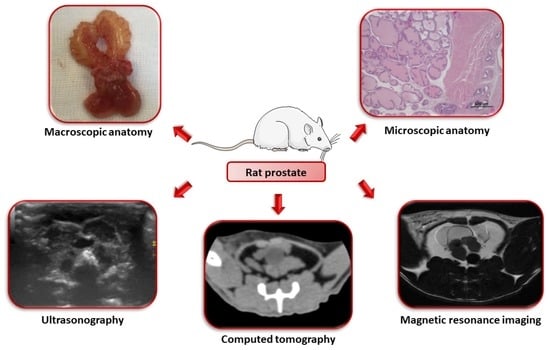
Anatomy and Imaging of Rat Prostate: Practical Monitoring in Experimental Cancer-Induced Protocols
Abstract
1. Introduction
2. Prostate Cancer Induction Protocol
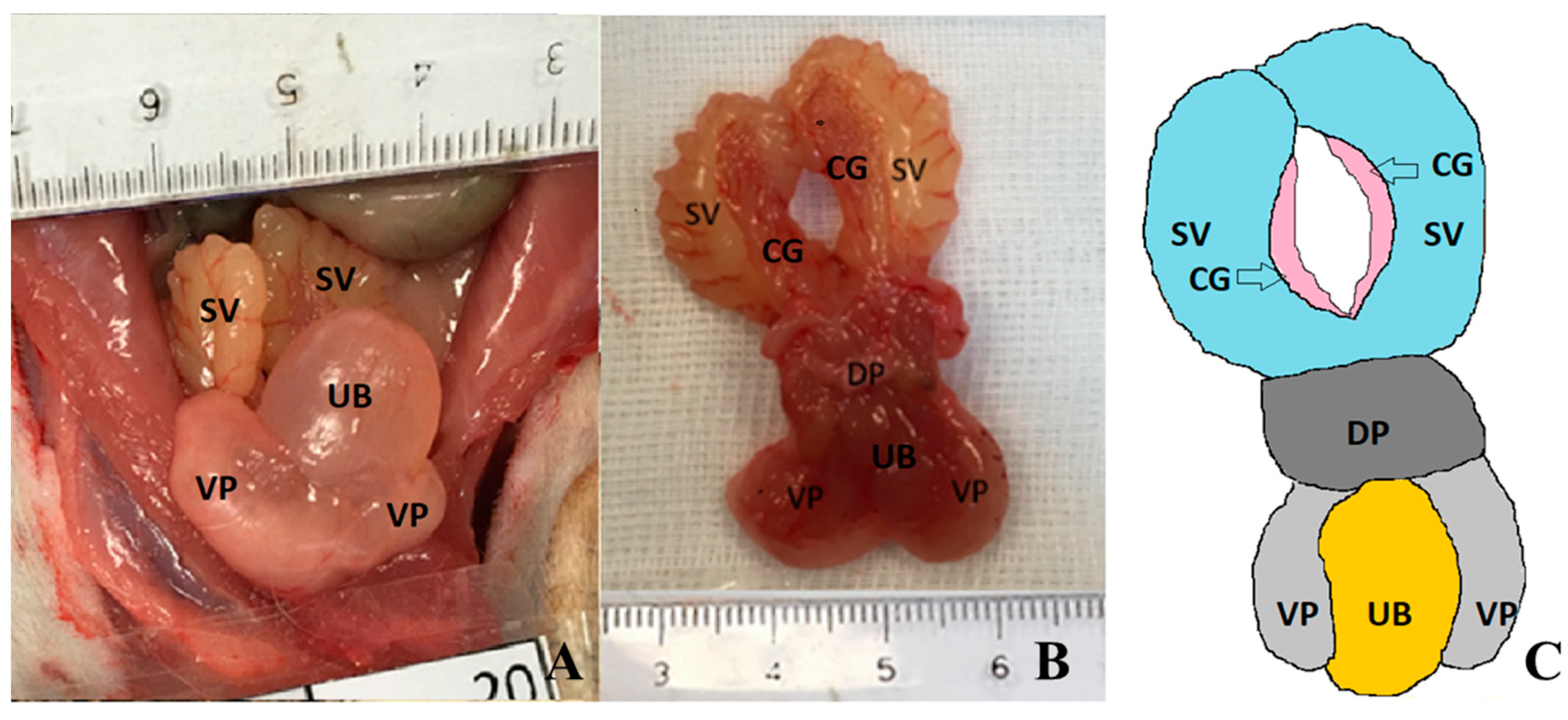
3. Macroscopic Anatomy
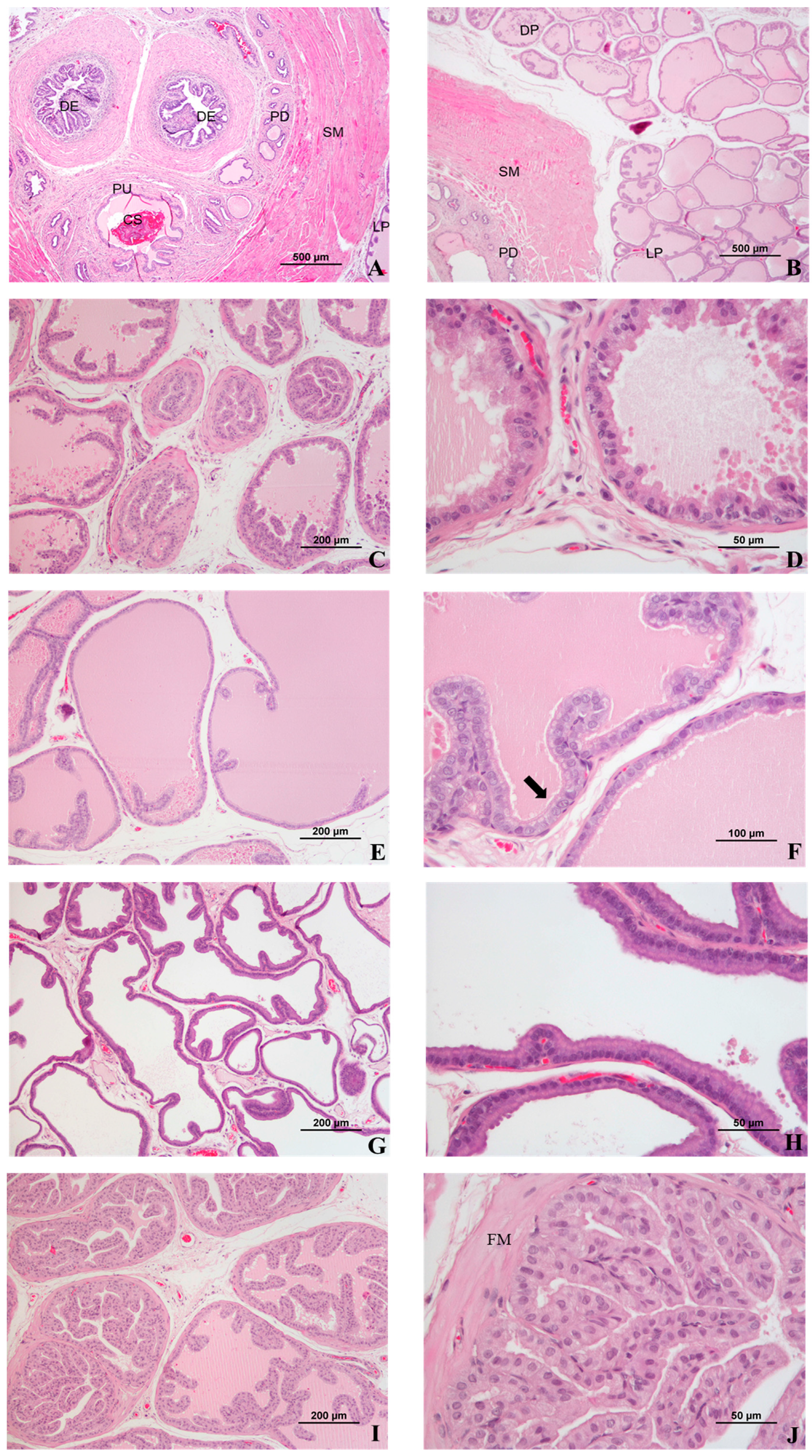
