FDG-PET/CT Versus Contrast-Enhanced CT for Response Evaluation in Metastatic Breast Cancer: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Systematic Literature Search
- Breast cancer AND
- Metastasis AND
- Response evaluation criteria in solid tumors OR PET response criteria in solid tumors
2.2. Selection of Literature
- Studies that focused on or included patients with MBC
- Scans were used to evaluate response to treatment
- Scans were evaluated by RECIST and PERCIST
- Included patients should have at least one FDG-PET/CT and one CE-CT-scan after diagnosis of metastases and before initiation of a new treatment regimen
- Patients with locally advanced breast cancer
- RECIST evaluations by MRI
2.3. Quality Assessment
3. Results
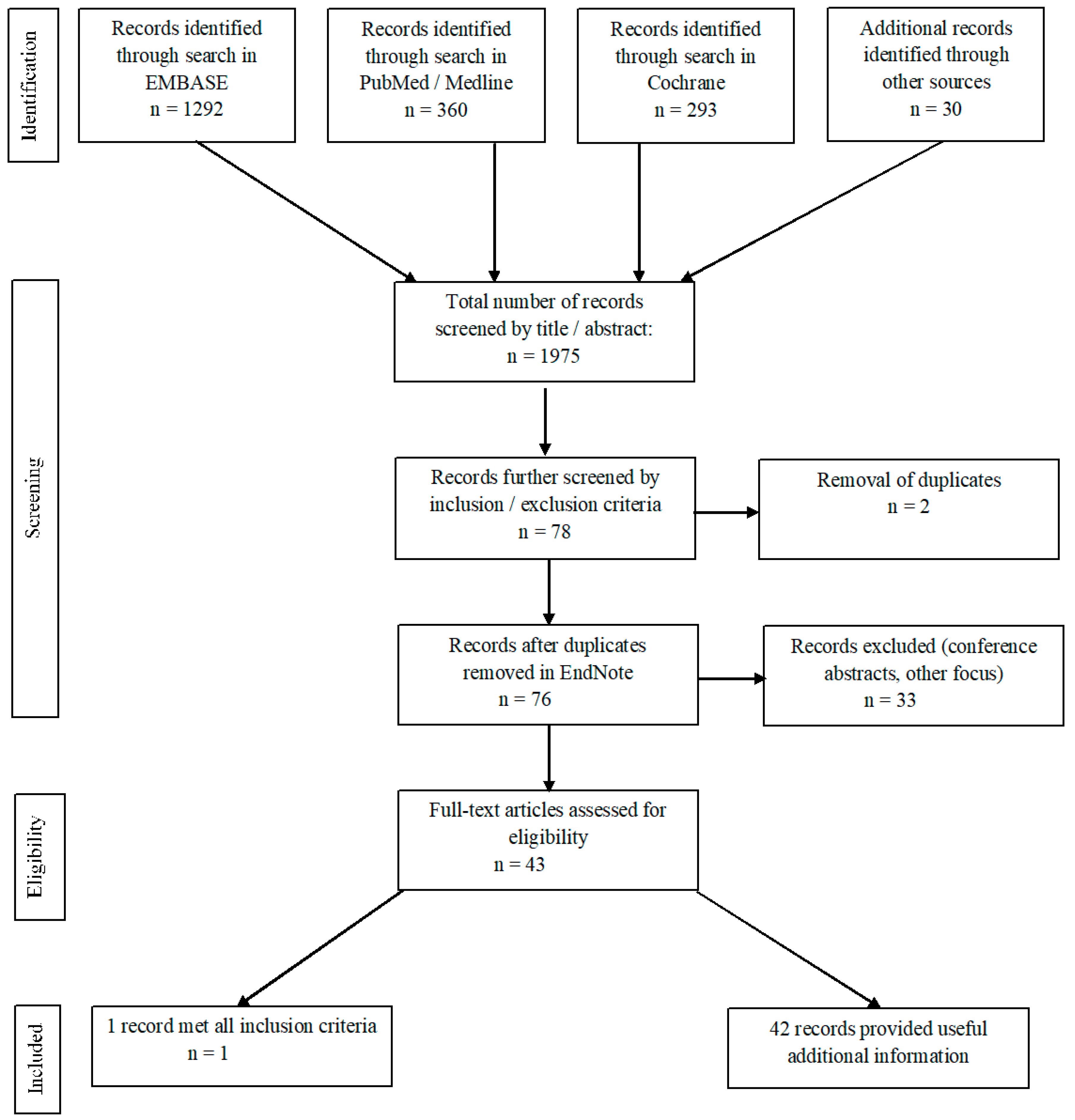
3.1. Systematic Literature Search
3.2. Selection of Literature
3.3. Quality Assessment
3.4. Summary Results: Main Article
4. Discussion
4.1. The RECIST and PERCIST Approaches
4.2. Patient Studies in MBC: Shortcomings of RECIST
4.3. Patient Studies in MBC: PERCIST May Be An Improvement over RECIST
4.4. Strengths and Limitations
4.5. Perspectives
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef]
- Brewster, A.M.; Hortobagyi, G.N.; Broglio, K.R.; Kau, S.W.; Santa-Maria, C.A.; Arun, B.; Buzdar, A.U.; Booser, D.J.; Valero, V.; Bondy, M.; et al. Residual risk of breast cancer recurrence 5 years after adjuvant therapy. J. Natl. Cancer Inst. 2008, 100, 1179–1183. [Google Scholar] [CrossRef] [PubMed]
- Chia, S.K.; Speers, C.H.; D’Yachkova, Y.; Kang, A.; Malfair-Taylor, S.; Barnett, J.; Coldman, A.; Gelmon, K.A.; O’Reilly S, E.; Olivotto, I.A. The impact of new chemotherapeutic and hormone agents on survival in a population-based cohort of women with metastatic breast cancer. Cancer 2007, 110, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Gennari, A.; Conte, P.; Rosso, R.; Orlandini, C.; Bruzzi, P. Survival of metastatic breast carcinoma patients over a 20-year period: A retrospective analysis based on individual patient data from six consecutive studies. Cancer 2005, 104, 1742–1750. [Google Scholar] [CrossRef] [PubMed]
- Bensch, F.; van Kruchten, M.; Lamberts, L.E.; Schroder, C.P.; Hospers, G.A.; Brouwers, A.H.; van Vugt, M.A.; de Vries, E.G. Molecular imaging for monitoring treatment response in breast cancer patients. Eur. J. Pharmacol. 2013, 717, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Danish Breast Cancer Cooperative Group. Retningslinjer Kap18 Behandling af Cancer Mammae med Fjernmetastaser -rev 25-07-2016. Available online: http://www.dbcg.dk/PDF%20Filer/Kap_18_Behandling_af_cancer_mammae_med_fjernmetastaser_Tyge_rev_25.07.2016.pdf (accessed on 17 December 2018).
