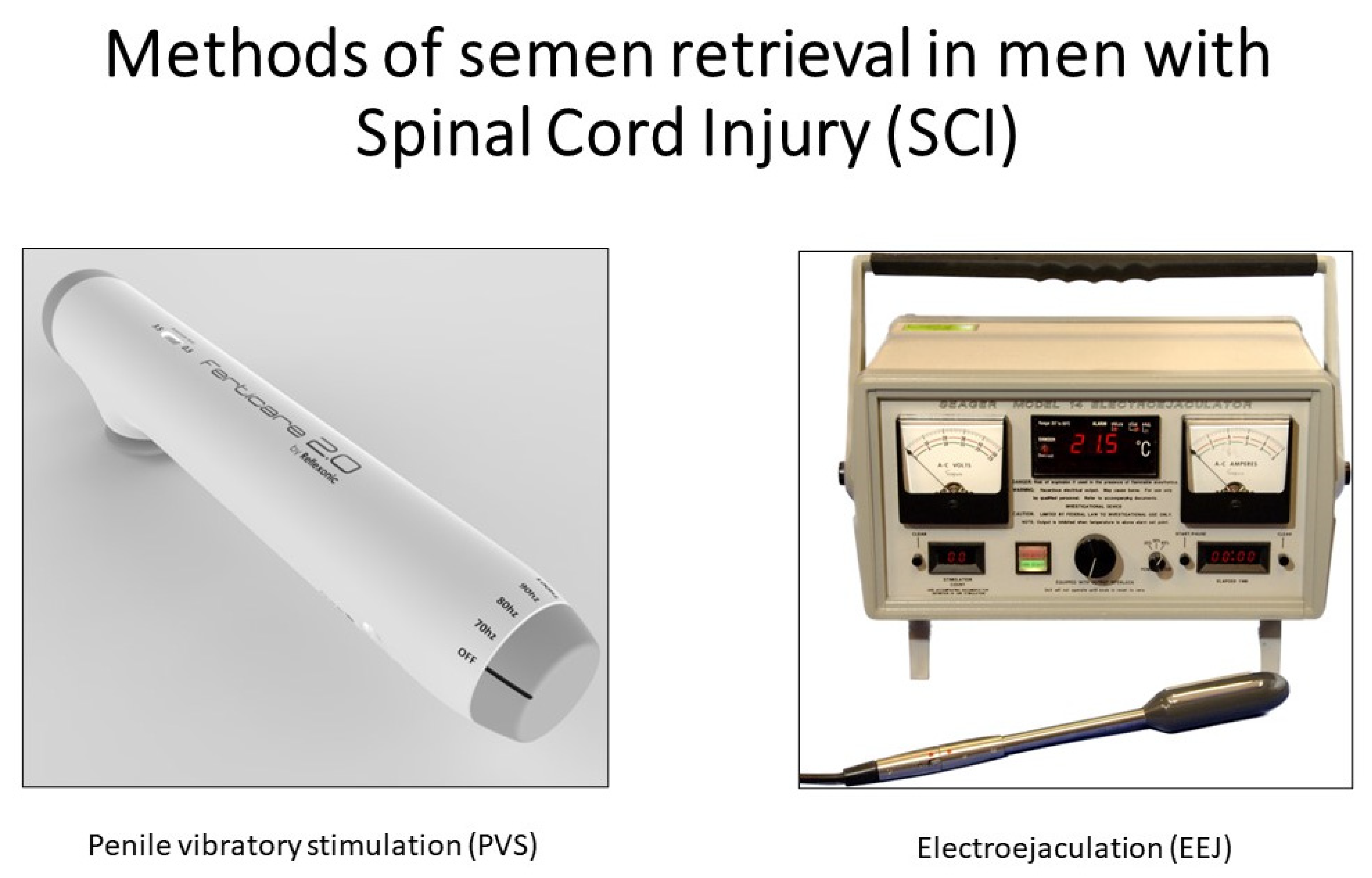
2.3. Penile Vibratory Stimulation (PVS)
2.3.1. Electroejaculation (EEJ)
2.3.2. Prostate Massage
2.3.3. Surgical Sperm Retrieval
2.4. Semen Quality/Motility Barriers
3. Reproductive Options after SCI
3.1. Intravaginal Insemination (IVI)
3.2. Intrauterine Insemination (IUI)
3.3. In Vitro Insemination/Intracytoplasmic Sperm Injection (IVF-ICSI)
4. Pursuit of Pleasure and Intimacy after SCI
5. Conclusions
6. Resources/Support
6.1. Book Readings
- -
- Regain That Feeling: Secrets to Sexual Self-Discovery: People Living with Spinal Cord Injuries Share Profound Insights into Sex, Pleasure, Relationships, Orgasm, and the Importance of Connectedness—Mitchell S. Tepper, Ph.D., MPH [110].