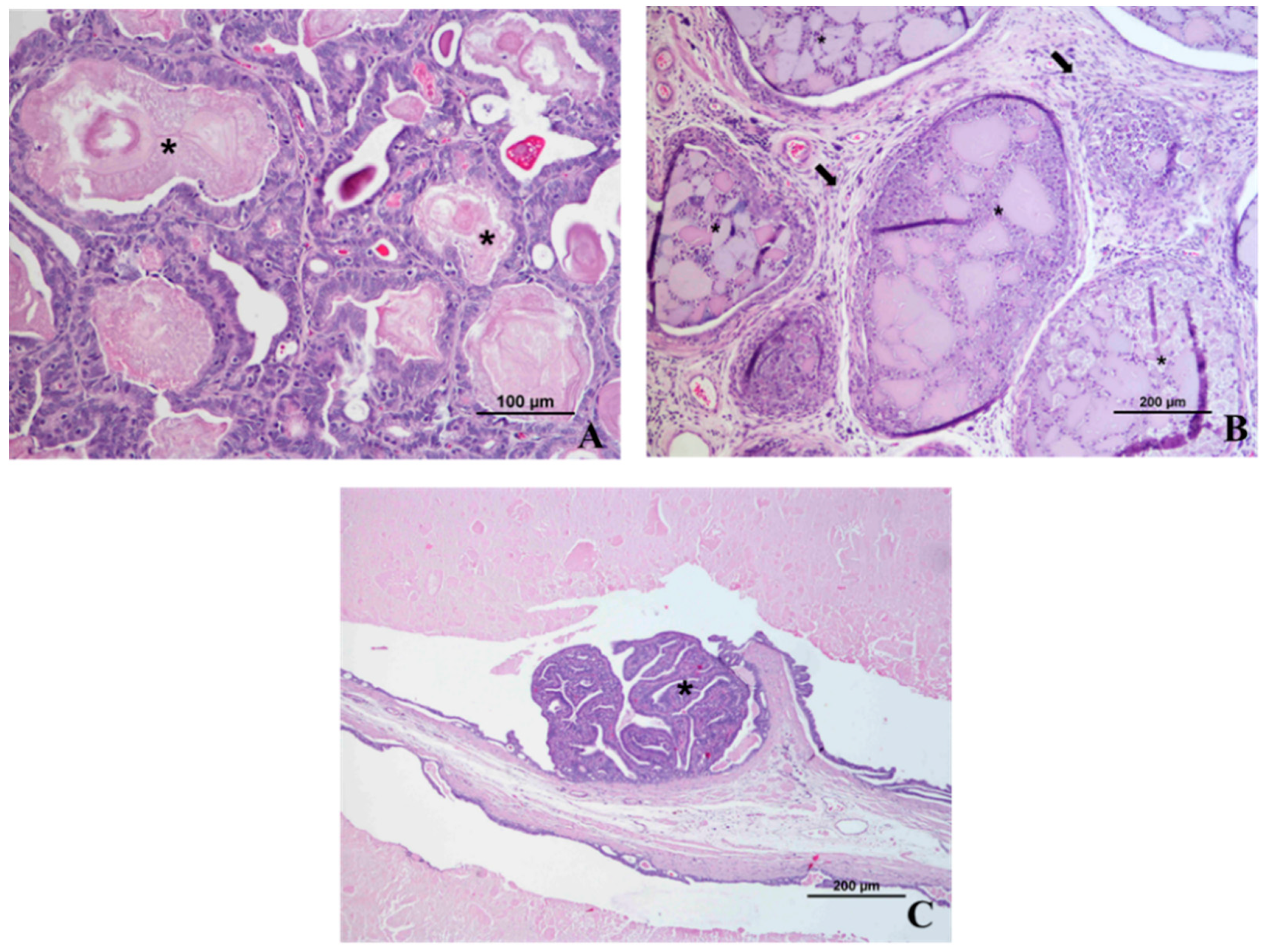
4. Microscopic Anatomy
5. Prostate Imaging
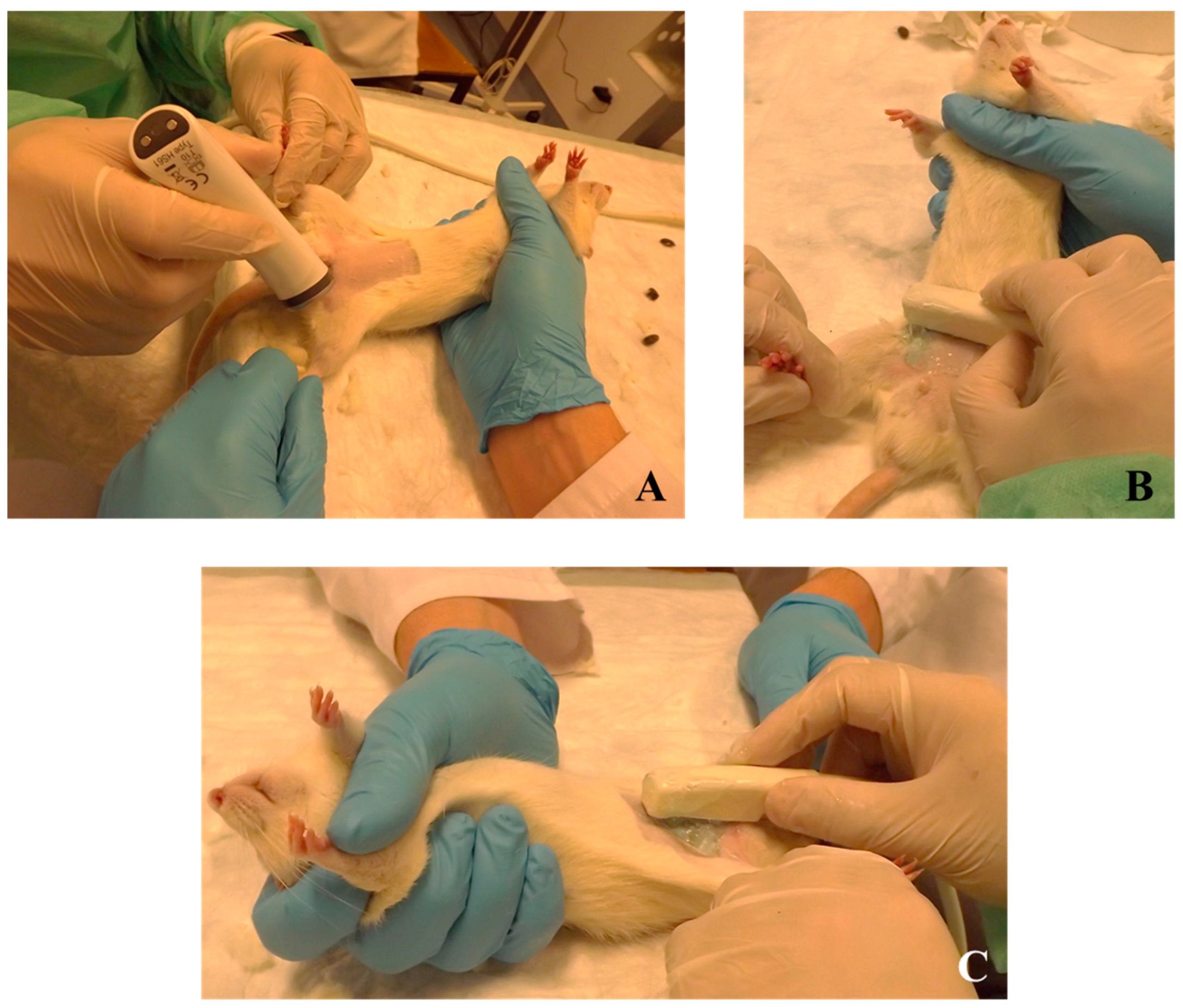
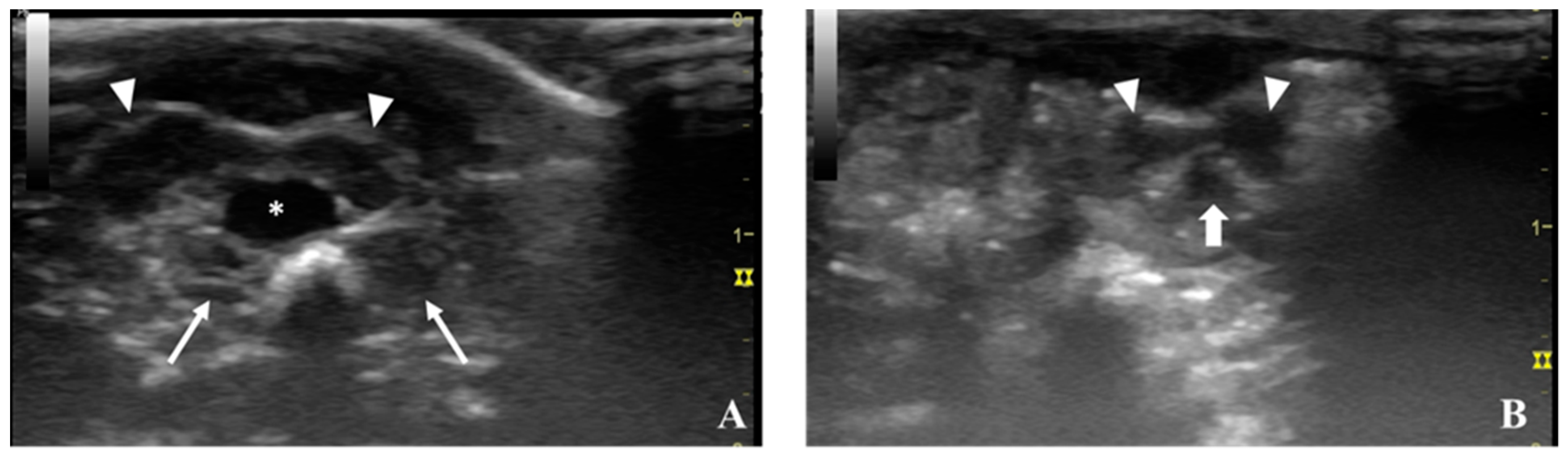
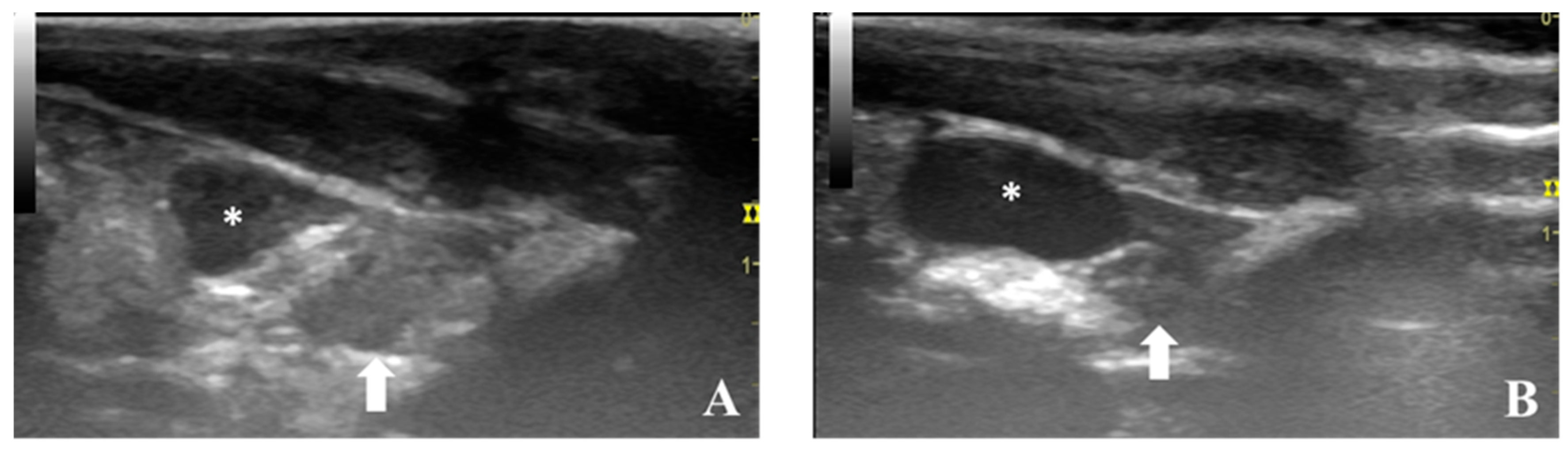
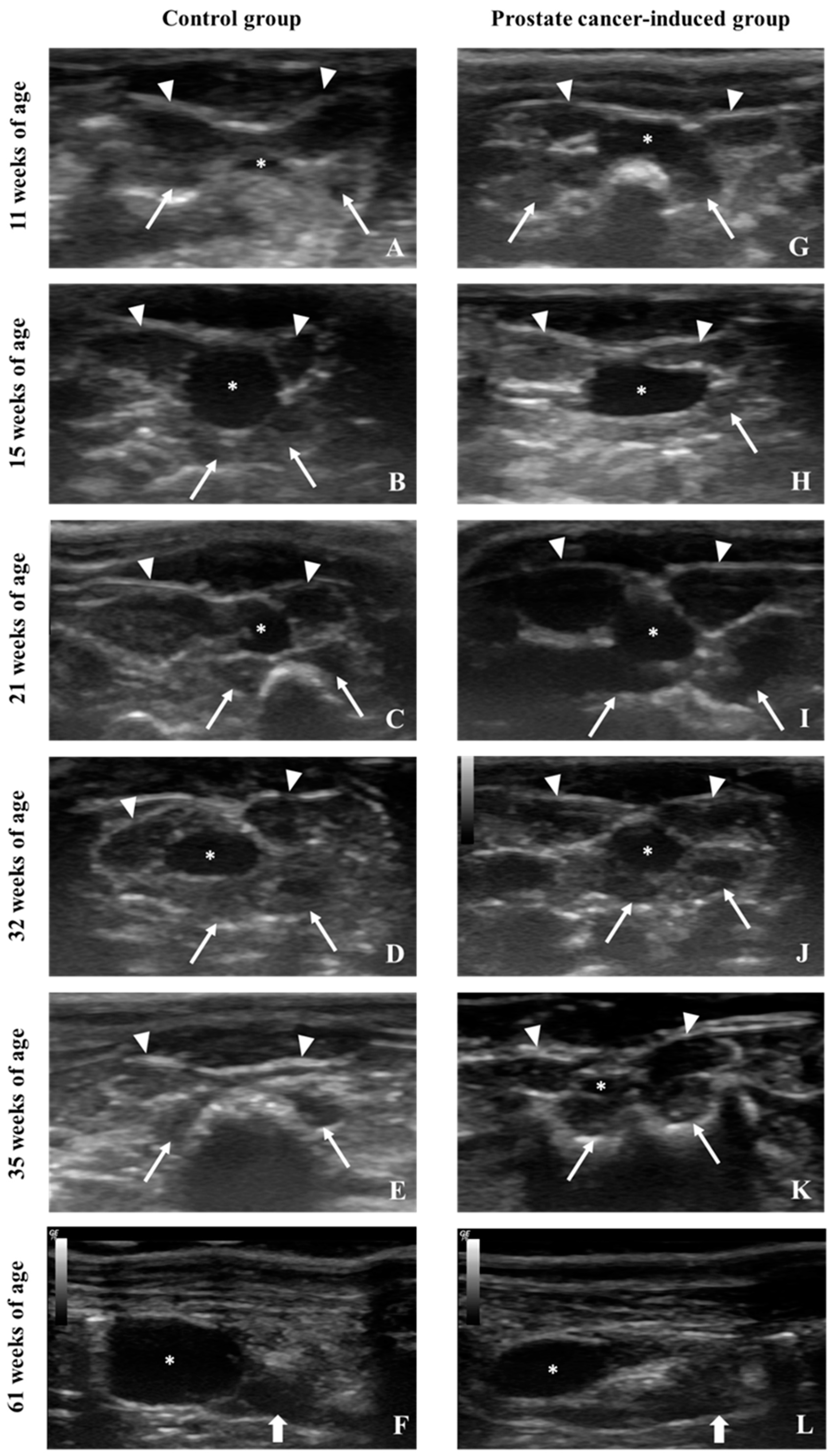
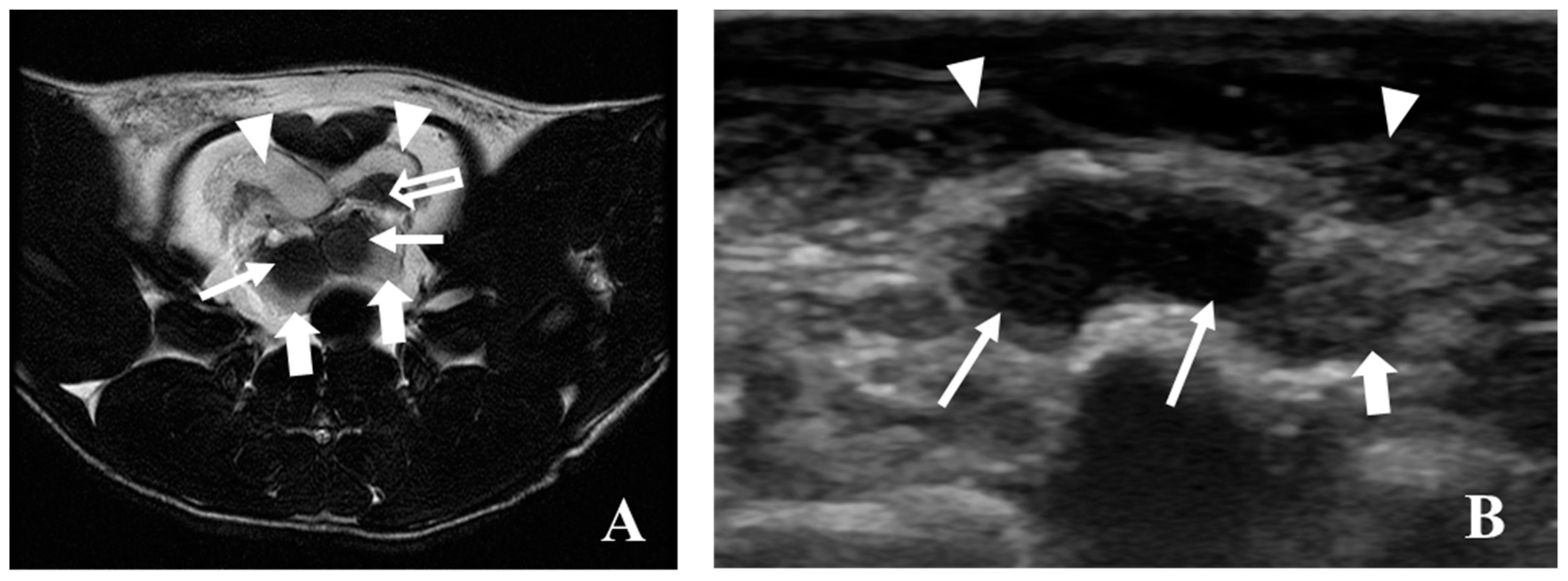
5.1. Ultrasonography
