Mechanisms Involved in Microglial-Interceded Alzheimer’s Disease and Nanocarrier-Based Treatment Approaches
Abstract
:1. Introduction
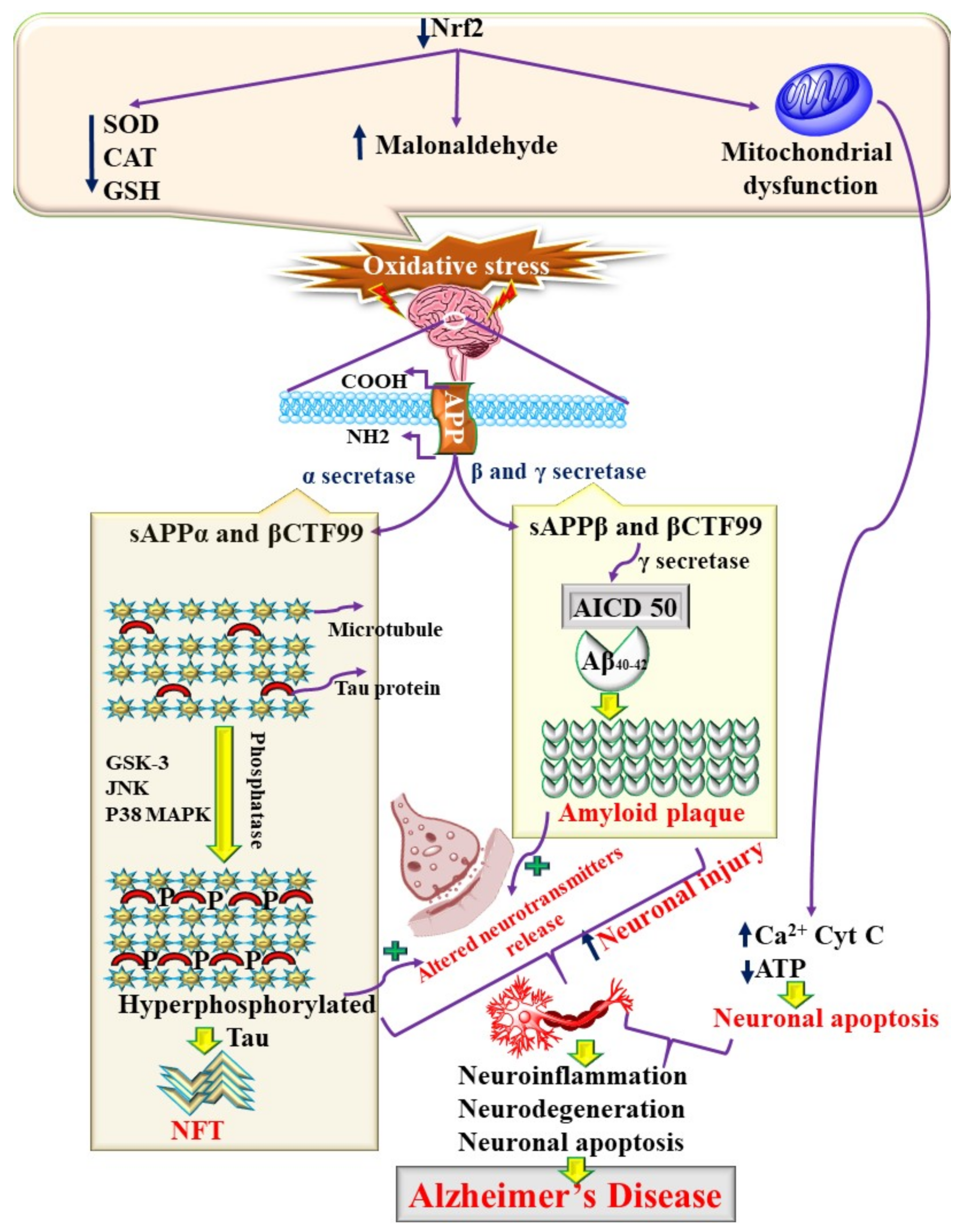
2. Molecular Pathogenesis of AD
3. Innate Immunity in AD
3.1. Glial Cells and Innate Immunity in AD
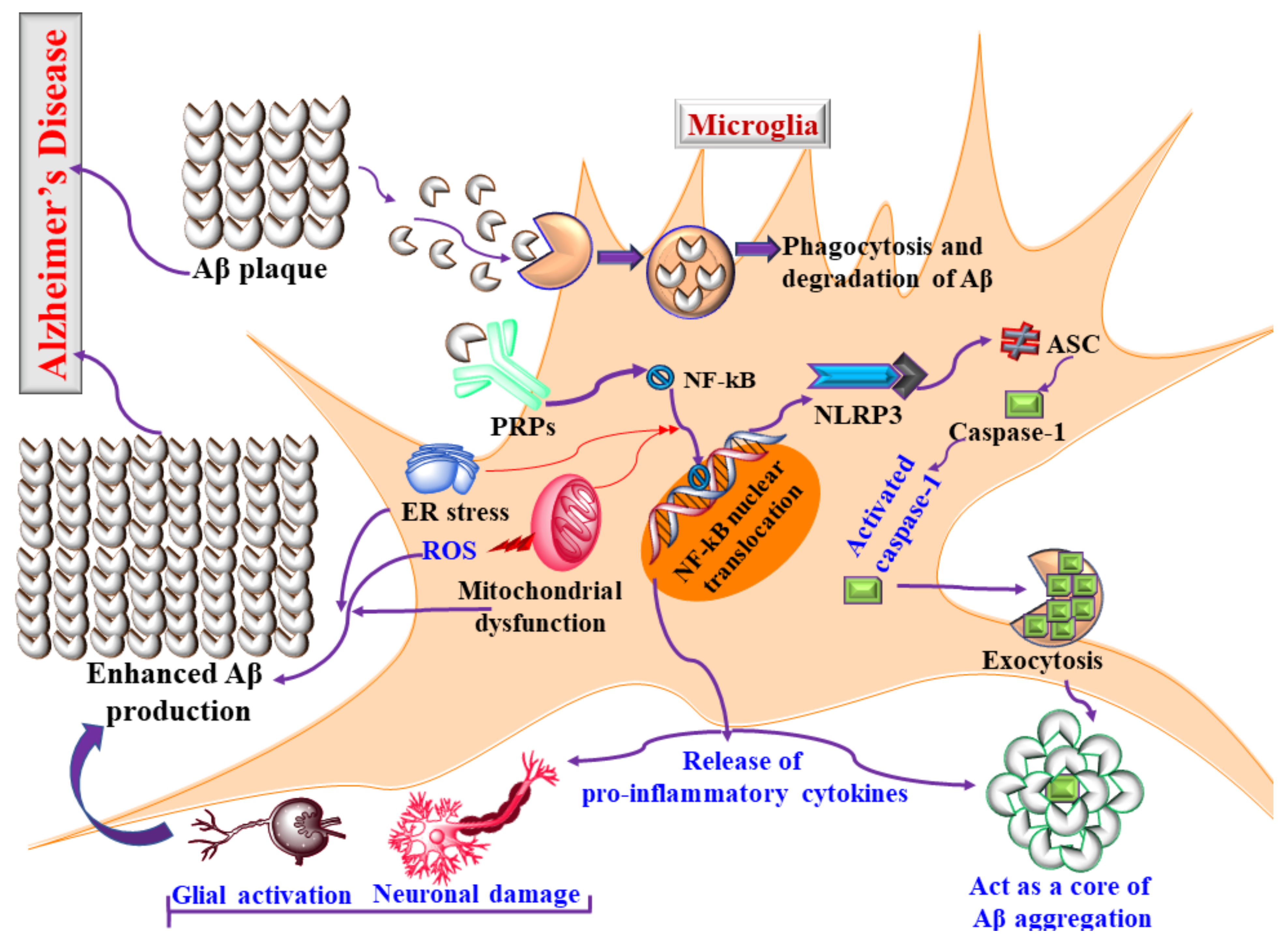
3.2. Microglia and Neuroinflammation in AD
3.3. Microglia and Tau Pathology
4. Therapeutic Approaches for the Management of Microglial-Activated AD
4.1. Drugs Targeting Inhibition of M1 Microglial Polarization
4.2. Drugs Targeting the M1 to M2 Phenotype Shift
4.3. Limitations of Existing Targeted Drugs to Combat AD
4.4. Natural Bioactives as Potential Modulators of Microglial-Mediated AD
5. Nanocarrier as a Potential Tool for Groundbreaking Drug Delivery in AD
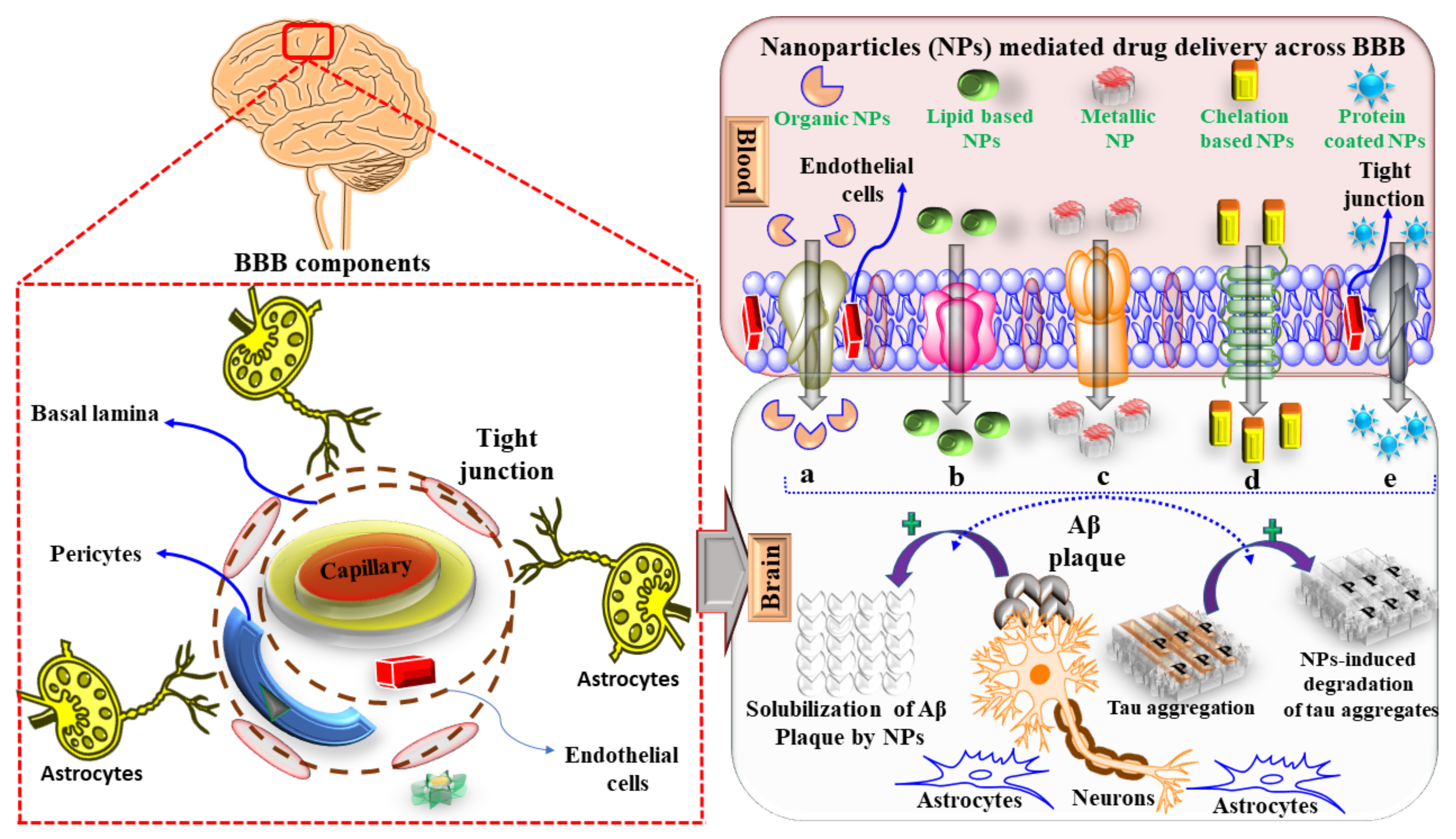
5.1. Targeted Drug Delivery into the CNS and Challenges to Cross the BBB
5.2. Microglia and the Uptake of Nanocarriers
5.3. Mechanism of Nanocarriers for Targeting Microglial Activation in AD
6. Nanocarrier Based Drug Delivery for the Treatment of AD
6.1. Polymeric-Based NPs
6.2. Nanomicellar
6.3. Dendrimers and Nanogels
6.4. Lipid-Based NPs for AD Therapy
6.4.1. Solid Lipid Nanoparticles
6.4.2. Liposomes
6.4.3. Niosomes
6.4.4. Nanoemulsion and Cubosomes
6.5. Metallic NPs
6.5.1. Selenium and Cerium NPs
6.5.2. Gold NPs
6.6. NP-Chelation-Based AD Therapy
6.7. Protein and Antibody-Coated NPs
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iqubal, A.; Rahman, S.O.; Ahmed, M.; Bansal, P.; Haider, R.; Iqubal, M.K.; Najmi, A.K.; Pottoo, F.H.; Haque, S.E. current quest in natural bioactive compounds for Alzheimer’s disease: Multi-targeted-designed-ligand based approach with preclinical and clinical based evidence. Curr. Drug Targets 2021, 22, 685–720. [Google Scholar] [CrossRef]
- Calabrò, M.; Rinaldi, C.; Santoro, G.; Crisafulli, C. The biological pathways of Alzheimer disease: A review. AIMS Neurosci. 2021, 8, 86–132. [Google Scholar] [CrossRef]
- Ashford, M.T.; Veitch, D.P.; Neuhaus, J.; Nosheny, R.L.; Tosun, D.; Weiner, M.W. The search for a convenient procedure to detect one of the earliest signs of Alzheimer’s disease: A systematic review of the prediction of brain amyloid status. Alzheimer’s Dement. 2021, 17, 866–887. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, D.; Nordberg, A.; Westman, E. Biological subtypes of Alzheimer disease: A systematic review and meta-analysis. Neurology 2021, 96, 238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leng, F.; Edison, P. Neuroinflammation and microglial activation in Alzheimer disease: Where do we go from here? Nat. Rev. Neurol. 2020, 17, 157–172. [Google Scholar] [CrossRef]
- Kabra, A.; Ramteke, A. Alzheimer disease—Etiology, pathophysiology and treatment: An overview. Ann. Rom. Soc. Cell Biol. 2021, 25, 3548–3556. [Google Scholar]
- Iqubal, A.; Iqubal, M.K.; Fazal, S.A.; Pottoo, F.H.; Haque, S.E. Nutraceuticals and their Derived Nano-formulations for the Prevention and Treatment of Alzheimer’s disease. Curr. Mol. Pharmacol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Yiannopoulou, K.G.; Papageorgiou, S.G. Current and future treatments in Alzheimer disease: An update. J. Cent. Nerv. Syst. Dis. 2020, 12. [Google Scholar] [CrossRef] [Green Version]
- Uddin, S.; Kabir, T.; Jeandet, P.; Mathew, B.; Ashraf, G.M.; Perveen, A.; Bin-Jumah, M.N.; Mousa, S.A.; Abdel-Daim, M.M. Novel anti-alzheimer’s therapeutic molecules targeting amyloid precursor protein processing. Oxidative Med. Cell. Longev. 2020, 2020, 7039138. [Google Scholar] [CrossRef] [PubMed]
- Nous, A.; Engelborghs, S.; Smolders, I. Melatonin levels in the Alzheimer’s disease continuum: A systematic review. Alzheimer’s Res. Ther. 2021, 13, 52. [Google Scholar] [CrossRef]
- Misrani, A.; Tabassum, S.; Yang, L. Mitochondrial dysfunction and oxidative stress in Alzheimer’s disease. Front. Aging Neurosci. 2021, 13, 57. [Google Scholar] [CrossRef]
- Iqubal, A.; Syed, M.A.; Najmi, A.K.; Ali, J.; Haque, S.E. Ameliorative effect of nerolidol on cyclophosphamide-induced gonadal toxicity in Swiss Albino mice: Biochemical-, histological-and immunohistochemical-based evidences. Andrologia 2020, 52, e13535. [Google Scholar] [CrossRef] [PubMed]
- Regen, F.; Hellmann-Regen, J.; Costantini, E.; Reale, M. Neuroinflammation and Alzheimer’s disease: Implications for microglial activation. Curr. Alzheimer Res. 2017, 14, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- Streit, W.J. Microglial activation and neuroinflammation in Alzheimer’s disease: A critical examination of recent history. Front. Aging Neurosci. 2010, 2, 22. [Google Scholar] [CrossRef] [Green Version]
- Schlachetzki, J.; Hull, M. Microglial activation in Alzheimer’s disease. Curr. Alzheimer Res. 2009, 6, 554–563. [Google Scholar] [CrossRef]
- Cheignon, C.; Tomas, M.; Bonnefont-Rousselot, D.; Faller, P.; Hureau, C.; Collin, F. Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Boil. 2018, 14, 450–464. [Google Scholar] [CrossRef] [PubMed]
- Simunkova, M.; Alwasel, S.H.; Alhazza, I.M.; Jomova, K.; Kollar, V.; Rusko, M.; Valko, M. Management of oxidative stress and other pathologies in Alzheimer’s disease. Arch. Toxicol. 2019, 93, 2491–2513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cassidy, L.; Fernandez, F.; Johnson, J.B.; Naiker, M.; Owoola, A.G.; Broszczak, D.A. Oxidative stress in Alzheimer’s disease: A review on emergent natural polyphenolic therapeutics. Complement. Ther. Med. 2020, 49, 102294. [Google Scholar] [CrossRef] [PubMed]
- Delgado, A.; Cholevas, C.; Theoharides, T.C. Neuroinflammation in Alzheimer’s disease and beneficial action of luteolin. Biofactors 2021, 47, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Nagele, R.G.; Wegiel, J.; Venkataraman, V.; Imaki, H.; Wang, K.-C.; Wegiel, J. Contribution of glial cells to the development of amyloid plaques in Alzheimer’s disease. Neurobiol. Aging 2004, 25, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Hansen, D.V.; Hanson, J.E.; Sheng, M. Microglia in Alzheimer’s disease. J. Cell Biol. 2018, 217, 459–472. [Google Scholar] [CrossRef] [PubMed]
- Solito, E.; Sastre, M. Microglia function in Alzheimer’s disease. Front. Pharmacol. 2012, 3, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiao, O.; Ji, H.; Zhang, Y.; Zhang, X.; Zhang, X.; Liu, N.; Huang, L.; Liu, C.; Gao, W. New insights in drug development for Alzheimer’s disease based on microglia function. Biomed. Pharmacother. 2021, 111703. [Google Scholar] [CrossRef]
- Perea, J.R.; Bolós, M.; Avila, J. Microglia in Alzheimer’s disease in the context of tau pathology. Biomolecules 2020, 10, 1439. [Google Scholar] [CrossRef] [PubMed]
- Orihuela, R.; McPherson, C.A.; Harry, G.J. Microglial M1/M2 polarization and metabolic states. Br. J. Pharmacol. 2016, 173, 649–665. [Google Scholar] [CrossRef]
- Yao, K.; Zu, H.-B. Microglial polarization: Novel therapeutic mechanism against Alzheimer’s disease. Inflammopharmacology 2020, 28, 95–110. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.-S.; Tan, Z.-X.; Wu, L.-Y.; Dong, F.; Zhang, F. The involvement of NLRP3 inflammasome in the treatment of Alzheimer’s disease. Ageing Res. Rev. 2020, 64, 101192. [Google Scholar] [CrossRef] [PubMed]
- Milner, M.T.; Maddugoda, M.; Götz, J.; Burgener, S.S.; Schroder, K. The NLRP3 inflammasome triggers sterile neuroinflammation and Alzheimer’s disease. Curr. Opin. Immunol. 2021, 68, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Hanslik, K.L.; Ulland, T.K. The role of microglia and the Nlrp3 Inflammasome in Alzheimer’s disease. Front. Neurol. 2020, 11, 1063. [Google Scholar] [CrossRef] [PubMed]
- Iqubal, A.; Syed, M.A.; Najmi, A.K.; Azam, F.; Barreto, G.E.; Iqubal, M.K.; Ali, J.; Haque, S.E. Nano-engineered nerolidol loaded lipid carrier delivery system attenuates cyclophosphamide neurotoxicity—Probable role of NLRP3 inflammasome and caspase-1. Exp. Neurol. 2020, 334, 113464. [Google Scholar] [CrossRef]
- He, X.-F.; Xu, J.-H.; Li, G.; Li, M.-Y.; Li, L.-L.; Pei, Z.; Zhang, L.-Y.; Hu, X.-Q. NLRP3-dependent microglial training impaired the clearance of amyloid-beta and aggravated the cognitive decline in Alzheimer’s disease. Cell Death Dis. 2020, 11, 849. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wise, L.; Fukuchi, K.-I. TLR4 Cross-talk with NLRP3 inflammasome and complement signaling pathways in Alzheimer’s disease. Front. Immunol. 2020, 11, 724. [Google Scholar] [CrossRef]
