Pain Improvement in Parkinson’s Disease Patients Treated with Safinamide: Results from the SAFINONMOTOR Study
Abstract
:1. Introduction
2. Material and Methods
2.1. Data Analysis
2.2. Standard Protocol Approvals, Registrations, and Patient Consents
2.3. Data Availability
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Financial Disclosures
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Bloem, B.R.; Okun, M.S.; Klein, C. Parkinson’s disease. Lancet 2021, 397, 2284–2303. [Google Scholar] [CrossRef]
- Fil, A.; Cano-de-la-Cuerda, R.; Muñoz-Hellín, E.; Vela, L.; Ramiro-González, M.; Fernández-de-Las-Peñas, C. Pain in Parkinson disease: A review of the literature. Parkinsonism Relat. Disord. 2013, 19, 285–294; discussion 285. [Google Scholar] [CrossRef] [Green Version]
- Ha, A.D.; Jankovic, J. Pain in Parkinson’s disease. Mov. Disord. 2012, 27, 485–491. [Google Scholar] [CrossRef]
- Beiske, A.G.; Loge, J.H.; Rønningen, A.; Svensson, E. Pain in Parkinson’s disease: Prevalence and characteristics. Pain 2009, 141, 173–177. [Google Scholar] [CrossRef]
- Ford, B. Pain in Parkinson’s disease. Mov. Disord. 2010, 25 (Suppl. 1), S98–S103. [Google Scholar] [CrossRef] [PubMed]
- Rukavina, K.; Cummins, T.M.; Chaudhuri, K.R.; Bannister, K. Pain in Parkinson’s disease: Mechanism-based treatment strategies. Curr. Opin. Support. Palliat. Care 2021, 15, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Boccella, S.; Marabese, I.; Guida, F.; Luongo, L.; Maione, S.; Palazzo, E. The Modulation of Pain by Metabotropic Glutamate Receptors 7 and 8 in the Dorsal Striatum. Curr. Neuropharmacol. 2020, 18, 34–50. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, M.; Rosa, M.M.; Abreu, D.; Ferreira, J.J. Clinical pharmacology review of safinamide for the treatment of Parkinson’s disease. Neurodegener. Dis. Manag. 2015, 5, 481–496. [Google Scholar] [CrossRef]
- Borgohain, R.; Szasz, J.; Stanzione, P.; Meshram, C.; Bhatt, M.; Chirilineau, D.; Stocchi, F.; Lucini, V.; Giuliani, R.; Forrest, E.; et al. Study 016 Investigators. Randomized trial of safinamide add-on to levodopa in Parkinson’s disease with motor fluctuations. Mov. Disord. 2014, 29, 229–237. [Google Scholar] [CrossRef]
- Borgohain, R.; Szasz, J.; Stanzione, P.; Meshram, C.; Bhatt, M.H.; Chirilineau, D.; Stocchi, F.; Lucini, V.; Giuliani, R.; Forrest, E.; et al. Study 018 Investigators. Mov. Disord. 2014, 29, 1273–1280. [Google Scholar] [CrossRef]
- Bianchi, M.L.E.; Riboldazzi, G.; Mauri, M.; Versino, M. Efficacy of safinamide on non-motor symptoms in a cohort of patients affected by idiopathic Parkinson’s disease. Neurol. Sci. 2019, 40, 275–279. [Google Scholar] [CrossRef]
