Forgetting Unwanted Memories: Active Forgetting and Implications for the Development of Psychological Disorders
Abstract
1. Introduction
2. Intentional and Incidental Forgetting in Post-Traumatic Stress Disorder
3. Intentional and Incidental Forgetting in Depression
4. Intentional and Incidental Forgetting in Schizophrenia
5. Intentional and Incidental Forgetting in Obsessive-Compulsive Disorder
6. Neurotransmitters and Active Forgetting
6.1. Glutamate
6.2. GABA
6.3. Acetylcholine
6.4. Dopamine
6.5. Noradrenaline
6.6. Glucocorticoids
6.7. Neurotransmitters, Inhibitory Control, and Active Forgetting: An Overview
7. Conclusions
Funding
Conflicts of Interest
References
- Brewin, C.R.; Gregory, J.D.; Lipton, M.; Burgess, N. Intrusive images in psychological disorders: Characteristics, neural mechanisms, and treatment implications. Psychol. Rev. 2010, 117, 210–232. [Google Scholar] [CrossRef]
- Brewin, C.R. Episodic memory, perceptual memory, and their interaction: Foundations for a theory of posttraumatic stress disorder. Psychol. Bull. 2014, 140, 69–97. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.B.; Watkins, E.; Mansell, W.; Shafran, R. Cognitive Behavioural Processes across Psychological Disorders: A Transdiagnostic Approach to Research and Treatment; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Hirsch, C.R.; Holmes, E.A. Mental imagery in anxiety disorders. Psychiatry 2007, 6, 161–165. [Google Scholar] [CrossRef]
- Speckens, A.E.M.; Ehlers, A.; Hackmann, A.; Ruths, F.A.; Clark, D.M. Intrusive memories and rumination in patients with post-traumatic stress disorder: A phenomenological comparison. Memory 2007, 15, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Visser, R.M. Why Do Certain Moments Haunt Us? Conceptualizing Intrusive Memories as Conditioned Responses. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2020, 5, 375–376. [Google Scholar] [CrossRef] [PubMed]
- Berntsen, D. Involuntary Autobiographical Memories; Cambridge University Press (CUP): Cambridge, UK, 2009. [Google Scholar]
- Mace, C. Mindfulness in psychotherapy: An introduction. Adv. Psychiatr. Treat. 2007, 13, 147–154. [Google Scholar] [CrossRef]
- Hu, X.; Bergström, Z.M.; Gagnepain, P.; Anderson, M.C. Suppressing Unwanted Memories Reduces Their Unintended Influences. Curr. Dir. Psychol. Sci. 2017, 26, 197–206. [Google Scholar] [CrossRef]
- Gagnepain, P.; Hulbert, J.; Anderson, M.C. Parallel Regulation of Memory and Emotion Supports the Suppression of Intrusive Memories. J. Neurosci. 2017, 37, 6423–6441. [Google Scholar] [CrossRef]
- Streb, M.; Mecklinger, A.; Anderson, M.C.; Lass-Hennemann, J.; Michael, T. Memory control ability modulates intrusive memories after analogue trauma. J. Affect. Disord. 2016, 192, 134–142. [Google Scholar] [CrossRef]
- Catarino, A.; Küpper, C.S.; Werner-Seidler, A.; Dalgleish, T.; Anderson, M.C. Failing to Forget. Psychol. Sci. 2015, 26, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, J.M.; Benoit, R.G.; Gagnepain, P.; Salman, A.; Bartholdy, S.; Bradley, C.; Chan, D.K.-Y.; Roche, A.; Brewin, C.R.; Anderson, M.C. The origins of repetitive thought in rumination: Separating cognitive style from deficits in inhibitory control over memory. J. Behav. Ther. Exp. Psychiatry 2015, 47, 1–8. [Google Scholar] [CrossRef]
- Hertel, P.T.; Maydon, A.; Ogilvie, A.; Mor, N. Ruminators (Unlike Others) Fail to Show Suppression-Induced Forgetting on Indirect Measures of Memory. Clin. Psychol. Sci. 2018, 6, 872–881. [Google Scholar] [CrossRef]
- Marzi, T.; Regina, A.; Righi, S. Emotions shape memory suppression in trait anxiety. Front. Psychol. 2014, 4, 1001. [Google Scholar] [CrossRef]
- Benoit, R.G.; Davies, D.J.; Anderson, M.C. Reducing future fears by suppressing the brain mechanisms underlying episodic simulation. Proc. Natl. Acad. Sci. USA 2016, 113, E8492–E8501. [Google Scholar] [CrossRef]
- Hertel, P.T.; Gerstle, M. Depressive Deficits in Forgetting. Psychol. Sci. 2003, 14, 573–578. [Google Scholar] [CrossRef]
- Noreen, S.; Ridout, N. Examining the impact of thought substitution on intentional forgetting in induced and naturally occurring dysphoria. Psychiatry Res. 2016, 241, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Noreen, S.; Ridout, N. Intentional forgetting in dysphoria: Investigating the inhibitory effects of thought substitution using independent cues. J. Behav. Ther. Exp. Psychiatry 2016, 52, 110–118. [Google Scholar] [CrossRef]
- Zhang, D.; Xie, H.; Liu, Y.; Luo, Y. Neural correlates underlying impaired memory facilitation and suppression of negative material in depression. Sci. Rep. 2016, 6, 37556. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.C.; Green, C. Suppressing unwanted memories by executive control. Nat. Cell Biol. 2001, 410, 366–369. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.L.; Zhong, Y. The Biology of Forgetting—A Perspective. Neuron 2017, 95, 490–503. [Google Scholar] [CrossRef]
- Medina, J.H. Neural, Cellular and Molecular Mechanisms of Active Forgetting. Front. Syst. Neurosci. 2018, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Hulbert, J.C.; Anderson, M.C. Does retrieving a memory insulate it against memory inhibition? A retroactive interference study. Memory 2020, 28, 293–308. [Google Scholar] [CrossRef] [PubMed]
- Erdelyi, M.H. The unified theory of repression. Behav. Brain Sci. 2006, 29, 499–511. [Google Scholar] [CrossRef]
- Nørby, S. Forgetting and emotion regulation in mental health, anxiety and depression. Memory 2018, 26, 342–363. [Google Scholar] [CrossRef]
- Schacter, D.L.; Bowers, J.; Booker, J. Intention, awareness, and implicit memory: The retrieval intentionality criterion. In Implicit Memory: Theoretical Issues; Lewandowsky, S., Dunn, J.C., Kirsner, K., Eds.; Lawrence Erlbaum Associates, Inc.: Mahwah, NJ, USA, 1989; pp. 47–65. [Google Scholar]
- Otgaar, H.; Howe, M.L.; Patihis, L.; Merckelbach, H.; Lynn, S.J.; Lilienfeld, S.O.; Loftus, E.F. The Return of the Repressed: The Persistent and Problematic Claims of Long-Forgotten Trauma. Perspect. Psychol. Sci. 2019, 14, 1072–1095. [Google Scholar] [CrossRef]
- Davis, P.J.; Schwartz, G.E. Repression and the inaccessibility of affective memories. J. Pers. Soc. Psychol. 1987, 52, 155–162. [Google Scholar] [CrossRef]
- Anderson, M. Rethinking interference theory: Executive control and the mechanisms of forgetting. J. Mem. Lang. 2003, 49, 415–445. [Google Scholar] [CrossRef]
- Anderson, M.C.; Levy, B.J. Theoretical issues in inhibition: Insights from research on human memory. In Inhibition in Cognition; American Psychological Association (APA): Washington, DC, USA, 2008; pp. 81–102. [Google Scholar]
- Raaijmakers, J.G.W. Inhibition in Memory. In Stevens’ Handbook of Experimental Psychology and Cognitive Neuroscience; Wiley: Hoboken, NJ, USA, 2018; pp. 1–34. [Google Scholar]
- Raaijmakers, J.G.W. Inhibition in memory. In Stevens’ Handbook of Experimental Psychology and Cognitive Neuroscience, Language and Memory, 4th ed.; Phelps, E.A., Davachi, L., Wixted, J.T., Eds.; Wiley: Hoboken, NJ, USA, 2018; Volume 1, pp. 251–284. [Google Scholar] [CrossRef]
- Murayama, K.; Miyatsu, T.; Buchli, D.; Storm, B.C. Forgetting as a consequence of retrieval: A meta-analytic review of retrieval-induced forgetting. Psychol. Bull. 2014, 140, 1383–1409. [Google Scholar] [CrossRef]
- Anderson, M.C.; Hanslmayr, S. Neural mechanisms of motivated forgetting. Trends Cogn. Sci. 2014, 18, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Geraerts, E.; McNally, J.R. Forgetting unwanted memories: Directed forgetting and thought suppression methods. Acta Psychol. 2008, 127, 614–622. [Google Scholar] [CrossRef] [PubMed]