- Maphis, N.; Xu, G.; Kokiko-Cochran, O.N.; Jiang, S.; Cardona, A.; Ransohoff, R.M.; Lamb, B.T.; Bhaskar, K. Reactive microglia drive tau pathology and contribute to the spreading of pathological tau in the brain. Brain 2015, 138, 1738–1755. [Google Scholar] [CrossRef]
- Carroll, J.C.; Iba, M.; Bangasser, D.A.; Valentino, R.J.; James, M.J.; Brunden, K.R.; Lee, V.M.-Y.; Trojanowski, J.Q.; Bangasser, D.A. Chronic stress exacerbates tau pathology, neurodegeneration, and cognitive performance through a corticotropin-releasing factor receptor-dependent mechanism in a transgenic mouse model of tauopathy. J. Neurosci. 2011, 31, 14436–14449. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ising, C.; Venegas, C.; Zhang, S.; Scheiblich, H.; Schmidt, S.V.; Vieira-Saecker, A.; Schwartz, S.; Albasset, S.; McManus, R.M.; Tejera, D.; et al. NLRP3 inflammasome activation drives tau pathology. Nature 2019, 575, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Pooler, A.M.; Polydoro, M.; Wegmann, S.; Nicholls, S.B.; Spires-Jones, T.L.; Hyman, B.T. Propagation of tau pathology in Alzheimer’s disease: Identification of novel therapeutic targets. Alzheimer’s Res. Ther. 2013, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Garcez, M.L.; Mina, F.; Bellettini-Santos, T.; Carneiro, F.G.; Luz, A.P.; Schiavo, G.L.; Andrighetti, M.S.; Scheid, M.G.; Bolfe, R.P.; Budni, J. Minocycline reduces inflammatory parameters in the brain structures and serum and reverses memory impairment caused by the administration of amyloid β (1-42) in mice. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2017, 77, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Elewa, H.F.; Hilali, H.; Hess, D.C.; Machado, S.; Fagan, S.C. Minocycline for short-term neuroprotection. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2006, 26, 515–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maniar, K.H.; Jones, I.A.; Gopalakrishna, R.; Vangsness, C.T., Jr. Lowering side effects of NSAID usage in osteoarthritis: Recent attempts at minimizing dosage. Expert Opin. Pharmacother. 2018, 19, 93–102. [Google Scholar] [CrossRef]
- Thawkar, B.S.; Kaur, G. Inhibitors of NF-κB and P2X7/NLRP3/Caspase 1 pathway in microglia: Novel therapeutic opportunities in neuroinflammation induced early-stage Alzheimer’s disease. J. Neuroimmunol. 2019, 326, 62–74. [Google Scholar] [CrossRef]
- Flores, J.; Noël, A.; Foveau, B.; Lynham, J.; Lecrux, C.; Leblanc, A.C. Caspase-1 inhibition alleviates cognitive impairment and neuropathology in an Alzheimer’s disease mouse model. Nat. Commun. 2018, 9, 3916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tobinick, E.L.; Gross, H. Rapid improvement in verbal fluency and aphasia following perispinal etanercept in Alzheimer’s disease. BMC Neurol. 2008, 8, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butchart, J.; Brook, L.; Hopkins, V.; Teeling, J.; Püntener, U.; Culliford, D.; Sharples, R.; Sharif, S.; McFarlane, B.; Raybould, R.; et al. Etanercept in Alzheimer disease: A randomized, placebo-controlled, double-blind, phase 2 trial. Neurology 2015, 84, 2161–2168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitazawa, M.; Cheng, D.; Tsukamoto, M.R.; Koike, M.A.; Wes, P.; Vasilevko, V.; Cribbs, D.H.; LaFerla, F.M. Blocking IL-1 signaling rescues cognition, attenuates tau pathology, and restores neuronal β-Catenin pathway function in an Alzheimer’s disease model. J. Immunol. 2011, 187, 6539–6549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grimaldi, L.M.E.; Zappalà, G.; Iemolo, F.; Castellano, A.E.; Ruggieri, S.; Bruno, G.; Paolillo, A. A pilot study on the use of interferon beta-1a in early Alzheimer’s disease subjects. J. Neuroinflamm. 2014, 11, 30. [Google Scholar] [CrossRef] [Green Version]
- Moussa, C.; Hebron, M.; Huang, X.; Ahn, J.; Rissman, R.A.; Aisen, P.S.; Turner, R. Resveratrol regulates neuro-inflammation and induces adaptive immunity in Alzheimer’s disease. J. Neuroinflamm. 2017, 14, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandrekar-Colucci, S.; Landreth, G.E. Nuclear receptors as therapeutic targets for Alzheimer’s disease. Expert Opin. Ther. Targets 2011, 15, 1085–1097. [Google Scholar] [CrossRef]
- Gold, M.; Alderton, C.; Zvartau-Hind, M.; Egginton, S.; Saunders, A.M.; Irizarry, M.; Craft, S.; Landreth, G.; Linnamägi, Ü.; Sawchak, S. Rosiglitazone monotherapy in mild-to-moderate Alzheimer’s disease: Results from a randomized, double-blind, placebo-controlled phase III study. Dement. Geriatr. Cogn. Disord. 2010, 30, 131–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yin, J.; Zhao, F.; Chojnacki, J.; Fulp, J.; Klein, W.L.; Zhang, S.; Zhu, X. NLRP3 inflammasome inhibitor ameliorates amyloid pathology in a mouse model of Alzheimer’s disease. Mol. Neurobiol. 2018, 55, 1977–1987. [Google Scholar] [CrossRef] [PubMed]
- Prins, N.D.; Harrison, J.E.; Chu, H.-M.; Blackburn, K.; Alam, J.J.; Scheltens, P. A phase 2 double-blind placebo-controlled 24-week treatment clinical study of the p38 alpha kinase inhibitor neflamapimod in mild Alzheimer’s disease. Alzheimer’s Res. Ther. 2021, 13, 106. [Google Scholar] [CrossRef]
- Wang, T.; Kuang, W.; Chen, W.; Xu, W.; Zhang, L.; Li, Y.; Li, H.; Peng, Y.; Chen, Y.; Wang, B.; et al. A phase II randomized trial of sodium oligomannate in Alzheimer’s dementia. Alzheimer’s Res. Ther. 2020, 12, 110. [Google Scholar] [CrossRef] [PubMed]
- Navarro, G.; Escuela, D.O.B.; Angelats, E.; Etayo, Í.; Reyes-Resina, I.; Pulido-Salgado, M.; Rodríguez-Pérez, A.I.; Canela, E.I.; Saura, J.; Lanciego, J.L.; et al. Receptor-heteromer mediated regulation of endocannabinoid signaling in activated microglia. Role of CB1 and CB2 receptors and relevance for Alzheimer’s disease and levodopa-induced dyskinesia. Brain Behav. Immun. 2018, 67, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, N.; Nadjafi, S.; Janani, L.; Faraji, Z.; Ashtari, B.; Jameie, S.B.; Farhadi, M.; Isfahani, B.O. A systematic review and meta-analysis of the role of toll-like receptor 9 in Alzheimer’s Disease: The protocol for a systematic review. Thrita 2021, 10, e114260. [Google Scholar] [CrossRef]
- Dasu, M.R.; Riosvelasco, A.C.; Jialal, I. Candesartan inhibits Toll-like receptor expression and activity both in vitro and in vivo. Atherosclerosis 2009, 202, 76–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acuña, L.; Hamadat, S.; Corbalán, N.S.; González-Lizárraga, F.; dos-Santos-Pereira, M.; Rocca, J.; Sepúlveda Díaz, J.; Del-Bel, E.; Papy-García, D.; Chehín, R.N.; et al. Rifampicin and its derivative rifampicin quinone reduce microglial inflammatory responses and neurodegeneration induced in vitro by α-synuclein fibrillary aggregates. Cells 2019, 8, 776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hughes, C.D.; Choi, M.L.; Ryten, M.; Hopkins, L.; Drews, A.; Botía, J.A.; Iljina, M.; Rodrigues, M.; Gagliano, S.A.; Gandhi, S.; et al. Picomolar concentrations of oligomeric alpha-synuclein sensitizes TLR4 to play an initiating role in Parkinson’s disease pathogenesis. Acta Neuropathol. 2019, 137, 103–120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ojha, S.; Javed, H.; Azimullah, S.; Haque, M.E. β-Caryophyllene, a phytocannabinoid attenuates oxidative stress, neuroinflammation, glial activation, and salvages dopaminergic neurons in a rat model of Parkinson disease. Mol. Cell. Biochem. 2016, 418, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Wan, H.-L.; Hong, X.-Y.; Zhao, Z.-H.; Li, T.; Zhang, B.-G.; Liu, Q.; Wang, Q.; Zhao, S.; Wang, J.-Z.; Shen, X.-F. STAT3 ameliorates cognitive deficits via regulation of NMDAR expression in an Alzheimer’s disease animal model. Theranostics 2021, 11, 5511. [Google Scholar] [CrossRef]
- Kim, B.-W.; Koppula, S.; Kumar, H.; Park, J.-Y.; Kim, I.-W.; More, S.V.; Kim, I.-S.; Han, S.-D.; Kim, S.-K.; Yoon, S.-H. α-Asarone attenuates microglia-mediated neuroinflammation by inhibiting NF kappa B activation and mitigates MPTP-induced behavioral deficits in a mouse model of Parkinson’s disease. Neuropharmacology 2015, 97, 46–57. [Google Scholar] [CrossRef]
- Essa, M.M.; Vijayan, R.K.; Castellano-Gonzalez, G.; Memon, M.A.; Braidy, N.; Guillemin, G.J. Neuroprotective Effect of Natural Products Against Alzheimer’s Disease. Neurochem. Res. 2012, 37, 1829–1842. [Google Scholar] [CrossRef]