- Santos García, D.; Labandeira Guerra, C.; Yáñez Baña, R.; Cimas Hernando, M.I.; Cabo López, I.; Paz Gonález, J.M.; Alonso Losada, M.G.; González Palmás, M.J.; Martínez Miró, C. Safinamide Improves Non-Motor Symptoms Burden in Parkinson´s Disease: An Open-Label Prospective Study. Brain Sci. 2021, 11, 316. [Google Scholar] [CrossRef]
- Liguori, C.; Stefani, A.; Ruffini, R.; Mercuri, N.B.; Pierantozzi, M. Safinamide effect on sleep disturbances and daytime sleepiness in motor fluctuating Parkinson’s disease patients: A validated questionnaires-controlled study. Park. Relat. Disord. 2018, 57, 80–81. [Google Scholar] [CrossRef] [PubMed]
- Peña, E.; Borrué, C.; Mata, M.; Martínez-Castrillo, J.C.; Alonso-Canovas, A.; Chico, J.L.; López-Manzanares, L.; Llanero, M.; Herreros-Rodríguez, J.; Esquivel, A.; et al. Impact of SAfinamide on Depressive Symptoms in Parkinson’s Disease Patients (SADness-PD Study): A Multicenter Retrospective Study. Brain Sci. 2021, 11, 232. [Google Scholar] [CrossRef]
- Gómez-López, A.; Sánchez-Sánchez, A.; Natera-Villalba, E.; Ros-Castelló, V.; Beltrán-Corbellini, Á.; Fanjul-Arbós, S.; Moreno, I.P.; Moreno, J.L.L.-S.; Castrillo, J.C.M.; Alonso-Canovas, A. SURINPARK: Safinamide for Urinary Symptoms in Parkinson’s Disease. Brain Sci. 2021, 11, 57. [Google Scholar] [CrossRef] [PubMed]
- Geroin, C.; Di Vico, I.A.; Squintani, G.; Segatti, A.; Bovi, T.; Tinazzi, M. Effects of safinamide on pain in Parkinson’s disease with motor fluctuations: An exploratory study. J. Neural. Transm. (Vienna) 2020, 127, 1143–1152. [Google Scholar] [CrossRef] [PubMed]
- Cattaneo, C.; Barone, P.; Bonizzoni, E.; Sardina, M. Effects of Safinamide on Pain in Fluctuating Parkinson’s Disease Patients: A Post-Hoc Analysis. J. Parkinsons Dis. 2017, 7, 95–101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cattaneo, C.; Kulisevsky, J.; Tubazio, V.; Castellani, P. Long-term Efficacy of Safinamide on Parkinson’s Disease Chronic Pain. Adv. Ther. 2018, 35, 515–522. [Google Scholar] [CrossRef] [Green Version]
- Qureshi, A.R.; Rana, A.Q.; Malik, S.H.; Rizvi, S.F.H.; Akhter, S.; Vannabouathong, C.; Sarfraz, Z.; Rana, R. Comprehensive Examination of Therapies for Pain in Parkinson’s Disease: A Systematic Review and Meta-Analysis. Neuroepidemiology 2018, 51, 190–206. [Google Scholar] [CrossRef]
- Daniel, S.E.; Lees, A.J. Parkinson’s Disease Society Brain Bank, London: Overview and research. J. Neural. Transm. Suppl. 1993, 39, 165–172. [Google Scholar]