- Sahakyan, L.; Delaney, P.F.; Nathaniel, L.; Abushanab, B. List-Method Directed Forgetting in Cognitive and Clinical Research: A Theoretical and Methodological Review. Psychol. Learn. Motiv. 2013, 59, 131–189. [Google Scholar]
- Bjork, R.A. Theoretical implication of directed forgetting. In Coding Processes in Human Memory; Melton, A.W., Martin, E., Eds.; Winston & Sons, Inc.: Washington, DC, USA, 1972; pp. 217–235. [Google Scholar]
- Sahakyan, L.; Delaney, P.F. Can encoding differences explain the benefits of directed forgetting in the list method paradigm? J. Mem. Lang. 2003, 48, 195–206. [Google Scholar] [CrossRef]
- Sahakyan, L.; Kelley, C.M. A contextual change account of the directed forgetting effect. J. Exp. Psychol. Learn. Mem. Cogn. 2002, 28, 1064–1072. [Google Scholar] [CrossRef] [PubMed]
- Rizio, A.A.; Dennis, N.A. The Neural Correlates of Cognitive Control: Successful Remembering and Intentional Forgetting. J. Cogn. Neurosci. 2013, 25, 297–312. [Google Scholar] [CrossRef] [PubMed]
- Hanslmayr, S.; Volberg, G.; Wimber, M.; Oehler, N.; Staudigl, T.; Hartmann, T.; Raabe, M.; Greenlee, M.W.; Bäuml, K.-H.T. Prefrontally Driven Downregulation of Neural Synchrony Mediates Goal-Directed Forgetting. J. Neurosci. 2012, 32, 14742–14751. [Google Scholar] [CrossRef]
- Murray, B.D.; Muscatell, K.A.; Kensinger, E.A. Effects of emotion and age on performance during a think/no-think memory task. Psychol. Aging 2011, 26, 940–955. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Yi, D.-J. Out of Mind, Out of Sight. Psychol. Sci. 2013, 24, 569–574. [Google Scholar] [CrossRef]
- Spataro, P.; Saraulli, D.; Cestari, V.; Costanzi, M.; Sciarretta, A.; Rossi-Arnaud, C. Implicit memory in schizophrenia: A meta-analysis. Compr. Psychiatry 2016, 69, 136–144. [Google Scholar] [CrossRef]
- Kang, E.; Wen, Z.; Song, H.; Christian, K.M.; Ming, G.-L. Adult Neurogenesis and Psychiatric Disorders. Cold Spring Harb. Perspect. Biol. 2016, 8, a019026. [Google Scholar] [CrossRef]
- Richards, B.A.; Frankland, P.W. The Persistence and Transience of Memory. Neuron 2017, 94, 1071–1084. [Google Scholar] [CrossRef]
- Liu, Y.; Lin, W.; Liu, C.; Luo, Y.; Wu, J.; Bayley, P.J.; Qin, S. Memory consolidation reconfigures neural pathways involved in the suppression of emotional memories. Nat. Commun. 2016, 7, 13375. [Google Scholar] [CrossRef] [PubMed]
- Nader, K.; Hardt, O.; Lanius, R. Memory as a new therapeutic target. Dialog Clin. Neurosci. 2013, 15, 475–486. [Google Scholar]
- Choudhury, A.; Sahu, T.; Ramanujam, P.L.; Banerjee, A.K.; Chakraborty, I.; Kumar, A.; Arora, N. Neurochemicals, Behaviours and Psychiatric Perspectives of Neurological Diseases. Neuropsychiatry 2018, 8, 395–424. [Google Scholar] [CrossRef]
- Kalueff, A.V.; Nutt, D.J. Role of GABA in anxiety and depression. Depress. Anxiety 2006, 24, 495–517. [Google Scholar] [CrossRef] [PubMed]
- Kaouane, N.; Porte, Y.; Vallée, M.; Brayda-Bruno, L.; Mons, N.; Calandreau, L.; Marighetto, A.; Piazza, P.V.; Desmedt, A. Glucocorticoids Can Induce PTSD-Like Memory Impairments in Mice. Science 2012, 335, 1510–1513. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, I.; Nuss, P. Anxiety disorders and GABA neurotransmission: A disturbance of modulation. Neuropsychiatr. Dis. Treat. 2015, 11, 165–175. [Google Scholar] [CrossRef]
- Poels, E.M.; Kegeles, L.S.; Kantrowitz, J.T.; Javitt, D.C.; Lieberman, J.A.; Abi-Dargham, A.; Girgis, R.R. Glutamatergic abnormalities in schizophrenia: A review of proton MRS findings. Schizophr. Res. 2014, 152, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Zheng, P.; Zeng, B.; Liu, M.; Chen, J.; Pan, J.; Han, Y.; Liu, Y.; Cheng, K.; Zhou, C.; Wang, H.; et al. The gut microbiome from patients with schizophrenia modulates the glutamate-glutamine-GABA cycle and schizophrenia-relevant behaviors in mice. Sci. Adv. 2019, 5, eaau8317. [Google Scholar] [CrossRef]
- Castellano, C.; Cabib, S.; Zocchi, A.; Puglisi-Allegra, S. Strain-dependent effects of cocaine on memory storage improvement induced by post-training physostigmine. Psychopharmacology 1996, 123, 340–345. [Google Scholar] [CrossRef]
- Furini, C.R.; Myskiw, J.C.; Benetti, F.; Izquierdo, I. New frontiers in the study of memory mechanisms. Rev. Bras. Psiquiatr. 2013, 35, 173–177. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gold, J.J.; Squire, L.R. Quantifying medial temporal lobe damage in memory-impaired patients. Hippocampus 2004, 15, 79–85. [Google Scholar] [CrossRef][Green Version]
- Meneses, A. Frameworking memory and serotonergic markers. Rev. Neurosci. 2017, 28, 455–497. [Google Scholar] [CrossRef] [PubMed]
- Ridley, R.; Baker, H.; Harder, J.; Pearson, C. Effects of lesions of different parts of the septo-hippocampal system in primates on learning and retention of information acquired before or after surgery. Brain Res. Bull. 1996, 40, 21–32. [Google Scholar] [CrossRef]
- Tellez, R.; Gomez-Viquez, L.; Meneses, A. GABA, glutamate, dopamine and serotonin transporters expression on memory formation and amnesia. Neurobiol. Learn. Mem. 2012, 97, 189–201. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; (DSM-5); American Psychiatric Association: Washington, DC, USA, 2013; p. 5. [Google Scholar]
- Ehlers, A. Understanding and Treating Unwanted Trauma Memories in Posttraumatic Stress Disorder. Z. Psychol. J. Psychol. 2010, 218, 141–145. [Google Scholar] [CrossRef] [PubMed]
- Millon, E.M.; Chang, H.Y.M.; Shors, T.J. Stressful Life Memories Relate to Ruminative Thoughts in Women with Sexual Violence History, Irrespective of PTSD. Front. Psychiatry 2018, 9, 311. [Google Scholar] [CrossRef] [PubMed]
- Brewin, C.R. The Nature and Significance of Memory Disturbance in Posttraumatic Stress Disorder. Annu. Rev. Clin. Psychol. 2011, 7, 203–227. [Google Scholar] [CrossRef]
- Brewin, C.R. Memory and Forgetting. Curr. Psychiatry Rep. 2018, 20, 1–8. [Google Scholar] [CrossRef]
- McNally, R.J. Experimental approaches to cognitive abnormality in posttraumatic stress disorder. Clin. Psychol. Rev. 1998, 18, 971–982. [Google Scholar] [CrossRef]
- Levy, B.J.; Anderson, M.C. Individual differences in the suppression of unwanted memories: The executive deficit hypothesis. Acta Psychol. 2008, 127, 623–635. [Google Scholar] [CrossRef]
- El Khoury-Malhame, M.; Lanteaume, L.; Beetz, E.M.; Roques, J.; Reynaud, E.; Samuelian, J.-C.; Blin, O.; Garcia, R.; Khalfa, S. Attentional bias in post-traumatic stress disorder diminishes after symptom amelioration. Behav. Res. Ther. 2011, 49, 796–801. [Google Scholar] [CrossRef] [PubMed]
- Fleurkens, P.; Rinck, M.; Van Minnen, A. Specificity and generalization of attentional bias in sexual trauma victims suffering from posttraumatic stress disorder. J. Anxiety Disord. 2011, 25, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Paunovic, N.; Lundh, L.-G.; Öst, L.-G. Attentional and memory bias for emotional information in crime victims with acute posttraumatic stress disorder (PTSD). J. Anxiety Disord. 2002, 16, 675–692. [Google Scholar] [CrossRef]
- Küpper, C.S.; Benoit, R.G.; Dalgleish, T.; Anderson, M.C. Direct suppression as a mechanism for controlling unpleasant memories in daily life. J. Exp. Psychol. Gen. 2014, 143, 1443–1449. [Google Scholar] [CrossRef]
- Baumann, M.; Zwissler, B.; Schalinski, I.; Ruf-Leuschner, M.; Schauer, M.; Kissler, J.M. Directed Forgetting in Post-Traumatic-Stress-Disorder: A Study of Refugee Immigrants in Germany. Front. Behav. Neurosci. 2013, 7, 94. [Google Scholar] [CrossRef]
