Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorders: Diagnosis, Treatment and Prevention
Abstract
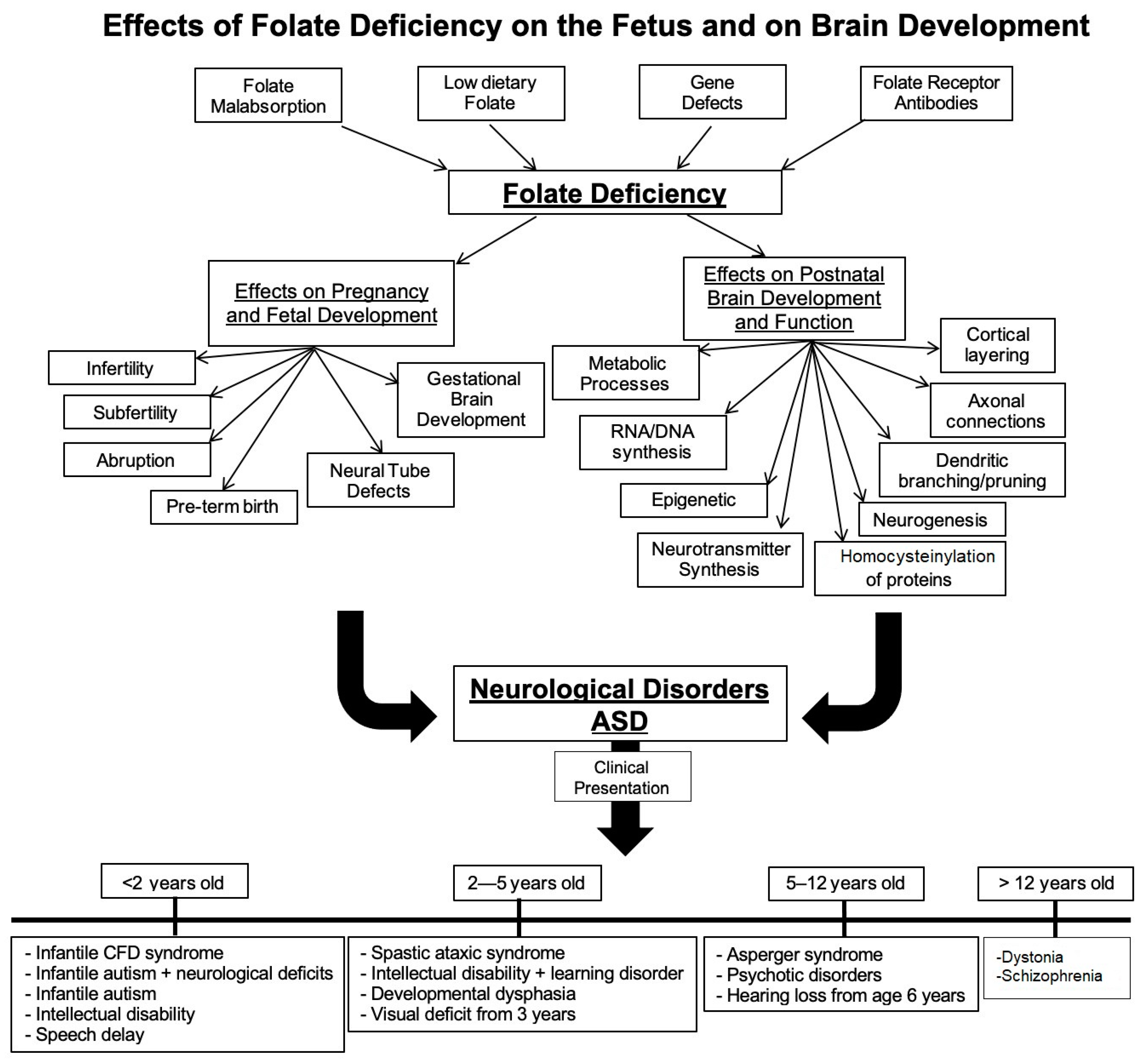
1. Background
2. Folate Requirements during Pregnancy
3. Folate and Fetal Brain Development
4. Folate and Neonatal Brain Development
5. Folate Receptors: Expression and Function
6. FRα Role in Maternofetal Transport of Folate
7. FRα Role in Folate Transport to the Brain
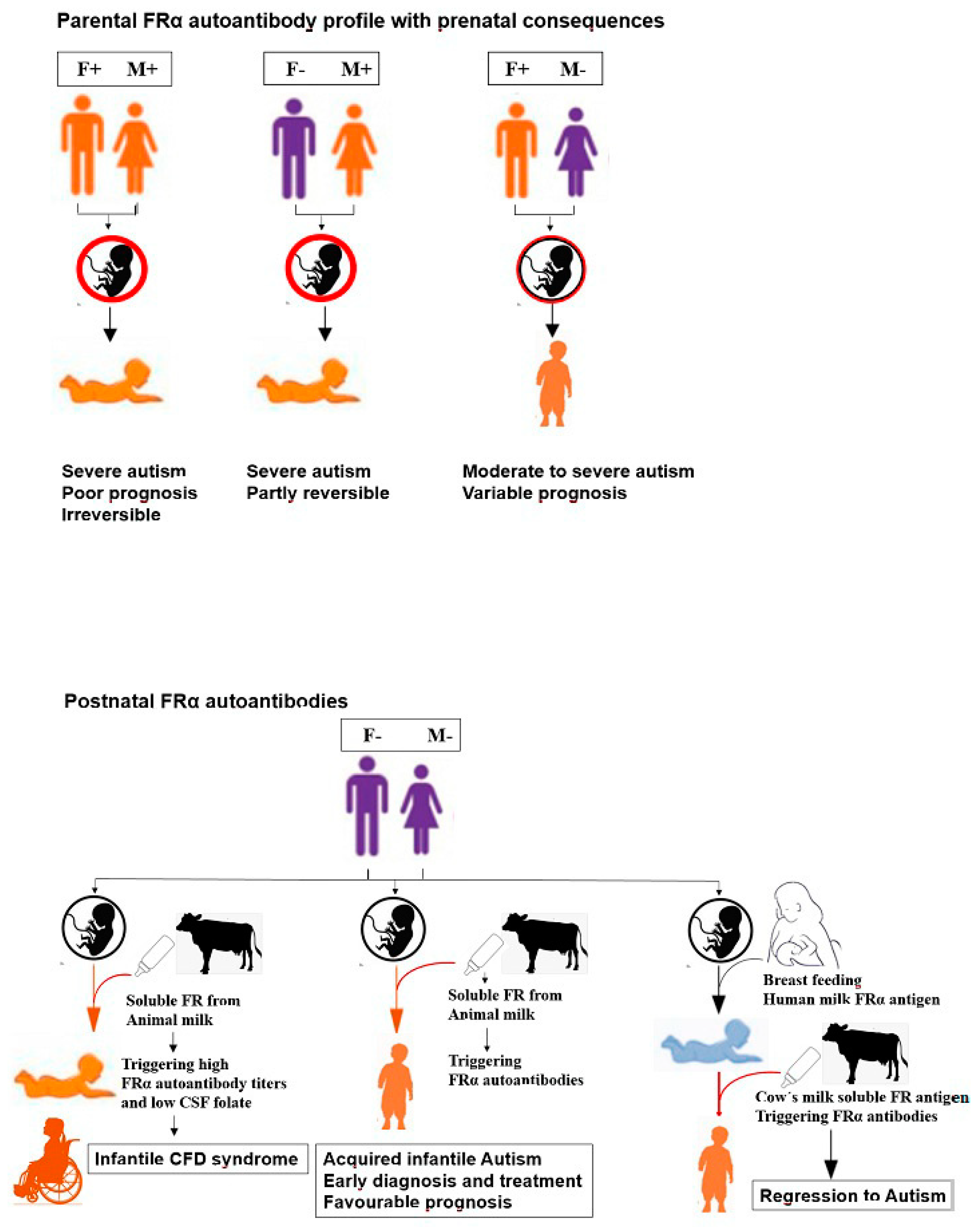
8. Folate Receptor Autoantibodies: Their Role in Disrupting Folate Transport
9. Pathologic Consequences of Folate Receptor Antibodies
10. Diagnosis of Folate Receptor Autoimmune Disorder
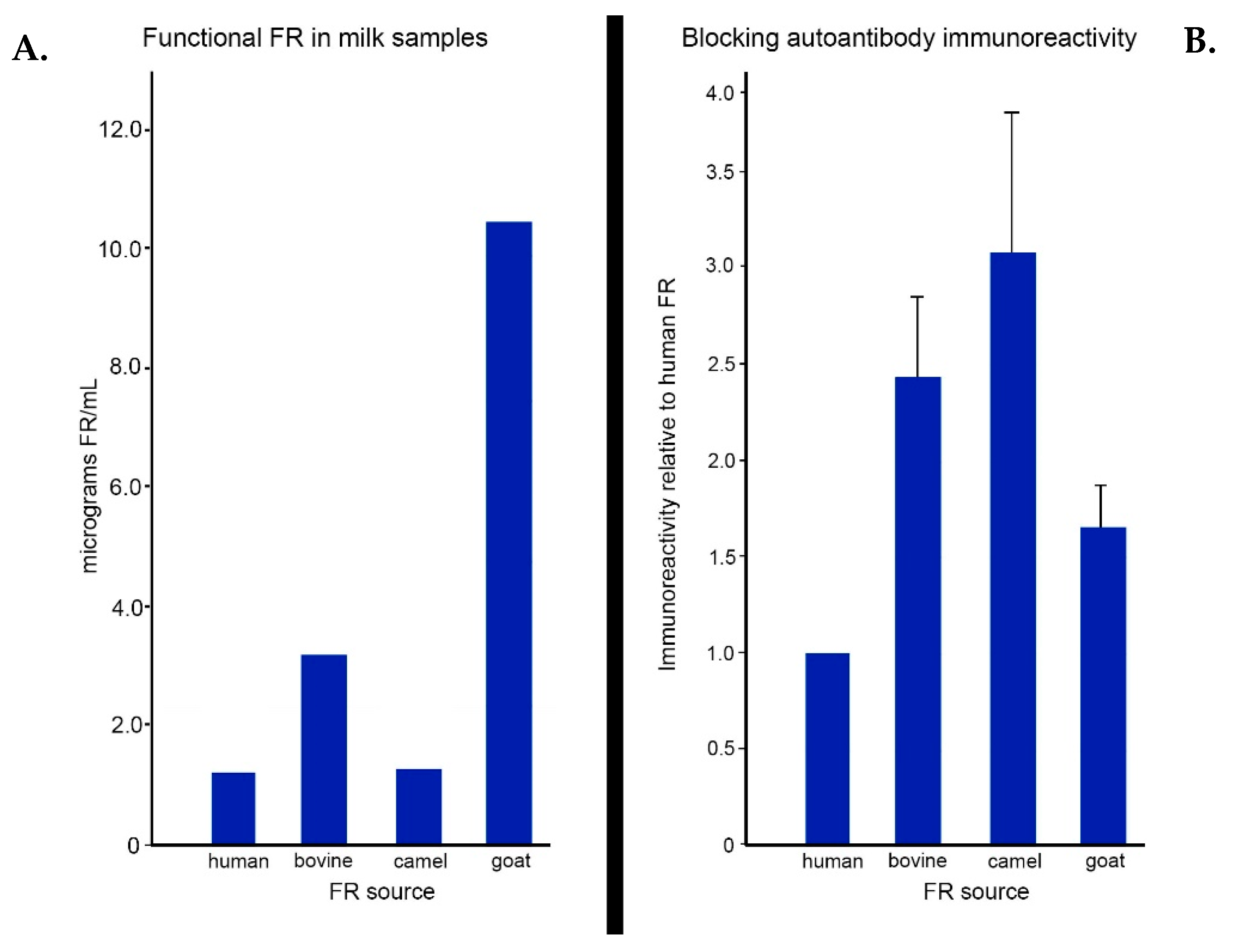
11. Assay for Blocking Antibodies
12. Assay for Binding Antibody
13. Treatment of FRα Autoimmune Disorder in ASD
14. Treatment of FR Autoimmune Disorder in Pregnancy
15. Prevention of ASD and Related Complications Due to FR Antibodies
16. Clinical Significance of the Findings
17. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rucker, R.B.; Zempleni, J.; Suttie, J.W.; McCormick, D.B. Handbook of Vitamins, 4th ed.; Taylor & Francis: Boca Raton, FL, USA, 2007; Available online: https://books.google.com/books?id=AasGngEACAAJ (accessed on 4 November 2019).
- Mikkelsen, K.; Apostolopoulos, V. Vitamin B12, Folic Acid, and the Immune System. In Nutrition and Immunity; Mahmoudi, M., Rezaei, N., Eds.; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Scott, J.M. Folate and vitamin B12. Proc. Nutr. Soc. 1999, 58, 441–448. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Blau, N. Cerebral folate deficiency. Dev. Med. Child. Neurol. 2004, 46, 843–851. [Google Scholar] [CrossRef]
- Cario, H.; Bode, H.; Debatin, K.M.; Opladen, T.; Schwarz, K. Congenital null mutations of the FOLR1 gene: A progressive neurologic disease and its treatment. Neurology 2009, 73, 2127–2129. [Google Scholar] [CrossRef]
- Pérez-Dueñas, B.; Toma, C.; Ormazábal, A.; Muchart, J.; Sanmartí, F.; Bombau, G.; Serrano, M.; García-Cazorla, A.; Cormand, B.; Artuch, R. Progressive ataxia and myoclonic epilepsy in a patient with a homozygous mutation in the FOLR1 gene. J. Inherit. Metab. Dis. 2010, 33, 795–802. [Google Scholar] [CrossRef]