- Dubois, B.; Burn, D.; Goetz, C.; Aarsland, D.; Brown, R.; Broe, G.A.; Dickson, D.W.; Duyckaerts, C.; Cummings, J.; Gauthier, S.; et al. Diagnostic procedures for Parkinson’s disease dementia: Recommendations from the movement disorder society task force. Mov. Disord. 2007, 22, 2314–2324. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, K.R.; Rizos, A.; Trenkwalder, C.; Rascol, O.; Pal, S.; Martino, D.; Carroll, C.; Paviour, D.; Falup-Pecurariu, C.; Kessel, B.; et al. King´s Parkinson´s disease pain scale, the first scale for pain in PD: An international validation. Mov. Disord. 2015, 30, 1623–1631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burckhardt, C.S.; Jones, K.D. Adult measures of pain: The McGill Pain Questionnaire (MPQ), Rheumatoid Arthritis Pain Scale (RAPS), Short Form McGill Pain Questionnaire (SF-MPQ), Verbal Descriptive Scale (VDS), Visual Analog Scale (VAS), and West Haven-Yale Multidisciplinary Pain Inventory (WHYMPI). Arthritis Rheum. 2003, 49, 96–104. [Google Scholar]
- Schade, S.; Mollenhauer, B.; Trenkwalder, C. Levodopa Equivalent Dose Conversion Factors: An Updated Proposal Including Opicapone and Safinamide. Mov. Disord. Clin. Pr. 2020, 7, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Valkovic, P.; Minar, M.; Singliarova, H.; Harsany, J.; Hanakova, M.; Martinkova, J.; Benetin, J. Pain in Parkinson’s Disease: A Cross-Sectional Study of Its Prevalence, Types, and Relationship to Depression and Quality of Life. PLoS ONE 2015, 10, e0136541. [Google Scholar] [CrossRef]
- Wasner, G.; Deuschl, G. Pains in Parkinson disease—Many syndromes under one umbrella. Nat. Rev. Neurol. 2012, 8, 284–294. [Google Scholar] [CrossRef]
- Chaudhuri, K.R.; Schapira, A.H. Non-motor symptoms of Parkinson’s disease: Dopaminergic pathophysiology and treatment. Lancet Neurol. 2009, 8, 464–474. [Google Scholar] [CrossRef]
- Barone, P.; Amboni, M.; Vitale, C.; Bonavita, V. Treatment of nocturnal disturbances and excessive daytime sleepiness in Parkinson’s disease. Neurology 2004, 63 (Suppl. 3), S35–S38. [Google Scholar] [CrossRef] [PubMed]
- Santos-García, D.; Fonticoba, T.D.D.; Castro, E.S.; Díaz, A.A.; McAfee, D.; Catalán, M.J.; Alonso-Frech, F.; Villanueva, C.; Jesús, S.; Mir, P.; et al. COPPADIS Study Group. Non-motor symptom burden is strongly correlated to motor complications in patients with Parkinson’s disease. Eur. J. Neurol. 2020, 27, 1210–1223. [Google Scholar] [CrossRef] [PubMed]
- LeWitt, P.A.; Chaudhuri, K.R. Unmet needs in Parkinson disease: Motor and non-motor. Parkinsonism Relat. Disord. 2020, 80 (Suppl. 1), S7–S12. [Google Scholar] [CrossRef]
- Ehrt, U.; Larsen, J.P.; Aarsland, D. Pain and its relationship to depression in Parkinson disease. Am. J. Geriatr. Psychiatry 2009, 17, 269–275. [Google Scholar] [CrossRef]
- Berardelli, A.; Conte, A.; Fabbrini, G.; Bologna, M.; Latorre, A.; Rocchi, L.; Suppa, A. Pathophysiology of pain and fatigue in Parkinson’s disease. Parkinsonism Relat. Disord. 2012, 18 (Suppl. 1), S226–S228. [Google Scholar] [CrossRef]