- -
- Enabling Romance: A Guide to Love, Sex and Relationships for People with Disabilities (and the People who Care About Them)—Ken Kroll and Erica Levy Klein [109].
6.2. Educational Materials
6.3. Organizations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National SCI Statistical Center. Traumatic Spinal Cord Injury Facts and Figures at a Glance 2022. Available online: https://msktc.org/sites/default/files/SCI-Facts-Figs-2022-Eng-508.pdf (accessed on 19 September 2022).
- Lee, B.B.; Cripps, R.A.; Fitzharris, M.; Wing, P.C. The global map for traumatic spinal cord injury epidemiology: Update 2011, global incidence rate. Spinal Cord 2013, 52, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Wyndaele, M.; Wyndaele, J.J. Incidence, prevalence and epidemiology of spinal cord injury: What learns a worldwide literature survey? Spinal Cord 2006, 44, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Polinder, S.; Meerding, W.J.; Mulder, S.; Petridou, E.; Van Beeck, E. Assessing the burden of injury in six European countries. Bull. World Heal. Organ. 2007, 85, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Rasouli, M.R.; Nouri, M.; Rahimi-Movaghar, V. Spinal cord injuries from road traffic crashes in southeastern Iran. Chin. J. Traumatol. (Engl. Ed.) 2007, 10, 323–326. [Google Scholar]
- Feng, H.-Y.; Ning, G.-Z.; Feng, S.-Q.; Yu, T.-Q.; Zhou, H.-X. Epidemiological profile of 239 traumatic spinal cord injury cases over a period of 12 years in Tianjin, China. J. Spinal Cord Med. 2011, 34, 388–394. [Google Scholar] [CrossRef] [PubMed]
- McCammon, J.R.; Ethans, K. Spinal cord injury in Manitoba: A provincial epidemiological study. J. Spinal Cord Med. 2011, 34, 6–10. [Google Scholar] [CrossRef]
- Ibrahim, E.M.; Lynne, C.M.; Brackett, N.L. Male fertility following spinal cord injury: An update. Andrology 2015, 4, 13–26. [Google Scholar] [CrossRef]
- Alexander, M.S.; Aisen, C.M.; Alexander, S.M.; Aisen, M.L. Sexual concerns after Spinal Cord Injury: An update on management. NeuroRehabilitation 2017, 41, 343–357. [Google Scholar] [CrossRef]
- Anderson, K.D. Targeting Recovery: Priorities of the Spinal Cord-Injured Population. J. Neurotrauma 2004, 21, 1371–1383. [Google Scholar] [CrossRef]
- Giuliano, F.; Sanchez-Ramos, A.; Löchner-Ernst, D.; Del Popolo, G.; Cruz, N.; Leriche, A.; Lombardi, G.; Reichert, S.; Dahl, P.; Elion-Mboussa, A.; et al. Efficacy and Safety of Tadalafil in Men with Erectile Dysfunction Following Spinal Cord Injury. Arch. Neurol. 2007, 64, 1584–1592. [Google Scholar] [CrossRef]
- Hess, M.J.; Hough, S. Impact of spinal cord injury on sexuality: Broad-based clinical practice intervention and practical application. J. Spinal Cord Med. 2012, 35, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Sinha, V.; Elliott, S.; Ibrahim, E.; Lynne, C.M.; Brackett, N.L. Reproductive Health of Men with Spinal Cord Injury. Top. Spinal Cord Inj. Rehabil. 2017, 23, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Monga, M.; Gordon, Z.; Rajasekaran, M. Spinal cord injury and male infertility. Zhonghua Nan Ke Xue Natl. J. Androl. 2002, 8, 235–240. [Google Scholar]
- Lue, T.F. Physiology of Penile Erection and Pathophysiology of Erectile Dysfunction. In Campbell-Walsh Urology, 10th ed.; Wein, A.J., Kavoussi, L.R., Novick, A.C., Partin, A.W., Peters, C.A., Eds.; Elsevier Saunders: Philadelphia, PA, USA, 2012; pp. 591–615. [Google Scholar]
- Alexander, M.S.; Marson, L. The neurologic control of arousal and orgasm with specific attention to spinal cord lesions: Integrating preclinical and clinical sciences. Auton. Neurosci. 2018, 209, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Ibrahim, E. Sexual Dysfunction After Spinal Cord Injury. Curr. Sex. Heal. Rep. 2020, 12, 244–250. [Google Scholar] [CrossRef]