- Liu, C.-Y.; Wang, X.; Liu, C.; Zhang, H.-L. Pharmacological targeting of microglial activation: New therapeutic approach. Front. Cell. Neurosci. 2019, 13, 514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, Y.C.; Shin, W.-H.; Baek, J.Y.; Cho, E.J.; Baik, H.H.; Kim, S.R.; Won, S.-Y.; Jin, B.K. CB2 receptor activation prevents glial-derived neurotoxic mediator production, BBB leakage and peripheral immune cell infiltration and rescues dopamine neurons in the MPTP model of Parkinson’s disease. Exp. Mol. Med. 2016, 48, e205. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Jing, H.; Yang, H.; Liu, Z.; Guo, H.; Chai, L.; Hu, L. Tanshinone I selectively suppresses pro-inflammatory genes expression in activated microglia and prevents nigrostriatal dopaminergic neurodegeneration in a mouse model of Parkinson’s disease. J. Ethnopharmacol. 2015, 164, 247–255. [Google Scholar] [CrossRef]
- Zhang, F.; Shi, J.-S.; Zhou, H.; Wilson, B.; Hong, J.-S.; Gao, H.-M. Resveratrol protects dopamine neurons against lipopolysaccharide-induced neurotoxicity through its anti-inflammatory actions. Mol. Pharmacol. 2010, 78, 466–477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moon, M.; Kim, H.G.; Hwang, L.; Seo, J.-H.; Kim, S.; Hwang, S.; Kim, S.; Lee, D.; Chung, H.; Oh, M.S.; et al. Neuroprotective effect of ghrelin in the 1-Methyl-4-Phenyl-1,2,3,6-Tetrahydropyridine mouse model of Parkinson’s disease by blocking microglial activation. Neurotox. Res. 2009, 15, 332–347. [Google Scholar] [CrossRef]
- Zhou, P.; Homberg, J.R.; Fang, Q.; Wang, J.; Li, W.; Meng, X.; Shen, J.; Luan, Y.; Liao, P.; Swaab, D.F.; et al. Histamine-4 receptor antagonist JNJ7777120 inhibits pro-inflammatory microglia and prevents the progression of Parkinson-like pathology and behaviour in a rat model. Brain Behav. Immun. 2019, 76, 61–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gordon, R.; Albornoz, E.A.; Christie, D.C.; Langley, M.R.; Kumar, V.; Mantovani, S.; Robertson, A.A.; Butler, M.S.; Rowe, D.B.; O’Neill, L.A. Inflammasome inhibition prevents α-synuclein pathology and dopaminergic neurodegeneration in mice. Sci. Transl. Med. 2018, 10, 1–26. [Google Scholar] [CrossRef] [Green Version]
- Schwenkgrub, J.; Joniec-Maciejak, I.; Sznejder-Pachołek, A.; Wawer, A.; Ciesielska, A.; Bankiewicz, K.; Czlonkowska, A.; Członkowski, A. Effect of human interleukin-10 on the expression of nitric oxide synthases in the MPTP-based model of Parkinson’s disease. Pharmacol. Rep. 2013, 65, 44–49. [Google Scholar] [CrossRef]
- Kinoshita, K.-I.; Muroi, Y.; Unno, T.; Ishii, T. Rolipram improves facilitation of contextual fear extinction in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced mouse model of Parkinson’s disease. J. Pharmacol. Sci. 2017, 134, 55–58. [Google Scholar] [CrossRef]
- Calvello, R.; Cianciulli, A.; Nicolardi, G.; De Nuccio, F.; Giannotti, L.; Salvatore, R.; Porro, C.; Trotta, T.; Panaro, M.A.; Lofrumento, D.D. Vitamin D treatment attenuates neuroinflammation and dopaminergic neurodegeneration in an animal model of parkinson’s disease, shifting M1 to M2 microglia responses. J. Neuroimmune Pharmacol. 2016, 12, 327–339. [Google Scholar] [CrossRef]
- Iqubal, A.; Iqubal, M.K.; Hoda, F.; Najmi, A.K.; Haque, S.E. COVID-19 and cardiovascular complications: An update from the underlying mechanism to consequences and possible clinical intervention. Expert Rev. Anti-Infect. Ther. 2021, 19, 1083–1092. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Zheng, J.; Li, S.; Li, H.; Zhou, Y.; Zheng, W.; Zhang, M.; Liu, L.; Chen, Z. GLP-1 improves the neuronal supportive ability of astrocytes in Alzheimer’s disease by regulating mitochondrial dysfunction via the cAMP/PKA pathway. Biochem. Pharmacol. 2021, 188, 114578. [Google Scholar] [CrossRef]
- Ghosh, M.; Xu, Y.; Pearse, D.D. Cyclic AMP is a key regulator of M1 to M2a phenotypic conversion of microglia in the presence of Th2 cytokines. J. Neuroinflamm. 2016, 13, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, S.; Yang, J.; Wang, L.; Peng, S.; Yin, J.; Jia, L.; Yang, X.; Yuan, Z.; Wu, C. NF-κB upregulates type 5 phosphodiesterase in N9 microglial cells: Inhibition by sildenafil and yonkenafil. Mol. Neurobiol. 2016, 53, 2647–2658. [Google Scholar] [CrossRef] [PubMed]
- Pifarre, P.; Prado, J.; Baltrons, M.A.; Giralt, M.; Gabarro, P.; Feinstein, D.L.; Hidalgo, J.; Garcia, A. Sildenafil (Viagra) ameliorates clinical symptoms and neuropathology in a mouse model of multiple sclerosis. Acta Neuropathol. 2011, 121, 499–508. [Google Scholar] [CrossRef]
- Xu, Y.; Zhu, N.; Xu, W.; Ye, H.; Liu, K.; Wu, F.; Zhang, M.; Ding, Y.; Zhang, C.; Zhang, H.; et al. Inhibition of phosphodiesterase-4 reverses Aβ-induced memory impairment by regulation of HPA axis related cAMP signaling. Front. Aging Neurosci. 2018, 10, 204. [Google Scholar] [CrossRef]
- Swanson, C.R.; Joers, V.; Bondarenko, V.; Brunner, K.; Simmons, H.A.; Ziegler, T.E.; Kemnitz, J.W.; Johnson, J.A.; Emborg, M.E. The PPAR-γ agonist pioglitazone modulates inflammation and induces neuroprotection in parkinsonian monkeys. J. Neuroinflamm. 2011, 8, 91. [Google Scholar] [CrossRef] [Green Version]
- Song, G.J.; Nam, Y.; Jo, M.; Jung, M.; Koo, J.Y.; Cho, W.; Koh, M.; Park, S.B.; Suk, K. A novel small-molecule agonist of PPAR-γ potentiates an anti-inflammatory M2 glial phenotype. Neuropharmacology 2016, 109, 159–169. [Google Scholar] [CrossRef]
- Joniec-Maciejak, I.; Ciesielska, A.; Wawer, A.; Sznejder-Pachołek, A.; Schwenkgrub, J.; Cudna, A.; Hadaczek, P.; Bankiewicz, K.S.; Członkowska, A.; Członkowski, A. The influence of AAV2-mediated gene transfer of human IL-10 on neurodegeneration and immune response in a murine model of Parkinson’s disease. Pharmacol. Rep. 2014, 66, 660–669. [Google Scholar] [CrossRef]
- Ouyang, Q.; Meng, Y.; Zhou, W.; Tong, J.; Cheng, Z.; Zhu, Q. New advances in brain-targeting nano-drug delivery systems for Alzheimer’s disease. J. Drug Target. 2021, 1–67. [Google Scholar] [CrossRef]
- Cunha, S.; Forbes, B.; Lobo, J.M.S.; Silva, A.C. Improving drug delivery for Alzheimer’s disease through nose-to-brain delivery using nanoemulsions, Nanostructured Lipid Carriers (NLC) and in situ hydrogels. Int. J. Nanomed. 2021, 16, 4373–4390. [Google Scholar] [CrossRef]
- Pottoo, F.H.; Sharma, S.; Javed, M.N.; Barkat, M.A.; Harshita; Alam, M.S.; Naim, M.J.; Alam, O.; Ansari, M.A.; Barreto, G.E. Lipid-based nanoformulations in the treatment of neurological disorders. Drug Metab. Rev. 2020, 52, 185–204. [Google Scholar] [CrossRef] [PubMed]
- Mohi-Ud-Din, R.; Mir, R.H.; Wani, T.U.; Shah, A.J.; Mohi-Ud-Din, I.; Dar, M.A.; Pottoo, F.H. Novel drug delivery system for curcumin: Implementation to improve therapeutic efficacy against neurological disorders. Comb. Chem. High Throughput Screen. 2021, 24, 1. [Google Scholar] [CrossRef]
- Mir, R.H.; Shah, A.J.; Mohi-Ud-Din, R.; Potoo, F.H.; Dar, M.A.; Jachak, S.M.; Masoodi, M.H. Natural Anti-inflammatory compounds as Drug candidates in Alzheimer’s disease. Curr. Med. Chem. 2020, 28, 4799–4825. [Google Scholar] [CrossRef]
- Javed, M.N.; Dahiya, E.S.; Ibrahim, A.M.; Alam, M.S.; Khan, F.A.; Pottoo, F.H. recent advancement in clinical application of nanotechnological approached targeted delivery of herbal drugs. In Nanophytomedicine; Springer: Berlin/Heidelberg, Germany, 2020; pp. 151–172. [Google Scholar]
- Sharma, S.; Rabbani, S.A.; Agarwal, T.; Baboota, S.; Pottoo, F.H.; Kadian, R. Nanotechnology driven approaches for the management of parkinson’s disease: Current status and future perspectives. Curr. Drug Metab. 2021, 22, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Zhao, J.; Wang, H.; Jiang, Y.; Yang, Q.; Fu, Y.; Zeng, H.; Hölscher, C.; Xu, J.; Zhang, Z. Magnolol alleviates Alzheimer’s disease-like pathology in transgenic C. elegans by promoting microglia phagocytosis and the degradation of beta-amyloid through activation of PPAR-γ. Biomed. Pharmacother. 2020, 124, 109886. [Google Scholar] [CrossRef]
- Yang, Z.; Kuboyama, T.; Tohda, C. Naringenin promotes microglial M2 polarization and Aβ degradation enzyme expression. Phytother. Res. 2019, 33, 1114–1121. [Google Scholar] [CrossRef]
- Huang, C.; Dong, D.; Jiao, Q.; Pan, H.; Ma, L.; Wang, R. Sarsasapogenin-AA 13 ameliorates Aβ-induced cognitive deficits via improving neuroglial capacity on Aβ clearance and antiinflammation. CNS Neurosci. Ther. 2017, 23, 498–509. [Google Scholar] [CrossRef] [Green Version]