- Blix, I.; Brennen, T. Intentional Forgetting of Emotional Words after Trauma: A Study with Victims of Sexual Assault. Front. Psychol. 2011, 2, 235. [Google Scholar] [CrossRef]
- Cottencin, O.; Vaiva, G.; Huron, C.; Devos, P.; Ducrocq, F.; Jouvent, R.; Goudemand, M.; Thomas, P. Directed forgetting in PTSD: A comparative study versus normal controls. J. Psychiatr. Res. 2006, 40, 70–80. [Google Scholar] [CrossRef] [PubMed]
- DePrince, A.P.; Freyd, J.J. Forgetting Trauma Stimuli. Psychol. Sci. 2004, 15, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Mary, A.; Dayan, J.; Leone, G.; Postel, C.; Fraisse, F.; Malle, C.; Vallée, T.; Klein-Peschanski, C.; Viader, F.; De La Sayette, V.; et al. Resilience after trauma: The role of memory suppression. Science 2020, 367, eaay8477. [Google Scholar] [CrossRef] [PubMed]
- Tudorache, A.-C.; El-Hage, W.; Tapia, G.; Goutaudier, N.; Kalenzaga, S.; Bouazzaoui, B.; Jaafari, N.; Clarys, D. Inhibitory control of threat remembering in PTSD. Memory 2019, 27, 1404–1414. [Google Scholar] [CrossRef]
- Waldhauser, G.T.; Dahl, M.J.; Ruf-Leuschner, M.; Müller-Bamouh, V.; Schauer, M.; Axmacher, N.; Elbert, T.; Hanslmayr, S. The neural dynamics of deficient memory control in heavily traumatized refugees. Sci. Rep. 2018, 8, 13132. [Google Scholar] [CrossRef] [PubMed]
- Van Schie, K.; Anderson, M.C. Successfully controlling intrusive memories is harder when control must be sustained. Memory 2017, 25, 1201–1216. [Google Scholar] [CrossRef] [PubMed]
- Zoellner, L.A.; Sacks, M.B.; Foa, E.B. Directed forgetting following mood induction in chronic posttraumatic stress disorder patients. J. Abnorm. Psychol. 2003, 112, 508–514. [Google Scholar] [CrossRef]
- Sauseng, P.; Klimesch, W.; Doppelmayr, M.; Pecherstorfer, T.; Freunberger, R.; Hanslmayr, S. EEG alpha synchronization and functional coupling during top-down processing in a working memory task. Hum. Brain Mapp. 2005, 26, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Cook, F.; Ciorciari, J.; Varker, T.; Devilly, G.J. Changes in long term neural connectivity following psychological trauma. Clin. Neurophysiol. 2009, 120, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Amir, N.; Taylor, C.T.; Bomyea, J.A.; Badour, C.L. Temporal allocation of attention toward threat in individuals with posttraumatic stress symptoms. J. Anxiety Disord. 2009, 23, 1080–1085. [Google Scholar] [CrossRef]
- Payne, A.; Kralj, A.; Young, J.; Meiser-Stedman, R. The prevalence of intrusive memories in adult depres- sion: A meta-analysis. J. Affect Diosord. 2019, 53, 193–202. [Google Scholar] [CrossRef]
- Power, M.; Dalgleish, T.; Claudio, V.; Tata, P.; Kentish, J. The directed forgetting task: Application to emotionally valent material. J. Affect. Disord. 2000, 57, 147–157. [Google Scholar] [CrossRef]
- Wingenfeld, K.; Terfehr, K.; Meyer, B.; Löwe, B.; Spitzer, C. Memory Bias for Emotional and Illness-Related Words in Patients with Depression, Anxiety and Somatization Disorders: An Investigation with the Directed Forgetting Task. Psychopathology 2013, 46, 22–27. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Manual for the Beck Depression Inventory–II; Psychological Corporation: San Antonio, TX, USA, 1996. [Google Scholar]
- Xie, H.; Jiang, D.; Zhang, D. Individuals with depressive tendencies experience difficulty in forgetting negative material: Two mechanisms revealed by ERP data in the directed forgetting paradigm. Sci. Rep. 2018, 8, 1–14. [Google Scholar] [CrossRef]
- Kuehl, L.K.; Wolf, O.T.; Driessen, M.; Schlosser, N.; Fernando, S.C.; Wingenfeld, K. Effects of cortisol on the memory bias for emotional words? A study in patients with depression and healthy participants using the Directed Forgetting task. J. Psychiatr. Res. 2017, 92, 191–198. [Google Scholar] [CrossRef]
- Groome, D.; Sterkaj, F. Retrieval-induced forgetting and clinical depression. Cogn. Emot. 2010, 24, 63–70. [Google Scholar] [CrossRef]
- Sacchet, M.D.; Levy, B.J.; Hamilton, J.P.; Maksimovskiy, A.; Hertel, P.T.; Joormann, J.; Anderson, M.C.; Wagner, A.D.; Gotlib, I.H. Cognitive and neural consequences of memory suppression in major depressive disorder. Cogn. Affect. Behav. Neurosci. 2017, 17, 77–93. [Google Scholar] [CrossRef]
- Noreen, S.; Cooke, R.; Ridout, N. Investigating the mediating effect of working memory on intentional forgetting in dysphoria. Psychol. Res. 2020, 84, 2273–2286. [Google Scholar] [CrossRef]
- Anticevic, A.; Repovs, G.; Corlett, P.R.; Barch, D.M. Negative and Nonemotional Interference with Visual Working Memory in Schizophrenia. Biol. Psychiatry 2011, 70, 1159–1168. [Google Scholar] [CrossRef]
- Diaz, M.T.; He, G.; Gadde, S.; Bellion, C.; Belger, A.; Voyvodic, J.T.; McCarthy, G. The influence of emotional distraction on verbal working memory: An fMRI investigation comparing individuals with schizophrenia and healthy adults. J. Psychiatr. Res. 2011, 45, 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- Horan, W.P.; Hajcak, G.; Wynn, J.K.; Green, M.F. Impaired emotion regulation in schizophrenia: Evidence from event-related potentials. Psychol. Med. 2013, 43, 2377–2391. [Google Scholar] [CrossRef] [PubMed]
- Strauss, G.P.; Kappenman, E.S.; Culbreth, A.J.; Catalano, L.T.; Ossenfort, K.L.; Lee, B.G.; Gold, J.M. Emotion regulation abnormalities in schizophrenia: Directed attention strategies fail to decrease the neurophysiological response to unpleasant stimuli. J. Abnorm. Psychol. 2015, 124, 288–301. [Google Scholar] [CrossRef]
- Brébion, G.; Gorman, J.M.; Malaspina, D.; Amador, X. A model of verbal memory impairments in schizophrenia: Two systems and their associations with underlying cognitive processes and clinical symptoms. Psychol. Med. 1999, 35, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Badcock, J.C.; Mahfouda, S.; Maybery, M.T. Hallucinations and inhibitory functioning in healthy young adults with high and low levels of hypomanic personality traits. Cogn. Neuropsychiatry 2015, 20, 254–269. [Google Scholar] [CrossRef] [PubMed]
- Brébion, G.; Stephan-Otto, C.; Ochoa, S.; Roca, M.; Nieto, L.; Usall, J. Impaired Self-Monitoring of Inner Speech in Schizophrenia Patients with Verbal Hallucinations and in Non-clinical Individuals Prone to Hallucinations. Front. Psychol. 2016, 7, 1381. [Google Scholar] [CrossRef]
- Müller, U.; Ullsperger, M.; Hammerstein, E.; Sachweh, S.; Becker, T. Directed forgetting in schizophrenia. Eur. Arch. Psychiatry Clin. Neurosci. 2004, 255, 251–257. [Google Scholar] [CrossRef]
- Patrick, R.E.; Kiang, M.; Christensen, B.K. Neurophysiological correlates of emotional directed-forgetting in persons with Schizophrenia: An event-related brain potential study. Int. J. Psychophysiol. 2015, 98, 612–623. [Google Scholar] [CrossRef] [PubMed]
- Patrick, R.E.; Christensen, B.K. Reduced directed forgetting for negative words suggests schizophrenia-related disinhibition of emotional cues. Psychol. Med. 2013, 43, 2289–2299. [Google Scholar] [CrossRef]
- Racsmány, M.; Conway, M.A.; Garab, E.A.; Cimmer, C.; Janka, Z.; Kurimay, T.; Pléh, C.; Szendi, I. Disrupted memory inhibition in schizophrenia. Schizophr. Res. 2008, 101, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Sahakyan, L.; Kwapil, T.R.; Jiang, L. Differential impairment of positive and negative schizotypy in list-method and item-method directed forgetting. J. Exp. Psychol. Gen. 2020, 149, 368–381. [Google Scholar] [CrossRef]