- Cardoso, F.; Costa, A.; Senkus, E.; Aapro, M.; Andre, F.; Barrios, C.H.; Bergh, J.; Bhattacharyya, G.; Biganzoli, L.; Cardoso, M.J.; et al. 3rd ESO-ESMO International Consensus Guidelines for Advanced Breast Cancer (ABC 3). Ann. Oncol. 2017, 28, 3111. [Google Scholar] [CrossRef] [PubMed]
- Bruzzi, P.; Del Mastro, L.; Sormani, M.P.; Bastholt, L.; Danova, M.; Focan, C.; Fountzilas, G.; Paul, J.; Rosso, R.; Venturini, M. Objective response to chemotherapy as a potential surrogate end point of survival in metastatic breast cancer patients. J. Clin. Oncol. 2005, 23, 5117–5125. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Cai, C.; Charnsangavej, C.; Theriault, R.L.; Green, M.; Quraishi, M.A.; Yang, W.T. Contrast-enhanced computed tomography evaluation of hepatic metastases in breast cancer patients before and after cytotoxic chemotherapy or targeted therapy. Can. Assoc. Radiol. J. 2015, 66, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Seyal, A.R.; Parekh, K.; Velichko, Y.S.; Salem, R.; Yaghmai, V. Tumor growth kinetics versus RECIST to assess response to locoregional therapy in breast cancer liver metastases. Acad. Radiol. 2014, 21, 950–957. [Google Scholar] [CrossRef]
- Mandrekar, S.J.; An, M.W.; Meyers, J.; Grothey, A.; Bogaerts, J.; Sargent, D.J. Evaluation of alternate categorical tumor metrics and cut points for response categorization using the RECIST 1.1 data warehouse. J. Clin. Oncol. 2014, 32, 841–850. [Google Scholar] [CrossRef]
- Willemsen, A.E.C.A.B.; Vlenterie, M.; Van Herpen, C.M.L.; Van Erp, N.P.; Van Der Graaf, W.T.A.; De Geus-Oei, L.F.; Oyen, W.J.G. Positron emission tomography response criteria in solid tumours criteria for quantitative analysis of [18F]-fluorodeoxyglucose positron emission tomography with integrated computed tomography for treatment response assessment in metastasised solid tumours: All that glitters is not gold. Eur. J. Cancer 2016, 56, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Wahl, R.L.; Jacene, H.; Kasamon, Y.; Lodge, M.A. From RECIST to PERCIST: Evolving Considerations for PET response criteria in solid tumors. J. Nucl. Med. 2009, 50, 122s–150s. [Google Scholar] [CrossRef] [PubMed]
- Joo Hyun, O.; Lodge, M.A.; Wahl, R.L. Practical PERCIST: A Simplified Guide to PET Response Criteria in Solid Tumors 1.0. Radiology 2016, 280, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- QUADAS-2 Steering Group. QUADAS-2. Available online: http://www.bristol.ac.uk/population-health-sciences/projects/quadas/quadas-2/ (accessed on 5 December 2018).
