Establishment of a Molecular Tumor Board (MTB) and Uptake of Recommendations in a Community Setting
Abstract
1. Introduction
2. Methods
2.1. Patients and Setting
2.2. CGP
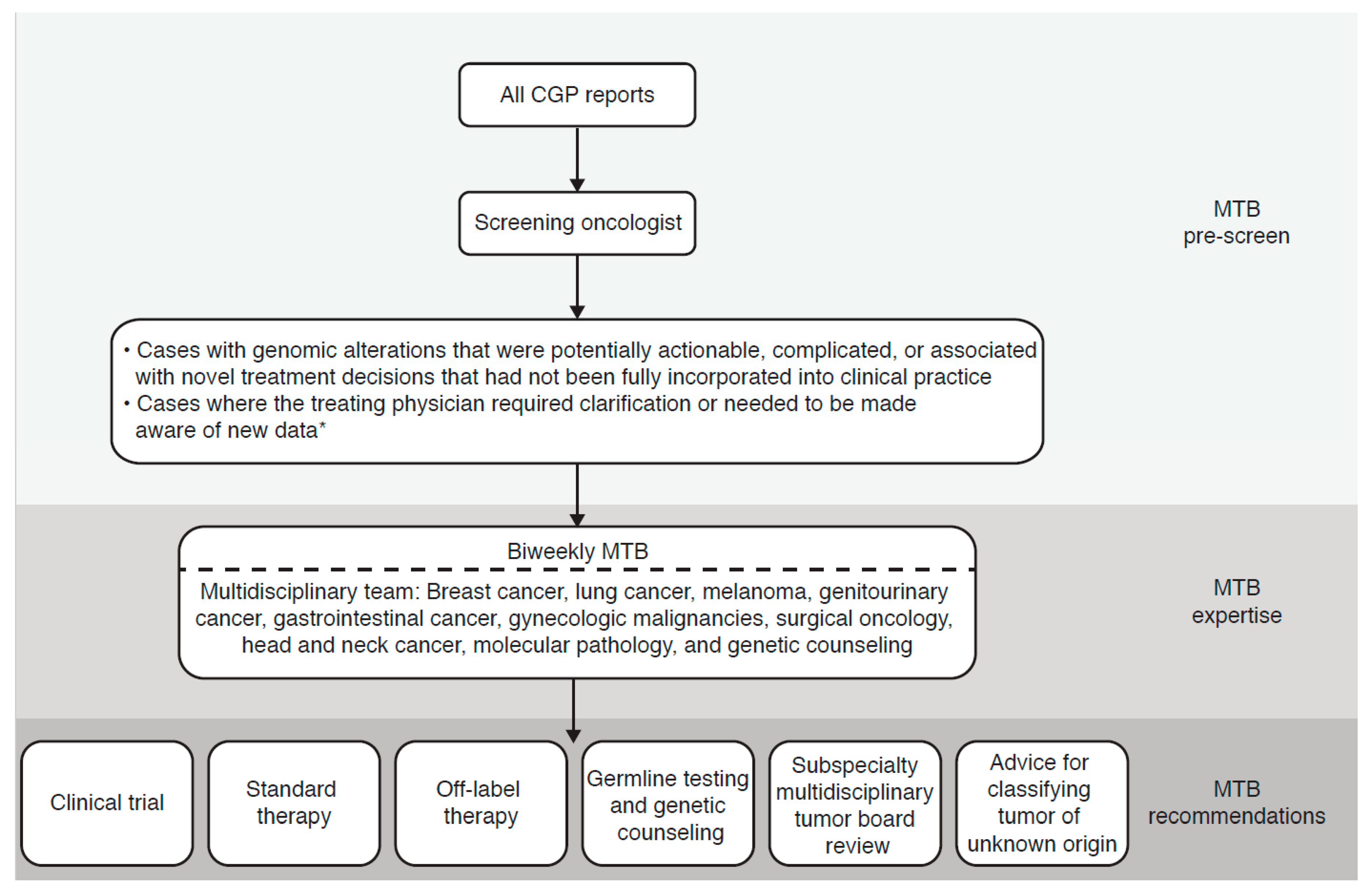
2.3. MTB
2.4. Analysis
3. Results
3.1. Patients and MTBs
3.2. MTB Recommendations and Adherence to Recommendations
3.3. The Value of MTBs beyond Standard Reporting
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Garraway, L.A. Genomics-driven oncology: Framework for an emerging paradigm. J. Clin. Oncol. 2013, 31, 1806–1814. [Google Scholar] [CrossRef] [PubMed]
- Sholl, L.M.; Do, K.; Shivdasani, P.; Cerami, E.; Dubuc, A.M.; Kuo, F.C.; Garcia, E.P.; Jia, Y.; Davineni, P.; Abo, R.P.; et al. Institutional implementation of clinical tumor profiling on an unselected cancer population. JCI Insight. 2016, 1, e87062. [Google Scholar] [CrossRef] [PubMed]
- Planchard, D.; Popat, S.; Kerr, K.; Novello, S.; Smit, E.F.; Faivre-Finn, C.; Mok, T.S.; Reck, M.; Van Schil, P.E.; Hellmann, M.D.; et al. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2019, 30, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Lindeman, N.I.; Cagle, P.T.; Aisner, D.L.; Arcila, M.E.; Beasley, M.B.; Bernicker, E.; Colasacco, C.; Dacic, S.; Hirsch, F.R.; Kerr, K.; et al. Updated molecular testing guideline for the selection of lung cancer patients for treatment with targeted tyrosine kinase inhibitors: Guideline from the College of American Pathologists, the International Association for the Study of Lung Cancer, and the Association for Molecular Pathology. Arch. Pathol. Lab. Med. 2018, 142, 321–346. [Google Scholar] [CrossRef] [PubMed]
