Endometrial Perivascular Progenitor Cells and Uterus Regeneration
Abstract
1. Introduction
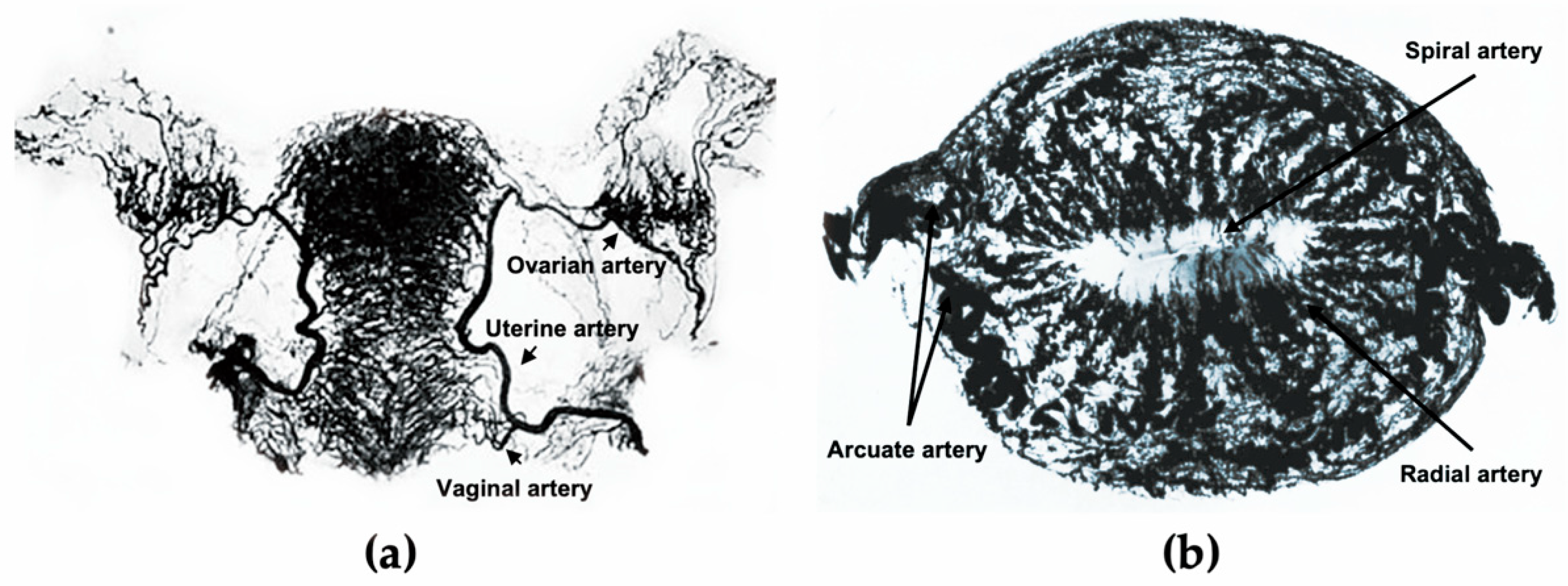
2. The Endometrial Vasculature
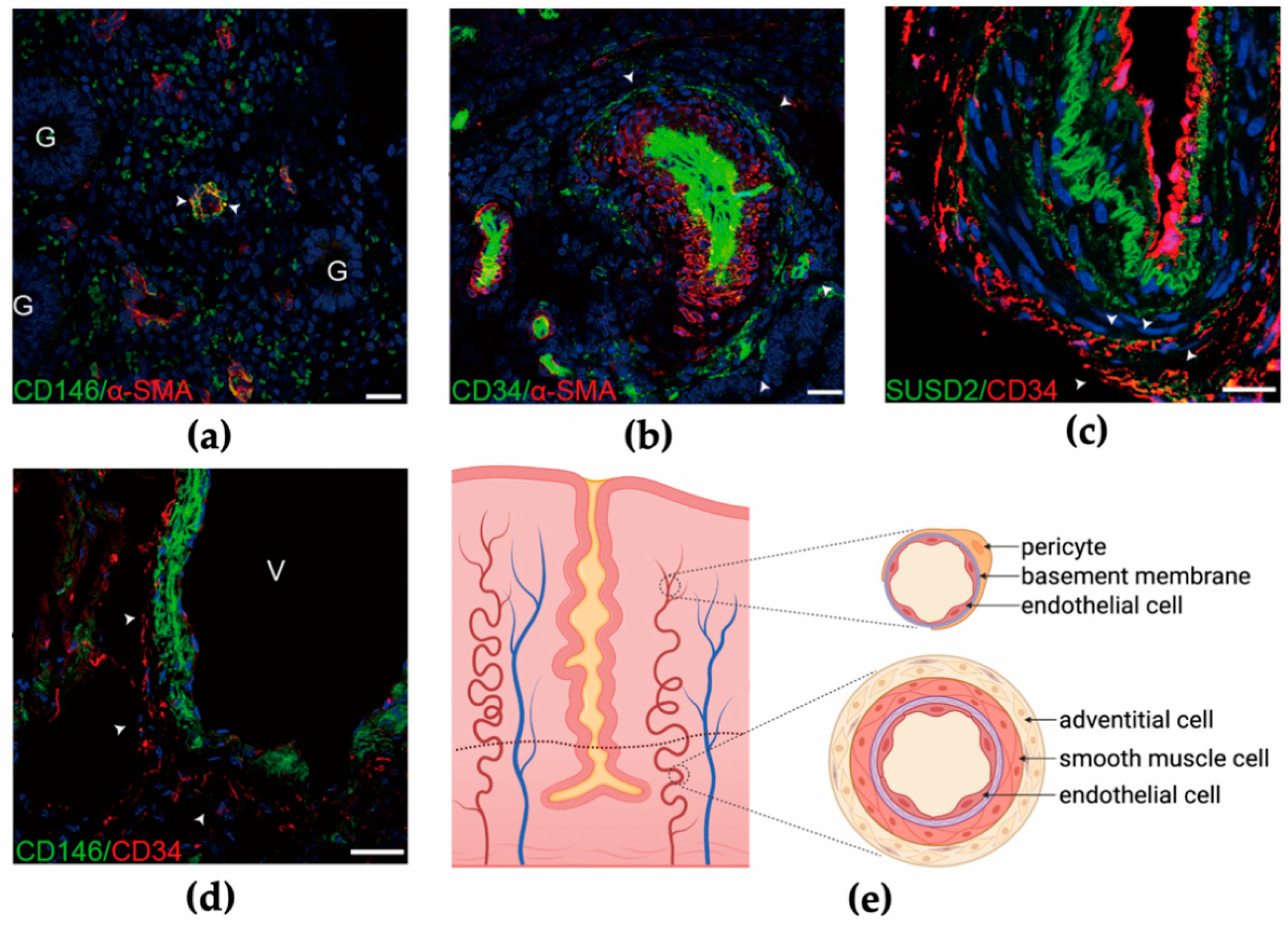
3. Endometrial Perivascular Cells as Native MSCs
4. Perivascular Cell-Based Angiogenetic Therapy for Asherman’s Syndrome (AS)
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Maybin, J.A.; Critchley, H.O.D. Menstrual Physiology: Implications for Endometrial Pathology and Beyond. Hum. Reprod. Update 2015, 21, 748–761. [Google Scholar] [CrossRef] [PubMed]
- Mihm, M.; Gangooly, S.; Muttukrishna, S. The Normal Menstrual Cycle in Women. Anim. Reprod. Sci. 2011, 124, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Samimi, M.; Pourhanifeh, M.H.; Mehdizadehkashi, A.; Eftekhar, T.; Asemi, Z. The Role of Inflammation, Oxidative Stress, Angiogenesis, and Apoptosis in the Pathophysiology of Endometriosis: Basic Science and New Insights Based on Gene Expression. J. Cell Physiol. 2019, 234, 19384–19392. [Google Scholar] [CrossRef] [PubMed]
- Buzzaccarini, G.; Vitagliano, A.; Andrisani, A.; Santarsiero, C.M.; Cicinelli, R.; Nardelli, C.; Ambrosini, G.; Cicinelli, E. Chronic Endometritis and Altered Embryo Implantation: A Unified Pathophysiological Theory from a Literature Systematic Review. J. Assist. Reprod Genet. 2020, 37, 2897–2911. [Google Scholar] [CrossRef]
- Gargett, C.E.; Chan, R.W.S.; Schwab, K.E. Endometrial Stem Cells. Curr. Opin. Obstet. Gynecol. 2007, 19, 377–383. [Google Scholar] [CrossRef]
- Friedler, S.; Margalioth, E.J.; Kafka, I.; Yaffe, H. Incidence of Post-Abortion Intra-Uterine Adhesions Evaluated by Hysteroscopy---A Prospective Study. Hum. Reprod 1993, 8, 442–444. [Google Scholar] [CrossRef]
- Badawy, S.Z.; Orbuch, L.; Khurana, K.K. Secondary Amenorrhea with Severe Intrauterine Adhesions and Chronic Uterine Torsion after Cesarean Section in a Teenage Girl. J. Pediatr. Adolesc. Gynecol. 1998, 11, 93–96. [Google Scholar] [CrossRef]
- Schenker, J.G.; Margalioth, E.J. Intrauterine Adhesions: An Updated Appraisal. Fertil. Steril. 1982, 37, 593–610. [Google Scholar] [CrossRef]
- Rabau, E.; David, A. Intrauterine adhesions: Etiology, prevention, and treatment. Obstet. Gynecol. 1963, 22, 626–629. [Google Scholar]
- Deane, J.A.; Gualano, R.C.; Gargett, C.E. Regenerating Endometrium from Stem/Progenitor Cells: Is It Abnormal in Endometriosis, Asherman’s Syndrome and Infertility? Curr. Opin. Obstet. Gynecol. 2013, 25, 193–200. [Google Scholar] [CrossRef]
- Arey, L.B. Developmental Anatomy: A Textbook and Laboratory Manual of Embryology; W. B. Saunders Company: Philadelphia, PA, USA, 1974; ISBN 978-0-7216-3443-2. [Google Scholar]
- Farrer-Brown, G.; Beilby, J.O.; Tarbit, M.H. The Blood Supply of the Uterus. 1. Arterial Vasculature. J. Obstet. Gynaecol. Br. Commonw. 1970, 77, 673–681. [Google Scholar] [CrossRef]