- Dean, R.C.; Lue, T.F. Physiology of Penile Erection and Pathophysiology of Erectile Dysfunction. Urol. Clin. North Am. 2005, 32, 379–395. [Google Scholar] [CrossRef]
- Burnett, A.L. Nitric oxide control of lower genitourinary tract functions: A review. Urology 1995, 45, 1071–1083. [Google Scholar] [CrossRef]
- Rosen, R.C.; Cappelleri, J.C.; Gendrano, N. The International Index of Erectile Function (IIEF): A state-of-the-science review. Int. J. Impot. Res. 2002, 14, 226–244. [Google Scholar] [CrossRef]
- Yafi, F.A.; Huynh, L.M.; Ahlering, T.; Rosen, R. What Is a “Validated Questionnaire”? A Critical Review of Erectile Function Assessment. J. Sex. Med. 2020, 17, 849–860. [Google Scholar] [CrossRef]
- Ghanem, H.M.; Salonia, A.; Martin-Morales, A. SOP: Physical Examination and Laboratory Testing for Men with Erectile Dysfunction. J. Sex. Med. 2013, 10, 108–110. [Google Scholar] [CrossRef]
- Varela, C.G.; Yeguas, L.A.M.; Rodríguez, I.C.; Vila, M.D.D. Penile Doppler Ultrasound for Erectile Dysfunction: Technique and Interpretation. Am. J. Roentgenol. 2020, 214, 1112–1121. [Google Scholar] [CrossRef] [PubMed]
- Alexander, M.S.; New, P.W.; Biering-Sørensen, F.; Courtois, F.; Del Popolo, G.; Elliott, S.; Kiekens, C.; Vogel, L.; Previnaire, J.G. International spinal cord injury male sexual function and female sexual and reproductive function basic data sets—Version 2.0. Spinal Cord Ser. Cases 2017, 3, 17050. [Google Scholar] [CrossRef] [PubMed]
- Bors, E.; Comarr, A.E. Neurological disturbances of sexual function with special reference to 529 patients with spinal cord injury. Urol. Surv. 1960, 10, 191–222. [Google Scholar]
- Madeira, C.R.; Tonin, F.S.; Fachi, M.M.; Borba, H.H.; Ferreira, V.L.; Leonart, L.P.; Bonetti, A.F.; Moritz, R.P.; Trindade, A.C.L.B.; Gonçalves, A.G.; et al. Efficacy and safety of oral phosphodiesterase 5 inhibitors for erectile dysfunction: A network meta-analysis and multicriteria decision analysis. World J. Urol. 2020, 39, 953–962. [Google Scholar] [CrossRef]
- Causanilles, A.; Cantillano, D.R.; Emke, E.; Bade, R.; Baz-Lomba, J.A.; Castiglioni, S.; Castrignanò, E.; Gracia-Lor, E.; Hernández, F.; Kasprzyk-Hordern, B.; et al. Comparison of phosphodiesterase type V inhibitors use in eight European cities through analysis of urban wastewater. Environ. Int. 2018, 115, 279–284. [Google Scholar] [CrossRef]
- Derry, F.A.; Dinsmore, W.W.; Fraser, M.; Gardner, B.P.; Glass, C.A.; Maytom, M.C.; Smith, M.D. Efficacy and safety of oral sildenafil (Viagra) in men with erectile dysfunction caused by spinal cord injury. Neurology 1998, 51, 1629–1633. [Google Scholar] [CrossRef]
- Hultling, C.; El Masry, W.S.; Smith, M.D.; Osterloh, I.H.; Orr, M.; Maytom, M. Sildenafil Study Group Randomized trial of sildenafil for the treatment of erectile dysfunction in spinal cord injury. Ann. Neurol. 1999, 46, 15–21. [Google Scholar] [CrossRef]
- Hultling, C.; Giuliano, F.; Quirk, F.; Peña, B.; Mishra, A.; Smith, M.D. Quality of life in patients with spinal cord injury receiving VIAGRA® (sildenafil citrate) for the treatment of erectile dysfunction. Spinal Cord 2000, 38, 363–370. [Google Scholar] [CrossRef]
- Maytom, M.C.; Derry, F.A.; Dinsmore, W.W.; Glass, C.A.; Smith, M.D.; Orr, M.; Osterloh, I.H. A two-part study of sildenafil (VIAGRA) in men with erectile dysfunction caused by spinal cord injury. Spinal Cord 1999, 37, 110–116. [Google Scholar] [CrossRef]
- Ohl, D.A.; Carlsson, M.; Stecher, V.J.; Rippon, G.A. Efficacy and Safety of Sildenafil in Men with Sexual Dysfunction and Spinal Cord Injury. Sex. Med. Rev. 2017, 5, 521–528. [Google Scholar] [CrossRef]