- He, P.; Yan, S.; Zheng, J.; Gao, Y.; Zhang, S.; Liu, Z.; Liu, X.; Xiao, C. Eriodictyol attenuates LPS-induced neuroinflammation, amyloidogenesis, and cognitive impairments via the inhibition of NF-κB in male C57BL/6J mice and BV2 microglial cells. J Agric. Food Chem. 2018, 66, 10205–10214. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Huo, X.; Liu, W.; Li, K.; Sun, Z.; Tian, J. Apigenin exhibits anti-inflammatory effects in LPS-stimulated BV2 microglia through activating GSK3β/Nrf2 signaling pathway. Immunopharmacol. Immunotoxicol. 2019, 42, 9–16. [Google Scholar] [CrossRef]
- Feng, J.; Wang, J.; Du, Y.; Liu, Y.; Zhang, W.; Chen, J.; Liu, Y.; Zheng, M.; Wang, K.; He, G. Dihydromyricetin inhibits microglial activation and neuroinflammation by suppressing NLRP 3 inflammasome activation in APP/PS 1 transgenic mice. CNS Neurosci. Ther. 2018, 24, 1207–1218. [Google Scholar] [CrossRef]
- Zhou, J.; Deng, Y.; Li, F.; Yin, C.; Shi, J.; Gong, Q. Icariside II attenuates lipopolysaccharide-induced neuroinflammation through inhibiting TLR4/MyD88/NF-κB pathway in rats. Biomed. Pharmacother. 2019, 111, 315–324. [Google Scholar] [CrossRef]
- Xie, L.; Gu, Z.; Liu, H.; Jia, B.; Wang, Y.; Cao, M.; Song, R.; Zhang, Z.; Bian, Y. The anti-depressive effects of hesperidin and the relative mechanisms based on the NLRP3 inflammatory signaling pathway. Front. Pharmacol. 2020, 11, 1251. [Google Scholar] [CrossRef]
- Jin, G.; Bai, D.; Yin, S.; Yang, Z.; Zou, D.; Zhang, Z.; Li, X.; Sun, Y.; Zhu, Q. Silibinin rescues learning and memory deficits by attenuating microglia activation and preventing neuroinflammatory reactions in SAMP8 mice. Neurosci. Lett. 2016, 629, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Hou, J.; Zhou, Z.; Ren, M.; Mo, Y.; Yang, G.; Qu, Z.; Hu, Y. Safflower yellow improves synaptic plasticity in APP/PS1 mice by regulating microglia activation phenotypes and BDNF/TrkB/ERK signaling pathway. NeuroMolecular Med. 2020, 22, 341–358. [Google Scholar] [CrossRef]
- Gao, F.; Shen, J.; Zhao, L.; Hao, Q.; Yang, Y. Curcumin alleviates lipopolysaccharide (LPS)-activated neuroinflammation via modulation of miR-199b-5p/IκB kinase β (IKKβ)/nuclear factor kappa B (NF-κB) pathway in microglia. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2019, 25, 9801. [Google Scholar] [CrossRef]
- Bao, Y.; Chen, Q.; Xie, Y.; Tao, Z.; Jin, K.; Chen, S.; Bai, Y.; Yang, J.; Shan, S. Ferulic acid attenuates oxidative DNA damage and inflammatory responses in microglia induced by benzo(a)pyrene. Int. Immunopharmacol. 2019, 77, 105980. [Google Scholar] [CrossRef]
- Moon, M.; Kim, H.G.; Choi, J.G.; Oh, H.; Lee, P.K.; Ha, S.K.; Kim, S.Y.; Park, Y.; Huh, Y.; Oh, M.S. 6-Shogaol, an active constituent of ginger, attenuates neuroinflammation and cognitive deficits in animal models of dementia. Biochem. Biophys. Res. Commun. 2014, 449, 8–13. [Google Scholar] [CrossRef]
- Singh, N.A.; Bhardwaj, V.; Ravi, C.; Ramesh, N.; Mandal, A.K.A.; Khan, Z.A. EGCG nanoparticles attenuate aluminum chloride induced neurobehavioral deficits, beta amyloid and tau pathology in a rat model of Alzheimer’s disease. Front. Aging Neurosci. 2018, 10, 244. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-P.; Yin, H.; Lin, Q.; Fang, S.-P.; Shen, J.-H.; Wu, Y.-F.; Su, S.-H.; Hai, J. Andrographolide enhances hippocampal BDNF signaling and suppresses neuronal apoptosis, astroglial activation, neuroinflammation, and spatial memory deficits in a rat model of chronic cerebral hypoperfusion. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2019, 392, 1277–1284. [Google Scholar] [CrossRef]
- Wang, X.; Gai, Y.-N.; Li, B.-B.; Huang, L.-L. Andalucin from Artemisia lannta suppresses the neuroinflammation via the promotion of Nrf2-mediated HO-1 levels by blocking the p65–p300 interaction in LPS-activated BV2 microglia. Phytomedicine 2018, 51, 226–232. [Google Scholar] [CrossRef]
- Castellano, J.M.; Garcia-Rodriguez, S.; Espinosa, J.M.; Millan-Linares, M.C.; Rada, M.; Perona, J.S. Oleanolic acid exerts a neuroprotective effect against microglial cell activation by modulating cytokine release and antioxidant defense systems. Biomolecules 2019, 9, 683. [Google Scholar] [CrossRef] [Green Version]
- Kim, N.; Do, J.; Bae, J.-S.; Jin, H.K.; Kim, J.-H.; Inn, K.-S.; Oh, M.S.; Lee, J.K. Piperlongumine inhibits neuroinflammation via regulating NF-κB signaling pathways in lipopolysaccharide-stimulated BV2 microglia cells. J. Pharmacol. Sci. 2018, 137, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Hou, J.; Mo, Y.; Ren, M.; Yang, G.; Qu, Z.; Hu, Y. Geniposidic acid ameliorates spatial learning and memory deficits and alleviates neuroinflammation via inhibiting HMGB-1 and downregulating TLR4/2 signaling pathway in APP/PS1 mice. Eur. J. Pharmacol. 2020, 869, 172857. [Google Scholar] [CrossRef]
- Chen, M.; Chang, Y.y.; Huang, S.; Xiao, L.h.; Zhou, W.; Zhang, L.y.; Li, C.; Zhou, R.p.; Tang, J.; Lin, L. Aromatic-Turmerone attenuates LPS-induced neuroinflammation and consequent memory impairment by targeting TLR4-Dependent signaling pathway. Mol. Nutr. Food Res. 2018, 62, 1700281. [Google Scholar] [CrossRef]
- Rankovic, Z. CNS drug design: Balancing physicochemical properties for optimal brain exposure. J. Med. Chem. 2015, 58, 2584–2608. [Google Scholar] [CrossRef] [PubMed]
- Peñalver, P.; Belmonte-Reche, E.; Adán, N.; Caro, M.; Mateos-Martín, M.L.; Delgado, M.; González-Rey, E.; Morales, J.C. Alkylated resveratrol prodrugs and metabolites as potential therapeutics for neurodegenerative diseases. Eur. J. Med. Chem. 2018, 146, 123–138. [Google Scholar] [CrossRef]
- Agarwal, M.; Alam, M.R.; Haider, M.K.; Malik, Z.; Kim, D.-K. Alzheimer’s disease: An overview of major hypotheses and therapeutic options in nanotechnology. Nanomaterials 2020, 11, 59. [Google Scholar] [CrossRef]
- Gonda, A.; Zhao, N.; Shah, J.V.; Calvelli, H.R.; Kantamneni, H.; Francis, N.L.; Ganapathy, V. Engineering tumor-targeting nanoparticles as vehicles for precision nanomedicine. Med. One 2019, 4, e190021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soares, S.; Sousa, J.; Pais, A.; Vitorino, C. Nanomedicine: Principles, properties, and regulatory issues. Front. Chem. 2018, 6, 360. [Google Scholar] [CrossRef]
- Jia, L.; Nie, X.-Q.; Ji, H.-M.; Yuan, Z.-X.; Li, R.-S. Multiple-Coated PLGA nanoparticles loading triptolide attenuate injury of a cellular model of Alzheimer’s disease. BioMed Res. Int. 2021, 2021, 8825640. [Google Scholar] [CrossRef]
- Petros, R.A.; DeSimone, J.M. Strategies in the design of nanoparticles for therapeutic applications. Nat. Rev. Drug Discov. 2010, 9, 615–627. [Google Scholar] [CrossRef]
- Chang, D.; Lim, M.; Goos, J.; Qiao, R.; Ng, Y.Y.; Mansfeld, F.M.; Jackson, M.; Davis, T.P.; Kavallaris, M. Biologically targeted magnetic hyperthermia: Potential and limitations. Front. Pharmacol. 2018, 9, 831. [Google Scholar] [CrossRef] [Green Version]
- Luan, X.; Sansanaphongpricha, K.; Myers, I.; Chen, H.; Yuan, H.; Sun, D. Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharmacol. Sin. 2017, 38, 754–763. [Google Scholar] [CrossRef] [Green Version]
- Chen, I.-C.; Hsiao, I.-L.; Lin, H.-C.; Wu, C.-H.; Chuang, C.-Y.; Huang, Y.-J. Influence of silver and titanium dioxide nanoparticles on in vitro blood-brain barrier permeability. Environ. Toxicol. Pharmacol. 2016, 47, 108–118. [Google Scholar] [CrossRef]
- Sela, H.; Cohen, H.; Elia, P.; Zach, R.; Karpas, Z.; Zeiri, Y. Spontaneous penetration of gold nanoparticles through the blood brain barrier (BBB). J. Nanobiotechnol. 2015, 13, 71. [Google Scholar] [CrossRef] [Green Version]
- Baghirov, H.; Snipstad, S.; Sulheim, E.; Berg, S.; Hansen, R.; Thorsen, F.; Mørch, Y.; Davies, C.D.L.; Åslund, A.K.O. Ultrasound-mediated delivery and distribution of polymeric nanoparticles in the normal brain parenchyma of a metastatic brain tumour model. PLoS ONE 2018, 13, e0191102. [Google Scholar] [CrossRef] [Green Version]
- Peviani, M.; Palmiero, U.C.; Cecere, F.; Milazzo, R.; Moscatelli, D.; Biffi, A. Biodegradable polymeric nanoparticles administered in the cerebrospinal fluid: Brain biodistribution, preferential internalization in microglia and implications for cell-selective drug release. Biomaterials 2019, 209, 25–40. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Chi, N.; Tang, X. Preparation of estradiol chitosan nanoparticles for improving nasal absorption and brain targeting. Eur. J. Pharm. Biopharm. 2008, 70, 735–740. [Google Scholar] [CrossRef]
