External Validation of COVID-19 Risk Scores during Three Waves of Pandemic in a German Cohort—A Retrospective Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
2.2. Study Population
- Patients not hospitalized for COVID-19 disease;
- Patients with a patient decree determining a DNR/DNI (do not resuscitate/do not intubate) situation;
- Patients transferred to our ICU from other hospitals, for example, due to the need for extracorporeal membrane oxygenation (ECMO).
2.3. Definition of Cohorts
2.4. Data Collection and Score Validation
2.5. Statistical Analysis
3. Results
3.1. Study Population
3.2. Comparison of Cohorts
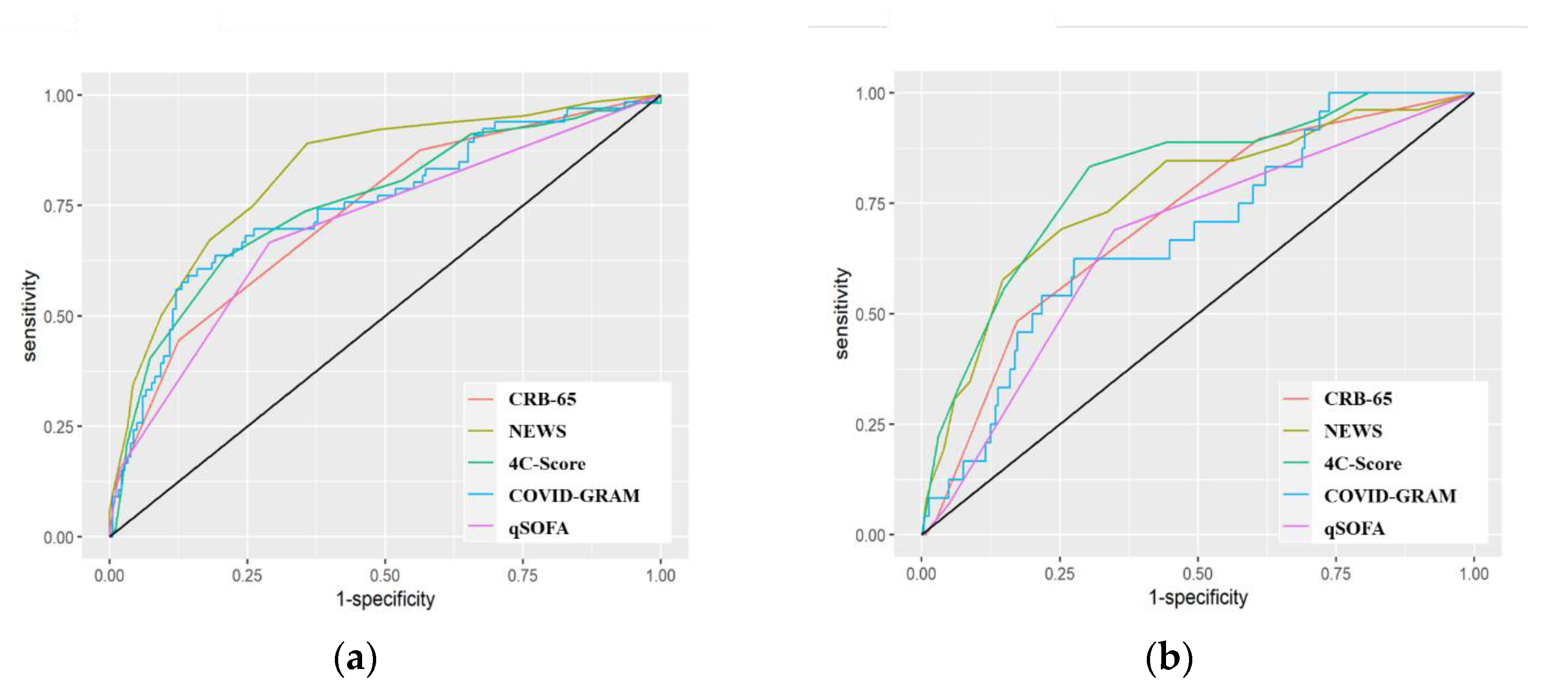
3.3. Predictive Performance of the Scores
3.4. Comparison between the Cohorts
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
References
- Cram, P.; Anderson, M.L.; Shaughnessy, E.E. All Hands on Deck: Learning to “Un-specialize” in the COVID-19 Pandemic. J. Hosp. Med. 2020, 15, 314–315. [Google Scholar] [CrossRef]
- Liang, W.; Liang, H.; Ou, L.; Chen, B.; Chen, A.; Li, C.; Li, Y.; Guan, W.; Sang, L.; Lu, J.; et al. Development and Validation of a Clinical Risk Score to Predict the Occurrence of Critical Illness in Hospitalized Patients With COVID-19. JAMA Intern. Med. 2020, 180, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Knight, S.R.; Ho, A.; Pius, R.; Buchan, I.; Carson, G.; Drake, T.M.; Dunning, J.; Fairfield, C.J.; Gamble, C.; Green, C.A.; et al. Risk stratification of patients admitted to hospital with COVID19 using the ISARIC WHO Clinical Characterisation Protocol: Development and validation of the 4C Mortality Score. BMJ 2020, 370, m3339. [Google Scholar] [CrossRef]
- Armiñanzas, C.; Arnaiz de Las Revillas, F.; Gutiérrez Cuadra, M.; Arnaiz, A.; Fernández Sampedro, M.; González-Rico, C.; Ferrer, D.; Mora, V.; Suberviola, B.; Latorre, M.; et al. Usefulness of the COVID-GRAM and CURB-65 scores for predicting severity in patients with COVID-19. Int. J. Infect. Dis. 2021, 108, 282–288. [Google Scholar] [CrossRef]
