miR-210 and miR-152 as Biomarkers by Liquid Biopsy in Invasive Ductal Carcinoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Groups
- Group Breast Cancer (BC): women with breast cancer (plasma/fragment: n = 30);
- Group Benign Breast Conditions (BBC): women with benign breast conditions (plasma/fragment: n = 5);
- Control: women with no atypical breast, and no history of BC in the family (plasma: n = 15; fragment: n = 5).
2.2. Collections
2.3. Determination of Diagnosis and Pathological Prognosis Staging
2.4. Analysis of miRNA Expression by RT-qPCR
2.5. Analysis of Target Protein Expression by Immunohistochemistry
2.6. Statistical Analysis
3. Results
3.1. Characterization of Study Population
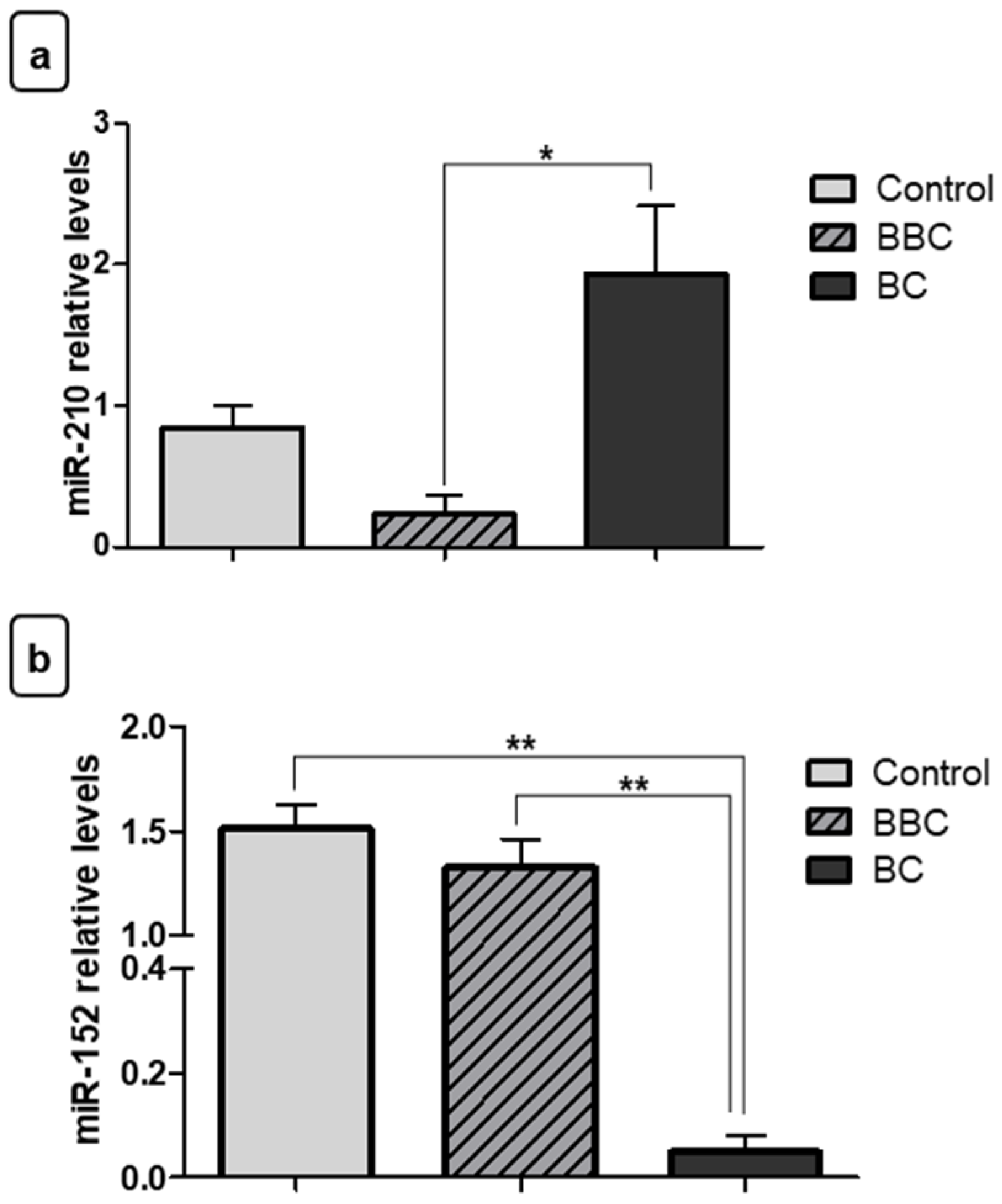
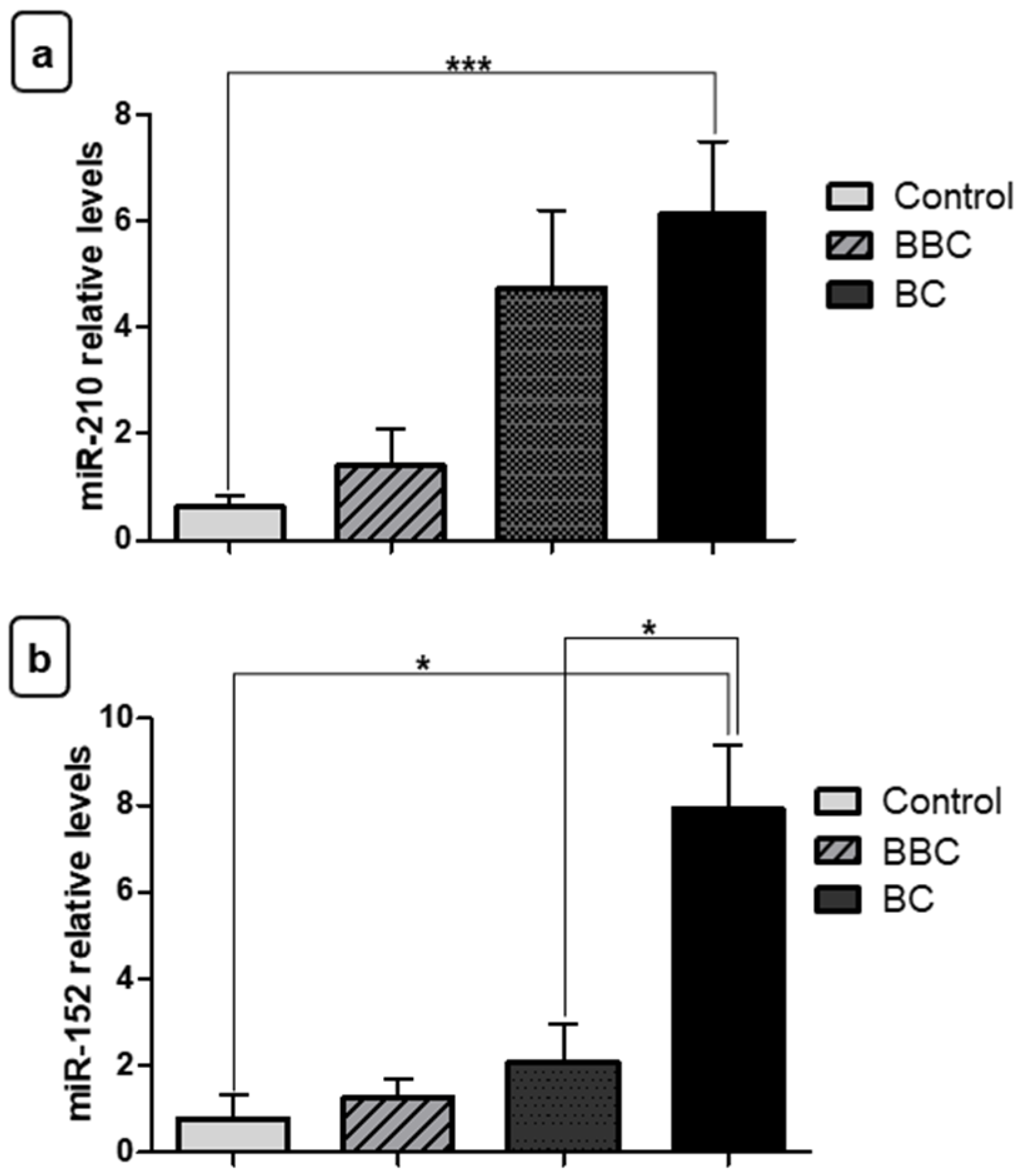
3.2. Expression Levels of miRNAs in Breast Cancer
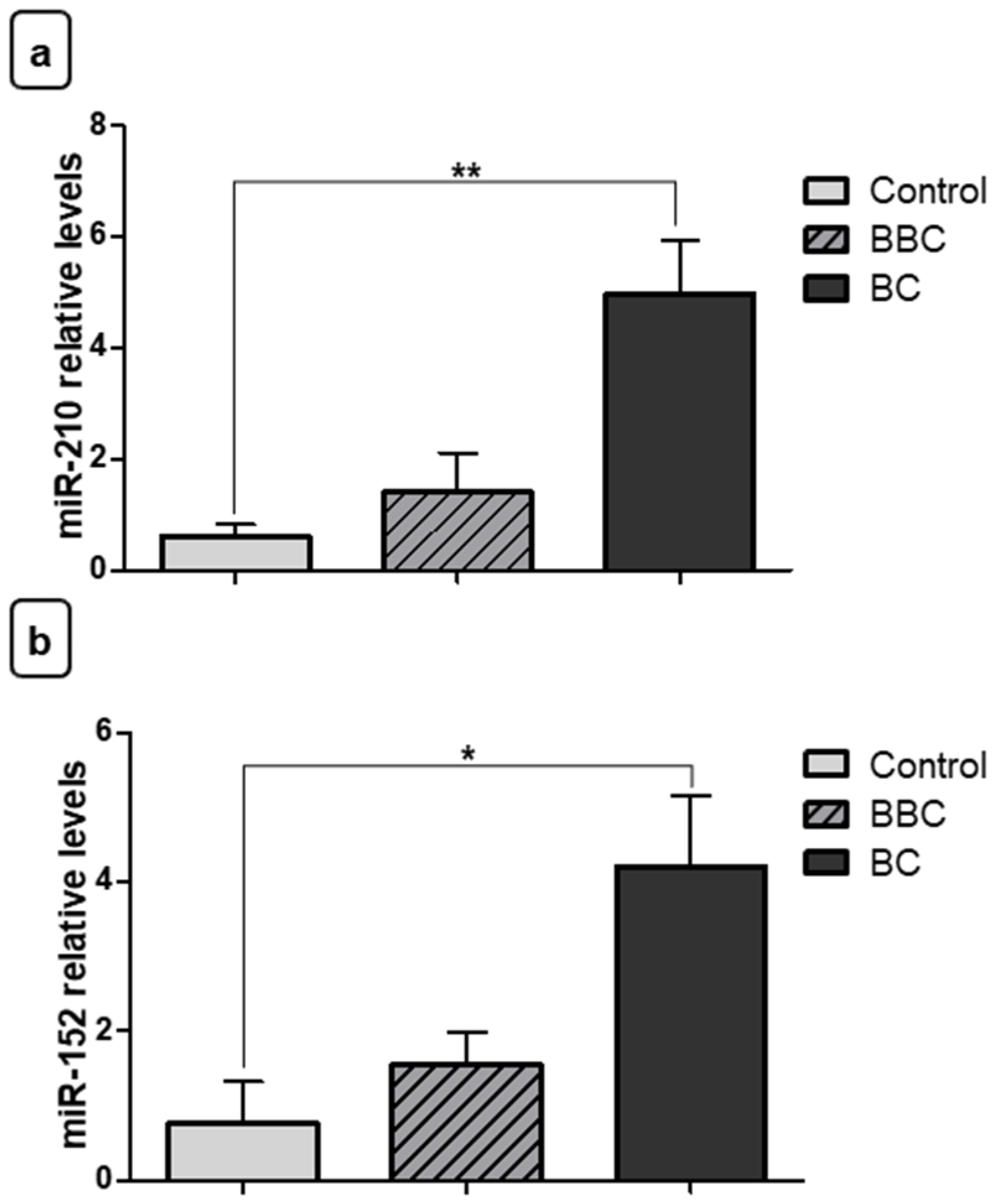
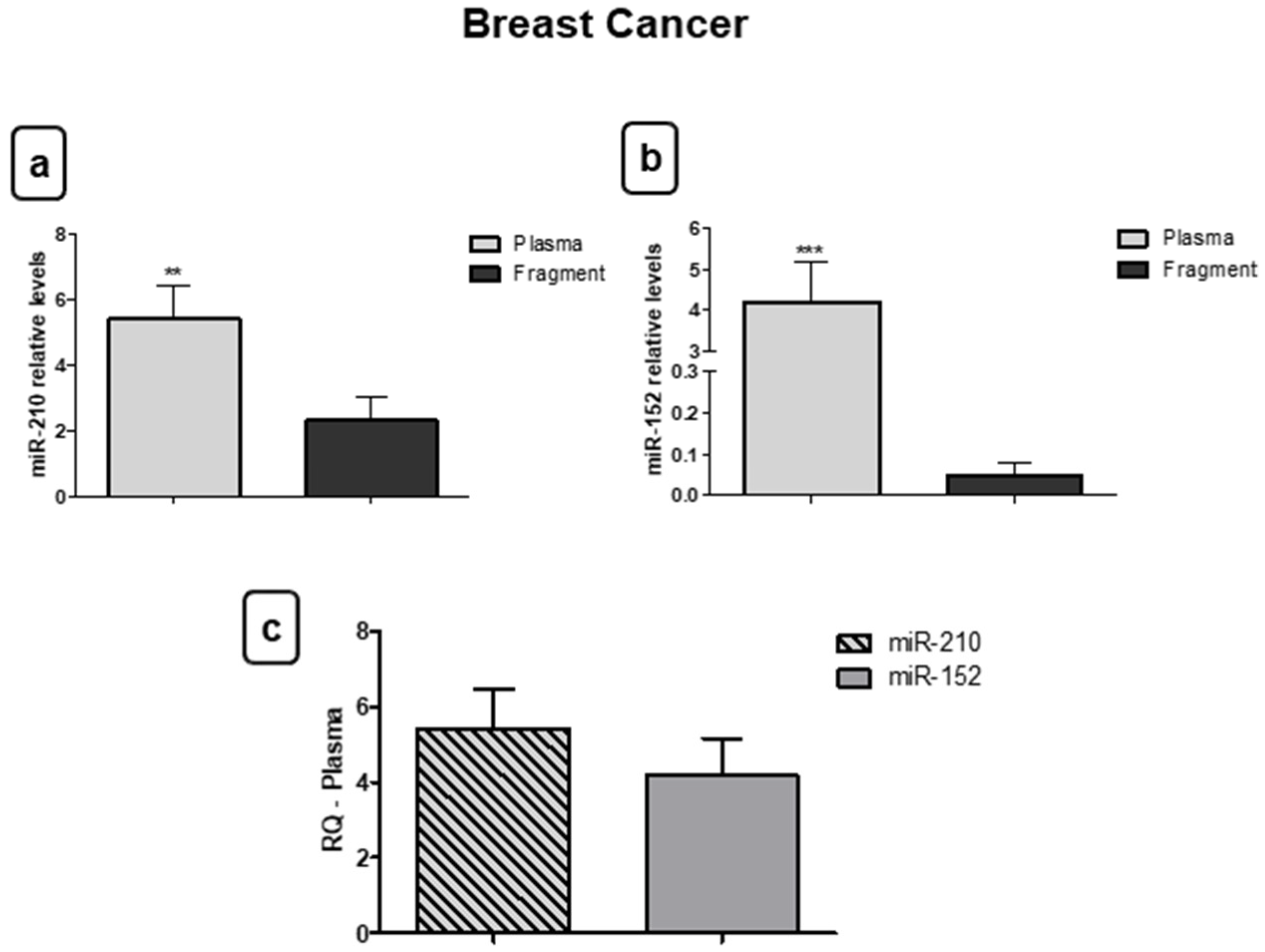
3.3. miR-210 and miR-152 Are Stable in Bloodstream
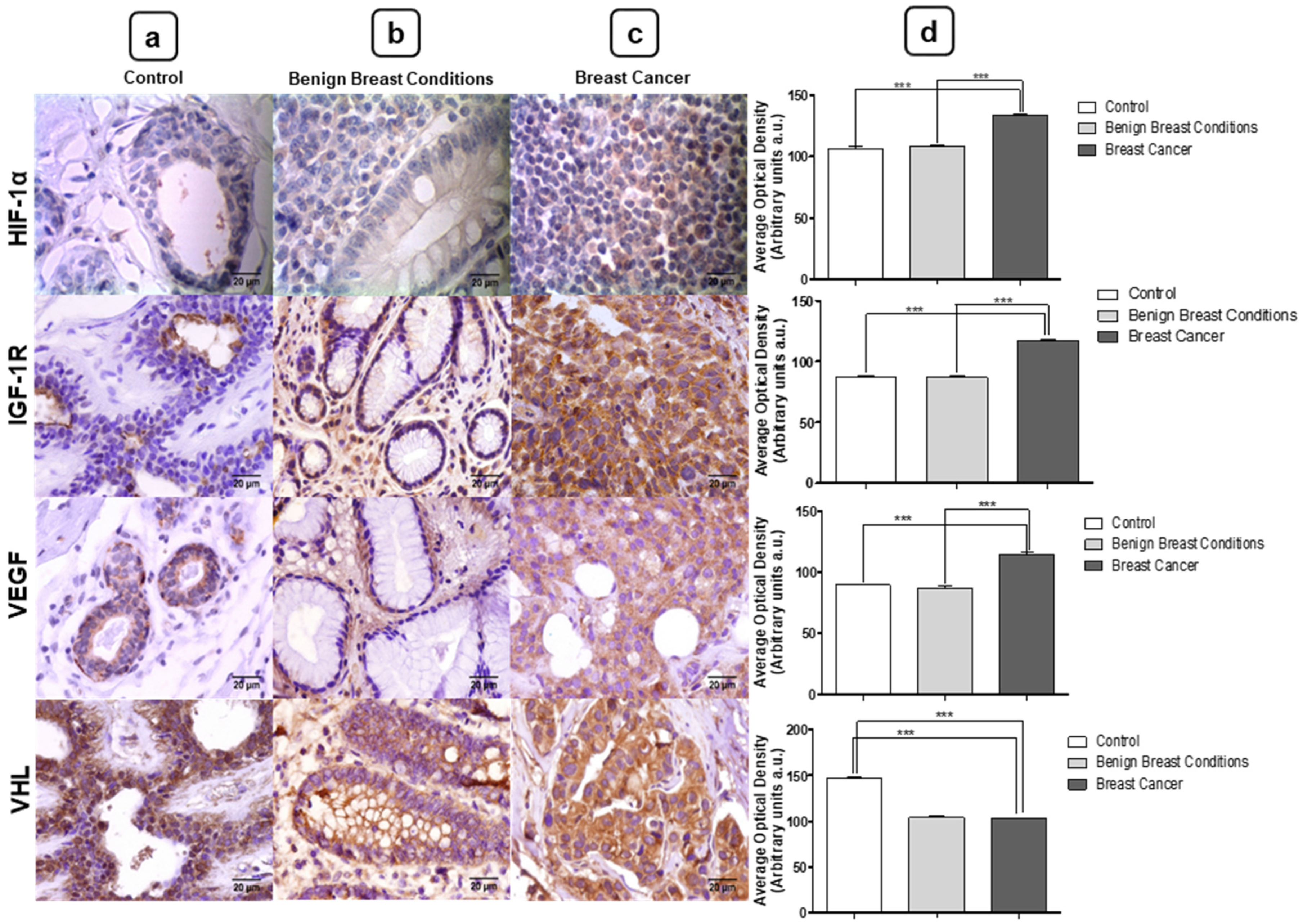
3.4. Expression of miRNA Target Proteins by Immunohistochemistry
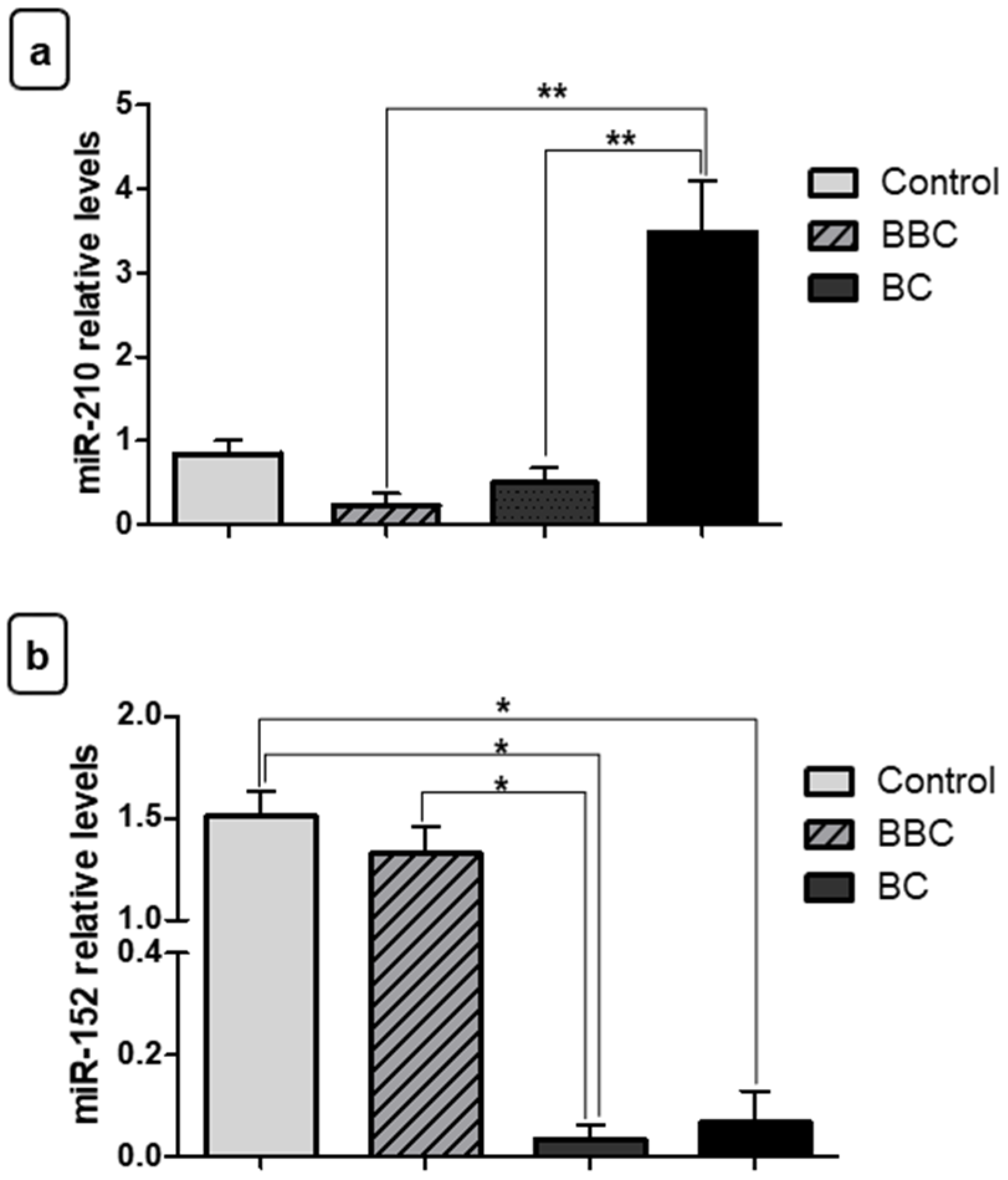
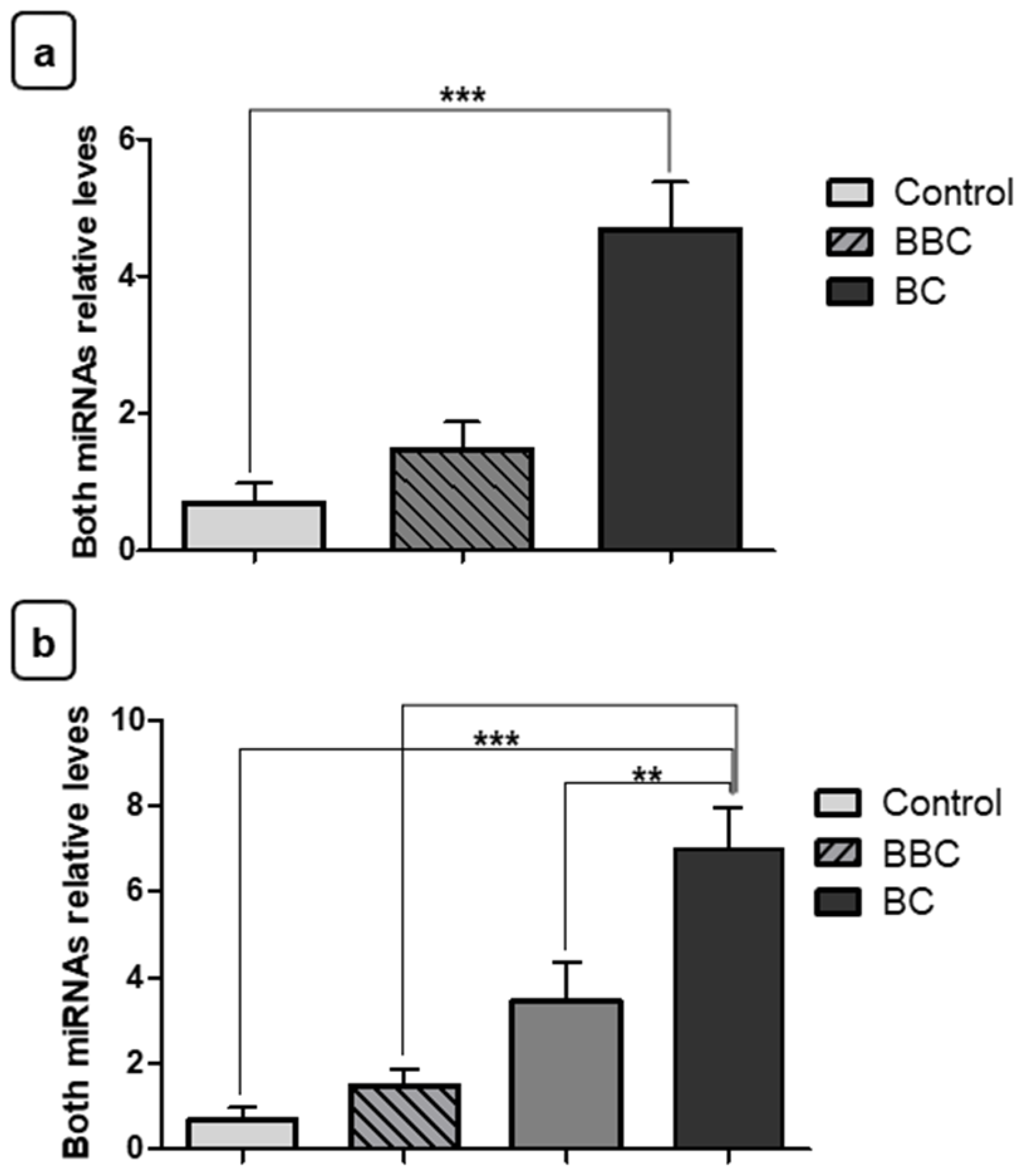
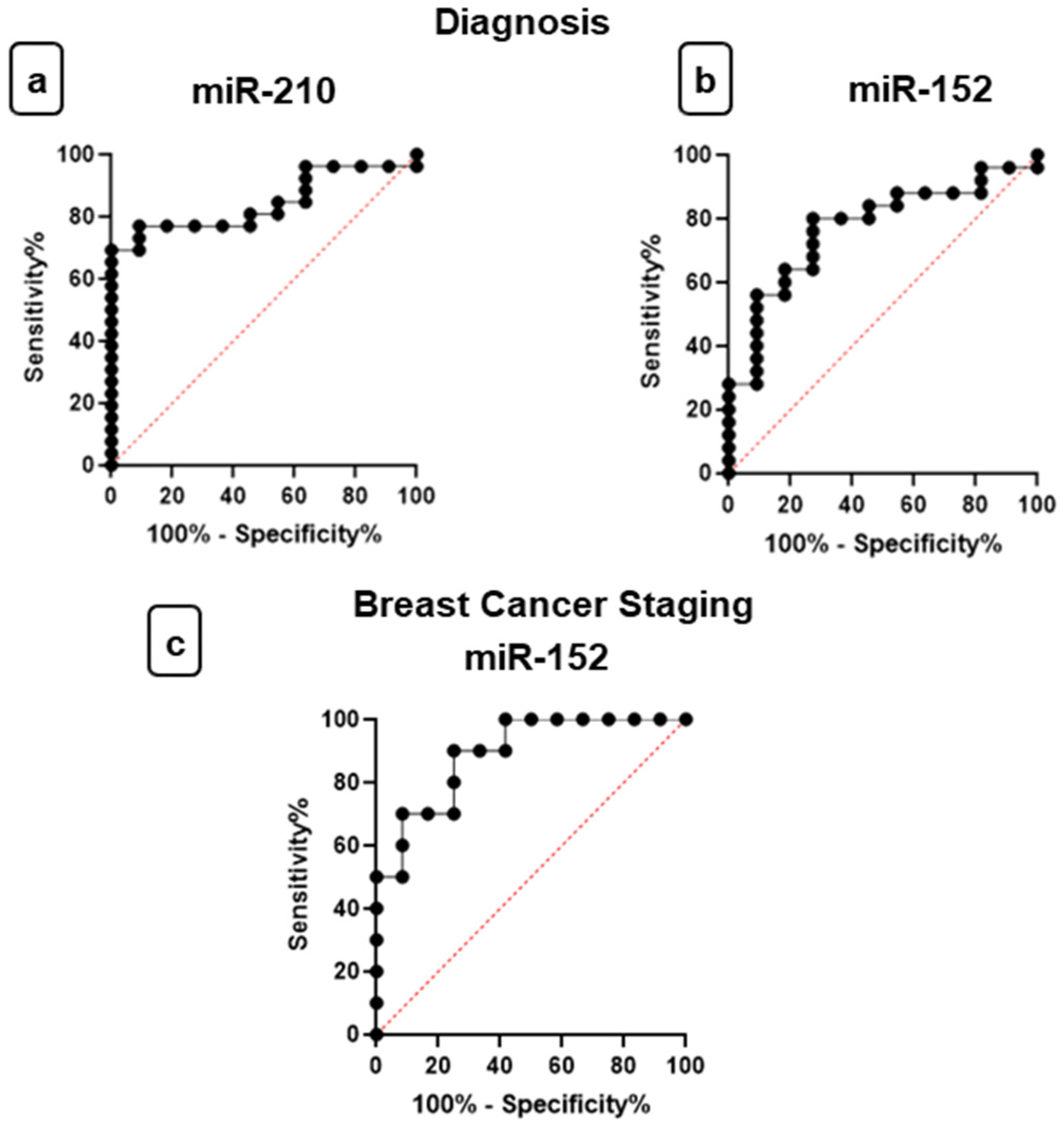
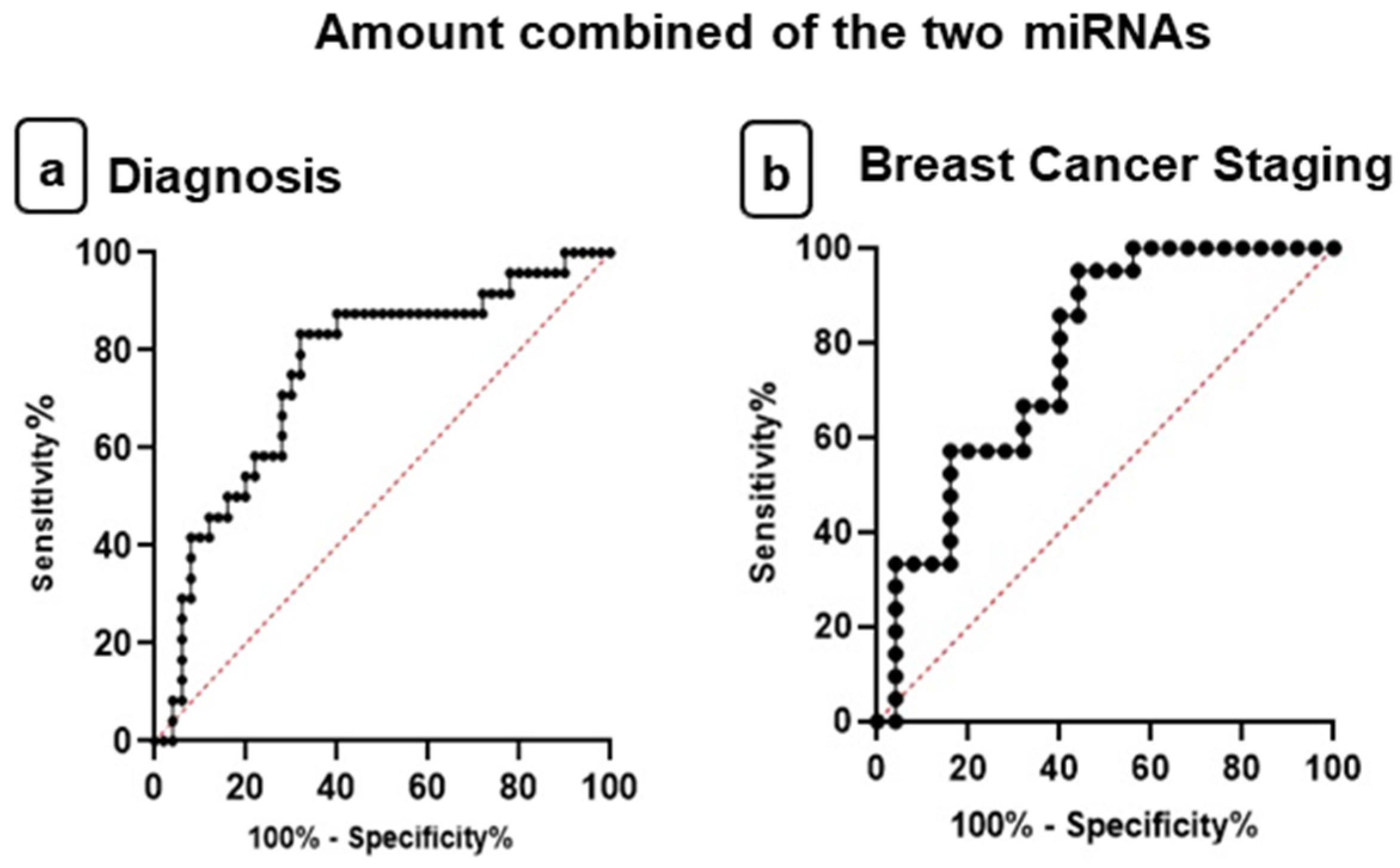
3.5. Evaluation of miR-210 and miR-152 as Potential Diagnostic and Staging Biomarkers
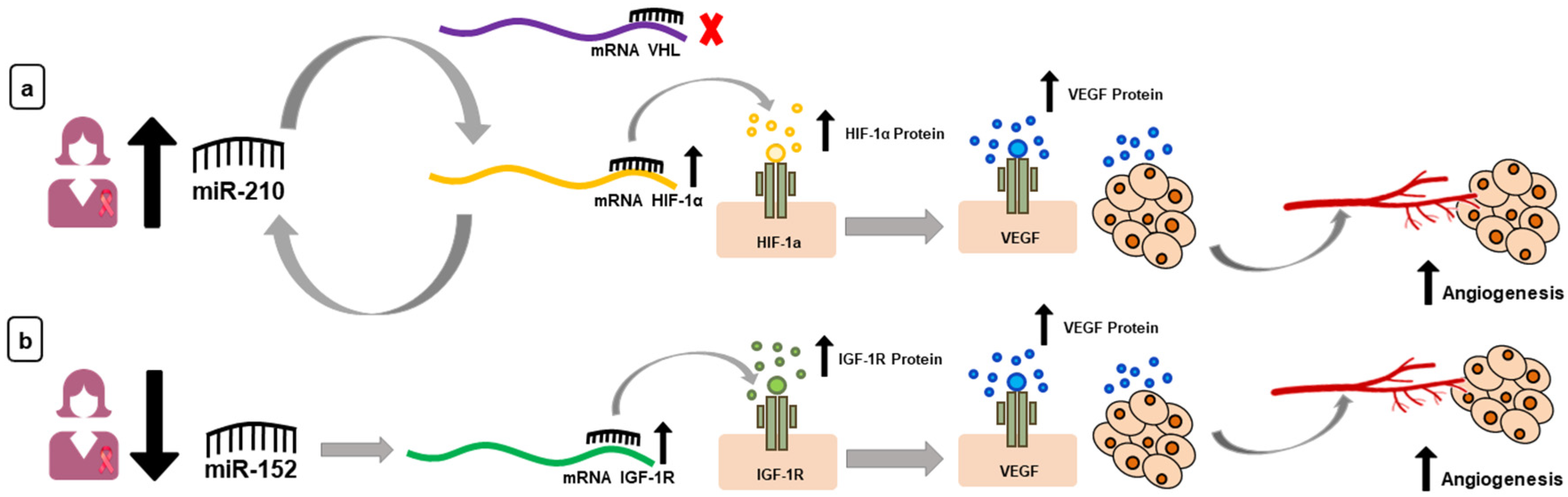
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crawford, Y.; Ferrara, N. VEGF inhibition: Insights from preclinical and clinical studies. Cell Tissue Res. 2009, 335, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Liu, K.; Li, T.; Fang, J.; Ding, Y.; Sun, L.; Tu, T.; Jiang, X.; Du, S.; Hu, J.; et al. miR-21: A gene of dual regulation in breast cancer. Int. J. Oncol. 