Ways to Address Perinatal Mast Cell Activation and Focal Brain Inflammation, including Response to SARS-CoV-2, in Autism Spectrum Disorder
Abstract
:1. Introduction
2. Infections and COVID-19
3. Psychological Stress
4. Mast Cell Activation
5. Mast Cells and Microglia
6. Treatment Approaches
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACE2 | Angiotensin-converting enzyme 2 |
| ADHD | Attention Deficit Hyperactivity Disorder |
| BBB | Blood–brain barrier |
| BDNF | Brain-derived neurotrophic factor |
| CNS | Central nervous system |
| CRH | Corticotropin-releasing hormone |
| DAMPs | Damage-associated molecular patterns |
| HPA | Hypothalamic–pituitary–adrenal |
| MCAS | Mast Cell Activation Syndrome |
| MCI | Mild cognitive impairment |
| mtDNA | Mitochondrial DNA |
| MIS | Multisystem Inflammatory Syndrome |
| NT | Neurotensin |
| PANS | Pediatric Acute Neuropsychiatric Syndrome |
| PAMPs | Pathogen-associated molecular patterns |
| PAF | Platelet-activating factor |
| SP | Substance P |
| TLR | Toll-like receptor |
References
- Johnson, C.P.; Myers, S.M. Identification and evaluation of children with autism spectrum disorders. Pediatrics 2007, 120, 1183–1215. [Google Scholar] [CrossRef] [Green Version]
- Lai, M.C.; Lombardo, M.V.; Baron-Cohen, S. Autism. Lancet 2014, 383, 896–910. [Google Scholar] [CrossRef]
- Howes, O.D.; Rogdaki, M.; Findon, J.L.; Wichers, R.H.; Charman, T.; King, B.H.; Loth, E.; McAlonan, G.M.; McCracken, J.T.; Parr, J.; et al. Autism spectrum disorder: Consensus guidelines on assessment, treatment and research from the British Association for Psychopharmacology. J. Psychopharmacol. 2018, 32, 3–29. [Google Scholar] [CrossRef] [PubMed]
- Braconnier, M.L.; Siper, P.M. Neuropsychological assessment in autism spectrum disorder. Curr. Psychiatry Rep. 2021, 23, 63. [Google Scholar] [CrossRef] [PubMed]
- Iles, A. Autism spectrum disorders. Prim. Care 2021, 48, 461–473. [Google Scholar] [CrossRef]
- Maenner, M.J.; Shaw, K.A.; Baio, J.; Washington, A.; Patrick, M.; DiRienzo, M.; Christensen, D.L.; Wiggins, L.D.; Pettygrove, S.; Andrews, J.G.; et al. Prevalence of autism spectrum disorder among children aged 8 years—Autism and developmental disabilities monitoring network, 11 sites, United States, 2016. MMWR Surveill. Summ. 2020, 69, 1–12. [Google Scholar] [CrossRef]
- Xu, G.; Strathearn, L.; Liu, B.; Bao, W. Prevalence of autism spectrum disorder among US children and adolescents, 2014–2016. JAMA 2018, 319, 81–82. [Google Scholar] [CrossRef] [Green Version]
- Baxter, A.J.; Brugha, T.S.; Erskine, H.E.; Scheurer, R.W.; Vos, T.; Scott, J.G. The epidemiology and global burden of autism spectrum disorders. Psychol. Med. 2015, 45, 601–613. [Google Scholar] [CrossRef]
- Leigh, J.P.; Du, J. Brief report: Forecasting the economic burden of autism in 2015 and 2025 in the United States. J. Autism Dev. Disord. 2015, 12, 4135–4139. [Google Scholar] [CrossRef] [PubMed]
- Buescher, A.V.; Cidav, Z.; Knapp, M.; Mandell, D.S. Costs of autism spectrum disorders in the United Kingdom and the United States. JAMA Pediatr. 2014, 168, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Rogge, N.; Janssen, J. The economic costs of autism spectrum disorder: A literature review. J. Autism Dev. Disord. 2019, 49, 2873–2900. [Google Scholar] [CrossRef]
- Xue, M.; Brimacombe, M.; Chaaban, J.; Zimmerman-Bier, B.; Wagner, G.C. Autism spectrum disorders: Concurrent clinical disorders. J. Child Neurol. 2008, 23, 6–13. [Google Scholar]
- Bauman, M.L. Medical comorbidities in autism: Challenges to diagnosis and treatment. Neurotherapeutics 2010, 7, 320–327. [Google Scholar] [CrossRef]
- Underwood, J.F.G.; Kendall, K.M.; Berrett, J.; Lewis, C.; Anney, R.; Van den Bree, M.B.; Hall, J. Autism spectrum disorder diagnosis in adults: Phenotype and genotype findings from a clinically derived cohort. Br. J. Psychiatry 2019, 215, 647–653. [Google Scholar] [CrossRef] [Green Version]
- Mesleh, A.G.; Abdulla, S.A.; El-Agnaf, O. Paving the way toward personalized medicine: Current advances and challenges in Multi-OMICS approach in autism spectrum disorder for biomarkers discovery and patient stratification. J. Pers. Med. 2021, 11, 41. [Google Scholar] [CrossRef]
- Angelidou, A.; Asadi, S.; Alysandratos, K.D.; Karagkouni, A.; Kourembanas, S.; Theoharides, T.C. Perinatal stress, brain inflammation and risk of autism—Review and proposal. BMC Pediatr. 2012, 12, 89. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Laszlo, K.D.; Gissler, M.; Li, F.; Zhang, J.; Yu, Y.; Li, J. Maternal hypertensive disorders and neurodevelopmental disorders in offspring: A population-based cohort in two Nordic countries. Eur. J. Epidemiol. 2021, 36, 519–530. [Google Scholar] [CrossRef]
- Barron, A.; McCarthy, C.M.; O’Keeffe, G.W. Preeclampsia and neurodevelopmental outcomes: Potential pathogenic roles for inflammation and oxidative stress? Mol. Neurobiol. 2021, 58, 2734–2756. [Google Scholar] [CrossRef] [PubMed]
- Katz, J.; Reichenberg, A.; Kolevzon, A. Prenatal and perinatal metabolic risk factors for autism: A review and integration of findings from population-based studies. Curr. Opin. Psychiatry 2021, 34, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Crump, C.; Sundquist, J.; Sundquist, K. Preterm or early term birth and risk of autism. Pediatrics 2021, 148, e2020032300. [Google Scholar] [CrossRef] [PubMed]
- Stephens, B.E.; Bann, C.M.; Watson, V.E.; Sheinkopf, S.J.; Peralta-Carcelen, M.; Bodnar, A.; Yolton, K.; Goldstein, R.F.; Dusick, A.M.; Wilson-Costello, D.E.; et al. Screening for autism spectrum disorders in extremely preterm infants. J. Dev. Behav. Pediatr. 2012, 33, 535–541. [Google Scholar] [CrossRef] [Green Version]
- McGowan, E.C.; Sheinkopf, S.J. Autism and preterm birth: Clarifying risk and exploring mechanisms. Pediatrics 2021, 148, e2021051978. [Google Scholar] [CrossRef]
- Croen, L.A.; Grether, J.K.; Yoshida, C.K.; Odouli, R.; Van de Water, J. Maternal autoimmune diseases, asthma and allergies, and childhood autism spectrum disorders: A case-control study. Arch. Pediatr. Adolesc. Med. 2005, 159, 151–157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, S.; Ding, Y.; Wu, F.; Li, R.; Xie, G.; Hou, J.; Mao, P. Family history of autoimmune diseases is associated with an increased risk of autism in children: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2015, 55, 322–332. [Google Scholar] [CrossRef]
- Lee, H.; Hsu, J.W.; Tsai, S.J.; Huang, K.L.; Bai, Y.M.; Su, T.P.; Chen, T.J.; Chen, M.H. Risk of attention deficit hyperactivity and autism spectrum disorders among the children of parents with autoimmune diseases: A nationwide birth cohort study. Eur. Child Adolesc. Psychiatry 2021, 1–9. [Google Scholar] [CrossRef]
- Matelski, L.; van de Water, J. Risk factors in autism: Thinking outside the brain. J. Autoimmun. 2016, 67, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Muhle, R.A.; Reed, H.E.; Stratigos, K.A.; Veenstra-Vanderweele, J. The emerging clinical neuroscience of autism spectrum disorder: A review. JAMA Psychiatry 2018, 75, 514–523. [Google Scholar] [CrossRef]
- Zimmerman, A.W.; Jyonouchi, H.; Comi, A.M.; Connors, S.L.; Milstien, S.; Varsou, A.; Heyes, M.P. Cerebrospinal fluid and serum markers of inflammation in autism. Pediatr. Neurol. 2005, 33, 195–201. [Google Scholar] [CrossRef]
- Estes, M.L.; McAllister, A.K. Immune mediators in the brain and peripheral tissues in autism spectrum disorder. Nat. Rev. Neurosci. 2015, 16, 469–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meltzer, A.; van de Water, J. The role of the immune system in autism spectrum disorder. Neuropsychopharmacology 2017, 42, 284–298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matta, S.M.; Hill-Yardin, E.L.; Crack, P.J. The influence of neuroinflammation in autism spectrum disorder. Brain Behav. Immun. 2019, 79, 75–90. [Google Scholar] [CrossRef]
- Hughes, H.K.; Mills, K.E.; Rose, D.; Ashwood, P. Immune dysfunction and autoimmunity as pathological mechanisms in autism spectrum disorders. Front. Cell Neurosci. 2018, 12, 405. [Google Scholar] [CrossRef] [Green Version]
- Kowal, C.; Athanassiou, A.; Chen, H.; Diamond, B. Maternal antibodies and developing blood-brain barrier. Immunol. Res. 2015, 63, 18–25. [Google Scholar] [CrossRef] [Green Version]
- Edmiston, E.; Ashwood, P.; van de Water, J. Autoimmunity, autoantibodies, and autism spectrum disorder. Biol. Psychiatry 2017, 81, 383–390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, K.L.; van de Water, J. Maternal autoantibody related autism: Mechanisms and pathways. Mol. Psychiatry 2019, 24, 252–265. [Google Scholar] [CrossRef] [PubMed]
- Mazon-Cabrera, R.; Vandormael, P.; Somers, V. Antigenic targets of patient and maternal autoantibodies in autism spectrum disorder. Front. Immunol. 2019, 10, 1474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theoharides, T.C.; Zhang, B. Neuro-inflammation, blood-brain barrier, seizures and autism. J. Neuroinflamm. 2011, 8, 168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theoharides, T.C.; Asadi, S.; Patel, A.B. Focal brain inflammation and autism. J. Neuroinflamm. 2013, 10, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theoharides, T.C.; Kavalioti, M.; Tsilioni, I. Mast cells, stress, fear and autism spectrum disorder. Int. J. Mol. Sci. 2019, 20, 3611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez, J.I.; Kern, J.K. Evidence of microglial activation in autism and its possible role in brain underconnectivity. Neuron Glia Biol. 2011, 7, 205–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, S.; Ellis, S.E.; Ashar, F.N.; Moes, A.; Bader, J.S.; Zhan, J.; West, A.B.; Arking, D.E. Transcriptome analysis reveals dysregulation of innate immune response genes and neuronal activity-dependent genes in autism. Nat. Commun. 2014, 5, 5748. [Google Scholar] [CrossRef]
- Koyama, R.; Ikegaya, Y. Microglia in the pathogenesis of autism spectrum disorders. Neurosci. Res. 2015, 100, 1–5. [Google Scholar] [CrossRef]
- Takano, T. Role of microglia in autism: Recent advances. Dev. Neurosci. 2015, 37, 195–202. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Asadi, S.; Panagiotidou, S.; Weng, Z. The “missing link” in autoimmunity and autism: Extracellular mitochondrial components secreted from activated live mast cells. Autoimmun. Rev. 2013, 12, 1136–1142. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Tsilioni, I.; Patel, A.B.; Doyle, R. Atopic diseases and inflammation of the brain in the pathogenesis of autism spectrum disorders. Transl. Psychiatry 2016, 6, e844. [Google Scholar] [CrossRef] [Green Version]
- Liao, T.C.; Lien, Y.T.; Wang, S.; Huang, S.L.; Chen, C.Y. Comorbidity of atopic disorders with autism spectrum disorder and attention deficit/hyperactivity disorder. J. Pediatr. 2016, 171, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C. Is a subtype of autism an “allergy of the brain”? Clin. Ther. 2013, 35, 584–591. [Google Scholar] [CrossRef]
- Magalhaes, E.S.; Pinto-Mariz, F.; Bastos-Pinto, S.; Pontes, A.T.; Prado, E.A.; Deazevedo, L.C. Immune allergic response in Asperger syndrome. J. Neuroimmunol. 2009, 216, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Jyonouchi, H. Autism spectrum disorders and allergy: Observation from a pediatric allergy/immunology clinic. Expert. Rev. Clin. Immunol. 2010, 6, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Lyall, K.; van de Water, J.; Ashwood, P.; Hertz-Picciotto, I. Asthma and allergies in children with autism spectrum disorders: Results from the charge study. Autism Res. 2015, 8, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Angelidou, A.; Alysandratos, K.D.; Asadi, S.; Zhang, B.; Francis, K.; Vasiadi, M.; Kalogeromitros, D.; Theoharides, T.C. Brief Report: “Allergic Symptoms” in children with Autism Spectrum Disorders. More than meets the eye? J. Autism Dev. Disord. 2011, 41, 1579–1585. [Google Scholar] [CrossRef] [PubMed]