- De Socio, G.V.; Gidari, A.; Sicari, F.; Palumbo, M.; Francisci, D. National Early Warning Score 2 (NEWS2) better predicts critical Coronavirus Disease 2019 (COVID-19) illness than COVID-GRAM, a multi-centre study. Infection 2021, 49, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.; Pitre, T.; Junek, M.; Kapralik, J.; Patel, R.; Feng, E.; Dawson, L.; Tsang, J.L.Y.; Duong, M.; Ho, T.; et al. External validation of the 4C mortality score among COVID-19 patients admitted to hospital in Ontario, Canada: A retrospective study. Sci. Rep. 2021, 11, 18638. [Google Scholar] [CrossRef] [PubMed]
- Covino, M.; de Matteis, G.; Burzo, M.L.; Russo, A.; Forte, E.; Carnicelli, A.; Piccioni, A.; Simeoni, B.; Gasbarrini, A.; Franceschi, F.; et al. Predicting In-Hospital Mortality in COVID-19 Older Patients with Specifically Developed Scores. J. Am. Geriatr. Soc. 2021, 69, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Satici, C.; Demirkol, M.A.; Sargin Altunok, E.; Gursoy, B.; Alkan, M.; Kamat, S.; Demirok, B.; Surmeli, C.D.; Calik, M.; Cavus, Z.; et al. Performance of pneumonia severity index and CURB-65 in predicting 30-day mortality in patients with COVID-19. Int. J. Infect. Dis. 2020, 98, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Gude-Sampedro, F.; Fernández-Merino, C.; Ferreiro, L.; Lado-Baleato, Ó.; Espasandín-Domínguez, J.; Hervada, X.; Cadarso, C.M.; Valdés, L. Development and validation of a prognostic model based on comorbidities to predict COVID-19 severity: A population-based study. Int. J. Epidemiol. 2021, 50, 64–74. [Google Scholar] [CrossRef]
- Doğanay, F.; Ak, R. Performance of the CURB-65, ISARIC-4C and COVID-GRAM scores in terms of severity for COVID-19 patients. Int. J. Clin. Pract. 2021, 75, e14759. [Google Scholar] [CrossRef]
- Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Dexamethasone in Hospitalized Patients with COVID19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Boehm, E.; Kronig, I.; Neher, R.A.; Eckerle, I.; Vetter, P.; Kaiser, L. Novel SARS-CoV-2 variants: The pandemics within the pandemic. Clin. Microbiol. Infect. 2021, 27, 1109–1117. [Google Scholar] [CrossRef] [PubMed]
- Kluge, S.; Janssens, U.; Welte, T.; Weber-Carstens, S.; Schälte, G.; Spinner, C.D.; Malin, J.J.; Gastmeier, P.; Langer, F.; Bracht, H.; et al. S3-Leitlinie—Empfehlungen zur Stationären Therapie von Patienten Mit COVID-19. Available online: https://www.awmf.org/uploads/tx_szleitlinien/113-001LGl_S3_Empfehlungen-zur-stationaeren-Therapie-von-Patienten-mit-COVID-19_2022-03.pdf (accessed on 14 April 2022).