- Alderson-Day, B.; Smailes, D.; Moffatt, J.; Mitrenga, K.; Moseley, P.; Fernyhough, C. Intentional inhibition but not source memory is related to hallucination-proneness and intrusive thoughts in a university sample. Cortex 2019, 113, 267–278. [Google Scholar] [CrossRef]
- Debbané, M.; Glaser, B.; Eliez, S. Encoding and retrieval processes in velo-cardio-facial syndrome (VCFS). Neuropsychology 2008, 22, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Okruszek, Ł.; Jarkiewicz, M.; Szrubarz, P.; Wichniak, A.; Michałowski, J.; Marchewka, A.; Łojek, E. Many ways to forget—Neurophysiology of directed forgetting mechanisms in schizophrenia. Psychiatry Res. 2019, 274, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Nestor, P.G.; Piech, R.; Allen, C.; Niznikiewicz, M.; Shenton, M.; McCarley, R.W. Retrieval-induced forgetting in schizophrenia. Schizophr. Res. 2005, 75, 199–209. [Google Scholar] [CrossRef] [PubMed]
- AhnAllen, C.G.; Nestor, P.G.; McCarley, R.W.; Shenton, M.E. The role of retrieval inhibition in the associative memory impairment of schizophrenia. Psychiatry Res. 2007, 150, 43–50. [Google Scholar] [CrossRef]
- Soriano, M.F.; Jimenez, J.F.; Roman, P.; Bajo, M.T. Inhibitory processes in memory are impaired in schizophrenia: Evidence from retrieval induced forgetting. Br. J. Psychol. 2009, 100, 661–673. [Google Scholar] [CrossRef]
- Benzina, N.; Mallet, L.; Burguière, E.; N’Diaye, K.; Pelissolo, A. Cognitive Dysfunction in Obsessive-Compulsive Disorder. Curr. Psychiatry Rep. 2016, 18, 1–11. [Google Scholar] [CrossRef]
- Fullana, M.A.; Abramovitch, A.; Via, E.; López-Sola, C.; Goldberg, X.; Reina, N.; Fortea, L.; Solanes, A.; Buckley, M.J.; Ramella-Cravaro, V.; et al. Diagnostic biomarkers for obsessive-compulsive disorder: A reasonable quest or ignis fatuus? Neurosci. Biobehav. Rev. 2020, 118, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Lipton, M.G.; Brewin, C.R.; Linke, S.; Halperin, J. Distinguishing features of intrusive images in obsessive–compulsive disorder. J. Anxiety Disord. 2010, 24, 816–822. [Google Scholar] [CrossRef]
- Cavedini, P.; Zorzi, C.; Piccinni, M.; Cavallini, M.C.; Bellodi, L. Executive Dysfunctions in Obsessive-Compulsive Patients and Unaffected Relatives: Searching for a New Intermediate Phenotype. Biol. Psychiatry 2010, 67, 1178–1184. [Google Scholar] [CrossRef]
- Demeter, G.; Keresztes, A.; Harsanyi, A.; Csigò, K. Obsessed not to forget: Lack of retrieval-induced sup- pression effect in obsessive-compulsive disorder. Psychiatry Res. 2014. [Google Scholar] [CrossRef]
- Olley, A.; Malhi, G.; Sachdev, P. Memory and executive functioning in obsessive–compulsive disorder: A selective review. J. Affect. Disord. 2007, 104, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Rao, N.P.; Reddy, Y.J.; Kumar, K.J.; Kandavel, T.; Chandrashekar, C. Are neuropsychological deficits trait markers in OCD? Prog. Neuro Psychopharmacol. Biol. Psychiatry 2008, 32, 1574–1579. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, S.; Blackwell, A.; Fineberg, N.; Robbins, T.; Sahakian, B. The neuropsychology of obsessive compulsive disorder: The importance of failures in cognitive and behavioural inhibition as candidate endophenotypic markers. Neurosci. Biobehav. Rev. 2005, 29, 399–419. [Google Scholar] [CrossRef]
- Ewood, J.; Ahmari, S.E. A Framework for Understanding the Emerging Role of Corticolimbic-Ventral Striatal Networks in OCD-Associated Repetitive Behaviors. Front. Syst. Neurosci. 2015, 9, 171. [Google Scholar] [CrossRef]
- Wilhelm, S.; McNally, R.J.; Baer, L.; Florin, I. Directed forgetting in obsessive-compulsive disorder. Behav. Res. Ther. 1996, 34, 633–641. [Google Scholar] [CrossRef]
- Bohne, A.; Savage, C.R.; Deckersbach, T.; Keuthen, N.J.; Jenike, M.A.; Tuschen-Caffier, B.; Wilhelm, S. Visuospatial Abilities, Memory, and Executive Functioning in Trichotillomania and Obsessive-Compulsive Disorder. J. Clin. Exp. Neuropsychol. 2005, 27, 385–399. [Google Scholar] [CrossRef]
- Tolin, D.F.; Hamlin, C.; Foa, E.B. Directed forgetting in obsessive-compulsive disorder: Replication and extension. Behav. Res. Ther. 2002, 40, 793–803. [Google Scholar] [CrossRef]
- Moritz, S.; Hottenrott, B.; Jelinek, L.; Brooks, A.M.; Scheurich, A. Effects of Obsessive-Compulsive Symptoms on Neuropsychological Test Performance: Complicating an Already Complicated Story. Clin. Neuropsychol. 2012, 26, 31–44. [Google Scholar] [CrossRef]
- Konishi, M.; Shishikura, K.; Nakaaki, S.; Komatsu, S.-I.; Mimura, M. Remembering and forgetting: Directed forgetting effect in obsessive-compulsive disorder. Neuropsychiatr. Dis. Treat. 2011, 7, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Jelinek, L.; Rietschel, L.; Kellner, M.; Muhtz, C.; Moritz, S. The effect of practice on the recall of salient information in obsessive–compulsive disorder. Psychiatry Res. 2012, 198, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Niciu, M.J.; Kelmendi, B.; Sanacora, G. Overview of glutamatergic neurotransmission in the nervous system. Pharmacol. Biochem. Behav. 2012, 100, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Castellano, C.; Cestari, V.; Ciamei, A. NMDA receptors and learning and memory processes. Curr. Drug Targets 2001, 2, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Rezvani, A.H.; Levin, E.D.; Buccafusco, J.J. Involvement of the NMDA System in Learning and Memory; CRC Press/Taylor & Francis: Abingdon, UK, 2006; Chapter 4. [Google Scholar]
- Woodside, B.; Borroni, A.; Hammonds, M.; Teyler, T. NMDA receptors and voltage-dependent calcium channels mediate different aspects of acquisition and retention of a spatial memory task. Neurobiol. Learn. Mem. 2004, 81, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Walker, D.L.; Davis, M. Involvement of NMDA receptors within the amygdala in short- versus long-term memory for fear condiDoning as assessed with fear-potenDated startle. Behav. Neurosci. 2000, 114, 1019–1033. [Google Scholar] [CrossRef] [PubMed]
- Quartermain, D.; Nuygen, T.; Sheu, J.; Herting, R.L. Milacemide enhances memory storage and alleviates spontaneous forgetting in mice. Pharmacol. Biochem. Behav. 1991, 39, 31–35. [Google Scholar] [CrossRef]
- Sachser, R.M.; Santana, F.; Crestani, A.P.; Lunardi, P.; Pedraza, L.K.; Quillfeldt, J.A.; Hardt, O.; Alvares, L.D.O. Forgetting of long-term memory requires activation of NMDA receptors, L-type voltage-dependent Ca2+ channels, and calcineurin. Sci. Rep. 2016, 6, 22771. [Google Scholar] [CrossRef] [PubMed]
- Sachser, R.M.; Haubrich, J.; Lunardi, P.S.; Alvares, L.D.O. Forgetting of what was once learned: Exploring the role of postsynaptic ionotropic glutamate receptors on memory formation, maintenance, and decay. Neuropharmacology 2017, 112, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Migues, P.V.; Liu, L.; Archbold, G.E.B.; Einarsson, E.Ö.; Wong, J.; Bonasia, K.; Ko, S.H.; Wang, Y.T.; Hardt, O. Blocking Synaptic Removal of GluA2-Containing AMPA Receptors Prevents the Natural Forgetting of Long-Term Memories. J. Neurosci. 2016, 36, 3481–3494. [Google Scholar] [CrossRef]
- Hardt, O.; Nader, K.; Nadel, L. Decay happens: The role of active forgetting in memory. Trends Cogn. Sci. 2013, 17, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, K.; Hata, T. Post-acquisition hippocampal blockade of the NMDA receptor subunit GluN2A but not GluN2B sustains spatial reference memory retention. Neurobiol. Learn. Mem. 2018, 147, 1–8. [Google Scholar] [CrossRef]
- Wong, C.G.T.; Bottiglieri, T.; Snead, O.C. GABA, γ-hydroxybutyric acid, and neurological disease. Ann. Neurol. 2003, 54, S3–S12. [Google Scholar] [CrossRef]
