Trust and Trade-Offs in Sharing Data for Precision Medicine: A National Survey of Singapore
Abstract
1. Introduction
2. Methods
2.1. Survey Respondents
2.2. Survey Instrument
- Uses—the purposes respondents are willing to share data for.
- Health research;
- Quality improvement;
- Commercial uses.
- Users—with whom respondents are willing to share data.
- Government agencies;
- Private insurers;
- Pharmaceutical/biotech companies;
- Universities/research institutes;
- Hospitals.
- Data sensitivity—the types of data respondents would be sharing.
- Identifiable genetic data and your medical history;
- Identifiable genetic data without your medical history;
- De-identified genetic data and your de-identified medical history;
- De-identified genetic data without your medical history.
- Consent—the degree of control over the data.
- Consent for every study;
- One time consent only for all studies;
- Consent for some specific studies.
2.3. Data Analysis
2.4. Ethics Oversight
3. Results
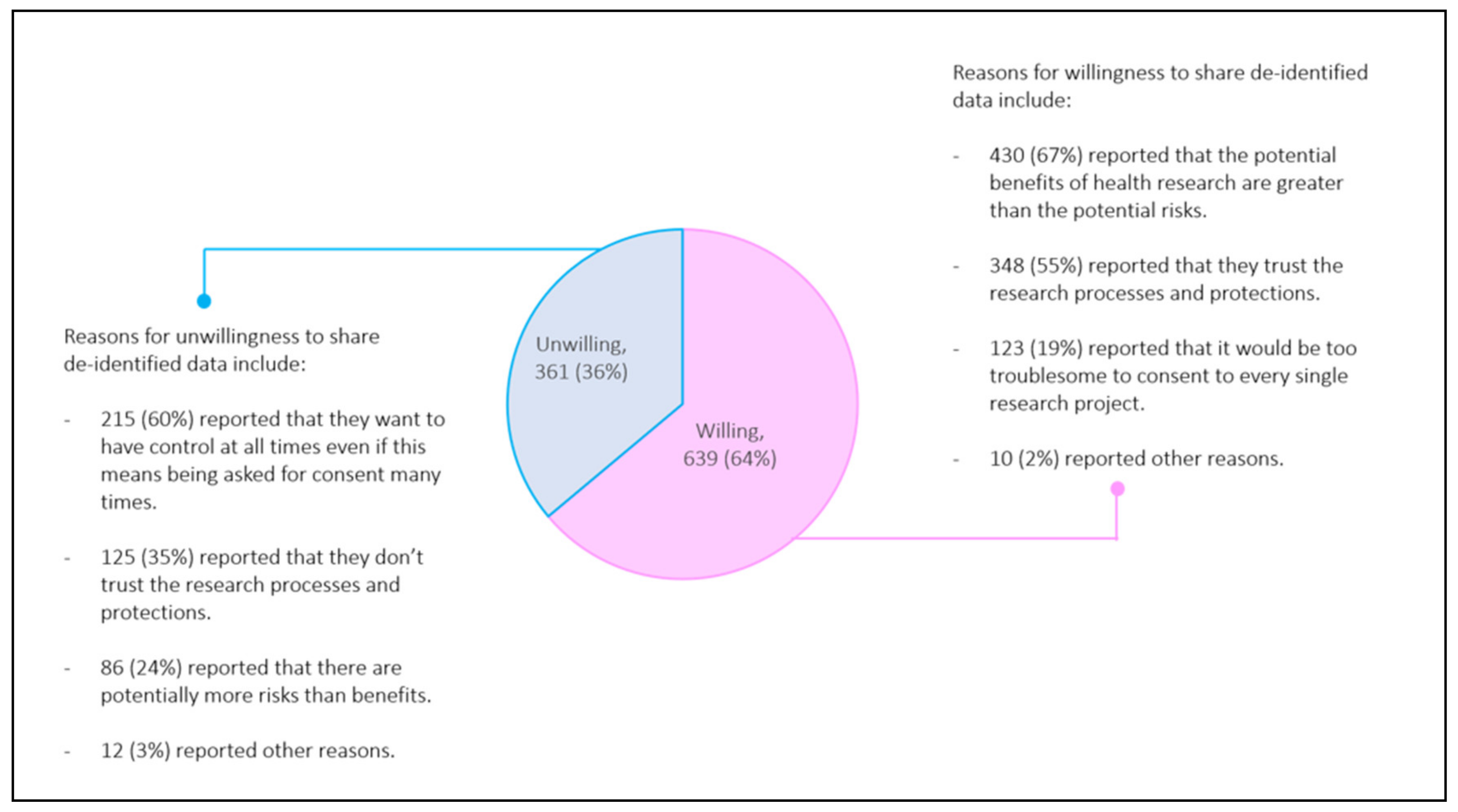
3.1. Population Demographics and Willingness to Share Data
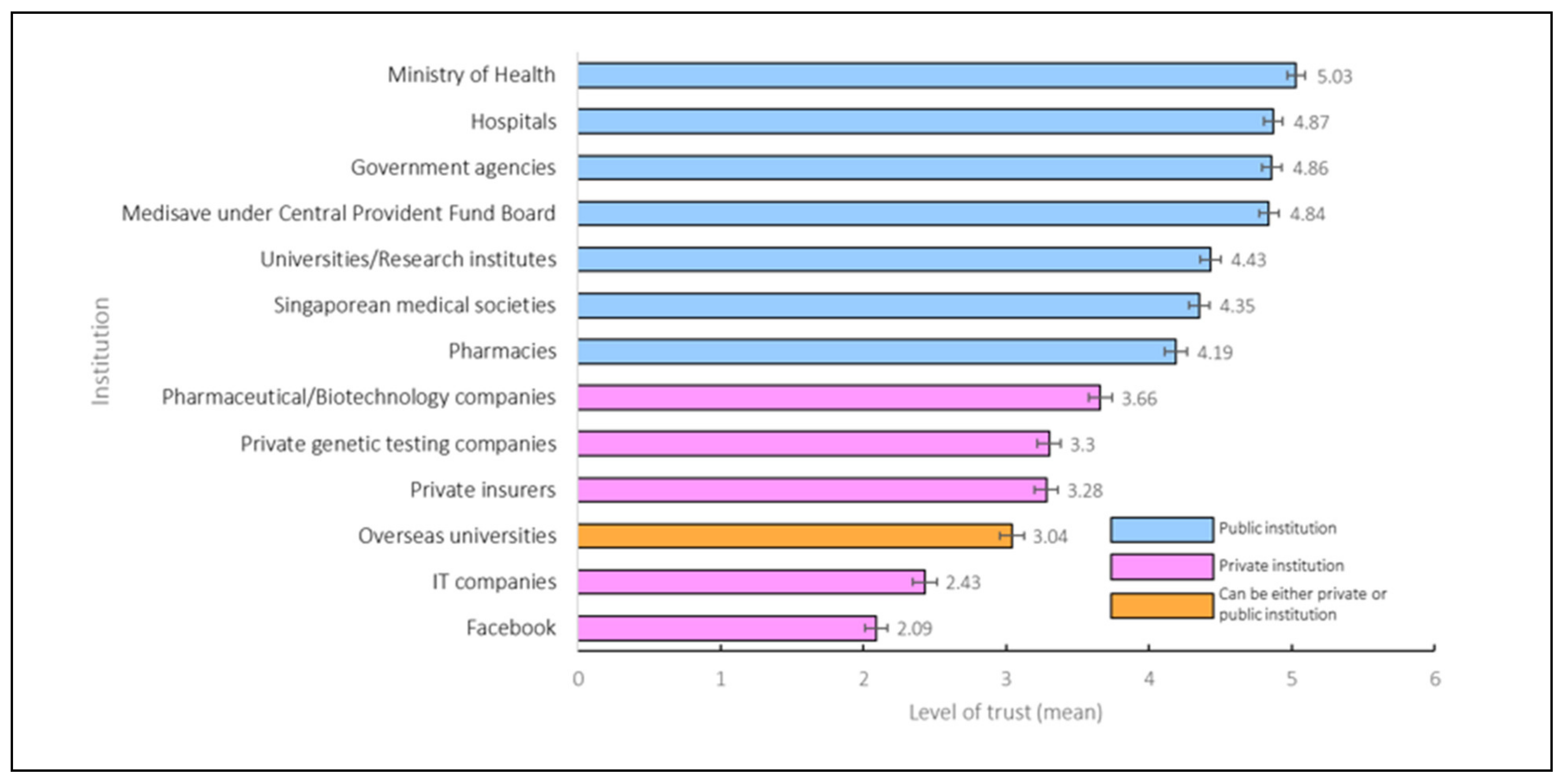
3.2. Trust in Institutions
3.3. Trade-Offs in Data Sharing Arrangements
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chowkwanyun, M.; Bayer, R.; Galea, S. “Precision” Public Health—Between Novelty and Hype. N. Engl. J. Med. 2018, 379, 1398–1400. [Google Scholar] [CrossRef] [PubMed]
- Aron, D.C. Precision medicine in an imprecise and complex world: Magic bullets, hype, and the fuzzy line between health and disease. J. Eval. Clin. Pract. 2020, 26, 1534–1538. [Google Scholar] [CrossRef] [PubMed]
- What Is the Precision Medicine Initiative? Available online: https://ghr.nlm.nih.gov/primer/precisionmedicine/initiative (accessed on 7 June 2021).