- Ramos, A.S.; Vidal, J.; Jáuregui, M.L.; La Barrera, S.S.-D.; Recio, C.; Giner, M.; Toribio, L.; Salvador, S.; Sanmartín, A.; De La Fuente, M.; et al. Efficacy, safety and predictive factors of therapeutic success with sildenafil for erectile dysfunction in patients with different spinal cord injuries. Spinal Cord 2001, 39, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, F.; Rubio-Aurioles, E.; Kennelly, M.; Montorsi, F.; Kim, E.D.; Finkbeiner, A.E.; Pommerville, P.J.; Colopy, M.W.; Wachs, B.H. Vardenafil Improves Ejaculation Success Rates and Self-confidence in Men with Erectile Dysfunction due to Spinal Cord Injury. Spine 2008, 33, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, F.; Rubio-Aurioles, E.; Kennelly, M.; Montorsi, F.; Kim, E.D.; Finkbeiner, A.E.; Pommerville, P.J.; Colopy, M.W.; Wilkins, H.J.; Wachs, B.H.; et al. Efficacy and safety of vardenafil in men with erectile dysfunction caused by spinal cord injury. Neurology 2006, 66, 210–216. [Google Scholar] [CrossRef]
- Denil, J.; Ohl, D.A.; Smythe, C. Vacuum erection device in spinal cord injured men: Patient and partner satisfaction. Arch. Phys. Med. Rehabil. 1996, 77, 750–753. [Google Scholar] [CrossRef]
- Lloyd, E.; Tóth, L.; Perkash, I. Vacuum tumescence: An option for spinal cord injured males with erectile dysfunction. SCI Nurs. Publ. Am. Assoc. Spinal Cord Inj. Nurses 1989, 6, 25–28. [Google Scholar]
- Rivas, D.A.; Chancellor, M.B. Complications Associated with the Use of Vacuum Constriction Devices for Erectile Dysfunction in the Spinal Cord Injured Population. J. Am. Paraplegia Soc. 1994, 17, 136–139. [Google Scholar] [CrossRef]
- Bodner, D.R.; Haas, C.A.; Krueger, B.; Seftel, A.D. Intraurethral alprostadil for treatment of erectile dysfunction in patients with spinal cord injury. Urology 1999, 53, 199–202. [Google Scholar] [CrossRef]
- Lee, M.; Sharifi, R. Non-invasive Management Options for Erectile Dysfunction When a Phosphodiesterase Type 5 Inhibitor Fails. Drugs Aging 2018, 35, 175–187. [Google Scholar] [CrossRef]
- Del Popolo, G.; Cito, G.; Gemma, L.; Natali, A. Neurogenic Sexual Dysfunction Treatment: A Systematic Review. Eur. Urol. Focus 2020, 6, 868–876. [Google Scholar] [CrossRef]
- Zermann, D.-H.; Kutzenberger, J.; Sauerwein, D.; Schubert, J.; Loeffler, U. Penile Prosthetic Surgery in Neurologically Impaired Patients: Long-Term Followup. J. Urol. 2006, 175, 1041–1044. [Google Scholar] [CrossRef]
- Kim, Y.D.; Yang, S.O.; Lee, J.K.; Jung, T.Y.; Shim, H.B. Usefulness of a malleable penile prosthesis in patients with a spinal cord injury. Int. J. Urol. 2008, 15, 919–923. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, F.; Clement, P. Neuroanatomy and physiology of ejaculation. Annu. Rev. Sex Res. 2005, 16, 190–216. [Google Scholar] [PubMed]
- Chéhensse, C.; Facchinetti, P.; Bahrami, S.; Andrey, P.; Soler, J.-M.; Chrétien, F.; Bernabé, J.; Clément, P.; Denys, P.; Giuliano, F. Human spinal ejaculation generator. Ann. Neurol. 2016, 81, 35–45. [Google Scholar] [CrossRef]
- Coolen, L.M.; Allard, J.; Truitt, W.A.; McKenna, K.E. Central regulation of ejaculation. Physiol. Behav. 2004, 83, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.; Hill, S.; Baker, H. Male fertility and sexual function after spinal cord injury. Prog. Brain Res. 2006, 152, 427–439. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, E.; Brackett, N.L.; Lynne, C.M. Penile Vibratory Stimulation for Semen Retrieval in Men with Spinal Cord Injury: Patient Perspectives. Res. Rep. Urol. 2022, 14, 149–157. [Google Scholar] [CrossRef]
- Fode, M.; Ohl, D.A.; Sønksen, J. A step-wise approach to sperm retrieval in men with neurogenic anejaculation. Nat. Rev. Urol. 2015, 12, 607–616. [Google Scholar] [CrossRef]
- Brackett, N.L.; Lynne, C.M.; Sonksen, J.; Ohl, D. Penile Vibratory Stimulation. In Surgical and Medical Management of Male Infertility, 1st ed.; Goldstein, M., Schlegel, P.N., Eds.; Cambridge University Press: New York, NY, USA, 2013; pp. 119–129. [Google Scholar]
- Ibrahim, E.; Jensen, C.F.S.; Sunara, I.; Khodamoradi, K.; Aballa, T.C.; Elliott, S.; Sonksen, J.; Ohl, D.A.; Hultling, C.; Lynne, C.M.; et al. Evaluation of a re-engineered device for penile vibratory stimulation in men with spinal cord injury. Spinal Cord 2020, 59, 151–158. [Google Scholar] [CrossRef]