- Saitoh, B.Y.; Tanaka, E.; Yamamoto, N.; Kruining, D.V.; Iinuma, K.; Nakamuta, Y.; Yamaguchi, H.; Yamasaki, R.; Matsumoto, K.; Kira, J.I. Early postnatal allergic airway inflammation induces dystrophic microglia leading to excitatory postsynaptic surplus and autism-like behavior. Brain Behav Immun. 2021, 95, 362–380. [Google Scholar] [CrossRef] [PubMed]
- Kotey, S.; Ertel, K.; Whitcomb, B. Co-occurrence of autism and asthma in a nationally-representative sample of children in the United States. J. Autism Dev. Disord. 2014, 44, 3083–3088. [Google Scholar] [CrossRef] [PubMed]
- Billeci, L.; Tonacci, A.; Tartarisco, G.; Ruta, L.; Pioggia, G.; Gangemi, S. Association between atopic dermatitis and autism spectrum disorders: A systematic review. Am. J. Clin. Dermatol. 2015, 16, 371–388. [Google Scholar] [CrossRef]
- Patel, S.; Cooper, M.N.; Jones, H.; Whitehouse, A.J.O.; Dale, R.C.; Guastella, A.J. Maternal immune-related conditions during pregnancy may be a risk factor for neuropsychiatric problems in offspring throughout childhood and adolescence. Psychol. Med. 2020, 1–11. [Google Scholar] [CrossRef]
- Angelidou, A.; Sullivan, K.; Melvin, P.R.; Shui, J.E.; Goldfarb, I.T.; Bartolome, R.; Chaudhary, N.; Vaidya, R.; Culic, I.; Singh, R.; et al. Association of maternal perinatal SARS-CoV-2 infection with neonatal outcomes during the COVID-19 pandemic in Massachusetts. JAMA Netw. Open 2021, 4, e217523. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Conti, P. COVID-19 and multisystem inflammatory syndrome, or is it mast cell activation syndrome? J. Biol. Regul. Homeost. Agents 2020, 34, 1633–1636. [Google Scholar]
- Holingue, C.; Brucato, M.; Ladd-Acosta, C.; Hong, X.; Volk, H.; Mueller, N.T.; Wang, X.; Fallin, M.D. Interaction between maternal immune activation and antibiotic use during pregnancy and child risk of autism spectrum disorder. Autism Res. 2020, 13, 2230–2241. [Google Scholar] [CrossRef]
- Wilkerson, D.S.; Volpe, A.G.; Dean, R.S.; Titus, J.B. Perinatal complications as predictors of infantile autism. Int. J. Neurosci. 2002, 112, 1085–1098. [Google Scholar] [CrossRef]
- Tioleco, N.; Silberman, A.E.; Stratigos, K.; Banerjee-Basu, S.; Spann, M.N.; Whitaker, A.H.; Turner, J.B. Prenatal maternal infection and risk for autism in offspring: A meta-analysis. Autism Res. 2021, 14, 1296–1316. [Google Scholar] [CrossRef]
- Yao, Z.H.; Yao, X.L.; Zhang, Y.; Zhang, S.F.; Hu, J.C. Luteolin could improve cognitive dysfunction by inhibiting neuroinflammation. Neurochem. Res. 2018, 43, 806–820. [Google Scholar] [CrossRef]
- Shen, Q.; Zhang, Q.; Zhao, J.; Huang, Z.; Wang, X.; Ni, M.; Tang, Z.; Liu, Z. Association between maternal perceived stress in all trimesters of pregnancy and infant atopic dermatitis: A prospective birth cohort study. Front. Pediatr. 2020, 8, 526994. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C. Autism spectrum disorders and mastocytosis. Int. J. Immunopathol. Pharmacol. 2009, 22, 859–865. [Google Scholar] [CrossRef] [Green Version]
- Theoharides, T.C. Effect of stress on neuroimmune processes. Clin. Ther. 2020, 42, 1007–1014. [Google Scholar] [CrossRef]
- Ronald, A.; Pennell, C.E.; Whitehouse, A.J. Prenatal maternal stress associated with ADHD and autistic traits in early childhood. Front. Psychol. 2010, 1, 223. [Google Scholar] [CrossRef] [Green Version]
- Okano, L.; Ji, Y.; Riley, A.W.; Wang, X. Maternal psychosocial stress and children’s ADHD diagnosis: A prospective birth cohort study. J. Psychosom. Obstet. Gynaecol. 2019, 40, 217–225. [Google Scholar] [CrossRef] [PubMed]
- MacKinnon, N.; Kingsbury, M.; Mahedy, L.; Evans, J.; Colman, I. The association between prenatal stress and externalizing symptoms in childhood: Evidence from the avon longitudinal study of parents and children. Biol. Psychiatry 2018, 83, 100–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beversdorf, D.Q.; Manning, S.E.; Hillier, A.; Anderson, S.L.; Nordgren, R.E.; Walters, S.E.; Nagaraja, H.N.; Cooley, W.C.; Gaelic, S.E.; Bauman, M.L. Timing of prenatal stressors and autism. J. Autism. Dev. Disord. 2005, 35, 471–478. [Google Scholar] [CrossRef]
- Crafa, D.; Warfa, N. Maternal migration and autism risk: Systematic analysis. Int. Rev. Psychiatry 2015, 27, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Beversdorf, D.Q.; Stevens, H.E.; Margolis, K.G.; van de Water, J. Prenatal stress and maternal immune dysregulation in autism spectrum disorders: Potential points for intervention. Curr. Pharm. Des. 2019, 25, 4331–4343. [Google Scholar] [CrossRef]
- Evans, D.W.; Canavera, K.; Kleinpeter, F.L.; Maccubbin, E.; Taga, K. The fears, phobias and anxieties of children with autism spectrum disorders and Down syndrome: Comparisons with developmentally and chronologically age matched children. Child Psychiatry Hum. Dev. 2005, 36, 3–26. [Google Scholar] [CrossRef] [PubMed]
- Beversdorf, D.Q.; Stevens, H.E.; Jones, K.L. Prenatal stress, maternal immune dysregulation, and their association with autism spectrum disorders. Curr. Psychiatry Rep. 2018, 20, 76. [Google Scholar] [CrossRef] [PubMed]
- Fuentes-Zacarias, P.; Murrieta-Coxca, J.M.; Gutierrez-Samudio, R.N.; Schmidt, A.; Markert, U.R.; Morales-Prieto, D.M. Pregnancy and pandemics: Interaction of viral surface proteins and placenta cells. Biochim. Biophys. Acta Mol. Basis. Dis. 2021, 1867, 166218. [Google Scholar] [CrossRef] [PubMed]
- Facchetti, F.; Bugatti, M.; Drera, E.; Tripodo, C.; Sartori, E.; Cancila, V.; Papaccio, M.; Castellani, R.; Casola, S.; Boniotti, M.B.; et al. SARS-CoV2 vertical transmission with adverse effects on the newborn revealed through integrated immunohistochemical, electron microscopy and molecular analyses of Placenta. EBioMedicine 2020, 59, 102951. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Seth, S.; Sharma, R.; Yadav, S.; Mishra, P.; Mukhopadhyay, S. Perinatal outcome and possible vertical transmission of coronavirus disease 2019: Experience from North India. Clin. Exp. Pediatr. 2021, 64, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Kaklamanos, E.G.; Menexes, G.; Makrygiannakis, M.A.; Topitsoglou, V.; Kalfas, S. Tooth wear in a sample of community-dwelling elderly Greeks. Oral Health Prev. Dent. 2020, 18, 133–138. [Google Scholar]
- Moosavi, F.; Hosseini, R.; Saso, L.; Firuzi, O. Modulation of neurotrophic signaling pathways by polyphenols. Drug Des. Devel. Ther. 2016, 10, 23–42. [Google Scholar]
- Theoharides, T.C.; Conti, P. Be aware of SARS-CoV-2 spike protein: There is more than meets the eye. J. Biol. Regul. Homeost. Agents 2021, 35, 833–838. [Google Scholar]
- Villar, J.; Ariff, S.; Gunier, R.B.; Thiruvengadam, R.; Rauch, S.; Kholin, A.; Roggero, P.; Prefumo, F.; do Vale, M.S.; Cardona-Perez, J.A. Maternal and neonatal morbidity and mortality among pregnant women with and without COVID-19 infection: The INTERCOVID multinational cohort study. JAMA Pediatr. 2021, 175, 817–826. [Google Scholar] [CrossRef]
- Lu-Culligan, A.; Chavan, A.R.; Vijayakumar, P.; Irshaid, L.; Courchaine, E.M.; Milano, K.M.; Tang, Z.; Pope, S.D.; Song, E.; Vogels, C.F. SARS-CoV-2 infection in pregnancy is associated with robust inflammatory response at the maternal-fetal interface. medRxiv 2021. [Google Scholar] [CrossRef]
- Narang, K.; Enninga, E.A.L.; Gunaratne, M.D.S.K.; Ibirogba, E.R.; Trad, A.T.A.; Elrefaei, A.; Theiler, R.N.; Ruano, R.; Szymanski, L.M.; Chakraborty, R. SARS-CoV-2 infection and COVID-19 during pregnancy: A multidisciplinary review. Mayo Clin. Proc. 2020, 95, 1750–1765. [Google Scholar] [CrossRef]
- Grammatopoulos, D.K.; Hillhouse, E.W. Role of corticotropin-releasing hormone in onset of labour. Lancet 1999, 354, 1546–1549. [Google Scholar] [CrossRef]
- Ng, E.K.; Leung, T.N.; Tsui, N.B.; Lau, T.K.; Panesar, N.S.; Chiu, R.W.; Lo, Y.M. The concentration of circulating corticotropin-releasing hormone mRNA in maternal plasma is increased in preeclampsia. Clin. Chem. 2003, 49, 727–731. [Google Scholar] [CrossRef] [Green Version]
- Chrousos, G.P. The hypothalamic-pituitary-adrenal axis and immune-mediated inflammation. N. Engl. J. Med. 1995, 332, 1351–1362. [Google Scholar] [CrossRef] [PubMed]
- She, J.; Liu, L.; Liu, W. COVID-19 epidemic: Disease characteristics in children. J. Med. Virol. 2020, 92, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Mo, X.; Hu, Y.; Qi, X.; Jiang, F.; Jiang, Z.; Tong, S. Epidemiology of COVID-19 among Children in China. Pediatrics 2020, 145, e20200702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ludvigsson, J.F. Systematic review of COVID-19 in children shows milder cases and a better prognosis than adults. Acta Paediatr. 2020, 109, 1088–1095. [Google Scholar] [CrossRef]
- Tian, S.; Hu, N.; Lou, J.; Chen, K.; Kang, X.; Xiang, Z.; Chen, H.; Wang, D.; Liu, N.; Liu, D. Characteristics of COVID-19 infection in Beijing. J. Infect. 2020, 80, 401–406. [Google Scholar] [CrossRef] [Green Version]
- Ciotti, M.; Angeletti, S.; Minieri, M.; Giovannetti, M.; Benvenuto, D.; Pascarella, S.; Sagnelli, C.; Bianchi, M.; Bernardini, S.; Ciccozzi, M. COVID-19 outbreak: An overview. Chemotherapy 2019, 64, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Wang, Y.; Chung, H.T.; Chen, C.J. Clinical characteristics of novel coronavirus disease 2019 (COVID-19) in newborns, infants and children. Pediatr. Neonatol. 2020, 61, 131–132. [Google Scholar] [CrossRef]
- Castagnoli, R.; Votto, M.; Licari, A.; Brambilla, I.; Bruno, R.; Perlini, S.; Rovida, F.; Baldanti, F.; Marseglia, G.L. Severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2) infection in children and adolescents: A systematic review. JAMA Pediatr. 2020, 174, 882–889. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greene, A.G.; Saleh, M.; Roseman, E.; Sinert, R. Toxic shock-like syndrome and COVID-19: A case report of multisystem inflammatory syndrome in children (MIS-C). Am. J. Emerg. Med. 2020, 38, 30492–30497. [Google Scholar] [CrossRef] [PubMed]
- Levin, M. Childhood multisystem inflammatory syndrome—A new challenge in the pandemic. N. Engl. J. Med. 2020, 383, 393–395. [Google Scholar] [CrossRef]
- Feldstein, L.R.; Rose, E.B.; Horwitz, S.M.; Collins, J.P.; Newhams, M.M.; Son, M.B.F.; Newburger, J.W.; Kleinman, L.C.; Heidemann, S.M.; Martin, A.A. Multisystem inflammatory syndrome in U.S. children and adolescents. N. Engl. J. Med. 2020, 383, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Tang, K.; Levin, M.; Irfan, O.; Morris, S.K.; Wilson, K.; Klein, J.D.; Bhutta, Z.A. COVID-19 and multisystem inflammatory syndrome in children and adolescents. Lancet Infect. Dis. 2020, 20, e276–e288. [Google Scholar] [CrossRef]
- Rowley, A.H. Understanding SARS-CoV-2-related multisystem inflammatory syndrome in children. Nat. Rev. Immunol. 2020, 20, 453–454. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, L.B.; Bradford, T.R.; Littman, B.H.; Wintroub, B.U. The fibrinogenolytic activity of purified tryptase from human lung mast cells. J. Immunol. 1985, 135, 2762–2767. [Google Scholar]