- The Lancet. Personalised medicine in the UK. Lancet 2018, 391, e1. [Google Scholar] [CrossRef]
- Cyranoski, D. China embraces precision medicine on a massive scale. Nature 2016, 529, 9–10. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.; Adams, C.; Flack, F. The role of data custodians in establishing and maintaining social licence for health research. Bioethics 2019, 33, 502–510. [Google Scholar] [CrossRef] [PubMed]
- Kalkman, S.; van Delden, J.; Banerjee, A.; Tyl, B.; Mostert, M.; van Thiel, G. Patients’ and public views and attitudes towards the sharing of health data for research: A narrative review of the empirical evidence. J. Med. Ethics 2019, 105651. [Google Scholar] [CrossRef]
- Garrison, N.A.; Sathe, N.A.; Antommaria, A.H.; Holm, I.A.; Sanderson, S.C.; Smith, M.E.; McPheeters, M.L.; Clayton, E.W. A systematic literature review of individuals’ perspectives on broad consent and data sharing in the United States. Genet. Med. 2016, 18, 663–671. [Google Scholar] [CrossRef]
- Vidgen, M.E.; Kaladharan, S.; Malacova, E.; Hurst, C.; Waddell, N. Sharing genomic data from clinical testing with researchers: Public survey of expectations of clinical genomic data management in Queensland, Australia. BMC Med. Ethics 2020, 21, 119. [Google Scholar] [CrossRef]
- Ludman, E.J.; Fullerton, S.M.; Spangler, L.; Trinidad, S.B.; Fujii, M.M.; Jarvik, G.P.; Larson, E.B.; Burke, W. Glad You Asked: Participants’ Opinions of Re-Consent for DbGap Data Submission. J. Empir. Res. Hum. Res. Ethics 2010, 5, 9–16. [Google Scholar] [CrossRef]
- Shah, N.; Coathup, V.; Teare, H.; Forgie, I.; Giordano, G.N.; Hansen, T.H.; Groeneveld, L.; Hudson, M.; Pearson, E.; Ruetten, H.; et al. Sharing data for future research—engaging participants’ views about data governance beyond the original project: A DIRECT Study. Genet. Med. 2019, 21, 1131–1138. [Google Scholar] [CrossRef]
- Willison, D.J.; Keshavjee, K.; Nair, K.; Goldsmith, C.; Holbrook, A.M. Patients’ consent preferences for research uses of information in electronic medical records: Interview and survey data. BMJ 2003, 326, 373. [Google Scholar] [CrossRef]
- Mathews, D.J.H.; Rabin, J.T.; Quain, K.; Campbell, E.; Collyar, D.; Hlubocky, F.J.; Isakoff, S.; Peppercorn, J. Secondary Use of Patient Tissue in Cancer Biobanks. Oncologist 2019, 24, 1577–1583. [Google Scholar] [CrossRef]
- Nair, K.; Willison, D.; Holbrook, A.; Keshavjee, K. Patients’ consent preferences regarding the use of their health information for research purposes: A qualitative study. J. Health Serv. Res. Policy 2004, 9, 22–27. [Google Scholar] [CrossRef]
- Taylor, M.J.; Taylor, N. Health research access to personal confidential data in England and Wales: Assessing any gap in public attitude between preferable and acceptable models of consent. Life Sci. Soc. Policy 2014, 10, 15. [Google Scholar] [CrossRef]
- Pullman, D.; Etchegary, H.; Gallagher, K.; Hodgkinson, K.; Keough, M.; Morgan, D. Street C: Personal privacy, public benefits, and biobanks: A conjoint analysis of policy priorities and public perceptions. Genet. Med. 2012, 14, 229–235. [Google Scholar] [CrossRef]
- Grande, D.; Mitra, N.; Shah, A.; Wan, F.; Asch, D.A. Public preferences about secondary uses of electronic health information. JAMA Intern. Med. 2013, 17, 1798–1806. [Google Scholar] [CrossRef]