- Gerdelat-Mas, A.; Simonetta-Moreau, M.; Thalamas, C.; Ory-Magne, F.; Slaoui, T.; Rascol, O.; Brefel-Courbon, C. Levodopa raises objective pain threshold in Parkinson’s disease: A RIII reflex study. J. Neurol. Neurosurg. Psychiatry 2007, 78, 1140–1142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Defazio, G.; Berardelli, A.; Fabbrini, G.; Martino, D.; Fincati, E.; Fiaschi, A.; Moretto, G.; Abbruzzese, G.; Marchese, R.; Bonuccelli, U.; et al. Pain as a nonmotor symptom of Parkinson disease: Evidence from a case-control study. Arch. Neurol. 2008, 65, 1191–1194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chudler, E.H.; Dong, W.K. The role of the basal ganglia in nociception and pain. Pain 1995, 60, 3–38. [Google Scholar] [CrossRef]
- Kim, H.-J.; Paek, S.H.; Kim, J.-Y.; Lee, J.-Y.; Lim, Y.H.; Kim, M.-R.; Kim, D.G.; Jeon, B.S. Chronic subthalamic deep brain stimulation improves pain in Parkinson disease. J. Neurol. 2008, 255, 1889–1894. [Google Scholar] [CrossRef] [PubMed]
- Stocchi, F.; Vacca, L.; Grassini, P.; Tomino, C.; Caminiti, G.; Casali, M.; D’Antoni, V.; Volterrani, M.; Torti, M. Overnight switch from rasagiline to safinamide in Parkinson’s disease patients with motor fluctuations: A tolerability and safety study. Eur. J. Neurol. 2021, 28, 349–354. [Google Scholar] [CrossRef]
- Stocchi, F.; Torti, M. Adjuvant therapies for Parkinson’s disease: Critical evaluation of safinamide. Drug Des. Devel 2016, 10, 609–618. [Google Scholar] [CrossRef] [Green Version]
- Santos-García, D.; de la Fuente-Fernández, R. Impact of non-motor symptoms on health-related and perceived quality of life in Parkinson’s disease. J. Neurol. Sci. 2013, 332, 136–140. [Google Scholar] [CrossRef]
- Santos-García, D.; Oreiro, M.; Pérez, P.; Fanjul, G.; González, J.M.P.; Painceiras, M.J.F.; Bartolomé, C.C.; Aymerich, L.V.; Sancho, C.G.; Rodrigo, M.D.M.C. Impact of Coronavirus Disease 2019 Pandemic on Parkinson’s Disease: A Cross-Sectional Survey of 568 Spanish Patients. Mov. Disord. 2020, 35, 1712–1716. [Google Scholar] [CrossRef]
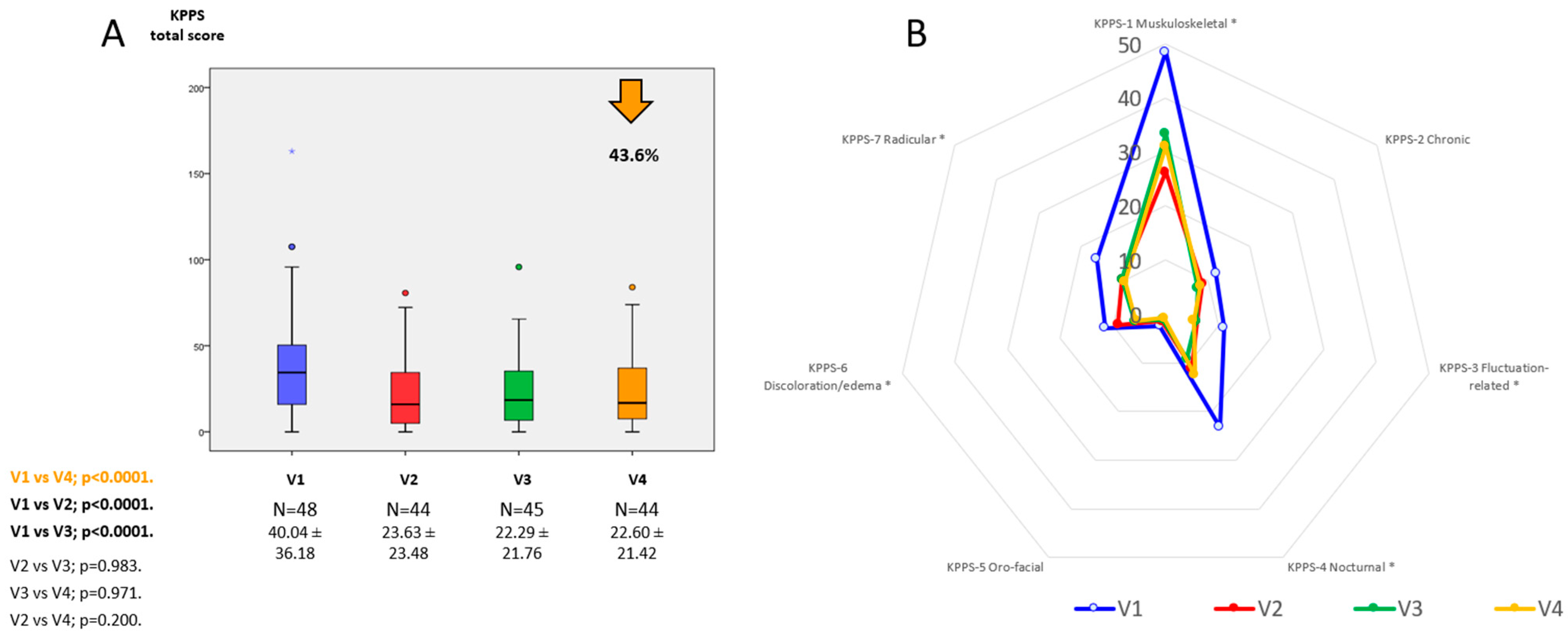

| V1 | V2 | V3 | V4 | p a | p b | p c | p d | |
|---|---|---|---|---|---|---|---|---|
| KPPS | 40.04 ± 36.18 | 23.63 ± 23.48 | 22.29 ± 21.76 | 22.60 ± 21.42 | <0.0001 | <0.0001 | <0.0001 | 0.2 |
| - Musculo-skeletal pain | 48.44 ± 39.74 | 26.32 ± 28.97 | 33.33 ± 31.38 | 31.06 ± 30.15 | 0.009 | 0.006 | <0.0001 | 0.009 |
| - Chronic pain | 11.89 ± 21.14 | 8.8 ± 13.39 | 7.68 ± 15.04 | 8.24 ± 15.24 | 0.636 | 0.326 | 0.406 | 0.872 |
| - Fluctuation-related pain | 11.11 ± 15.08 | 5.99 ± 12.04 | 5.86 ± 13.35 | 5.37 ± 10.74 | 0.02 | 0.04 | 0.03 | 0.02 |
| - Nocturnal pain | 23.18 ± 27.30 | 11.17 ± 17.62 | 9.25 ± 14.91 | 12.50 ± 26.17 | 0.001 | 0.002 | 0.002 | 0.087 |