- Delmelle, F.; Thöny, B.; Clapuyt, P.; Blau, N.; Nassogne, M.C. Neurological improvement following intravenous high-dose folinic acid for cerebral folate transporter deficiency caused by FOLR-1 mutation. Eur. J. Paediatr. Neurol. 2016, 20, 709–713. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Rothenberg, S.P.; Sequeira, J.M.; Opladen, T.; Blau, N.; Quadros, E.V.; Selhub, J. Autoantibodies to folate receptors in the cerebral folate deficiency syndrome. N. Engl. J. Med. 2005, 352, 1985–1991. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Segers, K.; Sequeira, J.M.; Koenig, M.; Van Maldergem, L.; Bours, V.; Kornak, U.; Quadros, E.V. Genetic assessment and folate receptor autoantibodies in infantile-onset cerebral folate deficiency (CFD) syndrome. Mol. Genet. Metab. 2018, 124, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Wolf, A.; Kim, S.E.; Cabrera, R.M.; Wlodarczyk, B.J.; Zhu, H.; Parker, M.; Lin, Y.; Steele, J.W.; Han, X.; et al. CIC de novo loss of function variants contribute to cerebral folate deficiency by downregulating FOLR1 expression. J. Med. Genet. 2020, 1–11. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Blau, N.; Sequeira, J.M.; Nassogne, M.C.; Quadros, E.V. Folate receptor autoimmunity and cerebral folate deficiency in low-functioning autism with neurological deficits. Neuropediatrics 2007, 38, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Ramaekers, V.T.; Sequeira, J.M.; Thöny, B.; Quadros, E.V. Oxidative Stress, Folate Receptor Autoimmunity, and CSF Findings in Severe Infantile Autism. Autism Res. Treat. 2020, 2020, 9095284. [Google Scholar] [CrossRef] [PubMed]
- Fekete, K.; Berti, C.; Trovato, M.; Lohner, S.; Dullmeijer, C.; Souverein, O.W.; Cetin, I.; Decsi, T. Effect of folate intake on health outcomes in pregnancy: A systematic review and meta-analysis on birth weight, placental weight and length of gestation. Nutr. J. 2012, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, J.A.; Bell, S.J.; Guan, Y.; Yu, Y. Folic acid supplementation and pregnancy—More than just neural tube defect prevention. Rev. Obs. Gynecol. 2011, 4, 52–59. [Google Scholar]
- Bailey, L.B.; Stover, P.J.; McNulty, H.; Fenech, M.F.; Gregory, J.F., 3rd; Mills, J.L.; Pfeiffer, C.M.; Fazili, Z.; Zhang, M.; Ueland, P.M.; et al. Biomarkers of nutrition for development-Folate Review. J. Nutr. 2015, 145, 1636S–1680S. [Google Scholar] [CrossRef]
- Rothenberg, S.P.; da Costa, M.P.; Sequeira, J.M.; Cracco, J.; Roberts, J.L.; Weedon, J.; Quadros, E.V. Autoantibodies against folate receptors in women with a pregnancy complicated by a neural-tube defect. N. Engl. J. Med. 2004, 350, 134–142. [Google Scholar] [CrossRef]
