MRI Outcomes Achieved by Simple Flow Blockage Technique in Symptomatic Carotid Artery Stenosis Stenting
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Inclusion Criteria
2.3. Patient Management
2.3.1. Medical Management
2.3.2. Simple Flow Blockage (SFB) Technique
2.3.3. Follow-Up and Outcome
2.4. MRI Evaluation
2.5. Data Collection
2.6. Statistical Analysis
3. Results
3.1. Patient Characteristics and Angiographic Patterns
3.2. Clinical and DWI Outcomes
4. Discussion
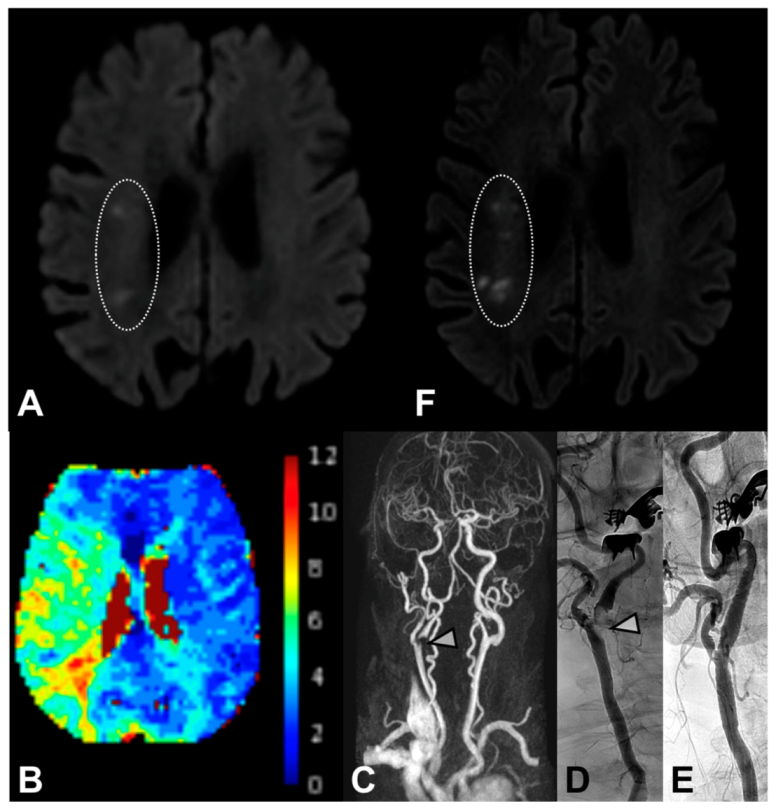
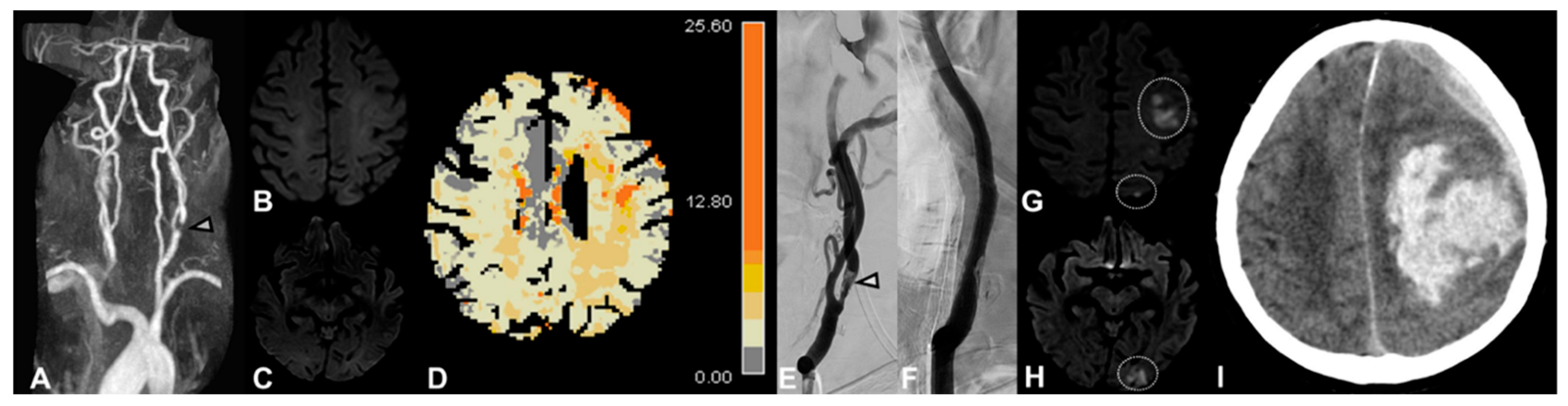
4.1. DWI Lesions
4.2. Irregular/Ulcerated Stenosis and Degree of Stenosis
4.3. Timing of Angioplasty
4.4. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
Appendix A.1. Data Collection
Appendix A.2. Patients’ Management
Appendix A.2.1. Medical Management and Simple Flow Blockage Technique (SFB)
Appendix A.2.2. Follow-Up and Outcome
Appendix B
References
- North American Symptomatic Carotid Endarterectomy Trial Collaborators. Beneficial Effect of Carotid Endarterectomy in Symptomatic Patients with High-Grade Carotid Stenosis. N. Engl. J. Med. 1991, 325, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Kernan, W.N.; Ovbiagele, B.; Black, H.R.; Bravata, D.M.; Chimowitz, M.I.; Ezekowitz, M.D.; Fang, M.C.; Fisher, M.; Furie, K.L.; Heck, D.V.; et al. Guidelines for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke 2014, 45, 2160–2236. [Google Scholar] [CrossRef]
- Mas, J.-L.; Chatellier, G.; Beyssen, B.; Branchereau, A.; Moulin, T.; Becquemin, J.-P.; Larrue, V.; Lièvre, M.; Leys, D.; Bonneville, J.-F.; et al. Endarterectomy versus Stenting in Patients with Symptomatic Severe Carotid Stenosis. N. Engl. J. Med. 2006, 355, 1660–1671. [Google Scholar] [CrossRef] [PubMed]
- Ringleb, A.P.; Allenberg, J.R.; Bruckmann, H.; Eckstein, H.H.; Fraedrich, G.; Hartmann, M.; Hennerici, M.G.; Jansen, O.; Klein, E.G.; Kunze, A.; et al. 30 day results from the SPACE trial of stent-protected angioplasty versus carotid endarterectomy in symptomatic patients: A randomised non-inferiority trial. Lancet 2006, 368, 1239–1247. [Google Scholar] [CrossRef]
- International Carotid Stenting Study investigators; Ederle, J.; Dobson, J.; Featherstone, R.L.; Bonati, L.H.; Van Der Worp, H.B.; De Borst, G.J.; Lo, T.H.; Gaines, P.; Dorman, P.J.; et al. Carotid artery stenting compared with endarterectomy in patients with symptomatic carotid stenosis (International Carotid Stenting Study): An interim analysis of a randomised controlled trial. Lancet 2010, 375, 985–997. [Google Scholar] [CrossRef] [PubMed]
