Unconventional Therapy with IgY in a Psoriatic Mouse Model Targeting Gut Microbiome
Abstract
:1. Introduction
2. Materials and Methods
2.1. Immunoglobulin Y
2.2. Animal Model
- -
- Ps group (8 mice-1:1 sex ratio, with a mean weight 20.4 ± 2.9 g) received a daily topical dose of 62.5 mg IMQ-based cream (5% Aldara Cream, Meda AB Sweden) on the shaved back region, for 6 consecutive days. The daily dose contains 3.125 mg of active compound. The mice that were designed for clinical and immunological evaluation were sacrificed on day 7 of the experiment;
- -
- IgY-treated Ps group (12 mice-1:1 sex ratio, with a mean weight 21.33 ± 2.14 g) with induced psoriatic dermatitis as described above, received (starting with day 7) a gavage dose of 37.5 µg IgY, for 5 consecutive days; the dose matches the dose of IgY given to a human adult (g/kg) according to a study case [48]. Mice were sacrificed on day 20, the day on which it was macroscopically assessed that experimental psoriatic dermatitis was remitted;
- -
- Naturally remitted Ps group (8 mice—1:1 sex ratio, with a mean weight 17.87 ± 0.81 g) with induced psoriatic dermatitis were allowed to heal naturally and were sacrificed on day 22—the day on which the natural remission was assessed macroscopically;
- -
- Control group (8 mice—1:1 sex ratio, with a mean weight 20.26 ± 1.36 g). Healthy mice with no treatment housed and fed in the same room with all the presented experimental groups and subjected to the same manipulation as the IgY-treated group but with sham gavage.
2.3. Scoring Severity of IMQ-Induced Skin Inflammation and Healing Assesment
2.4. Sampling of Biological Material and Processing of Samples
2.5. Flow Cytometry Analysis
2.6. Statistical Analysis
3. Results
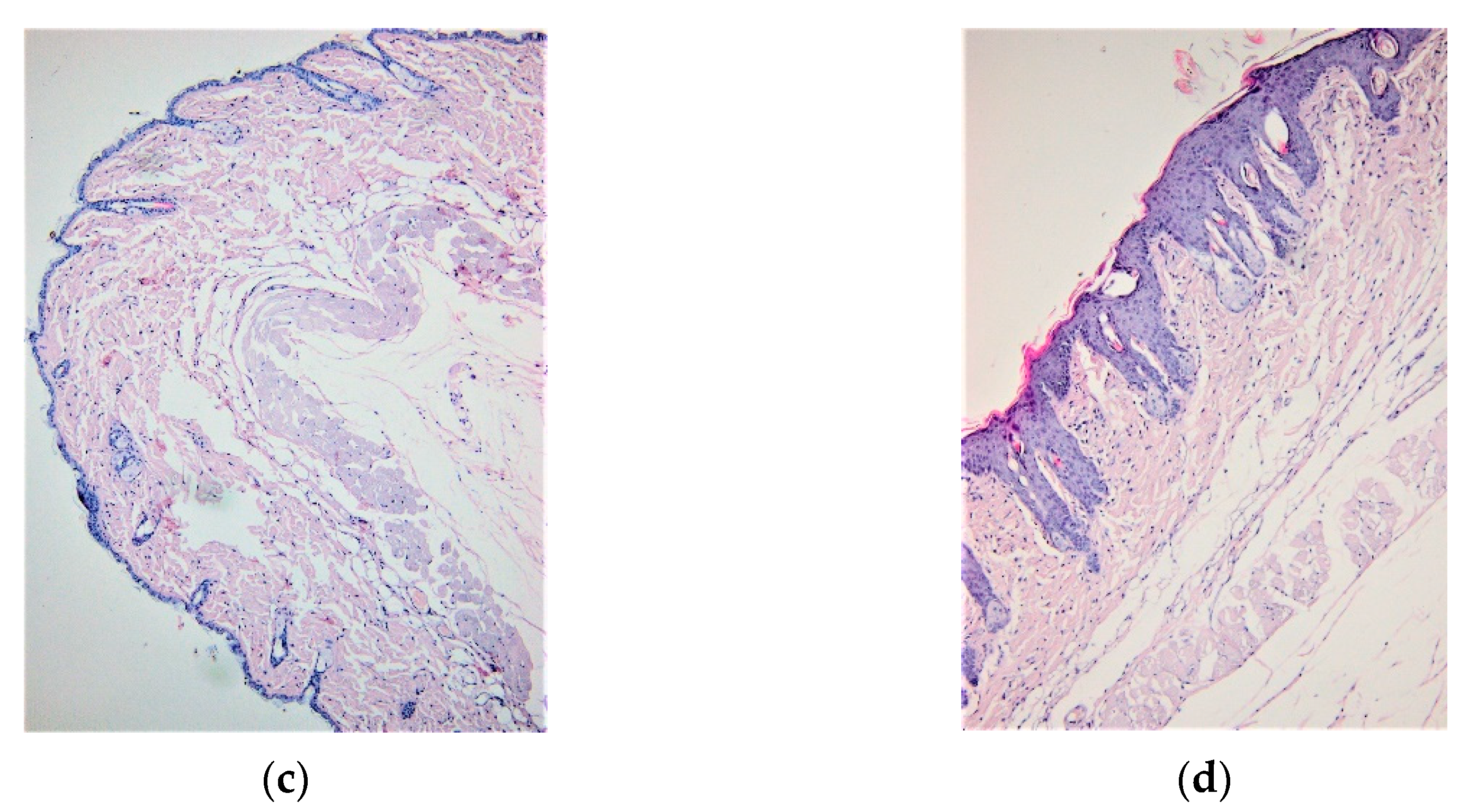
3.1. IMQ-Based Experimental Murine Model of Psoriatic Dermatitis
3.2. IgY Treatment-Induced Changes in Experimental Murine Model of Psoriatic Dermatitis
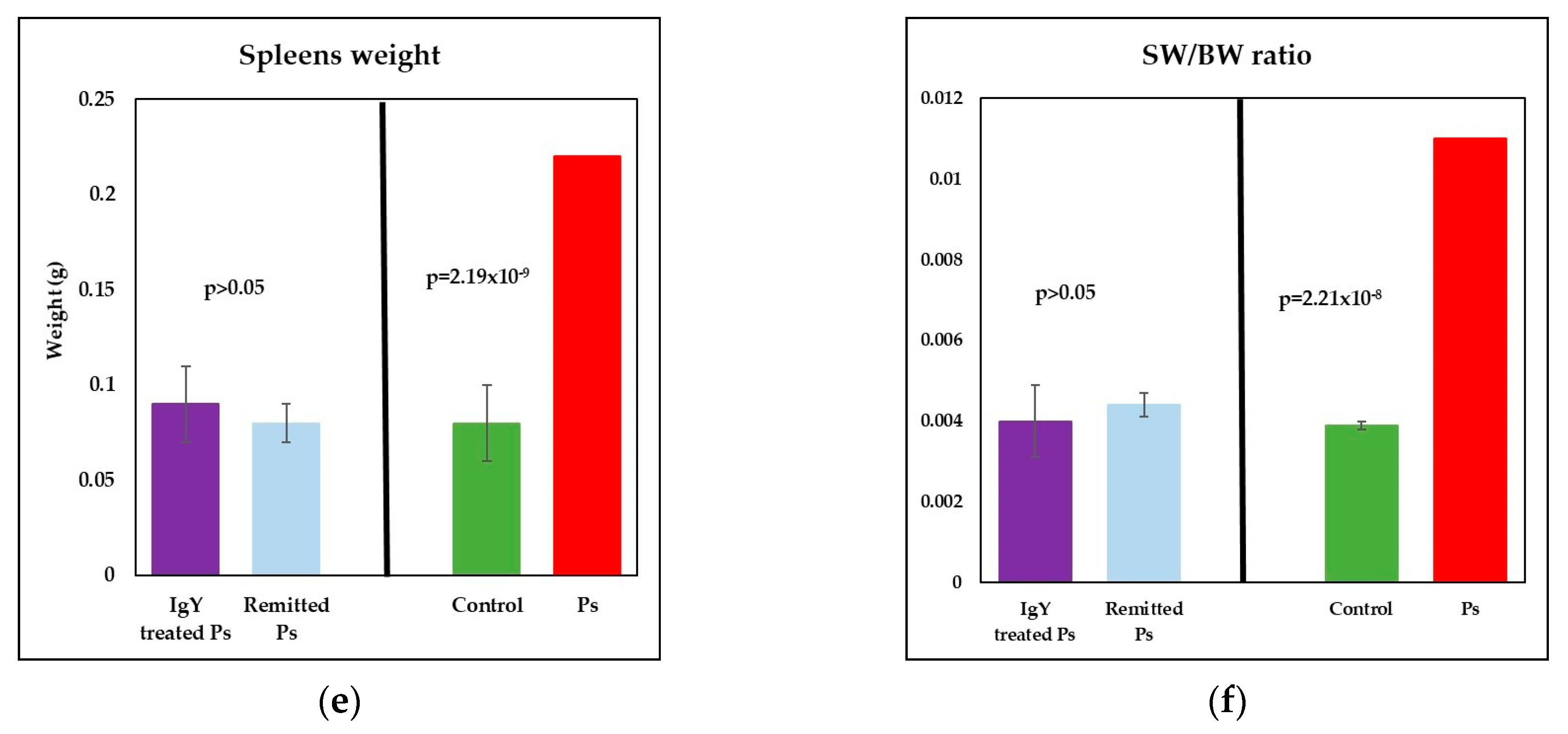
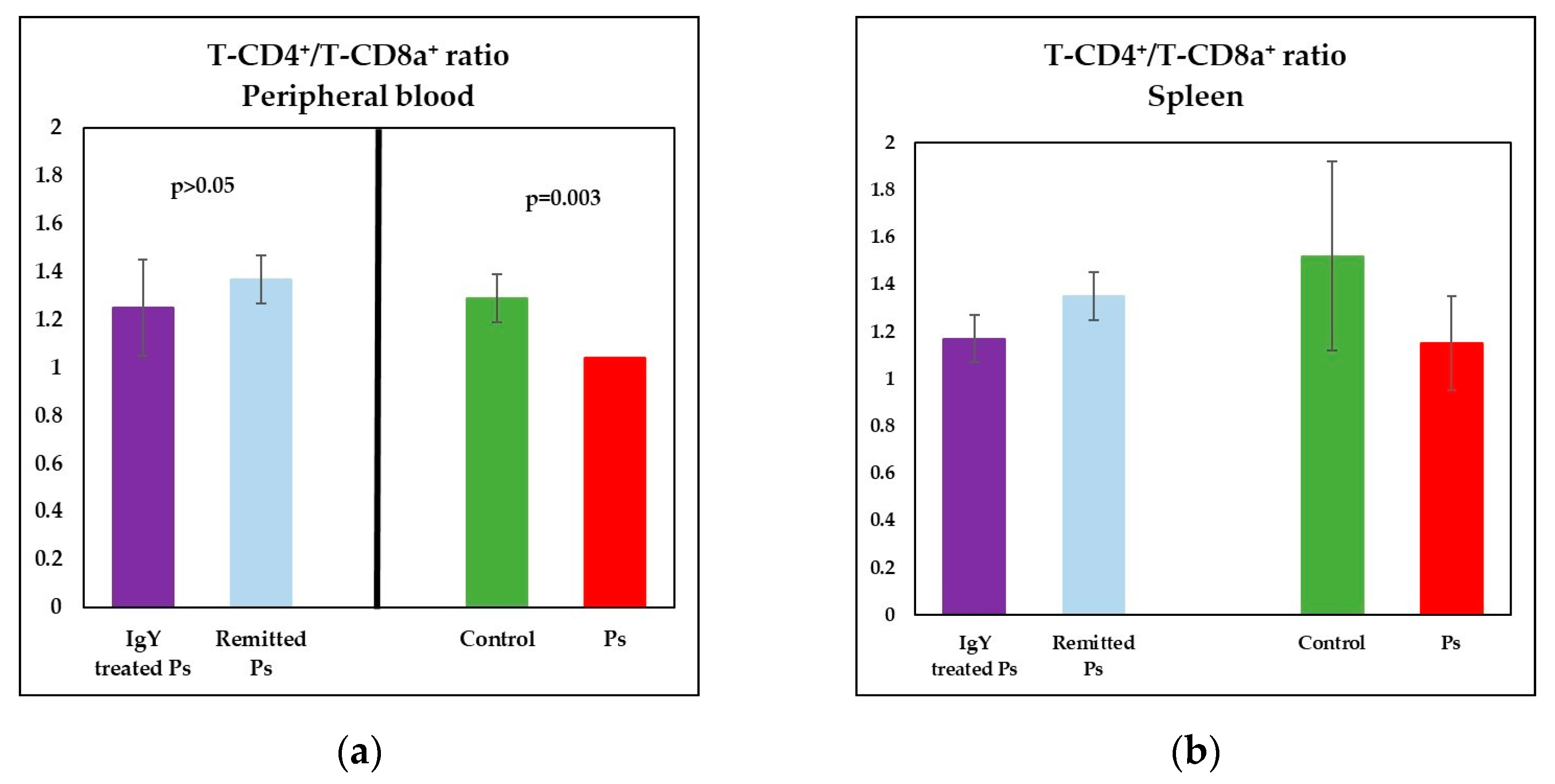
3.3. IgY Treatment-Induced Changes in Lymphocyte Distribution in Peripheral Blood and Spleen Cell Suspensions in Experimental Murine Model of Psoriatic Dermatitis
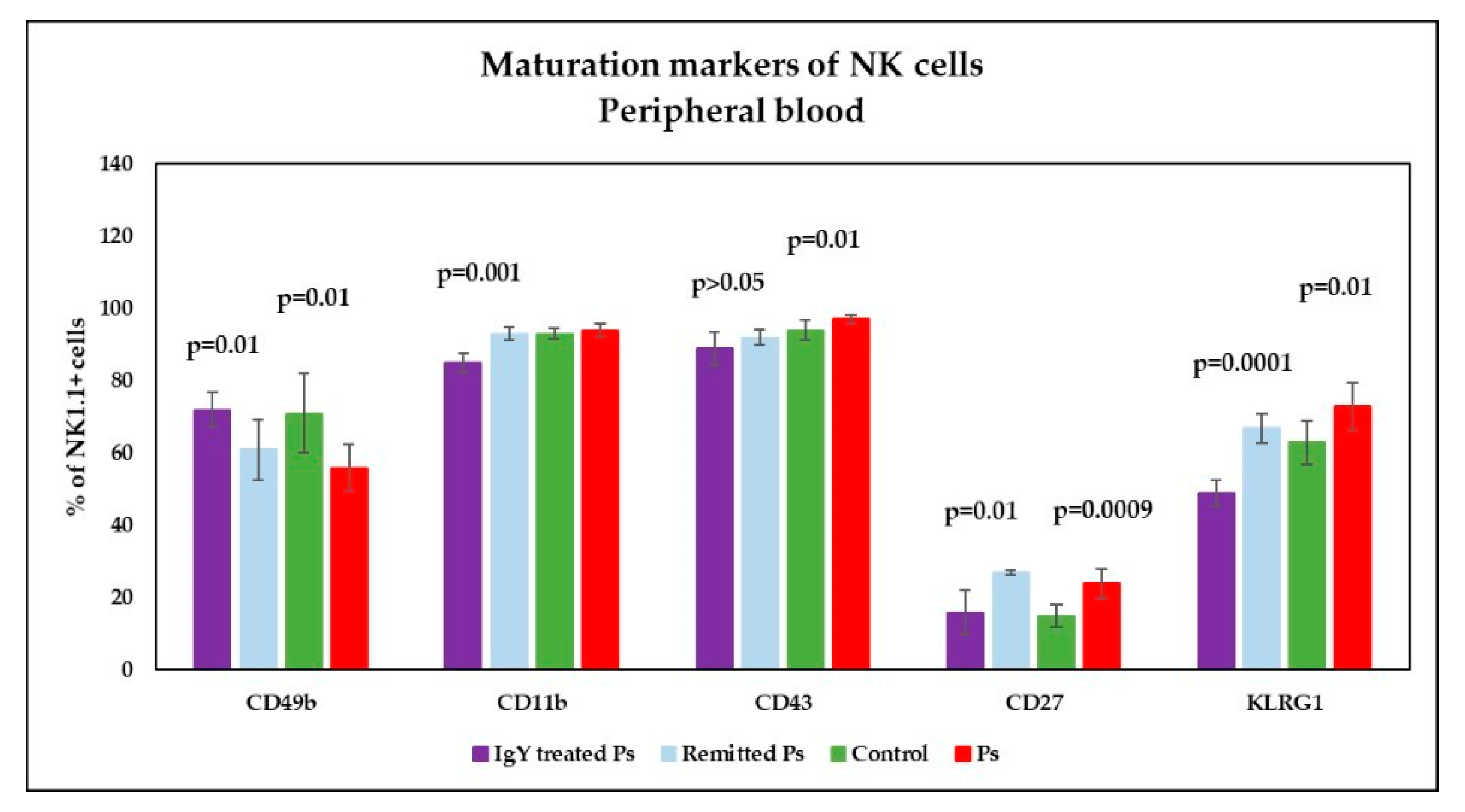
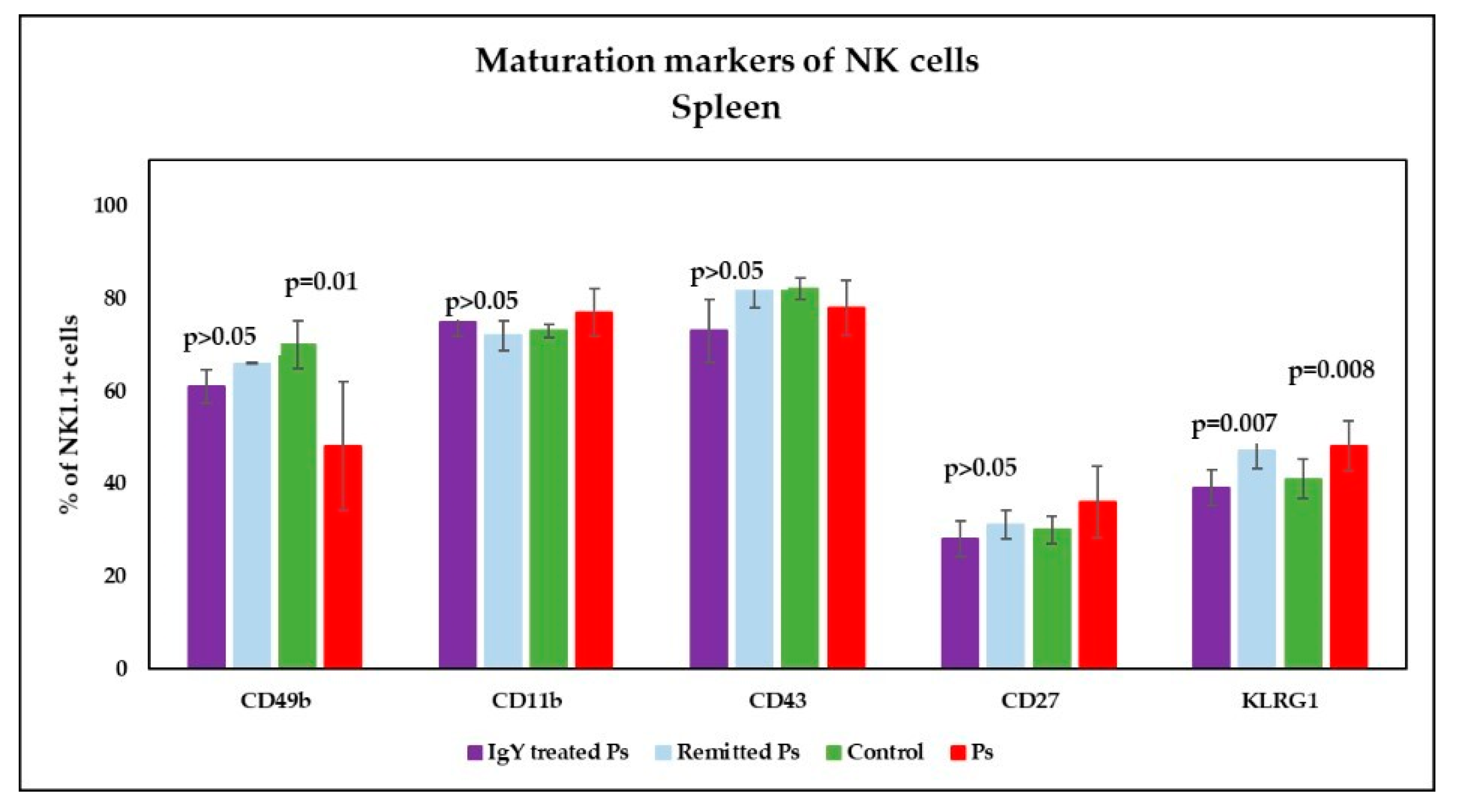
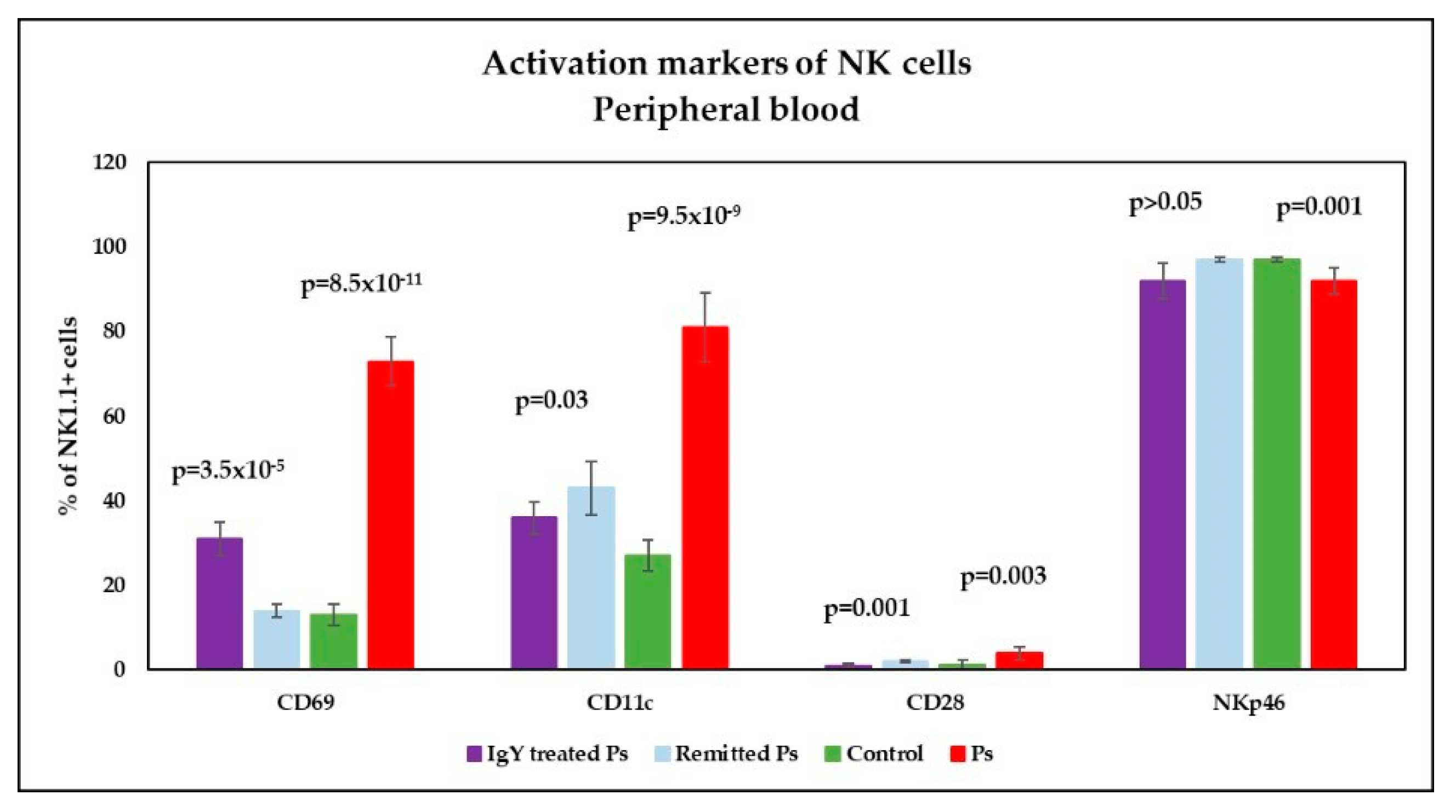
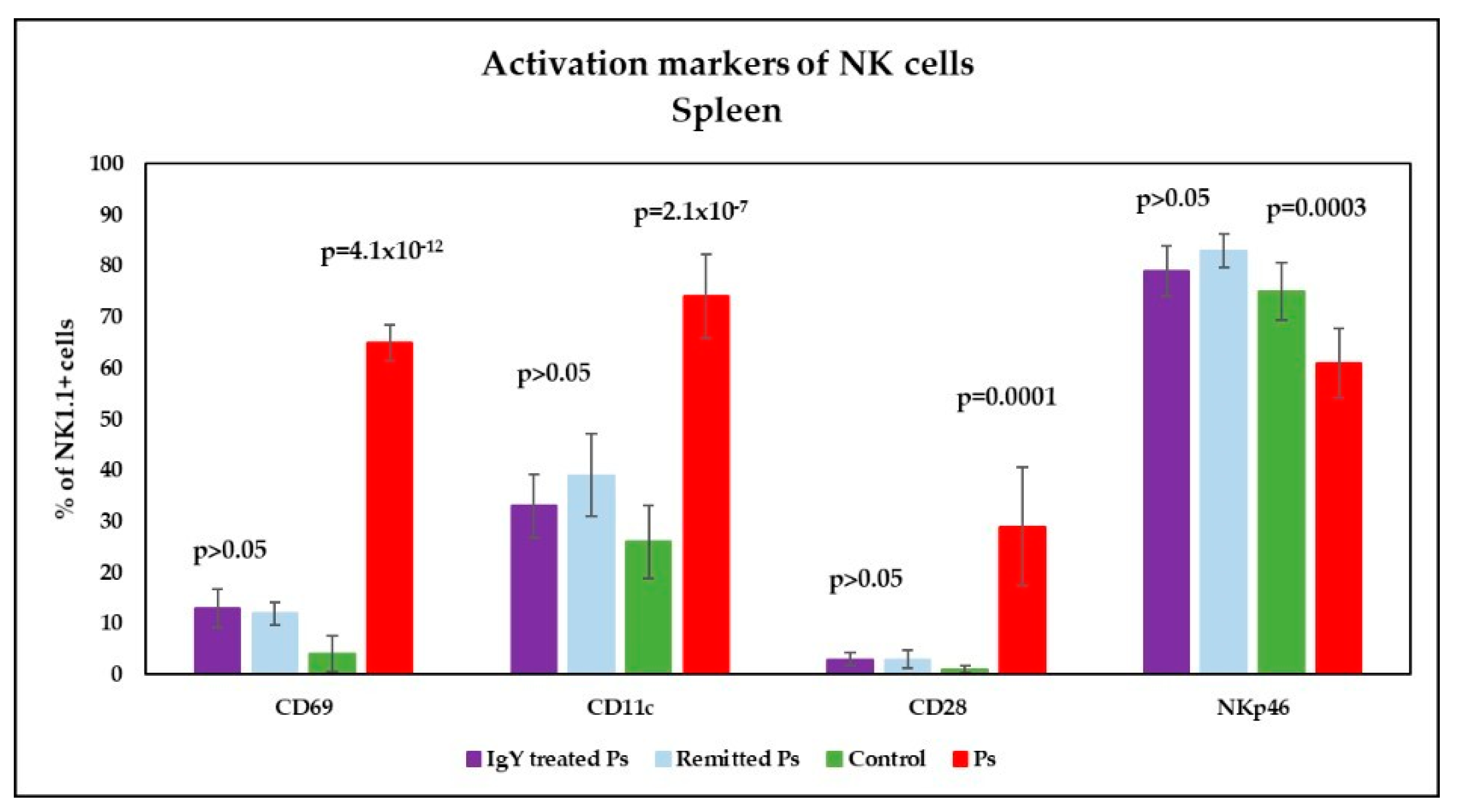
3.4. IgY Treatment-Induced Changes in NK Phenotype in Peripheral Blood and Spleen Cell Suspensions in Experimental Murine Model of Psoriatic Dermatitis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Guo, R.; Zhang, T.; Meng, X.; Lin, Z.; Lin, J.; Gong, Y.; Liu, X.; Yu, Y.; Zhao, G.; Ding, X.; et al. Lymphocyte Mass Cytometry Identifies a CD3−CD4+ Cell Subset with a Potential Role in Psoriasis. JCI Insight 2019, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nestle, F.O.; Kaplan, D.H.; Barker, J. Psoriasis. N. Engl. J. Med. 2009, 361, 496–509. [Google Scholar] [CrossRef] [PubMed]