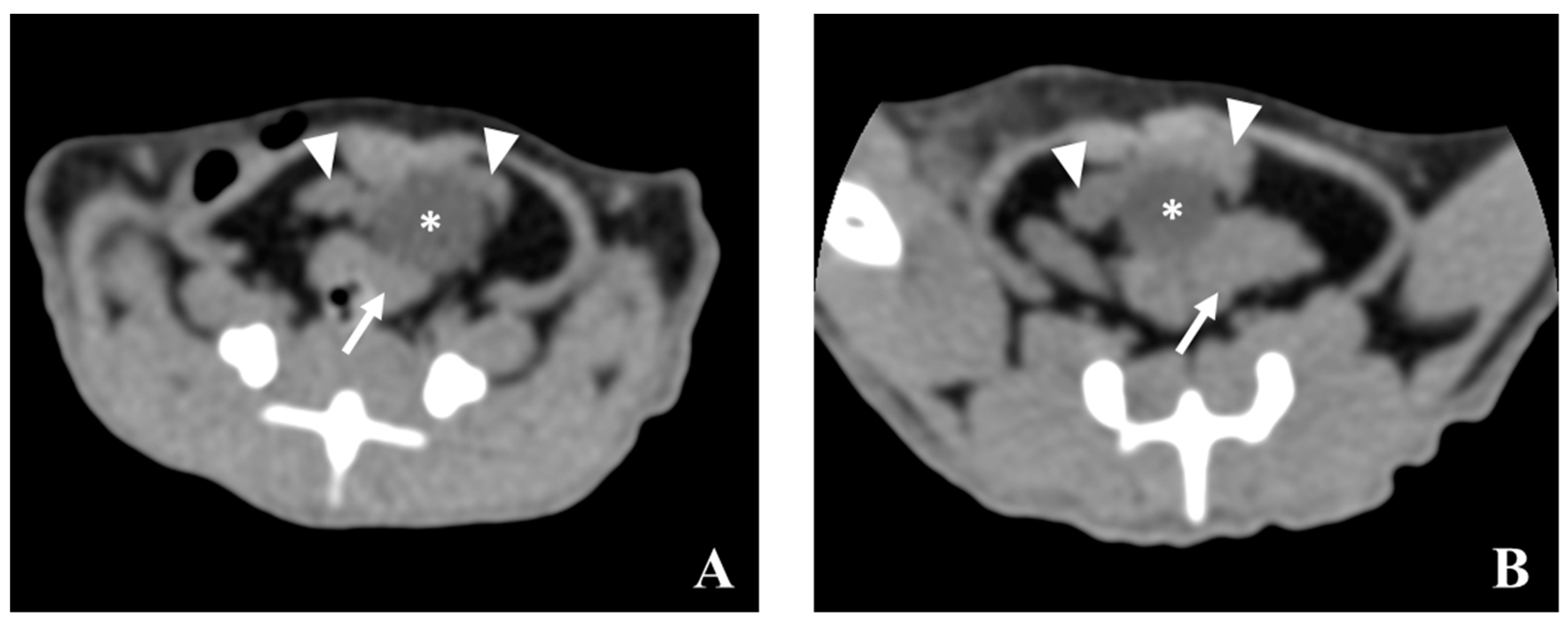
5.2. Computed Tomography (CT)
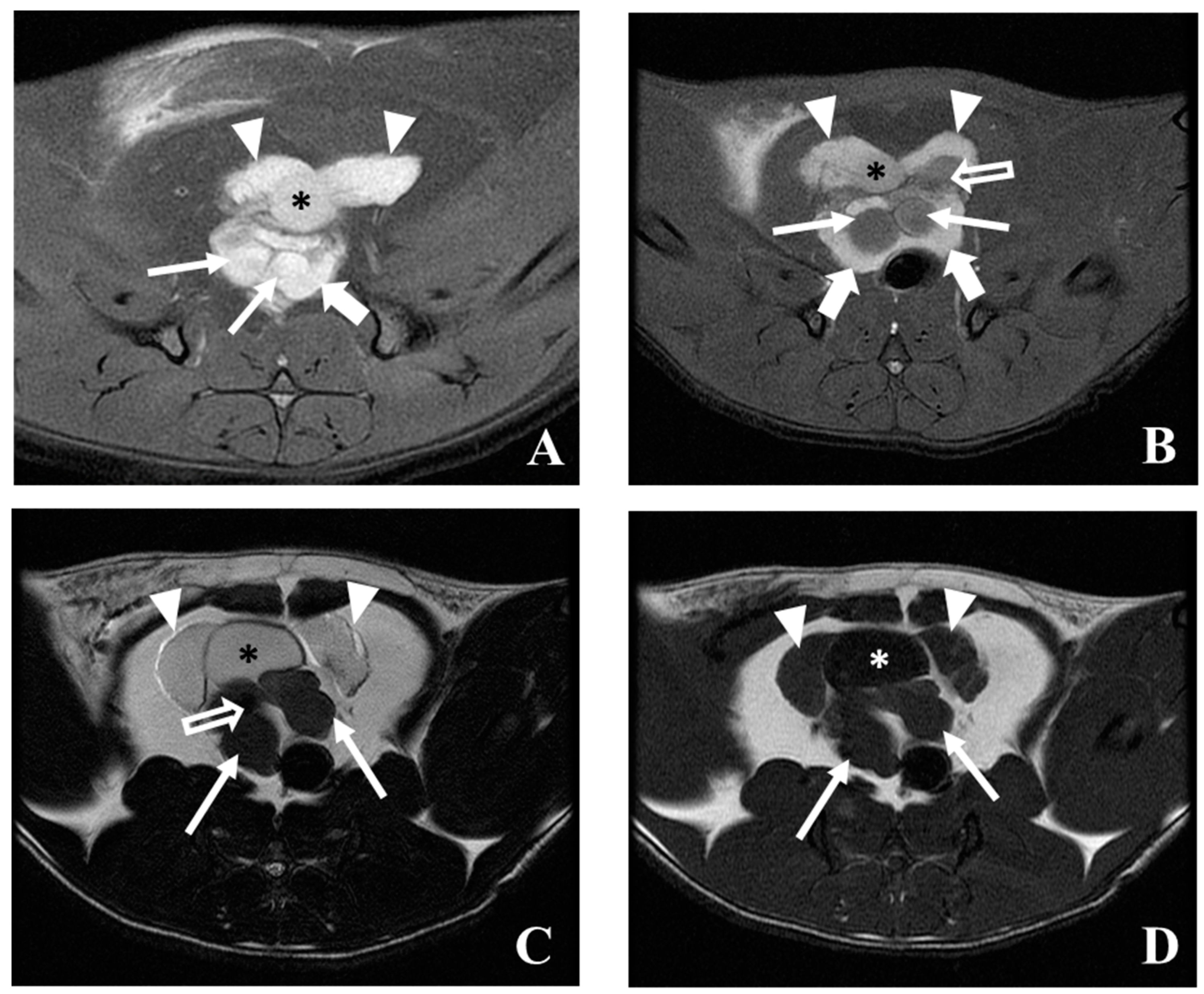
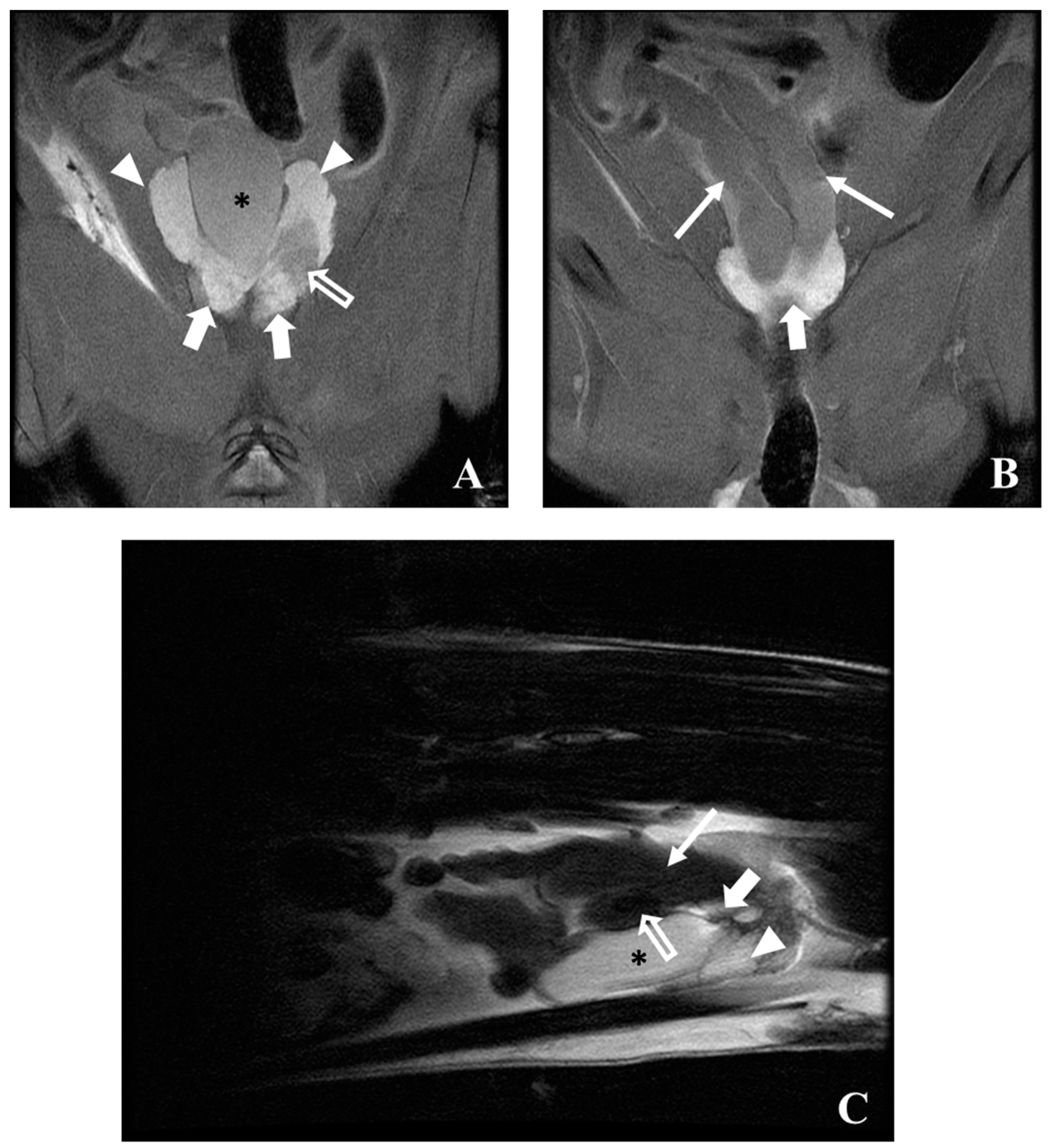
5.3. Magnetic Resonance Imaging (MRI)
5.4. Positron Emission Tomography/Computed Tomography (PET/CT)
6. Discussion
7. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, C.H.; Akin-Olugbade, O.; Kirschenbaum, A. Overview of Prostate Anatomy, Histology, and Pathology. Endocrinol. Metab. Clin. North Am. 2011, 40, 565–575. [Google Scholar] [CrossRef] [PubMed]
- Bhavsar, A.; Verma, S. Anatomic Imaging of the Prostate. Biomed. Res. Int. 2014, 2014, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Custodio, A.M.G.; Santos, F.C.A.; Campos, S.G.P.; Vilamaior, P.S.L.; Góes, R.M.; Taboga, S.R. Aging Effects on the Mongolian Gerbil Female Prostate (Skene’s Paraurethral Glands): Structural, Ultrastructural, Quantitative, and Hormonal Evaluations. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2008, 291, 463–474. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer, W.H.O. (WHO) Globocan 2012: Estimated cancer incidence, mortality and prevalence worldwide in 2012. Available online: http://gco.iarc.fr/today/data/factsheets/cancers/27-Prostate-fact-sheet.pdf (accessed on 29 June 2019).