- Haefely, W.; Martin, J.R.; Schoch, P. Novel anxiolytics that act as partial agonists at benzodiazepine receptors. Trends Pharmacol. Sci. 1990, 11, 452–456. [Google Scholar] [CrossRef]
- Olsen, R.W.; Spigelman, I. GABA-a Recaptor Plasticity in Alcohol Withdrawal. In JASPER’S Basic Mechanisms of the Epilepsies, 4th ed.; National Center for Biotechnology Information: Bethesda, MD, USA, 2012. [Google Scholar]
- Davis, M.; Campeau, S.; Kim, M.; Falls, W.A.; McGaugh, J.L.; Weinberger, N.M.; Lynch, G. Neural Systems of Emotion: The Amygdala’s Role in Fear and Anxiety. In Brain and Memory Modulation and Mediation of Neuroplasticity; Oxford University Press (OUP): Oxford, UK, 1995; pp. 3–40. [Google Scholar]
- Myers, K.M.; Davis, M. Systems-Level Reconsolidation. Neuron 2002, 36, 340–343. [Google Scholar] [CrossRef][Green Version]
- Davies, C.; Starkey, S.; Pozza, M.; Collingridge, G. GABA-B autoreceptors regulate the induction of LTP. Nature 1991, 349, 609–611. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; McNally, G.P.; Richardson, R. Recovery of fear memories in rats: Role of gamma-amino butyric acid (GABA) in infantile amnesia. Behav. Neurosci. 2006, 120, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Richardson, R. A developmental dissociation of context and GABA effects on extinguished fear in rats. Behav. Neurosci. 2007, 121, 131–139. [Google Scholar] [CrossRef]
- Tang, H.H.Y.; McNally, G.P.; Richardson, R. The effects of FG7142 on two types of forgetting in 18-day-old rats. Behav. Neurosci. 2007, 121, 1421–1425. [Google Scholar] [CrossRef] [PubMed]
- Zeman, A.; Hoefeijzers, S.; Milton, F.; Dewar, M.; Carr, M.; Streatfield, C. The GABAB receptor agonist, baclofen, contributes to three distinct varieties of amnesia in the human brain—A detailed case report. Cortex 2016, 74, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Bergstrom, H.C.; Pinard, C.R. Corticolimbic circuits in learning, memory, and disease. J. Neurosci. Res. 2017, 95, 795–796. [Google Scholar] [CrossRef]
- Blake, M.G.; Boccia, M.M. Basal Forebrain Cholinergic System and Memory. Curr. Top. Behav. Neurosci. 2016, 253–273. [Google Scholar] [CrossRef]
- Bentley, P.; Driver, J.; Dolan, R.J. Cholinergic modulation of cognition: Insights from human pharmacological functional neuroimaging. Prog. Neurobiol. 2011, 94, 360–388. [Google Scholar] [CrossRef]
- Bertrand, D.; Wallace, T.L. A Review of the Cholinergic System and Therapeutic Approaches to Treat Brain Disorders. Curr. Top. Behav. Neurosci. 2020, 1–28. [Google Scholar] [CrossRef]
- Edginton, T.; Rusted, J.M. Separate and combined effects of scopolamine and nicotine on retrieval-induced forgetting. Psychopharmacology 2003, 170, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Rusted, J.M.; Alvares, T. Nicotine effects on retrieval-induced forgetting are not attributable to changes in arousal. Psychopharmacology 2008, 196, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.O.; Battagello, D.S.; Cardoso, A.R.; Hauser, D.N.; Bittencourt, J.C.; Correa, R.G. Dopamine: Functions, signaling, and association with neurological diseases. Cell. Mol. Nurobiol. 2019, 39, 31–59. [Google Scholar] [CrossRef] [PubMed]
- Grogan, J.P.; Bogacz, R.; Tsivos, D.; Whone, A.L.; Coulthard, E. Dopamine and Consolidation of Episodic Memory: Timing Is Everything. J. Cogn. Neurosci. 2015, 27, 2035–2050. [Google Scholar] [CrossRef] [PubMed]
- Karunakaran, S.; Chowdhury, A.; Donato, F.; Quairiaux, C.; Michel, C.M.; Caroni, P. PV plasticity sustained through D1/5 dopamine signaling required for long-term memory consolidation. Nat. Neurosci. 2016, 19, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Péczely, L.; Ollmann, T.; László, K.; Kovács, A.; Gálosi, R.; Kertes, E.; Zagorácz, O.; Kállai, V.; Karádi, Z.; Lénárd, L. Role of ventral pallidal D2 dopamine receptors in the consolidation of spatial memory. Behav. Brain Res. 2016, 313, 1–9. [Google Scholar] [CrossRef]
- Yamasaki, M.; Takeuchi, T. Locus Coeruleus and Dopamine-Dependent Memory Consolidation. Neural Plast. 2017, 2017, 1–15. [Google Scholar] [CrossRef]
- Berry, J.A.; Cervantes-Sandoval, I.; Nicholas, E.P.; Davis, R.L. Dopamine Is Required for Learning and Forgetting in Drosophila. Neuron 2012, 74, 530–542. [Google Scholar] [CrossRef]
- Berry, J.A.; Cervantes-Sandoval, I.; Chakraborty, M.; Davis, R.L. Sleep Facilitates Memory by Blocking Dopamine Neuron-Mediated Forgetting. Cell 2015, 161, 1656–1667. [Google Scholar] [CrossRef]
- Reichenbach, N.; Herrmann, U.; Kähne, T.; Schicknick, H.; Pielot, R.; Naumann, M.; Dieterich, D.C.; Gundelfinger, E.D.; Smalla, K.-H.; Tischmeyer, W. Differential effects of dopamine signalling on long-term memory formation and consolidation in rodent brain. Proteome Sci. 2015, 13, 13. [Google Scholar] [CrossRef]
- Rossignoli, M.T.; Lopes-Aguiar, C.; Ruggiero, R.N.; Da Silva, R.A.D.V.; Bueno-Junior, L.S.; Kandratavicius, L.; Peixoto-Santos, J.E.; Crippa, J.A.; Hallak, J.E.C.; Zuardi, A.W.; et al. Selective post-training time window for memory consolidation interference of cannabidiol into the prefrontal cortex: Reduced dopaminergic modulation and immediate gene expression in limbic circuits. Neuroscience 2017, 350, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Sandoval, I.; Chakraborty, M.; MacMullen, C.; Davis, R.L. Scribble Scaffolds a Signalosome for Active Forgetting. Neuron 2016, 90, 1230–1242. [Google Scholar] [CrossRef]
- Kaun, K.R.; Rothenfluh, A. Dopaminergic rules of engagement for memory in Drosophila. Curr. Opin. Neurobiol. 2017, 43, 56–62. [Google Scholar] [CrossRef]
- Himmelreich, S.; Masuho, I.; Berry, J.A.; MacMullen, C.; Skamangas, N.K.; Martemyanov, K.A.; Davis, R.L. Dopamine Receptor DAMB Signals via Gq to Mediate Forgetting in Drosophila. Cell Rep. 2017, 21, 2074–2081. [Google Scholar] [CrossRef]
- Shuai, Y.; Hirokawa, A.; Ai, Y.; Zhang, M.; Li, W.; Zhong, Y. Dissecting neural pathways for forgetting in Drosophila olfactory aversive memory. Proc. Natl. Acad. Sci. USA 2015, 112, E6663–E6672. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, M.K.; Carlsson, M.L. The monoaminergic stabilizer (−)-OSU6162 reverses delay-dependent natural forgetting and improves memory impairment induced by scopolamine in mice. Neuropharmacology 2013, 75, 399–406. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.J.; Paolieri, D.; Colzato, L.S.; Bajo, M.T. Directed forgetting of memories in cocaine users. Exp. Clin. Psychopharmacol. 2016, 24, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Papenberg, G.; Bäckman, L.; Nagel, I.E.; Nietfeld, W.; Schröder, J.; Bertram, L.; Heekeren, H.R.; Lindenberger, U.; Li, S.-C. Dopaminergic Gene Polymorphisms Affect Long-term Forgetting in Old Age: Further Support for the Magnification Hypothesis. J. Cogn. Neurosci. 2013, 25, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Wimber, M.; Schott, B.H.; Wendler, F.; Seidenbecher, C.I.; Behnisch, G.; Macharadze, T.; Bäuml, K.H.; Richardson-Klavehn, A. Prefrontal dopamine and the dynamic control of human long-term memory. Trans-cult. Psychiatry 2011, 1, 15. [Google Scholar]
- Dolan, R.J. Emotion, Cognition, and Behavior. Science 2002, 298, 1191–1194. [Google Scholar] [CrossRef]
- Phelps, E.A.; Delgado, M.R.; Nearing, K.I.; LeDoux, J.E. Extinction Learning in Humans. Neuron 2004, 43, 897–905. [Google Scholar] [CrossRef]
- McGaugh, J.L.; Izquierdo, I. The contribution of pharmacology to research on the mechanisms of memory formation. Trends Pharmacol. Sci. 2000, 21, 208–210. [Google Scholar] [CrossRef]