- Sonksen, J.; Ohl, D.A. Penile vibratory stimulation and electroejaculation in the treatment of ejaculatory dysfunction1. Int. J. Androl. 2002, 25, 324–332. [Google Scholar] [CrossRef]
- Brackett, N.L.; Ibrahim, E.; Iremashvili, V.; Aballa, T.C.; Lynne, C.M. Treatment for Ejaculatory Dysfunction in Men with Spinal Cord Injury: An 18-Year Single Center Experience. J. Urol. 2010, 183, 2304–2308. [Google Scholar] [CrossRef]
- Brackett, N.L.; Kafetsoulis, A.; Ibrahim, E.; Aballa, T.C.; Lynne, C.M. Application of 2 Vibrators Salvages Ejaculatory Failures to 1 Vibrator During Penile Vibratory Stimulation in Men with Spinal Cord Injuries. J. Urol. 2007, 177, 660–663. [Google Scholar] [CrossRef] [PubMed]
- Kafetsoulis, A.; Ibrahim, E.; Aballa, T.C.; Goetz, L.L.; Lynne, C.M.; Brackett, N.L. Abdominal electrical stimulation rescues failures to penile vibratory stimulation in men with spinal cord injury: A report of two cases. Urology 2006, 68, 204.e9–204.e11. [Google Scholar] [CrossRef] [PubMed]
- Goetz, L.L.; Stiens, S.A. Abdominal Electric Stimulation Facilitates Penile Vibratory Stimulation for Ejaculation After Spinal Cord Injury: A Single-Subject Trial. Arch. Phys. Med. Rehabil. 2005, 86, 1879–1883. [Google Scholar] [CrossRef]
- Krassioukov, A.; Warburton, D.; Teasell, R.; Eng, J. A Systematic Review of the Management of Autonomic Dysreflexia After Spinal Cord Injury. Arch. Phys. Med. Rehabil. 2009, 90, 682–695. [Google Scholar] [CrossRef]
- Sheel, A.W.; Krassioukov, A.V.; Inglis, J.T.; Elliott, S.L. Autonomic dysreflexia during sperm retrieval in spinal cord injury: Influence of lesion level and sildenafil citrate. J. Appl. Physiol. 2005, 99, 53–58. [Google Scholar] [CrossRef]
- Gunn, R.M.C. Fertility in sheep: Artificial production of seminal ejaculation and the characteristics of the spermatozoa contained therein. Aust. Commonw. Counc. Sci. Ind. Res. 1936, 94, 1–5. [Google Scholar]
- Brindley, G.S. Electroejaculation: Its technique, neurological implications and uses. J. Neurol. Neurosurg. Psychiatry 1981, 44, 9–18. [Google Scholar] [CrossRef]
- Halstead, L.S.; Vervoort, S.; Seager, S.W.J. Rectal probe electrostimulation in the treatment of anejaculatory spinal cord injured men. Spinal Cord 1987, 25, 120–129. [Google Scholar] [CrossRef]
- Ibrahim, E.; Aballa, T.C.; Brackett, N.L.; Lynne, C.M. Electroejaculation in men with spinal cord injury: A step-by-step video demonstration. Fertil. Steril. 2021, 115, 1344–1346. [Google Scholar] [CrossRef]
- Brackett, N.; Ibrahim, E.; Lynne, C. Advances in the management of infertility in men with spinal cord injury. Asian J. Androl. 2016, 18, 382–390. [Google Scholar] [CrossRef]
- Engin-Ustun, Y.; Korkmaz, C.; Duru, N.K.; Başer, I. Comparison of three sperm retrieval techniques in spinal cord-injured men: Pregnancy outcome. Gynecol. Endocrinol. 2006, 22, 252–255. [Google Scholar] [CrossRef]
- Marina, S.; Marina, F.; Alcolea, R.; Nadal, J.; Pons, M.C.; Grossmann, M.; Expósito, R.; Vidal, J. Triplet pregnancy achieved through intracytoplasmic sperm injection with spermatozoa obtained by prostatic massage of a paraplegic patient: Case report. Hum. Reprod. 1999, 14, 1546–1548. [Google Scholar] [CrossRef][Green Version]
- Arafa, M.; Zohdy, W.A.; Shamloul, R. Prostatic massage: A simple method of semen retrieval in men with spinal cord injury. Int. J. Androl. 2007, 30, 170–173. [Google Scholar] [CrossRef]
- Karpman, E.; Williams, D.H. Techniques of sperm retrieval. In Infertility in the Male, 4th ed.; Lipshultz, L.I., Howards, S.S., Niederberger, C.S., Eds.; Cambridge University Press: Cambridge, UK, 2009. [Google Scholar]