- Carotid Stenting Trialists’ Collaboration; Bonati, L.H.; Dobson, J.; Algra, A.; Branchereau, A.; Chatellier, G.; Fraedrich, G.; Mali, W.P.; Zeumer, H.; Brown, M.M.; et al. Short-term outcome after stenting versus endarterectomy for symptomatic carotid stenosis: A preplanned meta-analysis of individual patient data. Lancet 2010, 376, 1062–1073. [Google Scholar] [CrossRef]
- Haussen, D.C.; Grossberg, J.A.; Bouslama, M.; Pradilla, G.; Belagaje, S.; Bianchi, N.; Allen, J.W.; Frankel, M.; Nogueira, R.G. Carotid Web (Intimal Fibromuscular Dysplasia) Has High Stroke Recurrence Risk and Is Amenable to Stenting. Stroke 2017, 48, 3134–3137. [Google Scholar] [CrossRef]
- Sivan-Hoffmann, R.; Gory, B.; Armoiry, X.; Goyal, M.; Riva, R.; Lab, C.; Lukaszewicz, A.-C.; Lehot, J.-J.; Derex, L.; Turjman, F. Stent-Retriever Thrombectomy for Acute Anterior Ischemic Stroke with Tandem Occlusion: A Systematic Review and Meta-Analysis. Eur. Radiol. 2016, 27, 247–254. [Google Scholar] [CrossRef]
- Omran, J.; Mahmud, E.; White, C.; Aronow, H.D.; Drachman, D.E.; Gray, W.; Abdullah, O.; Abu-Fadel, M.; Firwana, B.; Mishkel, G.; et al. Proximal balloon occlusion versus distal filter protection in carotid artery stenting: A meta-analysis and review of the literature. Catheter. Cardiovasc. Interv. 2016, 89, 923–931. [Google Scholar] [CrossRef]
- Dargazanli, C.; Mahmoudi, M.; Cappucci, M.; Collemiche, F.-L.; Labreuche, J.; Habza, O.; Gascou, G.; Lefèvre, P.-H.; Eker, O.; Mourand, I.; et al. Angiographic Patterns and Outcomes Achieved by Proximal Balloon Occlusion in Symptomatic Carotid Artery Stenosis Stenting. Clin. Neuroradiol. 2019, 30, 363–372. [Google Scholar] [CrossRef]
- Dietz, A.; Berkefeld, J.; Theron, J.G.; Schmitz-Rixen, T.; Zanella, F.E.; Turowski, B.; Steinmetz, H.; Sitzer, M. Endovascular Treatment of Symptomatic Carotid Stenosis Using Stent Placement. Stroke 2001, 32, 1855–1859. [Google Scholar] [CrossRef] [PubMed]
- Voeks, J.H.; Howard, G.; Roubin, G.S.; Malas, M.B.; Cohen, D.J.; Sternbergh, I.W.C.; Aronow, H.D.; Eskandari, M.K.; Sheffet, A.J.; Lal, B.K.; et al. Age and Outcomes After Carotid Stenting and Endarterectomy. Stroke 2011, 42, 3484–3490. [Google Scholar] [CrossRef]
- Brott, T.G.; Hobson, R.W.; Howard, G.; Roubin, G.S.; Clark, W.M.; Brooks, W.; Mackey, A.; Hill, M.D.; Leimgruber, P.P.; Sheffet, A.J.; et al. Stenting versus Endarterectomy for Treatment of Carotid-Artery Stenosis. N. Engl. J. Med. 2010, 363, 11–23. [Google Scholar] [CrossRef]