- Shapira, I.; Sequeira, J.M.; Quadros, E.V. Folate receptor autoantibodies in pregnancy related complications. Birth Defects Res. Part A Clin. Mol. Teratol. 2015, 103, 1028–1030. [Google Scholar] [CrossRef]
- Peng, L.; Dreumont, N.; Coelho, D.; Guéant, J.L.; Arnold, C. Genetic animal models to decipher the pathogenic effects of vitamin B12 and folate deficiency. Biochimie 2016, 126, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Kappen, C. Folate supplementation in three genetic models: Implications for understanding folate-dependent developmental pathways. Am. J. Med. Genet. Part C Semin Med. Genet. 2005, 135C, 24–30. [Google Scholar] [CrossRef]
- Piedrahita, J.A.; Oetama, B.; Bennett, G.D.; van Waes, J.; Kamen, B.A.; Richardson, J.; Lacey, S.W.; Anderson, R.G.; Finnell, R.H. Mice lacking the folic acid-binding protein Folbp1 are defective in early embryonic development. Nat. Genet. 1999, 23, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.S.; Santillano, D.R.; Wlodarczyk, B.J.; Miranda, R.C.; Finnell, R.H. Role of Folbp1 in the regional regulation of apoptosis and cell proliferation in the developing neural tube and craniofacies. Am. J. Med. Genet. Part C Semin. Med. Genet. 2005, 135C, 48–58. [Google Scholar] [CrossRef]
- Craciunescu, C.N.; Brown, E.C.; Mar, M.H.; Albright, C.D.; Nadeau, M.R.; Zeisel, S.H. Folic acid deficiency during late gestation decreases progenitor cell proliferation and increases apoptosis in fetal mouse brain. J Nutr. 2004, 134, 162–166. [Google Scholar] [CrossRef] [PubMed]
- Berrocal-Zaragoza, M.I.; Sequeira, J.M.; Murphy, M.M.; Fernandez-Ballart, J.D.; Abdel Baki, S.G.; Bergold, P.J.; Quadros, E.V. Folate deficiency in rat pups during weaning causes learning and memory deficits. Br. J. Nutr. 2014, 112, 1323–1332. [Google Scholar] [CrossRef]
- Blaise, S.A.; Nédélec, E.; Schroeder, H.; Alberto, J.M.; Bossenmeyer-Pourié, C.; Guéant, J.L.; Daval, J.L. Gestational vitamin B deficiency leads to homocysteine-associated brain apoptosis and alters neurobehavioral development in rats. Am. J. Pathol. 2007, 170, 667–679. [Google Scholar] [CrossRef] [PubMed]
- da Costa, M.; Sequeira, J.M.; Rothenberg, S.P.; Weedon, J. Antibodies to Folate Receptors Impair Embryogenesis and Fetal Development in the Rat. Birth Defects Res. Part A Clin. Mol. Teratol. 2003, 67, 837–847. [Google Scholar] [CrossRef]