- Herbert, S.P.; Stainier, D.Y.R. Molecular Control of Endothelial Cell Behaviour during Blood Vessel Morphogenesis. Nat. Rev. Mol. Cell Biol. 2011, 12, 551–564. [Google Scholar] [CrossRef] [PubMed]
- Potente, M.; Mäkinen, T. Vascular Heterogeneity and Specialization in Development and Disease. Nat. Rev. Mol. Cell Biol. 2017, 18, 477–494. [Google Scholar] [CrossRef]
- Stratman, A.N.; Yu, J.A.; Mulligan, T.S.; Butler, M.G.; Sause, E.T.; Weinstein, B.M. Chapter 24—Blood Vessel Formation. In Principles of Developmental Genetics, 2nd ed.; Moody, S.A., Ed.; Academic Press: Oxford, UK, 2015; pp. 421–449. ISBN 978-0-12-405945-0. [Google Scholar]
- Folkman, J. Angiogenesis in Cancer, Vascular, Rheumatoid and Other Disease. Nat. Med. 1995, 1, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Flamme, I.; Frölich, T.; Risau, W. Molecular Mechanisms of Vasculogenesis and Embryonic Angiogenesis. J. Cell Physiol. 1997, 173, 206–210. [Google Scholar] [CrossRef]
- Bikfalvi, A. History and Conceptual Developments in Vascular Biology and Angiogenesis Research: A Personal View. Angiogenesis 2017, 20, 463–478. [Google Scholar] [CrossRef] [PubMed]
- Cicinelli, E.; Einer-Jensen, N.; Galantino, P.; Alfonso, R.; Nicoletti, R. The Vascular Cast of the Human Uterus: From Anatomy to Physiology. Ann. N. Y. Acad. Sci. 2004, 1034, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.K. Regulation of Angiogenesis in the Endometrium. Trends Endocrinol. Metab. 2001, 12, 147–151. [Google Scholar] [CrossRef]
- Gargett, C.E.; Rogers, P.A. Human Endometrial Angiogenesis. Reproduction 2001, 121, 181–186. [Google Scholar] [CrossRef]
- Risau, W. Mechanisms of Angiogenesis. Nature 1997, 386, 671–674. [Google Scholar] [CrossRef]
- Asahara, T.; Masuda, H.; Takahashi, T.; Kalka, C.; Pastore, C.; Silver, M.; Kearne, M.; Magner, M.; Isner, J.M. Bone Marrow Origin of Endothelial Progenitor Cells Responsible for Postnatal Vasculogenesis in Physiological and Pathological Neovascularization. Circ Res. 1999, 85, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Burri, P.H.; Djonov, V. Intussusceptive Angiogenesis--the Alternative to Capillary Sprouting. Mol. Asp. Med. 2002, 23, S1–S27. [Google Scholar] [CrossRef]
- Gambino, L.S.; Wreford, N.G.; Bertram, J.F.; Dockery, P.; Lederman, F.; Rogers, P.A.W. Angiogenesis Occurs by Vessel Elongation in Proliferative Phase Human Endometrium. Hum. Reprod. 2002, 17, 1199–1206. [Google Scholar] [CrossRef] [PubMed]
- Goodger, A.M.; Rogers, P.A. Endometrial Endothelial Cell Proliferation during the Menstrual Cycle. Hum. Reprod. 1994, 9, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Ferenczy, A.; Bertrand, G.; Gelfand, M.M. Proliferation Kinetics of Human Endometrium during the Normal Menstrual Cycle. Am. J. Obstet. Gynecol. 1979, 133, 859–867. [Google Scholar] [CrossRef]