- Parisi, R.; Symmons, D.P.M.; Griffiths, C.E.M.; Ashcroft, D.M.; Identification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) Project Team. Global Epidemiology of Psoriasis: A Systematic Review of Incidence and Prevalence. J. Investig. Dermatol. 2013, 133, 377–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parisi, R.; Iskandar, I.Y.K.; Kontopantelis, E.; Augustin, M.; Griffiths, C.E.M.; Ashcroft, D.M. National, Regional, and Worldwide Epidemiology of Psoriasis: Systematic Analysis and Modelling Study. BMJ 2020, 369, m1590. [Google Scholar] [CrossRef]
- Wolf, P.; Weger, W.; Patra, V.; Gruber-Wackernagel, A.; Byrne, S.N. Desired Response to Phototherapy vs Photoaggravation in Psoriasis: What Makes the Difference? Exp. Dermatol. 2016, 25, 937–944. [Google Scholar] [CrossRef]
- Fry, L.; Baker, B.S. Triggering Psoriasis: The Role of Infections and Medications. Clin. Dermatol. 2007, 25, 606–615. [Google Scholar] [CrossRef]
- Naldi, L. Psoriasis and Smoking: Links and Risks. Psoriasis 2016, 6, 65–71. [Google Scholar] [CrossRef] [Green Version]
- Wheatley, R.; Brooks, J.; Stumpf, B.; Boh, E. Obesity, Diet, and Inflammation in Psoriasis. J. Psoriasis Psoriatic Arthritis 2017, 2, 97–101. [Google Scholar] [CrossRef]
- Farkas, A.; Kemény, L. Alcohol, Liver, Systemic Inflammation and Skin: A Focus on Patients with Psoriasis. Skin Pharmacol. Physiol. 2013, 26, 119–126. [Google Scholar] [CrossRef]
- Wolk, K.; Mallbris, L.; Larsson, P.; Rosenblad, A.; Vingård, E.; Ståhle, M. Excessive Body Weight and Smoking Associates with a High Risk of Onset of Plaque Psoriasis. Acta Derm.-Venereol. 2009, 89, 492–497. [Google Scholar] [CrossRef]
- Snast, I.; Reiter, O.; Atzmony, L.; Leshem, Y.A.; Hodak, E.; Mimouni, D.; Pavlovsky, L. Psychological Stress and Psoriasis: A Systematic Review and Meta-Analysis. Br. J. Dermatol. 2018, 178, 1044–1055. [Google Scholar] [CrossRef]
- Finlay, A.Y.; Khan, G.K. Dermatology Life Quality Index (DLQI)—A Simple Practical Measure for Routine Clinical Use. Clin. Exp. Dermatol. 1994, 19, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Langley, R.G.; Ellis, C.N. Evaluating Psoriasis with Psoriasis Area and Severity Index, Psoriasis Global Assessment, and Lattice System Physician’s Global Assessment. J. Am. Acad. Dermatol. 2004, 51, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Georgescu, S.-R.; Tampa, M.; Caruntu, C.; Sarbu, M.-I.; Mitran, C.-I.; Mitran, M.-I.; Matei, C.; Constantin, C.; Neagu, M. Advances in Understanding the Immunological Pathways in Psoriasis. Int. J. Mol. Sci. 2019, 20, 739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Surcel, M.; Huica, R.; Constantin, C.; Ursaciuc, C.; Neagu, M. Biomarkers Insights in Psoriasis-Regulatory Cytokines. Curr. Biomark. 2017, 7, 3–11. [Google Scholar] [CrossRef]
