Augmented-Reality-Assisted K-Wire Placement for Glenoid Component Positioning in Reversed Shoulder Arthroplasty: A Proof-of-Concept Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Imaging Data
2.2. Procedure Planning
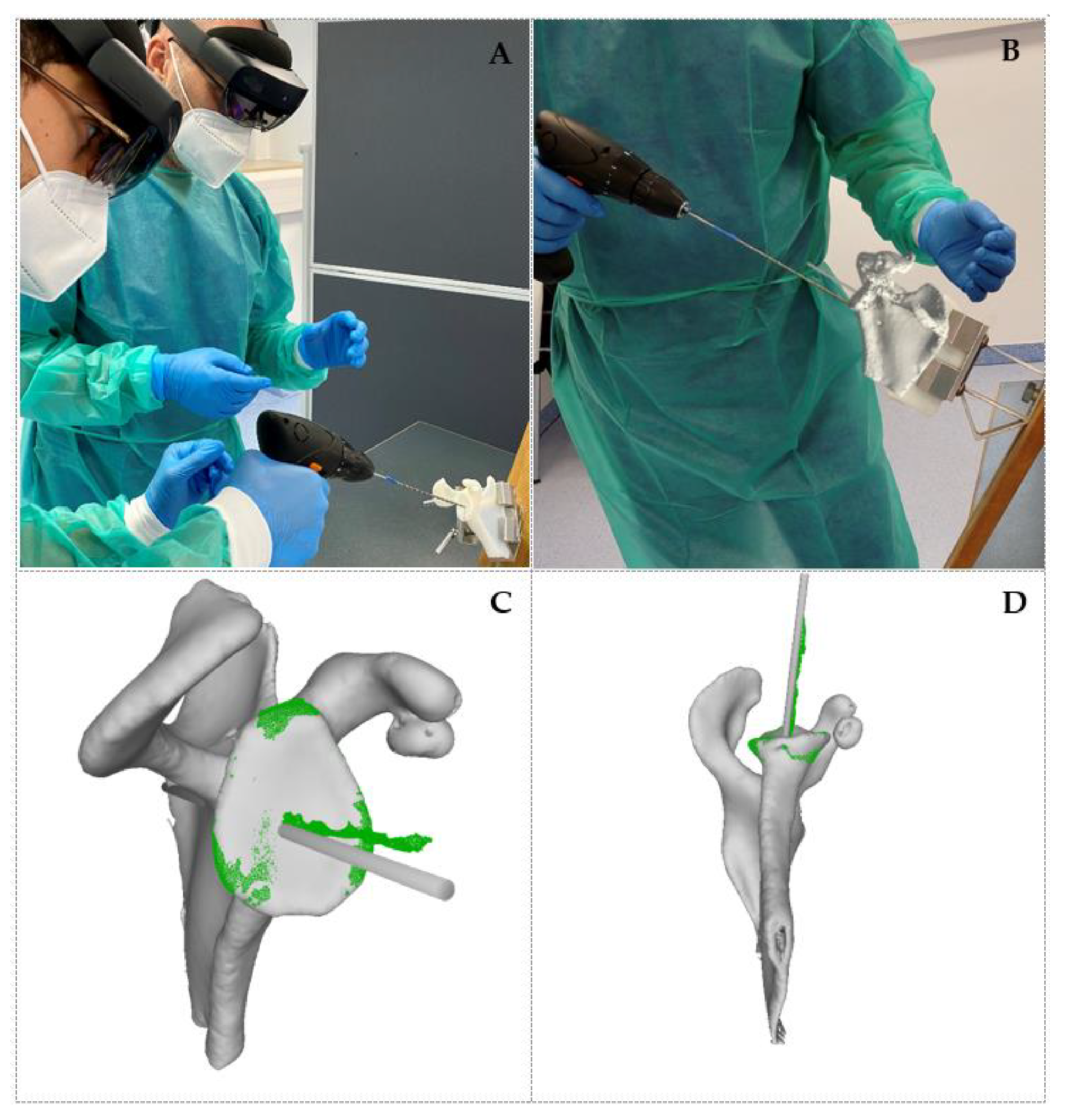
2.3. Procedure Execution
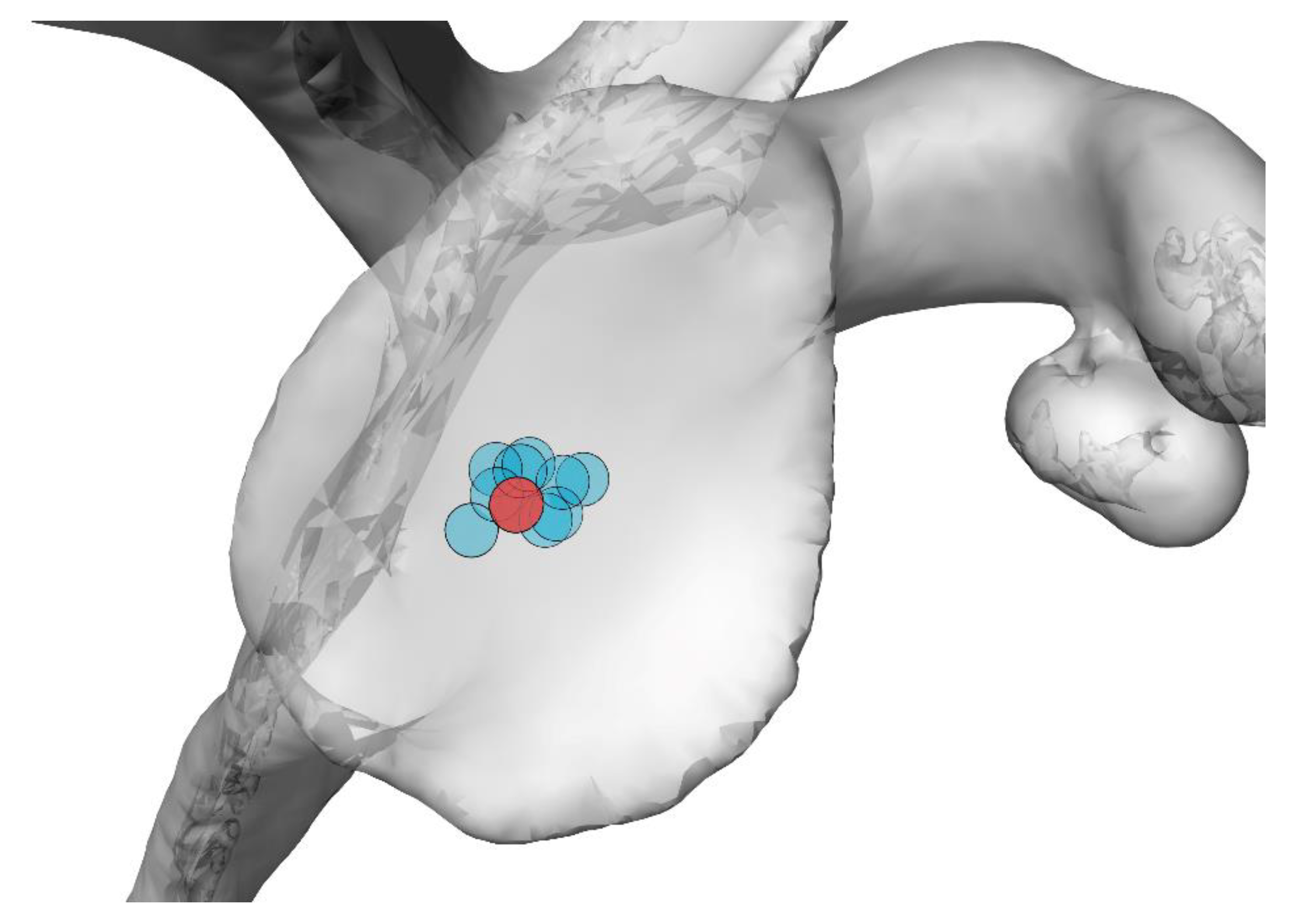
2.4. Error Measurement
3. Results
3.1. Qualitative Results
3.2. Quantitative Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Olaiya, O.R.; Nadeem, I.; Horner, N.S.; Bedi, A.; Leroux, T.; Alolabi, B.; Khan, M. Templating in shoulder arthroplasty—A comparison of 2D CT to 3D CT planning software: A systematic review. Shoulder Elb. 2020, 12, 303–314. [Google Scholar] [CrossRef]
- Lee, D.H.; Choi, Y.S.; Potter, H.G.; Endo, Y.; Sivakumaran, T.; Lim, T.K.; Chun, T.J. Reverse total shoulder arthroplasty: An imaging overview. Skelet. Radiol. 2019, 49, 19–30. [Google Scholar] [CrossRef]
- Chae, J.; Siljander, M.; Wiater, J.M. Instability in Reverse Total Shoulder Arthroplasty. J. Am. Acad. Orthop. Surg. 2018, 26, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Fevang, B.-T.S.; Lie, S.A.; Havelin, I.L.; Skredderstuen, A.; Furnes, O. Risk factors for revision after shoulder arthroplasty. Acta Orthop. 