- Consiglio, C.R.; Cotugno, N.; Sardh, F.; Pou, C.; Amodio, D.; Rodriguez, L.; Tan, Z.; Zicari, S.; Ruggiero, A.; Pascucci, G.R. The immunology of multisystem inflammatory syndrome in children with COVID-19. Cell 2020, 183, 968–981. [Google Scholar] [CrossRef]
- Hagberg, H.; Gressens, P.; Mallard, C. Inflammation during fetal and neonatal life: Implications for neurologic and neuropsychiatric disease in children and adults. Ann. Neurol. 2012, 71, 444–457. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.A.; Thomsen, C. The role of the innate immune system in psychiatric disorders. Mol. Cell Neurosci. 2013, 53, 52–62. [Google Scholar] [CrossRef]
- Chavarria, A.; Alcocer-Varela, J. Is damage in central nervous system due to inflammation? Autoimmun. Rev. 2004, 3, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Le Belle, J.E.; Sperry, J.; Ngo, A.; Ghochani, Y.; Laks, D.R.; Lopez-Aranda, M.; Silva, A.J.; Kornblum, H.I. Maternal inflammation contributes to brain overgrowth and autism-associated behaviors through altered redox signaling in stem and progenitor cells. Stem Cell Rep. 2014, 3, 725–734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.H.; Perl, D.P.; Nair, G.; Li, W.; Maric, D.; Murray, H.; Dodd, S.J.; Koretsky, A.P.; Watts, J.A.; Cheung, V.; et al. Microvascular injury in the brains of patients with Covid-19. N. Engl. J. Med. 2020, 384, 481–483. [Google Scholar] [CrossRef]
- Helms, J.; Kremer, S.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Kummerlen, C.; Collange, O.; Boulay, C.; Fafi-Kremer, S.; Ohana, M. Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 2020, 382, 2268–2270. [Google Scholar] [CrossRef] [PubMed]
- Fotuhi, M.; Mian, A.; Meysami, S.; Raji, C.A. Neurobiology of COVID-19. J. Alzheimers Dis. 2020, 76, 3–19. [Google Scholar] [CrossRef]
- Najjar, S.; Najjar, A.; Chong, D.J.; Pramanik, B.K.; Kirsch, C.; Kuzniecky, R.I.; Pacia, S.V.; Azhar, S. Central nervous system complications associated with SARS-CoV-2 infection: Integrative concepts of pathophysiology and case reports. J. Neuroinflamm. 2020, 17, 231. [Google Scholar] [CrossRef]
- Singh, A.K.; Bhushan, B.; Maurya, A.; Mishra, G.; Singh, S.K.; Awasthi, R. Novel coronavirus disease 2019 (COVID-19) and neurodegenerative disorders. Dermatol. Ther. 2020, 33, e13591. [Google Scholar] [CrossRef]
- Liotta, E.M.; Batra, A.; Clark, J.R.; Shlobin, N.A.; Hoffman, S.C.; Orban, Z.S.; Koralnik, I.J. Frequent neurologic manifestations and encephalopathy-associated morbidity in Covid-19 patients. Ann.Clin. Transl. Neurol. 2020, 7, 2221–2230. [Google Scholar] [CrossRef]
- Koralnik, I.J.; Tyler, K.L. COVID-19: A global threat to the nervous system. Ann. Neurol. 2020, 88, 1–11. [Google Scholar] [CrossRef]
- Nepal, G.; Rehrig, J.H.; Shrestha, G.S.; Shing, Y.K.; Yadav, J.K.; Ojha, R.; Pokhrel, G.; Tu, Z.L.; Huang, D.Y. Neurological manifestations of COVID-19: A systematic review. Crit. Care 2020, 24, 421. [Google Scholar] [CrossRef] [PubMed]
- Favas, T.T.; Dev, P.; Chaurasia, R.N.; Chakravarty, K.; Mishra, R.; Joshi, D.; Mishra, V.N.; Kumar, A.; Singh, V.K.; Pandey, M.; et al. Neurological manifestations of COVID-19: A systematic review and meta-analysis of proportions. Neurol. Sci. 2020, 41, 3437–3470. [Google Scholar] [CrossRef] [PubMed]
- Nazari, S.; Azari, J.A.; Mirmoeeni, S.; Sadeghian, S.; Heidari, M.E.; Assarzadegan, F.; Puormand, S.M.; Ebadi, H.; Fathi, D. Central nervous system manifestations in COVID-19 patients: A systematic review and meta-analysis. Brain Behav. 2021, 11, e02025. [Google Scholar] [CrossRef]
- Kempuraj, D.; Selvakumar, G.P.; Ahmed, M.E.; Raikwar, S.P.; Thangavel, R.; Khan, A.; Zaheer, S.A.; Iyer, S.S.; Burton, C.; James, D.; et al. COVID-19, mast cells, cytokine storm, psychological stress, and neuroinflammation. Neuroscientist 2020, 26, 402–414. [Google Scholar] [CrossRef]
- Schirinzi, T.; Landi, D.; Liguori, C. COVID-19: Dealing with a potential risk factor for chronic neurological disorders. J. Neurol. 2020, 268, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Ongur, D.; Perlis, R.; Goff, D. Psychiatry and COVID-19. JAMA 2020, 324, 1149–1150. [Google Scholar] [CrossRef]
- Vindegaard, N.; Benros, M.E. COVID-19 pandemic and mental health consequences: Systematic review of the current evidence. Brain Behav. Immun. 2020, 89, 531–542. [Google Scholar] [CrossRef]
- Pfefferbaum, B.; North, C.S. Mental health and the Covid-19 pandemic. N. Engl. J. Med. 2020, 383, 510–512. [Google Scholar] [CrossRef]
- Xiang, Y.-T.; Yang, Y.; Li, W.; Zhang, L.; Zhang, Q.; Cheung, T.; Ng, C. Timely mental health care for the 2019 novel coronavirus outbreak is urgently needed. Lancet Psychiatry 2020, 7, 228–229. [Google Scholar] [CrossRef] [Green Version]
- Gordon, J.A.; Borja, S.E. The COVID-19 pandemic: Setting the mental health research agenda. Biol. Psychiatry 2020, 88, 130–131. [Google Scholar] [CrossRef] [PubMed]
- Taquet, M.; Luciano, S.; Geddes, J.R.; Harrison, P.J. Bidirectional associations between COVID-19 and psychiatric disorder: Retrospective cohort studies of 62,354 COVID-19 cases in the USA. Lancet Psychiatry 2021, 8, 130–140. [Google Scholar] [CrossRef]
- Steardo, L., Jr.; Steardo, L.; Verkhratsky, A. Psychiatric face of COVID-19. Transl. Psychiatry 2020, 10, 261. [Google Scholar] [CrossRef]
- Shader, R.I. COVID-19 and depression. Clin. Ther. 2020, 42, 962–963. [Google Scholar] [CrossRef]
- Smith, C.M.; Komisar, J.R.; Mourad, A.; Kincaid, B.R. COVID-19-associated brief psychotic disorder. BMJ Case Rep. 2020, 13, e236940. [Google Scholar] [CrossRef]
- Druss, B.G. Addressing the COVID-19 pandemic in populations with serious mental illness. JAMA Psychiatry 2020, 77, 891–892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steinman, G. COVID-19 and autism. Med. Hypotheses 2020, 142, 109797. [Google Scholar] [CrossRef]
- Baig, A.M. Chronic COVID syndrome: Need for an appropriate medical terminology for long-COVID and COVID long-haulers. J. Med. Virol. 2020, 93, 2555–2556. [Google Scholar] [CrossRef]
- Moreno-Perez, O.; Merino, E.; Leon-Ramirez, J.M.; Andres, M.; Ramos, J.M.; renas-Jimenez, J.; Asensio, S.; Sanchez, R.; Ruiz-Torregrosa, P.; Galan, I. Post-acute COVID-19 Syndrome. Incidence and risk factors: A Mediterranean cohort study. J. Infect. 2021, 82, 378–383. [Google Scholar] [CrossRef]
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef]
- Montagne, A.; Nation, D.A.; Sagare, A.P.; Barisano, G.; Sweeney, M.D.; Chakhoyan, A.; Pachicano, M.; Joe, E.; Nelson, A.R.; D’Orazio, L.M.; et al. APOE4 leads to blood-brain barrier dysfunction predicting cognitive decline. Nature 2020, 581, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Baig, A.M. Deleterious outcomes in long-hauler COVID-19: The effects of SARS-CoV-2 on the CNS in chronic COVID syndrome. ACS Chem. Neurosci. 2020, 11, 4017–4020. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Higgins, V.; Sohaei, D.; Diamandis, E.P.; Prassas, I. COVID-19: From an acute to chronic disease? Potential long-term health consequences. Crit. Rev.Clin. Lab. Sci. 2020, 58, 297–310. [Google Scholar] [CrossRef]
- Townsend, L.; Dyer, A.H.; Jones, K.; Dunne, J.; Mooney, A.; Gaffney, F.; O’Connor, L.; Leavy, D.; O’Brien, K.; Dowds, J. Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS ONE 2020, 15, e0240784. [Google Scholar] [CrossRef]
- Theoharides, T.C. Stress, inflammation, and autoimmunity: The 3 modern erinyes. Clin. Ther. 2020, 42, 742–744. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C. The impact of psychological stress on mast cells. Ann. Allergy Asthma Immunol. Off. Publ. Am. Coll. Allergy Asthma Immunol. 2020, 125, 388–392. [Google Scholar] [CrossRef]
- Keller, F.; Persico, A.M. The neurobiological context of autism. Mol. Neurobiol. 2003, 28, 1–22. [Google Scholar] [CrossRef]
- El-Ansary, A.; Al-Ayadhi, L. Neuroinflammation in autism spectrum disorders. J. Neuroinflamm. 2012, 9, 265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Young, A.M.; Chakrabarti, B.; Roberts, D.; Lai, M.C.; Suckling, J.; Baron-Cohen, S. From molecules to neural morphology: Understanding neuroinflammation in autism spectrum condition. Mol. Autism 2016, 7, 9. [Google Scholar] [CrossRef] [Green Version]
- Prata, J.; Machado, A.S.; von Doellinger, O.; Almeida, M.I.; Barbosa, M.A.; Coelho, R.; Santos, S.G. The contribution of inflammation to autism spectrum disorders: Recent clinical evidence. Methods Mol. Biol. 2019, 2011, 493–510. [Google Scholar]
- Platt, M.P.; Agalliu, D.; Cutforth, T. Hello from the other side: How autoantibodies circumvent the blood-brain barrier in autoimmune encephalitis. Front. Immunol. 2017, 8, 442. [Google Scholar] [CrossRef] [Green Version]
- Theoharides, T.C.; Angelidou, A.; Alysandratos, K.D.; Zhang, B.; Asadi, S.; Francis, K.; Toniato, E.; Kalogeromitros, D. Mast cell activation and autism. Biochim. Biophys. Acta 2012, 1822, 34–41. [Google Scholar] [CrossRef] [Green Version]
- Theoharides, T.C.; Donelan, J.M.; Papadopoulou, N.; Cao, J.; Kempuraj, D.; Conti, P. Mast cells as targets of corticotropin-releasing factor and related peptides. Trends Pharmacol. Sci. 2004, 25, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, J.M.; Mortensen, E.L.; Christensen, D.S.; Rozing, M.; Brunsgaard, H.; Meincke, R.H.; Petersen, G.L.; Lund, R. Prenatal and early postnatal stress and later life inflammation. Psychoneuroendocrinology 2018, 88, 158–166. [Google Scholar] [CrossRef]
- Marsland, A.L.; Walsh, C.; Lockwood, K.; John-Henderson, N.A. The effects of acute psychological stress on circulating and stimulated inflammatory markers: A systematic review and meta-analysis. Brain Behav. Immun. 2017, 64, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, M.D.; Graham, A.M.; Feczko, E.; Miranda-Dominguez, O.; Rasmussen, J.M.; Nardos, R.; Entringer, S.; Wadhwa, P.D.; Buss, C.; Fair, D.A. Maternal IL-6 during pregnancy can be estimated from newborn brain connectivity and predicts future working memory in offspring. Nat. Neurosci. 2018, 21, 765–772. [Google Scholar] [CrossRef]
- Huang, M.; Pang, X.; Karalis, K.; Theoharides, T.C. Stress-induced interleukin-6 release in mice is mast cell-dependent and more pronounced in Apolipoprotein E knockout mice. Cardiovasc. Res. 2003, 59, 241–249. [Google Scholar] [CrossRef] [Green Version]
- Desai, A.; Jung, M.Y.; Olivera, A.; Gilfillan, A.M.; Prussin, C.; Kirshenbaum, A.S.; Beaven, M.A.; Metcalfe, D.D. IL-6 promotes an increase in human mast cell numbers and reactivity through suppression of suppressor of cytokine signaling 3. J. Allergy Clin. Immunol. 2016, 137, 1863–1871. [Google Scholar] [CrossRef] [Green Version]
- O’Keeffe, G.W. A new role for placental IL-6 signalling in determining neurodevelopmental outcome. Brain Behav. Immun. 2017, 62, 9–10. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Konstantinidou, A. Corticotropin-releasing hormone and the blood-brain-barrier. Front. Biosci. 2007, 12, 1615–1628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiorentino, M.; Sapone, A.; Senger, S.; Camhi, S.S.; Kadzielski, S.M.; Buie, T.M.; Kelly, D.L.; Cascella, N.; Fasano, A. Blood-brain barrier and intestinal epithelial barrier alterations in autism spectrum disorders. Mol. Autism 2016, 7, 49. [Google Scholar] [CrossRef] [Green Version]
- Kempuraj, D.; Papadopoulou, N.G.; Lytinas, M.; Huang, M.; Kandere-Grzybowska, K.; Madhappan, B.; Boucher, W.; Christodoulou, S.; Athanassiou, A.; Theoharides, T.C. Corticotropin-releasing hormone and its structurally related urocortin are synthesized and secreted by human mast cells. Endocrinology 2004, 145, 43–48. [Google Scholar] [CrossRef] [Green Version]
