A Brief Overview of Medical Fiber Optic Biosensors and Techniques in the Modification for Enhanced Sensing Ability
Abstract
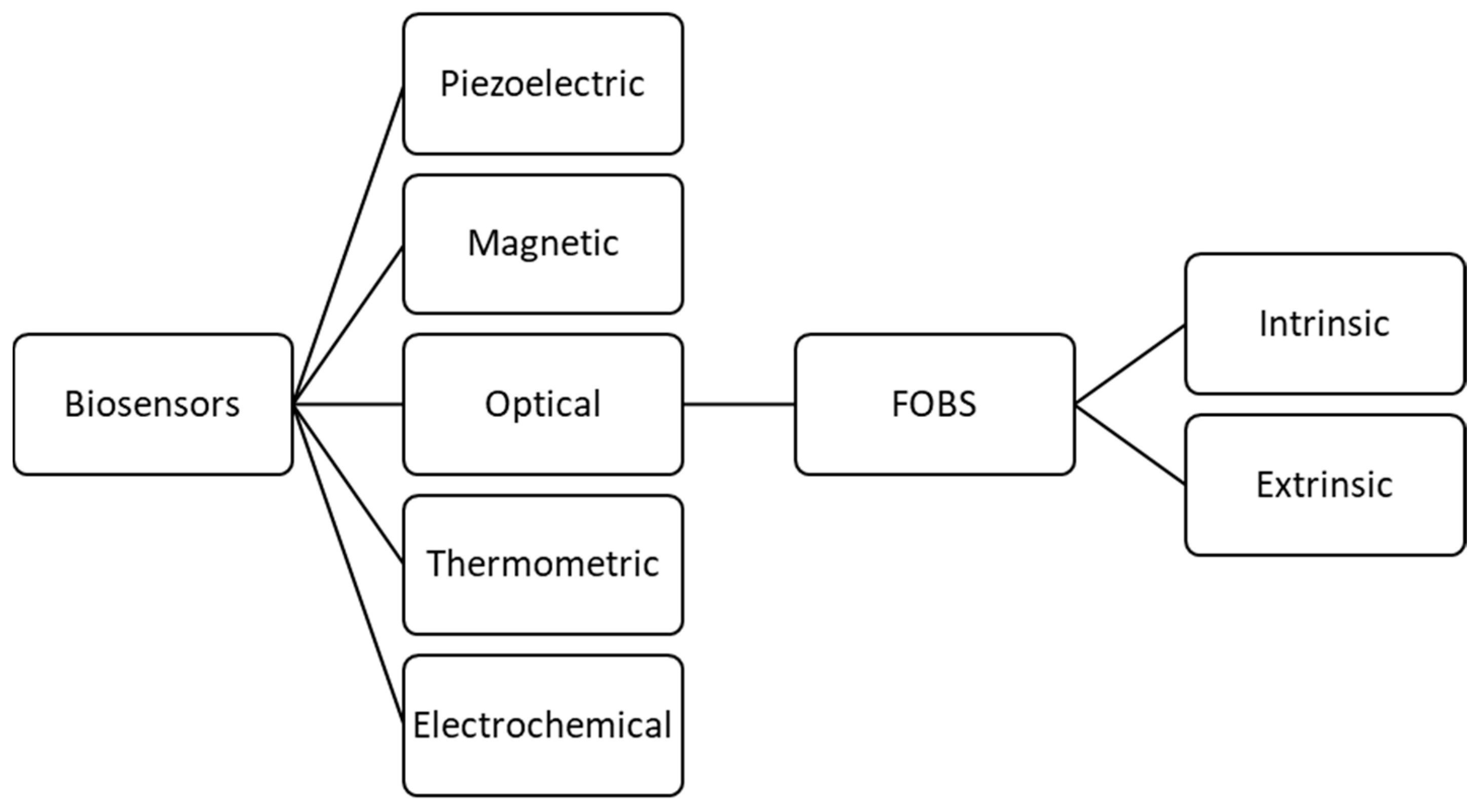
1. Introduction
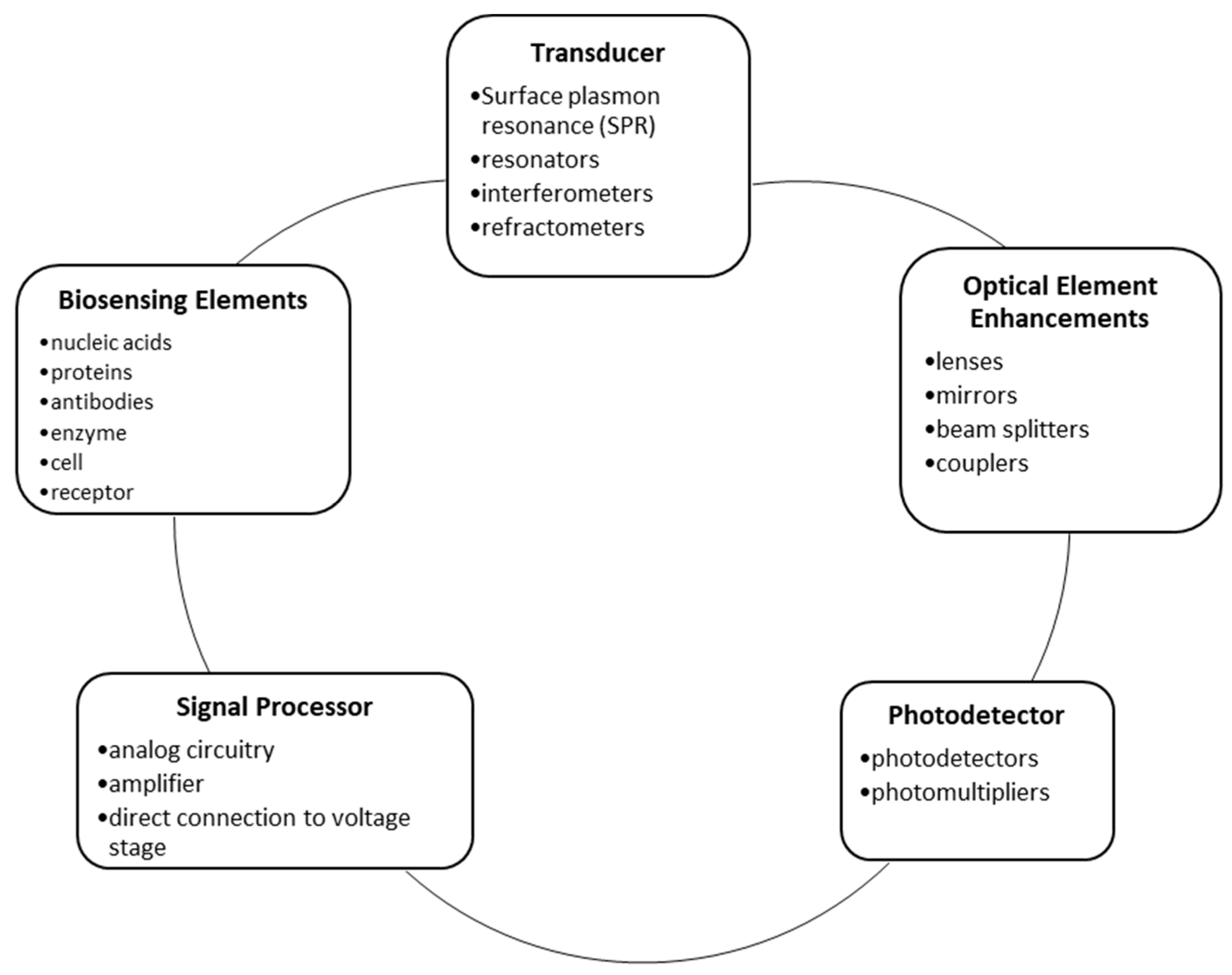
2. General Components and Modifications of FOBS
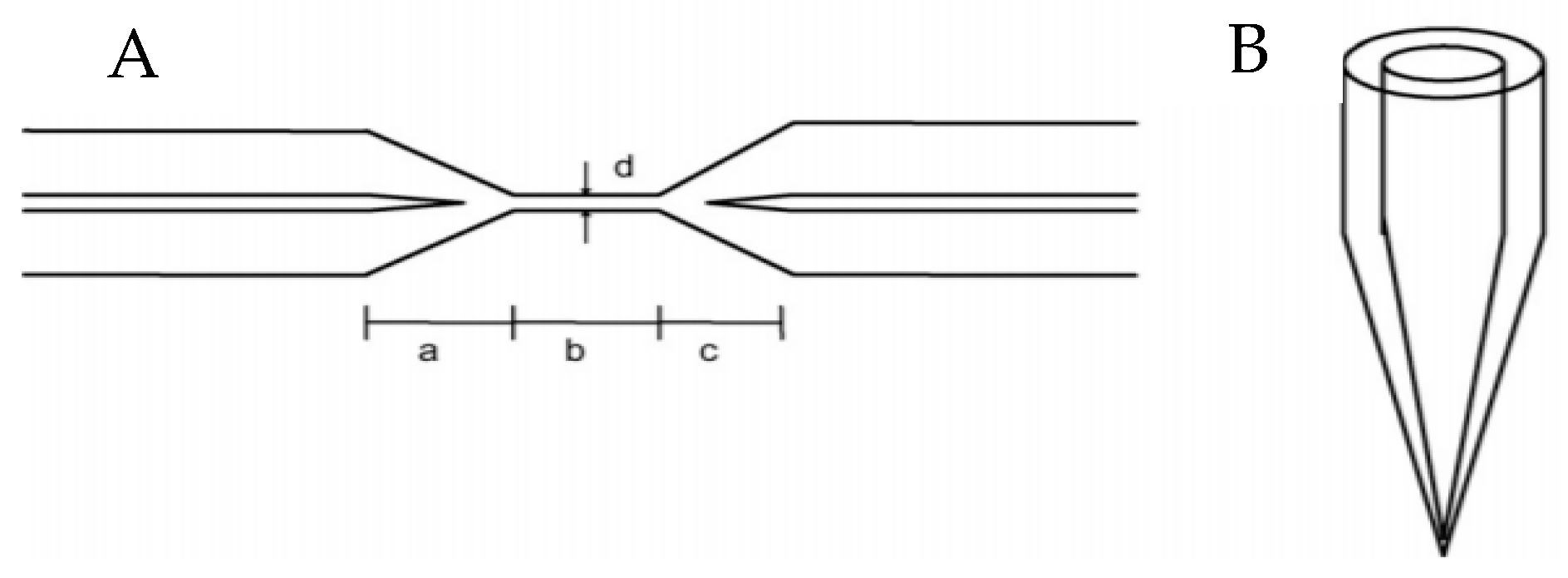
2.1. Fiber Optic Cables and Other Optical Elements
2.2. Biorecognition Elements
2.2.1. pH Biorecognition Elements
2.2.2. Enzymes as Biorecognition Elements
2.2.3. Immunosensors
2.2.4. Whole-Cell Biorecognition Elements