- Brott, T.G.; Howard, G.; Roubin, G.S.; Meschia, J.F.; Mackey, A.; Brooks, W.; Moore, W.S.; Hill, M.D.; Mantese, V.A.; Clark, W.M.; et al. Long-Term Results of Stenting versus Endarterectomy for Carotid-Artery Stenosis. N. Engl. J. Med. 2016, 374, 1021–1031. [Google Scholar] [CrossRef] [PubMed]
- Sardar, P.; Chatterjee, S.; Aronow, H.D.; Kundu, A.; Ramchand, P.; Mukherjee, D.; Nairooz, R.; Gray, W.A.; White, C.J.; Jaff, M.R.; et al. Carotid Artery Stenting Versus Endarterectomy for Stroke Prevention. J. Am. Coll. Cardiol. 2017, 69, 2266–2275. [Google Scholar] [CrossRef] [PubMed]
- Knappich, C.; Kuehnl, A.; Tsantilas, P.; Schmid, S.; Breitkreuz, T.; Kallmayer, M.; Zimmermann, A.; Eckstein, H.-H. The Use of Embolic Protection Devices Is Associated With a Lower Stroke and Death Rate After Carotid Stenting. JACC Cardiovasc. Interv. 2017, 10, 1257–1265. [Google Scholar] [CrossRef] [PubMed]
- Cole, T.S.; Mezher, A.W.; Catapano, J.S.; Godzik, J.; Baranoski, J.F.; Nakaji, P.; Albuquerque, F.C.; Lawton, M.T.; Little, A.S.; Ducruet, A.F. Nationwide Trends in Carotid Endarterectomy and Carotid Artery Stenting in the Post-CREST Era. Stroke 2020, 51, 579–587. [Google Scholar] [CrossRef]
- Brott, T.G.; Halperin, J.L.; Abbara, S.; Bacharach, J.M.; Barr, J.D.; Bush, R.L.; Cates, C.U.; Creager, M.A.; Fowler, S.B.; Friday, G.; et al. 2011 ASA/ACCF/AHA/AANN/AANS/ACR/ASNR/CNS/SAIP/SCAI/SIR/SNIS/SVM/SVS Guideline on the Management of Patients With Extracranial Carotid and Vertebral Artery Disease: Executive Summary. Circulation 2011, 124, 489–532. [Google Scholar] [CrossRef]
- Bonati, L.H.; Jongen, L.M.; Haller, S.; Flach, H.Z.; Dobson, J.; Nederkoorn, P.J.; Macdonald, S.; Gaines, A.P.; Waaijer, A.; Stierli, P.; et al. New ischaemic brain lesions on MRI after stenting or endarterectomy for symptomatic carotid stenosis: A substudy of the International Carotid Stenting Study (ICSS). Lancet Neurol. 2010, 9, 353–362. [Google Scholar] [CrossRef]
- Schnaudigel, S.; Gröschel, K.; Pilgram, S.M.; Kastrup, A. New Brain Lesions after Carotid Stenting versus Carotid Endarterectomy. Stroke 2008, 39, 1911–1919. [Google Scholar] [CrossRef]
- Gensicke, H.; Zumbrunn, T.; Jongen, L.M.; Nederkoorn, P.J.; Macdonald, S.; Gaines, P.A.; Lyrer, P.A.; Wetzel, S.G.; van der Lugt, A.; Mali, W.P.T.M.; et al. Characteristics of Ischemic Brain Lesions After Stenting or Endarterectomy for Symptomatic Carotid Artery Stenosis. Stroke 2013, 44, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Bendszus, M.; Koltzenburg, M.; Burger, R.; Warmuth-Metz, M.; Hofmann, E.; Solymosi, L. Silent embolism in diagnostic cerebral angiography and neurointerventional procedures: A prospective study. Lancet 1999, 354, 1594–1597. [Google Scholar] [CrossRef]