- Gall, J.S.; Kalb, R.E. Infliximab for the Treatment of Plaque Psoriasis. Biologics 2008, 2, 115–124. [Google Scholar] [CrossRef] [Green Version]
- Alwawi, E.A.; Mehlis, S.L.; Gordon, K.B. Treating Psoriasis with Adalimumab. Ther. Clin. Risk Manag. 2008, 4, 345–351. [Google Scholar] [CrossRef] [Green Version]
- Blauvelt, A.; Reich, K.; Lebwohl, M.; Burge, D.; Arendt, C.; Peterson, L.; Drew, J.; Rolleri, R.; Gottlieb, A.B. Certolizumab Pegol for the Treatment of Patients with Moderate-to-Severe Chronic Plaque Psoriasis: Pooled Analysis of Week 16 Data from Three Randomized Controlled Trials. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 546–552. [Google Scholar] [CrossRef]
- Farhi, D. Ustekinumab for the Treatment of Psoriasis: Review of Three Multicenter Clinical Trials. Drugs Today 2010, 46, 259–264. [Google Scholar] [CrossRef]
- Nogueira, M.; Torres, T. Guselkumab for the Treatment of Psoriasis—Evidence to Date. Drugs Context 2019, 8, 212594. [Google Scholar] [CrossRef]
- Witjes, H.; Khatri, A.; Diderichsen, P.M.; Mandema, J.; Othman, A.A. Meta-Analyses of Clinical Efficacy of Risankizumab and Adalimumab in Chronic Plaque Psoriasis: Supporting Evidence of Risankizumab Superiority. Clin. Pharmacol. Ther. 2020, 107, 435–442. [Google Scholar] [CrossRef] [Green Version]
- Blauvelt, A.; Sofen, H.; Papp, K.; Gooderham, M.; Tyring, S.; Zhao, Y.; Lowry, S.; Mendelsohn, A.; Parno, J.; Reich, K. Tildrakizumab Efficacy and Impact on Quality of Life up to 52 Weeks in Patients with Moderate-to-Severe Psoriasis: A Pooled Analysis of Two Randomized Controlled Trials. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 2305–2312. [Google Scholar] [CrossRef] [Green Version]
- López-Ferrer, A.; Vilarrasa, E.; Puig, L. Secukinumab (AIN457) for the Treatment of Psoriasis. Expert Rev. Clin. Immunol. 2015, 11, 1177–1188. [Google Scholar] [CrossRef]
- Papp, K.A.; Leonardi, C.L.; Blauvelt, A.; Reich, K.; Korman, N.J.; Ohtsuki, M.; Paul, C.; Ball, S.; Cameron, G.S.; Erickson, J.; et al. Ixekizumab Treatment for Psoriasis: Integrated Efficacy Analysis of Three Double-Blinded, Controlled Studies. Br. J. Dermatol. 2018, 178, 674–681. [Google Scholar] [CrossRef] [Green Version]
- Foulkes, A.C.; Warren, R.B. Brodalumab in Psoriasis: Evidence to Date and Clinical Potential. Drugs Context 2019, 8, 212570. [Google Scholar] [CrossRef] [PubMed]