- Strange, B.A.; Hurlemann, R.; Dolan, R.J. An emotion-induced retrograde amnesia in humans is amygdala- and -adrenergic-dependent. Proc. Natl. Acad. Sci. USA 2003, 100, 13626–13631. [Google Scholar] [CrossRef]
- Strange, B.A.; Dolan, R.J. Adrenergic modulation of emotional memory-evoked human amygdala and hippocampal responses. Proc. Natl. Acad. Sci. USA 2004, 101, 11454–11458. [Google Scholar] [CrossRef]
- Van Stegeren, A.H.; Goekoop, R.; Everaerd, W.; Scheltens, P.; Barkhof, F.; Kuijer, J.P.; Rombouts, S.A. Noradrenaline mediates amygdala activation in men and women during encoding of emotional material. NeuroImage 2005, 24, 898–909. [Google Scholar] [CrossRef] [PubMed]
- Harmer, C.J.; Hill, S.A.; Taylor, M.J.; Cowen, P.J.; Goodwin, G.M. Toward a Neuropsychological Theory of Antidepressant Drug Action: Increase in Positive Emotional Bias After Potentiation of Norepinephrine Activity. Am. J. Psychiatry 2003, 160, 990–992. [Google Scholar] [CrossRef]
- Harmer, C.J.; Shelley, N.C.; Cowen, P.J.; Goodwin, G.M. Increased Positive Versus Negative Affective Perception and Memory in Healthy Volunteers Following Selective Serotonin and Norepinephrine Reuptake Inhibition. Am. J. Psychiatry 2004, 161, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Hurlemann, R.; Hawellek, B.; Matusch, A.; Kolsch, H.; Wollersen, H.; Madea, B.; Vogeley, K.; Maier, W.; Dolan, R.J. Noradrenergic Modulation of Emotion-Induced Forgetting and Remembering. J. Neurosci. 2005, 25, 6343–6349. [Google Scholar] [CrossRef] [PubMed]
- Quartermain, D.; Leo, P. Strength of scopolamine-induced amnesia as a function of time between training and testing. Behav. Neural Biol. 1988, 50, 300–310. [Google Scholar] [CrossRef]
- Decker, M.W.; McGaugh, J.L. Effects of concurrent manipulations of cholinergic and noradrenergic function on learning and retention in mice. Brain Res. 1989, 477, 29–37. [Google Scholar] [CrossRef]
- Finsterwald, C.; Alberini, C.M. Stress and glucocorticoid receptor-dependent mechanisms in long-term memory: From adaptive responses to psychopathologies. Neurobiol. Learn. Mem. 2014, 112, 17–29. [Google Scholar] [CrossRef]
- Meir Drexler, S.; Wolf, O.T. The role of glucocorticoids in emotional memory reconsolidation. Neurobiol. Learn. Mem. 2016. [Google Scholar] [CrossRef]
- Wolf, O.T. Stress and memory in humans: Twelve years of progress? Brain Res. 2009, 1293, 142–154. [Google Scholar] [CrossRef]
- De Quervain, D.; Schwabe, L.; Roozendaal, B. Stress, glucocorticoids and memory:implications for treating fear-related disorders. Nat. Rev. Neurosci. 2017, 18, 7–19. [Google Scholar] [CrossRef]
- Het, S.; Ramlow, G.; Wolf, O. A meta-analytic review of the effects of acute cortisol administration on human memory. Psychoneuroendocrinology 2005, 30, 771–784. [Google Scholar] [CrossRef]
- Wolf, O.T.; Atsak, P.; De Quervain, D.J.; Roozendaal, B.; Wingenfeld, K. Stress and Memory: A Selective Review on Recent Developments in the Understanding of Stress Hormone Effects on Memory and Their Clinical Relevance. J. Neuroendocr. 2016, 28. [Google Scholar] [CrossRef] [PubMed]
- Koessler, S.; Steidle, L.; Engler, H.; Kissler, J. Stress eliminates retrieval-induced forgetting—Does the oral application of cortisol? Psychoneuroendocrinology 2013, 38, 94–106. [Google Scholar] [CrossRef] [PubMed]
- Koessler, S.; Engler, H.; Riether, C.; Kissler, J. No Retrieval-Induced Forgetting Under Stress. Psychol. Sci. 2009, 20, 1356–1363. [Google Scholar] [CrossRef] [PubMed]
- Quaedflieg, C.; Schneider, T.; Daume, J.; Engel, A.; Schwabe, L. Stress impairs intentional memory control through altered theta oscillations in lateral parietal cortex. J. Neurosci. 2020, 40, 7739–7748. [Google Scholar] [CrossRef]
- Engen, H.G.; Anderson, M.C. Memory Control: A Fundamental Mechanism of Emotion Regulation. Trends Cogn. Sci. 2018, 22, 982–995. [Google Scholar] [CrossRef]
- Barkley, R.A. Behavioral inhibition, sustained attention, and executive functions: Constructing a unifying theory of ADHD. Psychol. Bull. 1997, 121, 65–94. [Google Scholar] [CrossRef]
- Conway, M.A.; Fthenaki, A. Disruption of Inhibitory Control of Memory Following Lesions to the Frontal and Temporal Lobes. Cortex 2003, 39, 667–686. [Google Scholar] [CrossRef]
- Bari, A.; Robbins, T.W. Inhibition and impulsivity: Behavioral and neural basis of response control. Prog. Neurobiol. 2013, 108, 44–79. [Google Scholar] [CrossRef]
- Aron, A.R.; Robbins, T.W.; Poldrack, R.A. Inhibition and the right inferior frontal cortex: One decade on. Trends Cogn. Sci. 2014, 18, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.C.; Bunce, J.G.; Barbas, H. Prefrontal–hippocampal pathways underlying inhibitory control over memory. Neurobiol. Learn. Mem. 2016, 134, 145–161. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Nasca, C.; Gray, J.D. Stress Effects on Neuronal Structure: Hippocampus, Amygdala, and Prefrontal Cortex. Neuropsychopharmacology 2016, 41, 3–23. [Google Scholar] [CrossRef]
- Bartholomew, M.E.; Heller, W.; Miller, G.A. Inhibitory control of emotional processing: Theoretical and empirical considerations. Int. J. Psychophysiol. 2019. [Google Scholar] [CrossRef] [PubMed]
- McKlveen, J.M.; Moloney, R.D.; Scheimann, J.R.; Myers, B.; Herman, J.P. “Braking” the Prefrontal Cortex: The Role of Glucocorticoids and Interneurons in Stress Adaptation and Pathology. Biol. Psychiatry 2019, 86, 669–681. [Google Scholar] [CrossRef]
- Anderson, M.C.; Hulbert, J.C. Active Forgetting: Adaptation of Memory by Prefrontal Control. Annu. Rev. Psychol. 2021, 72, 1–36. [Google Scholar] [CrossRef]
- Del Arco, A.; Mora, F. Glutamate-dopamine in vivo interaction in the prefrontalcortex modulates the release of dopamine and acetylcholinein the nucleus accumbens of the awake rat. J. Neural Transm. 2005, 112, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Robbins, T.W.; Arnsten, A.F.T. The Neuropsychopharmacology of Fronto-Executive Function: Monoaminergic Modulation. Annu. Rev. Neurosci. 2009, 32, 267–287. [Google Scholar] [CrossRef] [PubMed]
- Duvarci, S.; Pare, D. Amygdala Microcircuits Controlling Learned Fear. Neuron 2014, 82, 966–980. [Google Scholar] [CrossRef]
- Schmitz, T.W.; Correia, M.M.; Ferreira, C.S.; Prescot, A.P.; Anderson, M.C. Hippocampal GABA enables inhibitory control over unwanted thoughts. Nat. Commun. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- Depue, B.E.; Curran, T.; Banich, M.T. Prefrontal Regions Orchestrate Suppression of Emotional Memories via a Two-Phase Process. Science 2007, 317, 215–219. [Google Scholar] [CrossRef]
- Ghashghaei, H.; Barbas, H. Pathways for emotion: Interactions of prefrontal and anterior temporal pathways in the amygdala of the rhesus monkey. Neuroscience 2002, 115, 1261–1279. [Google Scholar] [CrossRef]
- Sotres-Bayon, F.; Quirk, G.J. Prefrontal control of fear: More than just extinction. Curr. Opin. Neurobiol. 2010, 20, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Giustino, T.F.; Emaren, S. The Role of the Medial Prefrontal Cortex in the Conditioning and Extinction of Fear. Front. Behav. Neurosci. 2015, 9, 298. [Google Scholar] [CrossRef] [PubMed]
- Del Arco, A.; Mora, F. Neurotransmitters and prefrontal cortex–limbic system interactions: Implications for plasticity and psychiatric disorders. J. Neural. Transm. 2009, 116, 941–952. [Google Scholar] [CrossRef] [PubMed]
- Wellman, C.L. Dendritic reorganization in pyramidal neurons in medial prefrontal cortex after chronic corticosterone administration. J. Neurobiol. 2001, 49, 245–253. [Google Scholar] [CrossRef]