- Manahan, E.R.; Kuerer, H.M.; Sebastian, M.; Hughes, K.S.; Boughey, J.C.; Euhus, D.M.; Boolbol, S.K.; Taylor, W.A. Consensus guidelines on genetic testing for hereditary breast cancer from the American Society of Breast Surgeons. Ann. Surg Oncol. 2019, 26, 3025–3031. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration (FDA). ZEJULA® (niraparib) Prescribing Information. 2017. Available online: https://www.mdpi.com/journal/jpm/special_issues/personalized_oncology (accessed on 22 November 2020).
- Moore, K.N.; Secord, A.A.; Geller, M.A.; Miller, D.S.; Cloven, N.; Fleming, G.F.; Hendrickson, A.E.W.; Azodi, M.; DiSilvestro, P.; Oza, A.M.; et al. Niraparib monotherapy for late-line treatment of ovarian cancer (QUADRA): A multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2019, 20, 636–648. [Google Scholar] [CrossRef]
- Sepulveda, A.R.; Hamilton, S.R.; Allegra, C.J.; Grody, W.; Cushman-Vokoun, A.M.; Funkhouser, W.K.; Kopetz, S.E.; Lieu, C.; Lindor, N.M.; Minsky, B.D.; et al. Molecular biomarkers for the evaluation of colorectal cancer: Guideline from the American Society for Clinical Pathology, College of American Pathologists, Association for Molecular Pathology, and American Society of Clinical Oncology. J. Mol. Diagn. 2017, 19, 187–225. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.H.; Johnson, A.; Albacker, L.; Wang, K.; Chmielecki, J.; Frampton, G.; Gay, L.; Elvin, J.A.; Vergilio, J.A.; Ali, S.; et al. Comprehensive genomic profiling facilitates implementation of the National Comprehensive Cancer Network Guidelines for lung cancer biomarker testing and identifies patients who may benefit from enrollment in mechanism-driven clinical trials. Oncologist 2016, 21, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Vakiani, E. Molecular testing of colorectal cancer in the modern era: What are we doing and why? Surg. Pathol. Clin. 2017, 10, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration (FDA). FDA Fact Sheet: CDRH’S Approach to Tumor Profiling Next Generation Sequencing Tests, 2017. Available online: https://www.fda.gov/media/109050/download (accessed on 22 November 2020).