- Pandya, V.B.; Kumar, S.; Sachchidanand; Sharma, R.; Desai, R.C. Combating Autoimmune Diseases With Retinoic Acid Receptor-Related Orphan Receptor-γ (RORγ or RORc) Inhibitors: Hits and Misses. J. Med. Chem. 2018, 61, 10976–10995. [Google Scholar] [CrossRef] [PubMed]
- Bachelez, H.; Choon, S.-E.; Marrakchi, S.; Burden, A.D.; Tsai, T.-F.; Morita, A.; Turki, H.; Hall, D.B.; Shear, M.; Baum, P.; et al. Inhibition of the Interleukin-36 Pathway for the Treatment of Generalized Pustular Psoriasis. N. Engl. J. Med. 2019, 380, 981–983. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, D.M.; Kanno, Y.; Villarino, A.; Ward, M.; Gadina, M.; O’Shea, J.J. JAK Inhibition as a Therapeutic Strategy for Immune and Inflammatory Diseases. Nat. Rev. Drug Discov. 2017, 16, 843–862. [Google Scholar] [CrossRef] [PubMed]
- Page, K.M.; Suarez-Farinas, M.; Suprun, M.; Zhang, W.; Garcet, S.; Fuentes-Duculan, J.; Li, X.; Scaramozza, M.; Kieras, E.; Banfield, C.; et al. Molecular and Cellular Responses to the TYK2/JAK1 Inhibitor PF-06700841 Reveal Reduction of Skin Inflammation in Plaque Psoriasis. J. Investig. Dermatol. 2020, 140, 1546–1555.e4. [Google Scholar] [CrossRef]
- Zanin-Zhorov, A.; Weiss, J.M.; Trzeciak, A.; Chen, W.; Zhang, J.; Nyuydzefe, M.S.; Arencibia, C.; Polimera, S.; Schueller, O.; Fuentes-Duculan, J.; et al. Cutting Edge: Selective Oral ROCK2 Inhibitor Reduces Clinical Scores in Patients with Psoriasis Vulgaris and Normalizes Skin Pathology via Concurrent Regulation of IL-17 and IL-10. J. Immunol. 2017, 198, 3809–3814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaclavkova, A.; Chimenti, S.; Arenberger, P.; Holló, P.; Sator, P.-G.; Burcklen, M.; Stefani, M.; D’Ambrosio, D. Oral Ponesimod in Patients with Chronic Plaque Psoriasis: A Randomised, Double-Blind, Placebo-Controlled Phase 2 Trial. Lancet 2014, 384, 2036–2045. [Google Scholar] [CrossRef]
- Robbins, K.; Bissonnette, R.; Maeda-Chubachi, T.; Ye, L.; Peppers, J.; Gallagher, K.; Kraus, J.E. Phase 2, Randomized Dose-Finding Study of Tapinarof (GSK2894512 Cream) for the Treatment of Plaque Psoriasis. J. Am. Acad. Dermatol. 2019, 80, 714–721. [Google Scholar] [CrossRef]
- Rahman, S.; Van Nguyen, S.; Icatlo, F.C., Jr.; Umeda, K.; Kodama, Y. Oral Passive IgY-Based Immunotherapeutics. Hum. Vaccines Immunother. 2013, 9, 1039–1048. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torché, A.-M.; Le Dimna, M.; Le Corre, P.; Mesplède, A.; Le Gal, S.; Cariolet, R.; Le Potier, M.-F. Immune Responses after Local Administration of IgY Loaded-PLGA Microspheres in Gut-Associated Lymphoid Tissue in Pigs. Vet. Immunol. Immunopathol. 2006, 109, 209–217. [Google Scholar] [CrossRef]