- Rogers, P.A.; Lederman, F.; Taylor, N. Endometrial Microvascular Growth in Normal and Dysfunctional States. Hum. Reprod. Update 1998, 4, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Nayak, N.R.; Brenner, R.M. Vascular Proliferation and Vascular Endothelial Growth Factor Expression in the Rhesus Macaque Endometrium. J. Clin. Endocrinol. Metab. 2002, 87, 1845–1855. [Google Scholar] [CrossRef]
- Limbourg, A.; Korff, T.; Napp, L.C.; Schaper, W.; Drexler, H.; Limbourg, F.P. Evaluation of Postnatal Arteriogenesis and Angiogenesis in a Mouse Model of Hind-Limb Ischemia. Nat. Protoc. 2009, 4, 1737–1746. [Google Scholar] [CrossRef]
- Girling, J.E.; Lederman, F.L.; Walter, L.M.; Rogers, P.A.W. Progesterone, but Not Estrogen, Stimulates Vessel Maturation in the Mouse Endometrium. Endocrinology 2007, 148, 5433–5441. [Google Scholar] [CrossRef] [PubMed]
- Jabbour, H.N.; Kelly, R.W.; Fraser, H.M.; Critchley, H.O.D. Endocrine Regulation of Menstruation. Endocr. Rev. 2006, 27, 17–46. [Google Scholar] [CrossRef]
- Kooy, J.; Taylor, N.H.; Healy, D.L.; Rogers, P.A.W. Endothelial Cell Proliferation in the Endometrium of Women with Menorrhagia and in Women Following Endometrial Ablation. Hum. Reprod. 1996, 11, 1067–1072. [Google Scholar] [CrossRef]
- Nisolle, M.; Casanas-Roux, F.; Anaf, V.; Mine, J.M.; Donnez, J. Morphometric Study of the Stromal Vascularization in Peritoneal Endometriosis. Fertil. Steril. 1993, 59, 681–684. [Google Scholar] [CrossRef]
- Oosterlynck, D.J.; Meuleman, C.; Sobis, H.; Vandeputte, M.; Koninckx, P.R. Angiogenic Activity of Peritoneal Fluid from Women with Endometriosis. Fertil. Steril. 1993, 59, 778–782. [Google Scholar] [CrossRef]
- Jiang, P.; Tang, X.; Wang, H.; Dai, C.; Su, J.; Zhu, H.; Song, M.; Liu, J.; Nan, Z.; Ru, T.; et al. Collagen-Binding Basic Fibroblast Growth Factor Improves Functional Remodeling of Scarred Endometrium in Uterine Infertile Women: A Pilot Study. Sci. China Life Sci. 2019, 62, 1617–1629. [Google Scholar] [CrossRef] [PubMed]
- McLennan, C.E.; Rydell, A.H. Extent of Endometrial Shedding during Normal Menstruation. Obstet. Gynecol. 1965, 26, 605–621. [Google Scholar]
- Tempest, N.; Maclean, A.; Hapangama, D.K. Endometrial Stem Cell Markers: Current Concepts and Unresolved Questions. Int. J. Mol. Sci. 2018, 19, 3240. [Google Scholar] [CrossRef]
- Ferenczy, A. Studies on the Cytodynamics of Human Endometrial Regeneration. I. Scanning Electron Microscopy. Am. J. Obstet. Gynecol. 1976, 124, 64–74. [Google Scholar] [CrossRef]
- Padykula, H.A. Regeneration in the Primate Uterus: The Role of Stem Cells. Ann. N. Y. Acad. Sci. 1991, 622, 47–56. [Google Scholar] [CrossRef]
- Gurung, S.; Deane, J.; Masuda, H.; Maruyama, T.; Gargett, C. Stem Cells in Endometrial Physiology. Semin. Reprod. Med. 2015, 33, 326–332. [Google Scholar] [CrossRef]