- Shirai, T.; Takahashi, S.; Cui, L.; Futakuchi, M.; Kato, K.; Tamano, S.; Imaida, K. Experimental prostate carcinogenesis - rodent models. Mutat. Res. 2000, 462, 219–226. [Google Scholar] [CrossRef]
- Shirai, T. Significance of chemoprevention for prostate cancer development: Experimental in vivo approaches to chemoprevention. Pathol. Int. 2007, 58, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Nascimento-Gonçalves, E.; Faustino-Rocha, A.I.; Seixas, F.; Ginja, M.; Colaço, B.; Ferreira, R.; Fardilha, M.; Oliveira, P.A. Modelling human prostate cancer: Rat models. Life Sci. 2018, 203, 210–224. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.A.; Melchionna, R.H. Production of Tumors of the Prostate of the White Rat with 1:2-Benzpyrene. Am. J. Cancer 1937, 30, 731–741. [Google Scholar]
- Bosland, M.C. Chemical and hormonal induction of prostate cancer in animal models. Urol. Oncol. 1996, 2, 103–110. [Google Scholar] [CrossRef]
- Lee, C.; Holland, J. Anatomy, histology and ultrastructure (Correlation with Function), Prostate, Rat. In Genital System. Monographs on Pthology of Laboratory Animals; Jones, T., Mohr, U., Hunt, R., Eds.; Springer: Berlin/Heidelberg, Germany, 1987; pp. 239–240. [Google Scholar]
- Hayashi, N.; Sugimura, Y.; Kawamura, J.; Donjacour, A.A.; Cunha, G.R. Morphological and functional heterogeneity in the rat prostatic gland. Biol. Reprod. 1991, 45, 308–321. [Google Scholar] [CrossRef]
- Creasy, D.; Bube, A.; de Rijk, E.; Kandori, H.; Kuwahara, M.; Masson, R.; Nolte, T.; Reams, R.; Regan, K.; Rehm, S.; et al. Proliferative and Nonproliferative Lesions of the Rat and Mouse Male Reproductive System. Toxicol. Pathol. 2012, 40, 40S–121S. [Google Scholar] [CrossRef]
- Knoblaugh, S.; Tretiakova, M.; Hukkanen, R. Male reproductive system. In Comparative Anatomy and Histology: A Mouse, Rat and Human Atlas; Treuting, P., Dintzis, S., Montine, K., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 335–338. [Google Scholar]
- Jesik, C.J.; Holland, J.M.; Lee, C. An anatomic and histologic study of the rat prostate. Prostate 1982, 3, 81–97. [Google Scholar] [CrossRef]
- Suwa, T.; Nyska, A.; Peckham, J.C.; Hailey, J.R.; Mahler, J.F.; Haseman, J.K.; Maronpot, R.R. A Retrospective Analysis of Background Lesions and Tissue Accountability for Male Accessory Sex Organs in Fischer-344 Rats. Toxicol. Pathol. 2001, 29, 467–478. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Sensibar, J.A.; Dudek, S.M.; Hiipakka, R.A.; Liao, S.T. Prostatic ductal system in rats: Regional variation in morphological and functional activities. Biol. Reprod. 1990, 43, 1079–1086. [Google Scholar] [CrossRef] [PubMed]
- Whitney, K. Male accessory sex glands. In Boorman´s Pathology of the Rat: Reference and Atlas; Suttie, A., Leininger, J., Bradley, A., Eds.; Academic Press – Elsevier: Cambridge, MA, USA, 2018; pp. 579–586. [Google Scholar]
- Santamaría, L.; Ingelmo, I.; Alonso, L.; Pozuelo, J.; Rodriguez, R. The prostate of the rat. In Neuroendocrine Cells and Peptidergic Innervation in Human and Rat Prostate; Springer: Berlin/Heidelberg, Germany, 2007; pp. 39–42. [Google Scholar]
- Picut, C.; Remick, A. Male reproductive system. In Atlas of Histology of the Juvenile Rat; Parker, G., Picut, C., Eds.; Academic Press - Elsevier: Cambridge, MA, USA, 2016. [Google Scholar]
- Boorman, G.; Elwell, M.; Mitsumori, K. Male accessory sex glands, penis and scrotum. In Payhology of the Fischer Rat; Boorman, G., Ed.; Academic Press: Cambridge, MA, USA, 1990. [Google Scholar]
- Eastham, J. Prostate cancer screening. Investig. Clin. Urol. 2017, 58, 217–219. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.M.; Jorgensen, J.T.; Binderup, T.; Kjaer, A. Tumor volume in subcutaneous mouse xenografts measured by microCT is more accurate and reproducible than determined by 18F-FDG-microPET or external caliper. BMC Med. Imaging 2008, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, J.A. Ultrasound in sports medicine. Radiol. Clin. North Am. 2002, 40, 363–386. [Google Scholar] [CrossRef]
- Lento, P.H.; Primack, S. Advances and utility of diagnostic ultrasound in musculoskeletal medicine. Curr. Rev. Musculoskelet. Med. 2008, 1, 24–31. [Google Scholar] [CrossRef]
- Nazarian, L. The top 10 reasons musculoskeletal sonography in an important complementary or alternative tecnhique to MRI. Am. J. Roentgenol. 2008, 190, 1621–1626. [Google Scholar] [CrossRef]
- Rudin, M.; Qureshi, S.; Tolcsvai, L.; Siegel, R.A. Visualization and quantification of transplanted dunning prostate tumors in rats using magnetic resonance imaging. Prostate 1988, 12, 333–341. [Google Scholar] [CrossRef]
- Chen, Y.; Li, C.; Lu, Y.; Zhuang, H.; Gu, W.; Liu, B.; Liu, F.; Sun, J.; Yan, B.; Weng, D.; et al. IL-10-producing CD1dhiCD5+regulatory B cells may play a critical role in modulating immune homeostasis in silicosis patients. Front. Immunol. 2017, 8, 1613–1619. [Google Scholar] [CrossRef]
- Trabulsi, E.J.; Merriam, W.G.; Gomella, L.G. New imaging techniques in prostate cancer. Curr. Urol. Rep. 2006, 7, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Rubin, G.D. Computed Tomography: Revolutionizing the Practice of Medicine for 40 Years. Radiology 2014, 273, S45–S74. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.W. CT scans: Balancing health risks and medical benefits. Environ. Health Perspect. 2012, 120, A118–A121. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-X.; Min, P.-Q.; Song, B.; Xiao, B.-L.; Liu, Y.; Ge, Y.-H. Single-level dynamic spiral CT of hepatocellular carcinoma: Correlation between imaging features and density of tumor microvessels. World J. Gastroenterol. 2004, 10, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Jensen, H.; Doughty, R.W.; Grant, D.; Myhre, O. The Effects of the Iodinated X-Ray Contrast Media Iodixanol, Iohexol, Iopromide, and Ioversol on the Rat Kidney Epithelial Cell Line NRK 52-E. Ren. Fail. 2011, 33, 426–433. [Google Scholar] [CrossRef]