- Helmschrodt, C.; Höbel, S.; Schöniger, S.; Bauer, A.; Bonicelli, J.; Gringmuth, M.; Fietz, S.A.; Aigner, A.; Richter, A.; Richter, F. Polyethylenimine nanoparticle-mediated sirna delivery to reduce α-synuclein expression in a model of Parkinson’s disease. Mol. Ther.-Nucleic Acids 2017, 9, 57–68. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez, M.; Lapierre, J.; Ojha, C.R.; Kaushik, A.; Batrakova, E.; Kashanchi, F.; Dever, S.M.; Nair, M.; El-Hage, N. Intranasal drug delivery of small interfering RNA targeting Beclin1 encapsulated with polyethylenimine (PEI) in mouse brain to achieve HIV attenuation. Sci. Rep. 2017, 7, 1862. [Google Scholar] [CrossRef] [Green Version]
- Ulbrich, K.; Knobloch, T.; Kreuter, J. Targeting the insulin receptor: Nanoparticles for drug delivery across the blood–brain barrier (BBB). J. Drug Target. 2010, 19, 125–132. [Google Scholar] [CrossRef]
- Lalani, J.; Raichandani, Y.; Mathur, R.; Lalan, M.; Chutani, K.; Mishra, A.K.; Misra, A. Comparative receptor based brain delivery of tramadol-loaded Poly(lactic-co-glycolic acid) nanoparticles. J. Biomed. Nanotechnol. 2012, 8, 918–927. [Google Scholar] [CrossRef]
- Clark, A.J.; Davis, M.E. Increased brain uptake of targeted nanoparticles by adding an acid-cleavable linkage between transferrin and the nanoparticle core. Proc. Natl. Acad. Sci. USA 2015, 112, 12486–12491. [Google Scholar] [CrossRef] [Green Version]
- Fornaguera, C.; Dols-Perez, A.; Calderó, G.; García-Celma, M.; Camarasa, J.; Solans, C. PLGA nanoparticles prepared by nano-emulsion templating using low-energy methods as efficient nanocarriers for drug delivery across the blood–brain barrier. J. Control. Release 2015, 211, 134–143. [Google Scholar] [CrossRef]
- Ding, H.; Sagar, V.; Agudelo, M.; Pilakka-Kanthikeel, S.; Subba Rao Atluri, V.; Raymond, A.; Samikkannu, T.; Nair, M.P. Enhanced blood–brain barrier transmigration using a novel transferrin embedded fluorescent magneto-liposome nanoformulation. Nanotechnolgy 2014, 25, 055101. [Google Scholar] [CrossRef]
- Behzadi, S.; Serpooshan, V.; Tao, W.; Hamaly, M.A.; Alkawareek, M.Y.; Dreaden, E.C.; Brown, D.; Alkilany, A.M.; Farokhzad, O.C.; Mahmoudi, M. Cellular uptake of nanoparticles: Journey inside the cell. Chem. Soc. Rev. 2017, 46, 4218–4244. [Google Scholar] [CrossRef]
- Foroozandeh, P.; Aziz, A.A. Insight into Cellular Uptake and Intracellular Trafficking of Nanoparticles. Nanoscale Res. Lett. 2018, 13, 339. [Google Scholar] [CrossRef] [PubMed]
- Alnasser, Y.; Kambhampati, S.P.; Nance, E.; Rajbhandari, L.; Shrestha, S.; Venkatesan, A.; Kannan, R.M.; Kannan, S. Preferential and increased uptake of hydroxyl-terminated PAMAM dendrimers by activated microglia in rabbit brain mixed glial culture. Molecules 2018, 23, 1025. [Google Scholar] [CrossRef] [Green Version]
- Zabala, A.; Vazquez-Villoldo, N.; Rissiek, B.; Gejo, J.; Martin, A.; Palomino, A.; Perez-Samartín, A.; Pulagam, K.R.; Lukowiak, M.; Capetillo-Zarate, E.; et al. P2X4 receptor controls microglia activation and favors remyelination in autoimmune encephalitis. EMBO Mol. Med. 2018, 10, 8743. [Google Scholar] [CrossRef]
- Lu, X.; Xu, P.; Ding, H.-M.; Yu, Y.-S.; Huo, D.; Ma, Y.-Q. Tailoring the component of protein corona via simple chemistry. Nat. Commun. 2019, 10, 4520. [Google Scholar] [CrossRef]
- Saha, K.; Rahimi, M.; Yazdani, M.; Kim, S.T.; Moyano, D.F.; Hou, S.; Das, R.; Mout, R.; Rezaee, F.; Mahmoudi, M.; et al. Regulation of macrophage recognition through the interplay of nanoparticle surface functionality and protein corona. ACS Nano 2016, 10, 4421–4430. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Guan, J.; Jiang, Z.; Yang, Y.; Liu, J.; Hua, W.; Mao, Y.; Li, C.; Lu, W.; Qian, J.; et al. Brain-targeted drug delivery by manipulating protein corona functions. Nat. Commun. 2019, 10, 3561. [Google Scholar] [CrossRef] [Green Version]
- Ling, T.S.; Chandrasegaran, S.; Xuan, L.Z.; Suan, T.L.; Elaine, E.; Nathan, D.V.; Chai, Y.H.; Gunasekaran, B.; Salvamani, S. The potential benefits of nanotechnology in treating Alzheimer’s disease. BioMed Res. Int. 2021, 2021, 5550938. [Google Scholar] [CrossRef]
- Franco, R.; Lillo, A.; Rivas-Santisteban, R.; Reyes-Resina, I.; Navarro, G. Microglial adenosine receptors: From preconditioning to modulating the M1/M2 balance in activated cells. Cells 2021, 10, 1124. [Google Scholar] [CrossRef]
- Tiwari, R.K.; Singh, S.; Gupta, C.L.; Bajpai, P. Microglial TLR9: Plausible novel target for therapeutic regime against glioblastoma multiforme. Cell. Mol. Neurobiol. 2021, 41, 1391–1393. [Google Scholar] [CrossRef]
- Sarlus, H.; Heneka, M.T. Microglia in Alzheimer’s disease. J. Clin. Investig. 2017, 127, 3240–3249. [Google Scholar] [CrossRef]
- Shannahan, J.H.; Bai, W.; Brown, J.M. Implications of scavenger receptors in the safe development of nanotherapeutics. Recept. Clin. Investig. 2015, 2, e811. [Google Scholar] [CrossRef]
- Zhao, N.; Francis, N.L.; Calvelli, H.R.; Moghe, P.V. Microglia-targeting nanotherapeutics for neurodegenerative diseases. APL Bioeng. 2020, 4, 030902. [Google Scholar] [CrossRef]
- Yang, L.; Wang, W.; Chen, J.; Wang, N.; Zheng, G. A comparative study of resveratrol and resveratrol-functional selenium nanoparticles: Inhibiting amyloid β aggregation and reactive oxygen species formation properties. J. Biomed. Mater. Res. Part A 2018, 106, 3034–3041. [Google Scholar] [CrossRef]
- Ren, C.; Li, D.; Zhou, Q.; Hu, X. Mitochondria-targeted TPP-MoS2 with dual enzyme activity provides efficient neuroprotection through M1/M2 microglial polarization in an Alzheimer’s disease model. Biomaterials 2020, 232, 119752. [Google Scholar] [CrossRef]
- Vissers, C.; Ming, G.-L.; Song, H. Nanoparticle technology and stem cell therapy team up against neurodegenerative disorders. Adv. Drug Deliv. Rev. 2019, 148, 239–251. [Google Scholar] [CrossRef]
- Carradori, D.; Balducci, C.; Re, F.; Brambilla, D.; le Droumaguet, B.; Flores, O.; Gaudin, A.; Mura, S.; Forloni, G.; Ordoñez-Gutierrez, L. Antibody-functionalized polymer nanoparticle leading to memory recovery in Alzheimer’s disease-like transgenic mouse model, Nanomedicine: Nanotechnology. Biol. Med. 2018, 14, 609–618. [Google Scholar]
- Plaza-Oliver, M.; Santander-Ortega, M.J.; Lozano, M.V. Current approaches in lipid-based nanocarriers for oral drug delivery. Drug Deliv. Transl. Res. 2021, 11, 471–497. [Google Scholar] [CrossRef]
- Sánchez-López, E.; Ettcheto, M.; Egea, M.A.; Espina, M.; Cano, A.; Calpena, A.C.; Camins, A.; Carmona-Ule, N.; Silva, A.M.; Souto, E.B.; et al. Memantine loaded PLGA PEGylated nanoparticles for Alzheimer’s disease: In Vitro and in vivo characterization. J. Nanobiotechnol. 2018, 16, 32. [Google Scholar] [CrossRef]
- Jeon, S.G.; Cha, M.-Y.; Kim, J.-I.; Hwang, T.W.; Kim, K.A.; Kim, T.H.; Song, K.C.; Kim, J.-J.; Moon, M. Vitamin D-binding protein-loaded PLGA nanoparticles suppress Alzheimer’s disease-related pathology in 5XFAD mice. Nanotechnol. Biol. Med. 2019, 17, 297–307. [Google Scholar] [CrossRef]
- Vilella, A.; Belletti, D.; Sauer, A.K.; Hagmeyer, S.; Sarowar, T.; Masoni, M.; Stasiak, N.; Mulvihill, J.; Ruozi, B.; Forni, F.; et al. Reduced plaque size and inflammation in the APP23 mouse model for Alzheimer’s disease after chronic application of polymeric nanoparticles for CNS targeted zinc delivery. J. Trace Elem. Med. Biol. 2018, 49, 210–221. [Google Scholar] [CrossRef]
- Kaur, H.; Mohanta, G.C.; Gupta, V.; Kukkar, D.; Tyagi, S. Synthesis and characterization of ZIF-8 nanoparticles for controlled release of 6-mercaptopurine drug. J. Drug Deliv. Sci. Technol. 2017, 41, 106–112. [Google Scholar] [CrossRef]
- Meng, Q.; Wang, A.; Hua, H.; Jiang, Y.; Wang, Y.; Mu, H.; Wu, Z.; Sun, K. Intranasal delivery of Huperzine A to the brain using lactoferrin-conjugated N-trimethylated chitosan surface-modified PLGA nanoparticles for treatment of Alzheimer’s disease. Int. J. Nanomed. 2018, 13, 705–718. [Google Scholar] [CrossRef] [Green Version]
- Hanafy, A.S.; Farid, R.M.; ElGamal, S.S. Complexation as an approach to entrap cationic drugs into cationic nanoparticles administered intranasally for Alzheimer’s disease management: Preparation and detection in rat brain. Drug Dev. Ind. Pharm. 2015, 41, 2055–2068. [Google Scholar] [CrossRef]
- Xie, J.; Gonzalez-Carter, D.; Tockary, T.A.; Nakamura, N.; Xue, Y.; Nakakido, M.; Akiba, H.; Dirisala, A.; Liu, X.; Toh, K.; et al. Dual-Sensitive nanomicelles enhancing systemic delivery of therapeutically active antibodies specifically into the brain. ACS Nano 2020, 14, 6729–6742. [Google Scholar] [CrossRef]
- Tawfik, S.M.; Azizov, S.; Elmasry, M.R.; Sharipov, M.; Lee, Y.-I. Recent advances in nanomicelles delivery systems. Nanomaterials 2020, 11, 70. [Google Scholar] [CrossRef] [PubMed]