- Gray, S.W.; Hicks-Courant, K.; Cronin, A.; Rollins, B.J.; Weeks, J.C. Physicians’ attitudes about multiplex tumor genomic testing. J. Clin. Oncol. 2014, 32, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- van der Velden, D.L.; van Herpen, C.M.L.; van Laarhoven, H.W.M.; Smit, E.F.; Groen, H.J.M.; Willems, S.M.; Nederlof, P.M.; Langenberg, M.H.G.; Cuppen, E.; Sleijfer, S.; et al. Molecular tumor boards: Current practice and future needs. Ann. Oncol. 2017, 28, 3070–3075. [Google Scholar] [CrossRef] [PubMed]
- Tafe, L.J.; Gorlov, I.P.; de Abreu, F.B.; Lefferts, J.A.; Liu, X.; Pettus, J.R.; Marotti, J.D.; Bloch, K.J.; Memoli, V.A.; Suriawinata, A.A.; et al. Implementation of a molecular tumor board: The impact on treatment decisions for 35 patients evaluated at Dartmouth-Hitchcock Medical Center. Oncologist 2015, 20, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Farhangfar, C.J.; Morgan, O.; Concepcion, C.; Hwang, J.J.; Mileham, K.F.; Carrizosa, D.R.; Dellinger, B.; Farhangfar, F.; Kim, E.S. Utilization of consultative molecular tumor board in community setting. J. Clin. Oncol. 2017, 35, 6508. [Google Scholar] [CrossRef]
- Clark, D.F.; Maxwell, K.N.; Powers, J.; Lieberman, D.B.; Ebrahimzadeh, J.; Long, J.M.; McKenna, D.; Shah, P.; Bradbury, A.; Morrissette, J.J.D.; et al. Identification and confirmation of potentially actionable germline mutations in tumor-only genomic sequencing. JCO Prec. Oncol. 2019, 3, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Reitsma, M.; Fox, J.; Borre, P.V.; Cavanaugh, M.; Chudnovsky, Y.; Erlich, R.L.; Gribbin, T.E.; Anhorn, R. Effect of a collaboration between a health plan, oncology practice, and comprehensive genomic profiling company from the payer perspective. J. Manag. Care Spec. Pharm. 2019, 25, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Basse, C.; Morel, C.; Alt, M.; Sablin, M.P.; Franck, C.; Pierron, G.; Callens, C.; Melaabi, S.; Masliah-Planchon, J.; Bataillon, G.; et al. Relevance of a molecular tumour board (MTB) for patients’ enrolment in clinical trials: Experience of the Institut Curie. ESMO Open 2018, 3, e000339. [Google Scholar] [CrossRef] [PubMed]
- Schwaederle, M.; Parker, B.A.; Schwab, R.B.; Fanta, P.T.; Boles, S.G.; Daniels, G.A.; Bazhenova, L.A.; Subramanian, R.; Coutinho, A.C.; Ojeda-Fournier, H.; et al. Molecular tumor board: The University of California-San Diego Moores Cancer Center experience. Oncologist 2014, 19, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Harada, S.; Arend, R.; Dai, Q.; Levesque, J.A.; Winokur, T.S.; Guo, R.; Heslin, M.J.; Nabell, L.; Nabors, L.B.; Limdi, N.A.; et al. Implementation and utilization of the molecular tumor board to guide precision medicine. Oncotarget 2017, 8, 57845. [Google Scholar] [CrossRef] [PubMed]