2016, 48, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Boudreau, N.; Myers, C. Breast cancer-induced angiogenesis: Multiple mechanisms and the role of the microenvironment. Breast Cancer Res. 2003, 5, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.J.; Conley, Y.P.; Gorin, M.B.; Gensler, G.; Lai, C.Q.; Shang, F.; Taylor, A. Associations between Genetic Polymorphisms of Insulin-like Growth Factor Axis Genes and Risk for Age-related Macular Degeneration. Investig. Ophthalmol. Vis. Sci. 2011, 52, 9099–9107. [Google Scholar] [CrossRef]
- Rahimi, N. The Ubiquitin-Proteasome System Meets Angiogenesis. Mol. Cancer Ther. 2012, 11, 538–548. [Google Scholar] [CrossRef]
- Wu, F.T.; Lu, L.; Xu, W.; Li, J.Y. Circulating tumor DNA: Clinical roles in diffuse large B cell lymphoma. Ann. Hematol. 2018, 98, 255–269. [Google Scholar] [CrossRef]
- Qi, Z.H.; Xu, H.X.; Zhang, S.R.; Xu, J.Z.; Li, S.; Gao, H.L.; Jin, W.; Wang, W.Q.; Wu, C.T.; Ni, Q.X.; et al. The Significance of Liquid Biopsy in Pancreatic Cance. J. Cancer 2018, 9, 3417–3426. [Google Scholar] [CrossRef]
- Esquela-Kerscher, A.; Slack, F.J. Oncomirs-microRNAs with a role in cancer. Nat. Rev. Cancer 2006, 6, 259–269. [Google Scholar] [CrossRef]
- Wei, J.; Gao, W.; Zhu, C.J.; Liu, Y.K.; Mei, Z.; Cheng, T.; Shu, Y.Q. Identification of plasma microRNA-21 as biomarker for early detection and chemosensity of non-small cell lung cancer. Chin. J. Cancer 2011, 30, 407–414. [Google Scholar] [CrossRef]
- Bertoli, G.; Cava, C.; Castiglioni, I. MicroRNAs: New biomarkers for diagnosis, prognosis, therapy prediction and therapeutic tools for breast cancer. Theranostics 2015, 5, 1122–1143. [Google Scholar] [CrossRef]