- Riedl, C.C.; Pinker, K.; Ulaner, G.A.; Ong, L.T.; Baltzer, P.; Jochelson, M.S.; McArthur, H.L.; Gonen, M.; Dickler, M.; Weber, W.A. Comparison of FDG-PET/CT and contrast-enhanced CT for monitoring therapy response in patients with metastatic breast cancer. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1428–1437. [Google Scholar] [CrossRef]
- Siepe, B.; Hoilund-Carlsen, P.F.; Gerke, O.; Weber, W.A.; Motschall, E.; Vach, W. The move from accuracy studies to randomized trials in PET: Current status and future directions. J. Nucl. Med. 2014, 55, 1228–1234. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chen, S.; Ibrahim, N.K.; Yan, Y.; Wong, S.T.; Wang, H.; Wong, F.C. Risk stratification in patients with advanced-stage breast cancer by pretreatment [(18)F]FDG PET/CT. Cancer 2015, 121, 3965–3974. [Google Scholar] [CrossRef] [PubMed]
- Gerke, O.; Hermansson, R.; Hess, S.; Schifter, S.; Vach, W.; Hoilund-Carlsen, P.F. Cost-effectiveness of PET and PET/computed tomography: A systematic review. Pet. Clin. 2015, 10, 105–124. [Google Scholar] [CrossRef]
- Gunalp, B.; Oner, A.O.; Ince, S.; Alagoz, E.; Ayan, A.; Arslan, N. Evaluation of radiographic and metabolic changes in bone metastases in response to systemic therapy with (18)FDG-PET/CT. Radiol. Oncol. 2015, 49, 115–120. [Google Scholar] [CrossRef]
- Cook, G.J.; Azad, G.K.; Goh, V. Imaging Bone Metastases in Breast Cancer: Staging and Response Assessment. J. Nucl. Med. 2016, 57, 27s–33s. [Google Scholar] [CrossRef] [PubMed]
- Ulaner, G.A.; Eaton, A.; Morris, P.G.; Lilienstein, J.; Jhaveri, K.; Patil, S.; Fazio, M.; Larson, S.; Hudis, C.A.; Jochelson, M.S. Prognostic value of quantitative fluorodeoxyglucose measurements in newly diagnosed metastatic breast cancer. Cancer Med. 2013, 2, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Jia, Z.; Ragaz, J.; Zhang, Y.J.; Zhou, M.; Zhang, Y.P.; Li, G.; Wang, B.Y.; Wang, Z.H.; Hu, X.C. The maximum standardized uptake value of 18 F-FDG PET scan to determine prognosis of hormone-receptor positive metastatic breast cancer. BMC Cancer 2013, 13, 42. [Google Scholar] [CrossRef] [PubMed]
- Morris, P.G.; Ulaner, G.A.; Eaton, A.; Fazio, M.; Jhaveri, K.; Patil, S.; Evangelista, L.; Park, J.Y.; Serna-Tamayo, C.; Howard, J.; et al. Standardized uptake value by positron emission tomography/computed tomography as a prognostic variable in metastatic breast cancer. Cancer 2012, 118, 5454–5462. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi-Jehanno, N.; Giraudet, A.L.; Champion, L.; Lerebours, F.; Le Stanc, E.; Edeline, V.; Madar, O.; Bellet, D.; Pecking, A.P.; Alberini, J.L. Assessment of response to endocrine therapy using FDG PET/CT in metastatic breast cancer: A pilot study. Eur. J. Nucl. Med. Mol. Imaging 2012, 39, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Fojo, A.T.; Noonan, A.; Sharma, M.R.; Maitland, M.L.; Ratain, M.J. Why RECIST works and why it should stay—Counterpoint. Cancer Res. 2012, 72, 5151–5158. [Google Scholar] [CrossRef] [PubMed]