- Sequeira, J.M.; Desai, A.; Berrocal-Zaragoza, M.I.; Murphy, M.M.; Fernandez-Ballart, J.D.; Quadros, E.V. Exposure to folate receptor alpha antibodies during gestation and weaning leads to severe behavioral deficits in rats: A pilot study. PLoS ONE 2016, 11, e0152249. [Google Scholar] [CrossRef]
- Desai, A.; Sequeira, J.M.; Quadros, E.V. Prevention of behavioral deficits in rats exposed to folate receptor antibodies: Implication in autism. Mol. Psychiatry 2017, 22, 1291–1297. [Google Scholar] [CrossRef]
- Balashova, O.A.; Visina, O.; Borodinsky, L.N. Folate action in nervous system development and disease. Dev. Neurobiol. 2018, 78, 391–402. [Google Scholar] [CrossRef]
- Mann, A.; Portnoy, E.; Han, H.; Inbar, D.; Blatch, D.; Shmuel, M.; Ben-Hur, T.; Eyal, S.; Ekstein, D. Folate homeostasis in epileptic rats. Epilepsy Res. 2018, 142, 64–72. [Google Scholar] [CrossRef]
- Kruman, I.I.; Mouton, P.R.; Emokpae, R., Jr.; Cutler, R.G.; Mattson, M.P. Folate deficiency inhibits proliferation of adult hippocampal progenitors. NeuroReport. 2005, 16, 1055–1059. [Google Scholar] [CrossRef] [PubMed]
- Weng, Q.; Wang, J.; Wang, J.; Tan, B.; Wang, J.; Wang, H.; Zheng, T.; Lu, Q.R.; Yang, B.; He, Q. Folate Metabolism Regulates Oligodendrocyte Survival and Differentiation by Modulating AMPKα Activity. Sci. Rep. 2017, 7, 1705. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.B.; Chen, Y.; Kang, W.; Guo, J.; Payne, R.; Li, H.; Wei, Q.; Baker, J.; Dong, C.; Zhang, S.; et al. The critical chemical and mechanical regulation of folic acid on neural engineering. Biomaterials. 2018, 178, 504–516. [Google Scholar] [CrossRef]
- Mayanil, C.S.; Siddiqui, M.R.; Tomita, T. Novel functions of folate receptor alpha in CNS development and diseases. Neurosci. Discov. 2014, 2, 5. [Google Scholar] [CrossRef]
- Hou, Z.; Matherly, L.H. Biology of the major facilitative folate transporters SLC19A1 and SLC46A1. Curr. Top. Membr. 2014, 73, 175–204. [Google Scholar] [CrossRef]
- Antony, A.C. Folate receptors. Annu. Rev. Nutr. 1996, 16, 501–521. [Google Scholar] [CrossRef] [PubMed]
- Machacek, C.; Supper, V.; Leksa, V.; Mitulovic, G.; Spittler, A.; Drbal, K.; Suchanek, M.; Ohradanova-Repic, A.; Stockinger, H. Folate Receptor β Regulates Integrin CD11b/CD18 Adhesion of a Macrophage Subset to Collagen. J. Immunol. 2016, 197, 2229–2238. [Google Scholar] [CrossRef]