- Muthukumaran, K.; Kanwar, A.; Vegh, C.; Marginean, A.; Elliott, A.; Guilbeault, N.; Badour, A.; Sikorska, M.; Cohen, J.; Pandey, S. Ubisol-Q10 (a nanomicellar water-soluble formulation of CoQ10) treatment inhibits alzheimer-type behavioral and pathological symptoms in a double transgenic mouse (TgAPEswe, PSEN1dE9) model of Alzheimer’s disease. J. Alzheimer’s Dis. 2017, 61, 221–236. [Google Scholar] [CrossRef]
- Hagl, S.; Kocher, A.; Schiborr, C.; Kolesova, N.; Frank, J.; Eckert, G.P. Curcumin micelles improve mitochondrial function in neuronal PC12 cells and brains of NMRI mice—Impact on bioavailability. Neurochem. Int. 2015, 89, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Wang, W.; Dong, X.; Sun, Y. Nitrogen-Doped carbonized polymer dots: A potent scavenger and detector targeting Alzheimer’s β-Amyloid plaques. Small 2020, 16, 2002804. [Google Scholar] [CrossRef]
- Mlynarczyk, D.T.; Dlugaszewska, J.; Kaluzna-Mlynarczyk, A.; Goslinski, T. Dendrimers against fungi—A state of the art review. J. Control. Release 2020, 330, 599–617. [Google Scholar] [CrossRef]
- Gothwal, A.; Kumar, H.; Nakhate, K.; Uddin, A.; Dutta, A.; Borah, A.; Gupta, U. Lactoferrin coupled lower generation PAMAM Dendrimers for brain targeted delivery of memantine in aluminum-chloride-induced Alzheimer’s disease in mice. Bioconjugate Chem. 2019, 30, 2573–2583. [Google Scholar] [CrossRef]
- Igartúa, D.E.; Martinez, C.S.; Alonso, S.D.V.; Prieto, M.J. Combined therapy for alzheimer’s disease: Tacrine and PAMAM dendrimers co-administration reduces the side effects of the drug without modifying its activity. AAPS PharmSciTech 2020, 21, 110–114. [Google Scholar] [CrossRef]
- Patil, R.P.; Pawara, D.D.; Gudewar, C.S.; Tekade, A.R. Nanostructured cubosomes in an in situ nasal gel system: An alternative approach for the controlled delivery of donepezil HCl to brain. J. Liposome Res. 2018, 29, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Aderibigbe, B.A.; Naki, T. Design and efficacy of nanogels formulations for intranasal administration. Molecules 2018, 23, 1241. [Google Scholar] [CrossRef] [Green Version]
- Ashrafi, H.; Azadi, A.; Mohammadi-Samani, S.; Hamidi, M. New candidate delivery system for Alzheimer’s disease: Deferoxamine nanogels. Biointerface Res. Appl. Chem. 2020, 10, 7106–7119. [Google Scholar]
- Ikeda, K.; Okada, T.; Sawada, S.-I.; Akiyoshi, K.; Matsuzaki, K. Inhibition of the formation of amyloid β-protein fibrils using biocompatible nanogels as artificial chaperones. FEBS Lett. 2006, 580, 6587–6595. [Google Scholar] [CrossRef] [Green Version]
- Picone, P.; Sabatino, M.A.; Ditta, L.A.; Amato, A.; Biagio, P.L.S.; Mulè, F.; Giacomazza, D.; Dispenza, C.; Di Carlo, M. Nose-to-brain delivery of insulin enhanced by a nanogel carrier. J. Control. Release 2018, 270, 23–36. [Google Scholar] [CrossRef]
- Costa, C.P.; Moreira, J.N.; Lobo, J.M.S.; Silva, A.C. Intranasal delivery of nanostructured lipid carriers, solid lipid nanoparticles and nanoemulsions: A current overview of in vivo studies. Acta Pharm. Sin. B 2021, 11, 925–940. [Google Scholar] [CrossRef] [PubMed]
- Topal, G.R.; Mészáros, M.; Porkoláb, G.; Szecskó, A.; Polgár, T.F.; Siklós, L.; Deli, M.A.; Veszelka, S.; Bozkir, A. ApoE-targeting increases the transfer of solid lipid nanoparticles with donepezil cargo across a culture model of the blood–brain barrier. Pharmaceutics 2021, 13, 38. [Google Scholar] [CrossRef]
- Arduino, I.; Iacobazzi, R.M.; Riganti, C.; Lopedota, A.A.; Perrone, M.G.; Lopalco, A.; Cutrignelli, A.; Cantore, M.; Laquintana, V.; Franco, M.; et al. Induced expression of P-gp and BCRP transporters on brain endothelial cells using transferrin functionalized nanostructured lipid carriers: A first step of a potential strategy for the treatment of Alzheimer’s disease. Int. J. Pharm. 2020, 591, 120011. [Google Scholar] [CrossRef]
- Yasir, M.; Sara, U.V.S. Solid lipid nanoparticles for nose to brain delivery of haloperidol: In vitro drug release and pharmacokinetics evaluation. Acta Pharm. Sin. B 2014, 4, 454–463. [Google Scholar] [CrossRef] [Green Version]
- Yavarpour-Bali, H.; Ghasemi-Kasman, M.; Pirzadeh, M. Curcumin-loaded nanoparticles: A novel therapeutic strategy in treatment of central nervous system disorders. Int. J. Nanomed. 2019, 14, 4449–4460. [Google Scholar] [CrossRef] [Green Version]
- Malvajerd, S.S.; Izadi, Z.; Azadi, A.; Kurd, M.; Derakhshankhah, H.; Zadeh, M.S.; Javar, H.A.; Hamidi, M. Neuroprotective potential of curcumin-loaded nanostructured lipid carrier in an animal model of Alzheimer’s disease: Behavioral and biochemical evidence. J. Alzheimer’s Dis. 2019, 69, 671–686. [Google Scholar] [CrossRef]
- Malvajerd, S.S.; Azadi, A.; Izadi, Z.; Kurd, M.; Dara, T.; Dibaei, M.; Zadeh, M.S.; Javar, H.A.; Hamidi, M. Brain delivery of curcumin using solid lipid nanoparticles and nanostructured lipid carriers: Preparation, optimization, and pharmacokinetic evaluation. ACS Chem. Neurosci. 2019, 10, 728–739. [Google Scholar] [CrossRef]
- Sharma, A.; Sharma, U.S. Liposomes in drug delivery: Progress and limitations. Int. J. Pharm. 1997, 154, 123–140. [Google Scholar] [CrossRef]
- Rocha, S. Targeted drug delivery across the blood brain barrier in Alzheimer’s disease. Curr. Pharm. Des. 2013, 19, 6635–6646. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rip, J.; Chen, L.; Hartman, R.; Heuvel, A.V.D.; Reijerkerk, A.; Van Kregten, J.; Van Der Boom, B.; Appeldoorn, C.; De Boer, M.; Maussang, D.; et al. Glutathione PEGylated liposomes: Pharmacokinetics and delivery of cargo across the blood–brain barrier in rats. J. Drug Target. 2014, 22, 460–467. [Google Scholar] [CrossRef] [Green Version]
- Régina, A.; Demeule, M.; Ché, C.; Lavallée, I.; Poirier, J.; Gabathuler, R.; Béliveau, R.; Castaigne, J.-P. Antitumour activity of ANG1005, a conjugate between paclitaxel and the new brain delivery vector Angiopep-2. Br. J. Pharmacol. 2008, 155, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Hou, K.; Zhao, J.; Wang, H.; Li, B.; Li, K.; Shi, X.; Wan, K.; Ai, J.; Lv, J.; Wang, D.; et al. Chiral gold nanoparticles enantioselectively rescue memory deficits in a mouse model of Alzheimer’s disease. Nat. Commun. 2020, 11, 4790. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Li, X.-T.; Ni, Y.-N.; Xiao, H.-H.; Yao, Y.-J.; Wang, Y.-Y.; Ju, R.-J.; Li, H.-Y.; Liu, J.-J.; Fu, M.; et al. Transferrin-Modified Osthole pegylated liposomes travel the blood-brain barrier and mitigate alzheimer’s disease-related pathology in APP/PS-1 Mice. Int. J. Nanomed. 2020, 15, 2841–2858. [Google Scholar] [CrossRef] [Green Version]
- Joy, C.; Nair, S.K.; Kumar, K.K.; Dineshkumar, B. Niosomes as nano-carrier based targeted drug delivery system. J. Drug Deliv. Ther. 2021, 11, 166–170. [Google Scholar] [CrossRef]
- Ansari, M.; Eslami, H. Preparation and study of the inhibitory effect of nano-niosomes containing essential oil from artemisia absinthium on amyloid fibril formation. Nanomed. J. 2020, 7, 243–250. [Google Scholar]
- Rinaldi, F.; Hanieh, P.N.; Chan, L.K.N.; Angeloni, L.; Passeri, D.; Rossi, M.; Wang, J.T.-W.; Imbriano, A.; Carafa, M.; Marianecci, C. Chitosan glutamate-coated niosomes: A proposal for nose-to-brain delivery. Pharmaceutics 2018, 10, 38. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Bao, G.; Liu, D.; Yang, Y.; Li, X.; Cai, G.; Liu, Y.; Wu, Y. The Association between folate and Alzheimer’s Disease: A Systematic review and meta-analysis. Front. Neurosci. 2021, 15, 385. [Google Scholar]
- Ravouru, N.; Kondreddy, P.; Korakanchi, D.; Haritha, M. Formulation and evaluation of niosomal nasal drug delivery system of folic acid for brain targeting. Curr. Drug Discov. Technol. 2013, 10, 270–282. [Google Scholar] [CrossRef]
- Estabragh, M.A.R.; Hamidifar, Z.; Pardakhty, A. Formulation of rivastigmine niosomes for alzheimer disease. Int. Pharm. Acta 2018, 1, 104. [Google Scholar]
- Nirale, P.; Paul, A.; Yadav, K.S. Nanoemulsions for targeting the neurodegenerative diseases: Alzheimer’s, Parkinson’s and Prion’s. Life Sci. 2020, 245, 117394. [Google Scholar] [CrossRef]
- McShane, R.; Westby, M.J.; Roberts, E.; Minakaran, N.; Schneider, L.; Farrimond, L.E.; Maayan, N.; Ware, J.; Debarros, J. Memantine for dementia. Cochrane Database Syst. Rev. 2019, 3, CD003154. [Google Scholar] [CrossRef] [PubMed]