- Patil, S.; Lu, H.; Saunders, C.L.; Potoglou, D.; Robinson, N. Public preferences for electronic health data storage, access, and sharing—Evidence from a pan-European survey. J. Am. Med. Inform. Assoc. 2016, 23, 1096–1106. [Google Scholar] [CrossRef] [PubMed]
- Xafis, V. The acceptability of conducting data linkage research without obtaining consent: Lay people’s views and justifications. BMC Med. Ethics 2015, 16, 79. [Google Scholar] [CrossRef] [PubMed]
- Dang, J.H.T.; Rodriguez, E.M.; Luque, J.S.; Erwin, D.O.; Meade, C.D.; Chen, M.S., Jr. Engaging diverse populations about biospecimen donation for cancer research. J. Community Genet. 2014, 5, 313–327. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.L.; Chia, K.S.; Yam, W.M.; Teodoro, G.R.; Lau, K.W. Willingness to donate blood samples for genetic research: A survey from a community in Singapore. Clin. Genet. 2004, 65, 45–51. [Google Scholar] [CrossRef]
- Hate, K.; Meherally, S.; Shah More, N.; Jayaraman, A.; Bull, S.; Parker, M.; Osrin, D. Sweat, Skepticism, and Uncharted Territory: A Qualitative Study of Opinions on Data Sharing Among Public Health Researchers and Research Participants in Mumbai, India. J. Empir. Res. Hum. Res. Ethics 2015, 10, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Population and Population Structure. Available online: https://www.singstat.gov.sg/find-data/search-by-theme/population/population-and-population-structure/latest-data (accessed on 18 May 2021).
- Health and Biomedical Sciences. Available online: https://www.nrf.gov.sg/rie2020/health-and-biomedical-sciences (accessed on 7 June 2021).
- Precision Health Research, Singapore. Available online: https://www.npm.sg/ (accessed on 18 May 2021).
- Bylstra, Y.; Lysaght, T.; Thrivikraman, J.; Watson, S.; Tan, P. Ethical frameworks for obtaining informed consent in tumour profiling: An evidence-based case for Singapore. Hum. Genomics 2017, 11, 31. [Google Scholar] [CrossRef]
- Lysaght, T.; Ballantyne, A.; Xafis, V.; Ong, S.; Schaefer, O.G.; Ling, J.; Tai, E.S. “Who is watching the watchdog?”: Ethical perspectives of sharing health-related data for precision medicine in Singapore. BMC Med. Ethics 2020, 21, 1–11. [Google Scholar] [CrossRef]
- Sample Design and Selection Service for Household Surveys. Available online: https://www.singstat.gov.sg/our-services-and-tools/sampling-service-for-household-surveys (accessed on 18 May 2021).
- Ong, S.; Ling, J.; Ballantyne, A.; Lysaght, T.; Xafis, V. Perceptions of ‘Precision’ and ‘Personalised’ Medicine in Singapore and Associated Ethical Issues. Asian Bioeth. Rev. 2021, 13, 179–194. [Google Scholar] [CrossRef]
- Bridges, J.F.P.; Hauber, A.B.; Marshall, D.; Lloyd, A.; Prosser, L.A.; Regier, D.A.; Johnson, F.R.; Mauskopf, J. Conjoint Analysis Applications in Health—A Checklist: A Report of the ISPOR Good Research Practices for Conjoint Analysis Task Force. Value Health 2011, 14, 403–413. [Google Scholar] [CrossRef]
- Almario, C.V.; Keller, M.S.; Chen, M.; Lasch, K.; Ursos, L.; Shklovskaya, J.; Melmed, G.Y.; Spiegel, B.M.R. Optimizing Selection of Biologics in Inflammatory Bowel Disease: Development of an Online Patient Decision Aid Using Conjoint Analysis. Am. J. Gastroenterol. 2018, 113, 58–71. [Google Scholar] [CrossRef]
- The Adaptive Choice-Based Conjoint (ACBC) Technical Paper. Available online: https://sawtoothsoftware.com/resources/technical-papers/acbc-technical-paper (accessed on 7 June 2021).