2009, 80, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Guery, J.; Favard, L.; Sirveaux, F.; Oudet, D.; Mole, D.; Walch, G. Reverse Total Shoulder Arthroplasty. J. Bone Jt. Surg. Am. 2006, 88, 1742–1747. [Google Scholar] [CrossRef]
- Boileau, P. Complications and revision of reverse total shoulder arthroplasty. Orthop. Traumatol. Surg. Res. 2016, 102, S33–S43. [Google Scholar] [CrossRef]
- Wall, B.T.; Mottier, F.; Walch, G. 9: Complications and revision of the reverse prosthesis: A multicenter study of 457 cases. J. Shoulder Elb. Surg. 2007, 16, e55. [Google Scholar] [CrossRef]
- Gutiérrez, S.; Greiwe, R.M.; Frankle, M.; Siegal, S.; Lee, W.E. Biomechanical comparison of component position and hardware failure in the reverse shoulder prosthesis. J. Shoulder Elb. Surg. 2007, 16, S9–S12. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, P.N.; Boileau, P.; Romeo, A.A.; Tashjian, R.Z. Revision Reverse Shoulder Arthroplasty. J. Am. Acad. Orthop. Surg. 2019, 27, 426–436. [Google Scholar] [CrossRef]
- Liu, H.; Auvinet, E.; Giles, J.; Baena, F.R.Y. Augmented Reality Based Navigation for Computer Assisted Hip Resurfacing: A Proof of Concept Study. Ann. Biomed. Eng. 2018, 46, 1595–1605. [Google Scholar] [CrossRef] [Green Version]