- Mastorakos, G.; Chrousos, G.P.; Weber, J.S. Recombinant interleukin-6 activates the hypothalamic-pituitary-adrenal axis in humans. J. Clin. Endocrinol. Metab. 1993, 77, 1690–1694. [Google Scholar]
- Stolp, H.B.; Dziegielewska, K.M.; Ek, C.J.; Habgood, M.D.; Lane, M.A.; Potter, A.M.; Saunders, N.R. Breakdown of the blood-brain barrier to proteins in white matter of the developing brain following systemic inflammation. Cell Tissue Res. 2005, 320, 369–378. [Google Scholar] [CrossRef]
- Esposito, P.; Chandler, N.; Kandere-Grzybowska, K.; Basu, S.; Jacobson, S.; Connolly, R.; Tutor, D.; Theoharides, T.C. Corticotropin-releasing hormone (CRH) and brain mast cells regulate blood-brain-barrier permeability induced by acute stress. J. Pharmacol. Exp. Ther. 2002, 303, 1061–1066. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Doyle, R. Autism, gut-blood-brain barrier and mast cells. J. Clin. Psychopharm. 2008, 28, 479–483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rozniecki, J.J.; Sahagian, G.G.; Kempuraj, D.; Tao, K.; Jocobson, S.; Zhang, B.; Theoharides, T.C. Brain metastases of mouse mammary adenocarcinoma is increased by acute stress. Brain Res. 2010, 1366, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Rozniecki, J.J.; Sahagian, G.; Jocobson, S.; Kempuraj, D.; Conti, P.; Kalogeromitros, D. Impact of stress and mast cells on brain metastases. J. Neuroimmunol. 2008, 205, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Saunders, N.R. Ontogeny of the blood-brain barrier. Exp. Eye Res. 1977, 25, 523–550. [Google Scholar] [CrossRef]
- Saunders, N.R.; Liddelow, S.A.; Dziegielewska, K.M. Barrier mechanisms in the developing brain. Front Pharmacol. 2012, 3, 46. [Google Scholar] [CrossRef] [Green Version]
- Bueno, D.; Parvas, M.; Hermelo, I.; Garcia-Fernandez, J. Embryonic blood-cerebrospinal fluid barrier formation and function. Front. Neurosci. 2014, 8, 343. [Google Scholar] [CrossRef] [Green Version]
- Saunders, N.R.; Dziegielewska, K.M.; Mollgard, K.; Habgood, M.D. Recent developments in understanding barrier mechanisms in the developing brain: Drugs and drug transporters in pregnancy, susceptibility or protection in the fetal brain? Annu. Rev. Pharmacol. Toxicol. 2019, 59, 487–505. [Google Scholar] [CrossRef] [PubMed]
- Koehn, L.; Habgood, M.; Huang, Y.; Dziegielewska, K.; Saunders, N. Determinants of drug entry into the developing brain. F1000 Res. 2019, 8, 1372. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Azuine, R.E.; Zhang, Y.; Hou, W.; Hong, X.; Wang, G.; Riley, A.; Pearson, C.; Zuckerman, B.; Wang, X. Association of cord plasma biomarkers of in utero acetaminophen exposure with risk of attention-deficit/hyperactivity disorder and autism spectrum disorder in childhood. JAMA Psychiatry 2019, 77, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Ge, J.; Yang, M.; Feng, J.; Qiao, M.; Jiang, R.; Bi, J.; Zhan, G.; Xu, X.; Wang, L.; et al. Vicarious traumatization in the general public, members, and non-members of medical teams aiding in COVID-19 control. Brain Behav. Immun. 2020, 88, 916–919. [Google Scholar] [CrossRef]
- Zhang, K.; Zhou, X.; Liu, H.; Hashimoto, K. Treatment concerns for psychiatric symptoms in patients with COVID-19 with or without psychiatric disorders. Br. J. Psychiatry 2020, 217, 351. [Google Scholar] [CrossRef] [Green Version]
- Walton, M.; Murray, E.; Christian, M.D. Mental health care for medical staff and affiliated healthcare workers during the COVID-19 pandemic. Eur. Heart, J. Acute Cardiovasc. Care 2020, 9, 241–247. [Google Scholar] [CrossRef]
- Ren, Y.; Zhou, Y.; Qian, W.; Li, Z.; Liu, Z.; Wang, R.; Qi, L.; Yang, J.; Song, X.; Zeng, L.; et al. Letter to the Editor “A longitudinal study on the mental health of general population during the COVID-19 epidemic in China”. Brain Behav. Immunol. 2020, 87, 132–133. [Google Scholar] [CrossRef]
- Loades, M.E.; Chatburn, E.; Higson-Sweeney, N.; Reynolds, S.; Shafran, R.; Brigden, A.; Linney, C.; McManus, M.N.; Borwick, C.; Crawley, E. Rapid systematic review: The impact of social isolation and loneliness on the mental health of children and adolescents in the context of COVID-19. J. Am. Acad. Child Adolesc. Psychiatry 2020, 59, 1218–1239. [Google Scholar] [CrossRef]
- Peters, J.L.; Cohen, S.; Staudenmayer, J.; Hosen, J.; Platts-Mills, T.A.; Wright, R.J. Prenatal negative life events increases cord blood IgE: Interactions with dust mite allergen and maternal atopy. Allergy 2012, 67, 545–551. [Google Scholar] [CrossRef]
- Wang, I.J.; Wen, H.J.; Chiang, T.L.; Lin, S.J.; Guo, Y.L. Maternal psychologic problems increased the risk of childhood atopic dermatitis 1. Pediatr. Allergy Immunol. 2016, 27, 169–176. [Google Scholar] [CrossRef]
- Andersson, N.W.; Hansen, M.V.; Larsen, A.D.; Hougaard, K.S.; Kolstad, H.A.; Schlunssen, V. Prenatal maternal stress and atopic diseases in the child: A systematic review of observational human studies. Allergy 2016, 71, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Medsker, B.; Forno, E.; Simhan, H.; Celedon, J.C. Prenatal stress, prematurity, and asthma. Obstet. Gynecol. Surv. 2015, 70, 773–779. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosa, M.J.; Lee, A.G.; Wright, R.J. Evidence establishing a link between prenatal and early-life stress and asthma development. Curr. Opin. Allergy Clin. Immunol. 2018, 18, 148–158. [Google Scholar] [CrossRef] [PubMed]
- Van de Loo, K.F.; van Gelder, M.M.; Roukema, J.; Roeleveld, N.; Merkus, P.J.; Verhaak, C.M. Prenatal maternal psychological stress and childhood asthma and wheezing: A meta-analysis. Eur. Respir. J. 2016, 47, 133–146. [Google Scholar] [CrossRef]
- Postorino, V.; Kerns, C.M.; Vivanti, G.; Bradshaw, J.; Siracusano, M.; Mazzone, L. Anxiety disorders and obsessive-compulsive disorder in individuals with autism spectrum disorder. Curr. Psychiatry Rep. 2017, 19, 92. [Google Scholar] [CrossRef]
- Cai, R.Y.; Richdale, A.L.; Uljarevic, M.; Dissanayake, C.; Samson, A.C. Emotion regulation in autism spectrum disorder: Where we are and where we need to go. Autism Res. 2018, 11, 962–978. [Google Scholar] [CrossRef]
- Ye, Q.; Wang, B.; Mao, J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J. Infect. 2020, 80, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Wu, D.; Guo, W.; Cao, Y.; Huang, D.; Wang, H.; Wang, T.; Zhang, X.; Chen, H.; Yu, H.; et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Investig. 2020, 130, 2620–2629. [Google Scholar] [CrossRef] [Green Version]
- Conti, P.; Ronconi, G.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Frydas, I.; Kritas, S.K. Induction of pro-inflammatory cytokines (IL-1 and IL-6) and lung inflammation by Coronavirus-19 (COVI-19 or SARS-CoV-2): Anti-inflammatory strategies. J. Biol. Regul. Homeost. Agents 2020, 34, 327–331. [Google Scholar]
- Giamarellos-Bourboulis, E.J.; Netea, M.G.; Rovina, N.; Akinosoglou, K.; Antoniadou, A.; Antonakos, N.; Damoraki, G.; Gkavogianni, T.; Adami, M.-E.; Katsaounou, P.; et al. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host. Microbe 2020, 27, 992–1000. [Google Scholar] [CrossRef]
- Tang, Y.; Liu, J.; Zhang, D.; Xu, Z.; Ji, J.; Wen, C. Cytokine storm in COVID-19: The current evidence and treatment strategies. Front. Immunol. 2020, 11, 1708. [Google Scholar] [CrossRef]
- Paces, J.; Strizova, Z.; Smrz, D.; Cerny, J. COVID-19 and the immune system. Physiol. Res. 2020, 69, 379–388. [Google Scholar] [CrossRef]
- Ragab, D.; Salah, E.H.; Taeimah, M.; Khattab, R.; Salem, R. The COVID-19 cytokine storm; What we know so far. Front. Immunol. 2020, 11, 1446. [Google Scholar] [CrossRef]
- Brodin, P. Immune determinants of COVID-19 disease presentation and severity. Nat. Med. 2021, 27, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Canna, S.W.; Cron, R.Q. Highways to hell: Mechanism-based management of cytokine storm syndromes. J. Allergy Clin. Immunol. 2020, 146, 949–959. [Google Scholar] [CrossRef]
- Herold, T.; Jurinovic, V.; Arnreich, C.; Lipworth, B.J.; Hellmuth, J.C.; von Bergwelt-Baildon, M.; Klein, M.; Weinberger, T. Elevated levels of IL-6 and CRP predict the need for mechanical ventilation in COVID-19. J. Allergy Clin. Immunol. 2020, 146, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Ma, Q.; Li, C.; Liu, R.; Zhao, L.; Wang, W.; Zhang, P.; Liu, X.; Gao, G.; Liu, F. Profiling serum cytokines in COVID-19 patients reveals IL-6 and IL-10 are disease severity predictors. Emerg. Microbes. Infect. 2020, 9, 1123–1130. [Google Scholar] [CrossRef]
- Mazzoni, A.; Salvati, L.; Maggi, L.; Capone, M.; Vanni, A.; Spinicci, M.; Mencarini, J.; Caporale, R.; Peruzzi, B.; Antonelli, A.; et al. Impaired immune cell cytotoxicity in severe COVID-19 is IL-6 dependent. J. Clin. Investig. 2020, 130, 4694–4703. [Google Scholar] [CrossRef]
- Liu, F.; Li, L.; Xu, M.; Wu, J.; Luo, D.; Zhu, Y.; Li, B.; Song, X.; Zhou, X. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J. Clin. Virol. 2020, 127, 104370. [Google Scholar] [CrossRef] [PubMed]
- Copaescu, A.; Smibert, O.; Gibson, A.; Phillips, E.J.; Trubiano, J.A. The role of IL-6 and other mediators in the cytokine storm associated with SARS-CoV-2 infection. J. Allergy Clin. Immunol. 2020, 146, 518–534. [Google Scholar] [CrossRef] [PubMed]
- Conti, P.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Kritas, S.K.; Frydas, I.; Younes, A.; Ronconi, G. Coronavirus-19 (SARS-CoV-2) induces acute severe lung inflammation via IL-1 causing cytokine storm in COVID-19: A promising inhibitory strategy. J. Biol. Regul. Homeost. Agents 2020, 34, 1971–1975. [Google Scholar]
- Kritas, S.K.; Ronconi, G.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Conti, P. Mast cells contribute to coronavirus-induced inflammation: New anti-inflammatory strategy. J. Biol. Regul. Homeost. Agents 2020, 34, 9–14. [Google Scholar]
- Theoharides, T.C. COVID-19, pulmonary mast cells, cytokine storms, and beneficial actions of luteolin. Biofactors 2020, 46, 306–308. [Google Scholar] [CrossRef] [PubMed]
- Afrin, L.B.; Weinstock, L.B.; Molderings, G.J. Covid-19 hyperinflammation and post-Covid-19 illness may be rooted in mast cell activation syndrome. Int. J. Infect. Dis. 2020, 100, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C. Potential association of mast cells with COVID-19. Ann. Allergy Asthma Immunol. 2020, 126, 217–218. [Google Scholar] [CrossRef] [PubMed]
- Motta, J.D.S., Jr.; Miggiolaro, A.F.R.D.S.; Nagashima, S.; de Paula, C.B.V.; Baena, C.P.; Scharfstein, J.; de Noronha, L. Mast cells in alveolar septa of COVID-19 patients: A pathogenic pathway that may link interstitial edema to immunothrombosis. Front. Immunol. 2020, 11, 574862. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Valent, P.; Akin, C. Mast cells, mastocytosis, and related disorders. N. Engl. J. Med. 2015, 373, 163–172. [Google Scholar] [CrossRef]
- Fombonne, E. Epidemiology of pervasive developmental disorders. Pediatr. Res. 2009, 65, 591–598. [Google Scholar] [CrossRef]
- McPartland, J.; Volkmar, F.R. Autism and related disorders. Handb. Clin. Neurol. 2012, 106, 407–418. [Google Scholar] [PubMed] [Green Version]
- Chang, H.Y.; Seo, J.H.; Kim, H.Y.; Kwon, J.W.; Kim, B.J.; Kim, H.B.; Lee, S.Y.; Jang, G.C.; Song, D.J.; Kim, W.K. Allergic diseases in preschoolers are associated with psychological and behavioural problems. Allergy Asthma Immunol. Res. 2013, 5, 315–321. [Google Scholar] [CrossRef] [Green Version]