- Trustworthy Governance for Sharing Health-Related Data. Available online: https://medicine.nus.edu.sg/cbme/research/trustworthy-governance-for-sharing-health-related-data/ (accessed on 7 June 2021).
- What Are the Racial Proportions among Singapore Citizens? Available online: https://www.gov.sg/article/what-are-the-racial-proportions-among-singapore-citizens (accessed on 18 May 2021).
- Trinidad, S.B.; Fullerton, S.M.; Bares, J.M.; Jarvik, G.P.; Larson, E.B.; Burke, W. Informed Consent in Genome-Scale Research: What Do Prospective Participants Think? AJOB Prim. Res. 2012, 3, 3–11. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sanderson, S.C.; Brothers, K.B.; Mercaldo, N.D.; Clayton, E.W.; Antommaria, A.H.M.; Aufox, S.A.; Brilliant, M.H.; Campos, D.; Carrell, D.S.; Connolly, J.; et al. Public Attitudes toward Consent and Data Sharing in Biobank Research: A Large Multi-site Experimental Survey in the US. Am. J. Hum. Genet. 2017, 100, 414–427. [Google Scholar] [CrossRef] [PubMed]
- Platt, J.; Bollinger, J.; Dvoskin, R.; Kardia, S.L.; Kaufman, D. Public preferences regarding informed consent models for participation in population-based genomic research. Genet. Med. 2014, 16, 11–18. [Google Scholar] [CrossRef]
- Simon, C.M.; L’Heureux, J.; Murray, J.C.; Winokur, P.; Weiner, G.; Newbury, E.; Shinkunas, L.; Zimmerman, B. Active choice but not too active: Public perspectives on biobank consent models. Genet. Med. Off. J. Am. Coll. Med. Genet. 2011, 13, 821–831. [Google Scholar] [CrossRef] [PubMed]
- Hill, E.M.; Turner, E.L.; Martin, R.M.; Donovan, J.L. “Let’s get the best quality research we can”: Public awareness and acceptance of consent to use existing data in health research: A systematic review and qualitative study. BMC Med. Res. Methodol. 2013, 13, 72. [Google Scholar] [CrossRef] [PubMed]
- Department of Health and Human Services. NIH Genomic Data Sharing Policy. 2014. Available online: https://grants.nih.gov/grants/guide/notice-files/not-od-14-124.html (accessed on 15 September 2021).
- Kaye, J. The tension between data sharing and the protection of privacy in genomics research. Annu. Rev. Genomics Hum. Genet. 2012, 13, 415–431. [Google Scholar] [CrossRef]
- Tully, M.P.; Bozentko, K.; Clement, S.; Hunn, A.; Hassan, L.; Norris, R.; Oswald, M.; Peek, N. Investigating the Extent to Which Patients Should Control Access to Patient Records for Research: A Deliberative Process Using Citizens’ Juries. J. Med. Internet Res. 2018, 20, 112. [Google Scholar] [CrossRef]
- Public Attitudes to Data Sharing in Northern Ireland: Findings from the 2015 Northern Ireland Life and Times survey. Available online: https://www.ark.ac.uk/pdfs/Researchreports/Data_Sharing_Report.pdf (accessed on 18 May 2021).
- Tully, M.P.; Bernsten, C.; Aitken, M.; Vass, C. Public preferences regarding data linkage for research: A discrete choice experiment comparing Scotland and Sweden. BMC Med. Inform. Decis. Mak. 2020, 20, 109. [Google Scholar] [CrossRef] [PubMed]
- Aitken, M.; McAteer, G.; Davidson, S.; Frostick, C.; Cunningham-Burley, S. Public Preferences regarding Data Linkage for Health Research: A Discrete Choice Experiment. Int. J. Popul. Data Sci. 2018, 3, 429. [Google Scholar] [CrossRef]
- Dialogue on Data: Exploring the Public’s Views on Using Administrative Data for Research Purposes. Available online: https://www.ipsos.com/ipsos-mori/en-uk/dialogue-data-census-report (accessed on 18 May 2021).