- Kovacs-Nolan, J.; Mine, Y. Egg Yolk Antibodies for Passive Immunity. Annu. Rev. Food Sci. Technol. 2012, 3, 163–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akita, E.M.; Jang, C.B.; Kitts, D.D.; Nakai, S. Evaluation of Allergenicity of Egg Yolk Immunoglobulin Y and Other Egg Proteins by Passive Cutaneous Anaphylaxis. Food Agric. Immunol. 1999, 11, 191–201. [Google Scholar] [CrossRef] [Green Version]
- Vega, C.G.; Bok, M.; Vlasova, A.N.; Chattha, K.S.; Fernández, F.M.; Wigdorovitz, A.; Parreño, V.G.; Saif, L.J. IgY Antibodies Protect against Human Rotavirus Induced Diarrhea in the Neonatal Gnotobiotic Piglet Disease Model. PLoS ONE 2012, 7, e42788. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Li, X.; Jin, L.; Zhen, Y.; Lu, Y.; Li, S.; You, J.; Wang, L. Application of Chicken Egg Yolk Immunoglobulins in the Control of Terrestrial and Aquatic Animal Diseases: A Review. Biotechnol. Adv. 2011, 29, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Constantin, C.; Neagu, M.; Diana Supeanu, T.; Chiurciu, V.; Spandidos, D.A. IgY—Turning the Page toward Passive Immunization in COVID-19 Infection (Review). Exp. Ther. Med. 2020, 20, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Michael, A.; Meenatchisundaram, S.; Parameswari, G.; Subbraj, T.; Selvakumaran, R.; Ramalingam, S. Chicken Egg Yolk Antibodies (IgY) as an Alternative to Mammalian Antibodies. Indian J. Sci. Technol. 2010, 3, 468–474. [Google Scholar] [CrossRef]
- Yu, M.; Zhang, R.; Ni, P.; Chen, S.; Duan, G. Helicobacter Pylori Infection and Psoriasis: A Systematic Review and Meta-Analysis. Medicina 2019, 55, 645. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Jesús-Gil, C.; Sans-de San Nicolàs, L.; Ruiz-Romeu, E.; Ferran, M.; Soria-Martínez, L.; García-Jiménez, I.; Chiriac, A.; Casanova-Seuma, J.M.; Fernández-Armenteros, J.M.; Owens, S.; et al. Interplay between Humoral and CLA+T Cell Response against Candida albicans in Psoriasis. Int. J. Mol. Sci. 2021, 22, 1519. [Google Scholar] [CrossRef] [PubMed]
- Visser, M.J.E.; Kell, D.B.; Pretorius, E. Bacterial Dysbiosis and Translocation in Psoriasis Vulgaris. Front. Cell Infect. Microbiol. 2019, 9, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Surcel, M.; Huică, R.-I.; Munteanu, A.N.; Isvoranu, G.; Pîrvu, I.R.; Ciotaru, D.; Constantin, C.; Bratu, O.; Căruntu, C.; Neagu, M.; et al. Phenotypic Changes of Lymphocyte Populations in Psoriasiform Dermatitis Animal Model. Exp. Ther. Med. 2019, 17, 1030–1038. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Surcel, M.; Munteanu, A.N.; Huică, R.-I.; Isvoranu, G.; Pîrvu, I.R.; Constantin, C.; Bratu, O.; Căruntu, C.; Zaharescu, I.; Sima, L.; et al. Reinforcing Involvement of NK Cells in Psoriasiform Dermatitis Animal Model. Exp. Ther. Med. 2019, 18, 4956–4966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pătraşcu, I.V.; Chiurciu, V.; Chiurciu, C.; Topilescu, G. Procedure to Obtain and Use Hen Egg Immunoglobulins (IgY). RO129645 A0, 2014 00156, 30 July 2014. [Google Scholar]