- Blackwell, M.; Morgan, F.; DiGioia, A.M. Augmented Reality and Its Future in Orthopaedics. Clin. Orthop. Relat. Res. 1998, 354, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Molina, A.C.; Sciubba, D.M.; Greenberg, J.K.; Khan, M.; Witham, T. Clinical Accuracy, Technical Precision, and Workflow of the First in Human Use of an Augmented-Reality Head-Mounted Display Stereotactic Navigation System for Spine Surgery. Oper. Neurosurg. 2021, 20, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Lane, P.; Murphy, W.; Harris, S.; Murphy, S. Hipinsight: The World’s First Augmented Reality-Based Navigation System For Joint Arthroplasty. In Orthopaedic Proceedings; The British Editorial Society of Bone & Joint Surgery: London, UK, 2021; Volume 103-B, No. SUPP_9. [Google Scholar]
- Kriechling, P.; Roner, S.; Liebmann, F.; Casari, F.; Fürnstahl, P.; Wieser, K. Augmented reality for base plate component placement in reverse total shoulder arthroplasty: A feasibility study. Arch. Orthop. Trauma Surg. 2020. [CrossRef]
- BLUEPRINT™ Scan Protocol. Available online: https://www.wrightemedia.com/ProductFiles/Files/PDFs/AP-013380_EN_LR_LE.pdf (accessed on 17 May 2021).