- Willison, D.J.; Steeves, V.; Charles, C.; Schwartz, L.; Ranford, J.; Agarwal, G.; Cheng, J.; Thabane, L. Consent for use of personal information for health research: Do people with potentially stigmatizing health conditions and the general public differ in their opinions? BMC Med. Ethics 2009, 10, 10. [Google Scholar] [CrossRef] [PubMed]
- The One-Way Mirror: Public Attitudes to Commercial access to Health Data. Available online: https://wellcome.figshare.com/ndownloader/files/9778657 (accessed on 18 May 2021).
- Big Data: Public Views on Private Sector Data for Social Research. Available online: https://esrc.ukri.org/files/public-engagement/public-dialogues/public-dialogues-on-the-re-use-of-private-sector-data-for-social-research-report/ (accessed on 18 May 2021).
- Stockdale, J.; Cassell, J.; Ford, E. “Giving something back”: A systematic review and ethical enquiry into public views on the use of patient data for research in the United Kingdom and the Republic of Ireland. Wellcome Open Res. 2019, 3, 6. [Google Scholar] [CrossRef]
- Organisation for Economic Co-operation and Development (OECD). Trust and Public Policy; OECD Publishing: Paris, France, 2017. [Google Scholar]
- Ford, E.; Stockdale, J.; Jackson, R.; Cassell, J. For the greater good? Patient and public attitudes to use of medical free text data in research. In International Journal of Population Data Science, Proceedings of the International Population Data Linkage Network (IPDLN) Conference, Swansea, Wales, UK, 24–26 August 2016; PiSUI: Swansea, Wales, UK, 2017; Volume 229, pp. 2399–4908. [Google Scholar]
- Kaufman, D.J.; Murphy-Bollinger, J.; Scott, J.; Hudson, K.L. Public opinion about the importance of privacy in biobank research. Am. J. Hum. Genet. 2009, 85, 643–654. [Google Scholar] [CrossRef]
- Kim, H.; Kim, H.R.; Kim, S.; Kim, E.; Kim, S.Y.; Park, H.Y. Public Attitudes Toward Precision Medicine: A Nationwide Survey on Developing a National Cohort Program for Citizen Participation in the Republic of Korea. Front. Genet. 2020, 11, 283. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.-W. (Ed.) The Asian Developmental State: Ideas and Debates. In The Asian Developmental State: Reexaminations and New Departures; Palgrave Macmillan: New York, NY, USA, 2016; pp. 1–25. [Google Scholar]
- Liow, E.D. The Neoliberal-Developmental State: Singapore as Case Study. Crit. Sociol. 2012, 38, 241–264. [Google Scholar] [CrossRef]
- Wong, C.M.L.; Jensen, O. The paradox of trust: Perceived risk and public compliance during the COVID-19 pandemic in Singapore. J. Risk Res. 2020, 23, 1021–1030. [Google Scholar] [CrossRef]
- Woo, J.J. Policy capacity and Singapore’s response to the COVID-19 pandemic. Policy Soc. 2020, 39, 345–362. [Google Scholar] [CrossRef]
- Kuguyo, O.; Kengne, A.P.; Dandara, C. Singapore COVID-19 Pandemic Response as a Successful Model Framework for Low-Resource Health Care Settings in Africa? OMICS 2020, 24, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Edelman Trust Barometer Spring Update: Trust and the Covid-19 Pandemic. Available online: https://www.edelman.com/research/trust-2020-spring-update (accessed on 18 May 2021).