- Pătraşcu, I.V.; Chiurciu, V.; Chiurciu, C.; Topilescu, G. Method for Immunobiological Assay of Chicken Immunoglobulins Specific Activity. RO129677 A0, 2014 00179, 30 July 2014. [Google Scholar]
- van der Fits, L.; Mourits, S.; Voerman, J.S.A.; Kant, M.; Boon, L.; Laman, J.D.; Cornelissen, F.; Mus, A.-M.; Florencia, E.; Prens, E.P.; et al. Imiquimod-Induced Psoriasis-like Skin Inflammation in Mice Is Mediated via the IL-23/IL-17 Axis. J. Immunol. 2009, 182, 5836–5845. [Google Scholar] [CrossRef]
- Chiurciu, C.; Chiurciu, V.; Sima, L.; Mihai, I.; Patrascu, I.V. Production and Use of Personalized (Ovopatch) Hyperimmune Egg in the Treatment of Psoriasis. RO 130965 A0, 2015 00735, 30 March 2016. [Google Scholar]
- Chen, L.; Li, J.; Zhu, W.; Kuang, Y.; Liu, T.; Zhang, W.; Chen, X.; Peng, C. Skin and Gut Microbiome in Psoriasis: Gaining Insight Into the Pathophysiology of It and Finding Novel Therapeutic Strategies. Front. Microbiol. 2020, 11, 589726. [Google Scholar] [CrossRef]
- Takemoto, A.; Cho, O.; Morohoshi, Y.; Sugita, T.; Muto, M. Molecular Characterization of the Skin Fungal Microbiome in Patients with Psoriasis. J. Dermatol. 2015, 42, 166–170. [Google Scholar] [CrossRef]
- Salem, I.; Ramser, A.; Isham, N.; Ghannoum, M.A. The Gut Microbiome as a Major Regulator of the Gut-Skin Axis. Front. Microbiol. 2018, 9, 1459. [Google Scholar] [CrossRef] [Green Version]
- Haines Ely, P. Is psoriasis a bowel disease? Successful treatment with bile acids and bioflavonoids suggests it is. Clin. Dermatol. 2018, 36, 376–389. [Google Scholar] [CrossRef]
- Bocheńska, K.; Smolińska, E.; Moskot, M.; Jakóbkiewicz-Banecka, J.; Gabig-Cimińska, M. Models in the Research Process of Psoriasis. Int. J. Mol. Sci. 2017, 18, 2514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hawkes, J.E.; Adalsteinsson, J.A.; Gudjonsson, J.E.; Ward, N.L. Research Techniques Made Simple: Murine Models of Human Psoriasis. J. Investig. Dermatol. 2018, 138, e1–e8. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Ni, G.; Wang, T.; Mounsey, K.; Cavezza, S.; Pan, X.; Liu, X. Genital Warts Treatment: Beyond Imiquimod. Hum. Vaccines Immunother. 2018, 14, 1815–1819. [Google Scholar] [CrossRef] [PubMed]
- Jabeen, M.; Boisgard, A.-S.; Danoy, A.; El Kholti, N.; Salvi, J.-P.; Boulieu, R.; Fromy, B.; Verrier, B.; Lamrayah, M. Advanced Characterization of Imiquimod-Induced Psoriasis-Like Mouse Model. Pharmaceutics 2020, 12, 789. [Google Scholar] [CrossRef]