- Kastrup, A.; Gröschel, K.; Schnaudigel, S.; Nägele, T.; Schmidt, F.; Ernemann, U. Target lesion ulceration and arch calcification are associated with increased incidence of carotid stenting-associated ischemic lesions in octogenarians. J. Vasc. Surg. 2008, 47, 88–95. [Google Scholar] [CrossRef]
- Spagnoli, L.G.; Mauriello, A.; Sangiorgi, G.; Fratoni, S.; Bonanno, E.; Schwartz, R.S.; Piepgras, D.G.; Pistolese, R.; Ippoliti, A.; Holmes, D.R. Extracranial Thrombotically Active Carotid Plaque as a Risk Factor for Ischemic Stroke. JAMA 2004, 292, 1845–1852. [Google Scholar] [CrossRef]
- Naggara, O.; Touzé, E.; Beyssen, B.; Trinquart, L.; Chatellier, G.; Meder, J.-F.; Mas, J.-L. Anatomical and Technical Factors Associated With Stroke or Death During Carotid Angioplasty and Stenting. Stroke 2011, 42, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Eckstein, H.-H.; Ringleb, P.; Allenberg, J.-R.; Berger, J.; Fraedrich, G.; Hacke, W.; Hennerici, M.; Stingele, R.; Fiehler, J.; Zeumer, H.; et al. Results of the Stent-Protected Angioplasty versus Carotid Endarterectomy (SPACE) study to treat symptomatic stenoses at 2 years: A multinational, prospective, randomised trial. Lancet Neurol. 2008, 7, 893–902. [Google Scholar] [CrossRef]
- Bonati, L.H.; Dobson, J.; Featherstone, R.L.; Ederle, J.; van der Worp, H.B.; de Borst, G.J.; Mali, W.P.T.M.; Beard, J.D.; Cleveland, T.; Engelter, S.T.; et al. Long-term outcomes after stenting versus endarterectomy for treatment of symptomatic carotid stenosis: The International Carotid Stenting Study (ICSS) randomised trial. Lancet 2014, 385, 529–538. [Google Scholar] [CrossRef]
- Liu, H.; Chu, J.; Zhang, L.; Liu, C.; Yan, Z.; Zhou, S. Clinical Comparison of Outcomes of Early versus Delayed Carotid Artery Stenting for Symptomatic Cerebral Watershed Infarction due to Stenosis of the Proximal Internal Carotid Artery. BioMed Res. Int. 2016, 2016, 6241546. [Google Scholar] [CrossRef]
- Song, K.S.; Kwon, O.-K.; Hwang, G.; Bae, H.-J.; Han, M.-K.; Kim, B.J.; Bang, J.S.; Oh, C.W. Early carotid artery stenting for symptomatic carotid artery stenosis. Acta Neurochir. 2015, 157, 1873–1878. [Google Scholar] [CrossRef]
- Meschia, J.F.; Hopkins, L.N.; Altafullah, I.; Wechsler, L.R.; Stotts, G.; Gonzales, N.R.; Voeks, J.H.; Howard, G.; Brott, T.G. Time From Symptoms to Carotid Endarterectomy or Stenting and Perioperative Risk. Stroke 2015, 46, 3540–3542. [Google Scholar] [CrossRef]
- Kwolek, C.J.; Jaff, M.R.; Leal, J.I.; Hopkins, L.N.; Shah, R.M.; Hanover, T.M.; Macdonald, S.; Cambria, R.P.; Flores, A.; Leal, I.; et al. Results of the ROADSTER multicenter trial of transcarotid stenting with dynamic flow reversal. J. Vasc. Surg. 2015, 62, 1227–1234. [Google Scholar] [CrossRef] [PubMed]