- Kelemen, L.E. The role of folate receptor alpha in cancer development, progression and treatment: Cause, consequence or innocent bystander? Int. J. Cancer 2006, 119, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Spiegelstein, O.; Eudy, J.D.; Finnell, R.H. Identification of two putative novel folate receptor genes in humans and mouse. Gene 2000, 258, 117–125. [Google Scholar] [CrossRef]
- Holm, J.; Hansen, S.I. Characterization of soluble folate receptors (folate binding proteins) in humans. Biological roles and clinical potentials in infection and malignancy. Biochim. Biophys. Acta Proteins Proteom. 2020, 1868, 140466. [Google Scholar] [CrossRef] [PubMed]
- Alam, C.; Hoque, M.T.; Finnell, R.H.; Goldman, I.D.; Bendayan, R. Regulation of Reduced Folate Carrier (RFC) by Vitamin D Receptor at the Blood-Brain Barrier. Mol. Pharm. 2017, 14, 3848–3858. [Google Scholar] [CrossRef]
- Yasuda, S.; Hasui, S.; Yamamoto, C.; Yoshioka, C.; Kobayashi, M.; Itagaki, S.; Hirano, T.; Iseki, K. Placental folate transport during pregnancy. Biosci. Biotechnol. Biochem. 2008, 72, 2277–2284. [Google Scholar] [CrossRef] [PubMed]
- Grapp, M.; Wrede, A.; Schweizer, M.; Hüwel, S.; Galla, H.J.; Snaidero, N.; Simons, M.; Bückers, J.; Low, P.S.; Urlaub, H.; et al. Choroid plexus transcytosis and exosome shuttling deliver folate into brain parenchyma. Nat. Commun. 2013, 4, 2123. [Google Scholar] [CrossRef]
- Alam, C.; Kondo, M.; O’Connor, D.L.; Bendayan, R. Clinical Implications of Folate Transport in the Central Nervous System. Trends Pharmacol. Sci. 2020, 41, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.; Sequeira, J.M.; and Quadros, E.V. The metabolic basis for developmental disorders due to defective folate transport. Biochimie 2016, 126, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Berrocal-Zaragoza, M.I.; Fernandez-Ballart, J.D.; Murphy, M.M.; Cavallé-Busquets, P.; Sequeira, J.M.; Quadros, E.V. Association between blocking folate receptor autoantibodies and subfertility. Fertil. Steril. 2009, 91 (Suppl. 4), 1518–1521. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vo, H.D.; Sequeira, J.M.; Quadros, E.V.; Schwarz, S.M.; Perenyi, A.R. The role of folate receptor autoantibodies in preterm birth. Nutrition 2015, 31, 1224–1227. [Google Scholar] [CrossRef]