- Pinker, K.; Riedl, C.; Weber, W.A. Evaluating tumor response with FDG PET: Updates on PERCIST, comparison with EORTC criteria and clues to future developments. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 55–66. [Google Scholar] [CrossRef]
- Hildebrandt, M.G.; Gerke, O.; Baun, C.; Falch, K.; Hansen, J.A.; Farahani, Z.A.; Petersen, H.; Larsen, L.B.; Duvnjak, S.; Buskevica, I.; et al. [18F]Fluorodeoxyglucose (FDG)-Positron Emission Tomography (PET)/Computed Tomography (CT) in Suspected Recurrent Breast Cancer: A Prospective Comparative Study of Dual-Time-Point FDG-PET/CT, Contrast-Enhanced CT, and Bone Scintigraphy. J. Clin. Oncol. 2016, 34, 1889–1897. [Google Scholar] [CrossRef]
- Hoilund-Carlsen, P.F.; Gerke, O.; Vach, W. Demonstrating the benefits of clinical nuclear imaging: Is it time to add economic analysis? Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 1720–1722. [Google Scholar] [CrossRef]
- Goulon, D.; Necib, H.; Henaff, B.; Rousseau, C.; Carlier, T.; Kraeber-Bodere, F. Quantitative Evaluation of Therapeutic Response by FDG-PET-CT in Metastatic Breast Cancer. Front. Med. 2016, 3, 19. [Google Scholar] [CrossRef]
- Hoilund-Carlsen, P.F.; Hess, S.; Werner, T.J.; Alavi, A. Cancer metastasizes to the bone marrow and not to the bone: Time for a paradigm shift! Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 893–897. [Google Scholar] [CrossRef]
- Hayashi, N.; Costelloe, C.M.; Hamaoka, T.; Wei, C.; Niikura, N.; Theriault, R.L.; Hortobagyi, G.N.; Madewell, J.E.; Ueno, N.T. A prospective study of bone tumor response assessment in metastatic breast cancer. Clin. Breast Cancer 2013, 13, 24–30. [Google Scholar] [CrossRef]
- Al-Muqbel, K.M.; Yaghan, R.J. Effectiveness of 18F-FDG-PET/CT vs. bone scintigraphy in treatment response assessment of bone metastases in breast cancer. Medicine 2016, 95, e3753. [Google Scholar] [CrossRef]
- Coleman, R.; Body, J.J.; Aapro, M.; Hadji, P.; Herrstedt, J.; Group, E.G.W. Bone health in cancer patients: ESMO Clinical Practice Guidelines. Ann. Oncol. 2014, 25, iii124–iii137. [Google Scholar] [CrossRef]
- Catalano, O.A.; Nicolai, E.; Rosen, B.R.; Luongo, A.; Catalano, M.; Iannace, C.; Guimaraes, A.; Vangel, M.G.; Mahmood, U.; Soricelli, A.; et al. Comparison of CE-FDG-PET/CT with CE-FDG-PET/MR in the evaluation of osseous metastases in breast cancer patients. Br. J. Cancer 2015, 112, 1452–1460. [Google Scholar] [CrossRef]