- Prianishnikov, V.A. A Functional Model of the Structure of the Epithelium of Normal, Hyperplastic and Malignant Human Endometrium: A Review. Gynecol. Oncol. 1978, 6, 420–428. [Google Scholar] [CrossRef]
- Chan, R.W.S.; Schwab, K.E.; Gargett, C.E. Clonogenicity of Human Endometrial Epithelial and Stromal Cells. Biol. Reprod. 2004, 70, 1738–1750. [Google Scholar] [CrossRef] [PubMed]
- Gargett, C.E.; Schwab, K.E.; Zillwood, R.M.; Nguyen, H.P.T.; Wu, D. Isolation and Culture of Epithelial Progenitors and Mesenchymal Stem Cells from Human Endometrium. Biol. Reprod. 2009, 80, 1136–1145. [Google Scholar] [CrossRef] [PubMed]
- Schwab, K.E.; Gargett, C.E. Co-Expression of Two Perivascular Cell Markers Isolates Mesenchymal Stem-like Cells from Human Endometrium. Hum. Reprod. 2007, 22, 2903–2911. [Google Scholar] [CrossRef] [PubMed]
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.-W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A Perivascular Origin for Mesenchymal Stem Cells in Multiple Human Organs. Cell Stem. Cell 2008, 3, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Yu, F.; Yan, G.; Hu, Y.; Sun, H.; Ding, L. Human Endometrial Perivascular Stem Cells Exhibit a Limited Potential to Regenerate Endometrium after Xenotransplantation. Hum. Reprod. 2020. [Google Scholar] [CrossRef]
- Masuda, H.; Anwar, S.S.; Bühring, H.-J.; Rao, J.R.; Gargett, C.E. A Novel Marker of Human Endometrial Mesenchymal Stem-like Cells. Cell Transplant. 2012, 21, 2201–2214. [Google Scholar] [CrossRef]
- Betsholtz, C.; Lindblom, P.; Gerhardt, H. Role of Pericytes in Vascular Morphogenesis. In Mechanisms of Angiogenesis; Spring: Houston, TX, USA, 2005; pp. 115–125. [Google Scholar] [CrossRef]
- Dias, D.O.; Kim, H.; Holl, D.; Werne Solnestam, B.; Lundeberg, J.; Carlén, M.; Göritz, C.; Frisén, J. Reducing Pericyte-Derived Scarring Promotes Recovery after Spinal Cord Injury. Cell 2018, 173, 153–165.e22. [Google Scholar] [CrossRef]
- Crisan, M.; Corselli, M.; Chen, C.-W.; Péault, B. Multilineage Stem Cells in the Adult: A Perivascular Legacy? Organogenesis 2011, 7, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Spitzer, T.L.B.; Rojas, A.; Zelenko, Z.; Aghajanova, L.; Erikson, D.W.; Barragan, F.; Meyer, M.; Tamaresis, J.S.; Hamilton, A.E.; Irwin, J.C.; et al. Perivascular Human Endometrial Mesenchymal Stem Cells Express Pathways Relevant to Self-Renewal, Lineage Specification, and Functional Phenotype. Biol. Reprod. 2012, 86, 58. [Google Scholar] [CrossRef]
- Lehoux, S. Adventures in the Adventitia. Hypertension 2016, 67, 836–838. [Google Scholar] [CrossRef]
- Tinajero, M.G.; Gotlieb, A.I. Recent Developments in Vascular Adventitial Pathobiology: The Dynamic Adventitia as a Complex Regulator of Vascular Disease. Am. J. Pathol. 2020, 190, 520–534. [Google Scholar] [CrossRef] [PubMed]