- Ramaekers, V.; Sequeira, J.M.; Quadros, E.V. Clinical recognition and aspects of the cerebral folate deficiency syndromes. Clin. Chem. Lab. Med. 2013, 51, 497–511. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Slattery, J.; Delhey, L.; Furgerson, B.; Strickland, T.; Tippett, M.; Sailey, A.; Wynne, R.; Rose, S.; Melnyk, S.; et al. Folinic acid improves verbal communication in children with autism and language impairment: A randomized double-blind placebo-controlled trial. Mol. Psychiatry 2018, 23, 247–256. [Google Scholar] [CrossRef]
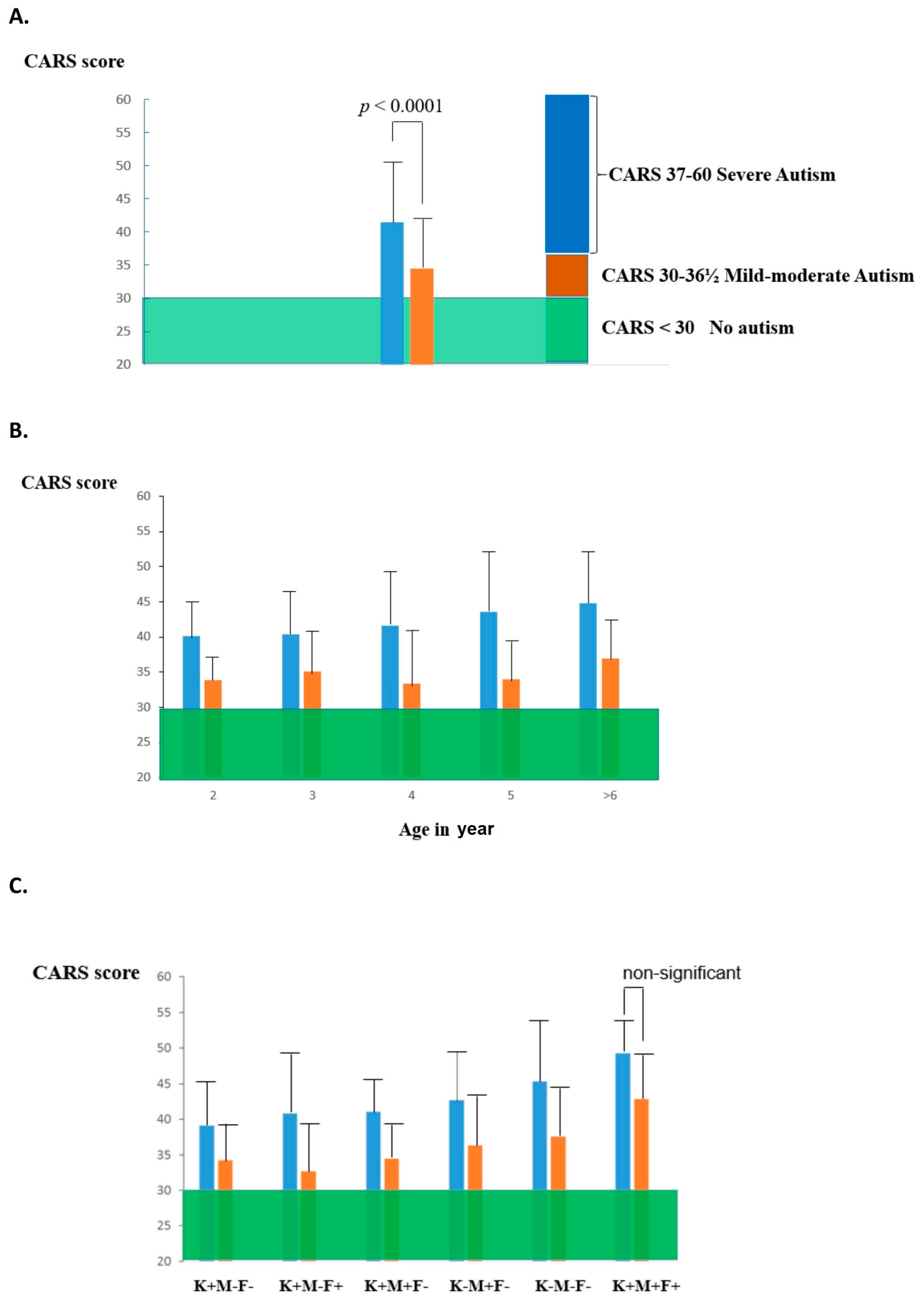
- Frye, R.E.; Delhey, L.; Slattery, J.; Tippett, M.; Wynne, R.; Rose, S.; Kahler, S.G.; Bennuri, S.C.; Stepan, M.; Sequeira, J.M.; et al. Blocking and binding folate receptor alpha autoantibodies identify novel autism spectrum disorder subgroups. Front. Neurosci. 2016, 10, 80. [Google Scholar] [CrossRef]
- Sequeira, J.M.; Ramaekers, V.T.; Quadros, E.V. The diagnostic utility of folate receptor autoantibodies in blood. Clin. Chem. Lab. Med. 2013, 51, 545–554. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Sequeira, J.M.; DiDuca, M.; Vrancken, G.; Thomas, A.; Philippe, C.; Peters, M.; Jadot, A.; Quadros, E.V. Improving Outcome in Infantile Autism with Folate Receptor Autoimmunity and Nutritional Derangements: A Self-Controlled Trial. Autism Res. Treat. 2019, 2019, 7486431. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liu, A.; He, F.; Jin, Y.; Zhou, S.; Xu, R.; Guo, H.; Zhou, W.; Wei, Q.; Wang, M. High prevalence of serum folate receptor autoantibodies in children with autism spectrum disorders. Biomarkers 2018, 23, 622–624. [Google Scholar] [CrossRef] [PubMed]
- Elsabbagh, M.; Divan, G.; Koh, Y.J.; Shin Kim, Y.; Kauchali, S.; Marcin, C.; Montiel-Nava, C.; Patel, P.; Paula, C.S.; Wang, C.; et al. Global Prevalence of Autism and Other Pervasive Developmental Disorders. Autism Res. 2012, 5, 160–179. [Google Scholar] [CrossRef]
- Chiarotti, F.; Venerosi, A. Epidemiology of Autism Spectrum Disorders: A Review of Worldwide Prevalence Estimates Since 2014. Brain Sci. 2020, 10, 274. [Google Scholar] [CrossRef] [PubMed]
- Ramaekers, V.T.; Sequeira, J.M.; Blau, N.; Quadros, E.V. A milk-free diet downregulates folate receptor autoimmunity in cerebral folate deficiency syndrome. Dev. Med. Child. Neurol. 2008, 50, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Whiteley, P.; Shattock, P.; Knivsberg, A.M.; Seim, A.; Reichelt, K.L.; Todd, L.; Carr, K.; Hooper, M. Gluten- and casein-free dietary intervention for autism spectrum conditions. Front. Hum. Neurosci. 2013, 6, 344. [Google Scholar] [CrossRef] [PubMed]