- Karakashev, S.V.; Reginato, M.J. Progress toward overcoming hypoxia-induced resistance to solid tumor therapy. Cancer Manag. Res. 2015, 7, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, R.; Pandey, P.; Jha, P.; Dwivedi, V.; Sarkar, C.; Kulshreshtha, R. Hypoxic signature of microRNAs in glioblastoma: Insights from small RNA deep sequencing. BMC Genom. 2014, 17, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.J.; Santarpia, L.; Kim, J.; Esteva, F.J.; Moretti, E.; Buzdar, A.U.; Di Leo, A.; Le, X.F.; Bast-Jr, R.C.; Park, S.T.; et al. Plasma microRNA 210 levels correlate with sensitivity to trastuzumab and tumor presence in breast cancer patients. Cancer 2012, 118, 2603–2614. [Google Scholar] [CrossRef] [PubMed]
- Dang, K.; Myers, K.A. The role of Hypoxia-Induced miR-210 in Cancer Progression. Int. J. Mol. Sci. 2015, 16, 6353–6372. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Xia, H.; Wang, F.; Chen, C.; Long, J. MicroRNA-210 interacts with FBXO31 to regulate cancer proliferation cell cycle and migration in human breast cancer. Onco Targets Ther. 2016, 9, 5245–5255. [Google Scholar] [CrossRef]
- Xu, Q.; Jiang, Y.; Yin, Y.; Li, Q.; He, J.; Jing, Y.; Qi, Y.-T.; Xu, Q.; Li, W.; Lu, B.; et al. A regulatory circuit of miR-148a/152 and DNMT1 in modulating cell transformation and tumor angiogenesis through IGF-IR and IRS1. J. Mol. Cell Biol. 2013, 5, 3–13. [Google Scholar] [CrossRef]
- Sengupta, D.; Deb, M.; Rath, S.K.; Kar, S.; Parbin, S.; Pradhan, N.; Patra, S.K. DNA methylation and not H3K4 trimethylation dictates the expression status of miR-152 gene which inhibits migration of breast cancer cells via DNMT1/CDH1 loop. Exp. Cell Res. 2016, 15, 176–187. [Google Scholar] [CrossRef]
- Liu, X.; Li, J.; Qin, F.; Dai, S. miR-152 as a tumor suppressor microRNA: Target recognition and regulation in cancer. Oncol. Lett. 2016, 11, 3911–3916. [Google Scholar] [CrossRef]