- Bryce, A.H.; Egan, J.B.; Borad, M.J.; Stewart, A.K.; Nowakowski, G.S.; Chanan-Khan, A.; Patnaik, M.M.; Ansell, S.M.; Banck, M.S.; Robinson, S.I.; et al. Experience with precision genomics and tumor board, indicates frequent target identification, but barriers to delivery. Oncotarget 2017, 8, 27145. [Google Scholar] [CrossRef] [PubMed]
- Singer, J.; Irmisch, A.; Ruscheweyh, H.J.; Singer, F.; Toussaint, N.C.; Levesque, M.P.; Stekhoven, D.J.; Beerenwinkel, N. Bioinformatics for precision oncology. Briefings Bioinforma. 2019, 20, 778–788. [Google Scholar] [CrossRef] [PubMed]
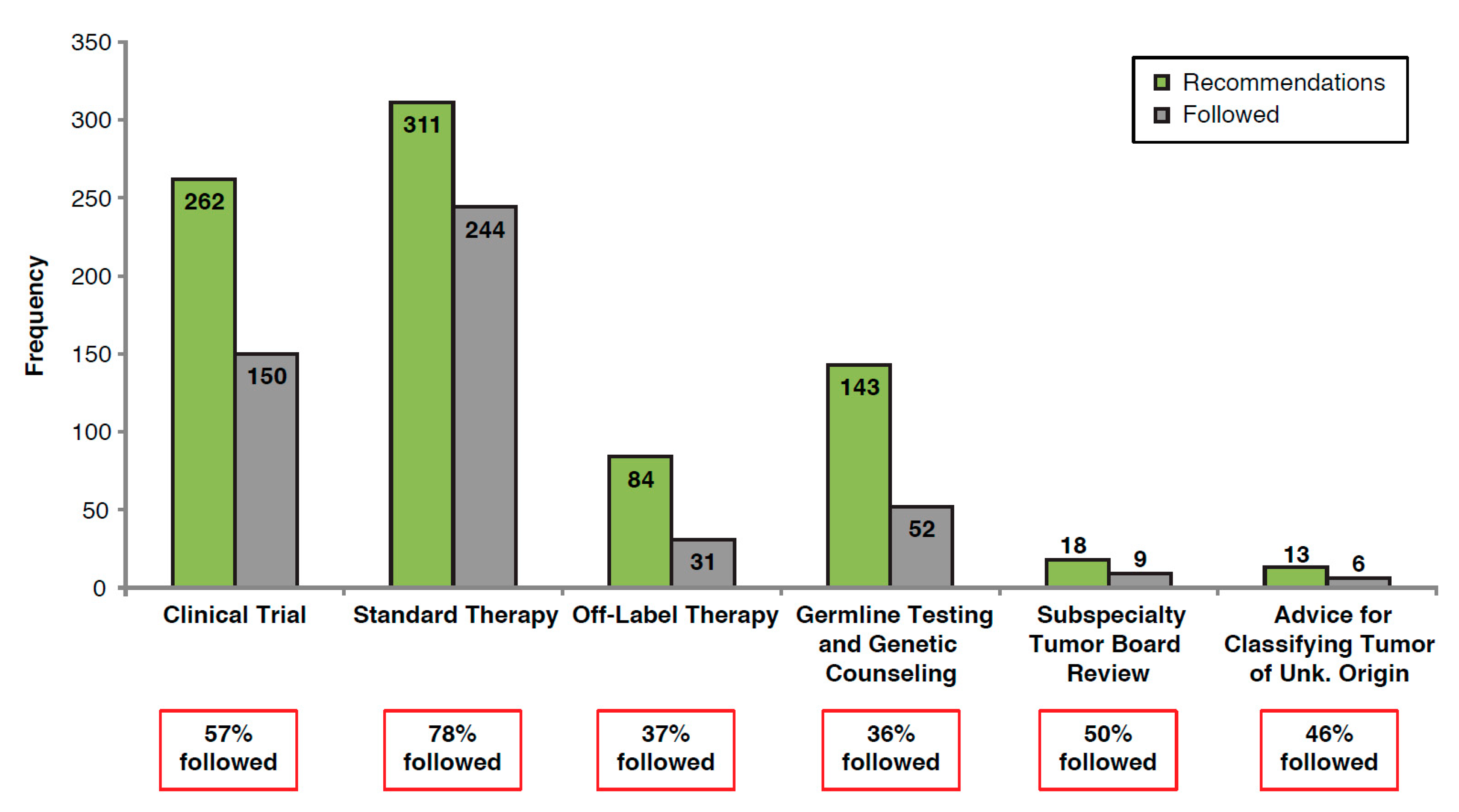
| Recommendation Category | Number of Recommendations | Proportion of Patients Receiving Recommendations 1 (%) |
|---|---|---|
| Clinical trial | 262 | 43 |
| Standard therapy | 311 | 51 |
| Off-label therapy | 84 | 14 |
| Germline testing and genetic counseling | 143 | 23 |
| Subspecialty multidisciplinary tumor board review | 18 | 3 |
| Advice for classifying tumor of unknown origin | 13 | 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
VanderWalde, A.; Grothey, A.; Vaena, D.; Vidal, G.; ElNaggar, A.; Bufalino, G.; Schwartzberg, L. Establishment of a Molecular Tumor Board (MTB) and Uptake of Recommendations in a Community Setting. J. Pers. Med. 2020, 10, 252. https://doi.org/10.3390/jpm10040252
VanderWalde A, Grothey A, Vaena D, Vidal G, ElNaggar A, Bufalino G, Schwartzberg L. Establishment of a Molecular Tumor Board (MTB) and Uptake of Recommendations in a Community Setting. Journal of Personalized Medicine. 2020; 10(4):252. https://doi.org/10.3390/jpm10040252
Chicago/Turabian StyleVanderWalde, Ari, Axel Grothey, Daniel Vaena, Gregory Vidal, Adam ElNaggar, Gabriella Bufalino, and Lee Schwartzberg. 2020. "Establishment of a Molecular Tumor Board (MTB) and Uptake of Recommendations in a Community Setting" Journal of Personalized Medicine 10, no. 4: 252. https://doi.org/10.3390/jpm10040252
APA StyleVanderWalde, A., Grothey, A., Vaena, D., Vidal, G., ElNaggar, A., Bufalino, G., & Schwartzberg, L. (2020). Establishment of a Molecular Tumor Board (MTB) and Uptake of Recommendations in a Community Setting. Journal of Personalized Medicine, 10(4), 252. https://doi.org/10.3390/jpm10040252