- Kramann, R.; Goettsch, C.; Wongboonsin, J.; Iwata, H.; Schneider, R.K.; Kuppe, C.; Kaesler, N.; Chang-Panesso, M.; Machado, F.G.; Gratwohl, S.; et al. Adventitial MSC-like Cells Are Progenitors of Vascular Smooth Muscle Cells and Drive Vascular Calcification in Chronic Kidney Disease. Cell Stem. Cell 2016, 19, 628–642. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Zhang, Z.; Torsney, E.; Afzal, A.R.; Davison, F.; Metzler, B.; Xu, Q. Abundant Progenitor Cells in the Adventitia Contribute to Atherosclerosis of Vein Grafts in ApoE-Deficient Mice. J. Clin. Investig. 2004, 113, 1258–1265. [Google Scholar] [CrossRef]
- Corselli, M.; Chen, C.-W.; Sun, B.; Yap, S.; Rubin, J.P.; Péault, B. The Tunica Adventitia of Human Arteries and Veins As a Source of Mesenchymal Stem Cells. Stem Cells Dev. 2012, 21, 1299–1308. [Google Scholar] [CrossRef] [PubMed]
- Sartore, S.; Chiavegato, A.; Faggin, E.; Franch, R.; Puato, M.; Ausoni, S.; Pauletto, P. Contribution of Adventitial Fibroblasts to Neointima Formation and Vascular Remodeling: From Innocent Bystander to Active Participant. Circ Res. 2001, 89, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Chen, S.J.; Oparil, S.; Chen, Y.F.; Thompson, J.A. Direct in Vivo Evidence Demonstrating Neointimal Migration of Adventitial Fibroblasts after Balloon Injury of Rat Carotid Arteries. Circulation 2000, 101, 1362–1365. [Google Scholar] [CrossRef]
- Wilcox, J.N.; Scott, N.A. Potential Role of the Adventitia in Arteritis and Atherosclerosis. Int J. Cardiol. 1996, 54, S21–S35. [Google Scholar] [CrossRef]
- Cervelló, I.; Gil-Sanchis, C.; Santamaría, X.; Faus, A.; Vallvé-Juanico, J.; Díaz-Gimeno, P.; Genolet, O.; Pellicer, A.; Simón, C. Leucine-Rich Repeat-Containing G-Protein-Coupled Receptor 5-Positive Cells in the Endometrial Stem Cell Niche. Fertil. Steril. 2017, 107, 510–519. [Google Scholar] [CrossRef]
- Kato, K.; Yoshimoto, M.; Kato, K.; Adachi, S.; Yamayoshi, A.; Arima, T.; Asanoma, K.; Kyo, S.; Nakahata, T.; Wake, N. Characterization of Side-Population Cells in Human Normal Endometrium. Hum. Reprod. 2007, 22, 1214–1223. [Google Scholar] [CrossRef]
- Meng, X.; Ichim, T.E.; Zhong, J.; Rogers, A.; Yin, Z.; Jackson, J.; Wang, H.; Ge, W.; Bogin, V.; Chan, K.W.; et al. Endometrial Regenerative Cells: A Novel Stem Cell Population. J. Transl. Med. 2007, 5, 57. [Google Scholar] [CrossRef]
- Tal, R.; Liu, Y.; Pluchino, N.; Shaikh, S.; Mamillapalli, R.; Taylor, H.S. A Murine 5-Fluorouracil-Based Submyeloablation Model for the Study of Bone Marrow-Derived Cell Trafficking in Reproduction. Endocrinology 2016, 157, 3749–3759. [Google Scholar] [CrossRef]
- Deans, R.; Abbott, J. Review of Intrauterine Adhesions. J. Minim. Invasive Gynecol. 2010, 17, 555–569. [Google Scholar] [CrossRef]
- The American Fertility Society. Classifications of Adnexal Adhesions, Distal Tubal Occlusion, Tubal Occlusion Secondary to Tubal Ligation, Tubal Pregnancies, Müllerian Anomalies and Intrauterine Adhesions. Fertil. Steril. 1988, 49, 944–955. [Google Scholar] [CrossRef]