- Alecu, M.; Ursaciuc, C.; Surcel, M.; Coman, G.; Ciotaru, D.; Dobre, M. CD28 T-cell costimulatory molecule expression in pemphigus vulgaris. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 288–291. [Google Scholar] [CrossRef]
- Prietl, B.; Treiber, G.; Mader, J.K.; Hoeller, E.; Wolf, M.; Pilz, S.; Graninger, W.B.; Obermayer-Pietsch, B.M.; Pieber, T.R. High-Dose Cholecalciferol Supplementation Significantly Increases Peripheral CD4+ Tregs in Healthy Adults without Negatively Affecting the Frequency of Other Immune Cells. Eur. J. Nutr. 2014, 53, 751–759. [Google Scholar] [CrossRef]
- Shin, S.-H.; Kim, H.-Y.; Yoon, H.-S.; Park, W.-J.; Adams, D.R.; Pyne, N.J.; Pyne, S.; Park, J.-W. A Novel Selective Sphingosine Kinase 2 Inhibitor, HWG-35D, Ameliorates the Severity of Imiquimod-Induced Psoriasis Model by Blocking Th17 Differentiation of Naïve CD4 T Lymphocytes. Int. J. Mol. Sci. 2020, 21, 8371. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Yan, Y.; Liu, H.; Qiu, F.; Liang, C.-L.; Zhang, Q.; Huang, R.-Y.; Han, L.; Lu, C.; Dai, Z. Dihydroartemisinin Ameliorates Psoriatic Skin Inflammation and Its Relapse by Diminishing CD8+ T-Cell Memory in Wild-Type and Humanized Mice. Theranostics 2020, 10, 10466–10482. [Google Scholar] [CrossRef]
- Kahlert, K.; Grän, F.; Muhammad, K.; Benoit, S.; Serfling, E.; Goebeler, M.; Kerstan, A. Aberrant B-Cell Subsets and Immunoglobulin Levels in Patients with Moderate-to-Severe Psoriasis. Acta Derm.-Venereol. 2019, 99, 226–227. [Google Scholar] [CrossRef] [Green Version]
- Alrefai, H.; Muhammad, K.; Rudolf, R.; Pham, D.A.T.; Klein-Hessling, S.; Patra, A.K.; Avots, A.; Bukur, V.; Sahin, U.; Tenzer, S.; et al. NFATc1 Supports Imiquimod-Induced Skin Inflammation by Suppressing IL-10 Synthesis in B Cells. Nat. Commun. 2016, 7, 11724. [Google Scholar] [CrossRef] [Green Version]
- Grän, F.; Kerstan, A.; Serfling, E.; Goebeler, M.; Muhammad, K. Current Developments in the Immunology of Psoriasis. Yale J. Biol. Med. 2020, 93, 97–110. [Google Scholar]
- Polese, B.; Zhang, H.; Thurairajah, B.; King, I.L. Innate Lymphocytes in Psoriasis. Front. Immunol. 2020, 11, 242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kucuksezer, U.C.; Aktas Cetin, E.; Esen, F.; Tahrali, I.; Akdeniz, N.; Gelmez, M.Y.; Deniz, G. The Role of Natural Killer Cells in Autoimmune Diseases. Front. Immunol. 2021, 12, 79. [Google Scholar] [CrossRef] [PubMed]
- Luci, C.; Gaudy-Marqueste, C.; Rouzaire, P.; Audonnet, S.; Cognet, C.; Hennino, A.; Nicolas, J.-F.; Grob, J.-J.; Tomasello, E. Peripheral Natural Killer Cells Exhibit Qualitative and Quantitative Changes in Patients with Psoriasis and Atopic Dermatitis. Br. J. Dermatol. 2012, 166, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Dunphy, S.E.; Sweeney, C.M.; Kelly, G.; Tobin, A.M.; Kirby, B.; Gardiner, C.M. Natural Killer Cells from Psoriasis Vulgaris Patients Have Reduced Levels of Cytotoxicity Associated Degranulation and Cytokine Production. Clin. Immunol. 2017, 177, 43–49. [Google Scholar] [CrossRef]