2.3. Transducers and Sensing Strategies
2.3.1. Surface Plasmon Resonance
2.3.2. Interferometers
2.4. Photodetectors and related Biosensing Strategies
2.4.1. Spectroscopy
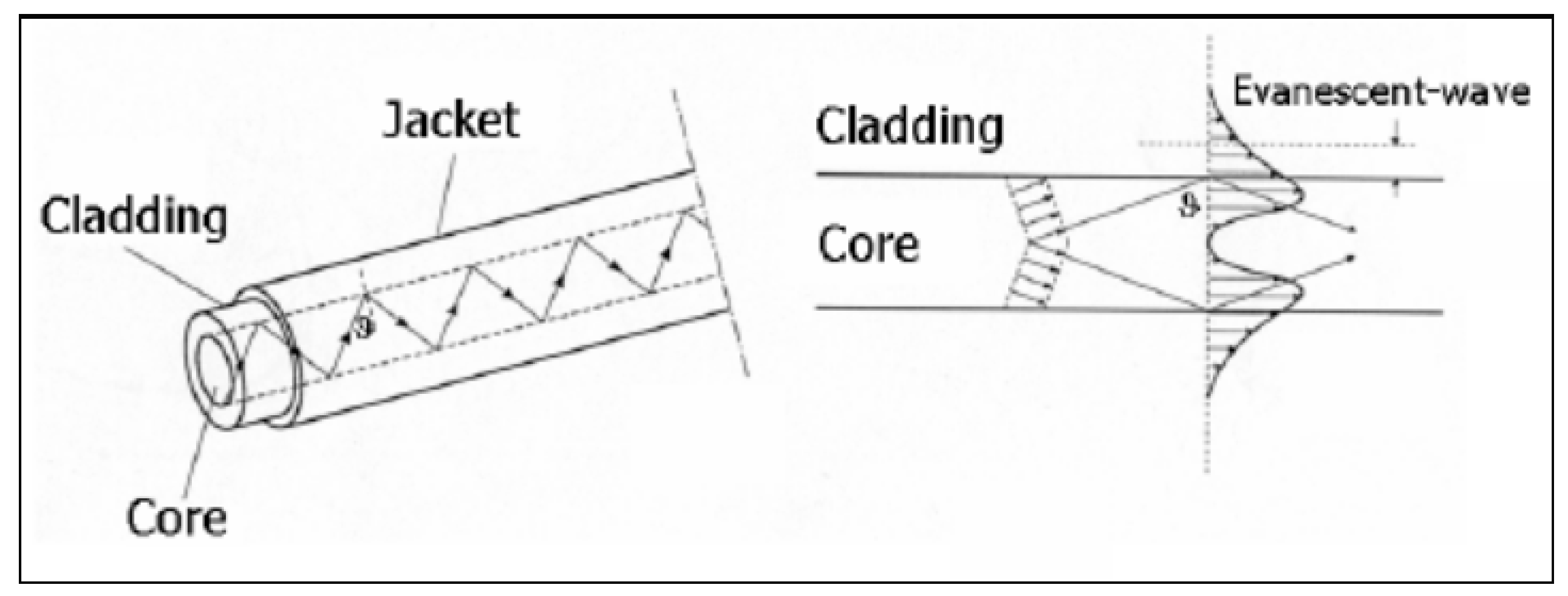
2.4.2. Evanescent Field Interaction
2.4.3. Photodetectors
2.5. Signal Processors
3. Discussion
4. Future Developments and Industry Trends
Funding
Conflicts of Interest
References
- Global Market Insights, Inc. Biosensors Market Revenue will Surpass $29 billion by 2024: Global Market Insights, Inc. 28 June 2018. Available online: https://globenewswire.com/news-release/2018/06/28/1530782/0/en/Biosensors-Market-revenue-will-surpass-29-billion-by-2024-Global-Market-Insights-Inc.html (accessed on 24 November 2018).
- Centers for Disease Control and Prevention. The Power of Prevention Chronic Disease … the Public Health Challenge of the 21st Century. 2009. Available online: https://www.cdc.gov/chronicdisease/pdf/2009-Power-of-Prevention.pdf (accessed on 9 September 2018).
- DeVol, R.; Bedroussian, A. An Unhealthy America: The Economic Burden of Chronic Disease. Milken Institute. 2007. Available online: http://www.milkeninstitute.org/pdf/ES_ResearchFindings.pdf (accessed on 9 September 2018).
- Laser Focus World. Medical Applications of Fiber Optics: Optical Fiber Sees Growth as Medical Sensors. 1 January 2011. Available online: https://www.laserfocusworld.com/articles/2011/01/medical-applications-of-fiber-optics-optical-fiber-sees-growth-as-medical-sensors.html (accessed on 9 September 2018).