- Schilling, J.; Buda, S.; Fischer, M.; Goerlitz, L.; Grote, U.; Haas, W.; Hamouda, O.; Prahm, K.; Tolksdorf, K. Retrospektive Phaseneinteilung der COVID-19-Pandemie in Deutschland bis Februar 2021; Robert Koch-Institut: Berlin, Germany, 2021. [CrossRef]
- Hippisley-Cox, J.; Coupland, C.A.; Mehta, N.; Keogh, R.H.; Diaz-Ordaz, K.; Khunti, K.; Lyons, R.A.; Kee, F.; Sheikh, A.; Rahman, S.; et al. Risk prediction of COVID19 related death and hospital admission in adults after COVID19 vaccination: National prospective cohort study. BMJ 2021, 374, n2244. [Google Scholar] [CrossRef] [PubMed]
- Schmid-Küpke, N.K.; Neufeind, J.; Wichmann, O.; Siedler, A. COVIMO: COVID-19 Vaccination Rate Monitoring in Germany. Available online: https://www.rki.de/EN/Content/infections/epidemiology/outbreaks/COVID-19/projects/covimo.html (accessed on 28 April 2022).
- Carpenter, J.; Bithell, J. Bootstrap confidence intervals: When, which, what? A practical guide for medical statisticians. Stat. Med. 2000, 19, 1141–1164. [Google Scholar] [CrossRef]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the Areas under Two or More Correlated Receiver Operating Characteristic Curves: A Nonparametric Approach. Biometrics 1988, 44, 837. [Google Scholar] [CrossRef]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.-C.; Müller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Hadley, W. Ggplot2: Elegrant Graphics for Data Analysis, 2nd ed.; Springer: Basel, Switzerland, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Davison, A.C.; Hinkley, D.V. Bootstrap Methods and Their Applications, Repr; Cambridge University Press: Cambridge, UK, 1999; ISBN 0-521-57391-2. [Google Scholar]
- Saito, T.; Rehmsmeier, M. Precrec: Fast and accurate precision-recall and ROC curve calculations in R. Bioinformatics 2017, 33, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Candel, B.G.; Duijzer, R.; Gaakeer, M.I.; ter Avest, E.; Sir, Ö.; Lameijer, H.; Hessels, R.; Reijnen, R.; van Zwet, E.W.; de Jonge, E.; et al. The association between vital signs and clinical outcomes in emergency department patients of different age categories. Emerg. Med. J. 2022. [Google Scholar] [CrossRef] [PubMed]
- Tobin, M.J.; Laghi, F.; Jubran, A. Why COVID-19 Silent Hypoxemia Is Baffling to Physicians. Am. J. Respir. Crit. Care Med. 2020, 202, 356–360. [Google Scholar] [CrossRef]
- .Karagiannidis, C.; Windisch, W.; McAuley, D.F.M.; Welte, T.; Busse, R. Major differences in ICU admissions during the first and second COVID-19 wave in Germany. Lancet Respir. Med. 2021, 9, e47–e48. [Google Scholar] [CrossRef]
- Karagiannidis, C.; Mostert, C.; Hentschker, C.; Voshaar, T.; Malzahn, J.; Schillinger, G.; Klauber, J.; Janssens, U.; Marx, G.; Weber-Carstens, S.; et al. Case characteristics, resource use, and outcomes of 10 021 patients with COVID-19 admitted to 920 German hospitals: An observational study. Lancet Respir. Med. 2020, 8, 853–862. [Google Scholar] [CrossRef]