| - Oro-facial pain | 2.49 ± 9.58 | 1.51 ± 6.94 | 1.23 ± 3.86 | 0.82 ± 3.20 | 1 | 0.673 | 0.673 | 1 |
| - Discoloration, edema/swelling | 11.46 ± 17.99 | 9.99 ± 15.25 | 5.83 ± 11.56 | 5.68 ± 11.65 | 0.009 | 0.031 | 0.454 | 0.009 |
| - Radicular pain | 16.32 ± 30.60 | 10.22 ± 23.49 | 10.37 ± 22.76 | 9.66 ± 22.15 | 0.048 | 0.192 | 0.137 | 0.677 |
| VAS-PAIN | 4.61 ± 3.22 | 3.64 ± 2.73 | 3.67 ± 2.52 | 3.67 ± 2.69 | 0.071 | 0.047 | 0.078 | 0.071 |
| PDQ-39SI Pain and discomfort | 44.56 ± 27.35 | 36.87 ± 25.64 | 29.62 ± 22.58 | 33.33 ± 19.93 | 0.018 | <0.0001 | 0.018 | 0.537 |
| Dose of safinamide (mg/day) | N. A. | 53.84 ± 13.49 | 96.15 ± 13.49 | 98.72 ± 8.00 |
| KPPS | KPPS-1 | KPPS-2 | KPPS-3 | KPPS-4 | KPPS-5 | KPPS-6 | KPPS-7 | |
|---|---|---|---|---|---|---|---|---|
| Total Score | Musculoskel. | Chronic | Fluctuation-Related | Nocturnal | Oro-Facial | Disc./Edema | Radicular | |
| MOTOR ASSESSMENT | ||||||||
| UPDRS-III-ON | 0.255 | 0.182 | 0.245 | 0.165 | −0.092 | 0.126 | 0.216 | 0.147 |
| UPDRS-IV | 0.031 | −0.010 | 0.377 *** | 0.385 *** | 0.370 *** | 0.11 | −0.124 | −0.217 |
| FOGQ | 0.106 | −0.061 | 0.21 | 0.343 *** | −0.135 | 0.172 | 0.094 | −0.280 |
| NON MOTOR ASSESSMENT | ||||||||
| NMSS total score | 0.577 * | 0.188 | 0.604 * | 0.520 * | 0.283 | 0.281 | 0.399 *** | −0.129 |
| ESS | 0.118 | 0.026 | −0.045 | 0.321 *** | 0.01 | −0.110 | 0.095 | −0.008 |
| PSQI | 0.062 | −0.201 | 0.187 | 0.238 | 0.065 | −0.034 | 0.113 | −0.270 |
| BDI-II | 0.05 | 0.109 | 0.478 * | 0.354 *** | 0.29 | 0.242 | 0.345 *** | 0.098 |
| VAS-PAIN | 0.438 * | 0.245 | 0.191 | 0.176 | 0.407 *** | 0.285 | 0.286 | 0.111 |
| VAFS-Physical | 0.339 *** | 0.1 | 0.182 | 0.089 | 0.489 ** | 0.272 | 0.104 | 0.024 |
| VAFS-Mental | 0.206 | −0.085 | 0.191 | 0.181 | 0.296 | 0.082 | 0.009 | 0.045 |
| QOL AND AUTONOMY | ||||||||
| PDQ-39SI | 0.326 *** | 0.072 | 0.247 | 0.439 *** | 0.069 | 0.055 | 0.315 *** | 0.019 |
| - Mobility | 0.166 | −0.187 | 0.285 | 0.335 *** | −0.061 | 0.004 | 0.243 | −0.070 |
| - Activities of daily living | 0.196 | 0.008 | 0.147 | 0.263 | 0.034 | 0.009 | 0.361 *** | −0.224 |
| - Emotional well-being | 0.252 | 0.241 | 0.211 | 0.229 | 0.027 | 0.069 | 0.135 | −0.026 |
| - Stigmatization | −0.147 | −0.154 | −0.117 | 0.035 | −0.020 | −0.153 | −0.187 | −0.023 |
| - Social support | 0.006 | 0.064 | −0.340 *** | 0.035 | 0.165 | 0.092 | 0.053 | −0.073 |
| - Cognition | 0.183 | 0.29 | −0.035 | 0.029 | 0.26 | −0.021 | −0.014 | 0.207 |
| - Communication | 0.15 | 0.325 *** | −0.033 | 0.235 | −0.049 | 0.097 | −0.051 | 0.149 |
| - Pain and discomfort | 0.426 * | 0.238 | 0.188 | 0.232 | 0.275 | 0.294 | 0.369 *** | 0.207 |
| ADLS | 0.005 | 0.011 | 0.016 | −0.095 | −0.054 | −0.050 | 0.025 | 0.276 |
| β a | β b | 95% IC a | 95% IC b | p a | p b | |