- Heijmans, J.; Cheng, L.; Wieringa, F. Optical fiber sensors for medical applications—Practical engineering considerations. In Proceedings of the 4th European Conference of the International Federation for Medical and Biological Engineering, Antwerp, Belgium, 23–27 November 2008; IFMBE Proceedings. Springer: Berlin/Heidelberg, Germany, 2008; Volume 22, pp. 2330–2334. [Google Scholar]
- Shaikh, M.; Patil, M.A. Analysis, Designing and Working Principal of Optical Fiber (OF) Biosensors. Int. J. Eng. Sci. 2012, 1, 52–57. Available online: https://pdfs.semanticscholar.org/6be4/259027827f649fff35ca9b5e4fdb105ed812.pdf (accessed on 13 September 2018).
- Turner, A.P. Biosensors: Sense and Sensitivity. Science 2000, 290, 1315–1317. Available online: https://0-www-jstor-org.libcat.widener.edu/stable/pdf/3078232.pdf?refreqid=excelsior:44250d6cef04e185434b36ec3e878f96 (accessed on 13 September 2018). [CrossRef] [PubMed]
- Martin, F.J.F.; Rodriguez, J.C.C.; Anton, J.C.A.; Perez, J.C.V.; Sanchez Barragan, I.; Costa-Fernandez, J.M.; Sanz-Medel, A. Design of a low-cost optical instrument for pH fluorescence measurements. IEEE Trans. Instrum. Meas. 2006, 55, 1215–1221. [Google Scholar]
- Leung, A.; Mohana Shankar, P.; Mutharasan, R. A review of fiber-optic biosensors. Sens. Actuators B 2007, 125, 688–703. [Google Scholar] [CrossRef]
- Yunianto, M.; Permata, A.N.; Eka, D.; Ariningrum, D.; Wahyuningsih, S.; Marzuki, A. Design of a Fiber Optic Biosensor for Cholesterol Detection in Human Blood. IOP Conf. Ser. Mater. Sci. Eng. 2017, 176, 012014. [Google Scholar] [CrossRef]
- Latifi, H.; Zibaii, M.; Hosseini, S.M.; Jorge, P. Nonadiabatic tapered optical fiber for biosensor applications. Photonic Sens. 2012, 2, 340–356. [Google Scholar] [CrossRef]
- Ferreira, A.P.; Werneck, M.M.; Ribeiro, R.M. Development of an evanescent-field fibre optic sensor for Escherichia coli O157: H7. Biosens. Bioelectron. 2001, 16, 399–408. [Google Scholar] [CrossRef]
- Haddock, H.S.; Shankar, P.M.; Mutharasan, R. Evanescent sensing of biomolecules and cells. Sens. Actuators B Chem. 2003, 88, 67–74. [Google Scholar] [CrossRef]
- Pospíšilová, M.; Kuncová, G.; Trögl, J. Fiber-Optic Chemical Sensors and Fiber-Optic Bio-Sensors. Sensors 2015, 15, 25208–25259. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, S.R.; Peterson, J.I.; Fitzgerald, R.V. A Miniature Fiber Optic pH Sensor for Physiological Use. J. Biomech. Eng. 1980, 102, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Marazuela, M.; Moreno-Bondi, M. Fiber-optic biosensors—An overview. Anal. Bioanal. Chem. 2002, 372, 664–682. [Google Scholar] [CrossRef] [PubMed]
- Rocchitta, G.; Spanu, A.; Babudieri, S.; Latte, G.; Madeddu, G.; Galleri, G.; Nuvoli, S.; Bagella, P.; Demartis, M.I.; Fiore, V.; et al. Enzyme Biosensors for Biomedical Applications: Strategies for Safeguarding Analytical Performances in Biological Fluids. Sensors 2016, 16, 780. [Google Scholar] [CrossRef] [PubMed]
- Wolfbeis, O.S.; Weis, L.J.; Leiner, M.J.P.; Ziegler, W.E. Fiber-optic fluorosensor for oxygen and carbon dioxide. Anal. Chem. 1988, 60, 2028–2030. [Google Scholar] [CrossRef]
- Pasic, A.; Koehler, H.; Kilmant, I.; Schaupp, L. Miniaturized fiber-optic hybrid sensor for continuous glucose monitoring in subcutaneous tissue. Sens. Actuators B Chem. 2007, 122, 60–68. [Google Scholar] [CrossRef]