- Whiteley, P.; Haracopos, D.; Knivsberg, A.M.; Reichelt, K.L.; Parlar, S.; Jacobsen, J.; Seim, A.; Pedersen, L.; Schondel, M.; Shattock, P. The ScanBrit randomised, controlled, single-blind study of a gluten- and casein-free dietary intervention for children with autism spectrum disorders. Nutr. Neurosci. 2010, 13, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Knivsberg, A.M.; Reichelt, K.L.; Høien, T.; Nødland, M. A randomised, controlled study of dietary intervention in autistic syndromes. Nutr. Neurosci. 2002, 5, 251–261. [Google Scholar] [CrossRef]
- Jarmołowska, B.; Bukało, M.; Fiedorowicz, E.; Cieślińska, A.; Kordulewska, N.K.; Moszyńska, M.; Świątecki, A.; Kostyra, E. Role of Milk-Derived Opioid Peptides and Proline Dipeptidyl Peptidase-4 in Autism Spectrum Disorders. Nutrients 2019, 11, 87. [Google Scholar] [CrossRef]
- Schwartz, R.S. Autoimmune folate deficiency and the rise and fall of “horror autotoxicus”. N. Engl. J. Med. 2005, 352, 1948–1950. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Wynne, R.; Rose, S.; Slattery, J.; Delhey, L.; Tippett, M.; Kahler, S.G.; Bennuri, S.C.; Melnyk, S.; Sequeira, J.M.; et al. Thyroid dysfunction in children with autism spectrum disorder is associated with folate receptor α autoimmune disorder. J. Neuroendocrinol. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Quadros, E.V.; Sequeira, J.M.; Brown, W.T.; Mevs, C.; Marchi, E.; Flory, M.; Jenkins, E.C.; Velinov, M.T.; Cohen, I.L. Folate receptor autoantibodies are prevalent in childrendiagnosed with autism spectrum disorder, their normal siblings and parents. Autism Res. 2018, 11, 707–712. [Google Scholar] [CrossRef]
- Cabrera, R.M.; Shaw, G.M.; Ballard, J.L.; Carmichael, S.L.; Yang, W.; Lammer, E.J.; Finnell, R.H. Autoantibodies to folate receptor during pregnancy and neural tube defect risk. J. Reprod. Immunol. 2008, 79, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Boyles, A.L.; Ballard, J.L.; Gorman, E.B.; McConnaughey, D.R.; Cabrera, R.M.; Wilcox, A.J.; Lie, R.T.; Finnell, R.H. Association between inhibited binding of folic acid to folate receptor alpha in maternal serum and folate-related birth defects in Norway. Hum. Reprod. 2011, 26, 2232–2238. [Google Scholar] [CrossRef]