- Likhtik, E.; Johansen, J.P. Neuromodulation in circuits of aversive emotional learning. Nat. Neurosci. 2019, 22, 1586–1597. [Google Scholar] [CrossRef]
- Bast, T.; Pezze, M.; McGarrity, S. Cognitive deficits caused by prefrontal cortical and hippocampal neural disinhibition. Br. J. Pharmacol. 2017, 174, 3211–3225. [Google Scholar] [CrossRef]
- Bloem, B.; Poorthuis, R.B.; Mansvelder, H.D. Cholinergic modulation of the medial prefrontal cortex: The role of nicotinic receptors in attention and regulation of neuronal activity. Front. Neural Circuits 2014, 8. [Google Scholar] [CrossRef]
- Parikh, V.; Bangasser, D.A. Cholinergic Signaling Dynamics and Cognitive Control of Attention. Behav. Neurobiol. Chronic Pain 2020, 71–87. [Google Scholar] [CrossRef]
- Kasparbauer, A.-M.; Petrovsky, N.; Schmidt, P.-M.; Trautner, P.; Weber, B.; Sträter, B.; Ettinger, U. Effects of nicotine and atomoxetine on brain function during response inhibition. Eur. Neuropsychopharmacol. 2019, 29, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Berridge, C.W.; Waterhouse, B.D. The locus coeruleus–noradrenergic system: Modulation of behavioral state and state-dependent cognitive processes. Brain Res. Rev. 2003, 42, 33–84. [Google Scholar] [CrossRef]
- Jouvet, M. Biogenic Amines and the States of Sleep. Science 1969, 163, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.A.; Rompre, P.P. Brain Dopamine and Reward. Annu. Rev. Psychol. 1989, 40, 191–225. [Google Scholar] [CrossRef]
- Aston-Jones, G.; Cohen, J.D. An Integrative Theory of Locus Coeruleus-Norepinephrine Function: Adaptive Gain and Optimal Performance. Annu. Rev. Neurosci. 2005, 28, 403–450. [Google Scholar] [CrossRef] [PubMed]
- Robinson, T.E. The neural basis of drug craving: An incentive-sensitization theory of addiction. Brain Res. Rev. 1993, 18, 247–291. [Google Scholar] [CrossRef]
- Mather, M.; Clewett, D.; Sakaki, M.; Harley, C.W. Norepinephrine ignites local hotspots of neuronal excitation: How arousal amplifies selectivity in perception and memory. Behav. Brain Sci. 2016, 39, e200. [Google Scholar] [CrossRef]
- Chmielewski, W.X.; Mückschel, M.; Ziemssen, T.; Beste, C. The norepinephrine system affects specific neurophysiological subprocesses in the modulation of inhibitory control by working memory demands. Hum. Brain Mapp. 2016, 38, 68–81. [Google Scholar] [CrossRef]
- Mückschel, M.; Dippel, G.; Beste, C. Distinguishing stimulus and response codes in theta oscillations in prefrontal areas during inhibitory control of automated responses. Hum. Brain Mapp. 2017, 38, 5681–5690. [Google Scholar] [CrossRef]
- Nieuwenhuis, S.; Aston-Jones, G.; Cohen, J.D. Decision making, the P3, and the locus coeruleus--norepinephrine system. Psychol. Bull. 2005, 131, 510–532. [Google Scholar] [CrossRef] [PubMed]
- Solanto, M.V. Neuropsychopharmacological mechanisms of stimulant drug action in attention-deficit hyperactivity disorder: A review and integration. Behav. Brain Res. 1998, 94, 127–152. [Google Scholar] [CrossRef]
- Dalley, J.W.; Cardinal, R.N.; Robbins, T.W. Prefrontal executive and cognitive functions in rodents: Neural and neurochemical substrates. Neurosci. Biobehav. Rev. 2004, 28, 771–784. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.J.; Fowler, J.S.; Logan, J.; Gatley, S.J.; Gifford, A.; Hitzemann, R.; Ding, Y.S.; Pappas, N. Prediction of reinforcing responses to psychostimulants in humans by brain dopamine D2 receptor levels. Am. J. Psychiatry 1999, 156, 1440–1443. [Google Scholar]
- Volkow, N.D.; Wang, G.-J.; Fowler, J.S.; Hitzemann, R.; Angrist, B.; Gatley, S.J.; Logan, J.; Ding, Y.-S.; Pappas, N. Association of Methylphenidate-Induced Craving with Changes in Right Striato-orbitofrontal Metabolism in Cocaine Abusers: Implications in Addiction. Am. J. Psychiatry 1999, 156, 19–26. [Google Scholar] [CrossRef]
- Eagle, D.M.; Tufft, M.R.A.; Goodchild, H.L.; Robbins, T.W. Differential effects of modafinil and methylphenidate on stop-signal reaction time task performance in the rat, and interactions with the dopamine receptor antagonist cis-flupenthixol. Psychopharmacology 2007, 192, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, S.R.; Hampshire, A.; Müller, U.; Rubia, K.; Del Campo, N.; Craig, K.; Regenthal, R.; Suckling, J.; Roiser, J.P.; Grant, J.E.; et al. Atomoxetine Modulates Right Inferior Frontal Activation During Inhibitory Control: A Pharmacological Functional Magnetic Resonance Imaging Study. Biol. Psychiatry 2009, 65, 550–555. [Google Scholar] [CrossRef]
- Mückschel, M.; Roessner, V.; Beste, C. Task experience eliminates catecholaminergic effects on inhibitory control—A randomized, double-blind cross-over neurophysiological study. Eur. Neuropsychopharmacol. 2020, 35, 89–99. [Google Scholar] [CrossRef]
- Xing, B.; Li, Y.-C.; Gao, W.-J. Norepinephrine versus dopamine and their interaction in modulating synaptic function in the prefrontal cortex. Brain Res. 2016, 1641, 217–233. [Google Scholar] [CrossRef]
- Belanoff, J.K.; Kalehzan, M.; Sund, B.; Fleming, F.S.; Schatzberg, A.F. Cortisol activity and cognitive changes in psychotic major depres- sion. Am. J. Psychiatry 2001, 158, 1612–1616. [Google Scholar] [CrossRef] [PubMed]
- Scholz, U.; La Marca, R.; Nater, U.M.; Aberle, I.; Ehlert, U.; Hornung, R.; Martin, M.; Kliegel, M. Go no-go performance under psychosocial stress: Beneficial effects of implementation intentions. Neurobiol. Learn. Mem. 2009, 91, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Arnsten, A.F.T.; Wang, M.; Paspalas, C.D. Dopamine’s Actions in Primate Prefrontal Cortex: Challenges for Treating Cognitive Disorders. Pharmacol. Rev. 2015, 67, 681–696. [Google Scholar] [CrossRef] [PubMed]
- Godsil, B.P.; Kiss, J.P.; Spedding, M.; Jay, T.M. The hippocampal–prefrontal pathway: The weak link in psychiatric disorders? Eur. Neuropsychopharmacol. 2013, 23, 1165–1181. [Google Scholar] [CrossRef]
- Cummings, J.L. Anatomic and Behavioral Aspects of Frontal-Subcortical Circuits. Ann. N. Y. Acad. Sci. 1995, 769, 1–14. [Google Scholar] [CrossRef]
- Volk, D.W.; Lewis, D.A. Impaired prefrontal inhibition in schizophrenia: Relevance for cognitive dysfunction. Physiol. Behav. 2002, 77, 501–505. [Google Scholar] [CrossRef]
- Lewis, D.A.; Volk, D.W.; Hashimoto, T. Selective alterations in prefrontal cortical GABA neurotransmission in schizophrenia: A novel target for the treatment of working memory dysfunction. Psychopharmacology 2003, 174, 143–150. [Google Scholar] [CrossRef]
- Hayes, D.J.; Jupp, B.; Sawiak, S.J.; Merlo, E.; Caprioli, D.; Dalley, J.W. Brain γ-aminobutyric acid: A neglected role in impulsivity. Eur. J. Neurosci. 2014, 39, 1921–1932. [Google Scholar] [CrossRef] [PubMed]
- Averill, L.A.; Purohit, P.; Averill, C.L.; Boesl, M.A.; Krystal, J.H.; Abdallah, C.G. Glutamate dysregulation and glutamatergic therapeutics for PTSD: Evidence from human studies. Neurosci. Lett. 2017, 649, 147–155. [Google Scholar] [CrossRef]
- Ousdal, O.T.; Milde, A.M.; Craven, A.R.; Ersland, L.; Endestad, T.; Melinder, A.; Huys, Q.J.; Hugdahl, K. Prefrontal glutamate levels predict altered amygdala–prefrontal connectivity in traumatized youths. Psychol. Med. 2018, 49, 1822–1830. [Google Scholar] [CrossRef] [PubMed]
- Sheth, C.; Prescot, A.P.; Legarreta, M.; Renshaw, P.F.; McGlade, E.; Yurgelun-Todd, D. Reduced gamma-amino butyric acid (GABA) and glutamine in the anterior cingulate cortex (ACC) of veterans exposed to trauma. J. Affect. Disord. 2019, 248, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, C.I.; Kegeles, L.S.; Levinson, A.; Ogden, R.T.; Mao, X.; Milak, M.S.; Vermes, D.; Xie, S.; Hunter, L.; Flood, P.; et al. In vivo effects of ketamine on glutamate-glutamine and gamma-aminobutyric acid in obsessive-compulsive disorder: Proof of concept. Psychiatry Res. Neuroimaging 2015, 233, 141–147. [Google Scholar] [CrossRef][Green Version]