- Brown, J.Q.; McShane, M.J. Modeling of spherical fluorescent glucose microsensor systems: Design of enzymatic smart tattoos. Biosens. Bioelectron. 2006, 21, 1760–1769. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.B.; Jones, E.R. Enzyme-based fiber optic zinc biosensor. Anal. Chem. 1993, 65, 730–734. [Google Scholar] [CrossRef]
- Ling, L.; Zhang, Z.; Jianzhong, L. A simplified enzyme-based fiber optic sensor for hydrogen peroxide and oxidase substrates. Talanta 1994, 41, 1999–2002. [Google Scholar] [CrossRef]
- Spohn, U.; Preuschoff, F.; Blankenstein, G.; Janasek, D.; Kula, M.R.; Hacker, A. Chemiluminometric enzyme sensors for flow-injection analysis. Anal. Chim. Acta 1995, 303, 109–120. [Google Scholar] [CrossRef]
- Andrade, J.D.; Vanwagenen, R.A.; Gregonis, D.E.; Newby, K.; Lin, J.N. Remote Fiber-optic Biosensors Based on Evanescent-Excited Fluoro-Immunoassay: Concept and Progress. IEEE Trans. Electron Dev. 1985, 32, 1175–1179. [Google Scholar] [CrossRef]
- Spiker, J.; Kyung, K. Preliminary study of real-time fiber optic based protein C biosensor. Biotechnol. Bioeng. 1999, 66, 158–163. [Google Scholar] [CrossRef]
- Close, D.M.; Ripp, S.; Sayler, G.S. Reporter proteins in whole-cell optical bioreporter detection systems, biosensor integrations, and biosensing applications. Sensors 2009, 9, 9147–9174. [Google Scholar] [CrossRef] [PubMed]
- Roda, A.; Cevenini, L.; Michelini, E.; Branchini, B.R. A portable bioluminescence engineered cell-based biosensor for on-site applications. Biosens. Bioelectron. 2011, 26, 3647–3653. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Mai, Z.; Chen, Y.; Wang, J.; Li, L.; Su, Q.; Li, X.; Hong, X. A label-free optic SPR biosensor for specific detection of C-reactive protein. Sci. Rep. 2017, 7, 16904. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; White, I.; Shopova, S.; Zhu, H.; Suter, J.; Sun, Y. Sensitive optical biosensors for unlabeled targets: A review. Anal. Chim. Acta 2008, 620, 8–26. [Google Scholar] [CrossRef] [PubMed]
- Pollet, J.; Delport, F.; Janssen, K.; Jans, K.; Maes, G.; Pfeiffer, H.; Wevers, M.; Lammertyn, J. Fiber optic SPR biosensing of DNA hybridization and DNA-protein interactions. Biosens. Bioelectron. 2009, 4, 864–869. [Google Scholar] [CrossRef] [PubMed]
- Udd, E.; Spillman, W. Fiber Optic Sensors: An Introduction for Engineers and Scientists, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar] [CrossRef]
- Speight, J. Sources and Types of Inorganic Pollutants. In Environmental Inorganic Chemistry for Engineers; Section 5.4.2; Butterworth-Heinemann: Oxford, UK, 2017; pp. 231–282. [Google Scholar]
- Ramakrishna, B.; Sai, V.V.R. Immunogold-silver staining (IGSS) based U-bent fiberoptic sandwich biosensor. In Proceedings of the SPIE BioPhotonics Australasia, Adelaide, Australia, 24 November 2016. 100130U. [Google Scholar] [CrossRef]
- Pospiskova, K.; Safarik, I.; Sebela, M.; Kuncova, G. Magnetic particles-based biosensor for biogenic amines using an optical oxygen sensor as a transducer. Microchim. Acta 2013, 180, 311–318. [Google Scholar] [CrossRef]
- Lee, B.H.; Kim, Y.H.; Park, K.S.; Eom, J.B.; Kim, M.J.; Rho, B.S.; Choi, H.Y. Interferometric fiber optic sensors. Sensors 2012, 12, 2467–2486. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, C.; Baptista, J.M.; Jorge, P. Refractometric Optical Fiber Platforms for Label Free Sensing. In Current Developments in Optical Fiber Technology; IntechOpen: London, UK, 2013; pp. 345–372. [Google Scholar] [CrossRef]
- Rose, K.; Dzyadevych, S.; Fernandez-Lafuente, R.; Jaffrezic, N.; Kuncova, G.; Matejec, V.; Scully, P. Hybrid coatings as transducers in optical biosensors. J. Coat. Technol. Res. 2008, 5, 491–496. [Google Scholar] [CrossRef]