- Md, S.; Gan, S.Y.; Haw, Y.H.; Ho, C.L.; Wong, S.; Choudhury, H. In vitro neuroprotective effects of naringenin nanoemulsion against β-amyloid toxicity through the regulation of amyloidogenesis and tau phosphorylation. Int. J. Biol. Macromol. 2018, 118, 1211–1219. [Google Scholar] [CrossRef]
- Kaur, A.; Nigam, K.; Srivastava, S.; Tyagi, A.; Dang, S. Memantine nanoemulsion: A new approach to treat Alzheimer’s disease. J. Microencapsul. 2020, 37, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Gul, R.; Jan, H.; Lalay, G.; Andleeb, A.; Usman, H.; Zainab, R.; Qamar, Z.; Hano, C.; Abbasi, B. Medicinal Plants and Biogenic Metal Oxide Nanoparticles: A Paradigm Shift to Treat Alzheimer’s Disease. Coatings 2021, 11, 717. [Google Scholar] [CrossRef]
- Ayaz, M.; Ovais, M.; Ahmad, I.; Sadiq, A.; Khalil, A.T.; Ullah, F. Biosynthesized metal nanoparticles as potential Alzheimer’s disease therapeutics. In Metal Nanoparticles for Drug Delivery and Diagnostic Applications; Elsevier: Amsterdam, The Netherlands, 2020; pp. 31–42. [Google Scholar] [CrossRef]
- Nazıroğlu, M.; Muhamad, S.; Pecze, L. Nanoparticles as potential clinical therapeutic agents in Alzheimer’s disease: Focus on selenium nanoparticles. Expert Rev. Clin. Pharmacol. 2017, 10, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Rajeshkumar, S.; Ganesh, L.; Santhoshkumar, J. Selenium Nanoparticles as Therapeutic Agents in Neurodegenerative Diseases. In Nanobiotechnology in Neurodegenerative Diseases; Springer: Berlin/Heidelberg, Germany, 2019; pp. 209–224. [Google Scholar] [CrossRef]
- Yin, T.; Yang, L.; Liu, Y.; Zhou, X.; Sun, J.; Liu, J. Sialic acid (SA)-modified selenium nanoparticles coated with a high blood–brain barrier permeability peptide-B6 peptide for potential use in Alzheimer’s disease. Acta Biomater. 2015, 25, 172–183. [Google Scholar] [CrossRef]
- Wahle, T.; Sofranko, A.; Dekkers, S.; Miller, M.R.; Heusinkveld, H.J.; Albrecht, C.; Cassee, F.R.; Schins, R.P. Evaluation of neurological effects of cerium dioxide nanoparticles doped with different amounts of zirconium following inhalation exposure in mouse models of Alzheimer’s and vascular disease. Neurochem. Int. 2020, 138, 104755. [Google Scholar] [CrossRef] [PubMed]
- Moskvin, M.; Marková, I.; Malínská, H.; Miklánková, D.; Hüttl, M.; Oliyarnyk, O.; Pop-Georgievski, O.; Zhigunov, A.; Petrovský, E.; Horák, D. Cerium oxide-decorated γ-Fe2O3 nanoparticles: Design, synthesis and in vivo effects on parameters of oxidative stress. Front. Chem. 2020, 8, 682. [Google Scholar] [CrossRef]
- Tian, E.-K.; Wang, Y.; Ren, R.; Zheng, W.; Liao, W. Gold nanoparticle: Recent progress on its antibacterial applications and mechanisms. J. Nanomater. 2021, 2021, 2501345. [Google Scholar] [CrossRef]
- Sanati, M.; Khodagholi, F.; Aminyavari, S.; Ghasemi, F.; Gholami, M.; Kebriaeezadeh, A.; Sabzevari, O.; Hajipour, M.J.; Imani, M.; Mahmoudi, M.; et al. Impact of gold nanoparticles on amyloid β-Induced Alzheimer’s disease in a rat animal model: Involvement of STIM proteins. ACS Chem. Neurosci. 2019, 10, 2299–2309. [Google Scholar] [CrossRef] [PubMed]
- Ali, T.; Kim, M.J.; Rehman, S.U.; Ahmad, A.; Kim, M.O. Anthocyanin-Loaded PEG-Gold nanoparticles enhanced the neuroprotection of anthocyanins in an Aβ1–42 mouse model of Alzheimer’s disease. Mol. Neurobiol. 2017, 54, 6490–6506. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, R.; Zhang, D.; Zhang, Z.; Zhu, J.; Xu, L.; Guo, Y. Neuroprotective effects of maize tetrapeptide-anchored gold nanoparticles in Alzheimer’s disease. Colloids Surf. B Biointerfaces 2021, 200, 111584. [Google Scholar] [CrossRef]
- Tramontin, N.D.S.; Da Silva, S.; Arruda, R.; Ugioni, K.S.; Canteiro, P.B.; Silveira, G.D.B.; Mendes, C.; Silveira, P.C.L.; Muller, A.P. Gold nanoparticles treatment reverses brain damage in Alzheimer’s disease model. Mol. Neurobiol. 2019, 57, 926–936. [Google Scholar] [CrossRef]
- Khan, N.H.; Mir, M.; Ngowi, E.E.; Zafar, U.; Khakwani, M.M.A.K.; Khattak, S.; Zhai, Y.-K.; Jiang, E.-S.; Zheng, M.; Duan, S.-F.; et al. Nanomedicine: A Promising way to manage Alzheimer’s disease. Front. Bioeng. Biotechnol. 2021, 9. [Google Scholar] [CrossRef]
- Lovell, M.; Robertson, J.; Teesdale, W.; Campbell, J.; Markesbery, W. Copper, iron and zinc in Alzheimer’s disease senile plaques. J. Neurol. Sci. 1998, 158, 47–52. [Google Scholar] [CrossRef]
- A Mandel, S.; Amit, T.; Machluf, M.; Youdim, M.B. Nanoparticles: A step forward for iron chelation in the brain. Futur. Neurol. 2007, 2, 265–269. [Google Scholar] [CrossRef]
- Farr, A.C.; Xiong, M.P. Challenges and opportunities of deferoxamine delivery for treatment of Alzheimer’s disease, Parkinson’s disease, and intracerebral hemorrhage. Mol. Pharm. 2021, 18, 593–609. [Google Scholar] [CrossRef] [PubMed]
- Mathys, Z.K.; White, A.R. Copper and Alzheimer’s disease. Neurotox. Met. 2017, 18, 199–216. [Google Scholar]
- Liu, G.; Men, P.; Perry, G.; Smith, M.A. Metal chelators coupled with nanoparticles as potential therapeutic agents for Alzheimer’s disease. J. Nanoneurosci. 2009, 1, 42–55. [Google Scholar] [CrossRef] [Green Version]
- Kreuter, J.; Shamenkov, D.; Petrov, V.; Ramge, P.; Cychutek, K.; Koch-Brandt, C.; Alyautdin, R. Apolipoprotein-mediated transport of nanoparticle-bound drugs across the blood-brain barrier. J. Drug Target. 2002, 10, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Kou, X.; Song, L.; Wang, Y.; Yu, Q.; Ju, H.; Yang, A.; Shen, R. Design, synthesis and anti-Alzheimer’s disease activity study of xanthone derivatives based on multi-target strategy. Bioorg. Med. Chem. Lett. 2020, 30, 126927. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, Y.; Hong, K.H.; Ning, Y.; Yu, P.; Ren, J.; Ji, M.; Cai, J. Design, synthesis and evaluation of a novel metal chelator as multifunctional agents for the treatment of Alzheimer’s disease. Bioorg. Chem. 2019, 87, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Choi, D.W.; Kim, H.N.; Park, C.G.; Lee, W.; Park, H.H. Protein-based nanoparticles as drug delivery systems. Pharmaceutics 2020, 12, 604. [Google Scholar] [CrossRef] [PubMed]
- Wong, H.L.; Wu, X.Y.; Bendayan, R. Nanotechnological advances for the delivery of CNS therapeutics. Adv. Drug Deliv. Rev. 2012, 64, 686–700. [Google Scholar] [CrossRef] [PubMed]
- Xuan, Z.; Gu, X.; Yan, S.; Xie, Y.; Zhou, Y.; Zhang, H.; Jin, H.; Hu, S.; Mak, M.S.H.; Zhou, D.; et al. Dimeric Tacrine(10)-hupyridone as a Multitarget-Directed ligand to treat Alzheimer’s disease. ACS Chem. Neurosci. 2021, 12, 2462–2477. [Google Scholar] [CrossRef] [PubMed]
- Luppi, B.; Bigucci, F.; Corace, G.; Delucca, A.; Cerchiara, T.; Sorrenti, M.; Catenacci, L.; Di Pietra, A.M.; Zecchi, V. Albumin nanoparticles carrying cyclodextrins for nasal delivery of the anti-Alzheimer drug tacrine. Eur. J. Pharm. Sci. 2011, 44, 559–565. [Google Scholar] [CrossRef]
- Zhao, Y.; Ran, W.; He, J.; Huang, Y.; Liu, Z.; Liu, W.; Tang, Y.; Zhang, L.; Gao, D.; Gao, F. High-Performance asymmetric supercapacitors based on multilayer MnO2/Graphene oxide nanoflakes and hierarchical porous carbon with enhanced cycling stability. Small 2015, 11, 1310–1319. [Google Scholar] [CrossRef]
- Gelinas, D.S.; DaSilva, K.; Fenili, D.; George-Hyslop, P.S.; McLaurin, J. Immunotherapy for Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 2004, 101, 14657–14662. [Google Scholar] [CrossRef] [Green Version]
- Moon, D.W.; Park, Y.H.; Lee, S.Y.; Lim, H.; Kwak, S.; Kim, M.S.; Kim, H.; Kim, E.; Jung, Y.; Hoe, H.-S.; et al. Multiplex protein imaging with secondary ion mass spectrometry using metal oxide nanoparticle-conjugated antibodies. ACS Appl. Mater. Interfaces 2020, 12, 18056–18064. [Google Scholar] [CrossRef]
- Liu, X.-G.; Zhang, L.; Lu, S.; Liu, D.-Q.; Zhang, L.-X.; Yu, X.-L.; Liu, R.-T. Multifunctional superparamagnetic iron oxide nanoparticles conjugated with Aβ Oligomer-Specific scFv antibody and class a scavenger receptor activator show early diagnostic potentials for Alzheimer’s disease. Int. J. Nanomed. 2020, 15, 4919–4932. [Google Scholar] [CrossRef]
- Kuo, Y.-C.; Tsai, H.-C. Rosmarinic acid- and curcumin-loaded polyacrylamide-cardiolipin-poly(lactide-co-glycolide) nanoparticles with conjugated 83-14 monoclonal antibody to protect β-amyloid-insulted neurons. Mater. Sci. Eng. C 2018, 91, 445–457. [Google Scholar] [CrossRef]
- Liu, H.; Jin, M.; Ji, M.; Zhang, W.; Liu, A.; Wang, T. Circ-Epc1 in Adipose-Derived Stem Cell Exosomes Can Improve Cognition by Shifting Microglial M1/M2 Polarization in Alzheimer’s Disease Mice Model. 2021. Available online: https://www.researchsquare.com/article/rs-445479/v1 (accessed on 5 May 2021).