- Haug, A.R.; Tiega Donfack, B.P.; Trumm, C.; Zech, C.J.; Michl, M.; Laubender, R.P.; Uebleis, C.; Bartenstein, P.; Heinemann, V.; Hacker, M. 18F-FDG PET/CT predicts survival after radioembolization of hepatic metastases from breast cancer. J. Nucl. Med. 2012, 53, 371–377. [Google Scholar] [CrossRef]
- Pinker, K.; Riedl, C.C.; Ong, L.; Jochelson, M.; Ulaner, G.A.; McArthur, H.; Dickler, M.; Gonen, M.; Weber, W.A. The Impact That Number of Analyzed Metastatic Breast Cancer Lesions Has on Response Assessment by 18F-FDG PET/CT Using PERCIST. J. Nucl. Med. 2016, 57, 1102–1104. [Google Scholar] [CrossRef][Green Version]
- Lin, N.U.; Guo, H.; Yap, J.T.; Mayer, I.A.; Falkson, C.I.; Hobday, T.J.; Dees, E.C.; Richardson, A.L.; Nanda, R.; Rimawi, M.F.; et al. Phase II Study of Lapatinib in Combination With Trastuzumab in Patients With Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer: Clinical Outcomes and Predictive Value of Early [18F]Fluorodeoxyglucose Positron Emission Tomography Imaging (TBCRC 003). J. Clin. Oncol. 2015, 33, 2623–2631. [Google Scholar] [CrossRef]
- Raphael, J.; Verma, S. Overall survival (OS) endpoint: An incomplete evaluation of metastatic breast cancer (MBC) treatment outcome. Breast Cancer Res. Treat. 2015, 150, 473–478. [Google Scholar] [CrossRef]
- Pantaleo, M.A.; Nannini, M.; Maleddu, A.; Fanti, S.; Ambrosini, V.; Nanni, C.; Boschi, S.; Biasco, G. Conventional and novel PET tracers for imaging in oncology in the era of molecular therapy. Cancer Treat. Rev. 2008, 34, 103–121. [Google Scholar] [CrossRef]
- Marcus, C.D.; Ladam-Marcus, V.; Cucu, C.; Bouche, O.; Lucas, L.; Hoeffel, C. Imaging techniques to evaluate the response to treatment in oncology: Current standards and perspectives. Crit. Rev. Oncol. Hematol. 2009, 72, 217–238. [Google Scholar] [CrossRef]
- Gong, C.; Yang, Z.; Sun, Y.; Zhang, J.; Zheng, C.; Wang, L.; Zhang, Y.; Xue, J.; Yao, Z.; Pan, H.; et al. A preliminary study of 18F-FES PET/CT in predicting metastatic breast cancer in patients receiving docetaxel or fulvestrant with docetaxel. Sci. Rep. 2017, 7, 6584. [Google Scholar] [CrossRef]
- Gebhart, G.; Lamberts, L.E.; Wimana, Z.; Garcia, C.; Emonts, P.; Ameye, L.; Stroobants, S.; Huizing, M.; Aftimos, P.; Tol, J.; et al. Molecular imaging as a tool to investigate heterogeneity of advanced HER2-positive breast cancer and to predict patient outcome under trastuzumab emtansine (T-DM1): The ZEPHIR trial. Ann. Oncol. 2016, 27, 619–624. [Google Scholar] [CrossRef]