- Besl, P.J.; McKay, N.D. A method for registration of 3-D shapes. IEEE Trans. Pattern Anal. Mach. Intell. 1992, 14, 239–256. [Google Scholar] [CrossRef]
- Cabarcas, B.C.; Cvetanovich, G.L.; Gowd, A.K.; Liu, J.N.; Manderle, B.J.; Verma, N.N. Accuracy of patient-specific instrumentation in shoulder arthroplasty: A systematic review and meta-analysis. JSES Open Access 2019, 3, 117–129. [Google Scholar] [CrossRef] [Green Version]
- Villatte, G.; Muller, A.-S.; Pereira, B.; Mulliez, A.; Reilly, P.; Emery, R. Use of Patient-Specific Instrumentation (PSI) for glenoid component positioning in shoulder arthroplasty. A systematic review and meta-analysis. PLoS ONE 2018, 13, e0201759. [Google Scholar] [CrossRef] [PubMed]
- Schoch, B.S.; Haupt, E.; Leonor, T.; Farmer, K.W.; Wright, T.W.; King, J.J. Computer navigation leads to more accurate glenoid targeting during total shoulder arthroplasty compared with 3-dimensional preoperative planning alone. J. Shoulder Elb. Surg. 2020, 29, 2257–2263. [Google Scholar] [CrossRef] [PubMed]
- Cercenelli, L.; Carbone, M.; Condino, S.; Cutolo, F.; Marcelli, E.; Tarsitano, A.; Marchetti, C.; Ferrari, V.; Badiali, G. The Wearable VOSTARS System for Augmented Reality-Guided Surgery: Preclinical Phantom Evaluation for High-Precision Maxillofacial Tasks. J. Clin. Med. 2020, 9, 3562. [Google Scholar] [CrossRef]
- Smith, T.J.; Siddiqi, A.; Forte, S.A.; Judice, A.; Sculco, P.K.; Vigdorchik, J.M.; Schwarzkopf, R.; Springer, B.D. Periprosthetic Fractures Through Tracking Pin Sites Following Computer Navigated and Robotic Total and Unicompartmental Knee Arthroplasty. JBJS Rev. 2021, 9, e20.00091. [Google Scholar] [CrossRef]
- Hu, X.; Cutolo, F.; Tatti, F.; Baena, F.R.Y. Automatic Calibration of Commercial Optical See-Through Head-Mounted Displays for Medical Applications. In Proceedings of the 2020 IEEE Conference on Virtual Reality and 3D User Interfaces Abstracts and Workshops (VRW), Atlanta, GA, USA, 22–26 March 2020; pp. 755–756. [Google Scholar] [CrossRef]
- Hu, X.; Baena, F.R.Y.; Cutolo, F. Alignment-Free Offline Calibration of Commercial Optical See-Through Head-Mounted Displays With Simplified Procedures. IEEE Access 2020, 8, 223661–223674. [Google Scholar] [CrossRef]
- Dibble, C.F.; Molina, C.A. Device profile of the XVision-spine (XVS) augmented-reality surgical navigation system: Overview of its safety and efficacy. Expert Rev. Med. Devices 2021, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
| Phantom ID | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 |
| Entry point (mm) | 2.8 | 1.9 | 1.2 | 1.8 | 2.3 | 2.8 | 2.3 | 3.9 | 2.3 |
| Orientation (°) | 9.0 | 5.3 | 6.7 | 2.3 | 2.1 | 1.8 | 4.2 | 2.2 | 1.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schlueter-Brust, K.; Henckel, J.; Katinakis, F.; Buken, C.; Opt-Eynde, J.; Pofahl, T.; Rodriguez y Baena, F.; Tatti, F. Augmented-Reality-Assisted K-Wire Placement for Glenoid Component Positioning in Reversed Shoulder Arthroplasty: A Proof-of-Concept Study. J. Pers. Med. 2021, 11, 777. https://doi.org/10.3390/jpm11080777
Schlueter-Brust K, Henckel J, Katinakis F, Buken C, Opt-Eynde J, Pofahl T, Rodriguez y Baena F, Tatti F. Augmented-Reality-Assisted K-Wire Placement for Glenoid Component Positioning in Reversed Shoulder Arthroplasty: A Proof-of-Concept Study. Journal of Personalized Medicine. 2021; 11(8):777. https://doi.org/10.3390/jpm11080777
Chicago/Turabian StyleSchlueter-Brust, Klaus, Johann Henckel, Faidon Katinakis, Christoph Buken, Jörg Opt-Eynde, Thorsten Pofahl, Ferdinando Rodriguez y Baena, and Fabio Tatti. 2021. "Augmented-Reality-Assisted K-Wire Placement for Glenoid Component Positioning in Reversed Shoulder Arthroplasty: A Proof-of-Concept Study" Journal of Personalized Medicine 11, no. 8: 777. https://doi.org/10.3390/jpm11080777
APA StyleSchlueter-Brust, K., Henckel, J., Katinakis, F., Buken, C., Opt-Eynde, J., Pofahl, T., Rodriguez y Baena, F., & Tatti, F. (2021). Augmented-Reality-Assisted K-Wire Placement for Glenoid Component Positioning in Reversed Shoulder Arthroplasty: A Proof-of-Concept Study. Journal of Personalized Medicine, 11(8), 777. https://doi.org/10.3390/jpm11080777