- Long, B.; Carius, B.M.; Chavez, S.; Liang, S.Y.; Brady, W.J.; Koyfman, A.; Gottlieb, M. Clinical update on COVID-19 for the emergency clinician: Presentation and evaluation. Am. J. Emerg. Med. 2022, 54, 46–57. [Google Scholar] [CrossRef] [PubMed]

| Cohort 1 | Cohort 2 | Cohort 3 | |
|---|---|---|---|
| Admission period | 1 March 2020–30 June 2020 | 1 July 2020–7 March 2021 | 8 March 2021–30 May 2021 |
| Variants | High prevalence of wild type | High prevalence of B.1.177 | High prevalence of alpha variant |
| Specific therapy | None | Remdesivir, steroids | Remdesivir, steroids |
| Vaccination | None | Completely vaccinated: 0.9–3.3% * (vaccination available since 26 December 2020) | Completely vaccinated: 6.9–27.4% * |
| qSOFA | CRB-65 | NEWS | COVID-GRAM | 4C-Score |
|---|---|---|---|---|
| Respiratory rate Consciousness Blood pressure | Confusion Respiratory rate Blood pressure Age | Respiratory rate Oxygen saturation Suppl. oxygen Temperature Blood pressure Heart rate Consciousness | X-ray abnormality Age Hemoptysis Dyspnea Unconsciousness Number of comorbidities Cancer history Neutrophil/lymphocytes Lactate dehydrogenase Direct bilirubin | Age Sex at birth Number of comorbidities Respiratory rate Oxygen saturation Glasgow Coma Scale Urea CRP level |
| Characteristics | Overall No (%) or Mean (Q1–Q3) | First Cohort No (%) or Mean (Q1–Q3) | Second Cohort No (%) or Mean (Q1–Q3) | Third Cohort No (%) or Mean (Q1–Q3) | p-Value * |
|---|---|---|---|---|---|
| Study population | 347 (100) | 134 (38.6) | 187 (53.9) | 26 (7.5) | - |
| In-hospital mortality | 32 (9.2) | 23 (17.2) | 9 (4.8) | 0 (0) | 0.044 |
| ICU admission | 82 (23.6) | 50 (37.3) | 30 (16.0) | 2 (7.7) | 0.002 |
| Average age | 65.4 (57.0–78.0) | 67.0 (58.5–80.0) | 65.1 (57.0–76.0) | 59.6 (51.8–68.3) | 1 |
| Women | 153 (44.1) | 57 (42.5) | 84 (44.9) | 12 (46.2) | 1 |
| COVID-19 vaccination | 1 (0.3) | 0 (0) | 0 (0) | 1 (3.8) | - |
| Respiratory disease | 72 (20.7) | 22 (16.4) | 44 (23.5) | 6 (23.1) | 1 |
| Cardiovascular disease | 246 (70.9) | 99 (73.9) | 131 (70.1) | 16 (61.5) | 1 |
| Diabetes | 82 (23.6) | 32 (23.9) | 46 (24.6) | 4 (15.3) | 1 |
| Hypertension | 204 (58.8) | 86 (64.2) | 106 (56.7) | 12 (46.2) | 1 |
| Liver disease | 27 (7.8) | 10 (7.5) | 12 (6.4) | 5 (19.2) | 1 |
| Chronic kidney disease | 41 (11.8) | 12 (9.7) | 25 (13.4) | 3 (11.5) | 1 |
| HIV or AIDS | 0 (0) | 0 (0) | 0 (0) | 0 (0) | - |
| Organ transplantation | 11 (3.2) | 4 (3.0) | 6 (3.2) | 1 (3.8) | 1 |
| Malignancy (active) | 45 (13.0) | 9 (6.7) | 34 (18.2) | 2 (7.7) | 0.236 |
| Malignancy (history) | 29 (8.4) | 20 (14.9) | 7 (3.7) | 2 (7.7) | 0.070 |
| Neurological conditions | 79 (22.8) | 29 (21.6) | 44 (23.5) | 6 (23.1) | 1 |
| COVID-19 findings | 173 (49.9) | 82 (61.2) | 79 (42.2) | 12 (46.2) | 0.272 |
| Oxygen saturation (%) | 93.4 (92.0–97.0) | 91.9 (90.0–96.0) | 94.4 (93.0–97.0) | 94.2 (93.0–97.0) | 0.034 |
| Heart rate (/min) | 86.5 (74.0–96.0) | 88.9 (76.0–100.0) | 85.1 (72.0–94.0) | 83.4 (69.0–91.0) | 1 |