- Ge, S.; Wang, D.; Kong, Q.; Gao, W.; Sun, J. Function of miR-152 as a Tumor Suppressor in Human Breast Cancer by Targeting PIK3CA. Oncol. Res. 2017, 25, 1363–1371. [Google Scholar] [CrossRef]
- Roth, C.; Rack, B.; Müller, V.; Janni, W.; Pantel, K.; Schwarzenbach, H. Circulating microRNAs as blood-based markers for patients with primary and metastatic breast cancer. Breast Cancer Res. 2010, 12, R90. [Google Scholar] [CrossRef]
- Duffy, M.J.; Harbeck, N.; Nap, M.; Molina, R.; Nicolini, A.; Senkus, E.; Cardoso, F. Clinical use of biomarkers in breast cancer: Updated guidelines from the European Group on Tumor Markers (EGTM). Eur. J. Cancer 2017, 75, 284–298. [Google Scholar] [CrossRef] [PubMed]
- Qin, Q.; Furong, W.; Baosheng, L. Multiple functions of hypoxia-regulated miR-210 in cancer. J. Exp. Clin. Cancer Res. 2014, 33, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Thakur, S.; Grover, R.K.; Gupta, S.; Yadav, A.K.; Das, B.C. Identification of Specific miRNA Signature in Paired Sera and Tissue Samples of Indian Women with Triple Negative Breast Cancer. PLoS ONE 2016, 11, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Ognibene, M.; Cangelosi, D.; Morini, M.; Segalerba, D.; Bosco, M.C.; Sementa, A.R.; Eva, A.; Varesio, L. Immunohistochemical analysis of PDK1, PHD3 and HIF-1α expression defines the hypoxic status of neuroblastoma tumors. PLoS ONE 2017, 12, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Hogner, A.; Krause, H.; Jandrig, B.; Kasim, M.; Fuller, T.F.; Schostak, M.; Erbersdobler, A.; Patzak, A.; Kilic, E. PBRM1 and VHL expression correlate in human clear cell renal cell carcinoma with differential association with patient’s overall survival. Urol. Oncol. Semin. Orig. Investig. 2018, 36, 94.e1–94.e14. [Google Scholar] [CrossRef]