- Ulaner, G.A.; Hyman, D.M.; Lyashchenko, S.K.; Lewis, J.S.; Carrasquillo, J.A. 89Zr-Trastuzumab PET/CT for Detection of Human Epidermal Growth Factor Receptor 2-Positive Metastases in Patients with Human Epidermal Growth Factor Receptor 2-Negative Primary Breast Cancer. Clin. Nucl. Med. 2017, 42, 912–917. [Google Scholar] [CrossRef]
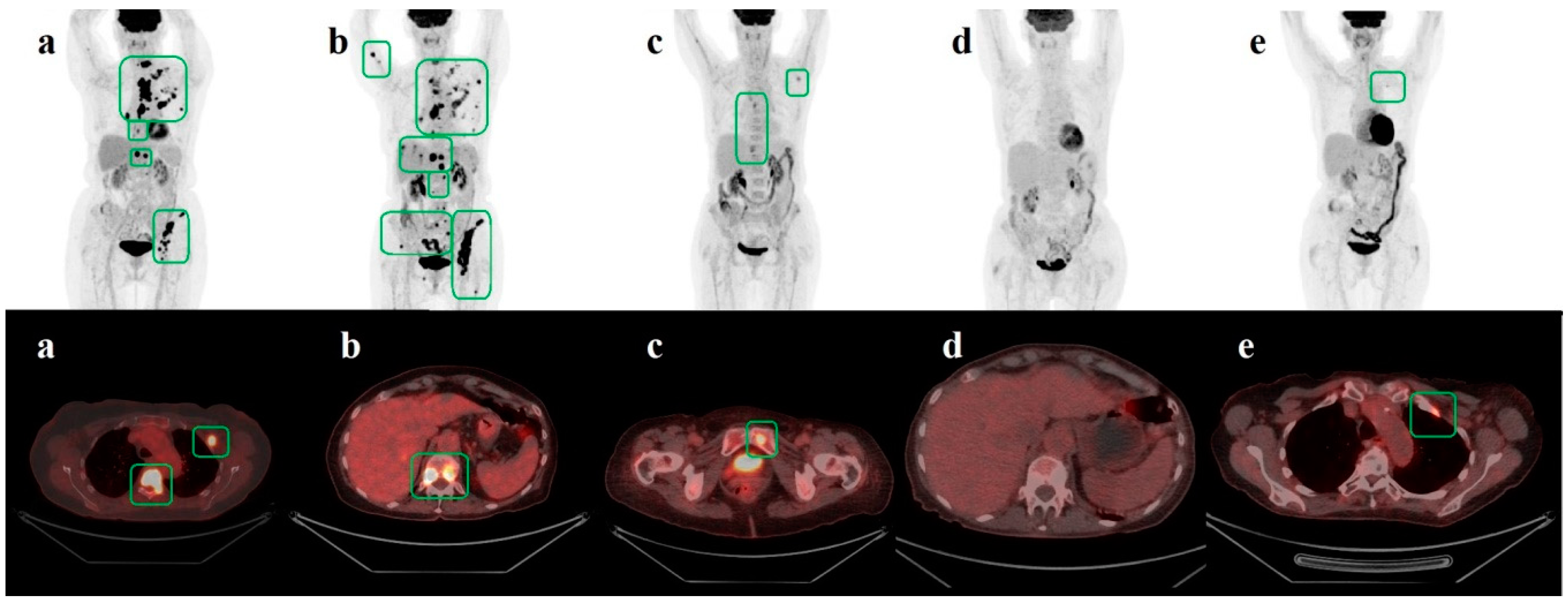
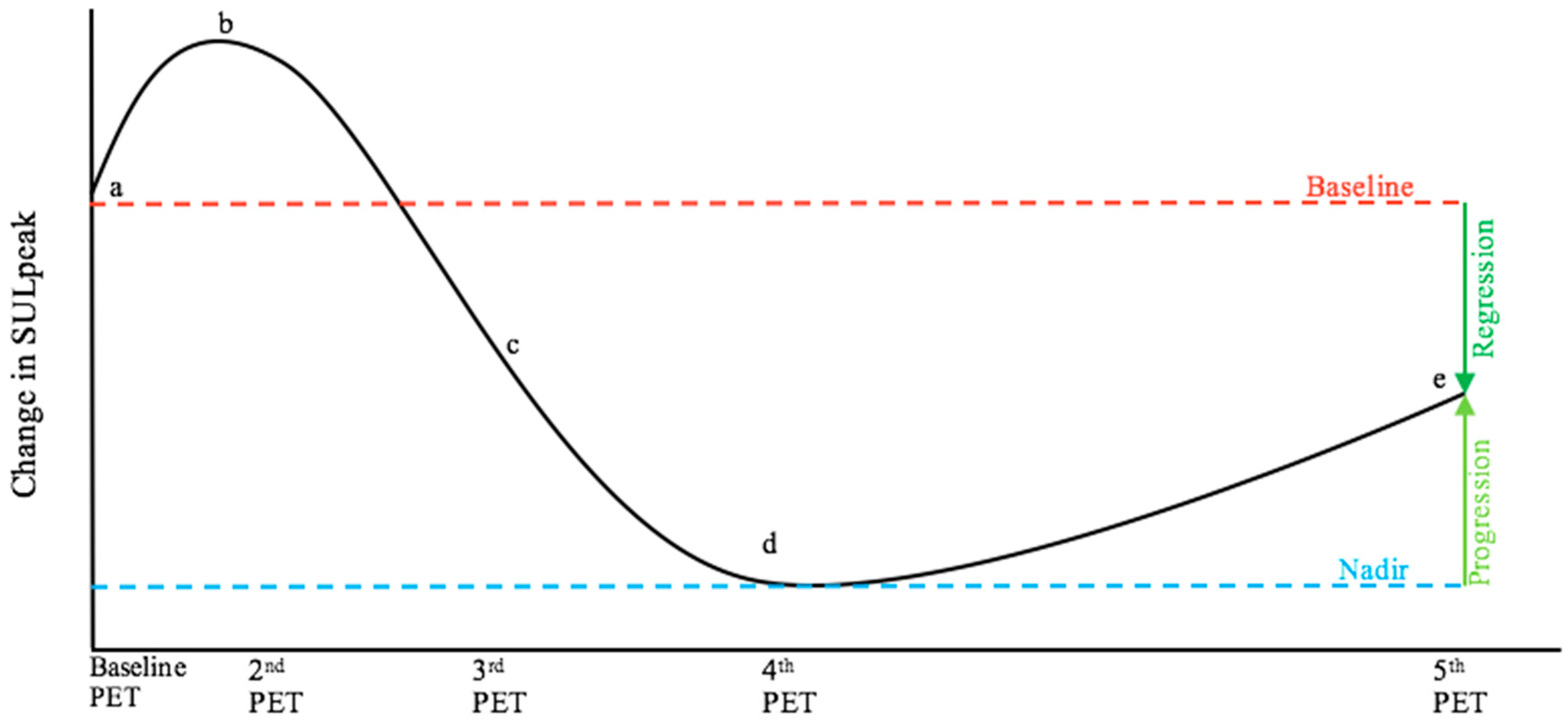
| Article title | Comparison of FDG-PET/CT and contrast-enhanced CT for monitoring therapy response in patients with metastatic breast cancer |
| First author | Christopher C. Riedl |
| Year of publication | 2017 |
| Study design | Retrospective cohort |
| Objectives | To compare FDG-PET/CT (PERCIST) and CE-CT (RECIST) for prediction of progression-free survival (PFS) and disease specific survival (DSS) in patients with stage IV breast cancer undergoing systemic therapy |
| Number of patients | 65 (aged 29–85 years, mean age 54 years) |
| Hormone/receptor status | ER+, HER2+ (n = 10) ER+, HER2- (n = 39) ER-, HER2+ (n = 5) ER-, HER2- (n = 11) |
| Histology | Invasive ductal (n = 54) Mixed ductal and lobular (n = 4) Invasive lobular (n = 7) |
| Metastatic location | Osseous + other metastases (n = 30) Only osseous metastases (n = 12) Only non-osseous metastases (n = 23) |
| Treatment at intervention point | Cytotoxic (n = 15) Immunotherapy + cytotoxic (n = 19) Immunotherapy (n = 14) Immunotherapy + hormone therapy (n = 13) Hormone therapy (anti-androgen) (n = 4) |
| Number and timing of scans | Baseline CE-CT and FDG-PET/CT within the 28 days prior to starting therapy CE-CT and FDG-PET/CT within the 90 days after start of therapy Follow-up with CE-CT every third month until progression, followed by routine follow-up until death |
| Response criteria used | CE-CT: response determined using RECIST 1.1 FDG-PET/CT: response determined using peak standardized uptake values normalized to lean body mass (SULpeak) and, in a separate analysis, by maximum standardized uptake (SUVmax). SULpeak and SUVmax were compared, and response categorization was based on PERCIST |
| Endpoint | Progression-free survival (PFS) Disease specific survival (DSS) |
| Criteria | RECIST 1.1 [16] | PERCIST 1.0 [14,15] |
|---|---|---|
| Standardization requirements | Anatomical coverage should at least include thorax, abdomen, and pelvis with a maximum slice thickness of 5 mm. Intra-patient (but not necessarily inter-patient) uniformity of contrast-administration should be sought, depending on patient needs, and available equipment. | Liver SUL must be within 20% range of baseline on follow-up scan. If liver is abnormal on follow-up, blood-pool SUL must be within 20% of baseline scan. Uptake time cannot diverge more than 15 min and must be started at least 50 min after injection. The same scanner or scanner model should be used for the same site. The injected dose, blood sugar, acquisition protocol, and software for reconstruction should be uniform. Scanners should produce reproducible data and be properly calibrated. |