- Kemal, F.; Hasan, E. Fiber Optic Sensors and Their Applications. In Proceedings of the 5th International Advanced Technologies Symposium (IATS’09), Karabuk, Turkey, 13–15 May 2009. [Google Scholar]
- Zibaii, M.; Latifi, H.; Asadollahi, A.; Bayat, A.; Dargahi, L.; Haghparast, A. Label Free FiberOptic Apta-Biosensor for in-vitro detection of Dopamine. J. Lightwave Technol. 2016, 34, 4516–4524. [Google Scholar] [CrossRef]
- Lao, J.; Han, L.; Wu, Z.; Zhang, X.; Huang, Y.; Tang, Y.; Guo, T. Gold Nanoparticle-Functionalized Surface Plasmon Resonance Optical Fiber Biosensor: In Situ Detection of Thrombin with 1 nM Detection Limit. J. Lightwave Technol. 2018. [Google Scholar] [CrossRef]
- Daems, D.; Pfeifer, W.; Rutten, I.; Sacca, B.; Spastic, D.; Lammertyn, J. Three-Dimensional DNA Origami as Programmable Anchoring Points for Bioreceptors in Fiber Optic Surface Plasmon Resonance Biosensing. ACS Appl. Mater. Interfaces 2018, 10, 23539–23547. [Google Scholar] [CrossRef] [PubMed]
- Mehrvar, M.; Bis, C.; Scharer, J.; Moo-Young, M.; Luong, J.H.T. Fiber-Optic Biosensors. Trends and Advances. Anal. Sci. 2000, 16. [Google Scholar] [CrossRef]
- Zhou, W.; Li, K.; Wei, Y.; Peng, H.; Mingbo, C.; Yongshun, L.; Yihui, W. Ultrasensitive label-free optical microfiber coupler biosensor for detection of cardiac troponin I based on interference turning point effect. Biosens. Bioelectr. 2018, 106, 99–104. [Google Scholar] [CrossRef] [PubMed]

| Component | Main Modification | Other Options | Application(s) | Advantages | Disadvantages | Reference(s) |
|---|---|---|---|---|---|---|
| Transducer | fiber optic cable | material (glass, silica, plastic) | medical, military, telecommunication, industrial, data storage, networking, and broadcast industries | cheaper, more flexible, more durable, easier to manufacture (depending on material type), can be miniaturized | less resistant to damage/unwanted scratching, easier to deform, require replacement faster (depending on material type) | [11,12,13,14] |
| core changes (tapering) | sensing | increased evanescent field interaction | --- | [11,12,13,14] | ||
| cladding changes (coating, tapering, scraping) | sensing | increased evanescent field interaction, increases sensitivity to specific wavelengths of light | --- | [11,12,13,14] | ||
| bending | sensing | increase in the evanescent field | loss of light signal | [11,12,13,14] | ||
| changing the launch angle | sensing | increase evanescent field depth | decreases applicability | [11,12,13,14] | ||
| increasing wavelength | sensing | increase evanescent field depth | --- | [11,12,13,14] | ||
| Biosensing element | extrinsic vs. intrinsic | sensing | extrinsic sensors guide the light to a separate sensing area (ideal for use with biosensors) | intrinsic sensors force the light signal to remain in the core of the fiber to detect changes | [16] | |
| pH | fluorescence | sensing | gels and other element fixing strategies are replaceable | needs a clear/large enough signal to excite a light generation or fluorescent signal | [9,17] | |
| enzyme | three options to initiate signal generation | sensing | well established in glucose monitoring, high sensitivity to low oxygen levels in intercellular fluid, is compatible with fluorescent indicators | reduced sensitivity to glucose signal resolution when miniaturized to a microsensor | [10,18,19,20,21,22,23] | |