- Zhang, H.; Lu, Y.; Wang, S.; Sheng, X.; Zhang, S. MicroRNA-152 Acts as a Tumor Suppressor MicroRNA by Inhibiting Kruppel-Like Factor 5 in Human Cervical Cancer. Oncol Res. Featur. Preclin. Clin. Cancer Ther. 2019, 27, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.Y.; Liu, W.T.; Sun, H.R.; Ge, X.; Shi, Z.M.; Wang, M.; Ling-Zhi, L.; Zhang, J.Y.; Liu, L.Z.; Jiang, B.H. IGF-1-mediated PKM2/beta-catenin/miR-152 regulatory circuit in breast cancer. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Sun, W.Y.; Yun, H.Y.; Song, Y.J.; Kim, H.; Lee, O.J.; Nam, S.J.; Koo, J.S. Insulin-like growth factor 1 receptor expression in breast cancer tissue and mammographic density. Mol. Clin. Oncol. 2015, 3, 572–580. [Google Scholar] [CrossRef]
- Cebe-Suarez, S.; Zehnder-Fjallman, A.; Ballmer-Hofer, K. The role of VEGF receptors in angiogenesis; complex partnerships. Cell Mol. Life Sci. 2006, 63, 601–615. [Google Scholar] [CrossRef]
- Maae, E.; Nielsen, M.; Steffensen, K.D.; Jakobsen, E.H.; Jakobsen, A.; Sorensen, F.B. Estimation of immunohistochemical expression of VEGF in ductal carcinomas of the breast. J. Histochem. Cytochem. 2011, 59, 750–760. [Google Scholar] [CrossRef]
- Wen, S.; Shao, G.; Zheng, J.; Zeng, H.; Luo, J.; Gu, D. Apatinib regulates the cell proliferation and apoptosis of liver cancer by regulation of VEGFR2/STAT3 signaling. Pathol. Res. Pract. 2019, 215, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liang, H.; Zhang, J.; Zen, K.; Zhang, C.Y. Secreted microRNAs: A new form of intercellular communication. Trends Cell Biolog. 