| Target lesions | Unidimensional longest diameter of tumor lesions is used. A minimum size of 10 mm is required at baseline. | Standardized uptake value corrected for lean body mass in the hottest single tumor lesion of a 1 mL spherical VOI (SULpeak) is used. A tumor lesion volume of minimum 1 mL is required at baseline. SULpeak must be at least 1.5 times + 2SD SULmean of a 3 cm diameter VOI of healthy liver at baseline. If the liver is abnormal, then background emission should be measured in a cylindrical VOI with 1 cm in diameter of blood-pool in the descending thoracic aorta, excluding the aortic wall. |
| Measurable node definition | A minimum size of 15 mm in short axis of lymph nodes is required. | No definition listed in the PERCIST criteria. |
| Requirements for measurable disease at baseline | Sum of longest target lesion diameters, short axis of nodes. Up to 5 measurable target lesions (maximum 2 per organ). Other lesions are mentioned non-target lesions. | SULpeak in the hottest one lesion or sum of SULpeak in up to 5 measurable target lesions (maximum 2 per organ). |
| Requirements for measurable disease at follow-up | Sum of the same target lesion diameters as determined on the baseline scan. | SULpeak in the single hottest lesion (not necessarily the same) or sum of SULpeak in up to 5 measurable target lesions (max. 2 per organ). |
| Follow-up measurements | Sum of longest diameters at baseline is used for comparison when assessing response. The smallest recorded sum lesion diameter (nadir) is used to assess progression. | Response evaluation is always compared to the baseline scan. |
| CR/CMR | Disappearance of all target lesions. Reduction in short axis of target lymph nodes to <10 mm. | Disappearance of all lesions on PET images, lesions are indistinguishable from background and less than SULmean of liver regardless of %-change from baseline and anatomical size. |
| PR/PMR | ≥30% decrease in sum of target lesion diameter sum. | ≥30% decrease in (sum of) target lesion(s) SUL and 1 SUL unit absolute change. |
| PD/PMR | ≥20% increase in sum of target lesion diameter and minimum 5 mm total increase, or new lesion, or unequivocal progression of non-target lesions. | ≥30% increase in sum of target lesion SUL and 1 SUL unit absolute change, or new FDG avid lesion, or unequivocal progression of non-target lesion (e.g., ≥30% increase), or unequivocal progression by RECIST. |