- Goldstein, M.S.P.N. Surgical and Medical Management of Male Infertility, 1st ed.; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Iwahata, T.; Shin, T.; Shimomura, Y.; Suzuki, K.; Kobayashi, T.; Miyata, A.; Kobori, Y.; Soh, S.; Okada, H. Testicular sperm extraction for patients with spinal cord injury-related anejaculation: A single-center experience. Int. J. Urol. 2016, 23, 1024–1027. [Google Scholar] [CrossRef]
- Raviv, G.; Madgar, I.; Elizur, S.E.; Zeilig, G.; Levron, J. Testicular sperm retrieval and intra cytoplasmic sperm injection provide favorable outcome in spinal cord injury patients, failing conservative reproductive treatment. Spinal Cord 2013, 51, 642–644. [Google Scholar] [CrossRef]
- Utida, C.; Truzzi, J.C.; Bruschini, H.; Simonetti, R.; Cedenho, A.P.; Srougi, M.; Ortiz, V. Male infertility in spinal cord trauma. Int. Braz. J. Urol. 2005, 31, 375–383. [Google Scholar] [CrossRef]
- Linsenmeyer, T.A. Male infertility following spinal cord injury. J. Am. Paraplegia Soc. 1991, 14, 116–121. [Google Scholar] [CrossRef]
- Denil, J.; Ohl, D.A.; Menge, A.C.; Keller, L.M.; McCabe, M. Functional Characteristics of Sperm Obtained by Electroejaculation. J. Urol. 1992, 147, 69–72. [Google Scholar] [CrossRef]
- Kathiresan, A.S.Q.; Ibrahim, E.; Modh, R.A.; Aballa, T.C.; Lynne, C.M.; Brackett, N.L. Semen quality in ejaculates produced by masturbation in men with spinal cord injury. Spinal Cord 2012, 50, 891–894. [Google Scholar] [CrossRef][Green Version]
- Wieder, J.A.; Lynne, C.M.; Ferrell, S.M.; Aballa, T.C.; Brackett, N.L. Brown-colored semen in men with spinal cord injury. J. Androl. 1999, 20, 594–600. [Google Scholar]
- Brackett, N.L.; Lynne, C.M.; Weizman, M.S.; Bloch, W.E.; Padron, O.F. Scrotal and oral temperatures are not related to semen quality of serum gonadotropin levels in spinal cord-injured men. J. Androl. 1994, 15, 614–619. [Google Scholar] [PubMed]
- Læssøe, L.; Sønksen, J.; Bagi, P.; Biering-Sørensen, F.; Ohl, D.A.; McGUIRE, E.J.; Kristensen, J.K. Effects of ejaculation by penile vibratory stimulation on bladder reflex activity in a spinal cord injured man. J. Urol. 2001, 166, 627. [Google Scholar] [CrossRef] [PubMed]
- Siösteen, A.; Forssman, L.; Steen, Y.; Sullivan, L.; Wickström, I. Quality of semen after repeated ejaculation treatment in spinal cord injury men. Spinal Cord 1990, 28, 96–104. [Google Scholar] [CrossRef]
- Das, S.; Dodd, S.; Soni, B.M.; Sharma, S.D.; Gazvani, R.; Lewis-Jones, D.I. Does repeated electro-ejaculation improve sperm quality in spinal cord injured men? Spinal Cord 2006, 44, 753–756. [Google Scholar] [CrossRef]
- Ohl, D.A.; Denil, J.; Fitzgerald-Shelton, K.; McCabe, M.; McGuire, E.J.; Menge, A.C.; Randolph, J.F. Fertility of Spinal Cord Injured Males: Effect of Genitourinary Infection and Bladder Management on Results of Electroejaculation. J. Am. Paraplegia Soc. 1992, 15, 53–59. [Google Scholar] [CrossRef]
- Ohl, D.A.; Sonksen, J.; Menge, A.C.; McCabe, M.; Keller, L.M. Electroejaculation Versus Vibratory Stimulation in Spinal Cord Injured Men: Sperm Quality and Patient Preference. J. Urol. 1997, 157, 2147–2149. [Google Scholar] [CrossRef]
- Brackett, N.L.; Padron, O.F.; Lynne, C.M. Semen quality of spinal cord injured men is better when obtained by vibratory stimulation versus electroejaculation. J. Urol. 1997, 157, 151–157. [Google Scholar] [CrossRef]
- Brackett, N.L.; Nash, M.S.; Lynne, C.M. Male Fertility Following Spinal Cord Injury: Facts and Fiction. Phys. Ther. 1996, 76, 1221–1231. [Google Scholar] [CrossRef]
- Brackett, N.L.; Ferrell, S.M.; Aballa, T.C.; Amador, M.J.; Lynne, C.M. Semen quality in spinal cord injured men: Does it progressively decline post-injury? Arch. Phys. Med. Rehabil. 1998, 79, 625–628. [Google Scholar] [CrossRef]
- Iremashvili, V.; Brackett, N.; Ibrahim, E.; Aballa, T.C.; Lynne, C.M. Semen Quality Remains Stable During the Chronic Phase of Spinal Cord Injury: A Longitudinal Study. J. Urol. 2010, 184, 2073–2077. [Google Scholar] [CrossRef]