|---|---|---|---|---|---|---|
| At V1 (baseline) | ||||||
| Age | 0.041 | N. A. | −0.848–1.102 | N. A. | 0.794 | N. A. |
| Gender | 0.326 | N. A. | 1.592–35.156 | N. A. | 0.033 | N. A. |
| Disease duration | 0.166 | N. A. | 0.019–1.499 | N. A. | 0.293 | N. A. |
| LEDD | 0.359 | N. A. | 0.003–0.037 | N. A. | 0.02 | N. A. |
| KPPS total score | 0.783 | 0.66 | 0.480–0.802 | 0.363–0.718 | <0.0001 | <0.0001 |
| Change at V4 (from V1 to V4) | ||||||
| UPDRS-III | 0.255 | N. A. | −0.237–1.698 | N. A. | 0.134 | N. A. |
| UPDRS-IV | 0.031 | N. A. | −3.984–4.710 | N. A. | 0.866 | N. A. |
| FOGQ | 0.106 | N. A. | −1.147–2.268 | N. A. | 0.511 | N. A. |
| NMSS | 0.577 | 0.242 | 0.280–0.731 | 0.022–0.402 | <0.0001 | 0.03 |
| ESS | 0.118 | N. A. | −1.233–2.695 | N. A. | 0.457 | N. A. |
| PSQI | 0.062 | N. A. | −0.369–0.215 | N. A. | 0.711 | N. A. |
| BDI-II | 0.508 | N. A. | 0.895–2.949 | N. A. | 0.001 | N. A. |
| VAFS-Physical | 0.339 | N. A. | 0.328–5.511 | N. A. | 0.028 | N. A. |
| VAFS-Mental | 0.206 | N. A. | −0.957–4.629 | N. A. | 0.192 | N. A. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos García, D.; Yáñez Baña, R.; Labandeira Guerra, C.; Cimas Hernando, M.I.; Cabo López, I.; Paz González, J.M.; Alonso Losada, M.G.; Gonzalez Palmás, M.J.; Cores Bartolomé, C.; Martínez Miró, C. Pain Improvement in Parkinson’s Disease Patients Treated with Safinamide: Results from the SAFINONMOTOR Study. J. Pers. Med. 2021, 11, 798. https://doi.org/10.3390/jpm11080798
Santos García D, Yáñez Baña R, Labandeira Guerra C, Cimas Hernando MI, Cabo López I, Paz González JM, Alonso Losada MG, Gonzalez Palmás MJ, Cores Bartolomé C, Martínez Miró C. Pain Improvement in Parkinson’s Disease Patients Treated with Safinamide: Results from the SAFINONMOTOR Study. Journal of Personalized Medicine. 2021; 11(8):798. https://doi.org/10.3390/jpm11080798
Chicago/Turabian StyleSantos García, Diego, Rosa Yáñez Baña, Carmen Labandeira Guerra, Maria Icíar Cimas Hernando, Iria Cabo López, Jose Manuel Paz González, Maria Gema Alonso Losada, Maria José Gonzalez Palmás, Carlos Cores Bartolomé, and Cristina Martínez Miró. 2021. "Pain Improvement in Parkinson’s Disease Patients Treated with Safinamide: Results from the SAFINONMOTOR Study" Journal of Personalized Medicine 11, no. 8: 798. https://doi.org/10.3390/jpm11080798
APA StyleSantos García, D., Yáñez Baña, R., Labandeira Guerra, C., Cimas Hernando, M. I., Cabo López, I., Paz González, J. M., Alonso Losada, M. G., Gonzalez Palmás, M. J., Cores Bartolomé, C., & Martínez Miró, C. (2021). Pain Improvement in Parkinson’s Disease Patients Treated with Safinamide: Results from the SAFINONMOTOR Study. Journal of Personalized Medicine, 11(8), 798. https://doi.org/10.3390/jpm11080798