- Dakour-Aridi, H.; Kashyap, V.S.; Wang, G.J.; Eldrup-Jorgensen, J.; Schermerhorn, M.L.; Malas, M.B. The impact of age on in-hospital outcomes after transcarotid artery revascularization, transfemoral carotid artery stenting, and carotid endarterectomy. J. Vasc. Surg. 2020, 72, 931–942. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | n | Values |
|---|---|---|
| Demographics | ||
| Age (years), median (IQR) | 82 | 68 (59 to 75) |
| Men | 82 | 37 (45.1) |
| Medical history | ||
| Hypertension | 82 | 67 (81.7) |
| Diabetes mellitus | 82 | 33 (40.2) |
| Dyslipidemia | 82 | 47 (57.3) |
| Current smoking | 82 | 45 (54.9) |
| Long-term antithrombotic use | 82 | 26 (31.7) |
| Lesion characteristics | ||
| Location of lesion | ||
| Right ICA | 82 | 47 (57.3) |
| Left ICA | 34 (41.5) | |
| Bilateral ICA | 1 (1.2) | |
| Degree of carotid stenosis, %, median (IQR) | 78 | 75 (70 to 90) |
| 0 to 49 (including web) | 6 (7.7) | |
| 50 to 69 | 13 (16.7) | |
| 70 to 99 | 59 (75.6) | |
| Etiology | ||
| Atherosclerosis | 82 | 78 (95.1) |
| Web | 4 (4.9) | |
| Carotid plaque surface characteristics | ||
| Regular | 77 | 28 (36.4) |
| Irregular without ulceration | 20 (26.0) | |
| Irregular with ulceration | 29 (37.7) | |
| Angiographic Pattern | ||
| Pattern 1 | 82 | 28 (34.3) |
| Pattern 2 | 27 (32.9) | |
| Pattern 3 | 27 (32.9) | |
| Treatment characteristics | ||
| Onset to endovascular procedure, days, median (IQR) | 82 | 7 (3 to 30) |
| Duration of endovascular procedure, minutes, median (IQR) | 82 | 35 (25 to 45) |
| Type of stent 1 | ||
| Casper (Microvention, Aliso Viejo, CA, USA) | 95 | 21 (22.1) |
| Xact (Abbott Vascular, Santa Clara, CA, USA) | 56 (59.6) | |
| Precise (Cordis, Milpitas, CA, USA) | 18 (18.9) |
| Outcomes | n (%) | 95%CI |
|---|---|---|
| DWI-MRI new lesions | ||
| Patients with ≥ 1 lesions | 33 (40.2) | 29.5 to 51.7 |
| Single | 11 (13.4) | |
| Multiple | 22 (26.8) | |
| Patients with punctiform lesions | 31 (37.8) | 27.3 to 49.2 |
| Patients with ipsilateral lesions | 31 (37.8) | 27.3 to 49.2 |
| Patients with contralateral lesions | 8 (9.8) | 4.3 to 18.3 |
| Patients with lesions > 10 mm | 7 (8.5) | 3.5 to 16.8 |
| Clinical outcomes | ||
| Composite endpoint at 30 days of intervention | 3 (3.7) | 1.3 to 12.0 |
| Death | 1 (1.2) | |
| Acute coronary syndrome | 0 (0.0) | |
| Symptomatic ischemic stroke | 3 (3.7) | |
| ≥1 Bleeding event | 1 (1.2) | |
| ≥1 procedural complications | 3 (3.7) | 1.3 to 12.0 |