- Jyonouchi, H. Food allergy and autism spectrum disorders: Is there a link? Curr. Allergy Asthma Rep. 2009, 9, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, H.; Chen, X.; Zhang, J.; Tong, G.; Sun, Y. Association of food hypersensitivity in children with the risk of autism spectrum disorder: A meta-analysis. Eur. J. Pediatr. 2021, 180, 999–1008. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Snetselaar, L.G.; Jing, J.; Liu, B.; Strathearn, L.; Bao, W. Association of food allergy and other allergic conditions with autism spectrum disorder in children. JAMA Netw. Open 2018, 1, e180279. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Thomas, S.; Lee, B.K. Parent-reported prevalence of food allergies in children with autism spectrum disorder: National health interview survey, 2011–2015. Autism Res. 2019, 12, 802–805. [Google Scholar] [CrossRef]
- Peretti, S.; Mariano, M.; Mazzocchetti, C.; Mazza, M.; Pino, M.C.; Verrotti Di, P.A.; Valenti, M. Diet: The keystone of autism spectrum disorder? Nutr. Neurosci. 2019, 22, 825–839. [Google Scholar] [CrossRef]
- Mostafa, G.A.; Al-Ayadhi, L.Y. The possible relationship between allergic manifestations and elevated serum levels of brain specific auto-antibodies in autistic children. J. Neuroimmunol. 2013, 261, 77–81. [Google Scholar] [CrossRef]
- Ravn, N.H.; Halling, A.S.; Berkowitz, A.G.; Rinnov, M.R.; Silverberg, J.I.; Egeberg, A.; Thyssen, J.P. How does parental history of atopic disease predict the risk of atopic dermatitis in a child? A systematic review and meta-analysis. J. Allergy Clin. Immunol. 2020, 145, 1182–1193. [Google Scholar] [CrossRef]
- Estes, M.L.; McAllister, A.K. Maternal immune activation: Implications for neuropsychiatric disorders. Science 2016, 353, 772–777. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zerbo, O.; Leong, A.; Barcellos, L.; Bernal, P.; Fireman, B.; Croen, L.A. Immune mediated conditions in autism spectrum disorders. Brain Behav. Immun. 2015, 46, 232–236. [Google Scholar] [CrossRef] [Green Version]
- Comi, A.M.; Zimmerman, A.W.; Frye, V.H.; Law, P.A.; Peeden, J.N. Familial clustering of autoimmune disorders and evaluation of medical risk factors in autism. J. Child Neurol. 1999, 14, 388–394. [Google Scholar] [CrossRef]
- Jarmołowska, B.; Bukało, M.; Fiedorowicz, E.; Cieślińska, A.; Kordulewska, N.K.; Moszyńska, M.; Świątecki, A.; Kostyra, E. Role of Milk-Derived Opioid Peptides and Proline Dipeptidyl Peptidase-4 in Autism Spectrum Disorders. Nutrients 2019, 11, 87. [Google Scholar] [CrossRef] [Green Version]
- Zhou, L.; Chen, L.; Li, X.; Li, T.; Dong, Z.; Wang, Y.T. Food allergy induces alteration in brain inflammatory status and cognitive impairments. Behav. Brain Res. 2019, 364, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Msallam, R.; Balla, J.; Rathore, A.P.S.; Kared, H.; Malleret, B.; Saron, W.A.A.; Liu, Z.; Hang, J.W.; Dutertre, C.A.; Larbi, A.; et al. Fetal mast cells mediate postnatal allergic responses dependent on maternal IgE. Science 2020, 370, 941–950. [Google Scholar] [CrossRef]
- Lenz, K.M.; Pickett, L.A.; Wright, C.L.; Davis, K.T.; Joshi, A.; McCarthy, M.M. Mast cells in the developing brain determine adult sexual behavior. J. Neurosci. 2018, 38, 8044–8059. [Google Scholar] [CrossRef] [Green Version]
- Theoharides, T.C.; Petra, A.I.; Taracanova, A.; Panagiotidou, S.; Conti, P. Targeting IL-33 in autoimmunity and inflammation. J. Pharmacol. Exp. Ther. 2015, 354, 24–31. [Google Scholar] [CrossRef] [Green Version]
- Theoharides, T.C.; Leeman, S.E. Effect of IL-33 on de novo synthesized mediators from human mast cells. J. Allergy Clin. Immunol. 2019, 143, 451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Dong, H.; Li, N.; Zhang, S.; Sun, J.; Zhang, S.; Qian, Y. Activated brain mast cells contribute to postoperative cognitive dysfunction by evoking microglia activation and neuronal apoptosis. J. Neuroinflamm. 2016, 13, 127. [Google Scholar] [CrossRef] [Green Version]
- Gurish, M.F.; Austen, K.F. Developmental origin and functional specialization of mast cell subsets. Immunity 2012, 37, 25–33. [Google Scholar] [CrossRef] [Green Version]
- Olivera, A.; Beaven, M.A.; Metcalfe, D.D. Mast cells signal their importance in health and disease. J. Allergy Clin. Immunol. 2018, 142, 381–393. [Google Scholar] [CrossRef] [Green Version]
- Galli, S.J.; Tsai, M.; Piliponsky, A.M. The development of allergic inflammation. Nature 2008, 454, 445–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theoharides, T.C.; Alysandratos, K.D.; Angelidou, A.; Delivanis, D.A.; Sismanopoulos, N.; Zhang, B.; Asadi, S.; Vasiadi, M.; Weng, Z.; Miniati, A.; et al. Mast cells and inflammation. Biochim. Biophys. Acta 2012, 1822, 21–33. [Google Scholar] [CrossRef] [Green Version]
- Mukai, K.; Tsai, M.; Saito, H.; Galli, S.J. Mast cells as sources of cytokines, chemokines, and growth factors. Immunol. Rev. 2018, 282, 121–150. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Alysandratos, K.D.; Angelidou, A.; Delivanis, D.A.; Sismanopoulos, N.; Zhang, B.; Asadi, S.; Vasiadi, M.; Weng, Z.; Miniati, A. Interleukin-1 family cytokines and mast cells: Activation and inhibition. J. Biol. Regul. Homeost. Agents 2019, 33, 1–6. [Google Scholar]
- Taracanova, A.; Tsilioni, I.; Conti, P.; Norwitz, E.R.; Leeman, S.E.; Theoharides, T.C. Substance P and IL-33 administered together stimulate a marked secretion of IL-1beta from human mast cells, inhibited by methoxyluteolin. Proc. Natl. Acad. Sci. USA 2018, 115, e9381–e9390. [Google Scholar] [CrossRef] [Green Version]
- Kandere-Grzybowska, K.; Letourneau, R.; Kempuraj, D.; Donelan, J.; Poplawski, S.; Boucher, W.; Athanassiou, A.; Theoharides, T.C. IL-1 induces vesicular secretion of IL-6 without degranulation from human mast cells. J. Immunol. 2003, 171, 4830–4836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taracanova, A.; Alevizos, M.; Karagkouni, A.; Weng, Z.; Norwitz, E.; Conti, P.; Leeman, S.E.; Theoharides, T.C. SP and IL-33 together markedly enhance TNF synthesis and secretion from human mast cells mediated by the interaction of their receptors. Proc. Natl. Acad. Sci. USA 2017, 114, e4002–e4009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rozniecki, J.J.; Dimitriadou, V.; Lambracht-Hall, M.; Pang, X.; Theoharides, T.C. Morphological and functional demonstration of rat dura mast cell-neuron interactions in vitro and in vivo. Brain Res. 1999, 849, 1–15. [Google Scholar] [CrossRef]
- Polyzoidis, S.; Koletsa, T.; Panagiotidou, S.; Ashkan, K.; Theoharides, T.C. Mast cells in meningiomas and brain inflammation. J. Neuroinflammation 2015, 12, 170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pang, X.; Letourneau, R.; Rozniecki, J.J.; Wang, L.; Theoharides, T.C. Definitive characterization of rat hypothalamic mast cells. Neuroscience 1996, 73, 889–902. [Google Scholar] [CrossRef]
- Kandere-Grzybowska, K.; Gheorghe, D.; Priller, J.; Esposito, P.; Huang, M.; Gerard, N.; Theoharides, T.C. Stress-induced dura vascular permeability does not develop in mast cell-deficient and neurokinin-1 receptor knockout mice. Brain Res. 2003, 980, 213–220. [Google Scholar] [CrossRef]
- Matsumoto, I.; Inoue, Y.; Shimada, T.; Aikawa, T. Brain mast cells act as an immune gate to the hypothalamic-pituitary-adrenal axis in dogs. J. Exp. Med. 2001, 194, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Bugajski, A.J.; Chlap, Z.; Gadek-Michalska, A.; Borycz, J.; Bugajski, J. Degranulation and decrease in histamine levels of thalamic mast cells coincides with corticosterone secretion induced by compound 48/80. Inflamm. Res. 1995, 44, S50–S51. [Google Scholar] [CrossRef] [PubMed]
- Kalogeromitros, D.; Syrigou, E.I.; Makris, M.; Kempuraj, D.; Stavrianeas, N.G.; Vasiadi, M.; Theoharides, T.C. Nasal provocation of patients with allergic rhinitis and the hypothalamic-pituitary-adrenal axis. Ann. Allergy Asthma Immunol. 2007, 98, 269–273. [Google Scholar] [CrossRef]
- Scaccianoce, S.; Lombardo, K.; Nicolai, R.; Affricano, D.; Angelucci, L. Studies on the involvement of histamine in the hypothalamic-pituitary-adrenal axis activation induced by nerve growth factor. Life Sci. 2000, 67, 3143–3152. [Google Scholar] [CrossRef]
- Alysandratos, K.; Asadi, S.; Angelidou, A.; Zhang, B.; Sismanopoulos, N.; Yang, H.; Critchfield, A.; Theoharides, T.C. Neurotensin and CRH interactions augment human mast cell activation. PLoS ONE 2012, 7, e48934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asadi, S.; Alysandratos, K.-D.; Angelidou, A.; Miniati, A.; Sismanopoulos, N.; Vasiadi, M.; Zhang, B.; Kalogeromitros, D.; Theoharides, T.C. Substance P (SP) induces expression of functional corticotropin-releasing hormone receptor-1 (CRHR-1) in human mast cells. J. Investig. Dermatol. 2012, 132, 324–329. [Google Scholar] [CrossRef] [Green Version]
- Marshall, J.S.; Portales-Cervantes, L.; Leong, E. Mast cell responses to viruses and pathogen products. Int. J. Mol. Sci. 2019, 20, 4241. [Google Scholar] [CrossRef] [Green Version]
- Ziegler, C.G.K.; Allon, S.J.; Nyquist, S.K.; Mbano, I.M.; Miao, V.N.; Tzouanas, C.N.; Cao, Y.; Yousif, A.S.; Bals, J.; Hauser, B.M. SARS-CoV-2 receptor ACE2 is an interferon-stimulated gene in human airway epithelial cells and is detected in specific cell subsets across tissues. Cell 2020, 181, 1016–1035. [Google Scholar] [CrossRef]
- Dietrich, N.; Rohde, M.; Geffers, R.; Kroger, A.; Hauser, H.; Weiss, S.; Gekara, N.O. Mast cells elicit proinflammatory but not type I interferon responses upon activation of TLRs by bacteria. Proc. Natl. Acad. Sci. USA 2010, 107, 8748–8753. [Google Scholar] [CrossRef] [Green Version]
- Bawazeer, M.A.; Theoharides, T.C. IL-33 stimulates human mast cell release of CCL5 and CCL2 via MAPK and NF-kappaB, inhibited by methoxyluteolin. Eur. J. Pharmacol. 2019, 865, 172760. [Google Scholar] [CrossRef]
- Saluja, R.; Khan, M.; Church, M.K.; Maurer, M. The role of IL-33 and mast cells in allergy and inflammation. Clin. Transl. Allergy 2015, 5, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conti, P.; Caraffa, A.; Tete, G.; Gallenga, C.E.; Ross, R.; Kritas, S.K.; Frydas, I.; Younes, A.; Di, E.P.; Ronconi, G. Mast cells activated by SARS-CoV-2 release histamine which increases IL-1 levels causing cytokine storm and inflammatory reaction in COVID-19. J. Biol. Regul. Homeost. Agents 2020, 34, 1629–1632. [Google Scholar]
- Zhang, B.; Asadi, S.; Weng, Z.; Sismanopoulos, N.; Theoharides, T.C. Stimulated human mast cells secrete mitochondrial components that have autocrine and paracrine inflammatory actions. PLoS ONE 2012, 7, e49767. [Google Scholar] [CrossRef]
- Collins, L.V.; Hajizadeh, S.; Holme, E.; Jonsson, I.M.; Tarkowski, A. Endogenously oxidized mitochondrial DNA induces in vivo and in vitro inflammatory responses. J. Leukoc. Biol. 2004, 75, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Sursal, T.; Adibnia, Y.; Zhao, C.; Zheng, Y.; Li, H.; Otterbein, L.E.; Hauser, C.J.; Itagaki, K. Mitochondrial DAMPs increase endothelial permeability through neutrophil dependent and independent pathways. PLoS ONE 2013, 8, e59989. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, B.; Angelidou, A.; Alysandratos, K.D.; Vasiadi, M.; Francis, K.; Asadi, S.; Theoharides, A.; Sideri, K.; Lykouras, L.; Kalogeromitros, D. Mitochondrial DNA and anti-mitochondrial antibodies in serum of autistic children. J. Neuroinflammation 2010, 7, 80. [Google Scholar] [CrossRef] [Green Version]
- Frye, R.E.; Cakir, J.; Rose, S.; Palmer, R.F.; Austin, C.; Curtin, P.; Arora, M. Mitochondria may mediate prenatal environmental influences in autism spectrum disorder. J. Pers. Med. 2021, 11, 218. [Google Scholar] [CrossRef] [PubMed]