- Basu, S.; Lynne, C.M.; Ruiz, P.; Aballa, T.C.; Ferrell, S.M.; Brackett, N.L. Cytofluorographic identification of activated T-cell subpopulations in the semen of men with spinal cord injuries. J. Androl. 2002, 23, 551–556. [Google Scholar] [PubMed]
- Aird, I.A.; Vince, G.S.; Bates, M.D.; Johnson, P.M.; Lewis-Jones, I.D. Leukocytes in semen from men with spinal cord injuries. Fertil. Steril. 1999, 72, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Trabulsi, E.J.; Shupp-Byrne, D.; Sedor, J.; Hirsch, I.H. Leukocyte subtypes in electroejaculates of spinal cord injured men. Arch. Phys. Med. Rehabil. 2002, 83, 31–34. [Google Scholar] [CrossRef]
- Parham, P. The Immune System, 2nd ed.; Garland Science: New York, NY, USA, 2005. [Google Scholar]
- Hoek, J.B.; Pastorino, J.G. Cellular signaling mechanisms in alchohol-induced liver damage. Sem. Liver Dis. 2004, 24, 257–272. [Google Scholar] [CrossRef]
- Yamaoka, J.; Kabashima, K.; Kawanishi, M.; Toda, K.M. Cytotoxicity of IFN-gamma and TNF-alpha for vascular endothelial cell is mediated by nitric oxide. Biochem. Biophys. Res. Commun. 2002, 291, 780–786. [Google Scholar] [CrossRef]
- Basu, S.; Aballa, T.C.; Ferrell, S.M.; Lynne, C.M.; Brackett, N.L. Inflammatory Cytokine Concentrations Are Elevated in Seminal Plasma of Men with Spinal Cord Injuries. J. Androl. 2004, 25, 250–254. [Google Scholar] [CrossRef]
- Cohen, D.R.; Basu, S.; Randall, J.M.; Aballa, T.C.; Lynne, C.M.; Brackett, N.L. Sperm Motility in Men with Spinal Cord Injuries Is Enhanced by Inactivating Cytokines in the Seminal Plasma. J. Androl. 2004, 25, 922–925. [Google Scholar] [CrossRef]
- Brackett, N.L.; Cohen, D.R.; Ibrahim, E.; Aballa, T.C.; Lynne, C.M. Neutralization of Cytokine Activity at the Receptor Level Improves Sperm Motility in Men with Spinal Cord Injuries. J. Androl. 2007, 28, 717–721. [Google Scholar] [CrossRef]
- Zhang, X.; Ibrahim, E.; Vaccari, J.P.D.R.; Lotocki, G.; Aballa, T.C.; Dietrich, W.D.; Keane, R.W.; Lynne, C.M.; Brackett, N.L. Involvement of the inflammasome in abnormal semen quality of men with spinal cord injury. Fertil. Steril. 2012, 99, 118–124.e2. [Google Scholar] [CrossRef]
- Ibrahim, E.; Aballa, T.C.; Lynne, C.M.; Brackett, N.L. Oral probenecid improves sperm motility in men with spinal cord injury. J. Spinal Cord Med. 2018, 41, 567–570. [Google Scholar] [CrossRef]
- Kathiresan, A.S.; Ibrahim, E.; Aballa, T.C.; Attia, G.R.; Lynne, C.M.; Brackett, N.L. Pregnancy outcomes by intravaginal and intrauterine insemination in 82 couples with male factor infertility due to spinal cord injuries. Fertil. Steril. 2011, 96, 328–331. [Google Scholar] [CrossRef] [PubMed]
- Leduc, B.E. Treatment of infertility in 31 men with spinal cord injury. Can. J. Urol. 2012, 19, 6432–6436. [Google Scholar] [PubMed]
- Sønksen, J.; Fode, M.; Löchner-Ernst, D.; Ohl, D.A. Vibratory ejaculation in 140 spinal cord injured men and home insemination of their partners. Spinal Cord 2011, 50, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Muthigi, A.; Jahandideh, S.; Bishop, L.A.; Naeemi, F.K.; Shipley, S.K.; O’Brien, J.E. Clarifying the relationship between total motile sperm counts and intrauterine insemination pregnancy rates. Fertil Steril 2021, 115, 1454–1460. [Google Scholar] [CrossRef]
- Hamilton, J.A.M.; Cissen, M.; Brandes, M.; Smeenk, J.M.J.; de Bruin, J.P.; Kremer, J.A.M.; Nelen, W.L.D.M.; Hamilton, C.J.C.M. Total motile sperm count: A better indicator for the severity of male factor infertility than the WHO sperm classification system. Hum. Reprod. 2015, 30, 1110–1121. [Google Scholar] [CrossRef]
- Kathiresan, A.S.; Ibrahim, E.; Aballa, T.C.; Attia, G.R.; Ory, S.J.; Hoffman, D.I.; Maxson, W.S.; Barrionuevo, M.J.; Lynne, C.M.; Brackett, N.L. Comparison of in vitro fertilization/intracytoplasmic sperm injection outcomes in male factor infertility patients with and without spinal cord injuries. Fertil. Steril. 2011, 96, 562–566. [Google Scholar] [CrossRef]