- Tangel, M.R.; Rastinehad, A.R. Advances in prostate cancer imaging. F1000Research 2018, 7, 1337. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Das, S. A Review of Imaging Methods for Prostate Cancer Detection. Biomed. Eng. Comput. Biol. 2016, 7, 1–15. [Google Scholar] [CrossRef]
- Hartwig, V.; Giovannetti, G.; Vanello, N.; Lombardi, M.; Landini, L.; Simi, S. Biological effects and safety in magnetic resonance imaging: A review. Int. J. Environ. Res. Public Health 2009, 6, 1778–1798. [Google Scholar] [CrossRef]
- Kristoffersen Wiberg, M.; Aspelin, P.; Perbeck, L.; Boné, B. Value of MR imaging in clinical evaluation of breast lesions. Acta Radiol. 2002, 43, 275–281. [Google Scholar] [CrossRef]
- Jadvar, H. Imaging evaluation of prostate cancer with 18F-fluorodeoxyglucose PET/CT: Utility and limitations. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, S5–S10. [Google Scholar] [CrossRef]
- Almuhaideb, A.; Papathanasiou, N.; Bomanji, J. 18F-FDG PET/CT Imaging In Oncology. Ann. Saudi Med. 2011, 31, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Wallitt, K.L.; Khan, S.R.; Dubash, S.; Tam, H.H.; Khan, S.; Barwick, T.D. Clinical PET Imaging in Prostate Cancer. RadioGraphics 2017, 37, 1512–1536. [Google Scholar] [CrossRef] [PubMed]
- Mason, B.R.; Eastham, J.A.; Davis, B.J.; Mynderse, L.A.; Pugh, T.J.; Lee, R.J.; Ippolito, J.E. Current Status of MRI and PET in the NCCN Guidelines for Prostate Cancer. J. Natl. Compr. Cancer Netw. 2019, 17, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, B.E.; Kramer, D.J.; Wiitala, L. Applications of musculoskeletal sonography. J. Clin. Ultrasound 1999, 27, 293–318. [Google Scholar] [CrossRef]
- De Visschere, P.J.L.; Standaert, C.; Fütterer, J.J.; Villeirs, G.M.; Panebianco, V.; Walz, J.; Maurer, T.; Hadaschik, B.A.; Lecouvet, F.E.; Giannarini, G.; et al. A Systematic Review on the Role of Imaging in Early Recurrent Prostate Cancer. Eur. Urol. Oncol. 2019, 2, 47–76. [Google Scholar] [CrossRef] [PubMed]
- Treglia, G.; Pereira Mestre, R.; Ferrari, M.; Bosetti, D.G.; Pascale, M.; Oikonomou, E.; De Dosso, S.; Jermini, F.; Prior, J.O.; Roggero, E.; et al. Radiolabelled choline versus PSMA PET/CT in prostate cancer restaging: A meta-analysis. Am. J. Nucl. Med. Mol. Imaging 2019, 9, 127–139. [Google Scholar] [PubMed]
- Mena-Romano, P.; Cheng, C.; Glowa, C.; Peschke, P.; Pan, L.; Haberkorn, U.; Dimitrakopoulou-Strauss, A.; Karger, C.P. Measurement of hypoxia-related parameters in three sublines of a rat prostate carcinoma using dynamic (18)F-FMISO-Pet-Ct and quantitative histology. Am. J. Nucl. Med. Mol. Imaging 2015, 5, 348–362. [Google Scholar]
- Lisova, K.; Sergeev, M.; Evans-Axelsson, S.; Stuparu, A.D.; Beykan, S.; Collins, J.; Jones, J.; Lassmann, M.; Herrmann, K.; Perrin, D.; et al. Microscale radiosynthesis, preclinical imaging and dosimetry study of [18F]AMBF 3 -TATE: A potential PET tracer for clinical imaging of somatostatin receptors. Nucl. Med. Biol. 2018, 61, 36–44. [Google Scholar] [CrossRef]
- Lucia, M.S.; Bostwick, D.G.; Bosland, M.; Cockett, A.T.K.; Knapp, D.W.; Leav, I.; Pollard, M.; Rinker-Schaeffer, C.; Shirai, T.; Watkins, B.A. Workgroup I: Rodent models of prostate cancer. Prostate 1998, 36, 49–55. [Google Scholar] [CrossRef]
- Fagundes, D.J.; Taha, M.O. Modelo animal de doença: Critérios de escolha e espécies de animais de uso corrente. Acta Cirúrgica Bras. 2004, 19, 59–65. [Google Scholar] [CrossRef]
- Hodge, K.K.; McNeal, J.E.; Stamey, T.A. Ultrasound guided transrectal core biopsies of the palpably abnormal prostate. J. Urol. 1989, 142, 66–70. [Google Scholar] [CrossRef]
- Golan, S.; Nidam, M.; Bernstine, H.; Baniel, J.; Groshar, D. Dynamic 11C-Choline PET/CT for the primary diagnosis of prostate cancer. Int. Braz. J. Urol. 2018, 44, 900–905. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ginja, M.; Pires, M.J.; Gonzalo-Orden, J.M.; Seixas, F.; Correia-Cardoso, M.; Ferreira, R.; Fardilha, M.; Oliveira, P.A.; Faustino-Rocha, A.I. Anatomy and Imaging of Rat Prostate: Practical Monitoring in Experimental Cancer-Induced Protocols. Diagnostics 2019, 9, 68. https://doi.org/10.3390/diagnostics9030068
Ginja M, Pires MJ, Gonzalo-Orden JM, Seixas F, Correia-Cardoso M, Ferreira R, Fardilha M, Oliveira PA, Faustino-Rocha AI. Anatomy and Imaging of Rat Prostate: Practical Monitoring in Experimental Cancer-Induced Protocols. Diagnostics. 2019; 9(3):68. https://doi.org/10.3390/diagnostics9030068
Chicago/Turabian StyleGinja, Mário, Maria J. Pires, José M. Gonzalo-Orden, Fernanda Seixas, Miguel Correia-Cardoso, Rita Ferreira, Margarida Fardilha, Paula A. Oliveira, and Ana I. Faustino-Rocha. 2019. "Anatomy and Imaging of Rat Prostate: Practical Monitoring in Experimental Cancer-Induced Protocols" Diagnostics 9, no. 3: 68. https://doi.org/10.3390/diagnostics9030068
APA StyleGinja, M., Pires, M. J., Gonzalo-Orden, J. M., Seixas, F., Correia-Cardoso, M., Ferreira, R., Fardilha, M., Oliveira, P. A., & Faustino-Rocha, A. I. (2019). Anatomy and Imaging of Rat Prostate: Practical Monitoring in Experimental Cancer-Induced Protocols. Diagnostics, 9(3), 68. https://doi.org/10.3390/diagnostics9030068