| SD/SMD | Does not meet other criteria. | Does not meet other criteria. |
| Advantages | Disadvantages | |
|---|---|---|
| RECIST | Well-defined, well-documented criteria [28] Less patient preparation needed Scan is less expensive than FDG-PET/CT High global availability Time-efficient High degree of repeatability | Difficulties in distinguishing viable from non-viable residual tumor tissue [18] Osseous metastases are considered non-measurable [18] Lack of concordance between response evaluation and time-to-event outcome (e.g., PFS, OS, DSS) [29] Difficulties in distinguishing PD and SD [11] Slightly higher inter-observer variability [18] |
| PERCIST | High degree of repeatabilityLess inter-observer variability [18] Differentiates active tumor from post-therapeutic changes [18] Able to assess metabolic activity in osseous metastases [30] Detects response/progression earlier Classifies patients with SD and PD on an anatomical scan more accurately [29] Good correlation to time-to-event measures [18] | Complex analysis due to multitude of data [12] Technical challenges, e.g., partial volume effects, physiological variations, acquisition errors, suboptimal signal-to-noise ratio [12] Various scanners, software, criteria not yet standardized for all solid tumors or subgroups [12] Time-consuming, requires highly trained personnel Not yet globally available |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Helland, F.; Hallin Henriksen, M.; Gerke, O.; Vogsen, M.; Høilund-Carlsen, P.F.; Hildebrandt, M.G. FDG-PET/CT Versus Contrast-Enhanced CT for Response Evaluation in Metastatic Breast Cancer: A Systematic Review. Diagnostics 2019, 9, 106. https://doi.org/10.3390/diagnostics9030106
Helland F, Hallin Henriksen M, Gerke O, Vogsen M, Høilund-Carlsen PF, Hildebrandt MG. FDG-PET/CT Versus Contrast-Enhanced CT for Response Evaluation in Metastatic Breast Cancer: A Systematic Review. Diagnostics. 2019; 9(3):106. https://doi.org/10.3390/diagnostics9030106
Chicago/Turabian StyleHelland, Fredrik, Martine Hallin Henriksen, Oke Gerke, Marianne Vogsen, Poul Flemming Høilund-Carlsen, and Malene Grubbe Hildebrandt. 2019. "FDG-PET/CT Versus Contrast-Enhanced CT for Response Evaluation in Metastatic Breast Cancer: A Systematic Review" Diagnostics 9, no. 3: 106. https://doi.org/10.3390/diagnostics9030106
APA StyleHelland, F., Hallin Henriksen, M., Gerke, O., Vogsen, M., Høilund-Carlsen, P. F., & Hildebrandt, M. G. (2019). FDG-PET/CT Versus Contrast-Enhanced CT for Response Evaluation in Metastatic Breast Cancer: A Systematic Review. Diagnostics, 9(3), 106. https://doi.org/10.3390/diagnostics9030106