2012, 22, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Madhavan, D.; Peng, C.; Wallwiener, M.; Zucknick, M.; Nees, J.; Schott, S. Circulating miRNAs with prognostic value in metastatic breast cancer and for early detection of metastasis. Carcinogenesis 2016, 37, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Markou, A.; Zavridou, M.; Sourvinou, I.; Yousef, G.; Kounelis, S.; Malamos, N.; Georgoulias, V.; Lianidou, E. Direct Comparison of Metastasis-Related miRNAs Expression Levels in Circulating Tumor Cells, Corresponding Plasma, and Primary Tumors of Breast Cancer Patients. Clin. Chem. 2016, 62, 1002–1011. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Qattan, A.M.; Intabli, H.; Alkhayal, W.; Eltabache, C.; Tweigieri, T.; Amer, S.B. Robust expression of tumor suppressor miRNA’s let-7 and miR-195 detected in plasma of Saudi female breast cancer patients. BMC Cancer 2017, 17, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yoshioka, Y.; Katsuda, T.; Ochiya, T. Extracellular vesicles and encapusulated miRNAs as emerging cancer biomarkers for novel liquid biopsy. Jpn. J. Clin. Oncol. 2018, 48, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Adouh, M.; Hamam, D.; Gao, Z.H.; Arena, V.; Arena, M.; Arena, G.O. Exosomes isolated from cancer patients sera transfer malignant traits and confer the same phenotype of primary tumors to oncosuppressor-mutated cells. J. Exp. Clin. Cancer Res. 2017, 36, 1–15. [Google Scholar] [CrossRef]
- Dilsiz, N. Role of exosomes and exosomal microRNAs in cancer. Future Sci. 2020, 6, FSO465. [Google Scholar] [CrossRef]
- Chen, H.; Liu, H.; Zou, H.; Chen, R.; Dou, Y.; Sheng, S.; Dai, S.; Ai, J.; Melson, J.; Kittles, R.A.; et al. Evaluation of Plasma miR-21 and miR-152 as Diagnostic Biomarkers for Common Types of Human Cancers. J. Cancer 2016, 7, 490–499. [Google Scholar] [CrossRef]