| Respiratory rate (/min) | 21.1 (16.0–24.0) | 22.4 (17.0–26.0) | 20.6 (16.3–24.0) | 17.8 (15.0–20.0) | 1 |
| Syst. Blood pressure (mmHg) | 132.0 (118.0–145.0) | 129.3 (110.0–140.0) | 133.8 (120.0–148.0) | 127.1 (116.3–138.3) | 1 |
| Temperature (°C) | 37.1 (36.4–37.7) | 37.3 (36.5–38.1) | 36.8 (36.2–37.4) | 37.5 (36.8–38.3) | 0.036 |
| GCS | 14.8 (15.0–15.0) | 14.8 (15.0–15.0) | 14.9 (15.0–15.0) | 14.8 (15.0–15.0) | 1 |
| Dyspnea | 184 (53.0) | 71 (53.0) | 103 (55.1) | 10 (38.5) | 1 |
| Cough | 179 (51.6) | 84 (62.7) | 88 (47.1) | 7 (26.9) | 0.751 |
| Fever | 181 (52.2) | 86 (64.2) | 80 (42.8) | 15 (57.7) | 0.026 |
| Leukocyte count (/µL) | 6828.2 (4355.0–8355.0) | 6586.8 (4385.0–7970.0) | 7064.0 (4305.0–8575.0) | 6389.1 (4543.0–7665.0) | 1 |
| Neutrophil count (103/µL) | 5.2 (3.0–6.4) | 4.9 (3.1–6.3) | 5.4 (2.9–6.8) | 5.3 (3.4–6.4) | 1 |
| Lymphocyte count (103/µL) | 1.1 (0.6–1.2) | 1.0 (0.6–1.1) | 1.3 (0.6–1.3) | 1.0 (1.0–1.2) | 1 |
| Urea (mmol/L) | 49.5 (27.0–62.0) | 47.1 (24.8–62.0) | 52.2 (28.0–65.0) | 43.5 (29.0–56.8) | 1 |
| CRP (mg/dL) | 8.0 (1.0–11.9) | 8.9 (2.6–13.3) | 7.7 (1.7–11.2) | 6.3 (1.9–11.5) | 1 |
| Bilirubin (mg/dL) | 0.7 (0.4–0.8) | 0.7 (0.5–0.9) | 0.6 (0.4–0.8) | 0.6 (0.4–0.8) | 0.507 |
| LDH (U/L) | 346.6 (229.8–378.5) | 328.6 (229.5–389.0) | 361.4 (225.0–365.5 | 344.0 (240.0–411.0) | 1 |
| Steroids | 146 (42.1) | 24 (17.9) | 103 (59.9) | 19 (73.1) | - |
| Remdesivir | 131 (37.8) | 0 (0) | 112 (55.1) | 19 (73.1) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Häger, L.; Wendland, P.; Biergans, S.; Lederer, S.; de Arruda Botelho Herr, M.; Erhardt, C.; Schmauder, K.; Kschischo, M.; Malek, N.P.; Bunk, S.; et al. External Validation of COVID-19 Risk Scores during Three Waves of Pandemic in a German Cohort—A Retrospective Study. J. Pers. Med. 2022, 12, 1775. https://doi.org/10.3390/jpm12111775
Häger L, Wendland P, Biergans S, Lederer S, de Arruda Botelho Herr M, Erhardt C, Schmauder K, Kschischo M, Malek NP, Bunk S, et al. External Validation of COVID-19 Risk Scores during Three Waves of Pandemic in a German Cohort—A Retrospective Study. Journal of Personalized Medicine. 2022; 12(11):1775. https://doi.org/10.3390/jpm12111775
Chicago/Turabian StyleHäger, Lukas, Philipp Wendland, Stephanie Biergans, Simone Lederer, Marius de Arruda Botelho Herr, Christian Erhardt, Kristina Schmauder, Maik Kschischo, Nisar Peter Malek, Stefanie Bunk, and et al. 2022. "External Validation of COVID-19 Risk Scores during Three Waves of Pandemic in a German Cohort—A Retrospective Study" Journal of Personalized Medicine 12, no. 11: 1775. https://doi.org/10.3390/jpm12111775
APA StyleHäger, L., Wendland, P., Biergans, S., Lederer, S., de Arruda Botelho Herr, M., Erhardt, C., Schmauder, K., Kschischo, M., Malek, N. P., Bunk, S., Bitzer, M., Gladstone, B. P., & Göpel, S. (2022). External Validation of COVID-19 Risk Scores during Three Waves of Pandemic in a German Cohort—A Retrospective Study. Journal of Personalized Medicine, 12(11), 1775. https://doi.org/10.3390/jpm12111775