- Hadad, U.; Thauland, T.; Butte, M.; Porgador, A.; Martinez, O.; Krams, S. NKp46 Clusters at the NK Cell Immune Synapse Regulate Specific Effector Functions. In Proceedings of the 2015 American Transplant Congress, Philadelphia, PA, USA, 2–6 May 2015. [Google Scholar]
- Abel, A.M.; Yang, C.; Thakar, M.S.; Malarkannan, S. Natural Killer Cells: Development, Maturation, and Clinical Utilization. Front. Immunol. 2018, 9, 1869. [Google Scholar] [CrossRef] [Green Version]
- Ramírez-Ramírez, D.; Vadillo, E.; Arriaga-Pizano, L.A.; Mayani, H.; Estrada-Parra, S.; Velasco-Velázquez, M.A.; Pérez-Tapia, S.M.; Pelayo, R. Early Differentiation of Human CD11c+NK Cells with Γδ T Cell Activation Properties Is Promoted by Dialyzable Leukocyte Extracts. J. Immunol. Res. 2016, 2016, 4097642. [Google Scholar] [CrossRef] [Green Version]
- Hugenholtz, F.; de Vos, W.M. Mouse models for human intestinal microbiota research: A critical evaluation. Cell. Mol. Life Sci. 2018, 75, 149–160. [Google Scholar] [CrossRef] [Green Version]
- Xiao, L.; Feng, Q.; Liang, S.; Sonne, S.B.; Xia, Z.; Qiu, X.; Li, X.; Long, H.; Zhang, J.; Zhang, D.; et al. A catalog of the mouse gut metagenome. Nat. Biotechnol. 2015, 33, 1103–1108. [Google Scholar] [CrossRef]
- Wang, J.; Lang, T.; Shen, J.; Dai, J.; Tian, L.; Wang, X. Core Gut Bacteria Analysis of Healthy Mice. Front. Microbiol. 2019, 10, 887. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Surcel, M.; Munteanu, A.; Isvoranu, G.; Ibram, A.; Caruntu, C.; Constantin, C.; Neagu, M. Unconventional Therapy with IgY in a Psoriatic Mouse Model Targeting Gut Microbiome. J. Pers. Med. 2021, 11, 841. https://doi.org/10.3390/jpm11090841
Surcel M, Munteanu A, Isvoranu G, Ibram A, Caruntu C, Constantin C, Neagu M. Unconventional Therapy with IgY in a Psoriatic Mouse Model Targeting Gut Microbiome. Journal of Personalized Medicine. 2021; 11(9):841. https://doi.org/10.3390/jpm11090841
Chicago/Turabian StyleSurcel, Mihaela, Adriana Munteanu, Gheorghita Isvoranu, Alef Ibram, Constantin Caruntu, Carolina Constantin, and Monica Neagu. 2021. "Unconventional Therapy with IgY in a Psoriatic Mouse Model Targeting Gut Microbiome" Journal of Personalized Medicine 11, no. 9: 841. https://doi.org/10.3390/jpm11090841
APA StyleSurcel, M., Munteanu, A., Isvoranu, G., Ibram, A., Caruntu, C., Constantin, C., & Neagu, M. (2021). Unconventional Therapy with IgY in a Psoriatic Mouse Model Targeting Gut Microbiome. Journal of Personalized Medicine, 11(9), 841. https://doi.org/10.3390/jpm11090841