- Jiang, X.; Du, L.; Wang, L.; Li, J.; Liu, Y.; Zheng, G.; Qu, A.; Zhang, X.; Pan, H.; Yang, Y.; et al. Serum microRNA expression signatures identified from genome-wide microRNA profiling serve as novel noninvasive biomarkers for diagnosis and recurrence of bladder cancer. Int. J. Cancer 2015, 136, 854–862. [Google Scholar] [CrossRef]
- Lin, Y.; Lin, Z.; Fang, Z.; Li, H.; Zhi, X.; Zhang, Z. Plasma MicroRNA-34a as a Potential Biomarker for Early Diagnosis of Esophageal Cancer. Clin. Lab. 2019, 65. [Google Scholar] [CrossRef]
- Kosaka, N.; Iguchi, H.; Yoshioka, Y.; Takeshita, F.; Matsuki, Y.; Ochiya, T. Secretory Mechanisms and Intercellular Transfer of MicroRNAs in Living Cells. J. Biol. Chem. 2010, 285, 17442–17452. [Google Scholar] [CrossRef]
- Ohshima, K.; Inoue, K.; Fujiwara, A.; Hatakeyama, K.; Kanto, K.; Watanabe, Y.; Muramatsu, K.; Fukuda, Y.; Ogura, S.-I.; Yamaguchi, K.; et al. Let-7 microRNA family is selectively secreted into the extracellular environment via exosomes in a metastatic gastric cancer cell line. PLoS ONE 2010, 8, e13247. [Google Scholar] [CrossRef] [PubMed]
- Ostenfeld, M.S.; Jeppesen, D.K.; Laurberg, J.R.; Boysen, A.T.; Bramsen, J.B.; Primdal-Bengtson, B.; Hendrix, A.; Lamy, P.; Dagnaes-Hansen, F.; Rasmussen, M.H.; et al. Cellular Disposal of miR23b by RAB27-Dependent Exosome Release Is Linked to Acquisition of Metastatic Properties. Cancer Res. 2014, 74, 5758–5771. [Google Scholar] [CrossRef]
- Falcone, G.; Felsani, A.; D’Agnano, I. Signaling by exosomal microRNAs in cancer. J. Exp. Clin. Cancer Res. 2015, 34, 1–10. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, S.; Weber, J.; Baxter, D.; Galas, D.J. Export of microRNAs and microRNA-protective protein by mammalian cells. Nucleic Acids Res. 2010, 38, 7248–7259. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.S.; Parkin, R.K.; Kroh, E.M.; Fritz, B.R.; Wyman, S.K.; Pogosova-Agadjanyan, E.L.; Peterson, A.; Noteboom, J.; O’Briant, K.C.; Allen, A.; et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc. Natl. Acad. Sci. USA 2008, 105, 10513–10518. [Google Scholar] [CrossRef]