- Prainsack, B.; Buyx, A. A Solidarity-Based Approach to the Governance of Research Biobanks. Med. Law Rev. 2013, 21, 71–91. [Google Scholar] [CrossRef]
- Laurie, G. Reflexive governance in biobanking: On the value of policy led approaches and the need to recognise the limits of law. Hum. Genet. 2011, 130, 347. [Google Scholar] [CrossRef]
- Capps, B.J. Defining Variables of Access to UK Biobank: The Public Interest and the Public Good. Law Innov. Technol. 2013, 5, 113–139. [Google Scholar] [CrossRef]
- Simm, K. The concepts of common good and public interest: From Plato to biobanking. Camb. Q. Healthc. Ethics 2011, 20, 554–562. [Google Scholar] [CrossRef]
- Ballantyne, A.; Schaefer, G.O. Public interest in health data research: Laying out the conceptual groundwork. J. Med. Ethics 2020, 46, 610–616. [Google Scholar] [CrossRef]
- Lavrakas, P.J. Encyclopedia of Survey Research Methods, 1st ed.; SAGE Publications: Thousand Oaks, CA, USA, 2008. [Google Scholar]
- Cooke Bailey, J.N.; Crawford, D.C.; Goldenberg, A.; Slaven, A.; Pencak, J.; Schachere, M.; Bush, W.; Sedor, J.; O’Toole, J. Willingness to participate in a national precision medicine cohort: Attitudes of chronic kidney disease patients at a Cleveland public hospital. J. Pers. Med. 2018, 8, 21. [Google Scholar] [CrossRef] [PubMed]
- Middleton, A.; Milne, R.; Almarri, M.A.; Answer, S.; Atutornu, J.; Baranova, E.E.; Baven, P.; Cerezo, M.; Cong, Y.; Critchley, C.; et al. Global Public Perceptions of Genomic Data Sharing: What Shapes the Willingness to Donate DNA and Health Data? Am. J. Hum. Genet. 2020, 107, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Porteri, C.; Pasqualetti, P.; Togni, E.; Parker, M. Public’s attitudes on participation in a biobank for research: An Italian survey. BMC Med. Ethics 2014, 15, 81. [Google Scholar] [CrossRef] [PubMed]

| Characteristic | No. of Respondents |
|---|---|
| Age bracket | |
| 21–29 | 149 |
| 30–39 | 188 |
| 40–49 | 199 |
| 50–59 | 195 |
| 60–69 | 162 |
| 70–79 | 91 |
| ≥80 | 16 |
| Gender | |
| Female | 519 |
| Male | 481 |
| Ethnicity | |
| Chinese | 635 |
| Malay | 178 |
| Indian | 177 |
| Others | 10 |
| Religion | |
| Buddhism | 274 |
| Christianity | 155 |
| Hinduism | 111 |
| Islam | 219 |
| Sikhism | 41 |
| Taoism | 20 |
| No religion | 174 |
| Others | 6 |
| Self-rated health | |
| Poor | 11 |
| Fair | 110 |
| Good | 471 |
| Very good | 320 |
| Excellent | 88 |
| Monthly household income, SGD | |
| No income | 78 |
| ≤2999 | 216 |
| 3000–5999 | 195 |
| 6000–9999 | 156 |
| 10,000–14,999 | 80 |
| ≥15,000 | 30 |
| No response | 245 |
| Housing Type | |
| 1-room flat | 30 |
| 2-room flat | 53 |
| 3-room flat | 264 |
| 4-room flat | 374 |
| 5-room flat | 211 |
| Condominium | 33 |
| Landed property | 35 |
| Educational level | |
| No formal education | 40 |
| Primary | 139 |
| Secondary/Post-secondary | 291 |
| A-Level/Polytechnic | 166 |
| Tertiary education | 364 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lysaght, T.; Ballantyne, A.; Toh, H.J.; Lau, A.; Ong, S.; Schaefer, O.; Shiraishi, M.; van den Boom, W.; Xafis, V.; Tai, E.S. Trust and Trade-Offs in Sharing Data for Precision Medicine: A National Survey of Singapore. J. Pers. Med. 2021, 11, 921. https://doi.org/10.3390/jpm11090921
Lysaght T, Ballantyne A, Toh HJ, Lau A, Ong S, Schaefer O, Shiraishi M, van den Boom W, Xafis V, Tai ES. Trust and Trade-Offs in Sharing Data for Precision Medicine: A National Survey of Singapore. Journal of Personalized Medicine. 2021; 11(9):921. https://doi.org/10.3390/jpm11090921
Chicago/Turabian StyleLysaght, Tamra, Angela Ballantyne, Hui Jin Toh, Andrew Lau, Serene Ong, Owen Schaefer, Makoto Shiraishi, Willem van den Boom, Vicki Xafis, and E Shyong Tai. 2021. "Trust and Trade-Offs in Sharing Data for Precision Medicine: A National Survey of Singapore" Journal of Personalized Medicine 11, no. 9: 921. https://doi.org/10.3390/jpm11090921
APA StyleLysaght, T., Ballantyne, A., Toh, H. J., Lau, A., Ong, S., Schaefer, O., Shiraishi, M., van den Boom, W., Xafis, V., & Tai, E. S. (2021). Trust and Trade-Offs in Sharing Data for Precision Medicine: A National Survey of Singapore. Journal of Personalized Medicine, 11(9), 921. https://doi.org/10.3390/jpm11090921