- Molloy, A.M.; Quadros, E.V.; Sequeira, J.M.; Troendle, J.F.; Scott, J.M.; Kirke, P.N.; Mills, J.L. Lack of association between folate-receptor autoantibodies and neural-tube defects. N. Engl. J. Med. 2009, 361, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Harel-Gadassi, A.; Friedlander, E.; Yaari, M.; Bar-Oz, B.; Eventov-Friedman, S.; Mankuta, D.; Yirmiya, N. Risk for ASD in Preterm Infants: A Three-Year Follow-Up Study. Autism Res. Treat. 2018, 2018, 8316212. [Google Scholar] [CrossRef] [PubMed]
| Protein | Gene | Chromosome | GPI Anchor? | Localization | Cofactors? | Refs. |
|---|---|---|---|---|---|---|
| FRα | FOLR1 | 11q13.3 | Yes | Liver, kidney, uterus, placenta, choroid plexus, retinal pigment epithelium | LRP2 | [33,35,36,37,38] |
| FRβ | FOLR2 | 11q13.4 | Yes | Placenta, spleen, bone marrow, thymus, macrophages | NA | [33,35,36,37,38,39] |
| FRγ | FOLR3 | 11q13.4 | NA | Secretory granules of neutrophil granulocytes | NA | [33,35,36,37,38,39] |
| FRδ | FOLR4 | 11q14 | Yes | Oocytes | NA | [33,35,36,37,38,39] |
| RFC | SLC19A1 | 21q22.3 | No | Liver, kidney, placenta, choroid plexus, intestinal tract | Vitamin D, thiamine pyrophosphate | [34,40] |
| PCFT | SLC46A1 | 17q11.2 | No | Liver, kidney, choroid plexus, placenta, intestinal epithelium, human tumors | Proton gradient | [34,40] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bobrowski-Khoury, N.; Ramaekers, V.T.; Sequeira, J.M.; Quadros, E.V. Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorders: Diagnosis, Treatment and Prevention. J. Pers. Med. 2021, 11, 710. https://doi.org/10.3390/jpm11080710
Bobrowski-Khoury N, Ramaekers VT, Sequeira JM, Quadros EV. Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorders: Diagnosis, Treatment and Prevention. Journal of Personalized Medicine. 2021; 11(8):710. https://doi.org/10.3390/jpm11080710
Chicago/Turabian StyleBobrowski-Khoury, Natasha, Vincent T. Ramaekers, Jeffrey M. Sequeira, and Edward V. Quadros. 2021. "Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorders: Diagnosis, Treatment and Prevention" Journal of Personalized Medicine 11, no. 8: 710. https://doi.org/10.3390/jpm11080710
APA StyleBobrowski-Khoury, N., Ramaekers, V. T., Sequeira, J. M., & Quadros, E. V. (2021). Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorders: Diagnosis, Treatment and Prevention. Journal of Personalized Medicine, 11(8), 710. https://doi.org/10.3390/jpm11080710