- Carcieri, E.M.; Mona, L.R. Assessment and treatment of sexual health issues in rehabilitation: A patient-centered approach. In Practical Psychology in Medical Rehabilitation; Budd, M.A., Hough, S., Wegener, S.T., Stiers, W., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 287–294. [Google Scholar]
- Mona, L.R.; Syme, M.L.; Cameron, R.P.; Fraley, S.; Smith, K.; Bagget, L. Sexuality and disability: A disability affirmative approach to sex therapy. In Principles and Practices of Sex Therapy, 5th ed.; Binik, Y., Hall, K., Eds.; Guilford Press: New York, NY, USA, 2014; pp. 457–481. [Google Scholar]
- Tepper, M.S.; Owens, A.F. Volume 3: Moral and Cultural Foundations. In Sexual Health; Praeger: Westport, CT, USA, 2007. [Google Scholar]
- Hess, M.; Hicks, T.A.; Hough, S. Sex and chronic physical illness. In The Textbook of Clinical Sexual Medicine; Ishak, W.W., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 479–506. [Google Scholar]
- Hough, S.; Cordes, C.C.; Goetz, L.L.; Kuemmel, A.; Lieberman, J.A.; Mona, L.R.; Tepper, M.S.; Varghese, J.G. A Primary Care Provider’s Guide to Sexual Health for Individuals with Spinal Cord Injury. Top. Spinal Cord Inj. Rehabil. 2020, 26, 144–151. [Google Scholar] [CrossRef]
- Collier, L. Seeking Intimacy People with Physical Disabilities Fight Hurtful Stereotypes When Looking for Relationship Partners; 2017; 48(11). American Psychological Association. Available online: https://www.apa.org/monitor/2017/12/seeking-intimacy (accessed on 20 October 2022).
- Kroll, K.; Klein, E.L. Enabling Romance: A Guide to Love, Sex, and Relationships for People with Disabilities; No Limits Communications: Baltimore, MD, USA, 2001; p. 86. [Google Scholar]
- Tepper, M. Regain That Feeling: Secrets to Sexual Self-Discovery: People Living with Spinal Cord Injuries Share Profound Insights into Sex, Pleasure, Relationships, Orgasm, and the Importance of Connectedness; CreateSpace Independent Publishing Platform: Scotts Valley, CA, USA, 2015. [Google Scholar]
- Kassioukov, A.; MacHattie, E.; Naphtali, K.; Miller, W.C.; Elliott, S. Disabilities Health Research Network. PleasureABLE: Sexual Device Manual for Persons with Disabilities; University of British Columbia: Vancouver, BC, Canada, 2009. [Google Scholar]
- Hospital, M.S. Sex, Love and Intimacy after Spinal Cord Injury 2018. Available online: www.sexualitysci.org (accessed on 20 October 2022).
- The Miami Project to Cure Paralysis: University of Miami Miller School of Medicine. Available online: https://www.themiamiproject.org/ (accessed on 20 October 2022).
- United Spinal Association. Available online: https://unitedspinal.org/ (accessed on 20 October 2022).
- Christopher & Dana Reeve Foundation. Available online: https://www.christopherreeve.org/ (accessed on 20 October 2022).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zizzo, J.; Gater, D.R.; Hough, S.; Ibrahim, E. Sexuality, Intimacy, and Reproductive Health after Spinal Cord Injury. J. Pers. Med. 2022, 12, 1985. https://doi.org/10.3390/jpm12121985
Zizzo J, Gater DR, Hough S, Ibrahim E. Sexuality, Intimacy, and Reproductive Health after Spinal Cord Injury. Journal of Personalized Medicine. 2022; 12(12):1985. https://doi.org/10.3390/jpm12121985
Chicago/Turabian StyleZizzo, John, David R. Gater, Sigmund Hough, and Emad Ibrahim. 2022. "Sexuality, Intimacy, and Reproductive Health after Spinal Cord Injury" Journal of Personalized Medicine 12, no. 12: 1985. https://doi.org/10.3390/jpm12121985
APA StyleZizzo, J., Gater, D. R., Hough, S., & Ibrahim, E. (2022). Sexuality, Intimacy, and Reproductive Health after Spinal Cord Injury. Journal of Personalized Medicine, 12(12), 1985. https://doi.org/10.3390/jpm12121985