- Wamsteker, K.; De Block, S. Diagnostic hysteroscopy: Technique and documentation. In Endoscopic Surgery for Gynecologists; Sutton, C., Diamond, M., Eds.; WB Saunders: London, UK, 1998; pp. 511–524. [Google Scholar]
- Cao, M.; Pan, Y.; Zhang, Q.; You, D.; Feng, S.; Liu, Z. Predictive Value of Live Birth Rate Based on Different Intrauterine Adhesion Evaluation Systems Following TCRA. Reprod. Biol. Endocrinol. 2021, 19. [Google Scholar] [CrossRef]
- Yu, D.; Wong, Y.-M.; Cheong, Y.; Xia, E.; Li, T.-C. Asherman Syndrome—One Century Later. Fertil. Steril. 2008, 89, 759–779. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhang, H.; Wang, Q.; Xie, F.; Gao, S.; Song, Y.; Dong, J.; Feng, H.; Xie, K.; Sui, L. Reproductive Outcomes in Patients with Intrauterine Adhesions Following Hysteroscopic Adhesiolysis: Experience from the Largest Women’s Hospital in China. J. Minim. Invasive Gynecol. 2017, 24, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Gargett, C.E.; Ye, L. Endometrial Reconstruction from Stem Cells. Fertil. Steril. 2012, 98, 11–20. [Google Scholar] [CrossRef]
- Han, Y.; Li, X.; Zhang, Y.; Han, Y.; Chang, F.; Ding, J. Mesenchymal Stem Cells for Regenerative Medicine. Cells 2019, 8, 886. [Google Scholar] [CrossRef] [PubMed]
- Cervelló, I.; Gil-Sanchis, C.; Santamaría, X.; Cabanillas, S.; Díaz, A.; Faus, A.; Pellicer, A.; Simón, C. Human CD133+ Bone Marrow-Derived Stem Cells Promote Endometrial Proliferation in a Murine Model of Asherman Syndrome. Fertil. Steril. 2015, 104, 1552–1560. [Google Scholar] [CrossRef] [PubMed]
- Kilic, S.; Yuksel, B.; Pinarli, F.; Albayrak, A.; Boztok, B.; Delibasi, T. Effect of Stem Cell Application on Asherman Syndrome, an Experimental Rat Model. J. Assist. Reprod. Genet. 2014, 31, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, Y.; Guan, C.-Y.; Tian, S.; Lv, X.-D.; Li, J.-H.; Ma, X.; Xia, H.-F. Therapeutic Effect of Human Umbilical Cord-Derived Mesenchymal Stem Cells on Injured Rat Endometrium during Its Chronic Phase. Stem. Cell Res. Ther. 2018, 9, 36. [Google Scholar] [CrossRef]
- Yianni, V.; Sharpe, P.T. Perivascular-Derived Mesenchymal Stem Cells. J. Dent. Res. 2019, 98, 1066–1072. [Google Scholar] [CrossRef]
- Zhao, H.; Feng, J.; Seidel, K.; Shi, S.; Klein, O.; Sharpe, P.; Chai, Y. Secretion of Shh by a Neurovascular Bundle Niche Supports Mesenchymal Stem Cell Homeostasis in the Adult Mouse Incisor. Cell Stem. Cell 2014, 14, 160–173. [Google Scholar] [CrossRef]
- Caporarello, N.; D’Angeli, F.; Cambria, M.T.; Candido, S.; Giallongo, C.; Salmeri, M.; Lombardo, C.; Longo, A.; Giurdanella, G.; Anfuso, C.D.; et al. Pericytes in Microvessels: From “Mural” Function to Brain and Retina Regeneration. Int. J. Mol. Sci. 2019, 20, 6351. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, J.; Chang, L.; Meyers, C.A.; Zhang, L.; Broderick, K.; Lee, M.; Peault, B.; James, A.W. Relative Contributions of Adipose-Resident CD146+ Pericytes and CD34+ Adventitial Progenitor Cells in Bone Tissue Engineering. NPJ Regen Med. 2019, 4, 1. [Google Scholar] [CrossRef]