- Scozzi, D.; Cano, M.; Ma, L.; Zhou, D.; Zhu, J.H.; O’Halloran, J.A.; Goss, C.W.; Rauseo, A.M.; Liu, Z.; Sahu, S.K.; et al. Circulating mitochondrial DNA is an early indicator of severe illness and mortality from COVID-19. JCI Insight 2021, 6, e143299. [Google Scholar]
- Demopoulos, C.; Antonopoulou, S.; Theoharides, T.C. COVID-19, microthromboses, inflammation, and platelet activating factor. Biofactors 2020, 46, 927–933. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Antonopoulou, S.; Demopoulos, C.A. Coronavirus 2019, microthromboses, and platelet activating factor. Clin. Ther. 2020, 42, 1850–1852. [Google Scholar] [CrossRef]
- Damialis, A.; Gilles, S.; Sofiev, M.; Sofieva, V.; Kolek, F.; Bayr, D.; Plaza, M.P.; Leier-Wirtz, V.; Kaschuba, S.; Ziska, L.H. Higher airborne pollen concentrations correlated with increased SARS-CoV-2 infection rates, as evidenced from 31 countries across the globe. Proc. Natl. Acad. Sci. USA 2021, 118, e2019034118. [Google Scholar] [CrossRef]
- Subhramanyam, C.S.; Wang, C.; Hu, Q.; Dheen, S.T. Microglia-mediated neuroinflammation in neurodegenerative diseases. Semin. Cell Dev. Biol. 2019, 94, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Colonna, M.; Butovsky, O. Microglia function in the central nervous system during health and neurodegeneration. Annu. Rev. Immunol. 2017, 35, 441–468. [Google Scholar] [CrossRef]
- Voet, S.; Prinz, M.; van Loo, G. Microglia in central nervous system inflammation and multiple sclerosis pathology. Trends Mol. Med. 2019, 25, 112–123. [Google Scholar] [CrossRef]
- Perry, V.H.; Nicoll, J.A.; Holmes, C. Microglia in neurodegenerative disease. Nat. Rev. Neurol. 2010, 6, 193–201. [Google Scholar] [CrossRef]
- Ransohoff, R.M. How neuroinflammation contributes to neurodegeneration. Science 2016, 353, 777–783. [Google Scholar] [CrossRef]
- Hickman, S.; Izzy, S.; Sen, P.; Morsett, L.; El, K.J. Microglia in neurodegeneration. Nat. Neurosci. 2018, 21, 1359–1369. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.X.; Kim, G.H.; Tan, J.W.; Riso, A.E.; Sun, Y.; Xu, E.Y.; Liao, G.Y.; Xu, H.; Lee, S.H.; Do, N.Y. Elevated protein synthesis in microglia causes autism-like synaptic and behavioral aberrations. Nat. Commun. 2020, 11, 1797. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.B.; Tsilioni, I.; Leeman, S.E.; Theoharides, T.C. Neurotensin stimulates sortilin and mTOR in human microglia inhibitable by methoxyluteolin, a potential therapeutic target for autism. Proc. Natl. Acad. Sci. USA 2016, 113, e7049–e7058. [Google Scholar] [CrossRef] [Green Version]
- Vargas, G.; Medeiros Geraldo, L.H.; Gedeao, S.N.; Viana, P.M.; Regina Souza, L.F.; Carvalho Alcantara, G.F. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and glial cells: Insights and perspectives. Brain Behav. Immun. Health 2020, 7, 100127. [Google Scholar] [CrossRef]
- Tremblay, M.E.; Madore, C.; Bordeleau, M.; Tian, L.; Verkhratsky, A. Neuropathobiology of COVID-19: The role for glia. Front. Cell Neurosci. 2020, 14, 592214. [Google Scholar] [CrossRef]
- Lee, N.; Chan, P.K.; Ip, M.; Wong, E.; Ho, J.; Ho, C.; Cockram, C.S.; Hui, D.S. Anti-SARS-CoV IgG response in relation to disease severity of severe acute respiratory syndrome. J. Clin. Virol. 2006, 35, 179–184. [Google Scholar] [CrossRef]
- Sotgiu, S.; Manca, S.; Gagliano, A.; Minutolo, A.; Melis, M.C.; Pisuttu, G.; Scoppola, C.; Bolognesi, E.; Clerici, M.; Guerini, F.R.; et al. Immune regulation of neurodevelopment at the mother-foetus interface: The case of autism. Clin. Transl. Immunol. 2020, 9, e1211. [Google Scholar] [CrossRef] [PubMed]
- Hendriksen, E.; van Bergeijk, D.; Oosting, R.S.; Redegeld, F.A. Mast cells in neuroinflammation and brain disorders. Neurosci. Biobehav. Rev. 2017, 79, 119–133. [Google Scholar] [CrossRef]
- Skaper, S.D.; Facci, L.; Giusti, P. Neuroinflammation, microglia and mast cells in the pathophysiology of neurocognitive disorders: A review. CNS Neurol. Disord. Drug Targets 2014, 13, 1654–1666. [Google Scholar] [CrossRef]
- Skaper, S.D.; Facci, L.; Zusso, M.; Giusti, P. Neuroinflammation, mast cells, and glia: Dangerous liaisons. Neuroscientist 2017, 23, 478–498. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, Y.; Dong, H.; Xu, Y.; Zhang, S. Induction of microglial activation by mediators released from mast cells. Cell Physiol. Biochem. 2016, 38, 1520–1531. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Stewart, J.M.; Panagiotidou, S.; Melamed, I. Mast cells, brain inflammation and autism. Eur. J. Pharmacol. 2015, 778, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Zhang, X.; Wang, Y.; Zhou, X.; Qian, Y.; Zhang, S. Suppression of brain mast cells degranulation inhibits microglial activation and central nervous system inflammation. Mol. Neurobiol. 2017, 54, 997–1007. [Google Scholar] [CrossRef]
- Shaik-Dasthagirisaheb, Y.B.; Conti, P. The role of mast cells in Alzheimer’s disease. Adv. Clin. Exp. Med. 2016, 25, 781–787. [Google Scholar] [CrossRef] [Green Version]
- Kempuraj, D.; Mentor, S.; Thangavel, R.; Ahmed, M.E.; Selvakumar, G.P.; Raikwar, S.P.; Dubova, I.; Zaheer, S.; Iyer, S.S.; Zaheer, A. Mast cells in stress, pain, blood-brain barrier, neuroinflammation and Alzheimer’s disease. Front. Cell Neurosci. 2019, 13, 54. [Google Scholar] [CrossRef] [Green Version]
- Hansen, D.V.; Hanson, J.E.; Sheng, M. Microglia in Alzheimer’s disease. J. Cell Biol. 2018, 217, 459–472. [Google Scholar] [CrossRef] [PubMed]
- Hatziagelaki, E.; Adamaki, M.; Tsilioni, I.; Dimitriadis, G.; Theoharides, T.C. Myalgic encephalomyelitis/chronic fatigue syndrome-metabolic disease or disturbed homeostasis due to focal inflammation in the hypothalamus? J. Pharmacol. Exp. Ther. 2018, 367, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Breach, M.R.; Dye, C.N.; Joshi, A.; Platko, S.; Gilfarb, R.A.; Krug, A.R.; Franceschelli, D.V.; Galan, A.; Dodson, C.M.; Lenz, K.M. Maternal allergic inflammation in rats impacts the offspring perinatal neuroimmune milieu and the development of social play, locomotor behavior, and cognitive flexibility. Brain Behav Immun. 2021, 95, 269–286. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Ji, P.; Riopelle, R.J.; Dow, K.E. Functional expression of corticotropin-releasing hormone (CRH) receptor 1 in cultured rat microglia. J. Neurochem. 2002, 80, 287–294. [Google Scholar] [CrossRef] [Green Version]
- Podlesek, A.; Komidar, L.; Kavcic, V. The relationship between perceived stress and subjective cognitive decline during the COVID-19 epidemic. Front Psychol. 2021, 12, 647971. [Google Scholar] [CrossRef]
- Martin, S.; Dicou, E.; Vincent, J.P.; Mazella, J. Neurotensin and the neurotensin receptor-3 in microglial cells. J. Neurosci. Res. 2005, 81, 322–326. [Google Scholar] [CrossRef]
- Angelidou, A.; Francis, K.; Vasiadi, M.; Alysandratos, K.-D.; Zhang, B.; Theoharides, A.; Lykouras, L.; Sideri, K.; Kalogeromitros, D.; Theoharides, T.C. Neurotensin is increased in serum of young children with autistic disorder. J. Neuroinflam. 2010, 7, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsilioni, I.; Dodman, N.; Petra, A.I.; Taliou, A.; Francis, K.; Moon-Fanelli, A.; Shuster, L.; Theoharides, T.C. Elevated serum neurotensin and CRH levels in children with autistic spectrum disorders and tail-chasing Bull Terriers with a phenotype similar to autism. Transl. Psychiatry 2014, 4, e466. [Google Scholar] [CrossRef] [Green Version]
- Almehmadi, K.A.; Tsilioni, T.T.C. Increased expression of miR-155p5 in amygdala of children with Autism Spectrum Disorder. Autism Res. 2019, 13, 18–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsilioni, I.; Pantazopoulos, H.; Conti, P.; Leeman, S.E.; Theoharides, T.C. IL-38 inhibits microglial inflammatory mediators and is decreased in amygdala of children with autism spectrum disorder. Proc. Natl. Acad. Sci. USA 2020, 117, 16475–16480. [Google Scholar] [CrossRef] [PubMed]
- Jack, C.S.; Arbour, N.; Manusow, J.; Montgrain, V.; Blain, M.; McCrea, E.; Shapiro, A.; Antel, J.P. TLR signaling tailors innate immune responses in human microglia and astrocytes. J. Immunol. 2005, 175, 4320–4330. [Google Scholar] [CrossRef] [Green Version]
- Murta, V.; Villarreal, A.; Ramos, A.J. Severe acute respiratory syndrome Coronavirus 2 impact on the central nervous system: Are astrocytes and microglia main players or merely bystanders? ASN Neuro. 2020, 12, 1759091420954960. [Google Scholar] [CrossRef] [PubMed]
- Akin, C.; Valent, P.; Metcalfe, D.D. Mast cell activation syndrome: Proposed diagnostic criteria. J. Allergy Clin. Immunol. 2010, 126, 1099–1104. [Google Scholar] [CrossRef] [Green Version]
- Theoharides, T.C.; Tsilioni, I.; Ren, H. Recent advances in our understanding of mast cell activation—Or should it be mast cell mediator disorders? Expert. Rev. Clin. Immunol. 2019, 15, 639–656. [Google Scholar] [CrossRef] [PubMed]
- Enstrom, A.; Krakowiak, P.; Onore, C.; Pessah, I.N.; Hertz-Picciotto, I.; Hansen, R.L.; Van de Water, J.A.; Ashwood, P. Increased IgG4 levels in children with autism disorder. Brain Behav. Immun. 2009, 23, 389–395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef]
- Kumar, S.; Khodoun, M.; Kettleson, E.M.; McKnight, C.; Reponen, T.; Grinshpun, S.A.; Adhikari, A. Glyphosate-rich air samples induce IL-33, TSLP and generate IL-13 dependent airway inflammation. Toxicology 2014, 325, 42–51. [Google Scholar] [CrossRef] [Green Version]
- Rossignol, D.A.; Frye, R.E. A systematic review and meta-analysis of immunoglobulin g abnormalities and the therapeutic use of Intravenous Immunoglobulins (IVIG) in autism spectrum disorder. J. Pers. Med. 2021, 11, 488. [Google Scholar] [CrossRef]
- Church, M.K. Allergy, histamine and antihistamines. Handb. Exp. Pharmacol. 2017, 241, 321–331. [Google Scholar]
- Vasiadi, M.; Kalogeromitros, D.; Kempuraj, D.; Clemons, A.; Zhang, B.; Chliva, C.; Makris, M.; Wolfberg, A.; House, M.; Theoharides, T.C. Rupatadine inhibits proinflammatory mediator secretion from human mast cells triggered by different stimuli. Int. Arch. Allergy Immunol. 2010, 151, 38–45. [Google Scholar] [CrossRef]
- Alevizos, M.; Karagkouni, A.; Vasiadi, M.; Sismanopoulos, N.; Makris, M.; Kalogeromitros, D.; Theoharides, T.C. Rupatadine inhibits inflammatory mediator release from human LAD2 cultured mast cells stimulated by PAF. Ann. Allergy Asthma Immunol. 2013, 111, 524–527. [Google Scholar] [CrossRef] [PubMed]
- Siebenhaar, F.; Förtsch, A.; Krause, K.; Weller, K.; Metz, M.; Magerl, M.; Martus, P.; Church, M.K.; Maurer, M. Rupatadine improves quality of life in mastocytosis: A randomized, double-blind, placebo-controlled trial. Allergy 2013, 68, 949–952. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Stewart, J.M. Antihistamines and mental status. J. Clin. Psychopharmacol. 2016, 36, 195–197. [Google Scholar] [CrossRef] [PubMed]
- Zahiruddin, S.; Basist, P.; Parveen, A.; Parveen, R.; Khan, W.; Gaurav; Ahmad, S. Ashwagandha in brain disorders: A review of recent developments. J. Ethnopharmacol. 2020, 257, 112876. [Google Scholar] [CrossRef] [PubMed]
- Ng, Q.X.; Loke, W.; Foo, N.X.; Tan, W.J.; Chan, H.W.; Lim, D.Y.; Yeo, W.S. A systematic review of the clinical use of Withania somnifera (Ashwagandha) to ameliorate cognitive dysfunction. Phytother. Res. 2020, 34, 583–590. [Google Scholar] [CrossRef]
- Beversdorf, D.Q.; Saklayen, S.; Higgins, K.F.; Bodner, K.E.; Kanne, S.M.; Christ, S.E. Effect of propranolol on word fluency in autism. Cogn. Behav. Neurol. 2011, 24, 11–17. [Google Scholar] [CrossRef]