| antibody | antigen or antibody use | immunoassay | compatible with fluorescent molecules, wide variety of diagnostic assay capabilities | require an attachment agent in order to be affixed to the cable at the sensing area | [6,24,34] | |
| whole cell | netting or membranes, fluorescence | bioluminescent cells | use of bioluminescent cells or cells that have measurable metabolic processes, some matrices shown to last up to a month without replacement | not compatible inside the human body because of bacterial infection concerns | [6,26,32] | |
| Sensing Strategies | signal generation | surface plasmon resonance (wavelength modulated, flow injection…) | categorized based on detection limit and sample being tested | can detect specific proteins up to small detection limits | --- | [29] |
| fluorescence | medical applications, chemical sensing and measurements of physical parameters | available in different configurations for specific applications | require more components and a fluorescent based signal in order to produce results | [35] | ||
| interferometers | sensing, telecommunication | measurand can be determined by changes in wavelength, phase, intensity, frequency and bandwidth as sensing indicators | require beam splitting and beam combining components | [36] | ||
| refractometers | sensing, telecommunication | simple, versatile, self-referenced | require design of sensitive layers that change when in contact with the analyte | [35] | ||
| Photodetectors | spectroscopy | fluorescence and absorption | sensing, telecommunication | well established technology with unlimited variations in technique and applications | have to be sensitive enough and have an adequate noise level to be used with FOBS, require a reference detector | [6,13,27,37] |
| evanescent wave interaction | sensing, telecommunication | ability to sense light and other electromagnetic radiation | overlap of evanescent waves with a separate medium to measure the reflective index, requires a reference detector | [5,6,13,27] | ||
| Signal Processor | voltage signal | amplifier, analog circuitry, analog to digital converter, noise reduction, background subtraction | output to interface, sensors | optimized for little noise if impedance is low, can propagate over larger distances | adds current consumption | [38] |
| current signal | amplifier, analog circuitry, analog to digital converter, noise reduction, background subtraction | output to interface, sensors | optimized for sending weak signals in a noisy environment | --- | [38] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mowbray, S.E.; Amiri, A.M. A Brief Overview of Medical Fiber Optic Biosensors and Techniques in the Modification for Enhanced Sensing Ability. Diagnostics 2019, 9, 23. https://doi.org/10.3390/diagnostics9010023
Mowbray SE, Amiri AM. A Brief Overview of Medical Fiber Optic Biosensors and Techniques in the Modification for Enhanced Sensing Ability. Diagnostics. 2019; 9(1):23. https://doi.org/10.3390/diagnostics9010023
Chicago/Turabian StyleMowbray, Shannon E., and Amir M. Amiri. 2019. "A Brief Overview of Medical Fiber Optic Biosensors and Techniques in the Modification for Enhanced Sensing Ability" Diagnostics 9, no. 1: 23. https://doi.org/10.3390/diagnostics9010023
APA StyleMowbray, S. E., & Amiri, A. M. (2019). A Brief Overview of Medical Fiber Optic Biosensors and Techniques in the Modification for Enhanced Sensing Ability. Diagnostics, 9(1), 23. https://doi.org/10.3390/diagnostics9010023