- Zheng, J.-H.; Zhang, J.-K.; Kong, D.-S.; Song, Y.-B.; Zhao, S.-D.; Qi, W.-B.; Li, Y.-N.; Zhang, M.; Huang, X.-H. Quantification of the CM-Dil-Labeled Human Umbilical Cord Mesenchymal Stem Cells Migrated to the Dual Injured Uterus in SD Rat. Stem. Cell Res. Ther. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Park, M.; Hong, S.-H.; Park, S.H.; Kim, Y.S.; Yang, S.C.; Kim, H.-R.; Noh, S.; Na, S.; Lee, H.K.; Lim, H.J.; et al. Perivascular Stem Cell-Derived Cyclophilin A Improves Uterine Environment with Asherman’s Syndrome via HIF1α-Dependent Angiogenesis. Mol. Ther. 2020, 28, 1818–1832. [Google Scholar] [CrossRef]
- Caplan, A.I. Why Are MSCs Therapeutic? New Data: New Insight. J. Pathol. 2009, 217, 318–324. [Google Scholar] [CrossRef]
- Zhang, Z.-S.; Zhou, H.-N.; He, S.-S.; Xue, M.-Y.; Li, T.; Liu, L.-M. Research Advances in Pericyte Function and Their Roles in Diseases. Chin. J. Traumatol. 2020, 23, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Hess, D.L.; Kelly-Goss, M.R.; Cherepanova, O.A.; Nguyen, A.T.; Baylis, R.A.; Tkachenko, S.; Annex, B.H.; Peirce, S.M.; Owens, G.K. Perivascular Cell-Specific Knockout of the Stem Cell Pluripotency Gene Oct4 Inhibits Angiogenesis. Nat. Commun. 2019, 10. [Google Scholar] [CrossRef]
- Caporali, A.; Martello, A.; Miscianinov, V.; Maselli, D.; Vono, R.; Spinetti, G. Contribution of Pericyte Paracrine Regulation of the Endothelium to Angiogenesis. Pharmacol. Ther. 2017, 171, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yan, G.; Diao, Q.; Yu, F.; Li, X.; Sheng, X.; Liu, Y.; Dai, Y.; Zhou, H.; Zhen, X.; et al. Transplantation of Human Endometrial Perivascular Cells with Elevated CYR61 Expression Induces Angiogenesis and Promotes Repair of a Full-Thickness Uterine Injury in Rat. Stem. Cell Res. Ther. 2019, 10, 179. [Google Scholar] [CrossRef] [PubMed]
- Le Blanc, K.; Tammik, C.; Rosendahl, K.; Zetterberg, E.; Ringdén, O. HLA Expression and Immunologic Properties of Differentiated and Undifferentiated Mesenchymal Stem Cells. Exp Hematol. 2003, 31, 890–896. [Google Scholar] [CrossRef]
- Yin, M.; Zhou, H.J.; Lin, C.; Long, L.; Yang, X.; Zhang, H.; Taylor, H.; Min, W. CD34+KLF4+ Stromal Stem Cells Contribute to Endometrial Regeneration and Repair. Cell Rep. 2019, 27, 2709–2724.e3. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Luo, Q.; Zhang, X.; Qin, Y.; Hao, J.; Kong, D.; Wang, H.; Li, G.; Gu, X.; Wang, H. Clinical Efficacy and Safety of Stem Cell-Based Therapy in Treating Asherman Syndrome: A System Review and Meta-Analysis. Stem. Cells Int. 2020, 2020. [Google Scholar] [CrossRef] [PubMed]