- Chen, L.; Cao, H.; Huang, Q.; Xiao, J.; Teng, H. Absorption, metabolism and bioavailability of flavonoids: A review. Crit. Rev. Food Sci. Nutr. 2021, 1–13. [Google Scholar] [CrossRef]
- Abbas, M. Potential role of nanoparticles in treating the accumulation of amyloid-beta peptide in Alzheimer’s patients. Polymers 2021, 13, 1051. [Google Scholar] [CrossRef]
- Teleanu, D.M.; Chircov, C.; Grumezescu, A.M.; Volceanov, A.; Teleanu, R.I. Impact of nanoparticles on brain health: An up to date overview. J. Clin. Med. 2018, 7, 490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, B.; Zhang, Y.; Liu, J.; Feng, X.; Zhou, T.; Shao, L. Is neurotoxicity of metallic nanoparticles the cascades of oxidative stress? Nanoscale Res. Lett. 2016, 11, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Yusuf, M.; Khan, M.; Alrobaian, M.M.; Alghamdi, S.A.; Warsi, M.H.; Sultana, S.; Khan, R.A. Brain targeted Polysorbate-80 coated PLGA thymoquinone nanoparticles for the treatment of Alzheimer’s disease, with biomechanistic insights. J. Drug Deliv. Sci. Technol. 2021, 61, 102214. [Google Scholar] [CrossRef]
- Teleanu, D.M.; Chircov, C.; Grumezescu, A.M.; Teleanu, R.I. Neurotoxicity of Nanomaterials: An Up-to-Date Overview. Nanomaterials 2019, 9, 96. [Google Scholar] [CrossRef] [Green Version]
- Chang, X.; Li, J.; Niu, S.; Xue, Y.; Tang, M. Neurotoxicity of metal-containing nanoparticles and implications in glial cells. J. Appl. Toxicol. 2021, 41, 65–81. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Lai, S.K. Engineering Well-Characterized PEG-Coated Nanoparticles for Elucidating Biological Barriers to Drug Delivery. Methods Mol. Biol. 2017, 1530, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Farokhcheh, M.; Hejazian, L.; Akbarnejad, Z.; Pourabdolhossein, F.; Hosseini, S.M.; Mehraei, T.M.; Soltanpour, N. Geraniol improved memory impairment and neurotoxicity induced by zinc oxide nanoparticles in male wistar rats through its antioxidant effect. Life Sci. 2021, 282, 119823. [Google Scholar] [CrossRef] [PubMed]
| S. No | Drug | Class of Drug/Target | MOA | References |
|---|---|---|---|---|
| [1]. | Candesartan | TLR-2 inhibitor | Inhibit TL-2 and 4 expressions, inhibit microglial activation. | [54] |
| [2]. | Rifampicin | TLR-2 inhibitor TLR-2 inhibitor | Reduces oxidative stress, level of TNF-a, and inactivates microglia. | [55] |
| [3]. | TAK-242 | [56] | ||
| [4]. | β-caryophyllene | CB2 agonist | Inhibit microglial activation and exhibit an anti-inflammatory effect. | [57] |
| [5]. | JWH133 | Inhibit BBB damage, reduce iNOS expression, and level of pro-inflammatory cytokines. | [62] | |
| [6]. | α-asarone | Modulator of JAK/STAT or NF-kB pathways | Inhibit NF-kB activation, reduce the level of the pro-inflammatory cytokine, and improve behavioral function. | [59] |
| [7]. | Tanshinone I | NADPH oxidase modulator | Microglial inactivation reduces oxidative stress and neuroinflammation. | [63,64] |
| [8]. | Apocynin | |||
| [9]. | Resveratrol | |||
| [10]. | Diphenyleneiodonium | |||
| [11]. | Ghrelin | GHS-R1a ligand | Inhibit microglial activation, reduces the level of NO and ROS, and exhibit an anti-inflammatory effect. | [65] |
| [12]. | JNJ7777120 | Antagonist of H4R | Exhibit anti-inflammatory effect and improve behavioral dysfunction. | [66] |
| [13]. | MCC950 | NLRP3 inhibitor | Inhibit microglial activation, prevent AB deposition, tau phosphorylation and improve behavioral dysfunction. | [67] |
| S. No | Drug | Class of Drug/Target | MOA | References |
|---|---|---|---|---|
| [1]. | AAV2-hIL-10 | IL-10 agonist | Inhibit iNOS and NF-kB expression, and exhibit an anti-inflammatory effect. | [79] |
| [2]. | Rolipram | PDE-4 inhibitor | Increases the level of cyclic AMP, reduces oxidative stress, level of TNF-a, and improves the phagocytic activity of microglia. | [74] |
| [3]. | Sildenafil | [75] | ||
| [4]. | Yonkenafil | [76] | ||
| [5]. | Vitamin D | ERK inhibitor | Stimulate polarization state from M1 to M2. As a result, it inhibits M1 activation, reduces neuroinflammation, and improves cognitive dysfunctions. | [70] |
| [6]. | Pioglitazone | PPAR-g agonists | Shift M1 to M2 polarization inhibit NF-kB activation, reduces iNOS expression and inhibits TLR-4 activation. | [77] |
| [7]. | Rosiglitazone | [78] |
| S. No | Drug | Class/Source | MOA | References |
|---|---|---|---|---|
| [1]. | Magnolol | Lignan/Magnolia officialis | Stimulate microglial mediated phagocytosis of degradation of Aβ. | [87] |
| [2]. | Naringenin | Flavanone/Grapefruit | Stimulate the shift of M1 to M2. Inhibit M1 activation, Aβ aggregation, and improved cognitive dysfunction. | [88] |
| [3]. | Sarsasapogenin AA13 | Saponin Rhizoma Anemarrhenae | Mitigate the AD-induced neuroinflammation via promoting microglial phagocytosis and inhibit the Aβ aggregation. | [89] |
| [4]. | Eriodictyol | Flavanone/Yerba santa | Inhibit NF-kB/p38 MAPK and activate SIRT1 pathway. | [90] |
| [5]. | Apigenin | Flavone/Fruits | Reduced the level of PGE2 and NO. Inhibit p38 MAPK/JNK/ERK1/2 pathways. | [91] |
| [6]. | Dihydromyricetin | flavonoids/Hovenia dulcis | Inhibit the microglial mediated neuroinflammation via inhibition of JAK/STAT/NLRP3 pathways. | [92] |
| [7]. | Icariside II | Flavonoid/Epimedium brevicornum | Inhibit the microglial mediated neuroinflammation via inhibition of TLR4/MyD88/NF-κB pathways. | [93] |
| [8]. | Hesperidin | Bioflavonoid/citrus fruits | Inhibit the microglial mediated neuroinflammation via inhibition of NLRP3 and stimulate Akt/Nrf2 pathway. | [94] |
| [9]. | Silibinin | Flavonolignan/Silybum marianum | Inhibit the neuroinflammatory activities of JNK/p38 MAPK/NF-kB pathway. | [95] |
| [10]. | Safflower Yellow extract | Carthamus tinctorius | Reduce the expression of iNOS and increase the expression of arginase-1. Stimulate polarization state from M1 to M2. Inhibit M1 activation. | [96] |
| [11]. | Curcumin | Polyphenol/Curcuma longa | Inhibit the microglia-mediated NF-kB pathway. | [97] |
| [12]. | Ferulic acid | Grains, fruits, and vegetables | Inhibit the microglial-induced neuroinflammation via modulation of NLRP3/NFkB/MAPk/TLR4 pathways. | [98] |
| [13]. | 6-Shogaol | Zingiber officinale | Inhibit the neuroinflammation via modulation of NGF level. | [99] |
| [14]. | Epigallocatechin-3gallate | Catechin/Camellia sinensis | Reduce the level of TNF-α, IL-6, IL-1β, stimulate Nrf2/HO pathways, and inhibit microglial activation. | [100] |
| [15]. | Andrographolide | Diterpenoid/Andrographis paniculata | Inhibitor of MAPK pathway and restrict the nuclear translocation of NF-kB. | [101] |
| [16]. | Andalucin | Sesquiterpene lactone/Artemisia lannta | Reduce the level of TNF-α, IL-6, IL-1β, stimulate Nrf2/HO pathways and inhibit microglial activation. | [102] |
| [17]. | Oleanolic acid | Pentacyclic triterpenoid | Reduce the level of TNF-α, IL-6, IL-1β and exhibit the antioxidant effect. Inhibit microglial activation. | [103] |
| [18]. | Piperlongumine | Alkaloid/Piper longum | Inhibitor of NF-kB pathway, restrict the activities of β and γ secretases and inhibit the aggregation of Aβ. | [104] |
| [19]. | Geniposidic acid | Iridoid glucoside/Eucommia ulmoides | Inhibitor of NF-kB pathway reduces the expression of iNOS, reduces the aggregation of Aβ, and inhibits microglial activation. | [105] |
| [20]. | Aromatic turmerone | Essential oil/Curcuma longa | Inhibitor of NF-kB, TLR-4, and stimulator of Nrf2 pathways. | [106] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Md, S.; Alhakamy, N.A.; Alfaleh, M.A.; Afzal, O.; Altamimi, A.S.A.; Iqubal, A.; Shaik, R.A. Mechanisms Involved in Microglial-Interceded Alzheimer’s Disease and Nanocarrier-Based Treatment Approaches. J. Pers. Med. 2021, 11, 1116. https://doi.org/10.3390/jpm11111116
Md S, Alhakamy NA, Alfaleh MA, Afzal O, Altamimi ASA, Iqubal A, Shaik RA. Mechanisms Involved in Microglial-Interceded Alzheimer’s Disease and Nanocarrier-Based Treatment Approaches. Journal of Personalized Medicine. 2021; 11(11):1116. https://doi.org/10.3390/jpm11111116
Chicago/Turabian StyleMd, Shadab, Nabil A. Alhakamy, Mohamed A. Alfaleh, Obaid Afzal, Abdulmalik S. A. Altamimi, Ashif Iqubal, and Rasheed A. Shaik. 2021. "Mechanisms Involved in Microglial-Interceded Alzheimer’s Disease and Nanocarrier-Based Treatment Approaches" Journal of Personalized Medicine 11, no. 11: 1116. https://doi.org/10.3390/jpm11111116
APA StyleMd, S., Alhakamy, N. A., Alfaleh, M. A., Afzal, O., Altamimi, A. S. A., Iqubal, A., & Shaik, R. A. (2021). Mechanisms Involved in Microglial-Interceded Alzheimer’s Disease and Nanocarrier-Based Treatment Approaches. Journal of Personalized Medicine, 11(11), 1116. https://doi.org/10.3390/jpm11111116