- Beversdorf, D.Q.; Saklayen, S.; Higgins, K.F.; Bodner, K.E.; Kanne, S.M.; Christ, S.E. alpha2-adrenergic agonists or stimulants for preschool-age children with attention-deficit/hyperactivity disorder. JAMA 2021, 325, 2067–2075. [Google Scholar]
- Banas, K.; Sawchuk, B. Clonidine as a treatment of behavioural disturbances in autism spectrum disorder: A systematic literature review. J. Can. Acad. Child Adolesc. Psychiatry 2020, 29, 110–120. [Google Scholar]
- Reichow, B.; Volkmar, F.R.; Bloch, M.H. Systematic review and meta-analysis of pharmacological treatment of the symptoms of attention-deficit/hyperactivity disorder in children with pervasive developmental disorders. J. Autism Dev. Disord. 2013, 43, 2435–2441. [Google Scholar] [CrossRef] [Green Version]
- Politte, L.C.; Scahill, L.; Figueroa, J.; McCracken, J.T.; King, B.; McDougle, C.J. A randomized, placebo-controlled trial of extended-release guanfacine in children with autism spectrum disorder and ADHD symptoms: An analysis of secondary outcome measures. Neuropsychopharmacology 2018, 43, 1772–1778. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okazaki, K.; Yamamuro, K.; Iida, J.; Kishimoto, T. Guanfacine monotherapy for ADHD/ASD comorbid with Tourette syndrome: A case report. Ann. Gen. Psychiatry 2019, 18, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ming, X.; Gordon, E.; Kang, N.; Wagner, G.C. Use of clonidine in children with autism spectrum disorders. Brain Dev. 2008, 30, 454–460. [Google Scholar] [CrossRef]
- Del Giudice, E.; Rinaldi, L.; Passarotto, M.; Facchinetti, F.; D’Arrigo, A.; Guiotto, A.; Carbonare, M.D.; Battistin, L.; Leon, A. Cannabidiol, unlike synthetic cannabinoids, triggers activation of RBL-2H3 mast cells. J. Leukoc. Biol. 2007, 81, 1512–1522. [Google Scholar] [CrossRef] [PubMed]
- Caslin, H.; Kiwanuka, K.N.; Haque, T.T.; Taruselli, M.; Macknight, H.P.; Paranjape, A.; Ryan, J.J. Controlling mast cell activation and homeostasis: Work Influenced by Bill Paul that continues today. Front. Immunol. 2018, 9, 868. [Google Scholar] [CrossRef]
- Noda, S.; Krueger, J.G.; Guttman-Yassky, E. The translational revolution and use of biologics in patients with inflammatory skin diseases. J. Allergy Clin. Immunol. 2015, 135, 324–336. [Google Scholar] [CrossRef]
- Leonardi, C.L.; Powers, J.L.; Matheson, R.T.; Goffe, B.S.; Zitnik, R.; Wang, A.; Gottlieb, A.B. Etanercept as monotherapy in patients with psoriasis. N. Engl. J. Med. 2003, 349, 2014–2022. [Google Scholar] [CrossRef] [Green Version]
- Ruzicka, T.; Mihara, R. Anti-interleukin-31 receptor a antibody for atopic dermatitis. N. Engl. J. Med. 2017, 376, 2093. [Google Scholar] [CrossRef]
- Olivieri, I.; D’Angelo, S.; Palazzi, C.; Padula, A. Treatment strategies for early psoriatic arthritis. Expert. Opin. Pharmacother. 2009, 10, 271–282. [Google Scholar] [CrossRef]
- Heinrich, M.C.; Griffith, D.J.; Druker, B.J.; Wait, C.L.; Ott, K.A.; Zigler, A.J. Inhibition of c-kit receptor tyrosine kinase activity by STI 571, a selective tyrosine kinase inhibitor. Blood 2000, 96, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Gotlib, J.; Kluin-Nelemans, J.C.; George, T.I.; Akin, C.; Sotlar, K.; Hermine, O.; Awan, F.T.; Hexner, E.; Mauro, M.J.; Sternberg, D.W.; et al. Efficacy and safety of midostaurin in advanced systemic mastocytosis. N. Engl. J. Med. 2016, 374, 2530–2541. [Google Scholar] [CrossRef]
- Finn, D.F.; Walsh, J.J. Twenty-first century mast cell stabilizers. Br. J. Pharmacol. 2013, 170, 23–37. [Google Scholar] [CrossRef] [Green Version]
- Theoharides, T.C.; Sieghart, W.; Greengard, P.; Douglas, W.W. Antiallergic drug cromolyn may inhibit histamine secretion by regulating phosphorylation of a mast cell protein. Science 1980, 207, 80–82. [Google Scholar] [CrossRef]
- Oka, T.; Kalesnikoff, J.; Starkl, P.; Tsai, M.; Galli, S.J. Evidence questioning cromolyn’s effectiveness and selectivity as a ‘mast cell stabilizer’ in mice. Lab. Investig. 2012, 92, 1472–1482. [Google Scholar] [CrossRef] [PubMed]
- Weng, Z.; Zhang, B.; Asadi, S.; Sismanopoulos, N.; Butcher, A.; Fu, X.; Katsarou-Katsari, A.; Antoniou, C.; Theoharides, T.C. Quercetin is more effective than cromolyn in blocking human mast cell cytokine release and inhibits contact dermatitis and photosensitivity in humans. PLoS ONE 2012, 7, e33805-k. [Google Scholar] [CrossRef]
- Dos Santos, R.V.; Magerl, M.; Martus, P.; Zuberbier, T.; Church, M.; Escribano, L.; Maurer, M. Topical sodium cromoglicate relieves allergen- and histamine-induced dermal pruritus. Br. J. Dermatol. 2010, 162, 674–676. [Google Scholar] [CrossRef] [PubMed]
- Weng, Z.; Patel, A.B.; Panagiotidou, S.; Theoharides, T.C. The novel flavone tetramethoxyluteolin is a potent inhibitor of human mast cells. J. Allergy Clin. Immunol. 2015, 135, 1044–1052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Church, M.K.; Gradidge, C.F. Potentiation of histamine release by sodium cromoglycate. Life Sci. 1978, 23, 1899–1904. [Google Scholar] [CrossRef]
- Kempuraj, D.; Madhappan, B.; Christodoulou, S.; Boucher, W.; Cao, J.; Papadopoulou, N.; Cetrulo, C.L.; Theoharides, T.C. Flavonols inhibit proinflammatory mediator release, intracellular calcium ion levels and protein kinase C theta phosphorylation in human mast cells. Br. J. Pharmacol. 2005, 145, 934–944. [Google Scholar] [CrossRef] [Green Version]
- Seelinger, G.; Merfort, I.; Schempp, C.M. Anti-oxidant, anti-inflammatory and anti-allergic activities of luteolin. Planta Med. 2008, 74, 1667–1677. [Google Scholar] [CrossRef]
- Calis, Z.; Mogulkoc, R.; Baltaci, A.K. The roles of flavonoles/flavonoids in neurodegeneration and neuroinflammation. Mini. Rev. Med. Chem. 2020, 20, 1475–1488. [Google Scholar] [CrossRef] [PubMed]
- Jager, A.K.; Saaby, L. Flavonoids and the CNS. Molecules 2011, 16, 1471–1485. [Google Scholar] [CrossRef] [Green Version]
- Leyva-Lopez, N.; Gutierrez-Grijalva, E.P.; Ambriz-Perez, D.L.; Heredia, J.B. Flavonoids as cytokine modulators: A possible therapy for inflammation-related diseases. Int. J. Mol. Sci. 2016, 17, 921. [Google Scholar] [CrossRef]
- Harwood, M.; Nielewska-Nikiel, B.; Borzelleca, J.F.; Flamm, G.W.; Williams, G.M.; Lines, T.C. A critical review of the data related to the safety of quercetin and lack of evidence of in vivo toxicity, including lack of genotoxic/carcinogenic properties. Food Chem. Toxicol. 2007, 45, 2179–2205. [Google Scholar] [CrossRef]
- Okamoto, T. Safety of quercetin for clinical application (Review). Int. J. Mol. Med. 2005, 16, 275–278. [Google Scholar] [CrossRef]
- Andres, S.; Pevny, S.; Ziegenhagen, R.; Bakhiya, N.; Schäfer, B.; Hirsch-Ernst, K.; Lampen, A. Safety aspects of the use of quercetin as a dietary supplement. Mol. Nutr. Food Res. 2018, 62, 1700447. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Su, W.; Jin, J.; Chen, J.; Li, X.; Zhang, X.; Sun, M.; Sun, S.; Fan, P.; An, D.; et al. Identification of luteolin as enterovirus 71 and coxsackievirus A16 inhibitors through reporter viruses and cell viability-based screening. Viruses 2014, 6, 2778–2795. [Google Scholar] [CrossRef] [Green Version]
- Fan, W.; Qian, S.; Qian, P.; Li, X. Antiviral activity of luteolin against Japanese encephalitis virus. Virus Res. 2016, 220, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Ma, L.; Wang, H.; Wu, S.; Huang, H.; Gu, Z.; Jiang, J.; Li, Y. Luteolin decreases the yield of influenza A virus in vitro by interfering with the coat protein I complex expression. J. Nat. Med. 2019, 73, 487–496. [Google Scholar] [CrossRef]
- Russo, M.; Moccia, S.; Spagnuolo, C.; Tedesco, I.; Russo, G.L. Roles of flavonoids against coronavirus infection. Chem. Biol. Interact. 2020, 328, 109211. [Google Scholar] [CrossRef]
- Derosa, G.; Maffioli, P.; D’Angelo, A.; Di, P.F. A role for quercetin in coronavirus disease 2019 (COVID-19). Phytother. Res. 2020, 35, 1230–1236. [Google Scholar] [CrossRef]
- Rezai-Zadeh, K.; Ehrhart, J.; Bai, Y.; Sanberg, P.R.; Bickford, P.; Tan, J.; Shytle, R.D. Apigenin and luteolin modulate microglial activation via inhibition of STAT1-induced CD40 expression. J. Neuroinflammation 2008, 5, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, S.; Kelley, K.W.; Johnson, R.W. Luteolin reduces IL-6 production in microglia by inhibiting JNK phosphorylation and activation of AP-1. Proc. Natl. Acad. Sci. USA 2008, 105, 7534–7539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burton, M.D.; Rytych, J.L.; Amin, R.; Johnson, R.W. Dietary luteolin reduces proinflammatory microglia in the brain of senescent mice. Rejuvenation. Res. 2016, 19, 286–292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, A.B.; Theoharides, T.C. Methoxyluteolin inhibits neuropeptide-stimulated proinflammatory mediator release via mTOR activation from human mast cells. J. Pharmacol. Exp. Ther. 2017, 361, 462–471. [Google Scholar] [CrossRef] [Green Version]
- Dajas, F.; Rivera-Megret, F.; Blasina, F.; Arredondo, F.; Abin-Carriquiry, J.; Costa, G.; Echeverry, C.; Lafon, L.; Heizen, H.; Ferreira, M.; et al. Neuroprotection by flavonoids. Braz. J. Med. Biol. Res. 2003, 36, 1613–1620. [Google Scholar] [CrossRef] [Green Version]
- Kempuraj, D.; Thangavel, R.; Kempuraj, D.D.; Ahmed, M.E.; Selvakumar, G.P.; Raikwar, S.P.; Zaheer, S.A.; Iyer, S.S.; Govindarajan, R.; Chandrasekaran, P.N. Neuroprotective effects of flavone luteolin in neuroinflammation and neurotrauma. Biofactors 2020, 47, 190–197. [Google Scholar] [CrossRef]
- Lin, T.Y.; Lu, C.W.; Wang, S.J. Luteolin protects the hippocampus against neuron impairments induced by kainic acid in rats. Neuro Toxicol. 2016, 55, 48–57. [Google Scholar] [CrossRef]
- Ashaari, Z.; Hassanzadeh, G.; Alizamir, T.; Yousefi, B.; Keshavarzi, Z.; Mokhtari, T. The flavone luteolin improves central nervous system disorders by different mechanisms: A review. J. Mol. Neurosci. 2018, 65, 491–506. [Google Scholar] [CrossRef]
- Bernatoniene, J.; Kazlauskaite, J.A.; Kopustinskiene, D.M. Pleiotropic Effects of Isoflavones in Inflammation and Chronic Degenerative Diseases. Int J Mol Sci. 2021, 22, 5656. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Conti, P.; Economu, M. Brain inflammation, neuropsychiatric disorders, and immunoendocrine effects of luteolin. J. Clin. Psychopharmacol. 2014, 34, 187–189. [Google Scholar] [CrossRef] [Green Version]
- Silva Dos Santos, J.; Gonçalves Cirino, J.P.; de Oliveira Carvalho, P.; Ortega, M.M. The Pharmacological Action of Kaempferol in Central Nervous System Diseases: A Review. Front Pharmacol. 2021, 11, 565700. [Google Scholar] [CrossRef] [PubMed]
- Devi, S.A.; Chamoli, A. Polyphenols as an effective therapeutic intervention against cognitive decline during normal and pathological brain aging. Adv. Exp. Med. Biol. 2020, 1260, 159–174. [Google Scholar] [PubMed]
- Theoharides, T.C.; Stewart, J.M.; Hatziagelaki, E.; Kolaitis, G. Brain “fog”, inflammation and obesity: Key aspects of 2 neuropsychiatric disorders improved by luteolin. Front. Neurosci. 2015, 9, 225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rezai-Zadeh, K.; Douglas, S.R.; Bai, Y.; Tian, J.; Hou, H.; Mori, T.; Zeng, J.; Obregon, D.; Town, T.; Tan, J. Flavonoid-mediated presenilin-1 phosphorylation reduces Alzheimer’s disease beta-amyloid production. J. Cell Mol. Med. 2009, 13, 574–588. [Google Scholar] [CrossRef]
- Theoharides, T.C.; Cholevas, C.; Polyzoidis, K.; Politis, A. Long-COVID syndrome-associated brain fog and chemofog: Luteolin to the rescue. Biofactors 2021, 47, 232–241. [Google Scholar] [CrossRef]