- Nagori, C.B.; Panchal, S.Y.; Patel, H. Endometrial Regeneration Using Autologous Adult Stem Cells Followed by Conception by In Vitro Fertilization in a Patient of Severe Asherman’s Syndrome. J. Hum. Reprod. Sci. 2011, 4, 43–48. [Google Scholar] [CrossRef]
- Cao, Y.; Sun, H.; Zhu, H.; Zhu, X.; Tang, X.; Yan, G.; Wang, J.; Bai, D.; Wang, J.; Wang, L.; et al. Allogeneic Cell Therapy Using Umbilical Cord MSCs on Collagen Scaffolds for Patients with Recurrent Uterine Adhesion: A Phase I Clinical Trial. Stem Cell Res. Ther. 2018, 9, 192. [Google Scholar] [CrossRef]
- Yang, Y.; Yu, F.; Ding, L.; Sheng, X.; Sun, H. Comparison of angiogenesis ability and transplantation safety among mesenchymal stem cells derived from umbilical artery, umbilical vein and Wharton’s jelly. Chin. J. Comp. Med. 2020, 30, 43–53. [Google Scholar] [CrossRef]
- Lee, K.; Xue, Y.; Lee, J.; Kim, H.-J.; Liu, Y.; Tebon, P.; Sarikhani, E.; Sun, W.; Zhang, S.; Haghniaz, R.; et al. A Patch of Detachable Hybrid Microneedle Depot for Localized Delivery of Mesenchymal Stem Cells in Regeneration Therapy. Adv. Funct. Mater. 2020, 30. [Google Scholar] [CrossRef]
- Murphy, W.L.; McDevitt, T.C.; Engler, A.J. Materials as Stem Cell Regulators. Nat. Mater. 2014, 13, 547–557. [Google Scholar] [CrossRef]
- Ding, L.; Li, X.; Sun, H.; Su, J.; Lin, N.; Péault, B.; Song, T.; Yang, J.; Dai, J.; Hu, Y. Transplantation of Bone Marrow Mesenchymal Stem Cells on Collagen Scaffolds for the Functional Regeneration of Injured Rat Uterus. Biomaterials 2014, 35, 4888–4900. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yu, C.; Chang, T.; Zhang, M.; Song, S.; Xiong, C.; Su, P.; Xiang, W. In Situ Repair Abilities of Human Umbilical Cord-Derived Mesenchymal Stem Cells and Autocrosslinked Hyaluronic Acid Gel Complex in Rhesus Monkeys with Intrauterine Adhesion. Sci. Adv. 2020, 6, eaba6357. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.; Hou, B.; Lin, W.; Wang, L.; Zheng, W.; Li, W.; Zheng, J.; Wen, X.; He, P. 3D Bioprinting a Human IPSC-Derived MSC-Loaded Scaffold for Repair of the Uterine Endometrium. Acta Biomater. 2020, 116, 268–284. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Ding, L. Endometrial Perivascular Progenitor Cells and Uterus Regeneration. J. Pers. Med. 2021, 11, 477. https://doi.org/10.3390/jpm11060477
Li S, Ding L. Endometrial Perivascular Progenitor Cells and Uterus Regeneration. Journal of Personalized Medicine. 2021; 11(6):477. https://doi.org/10.3390/jpm11060477
Chicago/Turabian StyleLi, Shiyuan, and Lijun Ding. 2021. "Endometrial Perivascular Progenitor Cells and Uterus Regeneration" Journal of Personalized Medicine 11, no. 6: 477. https://doi.org/10.3390/jpm11060477
APA StyleLi, S., & Ding, L. (2021). Endometrial Perivascular Progenitor Cells and Uterus Regeneration. Journal of Personalized Medicine, 11(6), 477. https://doi.org/10.3390/jpm11060477