- Hosmer, D.W., Jr.; Lemeshow, S.; Sturdivant, R.X. Applied Logistic Regression, 3rd ed.; Wiley: Hoboken, NJ, USA, 2013; p. 177. [Google Scholar]
- Dasari, S.; Wudayagiri, R.; Valluru, L. Cervical Cancer: Biomarkers for Diagnosis and Treatment. Clin. Chim. Acta 2015, 445, 7–11. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopes, B.C.; Braga, C.Z.; Ventura, F.V.; de Oliveira, J.G.; Kato-Junior, E.M.; Bordin-Junior, N.A.; Zuccari, D.A.P.C. miR-210 and miR-152 as Biomarkers by Liquid Biopsy in Invasive Ductal Carcinoma. J. Pers. Med. 2021, 11, 31. https://doi.org/10.3390/jpm11010031
Lopes BC, Braga CZ, Ventura FV, de Oliveira JG, Kato-Junior EM, Bordin-Junior NA, Zuccari DAPC. miR-210 and miR-152 as Biomarkers by Liquid Biopsy in Invasive Ductal Carcinoma. Journal of Personalized Medicine. 2021; 11(1):31. https://doi.org/10.3390/jpm11010031
Chicago/Turabian StyleLopes, Beatriz C., Cristine Z. Braga, Fabrício V. Ventura, Jéssica G. de Oliveira, Edson M. Kato-Junior, Newton A. Bordin-Junior, and Debora A. P. C. Zuccari. 2021. "miR-210 and miR-152 as Biomarkers by Liquid Biopsy in Invasive Ductal Carcinoma" Journal of Personalized Medicine 11, no. 1: 31. https://doi.org/10.3390/jpm11010031
APA StyleLopes, B. C., Braga, C. Z., Ventura, F. V., de Oliveira, J. G., Kato-Junior, E. M., Bordin-Junior, N. A., & Zuccari, D. A. P. C. (2021). miR-210 and miR-152 as Biomarkers by Liquid Biopsy in Invasive Ductal Carcinoma. Journal of Personalized Medicine, 11(1), 31. https://doi.org/10.3390/jpm11010031