- Gratton, G.; Weaver, S.R.; Burley, C.V.; Low, K.A.; Maclin, E.L.; Johns, P.W.; Pham, Q.S.; Lucas, S.J.E.; Fabiani, M.; Rendeiro, C. Dietary flavanols improve cerebral cortical oxygenation and cognition in healthy adults. Sci. Rep. 2020, 10, 19409. [Google Scholar] [CrossRef]
- Yeh, T.S.; Yuan, C.; Ascherio, A.; Rosner, B.; Willett, W.; Blacker, D. Long-term dietary flavonoid intake and subjective cognitive decline in US men and women. Neurology 2021. [Google Scholar] [CrossRef]
- Du, X.; Hill, R.A. 7,8-Dihydroxyflavone as a pro-neurotrophic treatment for neurodevelopmental disorders. Neurochem. Int. 2015, 89, 170–180. [Google Scholar] [CrossRef]
- Xu, S.L.; Bi, C.W.; Choi, R.C.; Zhu, K.Y.; Miernisha, A.; Dong, T.T.; Tsim, K.W. Flavonoids induce the synthesis and secretion of neurotrophic factors in cultured rat astrocytes: A signaling response mediated by estrogen receptor. Evid. Based Complement Alternat. Med. 2013, 2013, 127075. [Google Scholar] [CrossRef] [Green Version]
- Theoharides, T.C.; Athanassiou, M.; Panagiotidou, S.; Doyle, R. Dysregulated brain immunity and neurotrophin signaling in Rett syndrome and autism spectrum disorders. J. Neuroimmunol. 2015, 279, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Yi, L.; Li, Z.; Yuan, K.; Qu, X.; Chen, J.; Wang, G.; Zhang, H.; Luo, H.; Zhu, L.; Jiang, P.; et al. Small molecules blocking the entry of severe acute respiratory syndrome coronavirus into host cells. J. Virol. 2004, 78, 11334–11339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jo, S.; Kim, S.; Shin, D.H.; Kim, M.S. Inhibition of SARS-CoV 3CL protease by flavonoids. J. Enzyme Inhib. Med. Chem. 2020, 35, 145–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xue, G.; Gong, L.; Yuan, C.; Xu, M.; Wang, X.; Jiang, L.; Huang, M. A structural mechanism of flavonoids in inhibiting serine proteases. Food Funct. 2017, 8, 2437–2443. [Google Scholar] [CrossRef]
- Richman, S.; Morris, M.C.; Broderick, G.; Craddock, T.J.A.; Klimas, N.G.; Fletcher, M.A. Pharmaceutical interventions in chronic fatigue syndrome: A literature-based commentary. Clin. Ther. 2019, 41, 798–805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yi, Y.S. Regulatory roles of flavonoids on inflammasome activation during inflammatory responses. Mol. Nutr. Food Res. 2018, 62, e1800147. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Zhang, J.; Guo, L.; Xu, Y.; Sun, L.; Wang, S.; Feng, Y.; Gou, L.; Zhang, L.; Liu, Y. Protective role of luteolin against cognitive dysfunction induced by chronic cerebral hypoperfusion in rats. Pharmacol. Biochem. Behav. 2014, 126, 122–130. [Google Scholar] [CrossRef]
- Theoharides, T.C. Luteolin supplements: All that glitters is not gold. Biofactors 2020, 47, 242–244. [Google Scholar] [CrossRef] [PubMed]
- Taliou, A.; Zintzaras, E.; Lykouras, L.; Francis, K. An open-label pilot study of a formulation containing the anti-inflammatory flavonoid luteolin and its effects on behavior in children with autism spectrum disorders. Clin. Ther. 2013, 35, 592–602. [Google Scholar] [CrossRef]
- Tsilioni, I.; Taliou, A.; Francis, K.; Theoharides, T.C. Children with Autism Spectrum Disorders, who improved with a luteolin containing dietary formulation, show reduced serum levels of TNF and IL-6. Transl. Psychiatry 2015, 5, e647. [Google Scholar] [CrossRef] [Green Version]
- Beauchamp, G.K.; Keast, R.S.; Morel, D.; Lin, J.; Pika, J.; Han, Q.; Lee, C.H.; Smith, A.B.; Breslin, P.A. Phytochemistry: Ibuprofen-like activity in extra-virgin olive oil. Nature 2005, 437, 45–46. [Google Scholar] [CrossRef] [PubMed]
- Angeloni, C.; Malaguti, M.; Barbalace, M.C.; Hrelia, S. Bioactivity of olive oil phenols in neuroprotection. Int. J. Mol. Sci. 2017, 18, 2230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casamenti, F.; Stefani, M. Olive polyphenols: New promising agents to combat aging-associated neurodegeneration. Expert. Rev. Neurother. 2017, 17, 345–358. [Google Scholar] [CrossRef] [PubMed]
- Omar, S.H.; Scott, C.J.; Hamlin, A.S.; Obied, H.K. Olive biophenols reduces Alzheimer’s pathology in SH-SY5Y cells and appswe mice. Int. J. Mol. Sci. 2018, 20, 125. [Google Scholar] [CrossRef] [Green Version]
- Calahorra, J.; Shenk, J.; Wielenga, V.H.; Verweij, V.; Geenen, B.; Dederen, P.J.; Peinado, M.A.; Siles, E.; Wiesmann, M.; Kiliaan, A.J. Hydroxytyrosol, the major phenolic compound of olive oil, as an acute therapeutic strategy after ischemic stroke. Nutrients 2019, 11, 2430. [Google Scholar] [CrossRef] [Green Version]
- Khalatbary, A.R. Olive oil phenols and neuroprotection. Nutr. Neurosci. 2013, 16, 243–249. [Google Scholar] [CrossRef]
- Marquez-Martin, A.; de La, P.R.; Fernandez-Arche, A.; Ruiz-Gutierrez, V.; Yaqoob, P. Modulation of cytokine secretion by pentacyclic triterpenes from olive pomace oil in human mononuclear cells. Cytokine 2006, 36, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Hornedo-Ortega, R.; Cerezo, A.B.; De Pablos, R.M.; Krisa, S.; Richard, T.; García-Parrilla, M.C.; Troncoso, A.M. Phenolic compounds characteristic of the mediterranean diet in mitigating microglia-mediated neuroinflammation. Front. Cell Neurosci. 2018, 12, 373. [Google Scholar] [CrossRef]
- Bertelli, M.; Kiani, A.K.; Paolacci, S.; Manara, E.; Kurti, D.; Dhuli, K.; Bushati, V.; Miertus, J.; Pangallo, D.; Baglivo, M.; et al. Hydroxytyrosol: A natural compound with promising pharmacological activities. J. Biotechnol. 2020, 309, 29–33. [Google Scholar] [CrossRef]
- Li, W.; Yin, N.; Tao, W.; Wang, Q.; Fan, H.; Wang, Z. Berberine suppresses IL-33-induced inflammatory responses in mast cells by inactivating NF-kappaB and p38 signaling. Int. Immunopharmacol. 2019, 66, 82–90. [Google Scholar] [CrossRef]
- Fu, S.; Ni, S.; Wang, D.; Fu, M.; Hong, T. Berberine suppresses mast cell-mediated allergic responses via regulating FcvarepsilonRI-mediated and MAPK signaling. Int. Immunopharmacol. 2019, 71, 1–6. [Google Scholar] [CrossRef]
- Zhu, J.; Cao, D.; Guo, C.; Liu, M.; Tao, Y.; Zhou, J.; Wang, F.; Zhao, Y.; Wei, J.; Zhang, Y.; et al. Berberine facilitates angiogenesis against ischemic stroke through modulating microglial polarization via AMPK signaling. Cell Mol. Neurobiol. 2019, 39, 751–768. [Google Scholar] [CrossRef] [PubMed]
- Sengenc, E.; Kiykim, E.; Saltik, S. Vitamin D levels in children and adolescents with autism. J. Int. Med. Res. 2020, 48, 300060520934638. [Google Scholar] [CrossRef] [PubMed]
- Petruzzelli, M.G.; Marzulli, L.; Margari, F.; De Giacomo, A.; Gabellone, A.; Giannico, O.V.; Margari, L. Vitamin D Deficiency in Autism Spectrum Disorder: A Cross-Sectional Study. Dis. Markers 2020, 2020, 9292560. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ding, R.; Wang, J. The association between vitamin D status and Autism Spectrum Disorder (ASD): A systematic review and meta-analysis. Nutrients 2020, 13, 86. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C. Vitamin D and atopy. Clin. Ther. 2017, 39, 880–883. [Google Scholar] [CrossRef] [Green Version]
- Chen, D.; Jia, T.; Zhang, Y.; Cao, M.; Loth, E.; Lo, C.Z.; Cheng, W.; Liu, Z.; Gong, W.; Sahakian, B.J.; et al. Neural Biomarkers Distinguish Severe from Mild Autism Spectrum Disorder Among High-Functioning Individuals. Front Hum Neurosci. 2021, 15, 657857. [Google Scholar] [CrossRef]
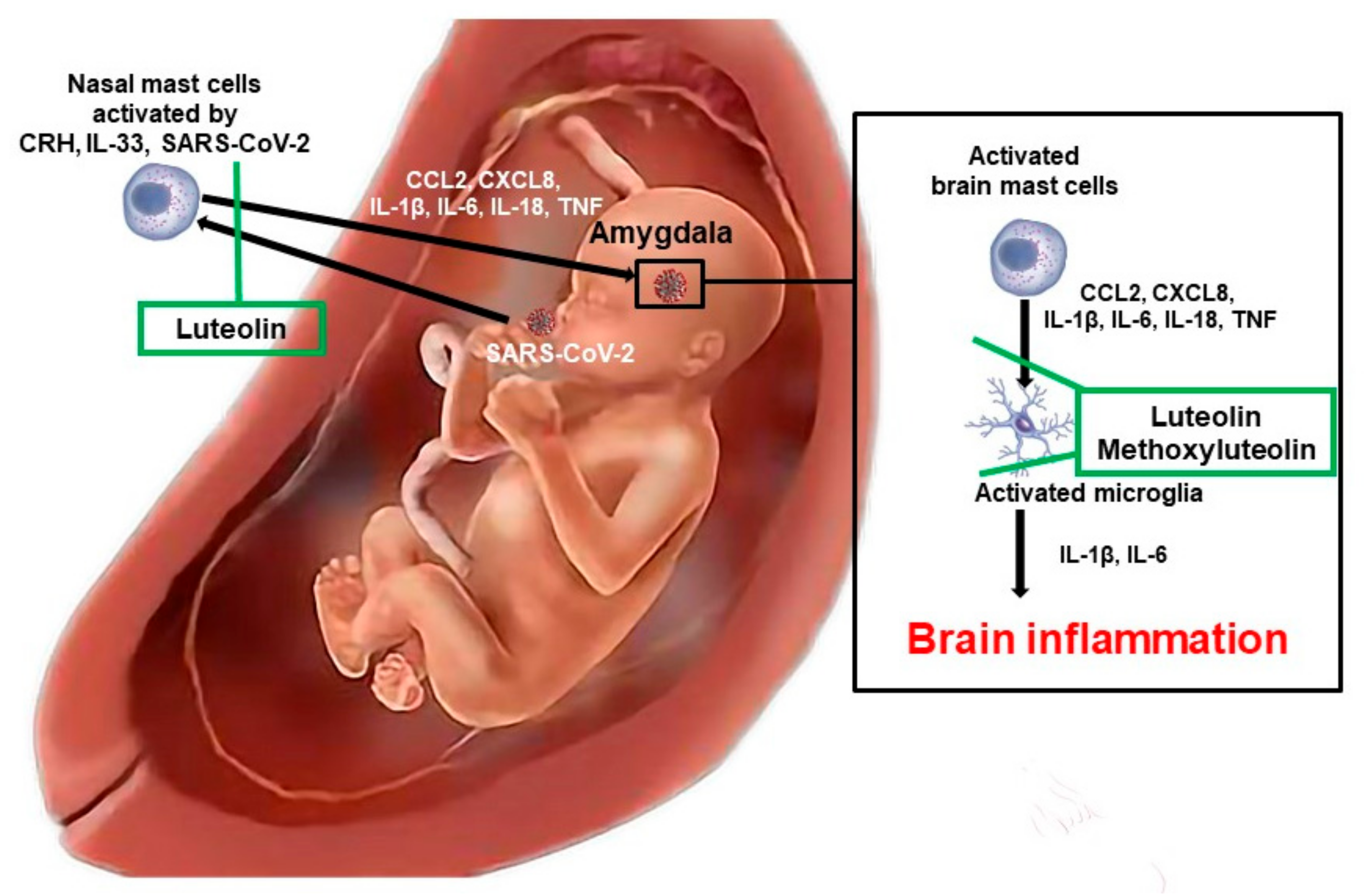
• Autoimmunity | [24,25,44] |
• Allergies | [45,46,47,48,49,50,51,52] |
• Asthma | [50,53] |
• Atopic dermatitis | [54,55] |
• COVID-19 | [56,57] |
• High fever | [58,59] |
• Hypothyroidism | [24] |
• Infection | [58,60,61] |
• Inflammation | [38,39] |
• Low birth weight/preterm birth | [16,20,21,22,62] |
• Pre-eclampsia | [17,18,19] |
• Mastocytosis | [63] |
• Psoriasis | [23,24,25] |
• Rheumatoid arthritis | [24] |
• Stress | [16,62,64,65,66,67,68,69,70,71,72] |
• Chemokines (CCL2, CxCl8) |
• Cytokines (IL-1β, IL-6, IL-18, TNF) |
• Histamine |
• Lipopolysaccharide (LPS) |
• Neuropeptides (CRH, HK-1, Neurotensin, SP) |
• Neurotransmitters |
• Pathogens (SARS-CoV-2) |
• Potassium |
• Prostaglandin D2 |
• Proteases (MMP-3, Thrombin, Tryptase) |
| Blood |
|---|
• IgA, IgG1, IgG4, IgE |
• Immune IgE (RAST for alpha-gal, casein, dust, dust mites, egg, fungi, grass, gluten, pollen) |
• Anti-IgE receptor antibody (basophil activation or histamine release test) |
• CCL2, CXCL8 (IL-8) |
• Chromogranin A * |
• Eosinophilic cationic protein (ECP) |
• Food Intolerance Panel |
• Heparin |
• IL-4, IL-6, IL-31, IL-33 |
• Prostaglandin D2 (PGD2) |
• Tryptase |
| Urine collected for 24 h or first morning void (must be kept and sent cold) |
• N-methylhistamine (NMH) or methylimidazole acetic acid (MIA) |
• PGD2 |
• 23BPG=2,3-Dinor-11β-PGF2α |
| Generic Drug | (Trade Name) |
|---|---|
| Bilastine * | Nonsedating, non-metabolized |
| Cetirizine | Nonsedating |
| Cyproheptadine | Antiserotonergic |
| Diphenhydramine | Sedating |
| Hydroxyzine | Anxiolytic |
| Ketotifen * | Anti-eosinophilic |
| Loratadine | Nonsedating |
| Rupatadine * | Anti-PAF (Platelet activating factor), mast cell inhibitor |
| Tricyclic Antidepressants | |
| Amitriptyline | Weight gain |
| Doxepin | Also histamine-2 receptor antagonist |
| Phenothiazines | |
| Promethazine | Antiemetic |
| Prochlorperazine | Antiemetic, mast cell inhibitor |
• Antagonizes SARS-CoV-2 Spike protein binding | [237,330,331,352] |
• Has broad antiviral properties | [327,328,329] |
• Improves cerebral cortical oxygenation and cognition | [347,348] |
• Induces the synthesis and secretion of BDNF | [77,349,350,351] |
• Inhibits serine proteases required for Spike protein processing | [353,354] |
• Inhibits neuroinflammation | [340,341,342] |
• Inhibits the release and action of PAF | [249,250] |
• Inhibits mast cell stimulation by different triggers | [317,335] |
• Inhibits microglia activation | [259,332,333,334] |
• Interferes with coronavirus replication | [355] |
• Is neuroprotective | [336,337,338,339] |
• Reduces cognitive decline | [61,343,344,345,346] |
• Reduces oxidative stress | [320] |
• Regulates inflammasome activation | [356] |
| Hyperactivity |
• Ashwagandha |
• Chamomile/Passiflora/Valerian extract |
• Clonidine or guanfacine |
• C-Acetyl cysteine (NAC) |
• Hydroxyzine |
• Propranolol |
| Allergic Inflammation |
• Berberine |
• Luteolin * |
• Rupatadine |
• Vitamin D3 |
| Neuronal fatigue |
• Folinate calcium or methylfolate |
• Glutathione |
• Methyl B12 |
• S-Adenosylmethionine (SAMe) |
| OCD |
• Aripiprazole |
• Risperidone |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Theoharides, T.C. Ways to Address Perinatal Mast Cell Activation and Focal Brain Inflammation, including Response to SARS-CoV-2, in Autism Spectrum Disorder. J. Pers. Med. 2021, 11, 860. https://doi.org/10.3390/jpm11090860
Theoharides TC. Ways to Address Perinatal Mast Cell Activation and Focal Brain Inflammation, including Response to SARS-CoV-2, in Autism Spectrum Disorder. Journal of Personalized Medicine. 2021; 11(9):860. https://doi.org/10.3390/jpm11090860
Chicago/Turabian StyleTheoharides, Theoharis C. 2021. "Ways to Address Perinatal Mast Cell Activation and Focal Brain Inflammation, including Response to SARS-CoV-2, in Autism Spectrum Disorder" Journal of Personalized Medicine 11, no. 9: 860. https://doi.org/10.3390/jpm11090860
APA StyleTheoharides, T. C. (2021). Ways to Address Perinatal Mast Cell Activation and Focal Brain Inflammation, including Response to SARS-CoV-2, in Autism Spectrum Disorder. Journal of Personalized Medicine, 11(9), 860. https://doi.org/10.3390/jpm11090860