- Yang, A.C.; Tsai, S.-J. New Targets for Schizophrenia Treatment beyond the Dopamine Hypothesis. Int. J. Mol. Sci. 2017, 18, 1689. [Google Scholar] [CrossRef]
- Vlček, P.; Polák, J.; Brunovský, M.; Horáček, J. Role of Glutamatergic System in Obsessive-Compulsive Disorder with Possible Therapeutic Implications. Pharmacopsychiatry 2018, 51, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, S.T.; Sanacora, G. A new generation of antidepressants: An update on the pharmaceutical pipeline for novel and rapid-acting therapeutics in mood disorders based on glutamate/GABA neurotransmitter systems. Drug Discov. Today 2019, 24, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Sanacora, G.; Krystal, J.H. Altered Connectivity in Depression: GABA and Glutamate Neurotransmitter Deficits and Reversal by Novel Treatments. Neuron 2019, 102, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Lavretsky, H.; Laird, K.T.; Krause-Sorio, B.; Heimberg, B.F.; Yeargin, J.; Grzenda, A.; Wu, P.; Thana-Udom, K.; Ercoli, L.M.; Siddarth, P. A Randomized Double-Blind Placebo-Controlled Trial of Combined Escitalopram and Memantine for Older Adults With Major Depression and Subjective Memory Complaints. Am. J. Geriatr. Psychiatry 2020, 28, 178–190. [Google Scholar] [CrossRef]
- Ketenci, S.; Acet, N.G.; Sarıdoğan, G.E.; Aydın, B.; Cabadak, H.; Gören, M.Z. The Neurochemical Effects of Prazosin Treatment on Fear Circuitry in a Rat Traumatic Stress Model. Clin. Psychopharmacol. Neurosci. 2020, 18, 219. [Google Scholar] [CrossRef]
- Foland-Ross, L.C.; Gotlib, I.H. Cognitive and Neural Aspects of Information Processing in Major Depressive Disorder: An Integrative Perspective. Front. Psychol. 2012, 3, 489. [Google Scholar] [CrossRef]
- Gvozdanovic, G.; Stämpfli, P.; Seifritz, E.; Rasch, B. Structural brain differences predict early traumatic memory processing. Psychophysiology 2019, 57, e13354. [Google Scholar] [CrossRef]
- Andrewes, D.G.; Jenkins, L.M. The Role of the Amygdala and the Ventromedial Prefrontal Cortex in Emotional Regulation: Implications for Post-traumatic Stress Disorder. Neuropsychol. Rev. 2019, 29, 220–243. [Google Scholar] [CrossRef]
- Davidson, R.J.; Pizzagalli, D.; Nitschke, J.B.; Putnam, K. Depression: Perspectives from Affective Neuroscience. Annu. Rev. Psychol. 2002, 53, 545–574. [Google Scholar] [CrossRef]
- Wolf, O.T. The influence of stress hormones on emotional memory: Relevance for psychopathology. Acta Psychol. 2008, 127, 513–531. [Google Scholar] [CrossRef]
- Itzhaky, L.; Levin, Y.; Fingerhut, H.; Solomon, Z. Depression and PTSD Co-Morbidity: What are We Missing? J. Depress. Anxiety 2014, 3, 1–6. [Google Scholar]
- Malhi, G.S.; Berk, M. Does dopamine dysfunction drive depression? Acta Psychiatr. Scand. 2007, 115, 116–124. [Google Scholar] [CrossRef]
- Gören, M.Z.; Cabadak, H. Noradrenaline and Post-traumatic Stress Disorder. In Comprehensive Guide to Post-Traumatic Stress Disorder; Metzler, J.B., Ed.; Springer: Cham, Switzerland, 2015; pp. 1–16. [Google Scholar]
- Dulawa, S.C.; Janowsky, D.S. Cholinergic regulation of mood: From basic and clinical studies to emerging therapeutics. Mol. Psychiatry 2019, 24, 694–709. [Google Scholar] [CrossRef]
- Hasselmo, M.E.; Stern, C.E. Mechanisms underlying working memory for novel information. Trends Cogn. Sci. 2006, 10, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.A.; Bourne, C. Inducing and modulating intrusive emotional memories: A review of the trauma film paradigm. Acta Psychol. 2008, 127, 553–566. [Google Scholar] [CrossRef]
- Maurer, S.V.; Williams, C.L. The Cholinergic System Modulates Memory and Hippocampal Plasticity via Its Interactions with Non-Neuronal Cells. Front. Immunol. 2017, 8, 1489. [Google Scholar] [CrossRef] [PubMed]
- Christodoulou, T.; Messinis, L.; Papathanasopoulos, P.; Frangou, S. The impact of familial risk for schizophrenia or bipolar disorder on cognitive control during episodic memory retrieval. Psychiatry Res. 2012, 197, 212–216. [Google Scholar] [CrossRef]
- Gotlib, I.H.; Joormann, J. Cognition and Depression: Current Status and Future Directions. Annu. Rev. Clin. Psychol. 2010, 6, 285–312. [Google Scholar] [CrossRef] [PubMed]
- Dreifus, L.; Engler, H.; Kissler, J. Retrieval-induced forgetting under psychosocial stress: No reduction by delayed stress and beta-adrenergic blockade. Neurobiol. Learn. Mem. 2014, 110, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Robbins, T.W.; Vaghi, M.M.; Banca, P. Review obsessive-compulsive disorder: Puzzles and prospects. Neuron 2019, 102, 27–47. [Google Scholar] [CrossRef] [PubMed]
- Tritsch, N.X.; Sabatini, B.L. Dopaminergic Modulation of Synaptic Transmission in Cortex and Striatum. Neuron 2012, 76, 33–50. [Google Scholar] [CrossRef] [PubMed]
- Glahn, D.C.; Knowles, E.E.M.; Pearlson, G.D. Genetics of cognitive control: Implications for Nimh’s research domain criteria initiative. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2015, 171, 111–120. [Google Scholar] [CrossRef]
- Iacono, W.G.; Malone, S.M.; Vrieze, S.I. Endophenotype best practices. Int. J. Psychophysiol. 2017, 111, 115–144. [Google Scholar] [CrossRef]
- James, E.L.; Lau-Zhu, A.; Clark, I.A.; Visser, R.M.; Hagenaars, M.A.; Holmes, E.A. The trauma film paradigm as an experimental psychopathology model of psychological trauma: Intrusive memories and beyond. Clin. Psychol. Rev. 2016, 47, 106–142. [Google Scholar] [CrossRef]
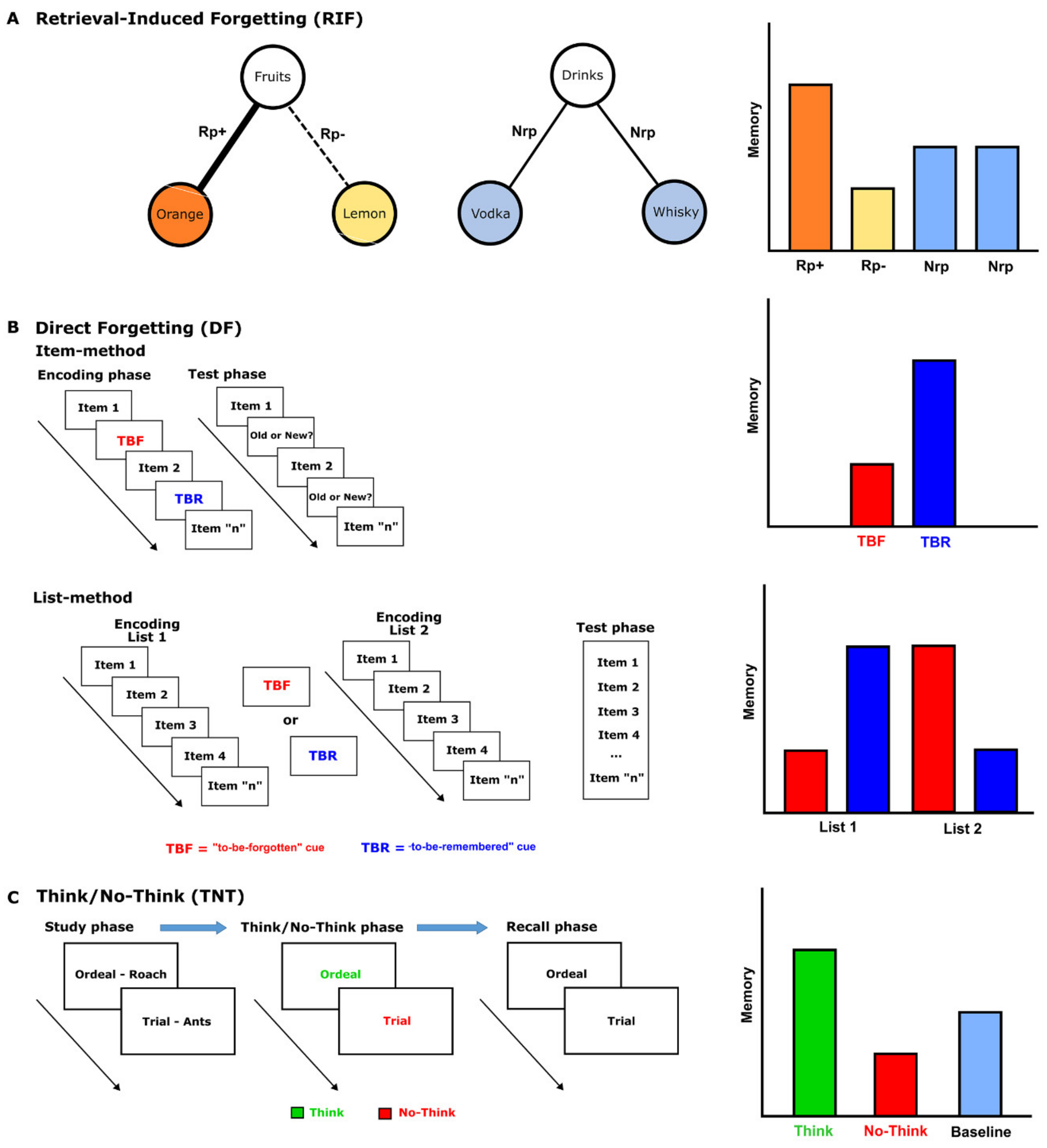
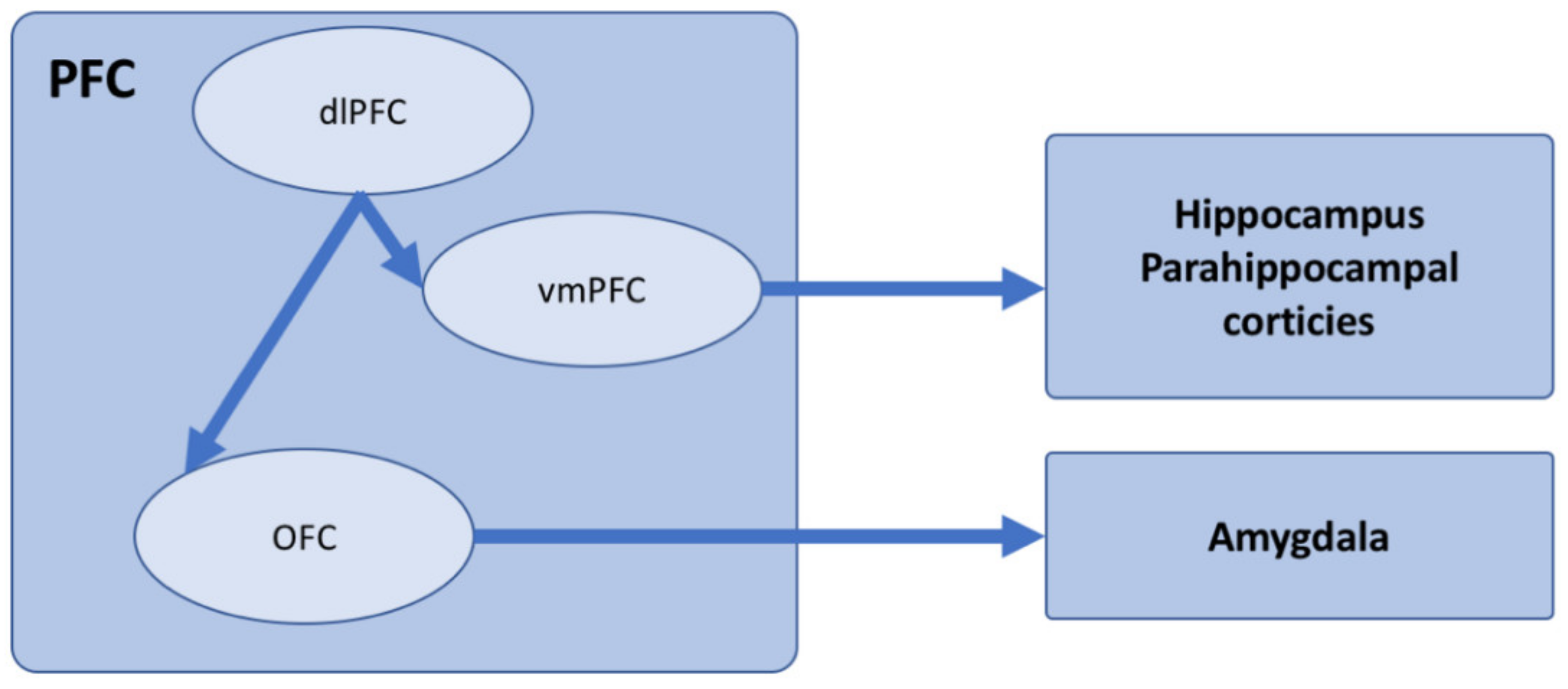
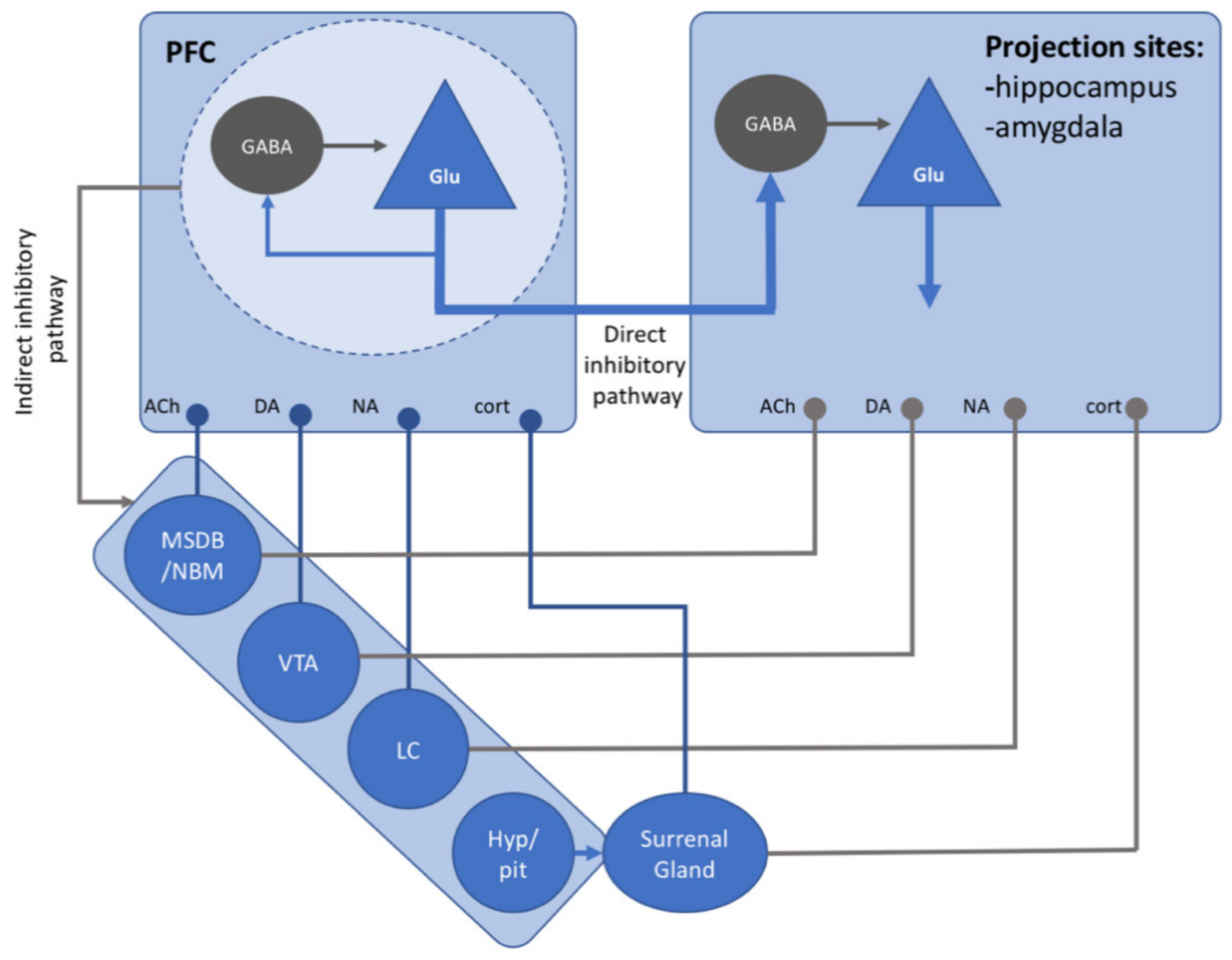
| Intentional Forgetting | Incidental Forgetting | ||
|---|---|---|---|
| DF | TNT | RIF | |
| Post-traumatic stress disorder | unaffected | affected | slightly affected |
| Depression * | affected | unaffected | affected |
| Schizophrenia | affected | n.a. | unaffected |
| Obsessive compulsive disorder | affected (only for disease-related stimuli) | n.a. | slightly affected (affected in OCD patients suffering of depressive symptoms) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costanzi, M.; Cianfanelli, B.; Santirocchi, A.; Lasaponara, S.; Spataro, P.; Rossi-Arnaud, C.; Cestari, V. Forgetting Unwanted Memories: Active Forgetting and Implications for the Development of Psychological Disorders. J. Pers. Med. 2021, 11, 241. https://doi.org/10.3390/jpm11040241
Costanzi M, Cianfanelli B, Santirocchi A, Lasaponara S, Spataro P, Rossi-Arnaud C, Cestari V. Forgetting Unwanted Memories: Active Forgetting and Implications for the Development of Psychological Disorders. Journal of Personalized Medicine. 2021; 11(4):241. https://doi.org/10.3390/jpm11040241
Chicago/Turabian StyleCostanzi, Marco, Beatrice Cianfanelli, Alessandro Santirocchi, Stefano Lasaponara, Pietro Spataro, Clelia Rossi-Arnaud, and Vincenzo Cestari. 2021. "Forgetting Unwanted Memories: Active Forgetting and Implications for the Development of Psychological Disorders" Journal of Personalized Medicine 11, no. 4: 241. https://doi.org/10.3390/jpm11040241
APA StyleCostanzi, M., Cianfanelli, B., Santirocchi, A., Lasaponara, S., Spataro, P., Rossi-Arnaud, C., & Cestari, V. (2021). Forgetting Unwanted Memories: Active Forgetting and Implications for the Development of Psychological Disorders. Journal of Personalized Medicine, 11(4), 241. https://doi.org/10.3390/jpm11040241