| Cerebral emboli | 3 (3.7) | |
| Groin hematoma | 0 (0.0) | |
| Dissection | 0 (0.0) |
| No DWI-MRI Lesions (n = 49) | ≥1 DWI-MRI Lesions (n = 33) | p-Value | |
|---|---|---|---|
| ICA lesion characteristics | |||
| Stenosis degree, % | |||
| 0 to 49 | 2 (4.4) | 4 (12.1) | 0.41 |
| 50 to 69 | 7 (15.6) | 6 (18.2) | |
| 70 to 99 | 36 (80.0) | 23 (69.7) | |
| Carotid plaque surface | |||
| Regular | 21 (46.7) | 7 (21.9) | 0.048 |
| Irregular without ulceration | 8 (17.8) | 12 (37.5) | |
| Irregular with ulceration | 16 (35.6) | 13 (40.6) | |
| Angiographic Pattern | |||
| Pattern 1 | 18 (40.0) | 8 (25.8) | 0.30 |
| Pattern 2 | 15 (33.3) | 10 (32.3) | |
| Pattern 3 | 12 (26.7) | 13 (41.9) | |
| Treatment characteristics | |||
| Number of stents | |||
| One | 43 (87.8) | 28 (84.9) | 0.75 |
| Two or three | 6 (12.2) | 5 (15.2) | |
| Duration of endovascular procedure, minutes, median (IQR) | 38 (30 to 45) | 29 (24 to 45) | 0.17 |
| Delay from symptoms onset to endovascular procedure, days, median (IQR) | 11 (3 to 30) | 5 (0.8 to 31) | 0.17 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hak, J.-F.; Arquizan, C.; Cagnazzo, F.; Mahmoudi, M.; Collemiche, F.-L.; Gascou, G.; Lefevre, P.-H.; Derraz, I.; Labreuche, J.; Mourand, I.; et al. MRI Outcomes Achieved by Simple Flow Blockage Technique in Symptomatic Carotid Artery Stenosis Stenting. J. Pers. Med. 2022, 12, 1564. https://doi.org/10.3390/jpm12101564
Hak J-F, Arquizan C, Cagnazzo F, Mahmoudi M, Collemiche F-L, Gascou G, Lefevre P-H, Derraz I, Labreuche J, Mourand I, et al. MRI Outcomes Achieved by Simple Flow Blockage Technique in Symptomatic Carotid Artery Stenosis Stenting. Journal of Personalized Medicine. 2022; 12(10):1564. https://doi.org/10.3390/jpm12101564
Chicago/Turabian StyleHak, Jean-François, Caroline Arquizan, Federico Cagnazzo, Mehdi Mahmoudi, Francois-Louis Collemiche, Gregory Gascou, Pierre-Henry Lefevre, Imad Derraz, Julien Labreuche, Isabelle Mourand, and et al. 2022. "MRI Outcomes Achieved by Simple Flow Blockage Technique in Symptomatic Carotid Artery Stenosis Stenting" Journal of Personalized Medicine 12, no. 10: 1564. https://doi.org/10.3390/jpm12101564
APA StyleHak, J.-F., Arquizan, C., Cagnazzo, F., Mahmoudi, M., Collemiche, F.-L., Gascou, G., Lefevre, P.-H., Derraz, I., Labreuche, J., Mourand, I., Gaillard, N., Corti, L., Charif, M., Costalat, V., & Dargazanli, C. (2022). MRI Outcomes Achieved by Simple Flow Blockage Technique in Symptomatic Carotid Artery Stenosis Stenting. Journal of Personalized Medicine, 12(10), 1564. https://doi.org/10.3390/jpm12101564






